Residual Maintenance Using Sodium Hypochlorite, Sodium Dichloroisocyanurate, and Chlorine Dioxide in Laboratory Waters of Varying Turbidity
Abstract
1. Introduction
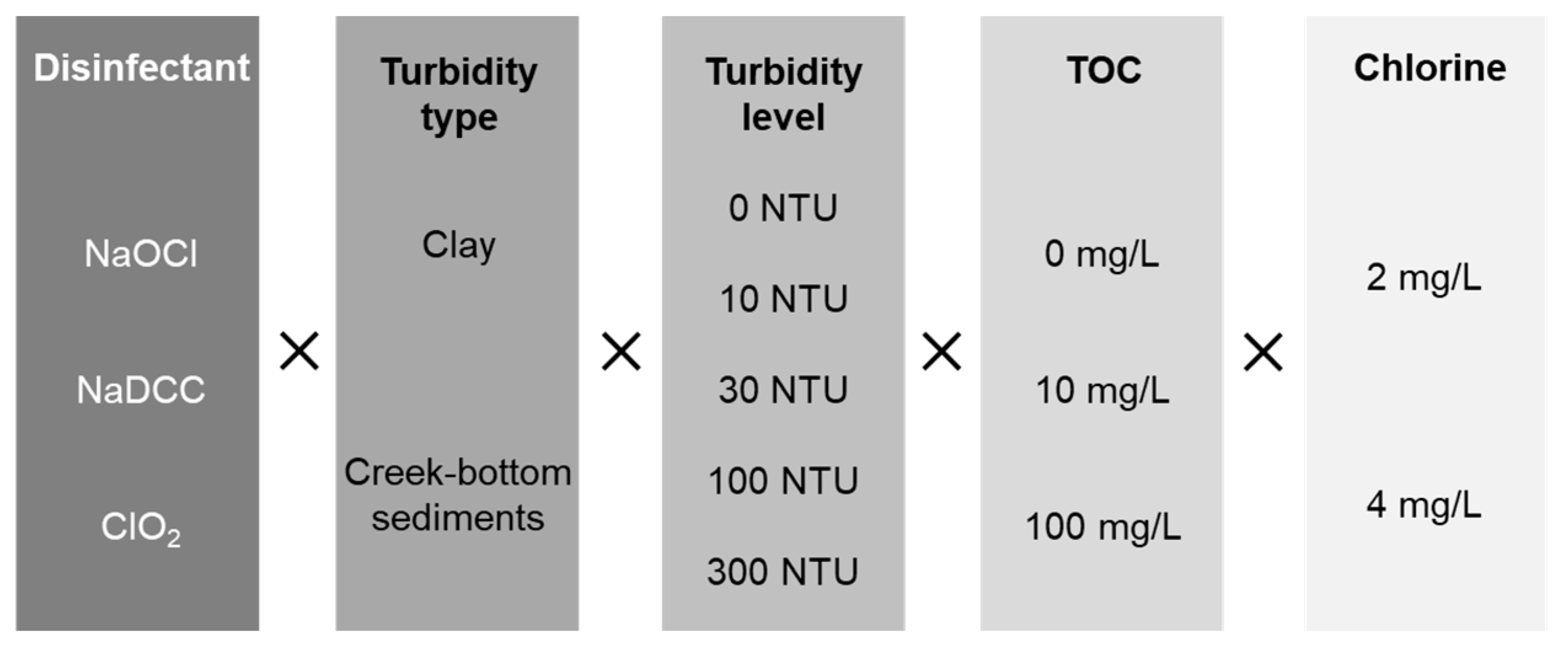
2. Materials and Methods
2.1. Turbidity
2.2. TOC
2.3. Disinfectants and Dose
2.4. Reactor Testing
2.5. Data Analysis
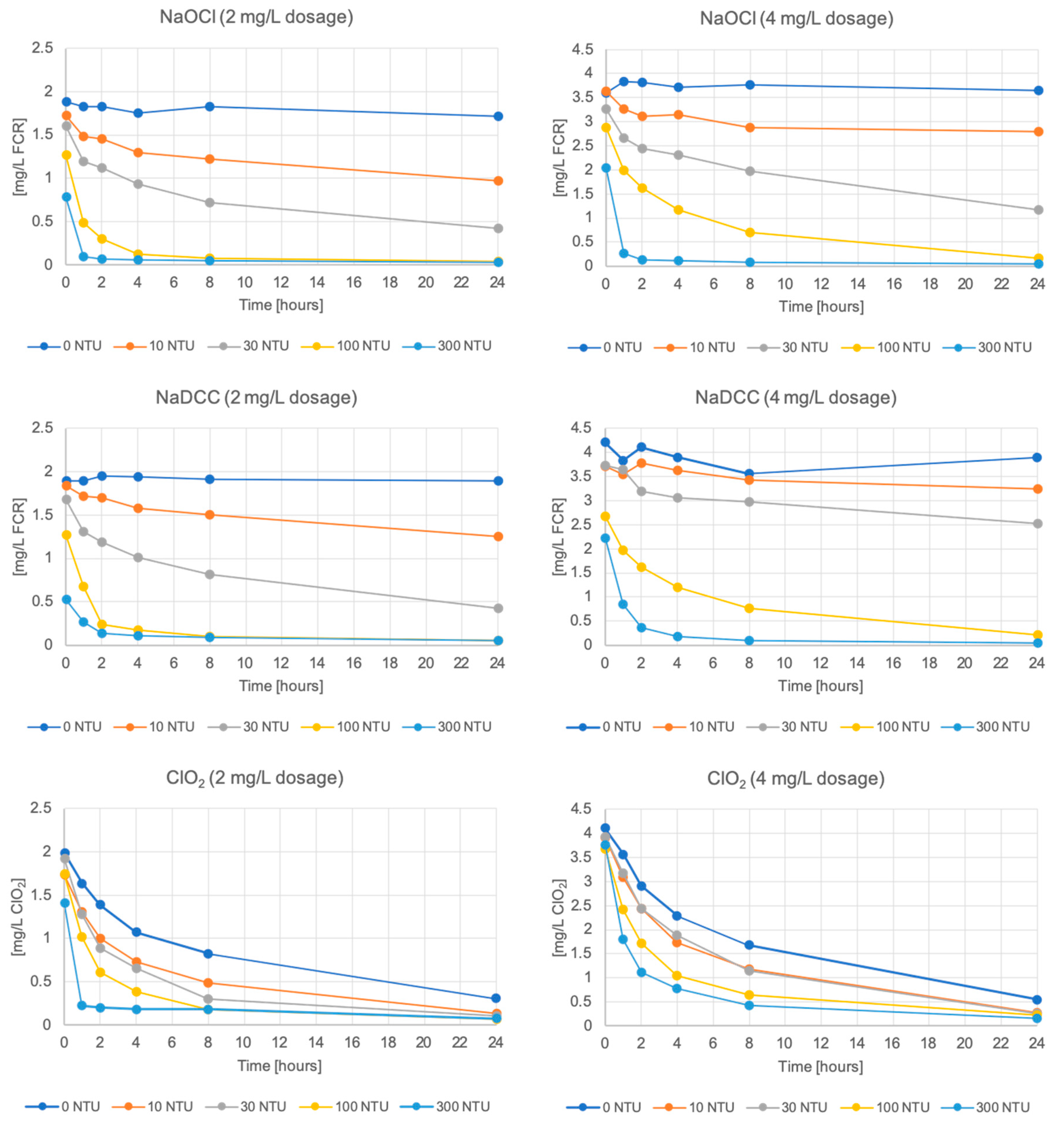
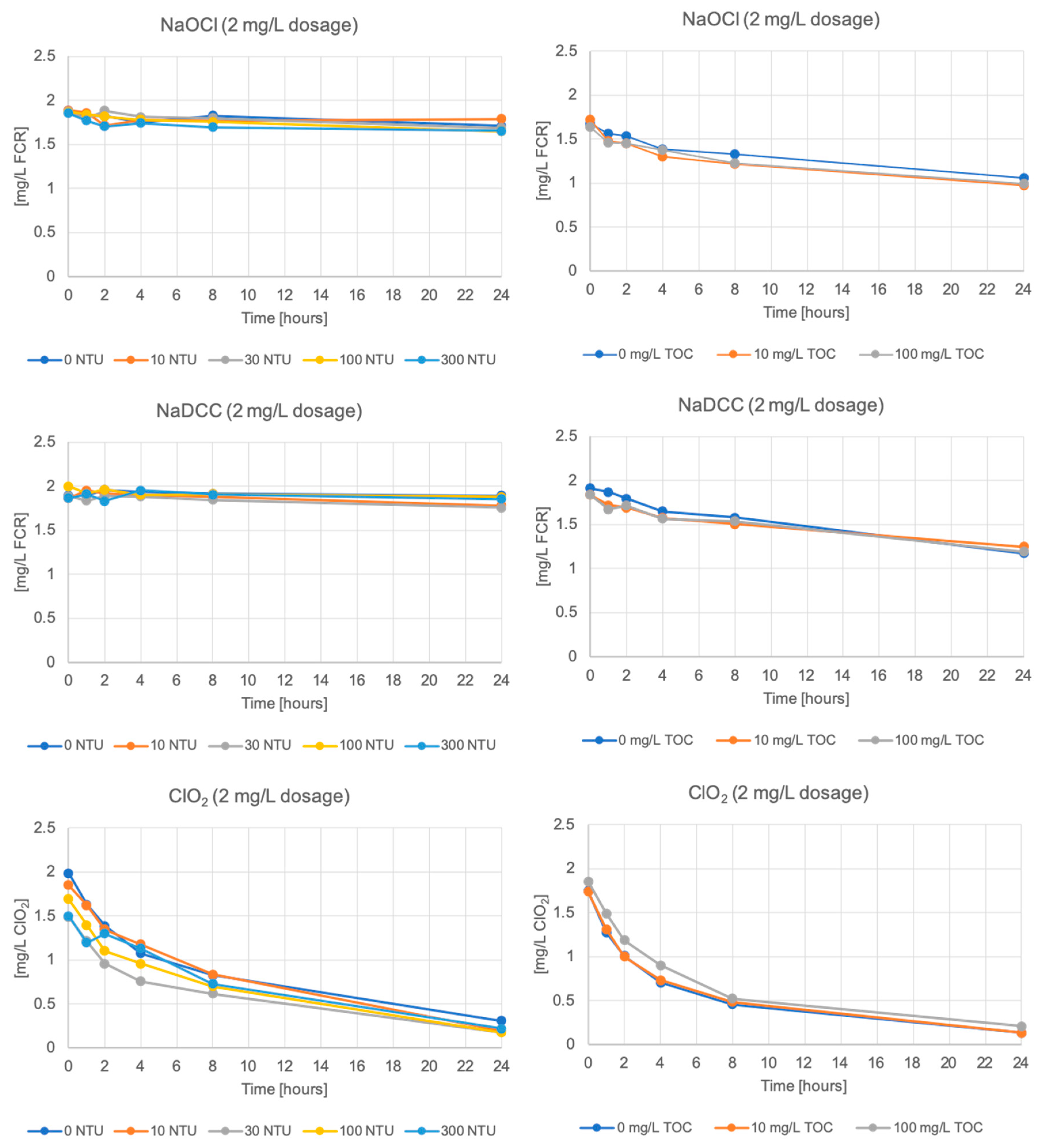
3. Results
3.1. Free Chlorine and Chlorine Dioxide Residual Decay
3.2. Meeting Minimim Standards
3.3. CT-factor Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO); United Nations Children’s Fund (UNICEF). Progress on Drinking Water, Sanitation and Hygiene: 2017 Update and SDG Baselines; WHO: Geneva, Switzerland; UNICEF: New York, NY, USA, 2017. [Google Scholar]
- Clasen, T.; Schmidt, W.P.; Rabie, T.; Roberts, I.; Cairncross, S. Interventions to improve water quality for preventing diarrhoea: systematic review and meta-analysis. BMJ 2007, 334, 782. [Google Scholar] [CrossRef] [PubMed]
- Clasen, T. Household water treatment and safe storage to prevent diarrheal disease in developing countries. Curr. Environ. Health Rep. 2015, 2, 69–74. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO); United Nations Children’s Fund (UNICEF). International Network on Household Water Treatment and Safe Stroage: Revised Strategy and Funding Proposal; WHO: Geneva, Switzerland; UNICEF: New York, NY, USA, 2011. [Google Scholar]
- World Health Organization (WHO). Considerations for Policy Development and Scaling-Up Household Water Treatment and Safe Storage with Communicable Disease Prevention Efforts; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Lantagne, D.; Quick, R.E.; Mintz, E. Household Water Treatment and Safe Storage Options in Developing Countries: A Review of Current Implementation Practices; Environmental Change and Security Program; Woodrow Wilson International Center for Scholars: Washington, DC, USA, 2006. [Google Scholar]
- Arnold, B.F.; Colford, J.M., Jr. Treating water with chlorine at point-of-use to improve water quality and reduce child diarrhea in developing countries: A systematic review and meta-analysis. Am. J. Trop. Med. Hyg. 2007, 76, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Medentech. Aquatabs® Water Disinfection Available Immediately. Available online: http://www.medentech.com/home/industries/water-purification/ (accessed on 4 January 2019).
- PSI. Water Treatment. Available online: http://www.psi.org/health-area/water-sanitation/water-treatment/#about (accessed on 4 January 2019).
- Lantagne, D.; Clasen, T. Use of household water treatment and safe storage methods in acute emergency response: Case study results from Nepal, Indonesia, Kenya, and Haiti. Environ. Sci. Technol. 2012, 46, 11352–11360. [Google Scholar] [CrossRef] [PubMed]
- LeChevallier, M.W.; Evans, T.M.; Seidler, R.J. Effect of Turbidity on Chlorine Efficiency and Bacterial Persistence in Drinking water. Appl. Environ. Microbiol. 1981, 42, 159–167. [Google Scholar] [PubMed]
- Black & Veatch Corporation. White’s Handbook of Chlorination and Alternative Disinfectants; John Wiley & Sons: Hoboken, NJ, USA, 2010. [Google Scholar]
- Deborde, M.; von Gunten, U. Reactions of chlorine with inorganic and organic compounds during water treatment—Kinetics and mechanisms: A critical review. J. Water Res. 2008, 42, 13–51. [Google Scholar] [CrossRef] [PubMed]
- American Water Works Association (AWWA). Water Quality and Treatment: A Handbook for Community Water Supplies; McGraw-Hill: Denver, CO, USA, 1999. [Google Scholar]
- American Public Health Association (APHA); American Water Works Association (AWWA); Water Environment Federation (WEF). Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017; ISBN 9780875532875. [Google Scholar]
- Lantagne, D. Sodium hypochlorite dosage for household and emergency water treatment. J. Am. Water Works Assoc. 2008, 100, 106–119. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Guidelines for Drinking-Water Quality, 4th ed.; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Safe Water System—Options for Pre-Treating Water in Chlorination Projects. Available online: https://www.cdc.gov/safewater/chlorination-pretreatment.html (accessed on 4 January 2019).
- Xinix. Freebact Disinfectant. Available online: http://www.xinix.se/products/freebact/ (accessed on 4 January 2019).
- Portable Aqua Chlorine Dioxide Water Purification Tablets. Available online: https://www.potableaqua.com/products/pa-chlorine-dioxide-purification-tablets-2/ (accessed on 4 January 2019).
- Scotmas. Safe, Reliable, and Cost-Effective ClO2 Solutions. Available online: https://www.scotmas.com/products/chemicals.aspx (accessed on 4 January 2019).
- Water Treatment Products, Ltd. Chlorine Dioxide Water Treatment. Available online: http://www.watertreatmentproducts.co.uk/product-category/chlorine-dioxide-water-treatment/ (accessed on 4 January 2019).
- Centers for Disease Control and Prevention (CDC). Safe Water for the Community: A Guide for Establishing A Community-Based Safe Water System Program; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2008.
- Lantagne, D.; Preston, K.; Blanton, E.; Kotlarz, N.; Gezagehn, H.; van Dusen, E.; Berens, J.; Jellison, K. Hypochlorite solution expiry and stability in household water treatment in developing countries. J. Environ. Eng. 2011, 137. [Google Scholar] [CrossRef]
- Rook, J.J. Formation of haloforms during chlorination of natural waters. Water Treat. Exam. 1974, 23, 234–243. [Google Scholar]
- Lantagne, D.; Blount, B.C.; Cardinali, F.; Quick, R.E. Disinfection by-product formation and mitigation strategies in point-of-use chlorination of turbid and non-turbid waters in western Kenya. J. Water Health 2008, 6, 67–82. [Google Scholar] [CrossRef]
- Clasen, T.; Edmonson, P. Sodium dichloroisocyanurate (NaDCC) tablets as an alternative to sodium hypochlorite for the routine treatment of drinking water at the household level. Int. J. Hyg. Environ. Health 2006, 209, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Kuszenof, P. Sodium Dichloroisocyanurate: Chemical and Technical Assessment; 61st JECFA; Food and Agriculture Organization (FAO) of the United Nations: Rome, Italy, 2004. [Google Scholar]
- Lantagne, D.; Cardinali, F.; Blount, B.C. Disinfection by-product formation and mitigation strategies in point-of-use chlorination with sodium dichloroisocyanurate in Tanzania. Am. J. Trop. Med. Hyg. 2010, 83, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Aieta, E.; Berg, J. A review of chlorine dioxide in drinking water treatment. J. Am. Water Works Assoc. 1986, 78, 62–72. [Google Scholar] [CrossRef]
- Mohamed, H.; Brown, J.; Njee, R.M.; Clasen, T.; Malebo, H.M.; Mbuligwe, S. Point-of-use chlorination of turbid water: results from a field study in Tanzania. J. Water Health 2015, 13, 544–552. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kotlarz, N.; Lantagne, D.; Preston, K.; Jellison, K. Turbidity and chlorine demand reduction using locally available physical water clarification mechanisms before household chlorination in developing countries. J. Water Health 2009, 7, 497–506. [Google Scholar] [CrossRef]
- Preston, K.; Lantagne, D.; Kotlarz, N.; Jellison, K. Turbidity and chlorine demand reduction using alum and moringa flocculation before household chlorination in developing countries. J. Water Health 2010, 8, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Safety Data Sheet—TOC Standard 10000 mg/L—Product Code #5847000—SDS #M02156. Available online: https://www.hach.com/toc-standard-solution-10000-mg-l-4-l/product-downloads?id=7640481435 (accessed on 25 June 2019).
- Kaplan, L.A.; Newbold, J.D. Surface and Subsurface Dissolved Organic Carbon. In Streams and Ground Waters; Academic Press: Cambridge, MA, USA, 2000. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). 4500-Cl: Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1992. [Google Scholar]
- American Public Health Association (APHA). 4500-ClO2: Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1992. [Google Scholar]
- Murray, A.; Lantagne, D. Accuracy, precision, usability, and cost of free chlorine residual testing methods. J. Water Health 2015, 13, 79–90. [Google Scholar] [CrossRef]
- Centers for Disease Control and Preventioen (CDC). Safe Water System (SWS)—Effect of Chlorination on Inactivating Selected Pathogens; Centers for Disease Control and Preventioen: Atlanta, GA, USA, 2008.
- Kotloff, K.L.; Nataro, J.P.; Blackwelder, W.C.; Nasrin, D.; Farag, T.H.; Panchalingam, S.; Wu, Y.; Sow, S.O.; Sur, D.; Breiman, R.F.; et al. Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): A prospective, case-control study. Lancet 2013, 382, 209–222. [Google Scholar] [CrossRef]
- La Fauci, V.; Costa, G.B.; Arena, A.; Ventura Spagnolo, E.; Genovese, C.; Palamara, M.A.; Squeri, R. Trend of MDR-microorganisms isolated from the biological samples of patients with HAI and from the surfaces around that patient. New Microbiol. 2018, 41, 42–46. [Google Scholar]
- D’Auria, F.D.; Simonetti, G.; Strippoli, V. Antimicrobial activity exerted by sodium dichloroisocyanurate. Annali di Igiene: Medicina Preventiva e di Comunita 1989, 1, 1445–1458. [Google Scholar]
- Schlosser, O.; Robert, C.; Bourderioux, C.; Rey, M.; de Roubin, M.R. Bacterial removal from inexpensive portable water treatment systems for travelers. J. Travel Med. 2001, 8, 12–18. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gava Mazzola, P.; Vessoni Pena, T.C.; Martins, A.M. Determination of decimal reduction time (D value) of chemical agents used in hospitals for disinfection purposes. BMC Infect. Dis. 2003, 3, 24. [Google Scholar] [CrossRef] [PubMed]
- LeChevallier, M.W.; Cawthon, D.C.; Lee, R.G. Inactivation of biofilm bacteria. Appl. Environ. Microbiol. 1988, 54, 2492–2499. [Google Scholar] [PubMed]
- Xue, B.; Jin, M.; Yang, D.; Guo, X.; Chen, Z.; Shen, Z.; Wang, X.; Qiu, Z.; Wang, J.; Zhang, B.; et al. Effects of chlorine and chlorine dioxide on human rotavirus infectivity and genome stability. Water Res. 2013, 47, 3329–3338. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.M.; Black, D.A.; Pien, S.H.; Read, E.J. Predicting the Inactivation of Giardia Lamblia: A Mathematical and Statistical Model; Environmental Protection Agency: Washington, DC, USA, 1990.
- US Environmental Protection Agency (US EPA). Guidance Manual for Compliance with the Filtration and Disinfection Requirements for Public Water Systems Using Surface Water Sources; Environmental Protection Agency: Washington, DC, USA, 1991.
- World Health Organization (WHO). Harmonized Testing Protocol: Technology Non-Specific. WHO International Scheme to Evaluate Household Water Treatment Technologies; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Aieta, E.M.; Roberts, P.V.; Hernandez, M. Determination of Chlorine Dioxide, Chlorine, Chlorite, and Chlorate in Water. J. Am. Water Works Assoc. 1984, 76, 64–70. [Google Scholar] [CrossRef]
- Wilhelm, N.; Kaufmann, A.; Blanton, E.; Lantagne, D. Sodium hypochlorite dosage for household and emergency water treatment: updated recommendations. J. Water Health 2018, 16, 112–126. [Google Scholar] [CrossRef]
| Compound | Chemical Formula | Mol. Weight (g/mol) | pH in Solution | Biocidal Compound | Typical Packaging |
|---|---|---|---|---|---|
| Sodium hypochlorite | NaOCl | 74.44 | 11–12 | HOCl | Liquid |
| Sodium dichloroisocyanurate | NaC3N3O3Cl2 | 219.95 | 6–7 | HOCl | Powder, tablets |
| Chlorine dioxide | ClO2 | 67.45 | n/a | ClO2 (g) | Liquid, tablets |
| Turbidity Type | Turbidity Level [41] | NaOCl k (h−1) | NaDCC k (h−1) | ClO2 k (h−1) | |||
|---|---|---|---|---|---|---|---|
| 2 mg/L | 4 mg/L | 2 mg/L | 4 mg/L | 2 mg/L | 4 mg/L | ||
| N/A | 0 | −0.0042 | −0.0012 | −0.0034 | −0.0039 | −0.0752 | −0.0824 |
| Clay | 10 | −0.0029 | −0.0013 | −0.0020 | −0.0021 | −0.0942 | −0.0985 |
| 30 | −0.0041 | −0.0021 | −0.0035 | −0.0019 | −0.0866 | −0.1009 | |
| 100 | −0.0038 | −0.0021 | −0.0028 | −0.0033 | −0.0966 | −0.0999 | |
| 300 | −0.0034 | −0.0013 | −0.0028 | −0.0027 | −0.0878 | −0.0942 | |
| Sediment | 10 | −0.0188 | −0.0110 | −0.0168 | −0.0069 | −0.0952 | −0.1005 |
| 30 | −0.0545* | −0.0515* | −0.0405* | −0.0258* | −0.1075 | −0.1008 | |
| 100 | −0.1392* | −0.1289* | −0.1327* | −0.0819* | −0.1004† | −0.1041† | |
| 300 | −0.1392 | −0.0986* | −0.1327 | −0.1267* | −0.1004 | −0.0943 | |
| Turbidity Type | Turbidity Level [41] | NaOCl (mg/L Cl2) | NaDCC (mg/L Cl2) | ClO2 (mg/L ClO2) | |||
|---|---|---|---|---|---|---|---|
| 2 mg/L | 4 mg/L | 2 mg/L | 4 mg/L | 2 mg/L | 4 mg/L | ||
| N/A | 0 | 1.7 (0.09) | 3.6 (0.13) | 1.8 (0.11) | 3.7 (0.18) | 0.29 (0.02) | 0.50 (0.05) |
| Clay | 10 | 1.7 (0.09) | 3.6 (0.09) | 1.9 (0.11) | 4.0 (0.14) | 0.17 (0.07) | 0.31 (0.13) |
| 30 | 1.6 (0.07) | 3.3 (0.21) | 1.8 (0.11) | 3.9 (0.10) | 0.19 (0.05) | 0.31 (0.15) | |
| 100 | 1.6 (0.08) | 3.5 (0.16) | 1.8 (0.11) | 3.8 (0.11) | 0.17 (0.10) | 0.32 (0.17) | |
| 300 | 1.6 (0.11) | 3.5 (0.17) | 1.8 (0.08) | 3.7 (0.17) | 0.20 (0.10) | 0.33 (0.16) | |
| Sediment | 10 | 1.0 (0.06) | 2.6 (0.19) | 1.2 (0.06) | 3.1 (0.16) | 0.16 (0.04) | 0.32 (0.09) |
| 30 | 0.39 (0.17) | 1.2 (0.06) | 0.64 (0.17) | 2.3 (0.19) | 0.12 (0.01) | 0.29 (0.07) | |
| 100 | 0.03 (0.01) | 0.12 (0.05) | 0.04 (0.01) | 0.39 (0.14) | 0.10 (0.03) | 0.21 (0.03) | |
| 300 | 0.03 (0.01) | 0.06 (0.01) | 0.05 (0.01) | 0.07 (0.02) | 0.12 (0.04) | 0.19 (0.03) | |
| Test Organism | Disinfectant | CT Factor (mg min/L) | Inactivation* | Temp. (°C) | Source |
|---|---|---|---|---|---|
| Escherichia coli | HOCl (NaOCl) | 0.04 | 2-log | 5 | [45] |
| ClO2 | 0.18–0.38 | 2-log | 15–25 | [45] | |
| Rotavirus | HOCl (oxidized HCl) | 1.25–3.57 | 3-log | 20 | [46] |
| ClO2 | 0.13–0.32 | 3-log | 20 | [46] | |
| Giardia lamblia | HOCl (unclear) | <9–12 | 2-log | 25 | [47] |
| ClO2 | 10 | 2-log | 20 | [48] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallandat, K.; Stack, D.; String, G.; Lantagne, D. Residual Maintenance Using Sodium Hypochlorite, Sodium Dichloroisocyanurate, and Chlorine Dioxide in Laboratory Waters of Varying Turbidity. Water 2019, 11, 1309. https://doi.org/10.3390/w11061309
Gallandat K, Stack D, String G, Lantagne D. Residual Maintenance Using Sodium Hypochlorite, Sodium Dichloroisocyanurate, and Chlorine Dioxide in Laboratory Waters of Varying Turbidity. Water. 2019; 11(6):1309. https://doi.org/10.3390/w11061309
Chicago/Turabian StyleGallandat, Karin, David Stack, Gabrielle String, and Daniele Lantagne. 2019. "Residual Maintenance Using Sodium Hypochlorite, Sodium Dichloroisocyanurate, and Chlorine Dioxide in Laboratory Waters of Varying Turbidity" Water 11, no. 6: 1309. https://doi.org/10.3390/w11061309
APA StyleGallandat, K., Stack, D., String, G., & Lantagne, D. (2019). Residual Maintenance Using Sodium Hypochlorite, Sodium Dichloroisocyanurate, and Chlorine Dioxide in Laboratory Waters of Varying Turbidity. Water, 11(6), 1309. https://doi.org/10.3390/w11061309





