Adsorption of Chloramphenicol on Commercial and Modified Activated Carbons
Abstract
:1. Introduction
2. Materials and Methods
2.1. Measurement of Chloramphenicol Adsorption
2.2. Models for Adsorption Kinetics and Isotherm
2.3. Activated Carbons Used in the Study
2.4. Modification of the WG-12 Carbon
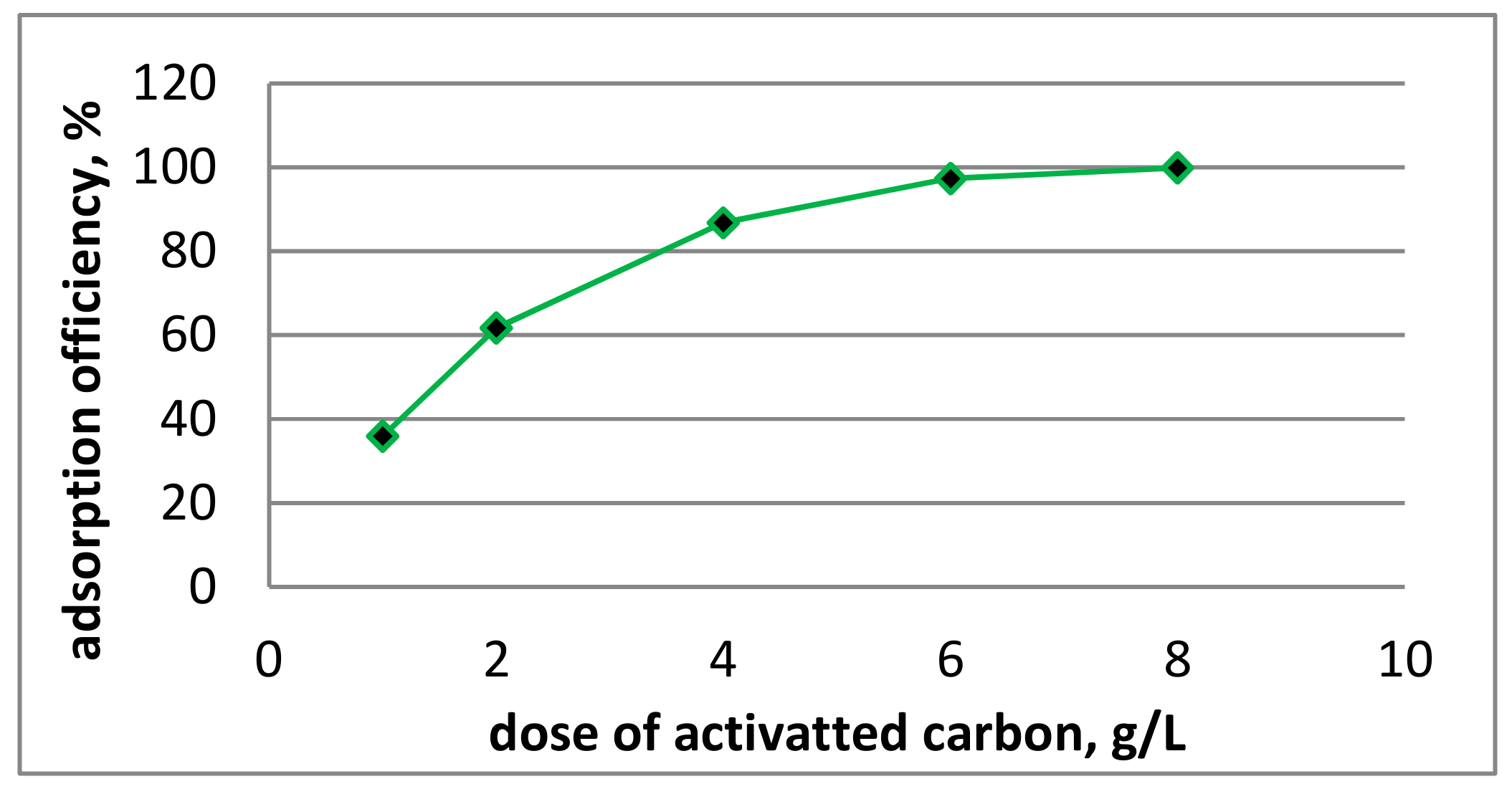
2.5. Research on Activated Carbons
3. Results and Discussion
3.1. Kinetics of Chloramphenicol Adsorption
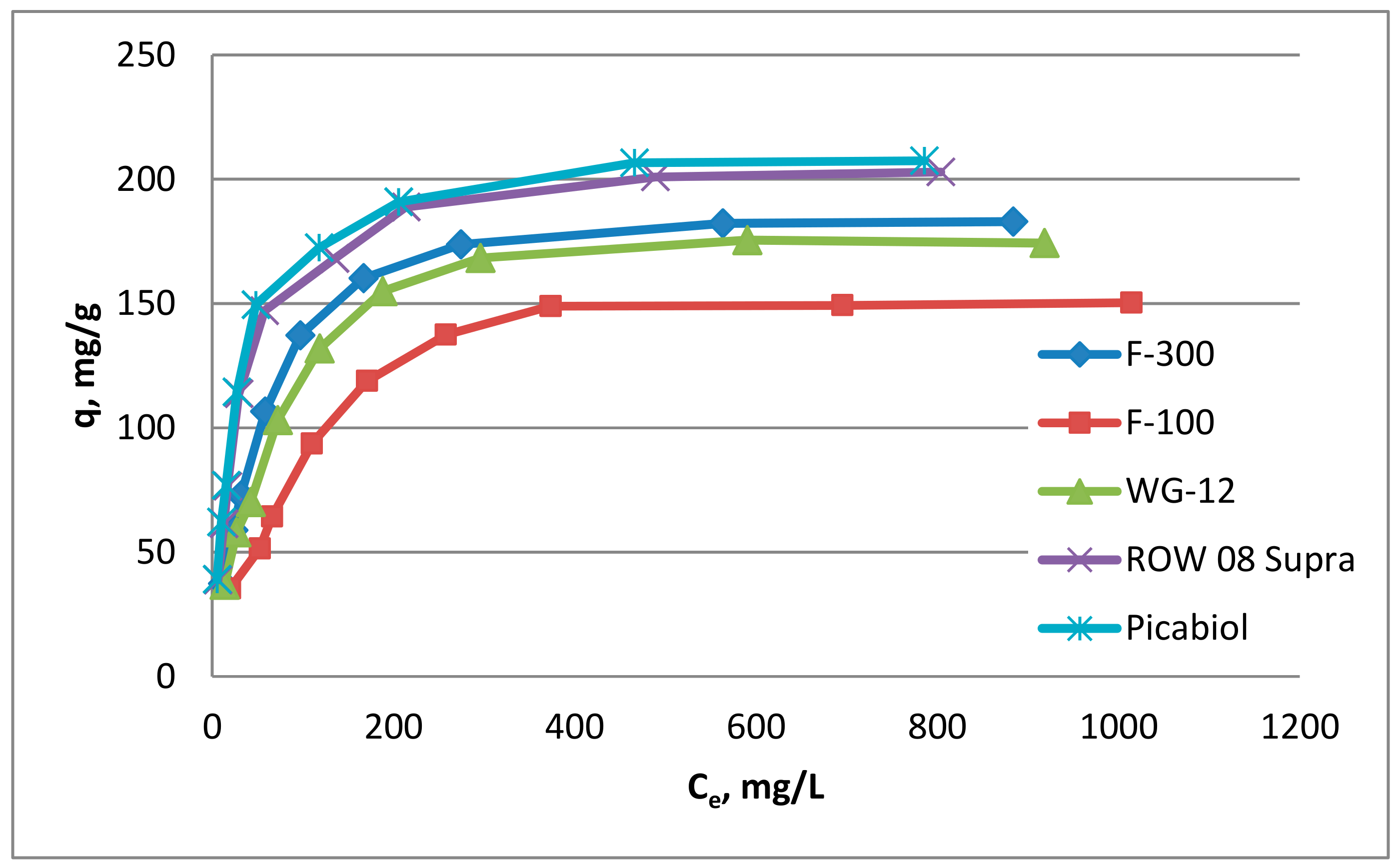
3.2. Isotherms of Chloramphenicol Adsorption on Commercial Activated Carbon
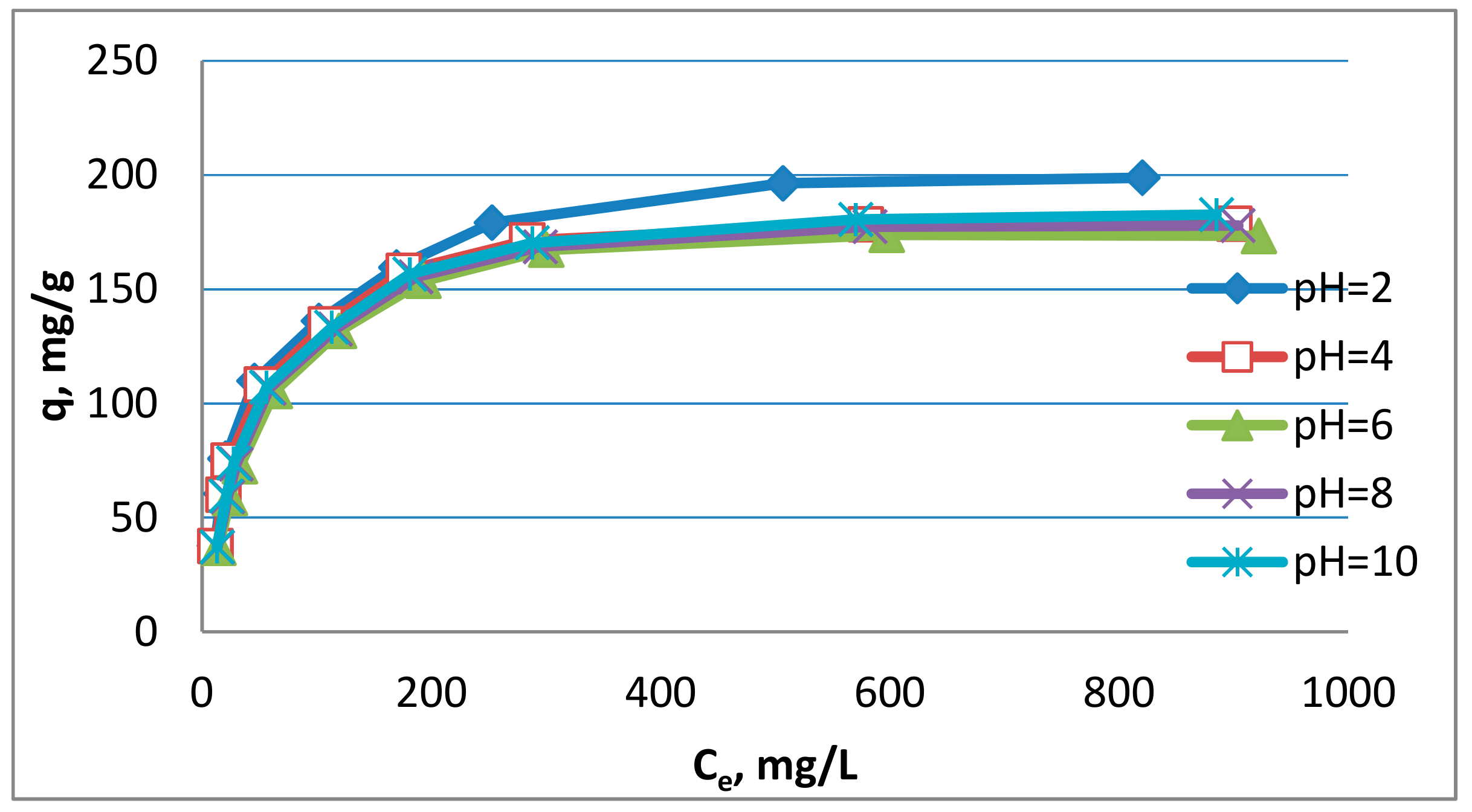
3.3. Impact of Process Conditions
- -
- pH about 1,5: AC-OH+ chloramphenicol = EDA interaction
- -
- pH = 4.0–4.25: AC-COO−.....H+…..-O2N-chloramphenicol = CABH formations with EDA interaction
- -
- pH obove 7.0: AC-OH+ chloramphenicol (H+/-OH/NO2/-NH-/-Cl) = H-bond formations repulsions interactions
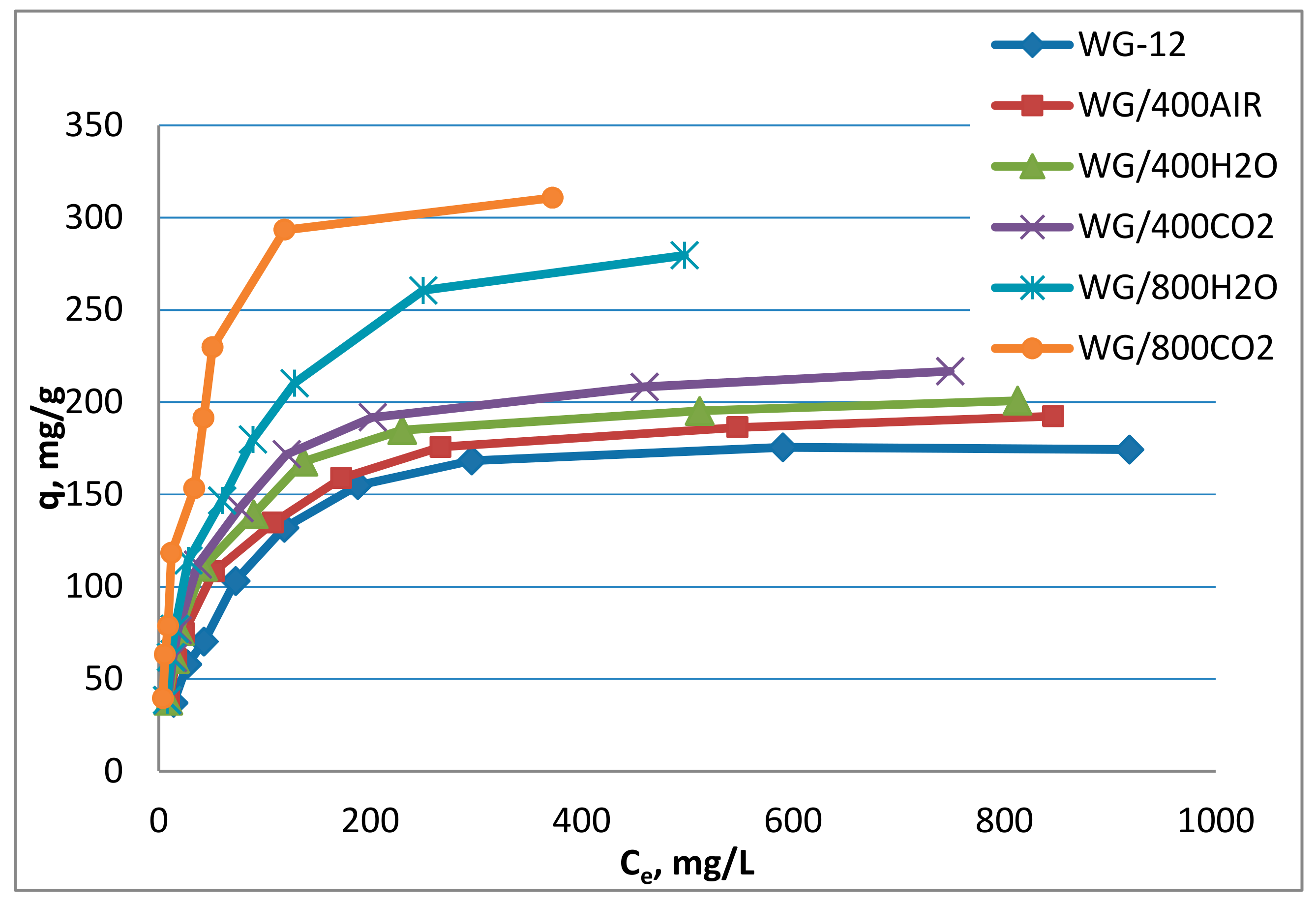
3.4. Chloramphenicol Adsorption on Modified Activated Carbons
4. Conclusions
- Commercial activated carbons (micro- or micro-mesoporous) such as F-100, F-300, ROW 08 Supra, WG-12, Picabiol can be used to remove chloramphenicol from water since they are characterized by quite a high adsorption capacity (qm from 174.68 to 214.91 mg/g). Activated carbons can be ranked according to their adsorption capacity in the following order: Picabiol > ROW 08 > F-300 > WG-12 > F-100.
- The order of activated carbons according to their adsorption capacity is consistent with their order according to the total volume of pores and the total volume of micro- and mesopores. This indicates the significance of the phenomenon of volumetric pore filling.
- The analysis of various adsorption kinetics equations (pseudo-first-order, pseudo-second-order, intraparticle diffusion model (Weber–Morris), Elovich) revealed that it is impossible to determine a single equation, which, for all activated carbons, would describe the results of the examinations with the highest correlation coefficient R2. Among the models used, the adsorption results are described the least efficiently by the pseudo-first order model.
- The insignificant effect of pH on adsorption efficiency was also found. It was observed that adsorption was better for acidic and alkaline solutions compared to solutions close to neutral. The maximum adsorption was obtained from solutions with pH = 2, but the adsorption capacity obtained was only 9% higher compared to adsorption from solutions with pH = 6.
- The high-temperature modification of activated carbon WG-12, adsorbents with much higher adsorption capacities were obtained for chloramphenicol. The highest monolayer capacity (qm = 343.12 mg/g) was obtained for carbon modified at 800 °C in the atmosphere of carbon dioxide and was almost twice as high as in the case of non-modified WG-12. Carbons modified at 800 °C were characterized by an increased specific surface area, volume of micropores and increased number of aerobic groups of alkaline character.
- It was found that among the analysed isotherms (Freundlich, Langnuir, Dubininin–Radushkevich, Temkin), the highest correlation coefficient in describing the results was obtained for the Langmuir model, whereas the lowest was for the Dubininin–Radushkevich. Temkin’s and Freundlich’s isotherms described the results well, but slightly worse than Langmuir’s isotherm.
Supplementary Materials
Funding
Conflicts of Interest
References
- Pramila, B.; Manikandan, S.; Anju, K.S.; Kannan, M.M.; Hong, S.; Maruthamuthu, S.; Subramanian, K. Elektrochemical decolorization and degradation of Turquoise Blue G (TBG) by pre-adapted petroleum degrading bacteria. Sep. Purif. Technol. 2014, 13, 719–727. [Google Scholar] [CrossRef]
- Osek, J.; Wieczorek, K. Spożycie leków przeciwbakteryjnych w Europie i występowanie oporności na te leki bakterii izolowanych od ludzi, zwierząt i z żywności w 2012 r. Życie Weter. 2015, 90, 601–603. (In Polish) [Google Scholar]
- European Centre for Disease Prevention and Control; European Food Safety Authority; European Medicines Agency. ECDC/EFSA/EMA first joint report on the integrated analysis of the consumption of antimicrobial agents and occurrence of antimicrobial resistance in bacteria from humans and food-producing animals. EFSA J. 2015, 13, 4006–4114. [Google Scholar] [CrossRef]
- Kuemmerer, K. Antibiotics in the aquatic environment—A review—Part I. Chemosphere 2009, 75, 417–434. [Google Scholar] [CrossRef]
- Baran, W.; Adamek, E.; Ziemianska, J.; Sobczak, A. Effects of the presence of sulfonamides in the environment and their influence on human health. J. Hazard. Mater. 2011, 196, 1–15. [Google Scholar] [CrossRef]
- Wu, M.; Que, C.; Xu, G.; Sun, Y.; Ma, J.; Xu, H.; Sun, R.; Tang, L. Occurrence, fate and interrelation of selected antibiotics in sewage treatment plants and their receiving surface water. Ecotoxicol. Environ. Saf. 2016, 132, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Golovko, O.; Kumar, V.; Fedorova, G.; Randak, T.; Grabic, R. Seasonal changes in antibiotics, antidepressants/psychiatric drugs, antihistamines and lipid regulators in a wastewater treatment plant. Chemosphere 2014, 111, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Michael, I.; Rizzo, L.; Mcardell, C.; Manaia, C.; Merlin, C.; Schwartz, T.; Dagot, C.; Fatta-Kassinos, D. Urban wastewater treatment plants as hotspots for the release of antibiotics in the environment: A review. Water Res. 2013, 47, 957–995. [Google Scholar] [CrossRef]
- Guerra, P.; Kim, M.; Shah, A.; Alaee, M.; Smyth, A. Occurrence and fate of antibiotic, analgesic/anti-inflammatory, and antifungal compounds in five wastewater treatment processes. Sci. Total Environ. 2014, 473–474, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Lach, J.; Stępniak, L.; Ociepa-Kubicka, A. Antibiotics in the Environment as one of the Barriers to Sustainable Development. Probl. Ekorozw. 2018, 13, 197–207. [Google Scholar]
- Watkinson, J.; Murby, E.; Costanzo, S. Removal of antibiotics in conventional and advanced wastewater treatment: Implications for environmental discharge and wastewater recycling. Water Res. 2007, 41, 4164–4176. [Google Scholar] [CrossRef] [PubMed]
- Kolpin, D.W.; Furlong, E.T.; Meyer, M.T.; Thurman, E.M.; Zaugg, S.D.; Barber, L.B.; Buxton, H.T. Pharmaceuticals, hormones, and others organic wastewater contaminants in US streams, 1999–2000: National reconnaissance. Environ. Sci. Technol. 2002, 36, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Zhang, R.; Wang, Y.; Pan, X.; Tang, J.; Zhang, G. Occurrence and distribution of antibiotics in the Beibu Gulf, China: Impacts of river discharge and aquaculture activities. Mar. Environ. Res. 2012, 78, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Grenni, P.; Ancona, V.; Caracciolo, A.B. Ecological effects of antibiotics on natural ecosystems: A review. Microchem. J. 2018, 136, 25–39. [Google Scholar] [CrossRef]
- Wollenberger, L.; Halling-Sørensen, B.; Kusk, K.O. Acute and chronic toxicity of veterinary antibiotics to Daphnia magna. Chemosphere 2000, 40, 723–730. [Google Scholar] [CrossRef]
- Yu, F.; Li, Y.; Han, S.; Ma, J. Adsorptive removal of antibiotics from aqueous solution using carbon materials. Chemosphere 2016, 153, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Loraine, G.; Pettigrove, M. Seasonal variations in concentrations of pharmaceuticals and personal care products in drinking water and reclaimed wastewater in southern California. Environ. Sci. Technol. 2006, 40, 687–695. [Google Scholar] [CrossRef]
- Wang, Q.J.; Mo, C.H.; Li, Y.W.; Gao, P.; Tai, Y.P.; Zhang, Y.; Ruan, Z.L.; Xu, J.W. Determination of four fluoroquinolone antibiotics in tap water in Guangzhou and Macao. Environ. Pollut. 2010, 158, 2350–2358. [Google Scholar] [CrossRef]
- Verlicchi, P.; Al Aukidy, M.; Zambello, E. Occurrence of pharmaceutical compounds in urban wastewater: Removal, mass load and environmental risk after a secondary treatment—A review. Sci. Total Environ. 2012, 429, 123–155. [Google Scholar] [CrossRef]
- Deo, R.P.; Halden, R.U. Pharmaceuticals in the Built and Natural Water Environment of the United States. Water 2013, 5, 1346–1365. [Google Scholar] [CrossRef]
- Jones, O.A.; Lester, J.N.; Voulvoulis, N. Pharmaceuticals: A threat to drinking water? Trends Biotechnol. 2005, 23, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Martínez, J.L. Environmental pollution by antibiotics and by antibiotic resistance determinants. Environ. Pollut. 2009, 157, 2893–2902. [Google Scholar] [CrossRef] [PubMed]
- WHO. Antimicrobial Resistance: Global Report on Surveillance (Geneva). 2014. Available online: https://apps.who.int/iris/bitstream/handle/10665/112642/9789241564748_eng.pdf;jsessionid=2F5AB71F63D62C312CD8298AF0ACB9A5?sequence=1 (accessed on 10 April 2019).
- ECDC/EMEA. The Bacterial Challenge: Time to React. 2009. Available online: https://ecdc.europa.eu/sites/portal/files/media/en/publications/Publications/0909_TER_The_Bacterial_Challenge_Time_to_React.pdf (accessed on 10 April 2019).
- Martinez-Huitle, C.A.; Brillas, E. Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods: A general review. Appl. Catal. B Environ. 2009, 87, 105–145. [Google Scholar] [CrossRef]
- Li, B.; Dong, Y.; Zou, C.; Xu, Y. Iron(III)-alginate fiber complex a highly effective and stable heterogeneous fenton photocatalyst for mineralization of organic dye. Ind. Eng. Chem. Res. 2014, 53, 4199–4206. [Google Scholar] [CrossRef]
- Adams, C.; Asce, M.; Wang, Y.; Loftin, Y.; Meyer, M. Removal of antibiotics from surface and distilled water in conventional water treatment processes. J. Environ. Eng. 2002, 128, 253–260. [Google Scholar] [CrossRef]
- Westerhoff, P.; Yoon, Y.; Snyder, S.; Wert, E. Fate of endocrine-disruptor, pharmaceutical, and personal care product chemicals during simulated drinking water treatment processes. Environ. Sci. Technol. 2005, 39, 6649–6663. [Google Scholar] [CrossRef]
- Zhang, Z. Porous Co3O4 nanorods-reduced graphene oxide with intrinsic peroxidase-like activity and catalysis in the degradation of methylene blue. ACS Appl. Mater. Interf. 2013, 5, 3809–3815. [Google Scholar] [CrossRef]
- Snyder, S.A.; Adham, S.; Redding, A.M.; Cannon, F.S.; DeCarolis, J.; Oppenheimer, J.; Wert, E.C.; Yoon, Y. Role of membranes and activated carbon in the removal of endocrine disruptors and pharmaceuticals. Desalination 2007, 202, 156–181. [Google Scholar] [CrossRef]
- Radjenovic, J.; Petrovic, M.; Ventura, F.; Barcelo, D. Rejection of pharmaceuticals in nanofiltration and reverse osmosis membrane drinking water treatment. Water Res. 2008, 42, 3601–3610. [Google Scholar] [CrossRef]
- Ngumba, E.; Gachanja, A.; Tuhkanen, T. Occurrence of selected antibiotics and antiretroviral drugs in Nairobi River Basin, Kenya. Sci. Total Environ. 2016, 539, 206–213. [Google Scholar] [CrossRef]
- Oturan, M.A.; Aron, J. Advanced oxidation processes in water/wastewater treatment: Principles and applications. A Review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2577–2641. [Google Scholar] [CrossRef]
- Zhang, X.; Gou, W.; Ngo, H.H.; Li, N.; Wu, W. Performance evaluation of powdered activated carbon for removing 28 types of antibiotics from water. J. Environ. Manag. 2016, 172, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Dai, J.; Jiang, Y.; Chang, Z.; Xie, A.; He, J.; Zhang, R.; Yan, Y. Facile preparation of intercrossed-stacked porous carbon originated from potassium citrate and their highly effective adsorption performance for chloramphenicol. J. Coll. Interf. Sci. 2017, 505, 858–869. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Zhou, Z.; Dai, J.; Ma, P.; Zhao, H.; He, J.; Xie, A.; Li, C.; Yan, Y. Novel N-doped hierarchically porous carbons derived from sustainable shrimp shell for high-performance removal of sulfamethazine and chloramphenicol. J. Taiwan Inst. Chem. Eng. 2016, 62, 228–238. [Google Scholar] [CrossRef]
- Zhang, R.; Zhou, Z.; Xie, A.; Dai, J.; Cui, J.; Lang, J.; Wei, M.; Dai, X.; Li, C.; Yan, Y. Preparation of hierarchical porous carbons from sodium carboxymethyl cellulose via halloysite template strategy coupled with KOH-activation for efficient removal of chloramphenicol. J. Taiwan Inst. Chem. Eng. 2017, 80, 424–433. [Google Scholar] [CrossRef]
- Xu, R.; Zhang, P.; Wang, Q.; Wang, X.; Yu, K.; Xue, T.; Wen, X. Influences of multi influent matrices on the retention of PPCPs by nanofiltration membranes. Sep. Purif. Technol. 2019, 212, 299–306. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, J.; Liu, H. Removal of chloramphenicol from aqueous solution using low-cost activated carbon prepared from Typha orientalis. Water 2018, 10, 351. [Google Scholar]
- Tran, H.N.; You, S.-J.; Nguyen, T.V.; Chao, H.-P. Insight into the adsorption mechanism of cationic dye onto biosorbents derived from agricultural wastes. Chem. Eng. Commun. 2017, 204, 1020–1036. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.-J.; Hosseini-Bandegharaei, A.; Chao, H.-P. Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: A critical review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef]
- Dada, A.O.; Olalekan, A.P.; Olatunya, A.M.; Dada, O. Langmuir, Freundlich and Dubinin-Radushkevich isotherms studies of equilibrium sorption of Zn+ unto phosphoric acid modified rice husk. IOSR J. Appl. Chem. 2012, 3, 38–45. [Google Scholar] [CrossRef]
- Pujol, J. The solution of nonlinear inverse problems and the Levenberg-Marquardt method. Geophysics 2007, 72, 1–16. [Google Scholar] [CrossRef]
- Buczek, B. The nature of active carbon surface oxidized in the gaseous phase. Zesz. Nauk. Akad. Ekon. W Krakowie 2005, 689. Available online: https://r.uek.krakow.pl/bitstream/123456789/411/1/93882657.pdf (accessed on 10 April 2019).
- Jankowska, H.; Świątkowski, A.; Starostin, L.; Ławrinienko-Omiecynska, J. Adsorpcja Jonów Na Węglu Aktywnym; Polish Scientific Publishers PWN: Warsaw, Poland, 1991; pp. 72–77. [Google Scholar]
- Boehm, H.P. Some aspects of the surface chemistry of carbon blacks and other carbons. Carbon 1994, 32, 759–769. [Google Scholar] [CrossRef]
- Pakuła, M.; Biniak, S.; Świątkowski, A.; Neffe, S. Influence of progressive surface oxidation of nitrogen-containing carbon on its electrochemical behaviour in phosphate buffer solutions. Carbon 2002, 40, 1873–1881. [Google Scholar] [CrossRef]
- Choma, J.; Jaroniec, M. Review of fundamental adsorption methods for evaluation surface and structural properties of activated carbon. Ochr. Sr. 2005, 27, 3–8. (In Polish) [Google Scholar]
- Horvath, G.; Kawazoe, K. Method for the calculation of effective pore size distribution in molecular sieve carbon. J. Chem. Eng. Jpn. 1983, 16, 470–475. [Google Scholar] [CrossRef]
- Zhu, X.; Gao, Y.; Yue, Q.; Song, Y.; Gao, B.; Xu, X. Facile synthesis of hierarchical porous carbon material by potassium tartrate activation for chloramphenicol removal. J. Taiwan Inst. Chem. Eng. 2018, 85, 141–144. [Google Scholar] [CrossRef]
- Ahmed, M.B.; Zhoua, J.L.; Ngo, H.H.; Guoa, W.; Johir, M.A.H.; Belhaj, D. Competitive sorption affinity of sulfonamides and chloramphenicol antibiotics toward functionalized biochar for water and wastewater treatment. Bioresour. Technol. 2017, 238, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Płaziński, W.; Rudziński, W. Adsorption Kinetics at solid/solution interfaces the meaning of the pseudo-first- and pseudo-second-order equations. Wiad. Chem. 2011, 65, 1055–1067. [Google Scholar]
- Tran, H.N.; Wang, Y.-F.; You, S.-J.; Chao, H.-P. Insights into the mechanism of cationic dye adsorption on activated charcoal: The importance of π–π interactions. Process Saf. Environ. 2017, 107, 168–180. [Google Scholar] [CrossRef]
- Din, M.A.T.; Ahmad, M.A.; Hameed, B.H. Ordered mesoporous carbons originated from non-edible polyethylene glycol 400 (PGE-400) for chloramphenicol antibiotic recovery from liquid phase. Chem. Eng. J. 2015, 260, 730–739. [Google Scholar] [CrossRef]
- Zhu, X.; Gao, Y.; Kan, Y.; Kong, W.; Gao, B. Preparation of green alga-based activated carbon with lower impregnation ratio and less activation time by potassium tartrate for adsorption of chloramphenicol. Ecotoxicol. Environ. Saf. 2017, 145, 289–294. [Google Scholar] [CrossRef]
- Liao, P.; Zhan, Z.; Dai, J.; Wu, X.; Zhang, W.; Wang, K.; Yuan, S. Adsorption of tetracycline and chloramphenicol in aqueous solutions by bamboo charcoal: A batch and fixed-bed column study. Chem. Eng. J. 2013, 228, 496–505. [Google Scholar] [CrossRef]
- Fan, Y.; Wang, B.; Yuan, S.; Wu, X.; Chen, J.; Wang, L. Adsorptive removal of chloramchenicol from wastewater by NaOH modified bamboo charcoal. Bioresour. Technol. 2010, 101, 7661–7664. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.-J.; Chao, H.-P. Fast and efficient adsorption of methylene green 5 on activated carbon prepared from new chemical activation method. J. Environ. Manag. 2017, 188, 322–336. [Google Scholar] [CrossRef] [PubMed]
- Bocos, E.; Alfaya, E.; Iglesias, O.; Pazos, M.; Sanroman, M.A. Application of new sandwich of granular activated and fiber carbon as cathode in the electrochemical advanced oxidation treatment of pharmaceutical effluents. Sep. Purif. Technol. 2015, 212, 299–306. [Google Scholar] [CrossRef]


| Molecular Structure | Molecular Formula | Molecular Weight, g/mol | logKow | pKa | Molecular Radius, nm |
|---|---|---|---|---|---|
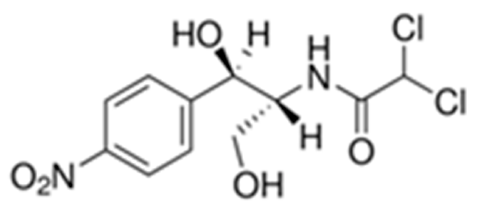 | C11H12Cl2N2O5 | 323.1 | 1.10 | 11.3 | 0.45 |
| Nonlinear Forma | Parameter |
|---|---|
| Pseudo-first-order | |
| qe, mg/g—is the amount of solute adsorbed et equilibrium and qt is the amount of solvent adsorbed at time t k1, h−1—is the rate constant for the pseudo-first-order kinetic model | |
| Pseudo-second-order | |
| qe, mg/g—is the amount of solute adsorbed et equilibrium and qt is the amount of solvent adsorbed at time t k2, h−1—is the rate constant for the pseudo-second-order kinetic model | |
| Elovich | |
| α, mg/(g∙h)—is the initial adsorption rate β, g/mg—is reflected the number of sites available for adsorption | |
| Intraparticle diffusion model (Weber-Morris) | |
| kp, mg/(g∙h0.5)—is the slope which refers to the intra-particle diffusion rate constant C, mg/g—is the intercept which is a constant related to the thickness of the boundary layer | |
| Nonlinear Forma | Linear Form | Parameter | Ref. |
|---|---|---|---|
| Freundlich | |||
| n—adsorption intensity KF, mg/g—Freundlich isotherm constant | [41] | ||
| Langmuir | |||
| qm, mg/g—solid phase concentration corresponding to the complete monolayer coverage of adsorption sites KL, L/mg—the constant related to the free energy of adsorption | [41] | ||
| Dubibin-Radushkevich | |||
| qs, mg/g—is a constant in the Dubinin–Radushkevich isotherm model whit are related to adsorption capacity KD, mol2/kJ2—is a constant in related to the mean free energy of adsorption ε—Dubibin–Radushkevich isotherm constant,
ε = RTln(1 + 1/Ce)
| [41] | ||
| Temkin | |||
| A, L/mg—Tempkin isotherm equilibrium binding constant B—Tempkin isotherm constant | [42] | ||
| where: Ce—equilibrium concentration of solute in bulk solution (mg/L), R is the gas constant, T is the absolute temperature. The constants of the adsorption kinetics and adsorption isotherm equations were determined from nonlinear forms using the Levenberg–Marquardt algorithm [43]. This algorithm was implemented by the scipy.optimize.curve_fit function, which is part of the SciPy library for the Python programming language. Furthermore, using the constant from the Langmuir equation, separator factor RL(10) [38] was calculated. | |||
| Parameter | Unit | Activated Carbon | ||||
|---|---|---|---|---|---|---|
| WG-12 | F-100 | F-300 | Picabiol | ROW 08 Supra | ||
| Surface area | m2/g | 1005 | 730 | 860 | 1344 | 796 |
| Bulk density | g/dm3 | 424 | 560 | 542 | 234 | 381 |
| Iodine adsorption, LI, | mg/g | 1050 | 850 | 1065 | 1071 | 1096 |
| Methylene blue number, LM | cm3 | 30 | 28 | 28 | 30 | 30 |
| Vmicr. Vmeso. Vtotal | cm3/g cm3/g cm3/g | 0.524 0.066 0.995 | 0.332 0.159 0.700 | 0.476 0.294 0.987 | 0.679 0.626 2.103 | 0.436 0.453 1.135 |
| Parameter | Unit | Activated Carbon | ||||
|---|---|---|---|---|---|---|
| F-300 | F-100 | WG-12 | ROW 08 Supra | Picabiol | ||
| Pseudo-first-order | ||||||
| qe | mg/g | 37.45 | 31.44 | 36.67 | 38.19 | 37.84 |
| k1 | h−1 | 0.3878 | 0.3807 | 0.3889 | 0.5004 | 0.7816 |
| R2 | - | 0.9538 | 0.9170 | 0.9405 | 0.9660 | 0.9499 |
| Pseudo-second-order | ||||||
| qe | mg/g | 46.61 | 38.86 | 45.45 | 45.77 | 43.015 |
| k2 | h−1 | 0.0088 | 0.0107 | 0.0092 | 0.0128 | 0.0251 |
| R2 | - | 0.9753 | 0.9542 | 0.9675 | 0.9883 | 0.9942 |
| Elovich | ||||||
| α | mg/(g∙h) | 30.83 | 26.79 | 31.26 | 50.55 | 139.6 3 |
| β | g/mg | 0.0866 | 0.1056 | 0.0898 | 0.0971 | 0.1261 |
| R2 | 0.9877 | 0.9819 | 0.9855 | 0.9884 | 0.9750 | |
| Intraparticle diffusion model (Weber-Morris) | ||||||
| Kp | mg/(g∙h0.5) | 10.8949 | 9.0064 | 10.5620 | 10.3495 | 8.4923 |
| C | mg/g | 5.43 | 4.80 | 5.61 | 9.17 | 15.75 |
| R2 | - | 0.9809 | 0.9959 | 0.9861 | 0.9451 | 0.8866 |
| Parameter | Unit | Activated Carbon | ||||
|---|---|---|---|---|---|---|
| F-300 | F-100 | WG-12 | ROW 08 Supra | Picabiol | ||
| Freundlich | ||||||
| 1/n | - | 0.3665 | 0.3981 | 0.3781 | 0.3288 | 0.3218 |
| KF | mg/g | 20.044 | 12.381 | 17.447 | 29.208 | 31.667 |
| R2 | - | 0.8784 | 0.8928 | 0.8918 | 0.8700 | 0.8698 |
| Langmuir | ||||||
| qm | mg/g | 200,67 | 174,68 | 195.45 | 212.29 | 214.91 |
| KL | L/mg | 0.0197 | 0.0106 | 0.0161 | 0.0354 | 0.0405 |
| R2 | - | 0.9931 | 0.9678 | 0.9873 | 0.9953 | 0.9961 |
| RL | - | 0.03–0.24 | 0.06–0.37 | 0.04–0.28 | 0.02–0.15 | 0.02–0.13 |
| Dubibin–Radushkevich | ||||||
| qs | mg/g | 168.21 | 145.64 | 162.13 | 182.87 | 185.53 |
| KD | mol2/kJ2 | 0.00014 | 0.00061 | 0.00022 | 0.00004 | 0.00003 |
| R2 | - | 0.8818 | 0.9032 | 0.8573 | 0.8886 | 0.8634 |
| Temkin | ||||||
| A | L/mg | 0.2690 | 0.1305 | 0.2690 | 0.5766 | 0.6982 |
| B | - | 37.36 | 34.40 | 37.07 | 36.42 | 36.29 |
| R2 | - | 0.9444 | 0.9206 | 0.9419 | 0.9534 | 0.9541 |
| Parameter | Unit | pH = 2 | pH = 4 | pH = 6 | pH = 8 | pH = 10 |
|---|---|---|---|---|---|---|
| Freundlich | ||||||
| 1/n | - | 0.336 | 0.3557 | 0.3593 | 0.3585 | 0.3566 |
| KF | mg/g | 23.409 | 20.40 | 19.45 | 22.59 | 20.78 |
| R2 | - | 0.871 | 0.8821 | 0.8776 | 0.8961 | 0.8852 |
| Langmuir | ||||||
| qm | mg/g | 207.61 | 190.41 | 189.80 | 192.14 | 195.28 |
| KL | L/mg | 0.0232 | 0.0251 | 0.0191 | 0.0204 | 0.0209 |
| R2 | - | 0.9902 | 0.9952 | 0.9951 | 0.9970 | 0.9974 |
| Dubibin-Radushkevich | ||||||
| qs | mg/g | 170.28 | 160.46 | 158.34 | 160.28 | 162.05 |
| KD | mol2/kJ2 | 0.00006 | 0.00006 | 0.00013 | 0.00010 | 0.00009 |
| R2 | - | 0.8519 | 0.8772 | 0.8765 | 0.87737 | 0.8702 |
| Temkin | ||||||
| A | L/mg | 0.3151 | 0.3739 | 0.2587 | 0.2791 | 0.2841 |
| B | - | 38.58 | 34.13 | 35.43 | 35.65 | 36.39 |
| R2 | - | 0.9800 | 0.9530 | 0.9497 | 0.9581 | 0.9648 |
| Parameter | WG-12 | WG/400AIR | WG/400H2O | WG/400CO2 | WG/800H2O | WG/800CO2 |
|---|---|---|---|---|---|---|
| FBET, m2 | 1098 | 1208 | 1163 | 1181 | 1199 | 1239 |
| Vmikro, cm3/g | 0.530 | 0.538 | 0.542 | 0.551 | 0.548 | 0.553 |
| Vmezo, cm3/g | 0.048 | 0.044 | 0.0455 | 0.0403 | 0.0468 | 0.051 |
| Vmakro, cm3/g | 0.417 | 0.413 | 0.413 | 0.343 | 0.321 | 0.370 |
| B *, mmol/g | 0.467 | 0.535 | 0.521 | 0.525 | 0.546 | 0.539 |
| A *, mmol/g | 0.586 | 0.629 | 0.585 | 0.853 | 0.562 | 0.549 |
| Parameter | WG-12 | WG400AIR | WG400H2O | WG400CO2 | WG800H2O | WG800CO2 |
|---|---|---|---|---|---|---|
| Freundlich | ||||||
| 1/n, - | 0.3781 | 0.3473 | 0.3503 | 0.3640 | 0.4602 | 0.457 |
| KF, mg/g | 17.45 | 23.07 | 24.66 | 24.92 | 20.47 | 29.80 |
| R2 | 0.8918 | 0.9136 | 0.8924 | 0.9007 | 0.9392 | 0.9021 |
| Langmuir | ||||||
| qm, mg/g | 195.45 | 199.81 | 210.58 | 225.64 | 306.54 | 343.12 |
| KL, L/mg | 0.0160 | 0.0235 | 0.0264 | 0.0264 | 0.0184 | 0.0340 |
| R2 | 0.9878 | 0.9950 | 0.9961 | 0.9971 | 0.9881 | 0.9751 |
| Dubibin–Radushkevich | ||||||
| qs | mg/g | 164.99 | 174.68 | 183.46 | 217.86 | 246.28 |
| KD | mol2/kJ2 | 0.00006 | 0.00005 | 0.00005 | 0.00005 | 0.00002 |
| R2 | - | 0.8248 | 0.8497 | 0.8354 | 0.7782 | 0.7885 |
| Temkin | ||||||
| A, L/mg | 0.2690 | 0.1305 | 0.2134 | 0.5766 | 0.6982 | 0.8671 |
| B, - | 37.069 | 36.26 | 38.01 | 41.31 | 60.05 | 65.11 |
| R2 | 0.9419 | 0.9808 | 0.9673 | 0.9785 | 0,9918 | 0.9606 |
| Parameter | SBET | Vmicro | Vmicro + meso | Acidic Groups | Basic Groups |
|---|---|---|---|---|---|
| qm | 0.69 | 0.75 | 0.93 | −0.50 | 0.63 |
| Activated Carbon | Adsorption Conditions | The Equations of Adsorption Kinetics Studied 1 | Adsorption Isotherms Equations Tested 1 | qm, mg/g | Ref. |
|---|---|---|---|---|---|
| AC Picabiol | T*—20 °C C0*—161–646 mg/L D*—400 mg AC/100 mL pH 2–10 | 1. PSO * 2. E * 3. PFO * 4. IPD * | 1.Langmuir 2. Temkin 3. Freundlich 4. D-R * | 214.91 | This work |
| AC WG-12 | 1. E 2. PSO 3. IPD 4. PFO | 195.45 | |||
| WG-12 modified | - | 343.12 | |||
| AC from waste | T—20, 40, 60 °C C0—300–1000 mg/L D—50 mg AC/50 mL pH 2–12 | 1. PSO 2. IPD 3. PFO | 1. Langmuir 2. Freundlich | 160–893 mg/g | [50] |
| AC PEG-400 | T—30, 40, 50 °C C0—100–400 mg/L D—200 mg AC/200 mL | 1. PSO 2. E 3. PFO | 1. Temkin 2. Freundlich 3. Langmuir | 170–209 mg/g | [54] |
| AC from potassium citrate | T—10, 20, 30 °C C0—30–300 mg/L D = 2 mg AC/10 mL pH 3–11 | 1. PSO 2. PFO | 1. Langmuir 2. Freundlich 3. Temkin | 329–506 mg/g | [35] |
| AC from eucalyptus wood biomass | T—25 °C C0—0.25–20 mg/L D—80 mg AC/1000mL pH 4–4.25 | 1. PSO 2. PFO 3. IPD | 1. Freundlich 2. Langmuir | 21 mg/g | [51] |
| AC from Typha orientalis | T—25 °C C0—32.3–96.9 mg/L D—30 mg AC/50 mL pH 2–8 | 1. PSO 2. PFO 3. IPD | 1. Freundlich 2. Langmuir | 137 | [39] |
| F-400 | 140 | ||||
| Bamboo charcoal | T—30 °C C0—5–100 mg/L D—20 mg BC/20 mL pH 7 | - | 1. Freundlich 2. D-R 3. Langmuir | 8.1 | [56] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lach, J. Adsorption of Chloramphenicol on Commercial and Modified Activated Carbons. Water 2019, 11, 1141. https://doi.org/10.3390/w11061141
Lach J. Adsorption of Chloramphenicol on Commercial and Modified Activated Carbons. Water. 2019; 11(6):1141. https://doi.org/10.3390/w11061141
Chicago/Turabian StyleLach, Joanna. 2019. "Adsorption of Chloramphenicol on Commercial and Modified Activated Carbons" Water 11, no. 6: 1141. https://doi.org/10.3390/w11061141
APA StyleLach, J. (2019). Adsorption of Chloramphenicol on Commercial and Modified Activated Carbons. Water, 11(6), 1141. https://doi.org/10.3390/w11061141




