Marine Ranching Construction and Management in East China Sea: Programs for Sustainable Fishery and Aquaculture
Abstract
1. Introduction
2. History of Marine Ranching Demonstration Region Construction in China
3. Marine Ranching Construction in ECS: Techniques and Case
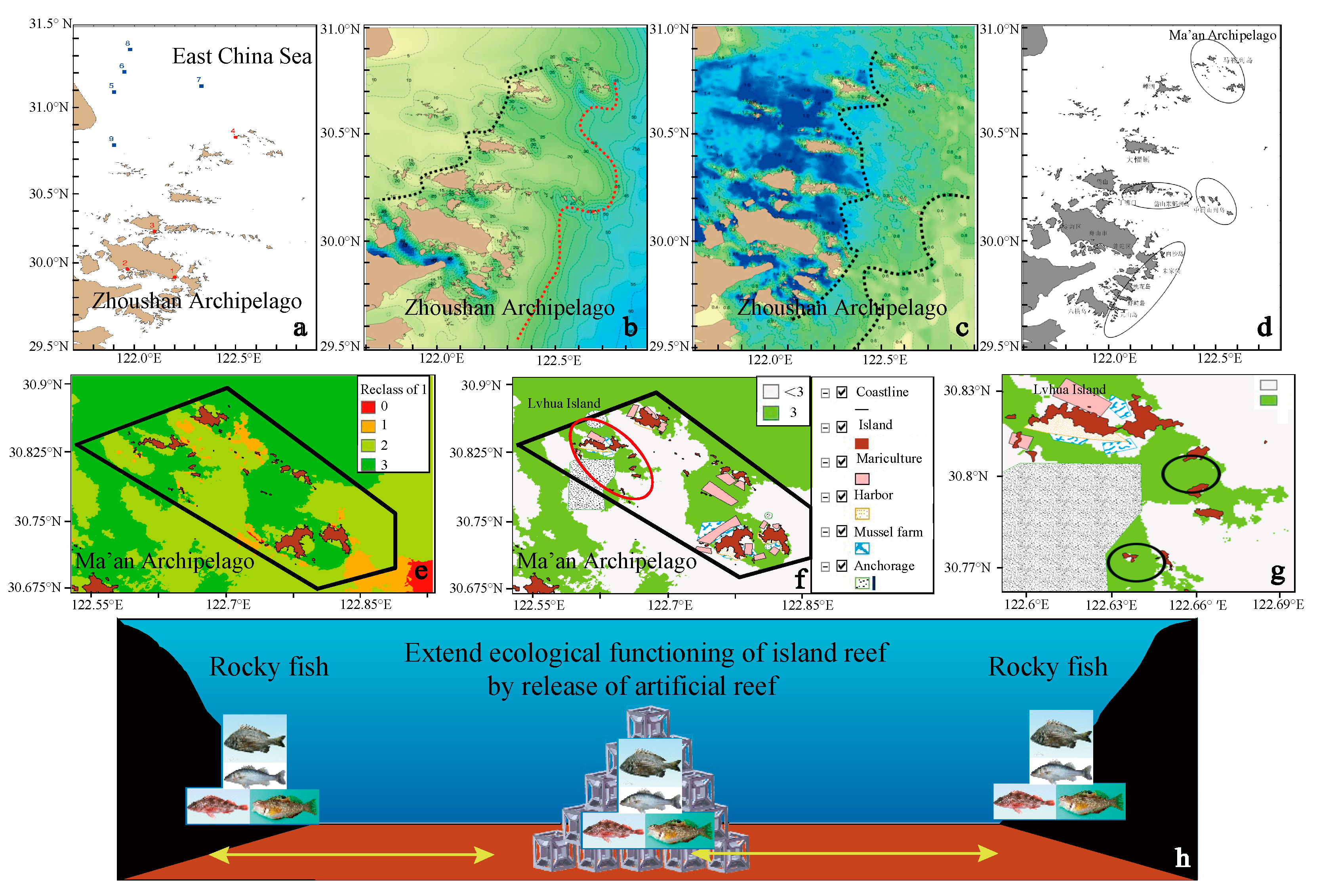
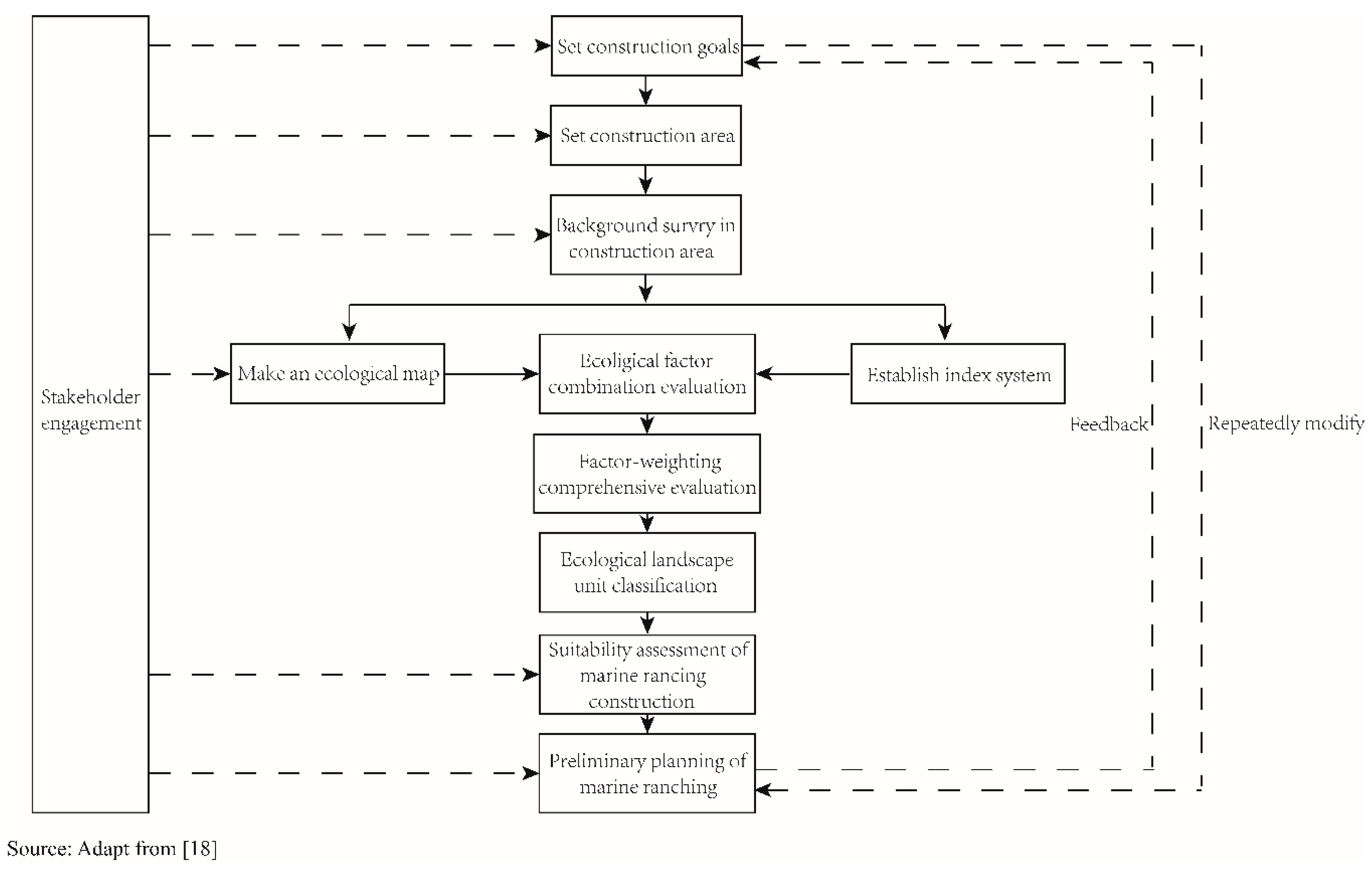
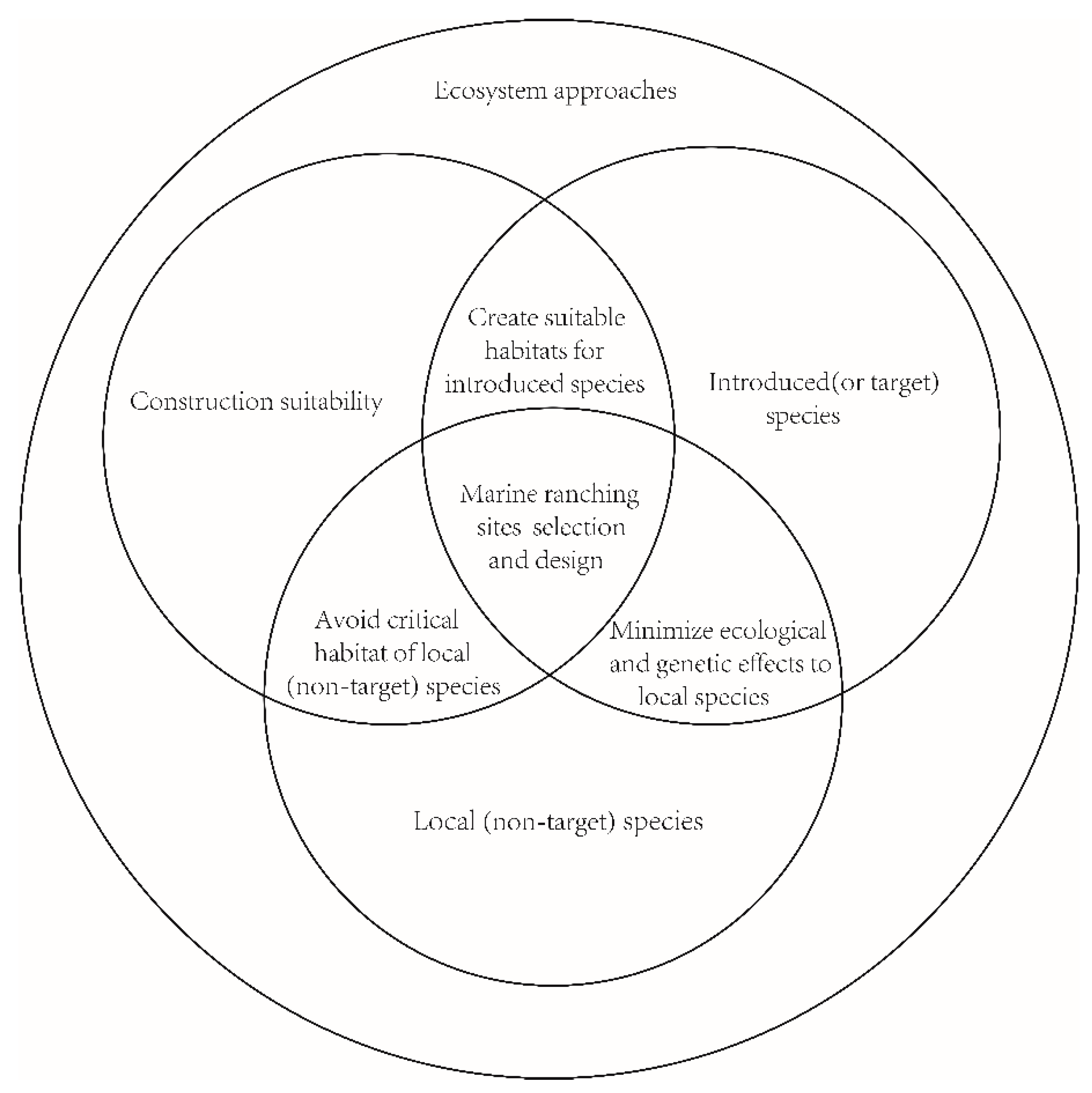
3.1. Marine Ranching Sites Selection and Design
3.2. Habitat Restoration and Construction Technologies
3.2.1. Overall Restoration and Construction Mode Use in the ECS
3.2.2. Restoration of Natural Habitat or Building Artificial Structure
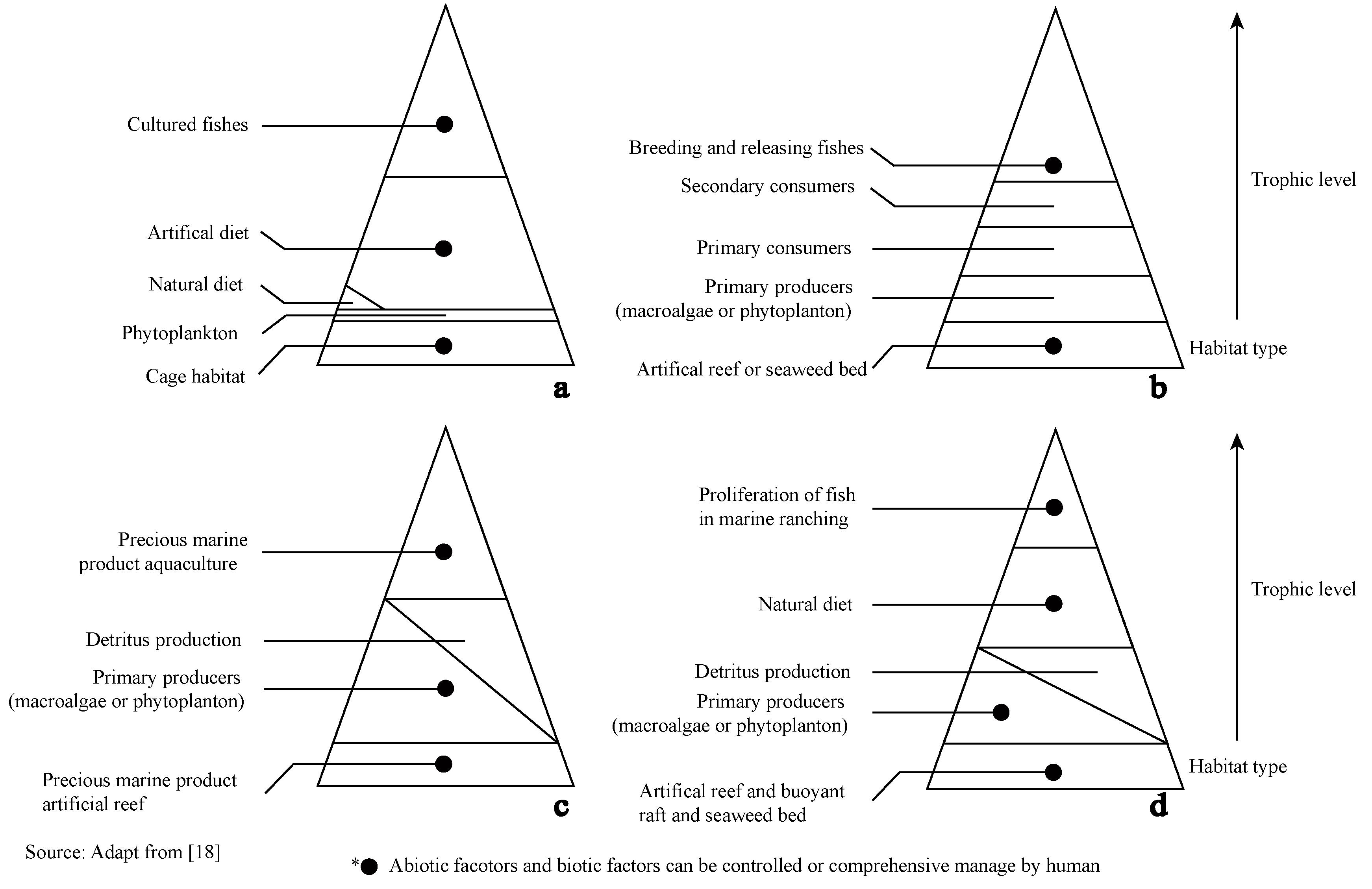
3.2.3. Constructing Biotic Habitats through Artificial Release of Biota of Different Trophic Levels in Artificial or Natural Habitats
3.3. Stock Enhancement and Behavioral Control of Fishery Resources
3.4. Marine Ranching Management
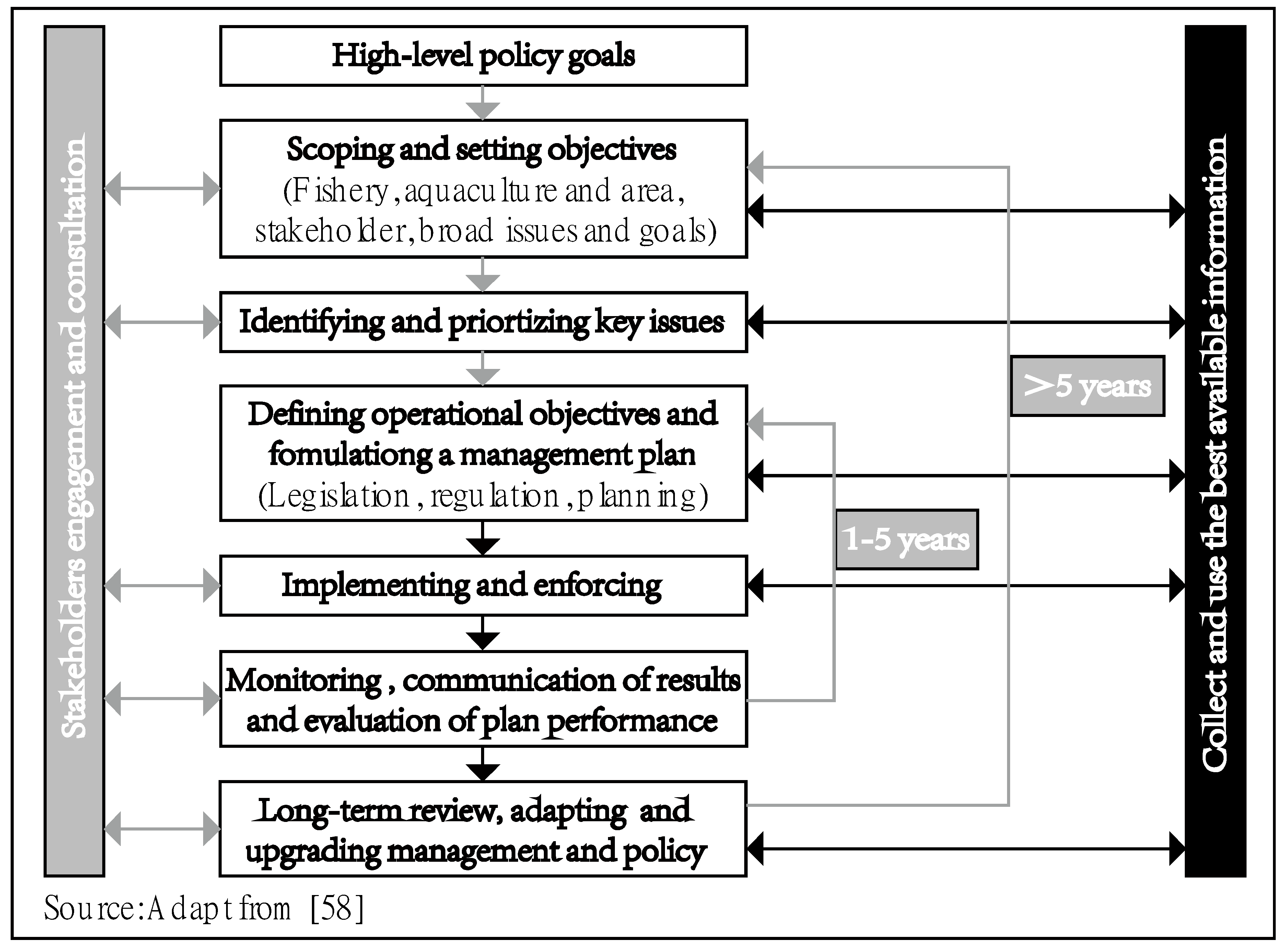
3.4.1. Marine Ranching Management Models
3.4.2. Real-Time Monitoring and Long-Range Data Communication
3.4.3. Management of Complex and Multi-Stakeholder Users and Uses
4. Conclusions
5. Synthesis and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Bank. Fish to 2030: Prospects for Fisheries and Aquaculture; World Bank Report No.83177-GLB, Agriculture and Environmental Services Discussion Paper 03; World Bank: Washington, DC, USA, 2013. [Google Scholar]
- FAO. The Future of Food and Agriculture—Trends and Challenges; FAO: Rome, Italy, 2017. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture 2018—Meeting the Sustainable Development Goals; FAO: Rome, Italy, 2018. [Google Scholar]
- FAO. The FAO Blue Growth Initiative: Strategy for the Development of Fisheries and Aquaculture in Eastern Africa; FAO Fisheries and Aquaculture Circular No. 1161; FAO: Rome, Italy, 2018. [Google Scholar]
- Lillebø, A.I.; Pita, C.; Rodrigues, J.G.; Ramos, S.; Villasante, S. How can marine ecosystem services support the blue growth agenda? Mar. Policy 2017, 81, 132–142. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture; FAO: Rome, Italy, 2016. [Google Scholar]
- Burgess, M.G.; Clemence, M.; McDermott, G.R.; Costello, C.; Gaines, S.D. Five rules for pragmatic blue growth. Mar. Policy 2018, 87, 331–339. [Google Scholar] [CrossRef]
- Massa, F.; Rigillo, R.; Bourdenet, D.; Fezzardi, D.; Nastasi, A.; Rizzotti, H.; Emam, W.; Carmignac, C. FAO Fisheries and Aquaculture Proceedings; No. 46, Proceedings of the Regional Conference Blue Growth in the Mediterranean and the Black Sea: Developing Sustainable Aquaculture for Food Security, Bari, Italy, 9–11 December 2014; FAO: Rome, Italy, 2017. [Google Scholar]
- Yang, H.S. Construction of marine ranching in China: Reviews and prospects. J. Fish. China 2016, 40, 1133–1140. (In Chinese) [Google Scholar]
- Morita, K.; Saito, T.; Miyakoshi, Y.; Fukuwaka, M.; Nagasawa, T.; Kaeriyame, M. A review of Pacific salmon hatchery programmes on Hokkaido Island, Japan. ICES J. Mar. Sci. 2006, 63, 1353–1363. [Google Scholar] [CrossRef]
- Loneragan, N.R.; Jenkins, G.I.; Taylor, M.D. Marine Stock Enhancement, Restocking, and Sea Ranching in Australia: Future Directions and a Synthesis of Two Decades of Research and Development. Rev. Fish. Sci. 2013, 21, 222–236. [Google Scholar] [CrossRef]
- Salvanes, A.G.V. Encyclopedia of Ocean Sciences, 2nd ed.; Academic Press: Cambridge, MA, USA, 2001; pp. 146–155. [Google Scholar]
- Bell, J.D.; Leber, K.M.; Blankenship, H.L.; Loneragan, N.R.; Masuda, R. A New Era for restocking, stock enhancement and sea ranching of coastal fisheries resources. Rev. Fish. Sci. 2008, 16, 1–9. [Google Scholar] [CrossRef]
- Mustafa, S. Stock enhancement and sea ranching: Objectives and potential. Rev. Fish. Biol. Fish. 2003, 13, 141–149. [Google Scholar] [CrossRef]
- Grant, W.S.; Jasper, J.; Bekkevold, D.; Adkison, M. Responsible genetic approach to stock restoration, sea ranching and stock enhancement of marine fishes and invertebrates. Rev. Fish. Biol. Fish. 2017, 27, 615–649. [Google Scholar] [CrossRef]
- Kitada, S. Economic, ecological and genetic impacts of marine stock enhancement and sea ranching: A systematic review. Fish Fish. 2018, 19, 511–532. [Google Scholar] [CrossRef]
- Lima, J.S.; Zalmon, I.R.; Love, M. Overview and trends of ecological and socioeconomic research on artificial reefs. Mar. Environ. Res. 2019, 145, 81–96. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Zhou, X.J.; Wang, K.; Lin, J.; Zhao, J.; Zhao, X.; Guo, Y.; Liu, S.R.; Cheng, X.P. Review of marine livestock ecological urbanization hypothesis and marine ranching construction key–technology against blue growth background. J. Fish. China 2019, 43, 81–96. (In Chinese) [Google Scholar]
- Ungson, J.R.; Matsuda, Y.; Hirata, H.; Shiihara, H. An economic assessment of the production and release of marine fish fingerlings for sea ranching. Aquaculture 1993, 118, 169–181. [Google Scholar] [CrossRef]
- Shi, H.H.; Zheng, W.; Zhang, X.L.; Zhu, M.Y.; Ding, D.W. Ecological–economic assessment of monoculture and integrated multi-trophic aquaculture in Sanggou Bay of China. Aquaculture 2013, 410, 172–178. [Google Scholar] [CrossRef]
- Que, H.Y.; Chen, Y.; Zhang, X.M.; Zhang, S.Y.; Zhang, G.F. Modern marine ranching: Status and development strategy. Eng. Sci. 2016, 18, 79–84. (In Chinese) [Google Scholar]
- Zeng, C.K.; Mao, H.L. Development, current situation and Prospect of Oceanographic. Sci. Bull. 1965, 10, 876–883. (In Chinese) [Google Scholar]
- Zeng, C.K. Fight for high speed to realize the modernization of China’s aquaculture business. Fish. Sci. Technol. Inf. 1978, 8, 1. (In Chinese) [Google Scholar]
- Liu, X.Z. Fishery should go to the road of farming--fishing experts talking about the development of marine fishery production. Outlook Wkly. 1984, 43, 25. (In Chinese) [Google Scholar]
- Mao, H.L. Advances of marine science in recent twenty years. Mar. Sci. 1979, 3, 1–9. (In Chinese) [Google Scholar]
- Huang, W.F. Theory and practice of fishery cultivation. Fujian Fish. Sci. Technol. 1979, 1, 6–21. (In Chinese) [Google Scholar]
- Wang, S.B. Achievements and prospects of marine biology. J. Liaoning Norm. Univ. 1979, 3, 57–60. (In Chinese) [Google Scholar]
- Si, Y.J. Legal Regulation of Artificial Reef; Ocean University of China: Qingdao, China, 2012. (In Chinese) [Google Scholar]
- Chinese State Council. Program of Action on the Conservation of Living Aquatic Resources of China; Ministry of Agriculture of the People’s Republic of China: Beijing, China, 2006. (In Chinese) [Google Scholar]
- Fisheries Administration Bureau of Ministry of Agriculture and Rural Affairs, Chinese Academy of Fishery Sciences. Research on the Development Strategy of Marine Ranching in China; China Agriculture Press: Beijing, China, 2017. (In Chinese) [Google Scholar]
- The 13th Five-year Plan Outline of the People’s Republic of China for National Economic and Social Development; People’s Publishing House: Beijing, China, 2016. (In Chinese)
- Ministry of Agriculture and Rural Affairs of People’s Republic of China. Ministry of Agriculture, Guiding Opinions on Speeding up the Transformation of Fishery Turnaround Structure. China Fisheries 2016, 6, 8–11. (In Chinese) [Google Scholar]
- Fishery Administration Bureau of Ministry of Agriculture. China Statistical Yearbook 2017; China Agriculture Press: Beijing, China, 2017. (In Chinese) [Google Scholar]
- Aguilar-Manjarrez, J.; Soto, D.; Brummett, R. Aquaculture Zoning, Site Selection and Area Management Under the Ecosystem Approach to Aquaculture: A Handbook; FAO: Rome, Italy; World Bank: Washington, DC, USA, 2017. [Google Scholar]
- Xu, Q.; Liu, S.B.; Xu, M.; Zhang, S.Y. Preliminary study on sites selection of marine ranching-take Zhoushan as an example. Fish. Mod. 2011, 38, 27–31. (In Chinese) [Google Scholar]
- Xu, Q.; Zhang, S.Y. Site selection evaluation of marine ranching in Zhoushan area based on AHP method. J. Shanghai Ocean. Univ. 2013, 22, 128–133. (In Chinese) [Google Scholar]
- Zeng, X.; Zhang, S.Y.; Lin, J.; Zhong, J.M. Site selection suitability assessment for protective artificial reefs in island area. J. Fish. China 2018, 42, 52–62. (In Chinese) [Google Scholar]
- Duan, D.Y.; Qin, C.X.; Ma, H.; Lin, H.J.; Chen, P.M. Analysis of marine ranching landscape elements from the perspective of landscape ecology. Mar. Environ. Sci. 2018, 37, 52–59. (In Chinese) [Google Scholar]
- Chesson, P. Mechanisms of maintenance of species diversity. Annu. Rev. Ecol. Syst. 2000, 31, 343–366. [Google Scholar] [CrossRef]
- Bulleri, F.; Chapman, M.G. The introduction of coastal infrastructure as a driver of change in marine environments. Appl. Ecol. 2010, 47, 26–35. [Google Scholar] [CrossRef]
- Firth, L.B.; Knights, A.M.; Bridger, D.; Evans, A.J.; Mieszkowska, N.; Moore, P.J.; O’Connor, N.E.; Sheehan, E.V.; Thompson, R.C.; Hawkins, S.J. Ocean sprawl: Challenges and opportunities for biodiversity management in a changing world. Oceanogr. Mar. Biol. Annu. Rev. 2016, 54, 193–269. [Google Scholar]
- Grossman, G.D.; Jones, G.P.; Seaman, W.J. Do artificial reefs increase regional fish do artificial reefs increase regional fish Production? A review of existing data. Fisheries 1997, 22, 17–23. [Google Scholar] [CrossRef]
- Smith, J.A.; Lowry, M.B.; Suthers, I.M. Fish attraction to artificial reefs not always harmful: A simulation study. Ecol. Evol. 2015, 5, 4590–4602. [Google Scholar] [CrossRef]
- Mills, K.A.; Hamer, P.A.; Quinn, G.P. Artificial reefs create distinct fish assemblages. Mar. Ecol. Prog. Ser. 2017, 585, 155–173. [Google Scholar] [CrossRef]
- Lee, M.O.; Otake, S.; Kim, J.K. Transition of artificial reefs (ARs) research and its prospects. Ocean Coast. Manag. 2018, 154, 55–65. [Google Scholar] [CrossRef]
- Brickhill, M.J.; Lee, S.Y.; Connolly, R.M. Fishes associated with artificial reefs: Attributing changes to attraction or production using novel approaches. J. Fish. Biol. 2005, 67, 53–71. [Google Scholar] [CrossRef]
- Valentine, J.F.; Heck, K.L.; Cinkovich, A.M. Impacts of seagrass food webs on marine ecosystems: A need for a broader perspective. Bull. Mar. Sci. Miami 2002, 71, 1361–1368. [Google Scholar]
- Aguilera, M.A.; Broitman, B.R.; Thiel, M. Artificial breakwaters as garbage bins: Structural complexity enhances anthropogenic litter accumulation in marine intertidal habitats. Environ. Pollut. 2016, 214, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Paxton, A.B.; Revels, L.W.; Rosemond, R.C.; Hoeck, R.V.; Lemoine, H.R.; Taylor, J.C.; Peterson, C.H. Convergence of fish community structure between a newly deployed and an established artificial reef along a five-month trajectory. Ecol. Eng. 2018, 123, 185–192. [Google Scholar] [CrossRef]
- Yang, X.Y.; Lin, C.G.; Song, X.Y.; Xu, M.; Yang, H.S. Effects of artificial reefs on the meiofaunal community and benthic environment—A case study in Bohai Sea, China. Mar. Pollut. Bull. 2019, 140, 179–187. [Google Scholar] [CrossRef]
- Peng, Y.S.; Zheng, M.X.; Zheng, Z.X.; Wu, G.C.; Chen, Y.C.; Xu, H.L.; Tian, G.H.; Peng, S.H.; Chen, G.Z.; Lee, S.Y. Virtual increase or latent loss? A reassessment of mangrove populations and their conservation in Guangdong, southern China. Mar. Pollut. Bull. 2016, 109, 691–699. [Google Scholar] [CrossRef]
- Zhang, X.M.; Zhou, Y.; Liu, P.; Wang, F.; Liu, B.J.; Liu, X.J.; Yang, H.S. Temporal pattern in biometrics and nutrient stoichiometry of the intertidal seagrass Zostera japonica and its adaptation to air exposure in a temperate marine lagoon (China): Implications for restoration and management. Mar. Pollut. Bull. 2015, 94, 103–113. [Google Scholar] [CrossRef]
- Liu, Z.Z.; Cui, B.S.; He, Q. Shifting paradigms in coastal restoration: Six decades’ lessons from China. Sci. Total Environ. 2016, 566, 205–214. [Google Scholar] [CrossRef]
- Ruiz, G.M.; Carlton, J.T.; Grosholz, E.D.; Hines, A.H. Global invasions of marine and estuarine habitats by non-indigenous species: Mechanisms, extent, and consequences. Integr. Comp. Biol. 1997, 37, 621–632. [Google Scholar] [CrossRef]
- Taylor, M.D.; Fairfax, A.V.; Suthers, I.M. The race for space: Using acoustic telemetry to understand density-dependent emigration and habitat selection in a released predatory. Fish. Rev. Fish. Sci. 2013, 21, 276–285. [Google Scholar] [CrossRef]
- Yang, J.; Gao, J.; Liu, B.; Zhang, W. Sediment deposits and organic carbon sequestration along mangrove coasts of the Leizhou Peninsula, southern China. Estuar. Coast. Shelf Sci. 2014, 136, 3–10. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, Y.; Zhang, S.; Wang, K.; Xu, Q. A comparative study of fish assemblages near aquaculture, artificial and natural habitats. J. Ocean. Univ. China 2015, 14, 149–160. [Google Scholar] [CrossRef]
- FAO. Aquanculture Development 6. Use of Wild Fishery Resources for Capture–Based Aquaculture. FAO Technical Guidelines for Responsible Fisheries; No. 5, Suppl. 6; FAO: Rome, Italy, 2011. [Google Scholar]
- Taranger, G.L.; Karlsen, O.; Bannister, R.J.; Glover, K.A.; Husa, V.; Karlsbakk, E.; Kvamme, B.O.; Boxaspen, K.K.; Bjorn, P.A.; Finstad, B.; et al. Risk assessment of the environmental impact of Norwegian Atlantic salmon farming. ICES J. Mar. Sci. 2015, 72, 997–1021. [Google Scholar] [CrossRef]
- Troell, M.; Joyce, A.; Chopin, T.; Neori, A.; Buschmann, A.H.; Fang, J.G. Ecological engineering in aquaculture–potential for integrated multi-trophic aquaculture (IMTA) in marine offshore systems. Aquaculture 2009, 297, 1–9. [Google Scholar] [CrossRef]
- Funge-Smith, S.; Lindebo, E.; Staples, D. Asian Fisheries Today: The Production and Use of Low Value/Trash Fish from Marine Fisheries in the Asia-Pacific Region; RAP Publication: Bangkok, Thailand; FAO: Bangkok, Thailand, 2005; Available online: www.fao.org/docrep/008/ae934e/ae934e00.htm (accessed on 10 December 2018).
- Hasan, M.R.; Halwart, M. (Eds.) Fish as Feed Inputs for Aquaculture: Practices, Sustainability and Implications; FAO Fisheries and Aquaculture Technical Paper No. 518; FAO: Rome, Italy, 2009; Available online: www.fao.org/docrep/012/i1140e/ i1140e.pdf (accessed on 14 November 2015).
- Dempster, T.; Arechavala-Lopez, P.; Barrett, L.T.; Fleming, L.A.; Sanchez-Jerez, P.; Uglem, I. Recapturing escaped fish from marine aquaculture is largely unsuccessful: Alternatives to reduce the number of escapees in the wild. Rev. Aquac. 2018, 10, 153–167. [Google Scholar] [CrossRef]
- Skilbrei, O.T.; Wennevik, V. The use of catch statistics to monitor the abundance of escaped farmed Atlantic salmon and rainbow trout in the sea. ICES J. Mar. Sci. 2006, 63, 1190–1200. [Google Scholar] [CrossRef][Green Version]
- Abrantes, K.G.; Lyle, J.M.; Nichols, P.D.; Semmens, J.M. Do exotic salmonids feed on native fauna after escaping from aquaculture cages in Tasmania, Australia? Can. J. Fish. Aquat. Sci. 2011, 68, 1539–1551. [Google Scholar] [CrossRef]
- Bolstad, G.H.; Hindar, K.; Robertsen, G.; Jonsson, B.; Sægrov, H.; Diserud, O.H.; Fishe, P.; Jensen, A.J.; Naesje, T.F.; Barlaup, B.T.; et al. Gene flow from domesticated escapes alters the life history of wild Atlantic salmon. Nat. Ecol. Evol. 2017, 1, 124. [Google Scholar] [CrossRef]
- Glover, K.A.; Solberg, M.F.; McGinnity, P.; Hindar, K.; Verspoor, E.; Coulson, M.W.; Hansen, M.M.; Araki, H.; Skaala, Y.; Svåsand, T. Half a century of genetic interaction between farmed and wild Atlantic salmon: Status of knowledge and unanswered questions. Fish Fish. 2017, 18, 890–927. [Google Scholar] [CrossRef]
- Hagen, I.J.; Jensen, A.J.; Bolstad, G.H.; Diserud, O.H.; Hindar, K.; Lo, H.; Karlsson, S. Supplementary stocking selects for domesticated genotypes. Nat. Commun. 2019, 10, 199. [Google Scholar] [CrossRef] [PubMed]
- Clavelle, T.; Lester, S.E.; Gentry, R.; Froehlich, H.E. Interactions and management for the future of marine aquaculture and capture fisheries. Fish Fish. 2019, 20, 368–388. [Google Scholar] [CrossRef]
- Tlusty, M.F.; Andrew, J.; Baldwin, K.; Bradley, T.M. Acoustic conditioning for recall/recapture of escaped Atlantic salmon and rainbow trout. Aquaculture 2008, 274, 57–64. [Google Scholar] [CrossRef]
- Blaxter, J.H. The enhancement of marine fish stocks. Adv. Mar. Biol. 2000, 38, 1–54. [Google Scholar]
- Cheng, J.H.; Jiang, Y.Z. Marine stock enhancement: Review and prospect. J. Fish. Sci. China 2010, 17, 610–617. (In Chinese) [Google Scholar]
- Kitada, S.; Nakajima, K.; Hamasaki, K.; Shishidou, H.; Waples, R.S.; Kishino, H. Rigorous monitoring of a large-scale marine stock enhancement program demonstrates the need for comprehensive management of fisheries and nursery habitat. Sci. Rep. 2019, 9, 5290. [Google Scholar] [CrossRef]
- Lorenzen, K.; Agnalt, A.L.; Blankenship, H.L.; Hines, A.H.; Leber, K.M.; Loneragan, N.R.; Taylor, M.D. Evolving context and maturing science: Aquaculture-based enhancement and restoration enter the marine fisheries management toolbox. Rev. Fish. Sci. 2013, 21, 213–221. [Google Scholar] [CrossRef]
- Mao, Y.Z.; Yang, H.S.; Zhou, Y.; Ye, N.H.; Fang, J.G. Potential of the seaweed Gracilaria lemaneiformis for integrated multi-trophic aquaculture with scallop Chlamys farreri in North China. J. Appl. Phycol. 2009, 21, 649–656. [Google Scholar] [CrossRef]
- Zhou, Y.; Yang, H.; Hu, H.; Liu, Y.; Mao, Y.; Zhou, H.; Xu, X.; Zhang, F. Bioremediation potential of the macroalga Gracilaria lemaneiformis (Rhodophyta) integrated into fed fish culture in coastal waters of north China. Aquaculture 2006, 252, 264–276. [Google Scholar] [CrossRef]
- Ottolenghi, F.; Silvestri, C.; Giordano, P.; Lovatelli, A.; New, M. Capture-Based Aquaculture: The Fattening of Eels, Groupers, Tunas and Yellowtails; FAO: Rome, Italy, 2004. [Google Scholar]
- Airoldi, L.; Balata, D.; Beck, M.W. The Gray zone: Relationships between habitat loss and marine diversity and their applications in conservation. J. Exp. Mar. Biol. Ecol. 2008, 366, 8–15. [Google Scholar] [CrossRef]
- Thompson, R.C.; Crowe, T.P.; Hawkins, S.J. Rocky intertidal communities: Past environmental changes, present status and predictions for the next 25 years. Environ. Conserv. 2002, 29, 168–191. [Google Scholar] [CrossRef]
- Clynick, B.G.; Chapman, M.G.; Underwood, A.J. Effects of Epibiota on assemblages of fish associated with urban structures. Mar. Ecol. Prog. Ser. 2007, 332, 201–210. [Google Scholar] [CrossRef]
- Hernández-Carmona, G.; Garcia, O.; Robledo, D.; Foster, M. Restoration techniques for Macrocystis pyrifera (Phaeophyceae) populations at the southern limit of their distribution in Mexico. Bot. Mar. 2000, 43, 273–284. [Google Scholar] [CrossRef]
- Bi, Y.; Feng, M.; Jiang, R.; Wu, Z.; Wang, W. The effects of sediment on Sargassum horneri in the early life stages on rocky subtidal reefs. Aquat. Bot. 2016, 132, 17–23. [Google Scholar] [CrossRef]
- Deregibus, D.; Quartino, M.L.; Campana, G.L.; Momo, F.R.; Wiencke, C.; Zacher, K. Photosynthetic light requirements and vertical distribution of macroalgae in newly ice-free areas in Potter Cove, South Shetland Islands. Antarct. Polar Biol. 2016, 39, 153–166. [Google Scholar] [CrossRef]
- Deysher, L.E.; Dean, T.A.; Grove, R.S.; Jahn, A. Design considerations for an artificial reef to grow giant kelp (Macrocystis pyrifera) in Southern California. ICES J. Mar. Sci. 2002, 59, S201–S207. [Google Scholar] [CrossRef]
- Pitcher, T.J.; Seaman, W., Jr. Petrarch’s Principle: How protected human-made reefs can help the reconstruction of fisheries and marine ecosystems. Fish 2010, 1, 73–81. [Google Scholar] [CrossRef]
- Chai, Z.Y.; Huo, Y.Z.; He, Q.; Huang, X.W.; Jiang, X.P.; He, P.M. Studies on breeding of Sargassum vachellianum on artificial reefs in Gouqi Island, China. Aquaculture 2014, 424, 189–193. [Google Scholar] [CrossRef]
- Schram, M.W. Factors influencing seaweed responses to eutrophication: Some results from EU-project EUMAC. J. Appl. Phycol. 1999, 11, 69–78. [Google Scholar] [CrossRef]
- Wells, R.J.D.; Steneck, R.S.; Palma, A.T. Three–dimensional resource partitioning between American lobster (Homarus americanus) and rock crab (Cancer irroratus) in a subtidal kelp forest. J. Exp. Mar. Biol. Ecol. 2010, 384, 1–6. [Google Scholar] [CrossRef]
- Bortone, S.A.; Martin, T.; Bundrick, C.M. Factors affecting fish assemblage development on a modular artificial reef in a northern Gulf of Mexico estuary. Bull. Mar. Sci. 1994, 55, 319–332. [Google Scholar]
- Duggins, D.O.; Gómez-Buckley, M.C.; Buckley, R.M.; Lowe, A.T.; Galloway, A.W.E.; Dethier, M.N. Islands in the stream: Kelp detritus as faunal magnets. Mar. Biol. 2016, 163, 17. [Google Scholar] [CrossRef]
- Duggins, D.O.; Eckman, J.E. The role of kelp detritus in the growth of benthic suspension feeders in an understory kelp forest. J. Exp. Mar. Biol. Ecol. 1994, 176, 53–68. [Google Scholar] [CrossRef]
- Garbary, D.J.; Galway, M.E.; Halat, L. Response to Ugarte et al.: Ascophyllum (Phaeophyceae) annually contributes over 100% of its vegetative biomass to detritus. Phycologia 2017, 56, 116–118. [Google Scholar] [CrossRef]
- Abdullah, M.I.; Fredriksen, S.; Christie, H. The impact of the kelp (Laminaria hyperborea) forest on the organic matter content in sediment of the west coast of Norway. Mar. Biol. Res. 2017, 13, 151–160. [Google Scholar] [CrossRef]
- Wang, W.D.; Liang, J.; Bi, Y.X.; Feng, M.P.; Zhou, S.S.; Yu, B.C. Status and Prospects of Marine Ranching Construction in Zhejiang Province. J. Zhejiang Ocean Univ. 2016, 3, 181–185. (In Chinese) [Google Scholar]
- Pursche, A.R.; Suthers, I.M.; Taylor, M.D. The effect of targeted stocking on behavior and space utilization of a released finfish. ICES J. Mar. Sci. 2014, 71, 1100–1106. [Google Scholar] [CrossRef]
- Bohnsack, J.A. Maintenance and recovery of reef fishery productivity. In Reef Fisheries; Polunin, N.V.C., Roberts, C.M., Eds.; Chapman and Hall: London, UK, 1996; pp. 283–313. [Google Scholar]
- Masuda, R.; Tsukamoto, K. Stock enhancement in Japan—Review and perspective. Bull. Mar. Sci. 1998, 62, 337–358. [Google Scholar]
- Munro, J.L.; Bell, J.D. Enhancement of marine fisheries resources. Rev. Fish. Sci. 1997, 5, 185–222. [Google Scholar] [CrossRef]
- Kristiansen, T.S. Enhancement studies of coastal cod (Gadus morhua, L.) in Nord-Trøndelag, Norway. In Stock Enhancement and Sea Ranching; Howell, B.R., Moksness, E., Svåsand, T., Eds.; Blackwell Scientific Publications: Oxford, UK, 1999; pp. 277–292. [Google Scholar]
- Gabriele, L.M.; Alessandro, L.; Francesco, S.; Giovanna, M. First release of hatchery juveniles of the dusky grouper Epinephelus marginatus (Lowe, 1834) (Serranidae: Teleostei) at artificial reefs in the Mediterranean: Results from a pilot study. Sci. Mar. 2008, 72, 743–756. [Google Scholar]
- Hu, Q.S.; Rahman, H.A.; Jiang, Y.Z.; Zhang, S.Y.; Shentu, J.K. Acoustic Conditioning System Development and Conditioning Experiments on Black Seabreams in the Xiangshan Bay Sea Ranch. J. Ocean Univ. China 2018, 17, 667–674. [Google Scholar] [CrossRef]
- Jiang, Z.Y.; Zhang, G.S.; Liang, Z.L. Acoustic taming on Chrysophrys major by rectangular continuant of 300 Hz. J. Fish. Sci. China 2008, 15, 86–90. (In Chinese) [Google Scholar]
- Dance, K.M.; Rooker, J.R.; Shipley, J.B.; Dance, M.A.; Wells, R.J.D. Feeding ecology of fishes associated with artificial reefs in the northwest Gulf of Mexico. PLoS ONE 2018, 13, e0203873. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.J.; Zhang, S.Y.; Wang, X.; Jiang, R.J.; Zhao, J. The feeding behaviour and ecological function during summer of one herbivore on seaweed bed in Gouqi Island: The gastropod, Turbo cornutus Solander. J. Fish. China 2015, 39, 511–519. [Google Scholar]
- Burke, N.C. Nocturnal foraging habitats of French and bluestriped grunts, Haemulon flavolineatum and H. sciurus, at Tobacco Caye, Belize. Env. Biol. Fishes 1995, 42, 365–374. [Google Scholar] [CrossRef]
- Rosemond, R.C.; Paxton, A.B.; Lemoine, H.R.; Fegley, S.R.; Peterson, C.H. Fish use of reef structures and adjacent sand flats: Implications for selecting minimum buffer zones between new artificial reefs and existing reefs. Mar. Ecol. Prog. 2018, 587, 187–199. [Google Scholar] [CrossRef]
- Xu, J.; Xia, M.; Ning, X.; Mathews, C.P. Stocking, enhancement, and mariculture of Penaeus orientalis and other species in Shanghai and Zhejiang provinces, China. Mar. Fish. Rev. 1997, 59, 8–14. [Google Scholar]
- Huang, X.Y.; Wang, Z.J.; Liu, Y.; Hu, W.T.; Ni, W. On the use of blast furnace slag and steel slag in the preparation of green artificial reef concrete. Constr. Build. Mater. 2016, 112, 241–246. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, Y.P.; Dong, G.H.; Guan, C.T.; Cui, Y.; Xu, T.J. A study of the flow field characteristics around star-shaped artificial reefs. J. Fluids Struct. 2013, 39, 27–40. [Google Scholar] [CrossRef]
- Wu, C.W.; Dong, Z.Y.; Chi, C.F.; Ding, F. Reproductive and spawning habits of Sepiella maindroni off Zhejiang, China. Oceanol. Limnol. Sin. 2010, 41, 39–46. (In Chinese) [Google Scholar]
- Wang, Q.Y.; Zhuang, Z.Z.; Deng, J.; Ye, Y.M. Stock enhancement and translocation of the shrimp Penaeus chinensis in China. Fish. Res. 2006, 80, 67–79. [Google Scholar] [CrossRef]
- Lü, H.; Xu, J.; Vander Haegen, G. Supplementing Marine Capture Fisheries in the East China Sea: Sea Ranching of Prawn Penaeus orientalis, Restocking of Large Yellow Croaker Pseudosciaena crocea, and Cage Culture. Rev. Fish. Sci. 2008, 16, 366–376. [Google Scholar] [CrossRef]
- Rillahan, C.; Chambers, M.D.; Howell, W.H.; Watson, W.H. The behavior of cod (Gadus morhua) in an offshore aquaculture net pen. Aquaculture 2011, 310, 361–368. [Google Scholar] [CrossRef]
- Bridger, C.J.; Booth, R.K.; McKinley, R.S.; Scruton, D.A.; Lindstrom, R.T. Monitoring fish behavior with a remote, combined acoustic/radio biotelemetry system. J. Appl. Ichthyol. 2001, 17, 126–129. [Google Scholar] [CrossRef]
- Zion, B.; Barki, A. Ranching fish using acoustic conditioning: Has it reached a dead end? Aquaculture 2012, 344, 3–11. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, S.Y.; Wang, W.D.; Yu, B.C. Experimental study on the juvenile Sparus macrocephalus’s tendency to different structural spaces of artificial reef. J. Fish. China 2010, 34, 1762–1768. [Google Scholar]
- Plagányi, É.E.; Punt, A.E.; Hillary, R.; Morello, E.B.; Thébaud, O.; Hutton, T.; Pillans, R.D.; Thorson, J.T.; Fulton, E.A.; Smith, A.D.; et al. Multispecies fisheries management and conservation: Tactical applications using models of intermediate complexity. Fish 2014, 15, 1–22. [Google Scholar] [CrossRef]
- Smith, A.D.M.; Fulton, E.J.; Hobday, A.J.; Smith, D.C.; Shoulder, P. Scientific tools to support the practical implementation of ecosystem-based fisheries management. ICES J. Mar. Sci. 2007, 64, 633–639. [Google Scholar] [CrossRef]
- FAO. Fisheries Technical Guidelines for Responsible Fisheries; No. 4, Suppl. 2, Add. 1; FAO: Rome, Italy, 2008. [Google Scholar]
- Ogle, K.; Tucker, C.; Cable, J.M. Beyond simple linear mixing models: Process-based isotope partitioning of ecological processes. Ecol. Appl. 2014, 24, 181–195. [Google Scholar] [CrossRef]
- Zhou, X.J.; Liu, Y.M.; Wang, K.; Zhao, J.; Zhao, X.; Zhang, S.Y. Re-Evaluation of the Impacts of Dietary Preferences on Macroinvertebrate Trophic Sources: An Analysis of Seaweed Bed Habitats Using the Integration of Stable Isotope and Observational Data. Sustainability 2018, 10, 2010. [Google Scholar] [CrossRef]
- Zhao, J.; Cao, J.; Tian, S.Q.; Chen, Y.; Zhang, S.Y.; Zhou, X.J. A comparison between two GAM models in quantifying relationships of environmental variables with fish richness and diversity indices. Aquat. Ecol. 2014, 48, 297–312. [Google Scholar] [CrossRef]
- Zhao, J.; Liang, J.L.; Zhou, X.J.; Zhao, X.; Zhang, S.Y. Research on relationship between dominant crustacean species and environmental factors based on GAM. South China Fish. Sci. 2017, 13, 26–35. (In Chinese) [Google Scholar]
- Lin, J.; Li, C.Y.; Zhang, S.Y. Hydrodynamic effect of a large offshore mussel suspended aquaculture farm. Aquaculture 2016, 451, 147–155. [Google Scholar] [CrossRef]
- Zeng, X.; Zhang, S.Y.; Wang, Z.H.; Lin, J.; Wang, K. Habitat suitability assessment of Sebasticus marmoratus in the rocky reef region of the Ma’an Archipelago. Acta Ecol. Sin. 2016, 36, 3765–3774. (In Chinese) [Google Scholar]
- Zeng, X.; Tanaka, K.R.; Chen, Y.; Wang, K.; Zhang, S.Y. Gillnet data enhance performance of rockfishes habitat suitability index model derived from bottom-trawl survey data: A case study with, Sebasticus marmoratus. Fish. Res. 2018, 204, 189–196. [Google Scholar] [CrossRef]
- Rademeyer, R.A.; Plagányi, É.E.; Butterworth, D.S. Tips and tricks in designing management procedures. ICES J. Mar. Sci. 2017, 64, 618–625. [Google Scholar] [CrossRef]
- Plagányi, É.E. Models for an Ecosystem Approach to Fisheries; FAO Technical Paper. No. 477; FAO: Rome, Italy, 2017. [Google Scholar]
- Zhao, S.; Song, K.; Gui, F.; Cai, H.; Jin, W.; Wu, C.W. The energy ecological footprint for small fish farm in China. Ecol. Indic. 2013, 29, 62–67. [Google Scholar] [CrossRef]
- Xu, G.F.; Cui, Y.P.; Liu, L. Assessment of eutrophication status by using the Pressure-Status-Response Model in Xiangshan Bay, China. J. Trop. Oceanogr. 2018, 37, 52–60. (In Chinese) [Google Scholar]
- Wei, C.; Ye, S.F.; Guo, Z.Y.; Liu, H.Q.; Deng, B.P.; Liu, X. Constructing an assessment indices system to analyze integrated regional carrying capacity in the coastal zones: A case in Nantong. Acta Ecol. Sin. 2013, 33, 5893–5904. (In Chinese) [Google Scholar] [CrossRef]
- Christensen, V.; Walters, C.; Pauly, D. Ecopath with Ecosim: A User’s Guide; Fisheries Centre; University of British Columbia: Vancouver, BC, Canada, 2005. [Google Scholar]
- Wang, X.; Zhao, X.; Zhang, S.Y.; Zhou, X.J. Distribution pattern of dissolved carbon, nitrogen and phosphorus in mussel culture areas of Gouqi Island. J. Fish. China 2015, 39, 1650–1664. (In Chinese) [Google Scholar]
- Jiang, R.J.; Zhang, S.Y.; Bi, Y.X.; Wang, Z.H.; Zhou, X.J.; Zhao, X.; Chen, L.R. Food sources of small invertebrates in the macroalgal bed of Gouqi Island. J. Fish. China 2015, 39, 1487–1498. (In Chinese) [Google Scholar]
- Zhao, J.; Zhang, S.Y.; Xu, M. The primary research of the energy flow in Gouqi kelp bed ecosystem. J. Shanghai Ocean Univ. 2010, 19, 98–104. (In Chinese) [Google Scholar]
- Shih, N.T.; Cai, Y.H.; Ni, I.H. A concept to protect fisheries recruits by seasonal closure during spawning periods for commercial fishes off Taiwan and the East China Sea. J. Appl. Ichthyol. 2009, 25, 676–685. [Google Scholar] [CrossRef]
- Zhao, J.; Cao, J.; Tian, S.Q.; Chen, Y.; Zhang, S.Y.; Zhou, X.J. Influence of sampling frequency on detectability of fish community and fish species in a fishery-independent survey. Aquac. Fish. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Zhao, J.; Cao, J.; Tian, S.Q.; Chen, Y.; Zhang, S.Y. Evaluating Sampling Designs for Demersal Fish Communities. Sustainability 2018, 10, 2585. [Google Scholar] [CrossRef]
- Shi, Y.; Li, H.; Yang, Y.Q.; Du, W.C.; Yang, Y.F.; Li, P.X. Design and Implementation of Multi-parameter Intelligent Monitoring System for Marine Ranching. Transducer Microsyst. Technol. 2017, 36, 70–76. [Google Scholar]
- Chen, X.Y.; Gao, H.W.; Yao, X.H.; Chen, Z.H.; Fang, H.D.; Ye, S.F. Ecosystem Health Assessment in the Pearl River Estuary of China by Considering Ecosystem Coordination. PLoS ONE 2013, 8, e70547. [Google Scholar] [CrossRef]
- Chen, X.J.; Li, G.; Feng, B.; Tian, S.Q. Habitat suitability index of Chub mackerel (Scomber japonicus) from July to September in the East China Sea. J. Oceanogr. 2009, 65, 93–102. [Google Scholar] [CrossRef]
- Yen, K.W.; Lu, H.J.; Chang, Y.; Lee, M.A. Using remote-sensing data to detect habitat suitability for yellowfin tuna in the Western and Central Pacific Ocean. Int. J. Remote Sens. 2012, 33, 7507–7522. [Google Scholar] [CrossRef]
- Kim, K.; Kim, Y.B.; Park, J.J.; Park, K.A.; Nam, S.H.; Chang, K., II. Long-term and real-time monitoring system of the East/Japan Sea. Ocean Sci. J. 2005, 40, 25–44. [Google Scholar] [CrossRef]
- Zhu, J. Overview of Regional and Coastal Systems. In Operational Oceanography in the 21st Century; Springer: Dutch, The Netherlands, 2011. [Google Scholar]
- Xu, H. Development Strategy for Aquaculture Facility and Deepwater Aquaculture Platform. Eng. Sci. 2016, 18, 37–42. (In Chinese) [Google Scholar]
- Wu, L.B.; Liu, Y.S.; Liu, X.Z.; Yang, J.D.; Zhang, G.S.; Li, S.D. Design of feeding system with remote monitoring in marine ranching. Fish. Mod. 2010, 37, 23–25. (In Chinese) [Google Scholar]
- Zhao, J.; Zhang, S.Y.; Lin, J.; Zhou, X.J. A comparative study of different sampling designs in fish community estimation. Chin. J. Appl. Ecol. 2014, 25, 1181–1187. (In Chinese) [Google Scholar]
- Maxwell, S.M.; Hazen, E.L.; Lewison, R.L.; Dunn, D.C.; Bailey, H.; Bograd, S.J.; Briscoe, D.K.; Fossette, S.; Hobday, A.J.; Bennett, M.; et al. Dynamic ocean management: Defining and conceptualizing real-time management of the ocean. Mar. Policy 2015, 58, 42–50. [Google Scholar] [CrossRef]
- McCann, K.S.; Rasmussen, J.B.; Umbanhowar, J. The dynamics of spatially coupled food webs. Ecol. Lett. 2005, 8, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Hooper, D.U.; Chapin, F.S.; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.H.; Lodge, D.M.; Loreau, M.; Naeem, S. Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecol. Monogr. 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Underwood, A.J.; Fairweather, P.G. Supply-side ecology and benthic marine assemblages. Trends Ecol. Evol. 1989, 4, 16–20. [Google Scholar] [CrossRef]
- France, K.E.; Duffy, J.E. Diversity and dispersal interactively affect predictability of ecosystem function. Nature 2006, 441, 1139–1143. [Google Scholar] [CrossRef]
- Aydin, K.Y.; McFarlane, G.A.; King, J.R.; Megrey, B.A.; Myers, K.W. Linking oceanic food webs to coastal production and growth rates of Pacific salmon (Oncorhynchus spp.), using models on three scales. Deep-Sea Res. Part II: Top. Stud. Oceanogr. 2005, 52, 757–780. [Google Scholar] [CrossRef]
- Walters, C.J.; Christensen, V.; Martell, S.J. Possible ecosystem impacts of applying MSY policies from single-species assessment. ICES J. Mar. Sci. 2005, 62, 558–568. [Google Scholar] [CrossRef]
- Martínez, M.L.; Intralawan, A.; Vázquez, G.; Pérez-Maqueo, O.; Sutton, P.; Landgrave, R. The coasts of our world: Ecological, economic and social importance. Ecol. Econ. 2007, 63, 254–272. [Google Scholar] [CrossRef]


| Aims | Tools |
|---|---|
| Conserve or protect ecological structures to enhance stock and biodiversity | Ecosystem approaches to fishery (EAF), marine ranching spatial planning |
| Protect ecologically valuable habitats and restore degraded habitats | Conservation and restoration of typical habitats (e.g., seaweed bed, mangrove, seagrass, etc.) |
| Promote appropriate use of marine space | Integrated multi-trophic aquaculture (IMTA), multi-trophic and spatial planning based marine ranching |
| Conserve vulnerable or regionally extinct species | Special marine protected areas (SMPA), species enhancement, and releasing |
| Construct high biological productivity habitats for the sustainable use of fishery resources | Artificial structure construction (e.g., artificial reefs, cages, buoyant rafts, artificial seaweed, mangroves, seagrass habitats). |
| Avoid and resolve social-economic conflicts | Spatial planning public or stakeholder engagement and regulatory system |
| Management Tools and Measures |
|---|
| Input control |
| Restriction or bans on certain fishing gear (including mesh size) or modes of fishing |
| Good stock enhancement practices, including artificial reefs and releasing |
| Time/areas closures |
| Zoning of areas of biological importance to wild caught seed or broodstock |
| Closed area (seasonal in typical habitats and permanently in MPA) |
| Protection and restoration of key seed settlements and nursery habitats |
| Output control |
| Restriction on certain protected or threatened species |
| Market-related measures |
| Traceability of products |
| Use of the Internet of Things and Internet + to consider of the potential power of consumer preferences |
| Good mariculture practices, including sustainable sourcing and use of feed |
| Certification system for geographical indications protection products |
| Other measures |
| Improve harvest, transport, and cultured practices to reduce wastage |
| Pollution controls |
| Control of disease |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, X.; Zhao, X.; Zhang, S.; Lin, J. Marine Ranching Construction and Management in East China Sea: Programs for Sustainable Fishery and Aquaculture. Water 2019, 11, 1237. https://doi.org/10.3390/w11061237
Zhou X, Zhao X, Zhang S, Lin J. Marine Ranching Construction and Management in East China Sea: Programs for Sustainable Fishery and Aquaculture. Water. 2019; 11(6):1237. https://doi.org/10.3390/w11061237
Chicago/Turabian StyleZhou, Xijie, Xu Zhao, Shouyu Zhang, and Jun Lin. 2019. "Marine Ranching Construction and Management in East China Sea: Programs for Sustainable Fishery and Aquaculture" Water 11, no. 6: 1237. https://doi.org/10.3390/w11061237
APA StyleZhou, X., Zhao, X., Zhang, S., & Lin, J. (2019). Marine Ranching Construction and Management in East China Sea: Programs for Sustainable Fishery and Aquaculture. Water, 11(6), 1237. https://doi.org/10.3390/w11061237




