A Review on the Current Knowledge and Prospects for the Development of Improved Detection Methods for Soil-Transmitted Helminth Ova for the Safe Reuse of Wastewater and Mitigation of Public Health Risks
Abstract
1. Introduction
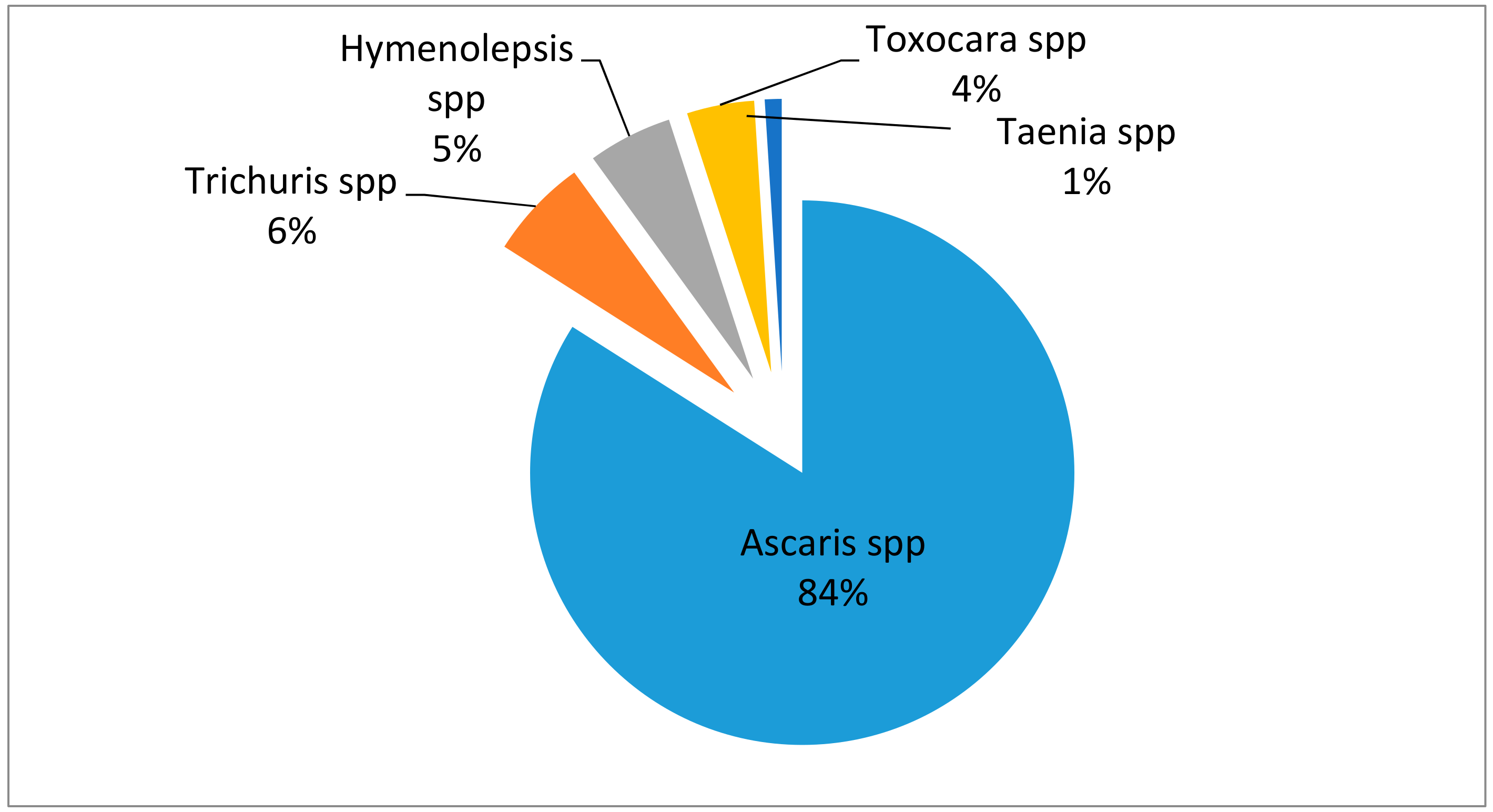
2. Soil-Transmitted Helminths: Prevalence and Health Risks to Humans
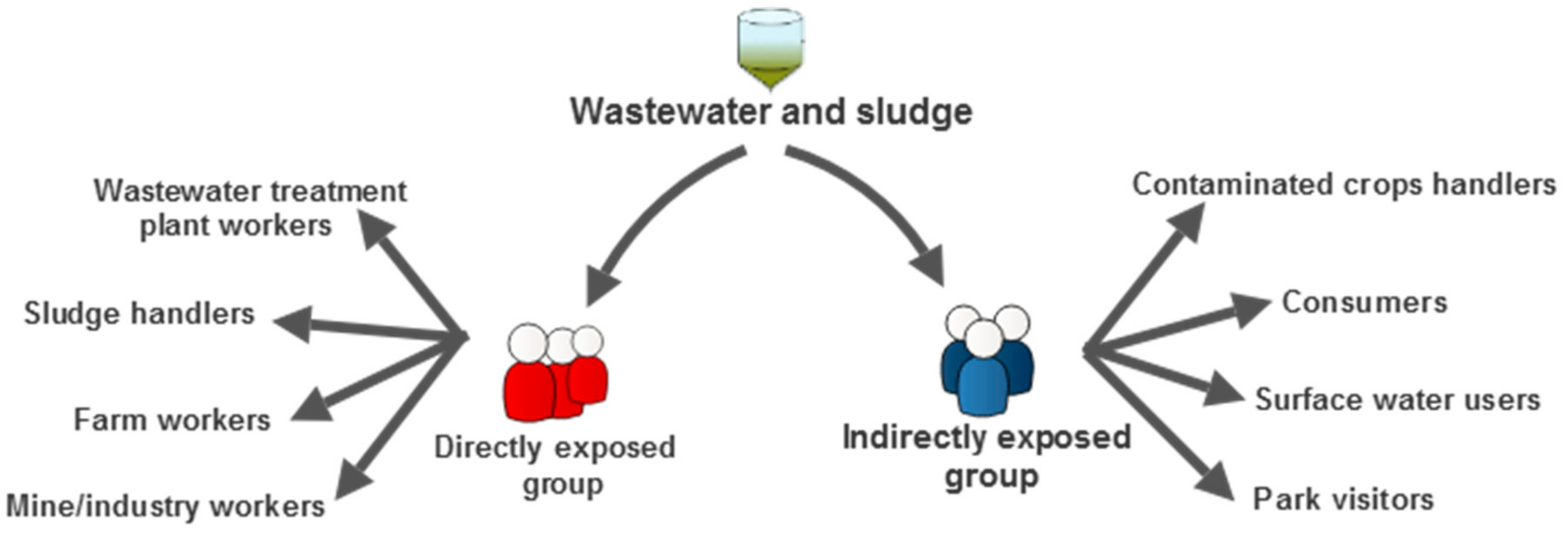
3. Mitigation of Public Health Risk
4. Recovery Methods for STH Ova
4.1. Sampling of Wastewater/Sludge
4.2. Ova Separation from Solid Matrix
4.3. Filtration
4.4. Sedimentation
4.5. Flotation
4.6. Phase Extraction
4.7. Factors Influencing Ova Recovery
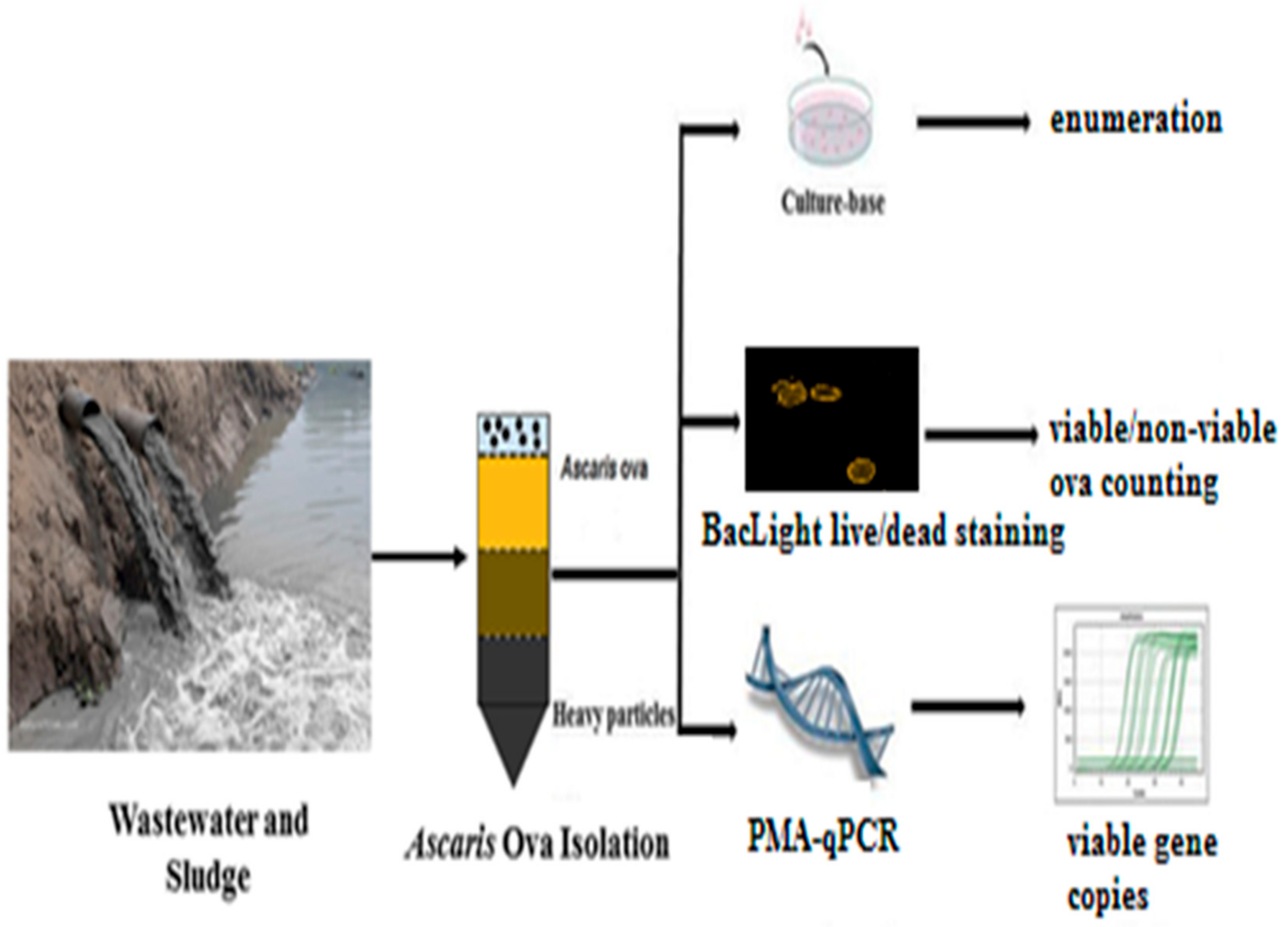
5. Detection Methods for Helminth Ova
5.1. Optical Microscopy
5.2. PCR-Based
5.3. Flow Cytometry
6. Implementation of Innovative Techniques
6.1. Digital PCR
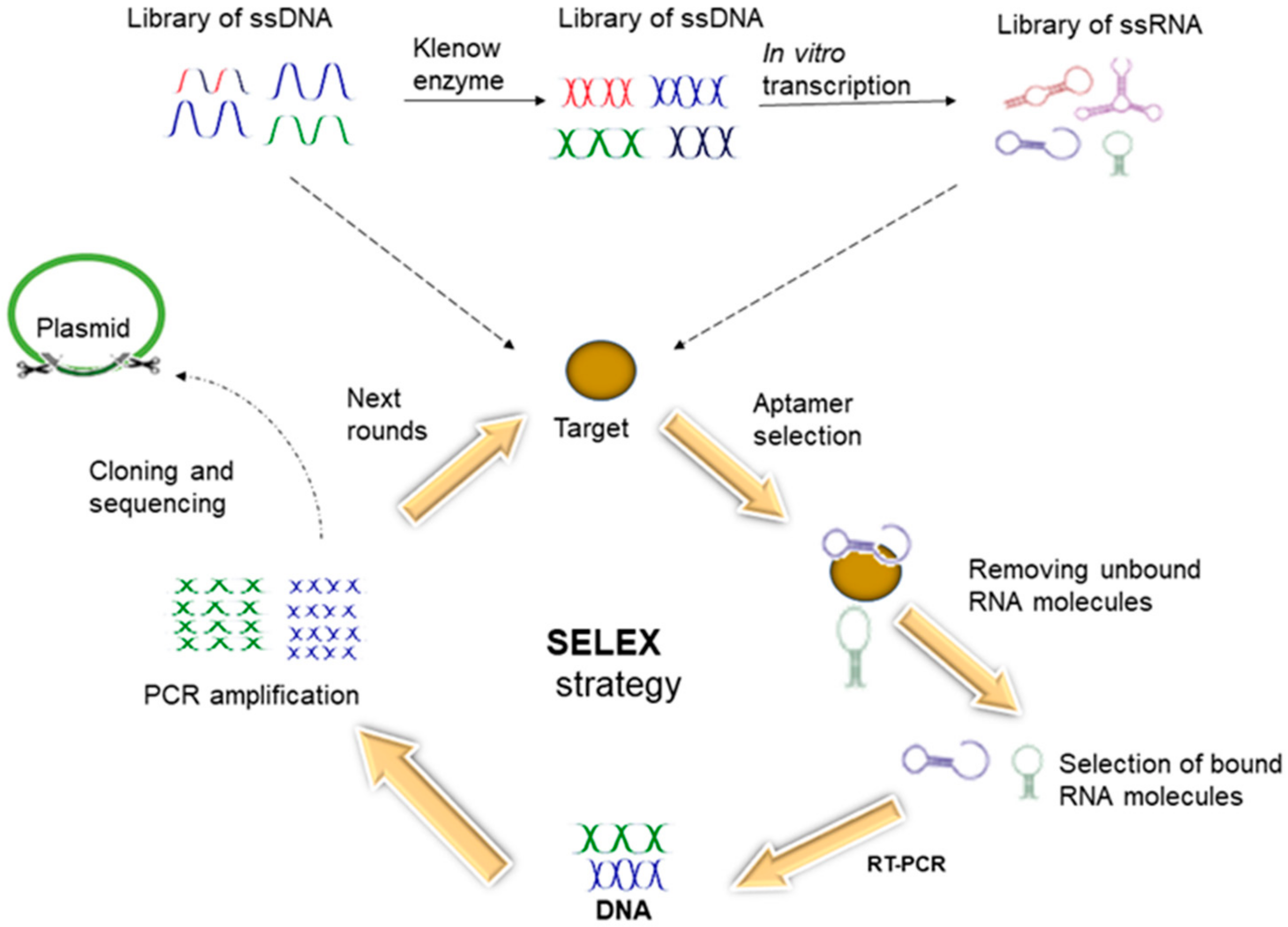
6.2. Aptamers
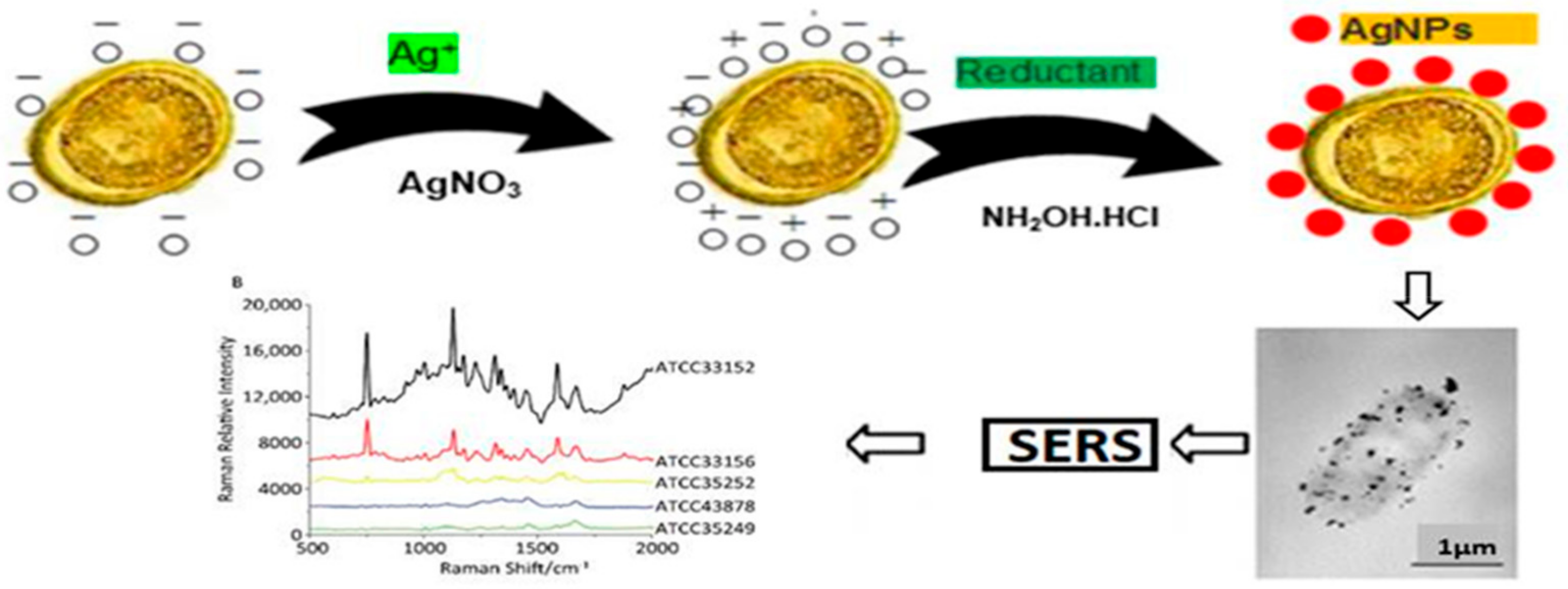
6.3. Gold Nanoparticle-Based Colorimetric Biosensors
6.4. Surface Enhanced Raman Scattering (SERS)
6.5. Smartphone-Based Detection
6.6. Isothermal Amplification Assays
6.7. Paper-Based Sensors
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gyawali, P. Infectious helminth ova in wastewater and sludge: A review on public health issues and current quantification practices. Water Sci. Technol. 2018, 77, 1048–1061. [Google Scholar] [CrossRef] [PubMed]
- Trang do, T.; van der Hoek, W.; Cam, P.D.; Vinh, K.T.; Hoa, N.V.; Dalsgaard, A. Low risk for helminth infection in wastewater-fed rice cultivation in Vietnam. J. Water Health 2006, 4, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, B. Helminth ova removal from wastewater for agriculture and aquaculture reuse. Water Sci. Technol. 2007, 55, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Navarro, I.; Jimenez, B.; Lucario, S.; Cifuentes, E. Application of Helminth ova infection dose curve to estimate the risks associated with biosolid application on soil. J. Water Health 2009, 7, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Stevens, D.P.; Surapaneni, A.; Thodupunuri, R.; O’Connor, N.A.; Smith, D. Helminth log reduction values for recycling water from sewage for the protection of human and stock health. Water Res. 2017, 125, 501–511. [Google Scholar] [CrossRef]
- Rocha, M.C.V.d.; Barés, M.E.; Braga, M.C.B. Quantification of viable helminth eggs in samples of sewage sludge. Water Res. 2016, 103, 245–255. [Google Scholar] [CrossRef] [PubMed]
- WHO. Integrated Guide to Sanitary Parasitology; WHO: Geneva, Switzerland, 2004. [Google Scholar]
- WHO. Guidelines foe the safe use of wastewater, excreta and greywater. In Wastewater Use in Agriculture; WHO: Geneva, Switzerland, 2006; Volume 2. [Google Scholar]
- Ravindran, V.B.; Surapaneni, A.; Crosbie, N.D.; Schmidt, J.; Soni, S.K.; Ball, A.S. A modified approach to recover and enumerate Ascaris ova in wastewater and sludge. PLoS Negl. Trop. Dis. 2019, 13, e0007020. [Google Scholar] [CrossRef]
- Collender, P.A.; Kirby, A.E.; Addiss, D.G.; Freeman, M.C.; Remais, J.V. Methods for Quantification of Soil-Transmitted Helminths in Environmental Media: Current Techniques and Recent Advances. Trends Parasitol. 2015, 31, 625–639. [Google Scholar] [CrossRef]
- Bethony, J.; Brooker, S.; Albonico, M.; Geiger, S.M.; Loukas, A.; Diemert, D.; Hotez, P.J. Soil-transmitted helminth infections: Ascariasis, trichuriasis, and hookworm. Lancet 2006, 367, 1521–1532. [Google Scholar] [CrossRef]
- Strunz, E.C.; Addiss, D.G.; Stocks, M.E.; Ogden, S.; Utzinger, J.; Freeman, M.C. Water, Sanitation, Hygiene, and Soil-Transmitted Helminth Infection: A Systematic Review and Meta-Analysis. PLoS Med. 2014, 11, e1001620. [Google Scholar] [CrossRef]
- Steinbaum, L.; Kwong, L.H.; Ercumen, A.; Negash, M.S.; Lovely, A.J.; Njenga, S.M.; Boehm, A.B.; Pickering, A.J.; Nelson, K.L. Detecting and enumerating soil-transmitted helminth eggs in soil: New method development and results from field testing in Kenya and Bangladesh. PLoS Negl. Trop. Dis. 2017, 11, e0005522. [Google Scholar] [CrossRef] [PubMed]
- Wardell, R.; Clements, A.C.; Lal, A.; Summers, D.; Llewellyn, S.; Campbell, S.J.; McCarthy, J.; Gray, D.J.; Nery, S.V. An environmental assessment and risk map of Ascaris lumbricoides and Necator americanus distributions in Manufahi District, Timor-Leste. PLoS Negl. Trop. Dis. 2017, 11, e0005565. [Google Scholar] [CrossRef] [PubMed]
- Jex, A.R.; Liu, S.; Li, B.; Young, N.D.; Hall, R.S.; Li, Y.; Yang, L.; Zeng, N.; Xu, X.; Xiong, Z.; et al. Ascaris suum draft genome. Nature 2011, 479, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Stolk, W.A.; Kulik, M.C.; le Rutte, E.A.; Jacobson, J.; Richardus, J.H.; de Vlas, S.J.; Houweling, T.A. Between-Country Inequalities in the Neglected Tropical Disease Burden in 1990 and 2010, with Projections for 2020. PLoS Negl. Trop. Dis. 2016, 10, e0004560. [Google Scholar] [CrossRef] [PubMed]
- Mascarini-Serra, L. Prevention of Soil-transmitted Helminth Infection. J. Glob. Infect. Dis. 2011, 3, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Hotez, P.J.; Alvarado, M.; Basáñez, M.G.; Bolliger, I.; Bourne, R.; Boussinesq, M.; Brooker, S.J.; Brown, A.S.; Buckle, G.; Budke, C.M.; et al. The Global Burden of Disease Study 2010: Interpretation and Implications for the Neglected Tropical Diseases. PLoS Negl. Trop. Dis. 2014, 8, e2865. [Google Scholar] [CrossRef] [PubMed]
- Hotez, P.J.; Damania, A.; Barua, A.; Stanaway, J. The first “London Declaration”: The Commonwealth and its neglected tropical diseases. PLoS Negl. Trop. Dis. 2017, 11, e0005321. [Google Scholar] [CrossRef] [PubMed]
- Jia, T.W.; Melville, S.; Utzinger, J.; King, C.H.; Zhou, X.N. Soil-Transmitted Helminth Reinfection after Drug Treatment: A Systematic Review and Meta-Analysis. PLoS Negl. Trop. Dis. 2012, 6, e1621. [Google Scholar] [CrossRef]
- Dunn, J.C.; Turner, H.C.; Tun, A.; Anderson, R.M. Epidemiological surveys of, and research on, soil-transmitted helminths in Southeast Asia: A systematic review. Parasit. Vectors 2016, 9, 31. [Google Scholar] [CrossRef]
- Jimenez, B. Helminth ova control in sludge: A review. Water Sci. Technol. 2007, 56, 147–155. [Google Scholar] [CrossRef]
- Sidhu, J.P.; Toze, S.G. Human pathogens and their indicators in biosolids: A literature review. Environ. Int. 2009, 35, 187–201. [Google Scholar] [CrossRef] [PubMed]
- Hanjra, M.A.; Blackwell, J.; Carr, G.; Zhang, F.; Jackson, T.M. Wastewater irrigation and environmental health: Implications for water governance and public policy. Int. J. Hyg. Environ. Health 2012, 215, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, D.L.; Penney, N.; McLaughlin, M.J.; Rigby, H.; Schwarz, K. Land application of sewage sludge (biosolids) in Australia: Risks to the environment and food crops. Water Sci. Technol. 2010, 62, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Karkashan, A.; Khallaf, B.; Morris, J.; Thurbon, N.; Rouch, D.; Smith, S.R.; Deighton, M. Comparison of methodologies for enumerating and detecting the viability of Ascaris eggs in sewage sludge by standard incubation-microscopy, the BacLight Live/Dead viability assay and other vital dyes. Water Res. 2015, 68, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Tran, Q.K.; Schwabe, K.A.; Jassby, D. Wastewater Reuse for Agriculture: Development of a Regional Water Reuse Decision-Support Model (RWRM) for Cost-Effective Irrigation Sources. Environ. Sci. Technol. 2016, 50, 9390–9399. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, N.A.; Surapaneni, A.; Smith, D.; Stevens, D. Occurrence and fate of Ascaris lumbricoides ova in biosolids in Victoria, Australia: A human health risk assessment of biosolids storage periods. Water Sci. Technol. 2017, 76, 1332–1346. [Google Scholar] [CrossRef] [PubMed]
- Maya, C.; Torner-Morales, F.J.; Lucario, E.S.; Hernández, E.; Jiménez, B. Viability of six species of larval and non-larval helminth eggs for different conditions of temperature, pH and dryness. Water Res. 2012, 46, 4770–4782. [Google Scholar] [CrossRef]
- Amoah, I.D.; Singh, G.; Stenström, T.A.; Reddy, P. Detection and quantification of soil-transmitted helminths in environmental samples: A review of current state-of-the-art and future perspectives. Acta Trop. 2017, 169, 187–201. [Google Scholar] [CrossRef]
- Wharton, D.A.; Jenkins, T. Structure and Chemistry of the Egg-Shell of a Nematode (Trichuris Suis). Tissue Cell 1978, 10, 427–440. [Google Scholar] [CrossRef]
- Wharton, D.A. The production and functional morphology of helminth egg-shells. Parasitology 1983, 86, 85–97. [Google Scholar] [CrossRef]
- Gupta, N.; Khan, D.K.; Santra, S.C. Prevalence of intestinal helminth eggs on vegetables grown in wastewater-irrigated areas of Titagarh, West Bengal, India. Food Control 2009, 20, 942–945. [Google Scholar] [CrossRef]
- Konaté, Y.; Maiga, A.H.; Basset, D.; Casellas, C.; Picot, B. Parasite removal by waste stabilisation pond in Burkina Faso, accumulation and inactivation in sludge. Ecol. Eng. 2013, 50, 101–106. [Google Scholar] [CrossRef]
- Sharafi, K.; Fazlzadehdavil, M.; Pirsaheb, M.; Derayat, J.; Hazrati, S. The comparison of parasite eggs and protozoan cysts of urban raw wastewater and efficiency of various wastewater treatment systems to remove them. Ecol. Eng. 2012, 244, 244–248. [Google Scholar] [CrossRef]
- Kouraa, A.; Fethi, F.; Fahde, A.; Lahlou, A.; Ouazzani, N. Reuse of urban wastewater treated by a combined stabilisation pond system in Benslimane (Morocco). Urban Water 2002, 4, 373–378. [Google Scholar] [CrossRef]
- Ayed, L.B.; Schijven, J.; Alouini, Z.; Jemli, M.; Sabbahi, S. Presence of parasitic protozoa and helminth in sewage and efficiency of sewage treatment in Tunisia. Parasitol. Res. 2009, 105, 393. [Google Scholar] [CrossRef] [PubMed]
- Gantzer, C.; Gaspard, P.; Galvez, L.; Huyard, A.; Dumouthier, N.; Schwartzbrod, J. Monitoring of bacterial and parasitological contamination during various treatment of sludge. Water Res. 2001, 35, 3763–3770. [Google Scholar] [CrossRef]
- Rivera, A.; Al-Shaiily, M.; Daag, A.; Al-Amri, M. Diversity and dimesnions of nematode ova: Atool in wastewater management. J. Appl. Sci. Environ. Sanit. 2012, 7, 21–28. [Google Scholar]
- Amoah, I.D.; Reddy, P.; Seidu, R.; Stenström, T.A. Removal of helminth eggs by centralized and decentralized wastewater treatment plants in South Africa and Lesotho: Health implications for direct and indirect exposure to the effluents. Environ. Sci. Pollut. Res. Int. 2018, 25, 12883–12895. [Google Scholar] [CrossRef] [PubMed]
- Gyawali, P.; Ahmed, W.; Sidhu, J.P.S.; Nery, S.V.; Clements, A.C.; Traub, R.; McCarthy, J.S.; Llewellyn, S.; Jagals, P.; Toze, S. Quantitative detection of viable helminth ova from raw wastewater, human feces, and environmental soil samples using novel PMA-qPCR methods. Environ. Sci. Pollut. Res. 2016, 23, 18639–18648. [Google Scholar] [CrossRef]
- AGWR. National Guidelines Foe Water Recycling: Managing Health and Environmental Risks. 2006. Available online: https://www.awa.asn.au/Documents/water-recycling-guidelines-health-environmental-21.pdf (accessed on 7 January 2019).
- Mes, T.H. Technical variability and required sample size of helminth egg isolation procedures. Vet. Parasitol. 2003, 115, 311–320. [Google Scholar] [CrossRef]
- Sengupta, M.E.; Thamsborg, S.M.; Andersen, T.J.; Olsen, A.; Dalsgaard, A. Sedimentation of helminth eggs in water. Water Res. 2011, 45, 4651–4660. [Google Scholar] [CrossRef] [PubMed]
- Levantesi, C.; La Mantia, R.; Masciopinto, C.; Böckelmann, U.; Ayuso-Gabella, M.N.; Salgot, M.; Tandoi, V.; Van Houtte, E.; Wintgens, T.; Grohmann, E. Quantification of pathogenic microorganisms and microbial indicators in three wastewater reclamation and managed aquifer recharge facilities in Europe. Sci. Total Environ. 2010, 408, 4923–4930. [Google Scholar] [CrossRef] [PubMed]
- Molleda, P.; Blanco, I.; Ansola, G.; de Luis, E. Removal of wastewater pathogen indicators in a constructed wetland in Leon, Spain. Ecol. Eng. 2008, 33, 252–257. [Google Scholar] [CrossRef]
- Bowman, D.D.; Little, M.D.; Reimers, R.S. Precision and accuracy of an assay for detecting Ascaris eggs in various biosolid matrices. Water Res. 2003, 37, 2063–2072. [Google Scholar] [CrossRef]
- Gyawali, P.; Sidhu, J.P.S.; Ahmed, W.; Jagals, P.; Toze, S. Rapid concentration and sensitive detection of hookworm ova from wastewater matrices using a real-time PCR method. Exp. Parasitol. 2015, 159, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Amoah, I.D.; Reddy, P.; Stenstrom, T.A. Effect of reagents used during detection and quantification of Ascaris suum in environmental samples on egg viability. Water Sci. Technol. 2017, 76, 2389–2400. [Google Scholar] [CrossRef] [PubMed]
- Shamma, M.; Al-Adawi, M.A. The morphological changes of Ascaris lumbricoides ova in sewage sludge water treated by gamma irradiation. Radiat. Phys. Chem. 2002, 65, 277–279. [Google Scholar] [CrossRef]
- Nelson, K.L.; Darby, J.L. Inactivation of Viable Ascaris Eggs by Reagents during Enumeration. Appl. Environ. Microbiol. 2001, 67, 5453–5459. [Google Scholar] [CrossRef]
- Koné, D.; Cofie, O.; Zurbrügg, C.; Gallizzi, K.; Moser, D.; Drescher, S.; Strauss, M. Helminth eggs inactivation efficiency by faecal sludge dewatering and co-composting in tropical climates. Water Res. 2007, 41, 4397–4402. [Google Scholar] [CrossRef]
- Gaspard, P.G.; Schwartzbrod, J. Parasite contamination (helminth eggs) in sludge treatment plants: Definition of a sampling strategy. Int. J. Hyg. Environ. Health 2003, 206, 117–122. [Google Scholar] [CrossRef]
- Amoah, I.D.; Reddy, P.; Seidu, R.; Stenström, T.A. Concentration of soil-transmitted helminth eggs in sludge from South Africa and Senegal: A probabilistic estimation of infection risks associated with agricultural application. J. Environ. Manag. 2018, 206, 1020–1027. [Google Scholar] [CrossRef] [PubMed]
- Reimers, R.S.; Little, M.D.; Englande, A.J.; Leftwich, D.B.; Bowman, D.D.; Wilkinson, R.F. Parasites in southern sludges and disinfection by standard sludge treatment. Water Sci. Technol. 1982, 14, 1568. [Google Scholar] [CrossRef]
- Trönnberg, L.; Hawksworth, D.; Hansen, A.; Archer, C.; Stenström, T.A. Household-based prevalence of helminths and parasitic protozoa in rural KwaZulu-Natal, South Africa, assessed from faecal vault sampling. Trans. R. Soc. Trop. Med. Hyg. 2010, 104, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Gaspard, P.; Wiart, J.; Schwartzbrod, J. A Method for Assessing the Viability of Nematode Eggs in Sludge. Environ. Technol. 1996, 17, 415–420. [Google Scholar] [CrossRef]
- Jaskoski, B.J. A Comparative Study of Detergent Effects on Ascarid Development. Am. Midl. Nat. 1954, 52, 142–148. [Google Scholar] [CrossRef]
- Engohang-Ndong, J.; Uribe, R.M.; Gregory, R.; Gangoda, M.; Nickelsen, M.G.; Loar, P. Effect of electron beam irradiation on bacterial and Ascaris ova loads and volatile organic compounds in municipal sewage sludge. Radiat. Phys. Chem. 2015, 112, 6–12. [Google Scholar] [CrossRef]
- Maya, C.; Ortiz, M.; Jimenez, B. Viability of Ascaris and other helminth genera non larval eggs in different conditions of temperature, lime (pH) and humidity. Water Sci. Technol. 2010, 62, 2616–2624. [Google Scholar] [CrossRef][Green Version]
- De Victorica, J.; Galván, M. Preliminary testing of a rapid coupled methodology for quantitation/viability determination of helminth eggs in raw and treated wastewater. Water Res. 2003, 37, 1278–1287. [Google Scholar] [CrossRef]
- Mara, D.; Sleigh, A. Estimation of Ascaris infection risks in children under 15 from the consumption of wastewater-irrigated carrots. J. Water Health 2010, 8, 35–38. [Google Scholar] [CrossRef]
- Zdybel, J.; Karamon, J.; Różycki, M.; Bilska-Zając, E.; Kłapeć, T.; Cencek, T. Characterisation of a new, highly effective method for detecting nematode eggs (Ascaris spp., Toxocara spp., Trichuris spp.) in sewage sludge containing flocculants. Exp. Parasitol. 2016, 170, 198–206. [Google Scholar] [CrossRef]
- Horiuchi, S.; Uga, S. Modified flotation method, an effective technique for recovering helminth eggs in soil. Parasitol. Int. 2016, 65, 576–579. [Google Scholar] [CrossRef] [PubMed]
- David, E.D.; Lindquist, W.D. Determination of the Specific Gravity of Certain Helminth Eggs Using Sucrose Density Gradient Centrifugation. J. Parasitol. 1982, 68, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Yaya-Beas, R.E.; Cadillo-La-Torre, E.A.; Kujawa-Roeleveld, K.; Van Lier, J.B.; Zeeman, G. Presence of helminth eggs in domestic wastewater and its removal at low temperature UASB reactors in Peruvian highlands. Water Res. 2016, 90, 286–293. [Google Scholar] [CrossRef] [PubMed]
- De Souza, G.S.; Rodrigues, L.A.; de Oliveira, W.J.; Chernicharo, C.A.; Guimarães, M.P.; Massara, C.L.; Grossi, P.A. Disinfection of domestic effluents by gamma radiation: Effects on the inactivation of Ascaris lumbricoides eggs. Water Res. 2011, 45, 5523–5528. [Google Scholar] [CrossRef] [PubMed]
- Mizgajska, H. The role of some environmental factors in the contamination of soil with Toxocara spp. and other geohelminth eggs. Parasitol. Int. 1997, 46, 67–72. [Google Scholar] [CrossRef]
- Cranston, I.; Teoh, P.J.; Baker, S.M.; Sengupta, M.E.; Ensink, J.H. Evaluating the efficacy of a centrifugation-flotation method for extracting Ascaris ova from soil. Trans. R. Soc. Trop. Med. Hyg. 2016, 110, 400–407. [Google Scholar] [CrossRef]
- Satchwell, M.G. An adaptation of concentration techniques for the enumeration of parasitic helminth eggs from sewage sludge. Water Res. 1986, 20, 813–816. [Google Scholar] [CrossRef]
- Shahsavari, E.; Schmidt, J.; Aburto-Medina, A.; Khallaf, B.; Balakrishnan, V.; Crosbie, N.D.; Aravind Surapanenid, A.; Andrew, S.; Balla, A.S. A modified assay for the enumeration of ascaris eggs in fresh raw sewage. MethodsX 2017, 4, 186–190. [Google Scholar] [CrossRef]
- Jeandron, A.; Ensink, J.H.; Thamsborg, S.M.; Dalsgaard, A.; Sengupta, M.E. A Quantitative Assessment Method for Ascaris Eggs on Hands. PLoS ONE 2014, 9, e96731. [Google Scholar] [CrossRef]
- Kazacos, K.R. Improved method for recovering ascarid and other helminth eggs from soil associated with epizootics and during survey studies. Am. J. Vet. Res. 1983, 44, 896–900. [Google Scholar]
- Roepstorff, A.; Nansen, P. Epidemiology, Diagnosis and Control of Helminth Parasites of Swine; Food and Agriculture Organization (FAO): Rome, Italy, 1998. [Google Scholar]
- Inpankaew, T.; Schär, F.; Dalsgaard, A.; Khieu, V.; Chimnoi, W.; Chhoun, C.; Sok, D.; Marti, H.; Muth, S.; Odermatt, P.; et al. High Prevalence of Ancylostoma ceylanicum Hookworm Infections in Humans, Cambodia, 2012. Emerg. Infect. Dis. 2014, 20, 976–982. [Google Scholar] [CrossRef] [PubMed]
- Bastos, V.K.; Cutolo, S.A.; Doria, M.D.C.O.; Razzolini, M.T.P. Detection and quantification of viable Ascaris sp. and other helminth eggs in sewage sludge. Int. J. Environ. Health Res. 2013, 23, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Shiraho, E.A.; Eric, A.L.; Mwangi, I.N.; Maina, G.M.; Kinuthia, J.M.; Mutuku, M.W.; Mugambi, R.M.; Mwandi, J.M.; Mkoji, G.M. Development of a Loop Mediated Isothermal Amplification for Diagnosis of Ascaris lumbricoides in Fecal Samples. J. Parasitol. Res. 2016, 2016, 7376207. [Google Scholar] [CrossRef]
- Gyawali, P.; Sidhu, J.P.S.; Ahmed, W.; Jagals, P.; Toze, S. Comparison of culture-based, vital stain and PMA-qPCR methods for the quantitative detection of viable hookworm ova. Water Sci. Technol. 2017, 75, 2615–2621. [Google Scholar] [CrossRef] [PubMed]
- Dabrowska, J.; Zdybel, J.; Karamon, J.; Kochanowski, M.; Stojecki, K.; Cencek, T.; Klapec, T. Assessment of viability of the nematode eggs (Ascaris, Toxocara, Trichuris) in sewage sludge with the use of LIVE/DEAD Bacterial Viability Kit. Ann. Agric. Environ. Med. 2014, 21, 35–41. [Google Scholar]
- Traub, R.J.; Robertson, I.D.; Irwin, P.; Mencke, N.; Thompson, R.A. Application of a species-specific PCR-RFLP to identify Ancylostoma eggs directly from canine faeces. Vet. Parasitol. 2004, 123, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Pilotte, N.; Papaiakovou, M.; Grant, J.R.; Bierwert, L.A.; Llewellyn, S.; McCarthy, J.S.; Williams, S.A. Improved PCR-Based Detection of Soil Transmitted Helminth Infections Using a Next-Generation Sequencing Approach to Assay Design. PLoS Negl. Trop. Dis. 2016, 10, e0004578. [Google Scholar] [CrossRef]
- Raynal, M.; Villegas, E.N.; Nelson, K.L. Enumeration of viable and non-viable larvated Ascaris eggs with quantitative PCR. J. Water Health 2012, 10, 594–604. [Google Scholar] [CrossRef][Green Version]
- Bag, S.; Saha, B.; Mehta, O.; Anbumani, D.; Kumar, N.; Dayal, M.; Pant, A.; Kumar, P.; Saxena, S.; Allin, K.H.; et al. An Improved Method for High Quality Metagenomics DNA Extraction from Human and Environmental Samples. Sci. Rep. 2016, 6, 26775. [Google Scholar] [CrossRef]
- Loreille, O.; Roumat, E.; Verneau, O.; Bouchet, F.; Hänni, C. Ancient DNA from Ascaris: Extraction amplification and sequences from eggs collected in coprolites. Int. J. Parasitol. 2001, 31, 1101–1106. [Google Scholar] [CrossRef]
- Deisingh, A.K.; Thompson, M. Biosensors for the detection of bacteria. Can. J. Microbiol. 2004, 50, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Hindson, C.M.; Chevillet, J.R.; Briggs, H.A.; Gallichotte, E.N.; Ruf, I.K.; Hindson, B.J.; Vessella, R.L.; Tewari, M. Absolute quantification by droplet digital PCR versus analog real-time PCR. Nat. Methods 2013, 10, 1003–1005. [Google Scholar] [CrossRef] [PubMed]
- Gordon, C.A.; Gray, D.J.; Gobert, G.N.; McManus, D.P. DNA amplification approaches for the diagnosis of key parasitic helminth infections of humans. Mol. Cell. Probes 2011, 25, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Llewellyn, S.; Inpankaew, T.; Nery, S.V.; Gray, D.J.; Verweij, J.J.; Clements, A.C.; Gomes, S.J.; Traub, R.; McCarthy, J.S. Application of a Multiplex Quantitative PCR to Assess Prevalence and Intensity Of Intestinal Parasite Infections in a Controlled Clinical Trial. PLoS Negl. Trop. Dis. 2016, 10, e0004380. [Google Scholar] [CrossRef] [PubMed]
- Gordon, C.A.; McManus, D.P.; Acosta, L.P.; Olveda, R.M.; Williams, G.M.; Ross, A.G.; Gray, D.J.; Goberta, G.N. Multiplex real-time PCR monitoring of intestinal helminths in humans reveals widespread polyparasitism in Northern Samar, the Philippines. Int. J. Parasitol. 2015, 45, 477–483. [Google Scholar] [CrossRef]
- Pecson, B.M.; Barrios, J.A.; Johnson, D.R.; Nelson, K.L. A Real-Time PCR Method for Quantifying Viable Ascaris Eggs Using the First Internally Transcribed Spacer Region of Ribosomal DNA. Appl. Environ. Microbiol. 2006, 72, 7864–7872. [Google Scholar] [CrossRef]
- Randazzo, W.; Khezri, M.; Ollivier, J.; Le Guyader, F.S.; Rodríguez-Díaz, J.; Aznar, R.; Sánchez, G. Optimization of PMAxx pretreatment to distinguish between human norovirus with intact and altered capsids in shellfish and sewage samples. Int. J. Food Microbiol. 2018, 266, 1–7. [Google Scholar] [CrossRef]
- Daranas, N.; Bonaterra, A.; Francés, J.; Cabrefiga, J.; Montesinos, E.; Badosa, E. Monitoring Viable Cells of the Biological Control Agent Lactobacillus plantarum PM411 in Aerial Plant Surfaces by Means of a Strain-Specific Viability Quantitative PCR Method. Appl. Environ. Microbiol. 2018, 84. [Google Scholar] [CrossRef]
- Hoffman, R.M.; Standridge, J.H.; Prieve, A.F.; Cucunato, J.C.; Bernhardt, M. Using flow cytometry to detect protozoa. J. Am. Water Work. Assoc. 1997, 89, 104–111. [Google Scholar] [CrossRef]
- Wang, Y.; Hammes, F.; De Roy, K.; Verstraete, W.; Boon, N. Past, present and future applications of flow cytometry in aquatic microbiology. Trends Biotechnol. 2010, 28, 416–424. [Google Scholar] [CrossRef]
- Barbosa, J.M.; Costa-de-Oliveira, S.; Rodrigues, A.G.; Hanscheid, T.; Shapiro, H.; Pina-Vaz, C. A flow cytometric protocol for detection of Cryptosporidium spp. Cytom. Part A 2008, 73, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, V.B.; Truskewycz, A.; Ball, A.S.; Soni, S.K. Detection of helminth ova genera using in-situ biosynthesis of gold nanoparticles. MethodsX 2019, 6, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Rajapaksha, P.; Elbourne, A.; Gangadoo, S.; Brown, R.; Cozzolino, D.; Chapman, J. A review of methods for the detection of pathogenic microorganisms. Analyst 2019, 144, 396–411. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.C.; Laperriere, G.; Germain, H. Droplet Digital PCR versus qPCR for gene expression analysis with low abundant targets: From variable nonsense to publication quality data. Sci. Rep. 2017, 7, 2409. [Google Scholar] [CrossRef] [PubMed]
- Ricchi, M.; Bertasio, C.; Boniotti, M.B.; Vicari, N.; Russo, S.; Tilola, M.; Bellotti, M.A.; Bertasi, B. Comparison among the Quantification of Bacterial Pathogens by qPCR, dPCR, and Cultural Methods. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Shehata, H.R.; Li, J.; Chen, S.; Redda, H.; Cheng, S.; Tabujara, N.; Li, H.; Warriner, K.; Hanner, R. Droplet digital polymerase chain reaction (ddPCR) assays integrated with an internal control for quantification of bovine, porcine, chicken and turkey species in food and feed. PLoS ONE 2017, 12, e0182872. [Google Scholar]
- Acosta, L.S.; Santisima-Trinidad, A.B.; Bornay-Llinares, F.J.; Gonzalez, M.M.; Valero, J.A.P.; Munoz, M.R. Quantitative PCR and digital PCR for detection of Ascaris lumbricoides eggs in reclaimed water. Biomed Res Int. 2017, 2017, 7515409. [Google Scholar] [CrossRef]
- Sivajothi, S.; Sudhakara Reddy, B. Molecular Diagnosis of Parasitic Diseases in Sheep—A Review. Int. J. Livest. Res. 2018, 8, 14–24. [Google Scholar] [CrossRef]
- Ospina-Villa, J.D.; Zamorano-Carrillo, A.; Castañón-Sánchez, C.A.; Ramírez-Moreno, E.; Marchat, L.A. Aptamers as a promising approach for the control of parasitic diseases. Braz. J. Infect. Dis. 2016, 20, 610–618. [Google Scholar] [CrossRef]
- Göringer, H.U. Parasite-specific aptamers as biosynthetic reagents and potential pharmaceuticals. Trends Parasitol. 2012, 28, 106–113. [Google Scholar] [CrossRef]
- Long, Y.; Qin, Z.; Duan, M.; Li, S.; Wu, X.; Lin, W.; Li, J.; Zhao, Z.; Liu, J.; Xiong, D. Screening and identification of DNA aptamers toward Schistosoma japonicum eggs via SELEX. Sci. Rep. 2016, 6, 24986. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, Z.; Yu, Y.; Wang, M.; Li, J.; Zhang, Z.; Liu, J.; Wu, X.; Lu, A.; Zhang, G.; Zhang, B. Recent Advances in SELEX Technology and Aptamer Applications in Biomedicine. Int. J. Mol. Sci. 2017, 18, 2142. [Google Scholar] [CrossRef] [PubMed]
- Aldewachi, H.; Chalati, T.; Woodroofe, M.N.; Bricklebank, N.; Sharrack, B.; Gardiner, P. Gold nanoparticle-based colorimetric biosensors. Nanoscale 2018, 10, 18–33. [Google Scholar] [CrossRef] [PubMed]
- Hutter, E.; Maysinger, D. Gold-nanoparticle-based biosensors for detection of enzyme activity. Trends Pharm. Sci. 2013, 34, 497–507. [Google Scholar] [CrossRef]
- Jongjinakool, S.; Palasak, K.; Bousod, N.; Teepoo, S. Gold Nanoparticles-based Colorimetric Sensor for Cysteine Detection. Energy Procedia 2014, 56, 10–18. [Google Scholar] [CrossRef]
- Lim, S.; Koo, O.K.; You, Y.S.; Lee, Y.E.; Kim, M.S.; Chang, P.S.; Kang, D.H.; Yu, J.-H.; Choi, Y.J.; Gunasekaran, S. Enhancing Nanoparticle-Based Visible Detection by Controlling the Extent of Aggregation. Sci. Rep. 2012, 2, 456. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Yang, D.; Ivleva, N.P.; Mircescu, N.E.; Niessner, R.; Haisch, C. SERS Detection of Bacteria in Water by in Situ Coating with Ag Nanoparticles. Anal. Chem. 2014, 86, 1525–1533. [Google Scholar] [CrossRef]
- Dina, N.E.; Zhou, H.; Colniţă, A.; Leopold, N.; Szoke-Nagy, T.; Coman, C.; Haisch, C. Rapid single-cell detection and identification of pathogens by using surface-enhanced Raman spectroscopy. Analyst 2017, 142, 1782–1789. [Google Scholar] [CrossRef]
- Xie, W.; Schlucker, S. Surface-enhanced Raman spectroscopic detection of molecular chemo- and plasmo-catalysis on noble metal nanoparticles. Chem. Commun. 2018, 54, 2326–2336. [Google Scholar] [CrossRef]
- Mocan, T.; Matea, C.T.; Pop, T.; Mosteanu, O.; Buzoianu, A.D.; Puia, C.; Iancu, C.; Mocan, L. Development of nanoparticle-based optical sensors for pathogenic bacterial detection. J. Nanobiotechnol. 2017, 15, 25. [Google Scholar] [CrossRef]
- Hernández-Neuta, I.; Neumann, F.; Brightmeyer, J.; Ba Tis, T.; Madaboosi, N.; Wei, Q.; Ozcan, A.; Nilsson, M. Smartphone-based clinical diagnostics: Towards democratization of evidence-based health care. J. Int. Med. 2019, 285, 19–39. [Google Scholar] [CrossRef] [PubMed]
- Ganguli, A.; Ornob, A.; Yu, H.; Damhorst, G.L.; Chen, W.; Sun, F.; Bhuiya, A.; Cunningham, B.T.; Bashir, R. Hands-free smartphone-based diagnostics for simultaneous detection of Zika, Chikungunya, and Dengue at point-of-care. Biomed. Microdevices 2017, 19, 73. [Google Scholar] [CrossRef] [PubMed]
- Sowerby, S.J.; Crump, J.A.; Johnstone, M.C.; Krause, K.L.; Hill, P.C. Smartphone Microscopy of Parasite Eggs Accumulated into a Single Field of View. Am. J. Trop. Med. Hyg. 2016, 94, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Macdonald, J.; von Stetten, F. Review: A comprehensive summary of a decade development of the recombinase polymerase amplification. Analyst 2019, 144, 31–67. [Google Scholar] [CrossRef] [PubMed]
- Daher, R.K.; Stewart, G.; Boissinot, M.; Bergeron, M.G. Recombinase Polymerase Amplification for Diagnostic Applications. Clin. Chem. 2016, 62, 947–958. [Google Scholar] [CrossRef] [PubMed]
- Parida, M.; Sannarangaiah, S.; Dash, P.K.; Rao, P.V.L.; Morita, K. Loop mediated isothermal amplification (LAMP): A new generation of innovative gene amplification technique; perspectives in clinical diagnosis of infectious diseases. Rev. Med. Virol. 2008, 18, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Salant, H.; Abbasi, I.; Hamburger, J. The development of a loop-mediated isothermal amplification method (LAMP) for Echinococcus granulosus [corrected] coprodetection. Am. J. Trop. Med. Hyg. 2012, 87, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Paris, D.H.; Imwong, M.; Faiz, A.M.; Hasan, M.; Yunus, E.B.; Silamut, K.; Lee, S.J.; Day, N.P.J.; Dondorp, A.M. Loop-mediated isothermal PCR (LAMP) for the diagnosis of falciparum malaria. Am. J. Trop. Med. Hyg. 2007, 77, 972–976. [Google Scholar] [CrossRef][Green Version]
- Notomi, T.; Okayama, H.; Masubuchi, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000, 28, E63. [Google Scholar] [CrossRef]
- Mugambi, R.M.; Agola, E.L.; Mwangi, I.N.; Kinyua, J.; Shiraho, E.A.; Mkoji, G.M. Development and evaluation of a Loop Mediated Isothermal Amplification (LAMP) technique for the detection of hookworm (Necator americanus) infection in fecal samples. Parasit. Vectors 2015, 8, 574. [Google Scholar] [CrossRef]
- Wang, J.; Liu, L.; Wang, J.; Sun, X.; Yuan, W. Recombinase Polymerase Amplification Assay-A Simple, Fast and Cost-Effective Alternative to Real Time PCR for Specific Detection of Feline Herpesvirus-1. PLoS ONE 2017, 12, e0166903. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Xing, W.; Yu, X.; Fu, W.; Wang, Y.; Zou, M.; Luo, Z.; Xu, D. Recombinase polymerase amplification combined with a lateral flow dipstick for rapid and visual detection of Schistosoma japonicum. Parasit. Vectors 2016, 9, 476. [Google Scholar] [CrossRef]
- Magro, L.; Jacquelin, B.; Escadafal, C.; Garneret, P.; Kwasiborski, A.; Manuguerra, J.C.; Monti, F.; Sakuntabhai, A.; Vanhomwegen, J.; Lafaye, P.; et al. Paper-based RNA detection and multiplexed analysis for Ebola virus diagnostics. Sci. Rep. 2017, 7, 1347. [Google Scholar] [CrossRef] [PubMed]
- Henderson, W.A.; Xiang, L.; Fourie, N.H.; Abey, S.K.; Ferguson, E.G.; Diallo, A.F.; Kenea, N.D.; Kim, C.H. Simple lateral flow assays for microbial detection in stool. Anal. Methods 2018, 10, 5358–5363. [Google Scholar] [CrossRef]
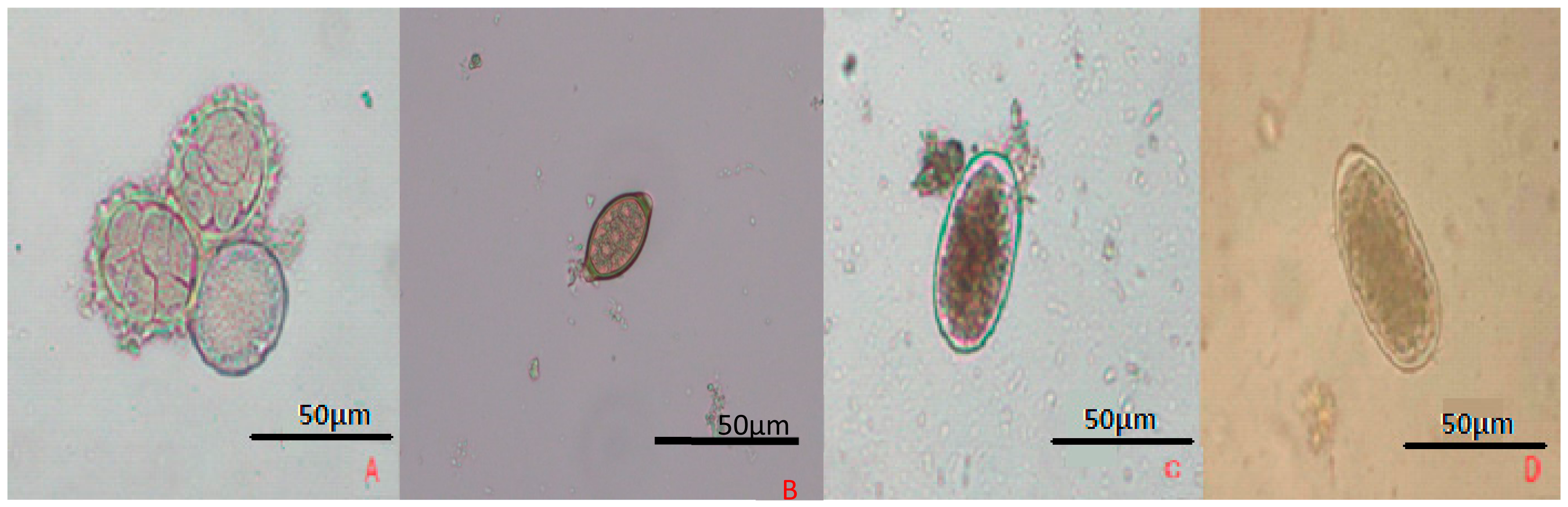
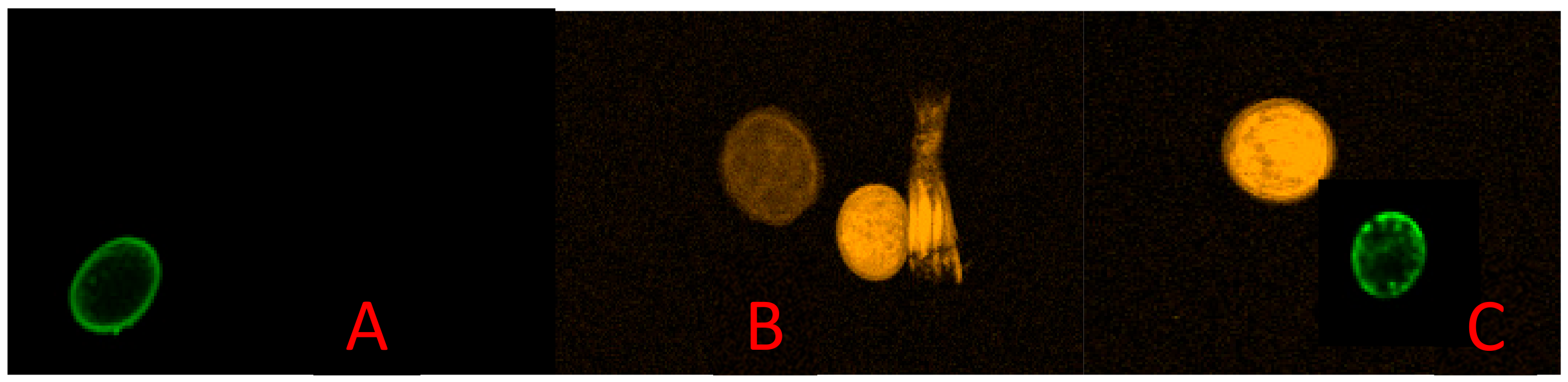
| STHs | Daily Ova Output per Worm | Ova Size and Shape | Shell of Ova | Infective Stage |
|---|---|---|---|---|
| Ascaris | 200,000 | 40 × 60 µm Golden brown colour for mammilated ova and clear for decorticated ova Oval shaped or slightly rounded | Smooth shell Has two layers, one thicker outer shell and one thinner inner shell Viable eggs have a defined space between inner and outer shells | Viable ova |
| Trichuris | 3000–5000 | 50 × 25 µm Lemon shaped with curved sides and rounded mucoid plugs | Smooth shell No space between inner and outer shell Mucoid plugs and shell are symmetric | Viable ova |
| Hookworm | 9000–15,000 | 65 × 40 µm Oval shaped | Smooth shell Outer shell appears as single, thin, black line Inner ovum is clearly segmented | Filariform larvae |
| Country | Untreated Wastewater (ova/L) * | Treated Wastewater (ova/L) * | Sludge (ova/g) * | Reference |
|---|---|---|---|---|
| India | 72A, 15H, 4T | 60A,9H,2T | NR | [33] |
| Iran | 12A, 3Ta, 2H | 1A | NR | |
| Burkina Faso | 10A, 4H, 1T | 1A | 1327A, 442H, 11T | [34] |
| Mexico | 330NS | NR | 177NS | [4] |
| Pakistan | 175A, 26T | 4A | NR | [35] |
| Morocco | 23NS | NR | NR | [36] |
| Tunisia | 960A, 208Ta | 240A, 52Ta | NR | [37] |
| France | NR | NR | 5NS | [38] |
| Oman | 517A, 33H, 18Ta | 45A, 18Ta | NR | [39] |
| Australia | NR | 41NS | NR | [1] |
| USA | 8NS | NR | 13NS | [4] |
| Sample | Flotation (S.G) | References |
|---|---|---|
| Wastewater/Sludge | NaCl (1.18) | [66] |
| Wastewater/Sludge | NaCl (1.27) | [44] |
| Wastewater/Sludge | ZnSO4 (1.18) | [67] |
| Wastewater/Sludge | ZnSO4 (1.20) | [29] |
| Wastewater/Sludge | MgSO4 (1.20) | [9,47] |
| Wastewater/Sludge | MgSO4 (1.29) | [52] |
| Wastewater/Sludge | NaNO3 (1.39) | [68] |
| Wastewater/Sludge | NaCl (1.19) | [57] |
| Method | Total Replicates | Average Numbers of Ova Recovered in 90 µL (Standard Deviation) | Average Ova Recovery (%) |
|---|---|---|---|
| Flotation method | |||
| Modified McMaster | 5 | 650 (90) | 65 |
| FLOTAC technique | 5 | 434 (83.0) | 43 |
| Tubes and pipettes | |||
| Non-coated Falcon tubes/glass pipettes | 5 | 586 (168) | 58 |
| NC Falcon tubes//C glass pipettes | 5 | 571 (171) | 57 |
| Coated Falcon tubes/glass pipettes | 3 | 335 (120.0) | 33 |
| Coated Falcon tubes/non-coated plastic pipettes | 3 | 374 (163) | 37 |
| Coated falcon tubes/ Non-coated glass pipettes | 5 | 356 (188) | 35 |
| Detergent | |||
| Deionized water | 5 | 742 (56) | 74 |
| 7X® 1% | 5 | 897 (228) | 89 |
| Tween 80 (0.1%) | 5 | 587 (180) | 58 |
| Benzenethonium chloride 0.1% | 5 | 870 (129) | 87 |
| Cetylpyridinium chloride 0.1% | 5 | 844 (178) | 84 |
| Methods | Advantages | Limitations | References |
|---|---|---|---|
| Optical microscopy | Viability possible Cost-effective Require less lab space Stains can differentiate viable and non-viable ova | Time-consuming Less sensitivity and specificity Possible false positive results in the determination of viability using stain-based methods Species differentiation is not possible | [47,61,96] |
| PCR-based | Fast, specific and sensitive Multiplex PCR is possible Quantitative detection (qPCR) of target pathogen is rapid | Not possible to distinguish viable and dead ovaneed for well-equipped laboratory Multiple primers required Requirement of skilled personnel | [79,90,96] |
| Flow cytometry | Accurate and reliable Differentiate cells based on complexity | Particle size detection limit ranging between 3 µm and 20 µm Expensive and require skilled personnel | [92,94] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravindran, V.B.; Soni, S.K.; Ball, A.S. A Review on the Current Knowledge and Prospects for the Development of Improved Detection Methods for Soil-Transmitted Helminth Ova for the Safe Reuse of Wastewater and Mitigation of Public Health Risks. Water 2019, 11, 1212. https://doi.org/10.3390/w11061212
Ravindran VB, Soni SK, Ball AS. A Review on the Current Knowledge and Prospects for the Development of Improved Detection Methods for Soil-Transmitted Helminth Ova for the Safe Reuse of Wastewater and Mitigation of Public Health Risks. Water. 2019; 11(6):1212. https://doi.org/10.3390/w11061212
Chicago/Turabian StyleRavindran, Vivek B., Sarvesh K. Soni, and Andrew S. Ball. 2019. "A Review on the Current Knowledge and Prospects for the Development of Improved Detection Methods for Soil-Transmitted Helminth Ova for the Safe Reuse of Wastewater and Mitigation of Public Health Risks" Water 11, no. 6: 1212. https://doi.org/10.3390/w11061212
APA StyleRavindran, V. B., Soni, S. K., & Ball, A. S. (2019). A Review on the Current Knowledge and Prospects for the Development of Improved Detection Methods for Soil-Transmitted Helminth Ova for the Safe Reuse of Wastewater and Mitigation of Public Health Risks. Water, 11(6), 1212. https://doi.org/10.3390/w11061212




