Community Response of Cladocera to Trophic Stress by Biomanipulation in a Shallow Oxbow Lake
Abstract
:1. Introduction
- (1)
- The separate analyses of the contemporary and subfossil Cladocera communities are not representative enough to specify the whole of the Cladocera communities.
- (2)
- Due to intensive fish introduction, the structure and quantitative relationship of the Cladocera community change in the biomanipulated area.
- (3)
- Due to the accumulation capacity of sediment, we may find more species in the subfossil community than in the contemporary community.
2. Materials and Methods
2.1. Processing of Cladocera Samples
2.2. Data Analysis
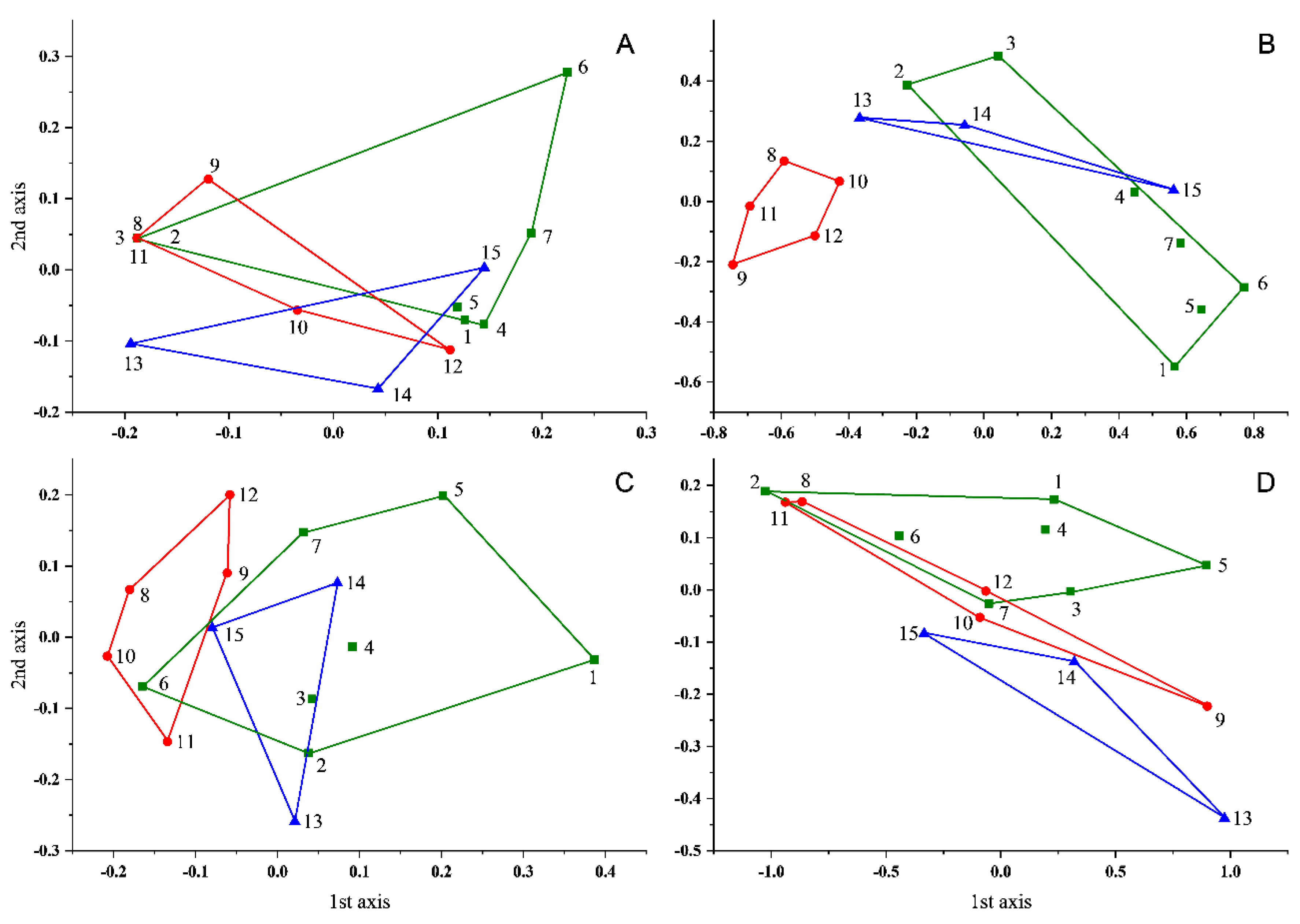
3. Results
3.1. Environmental Variables
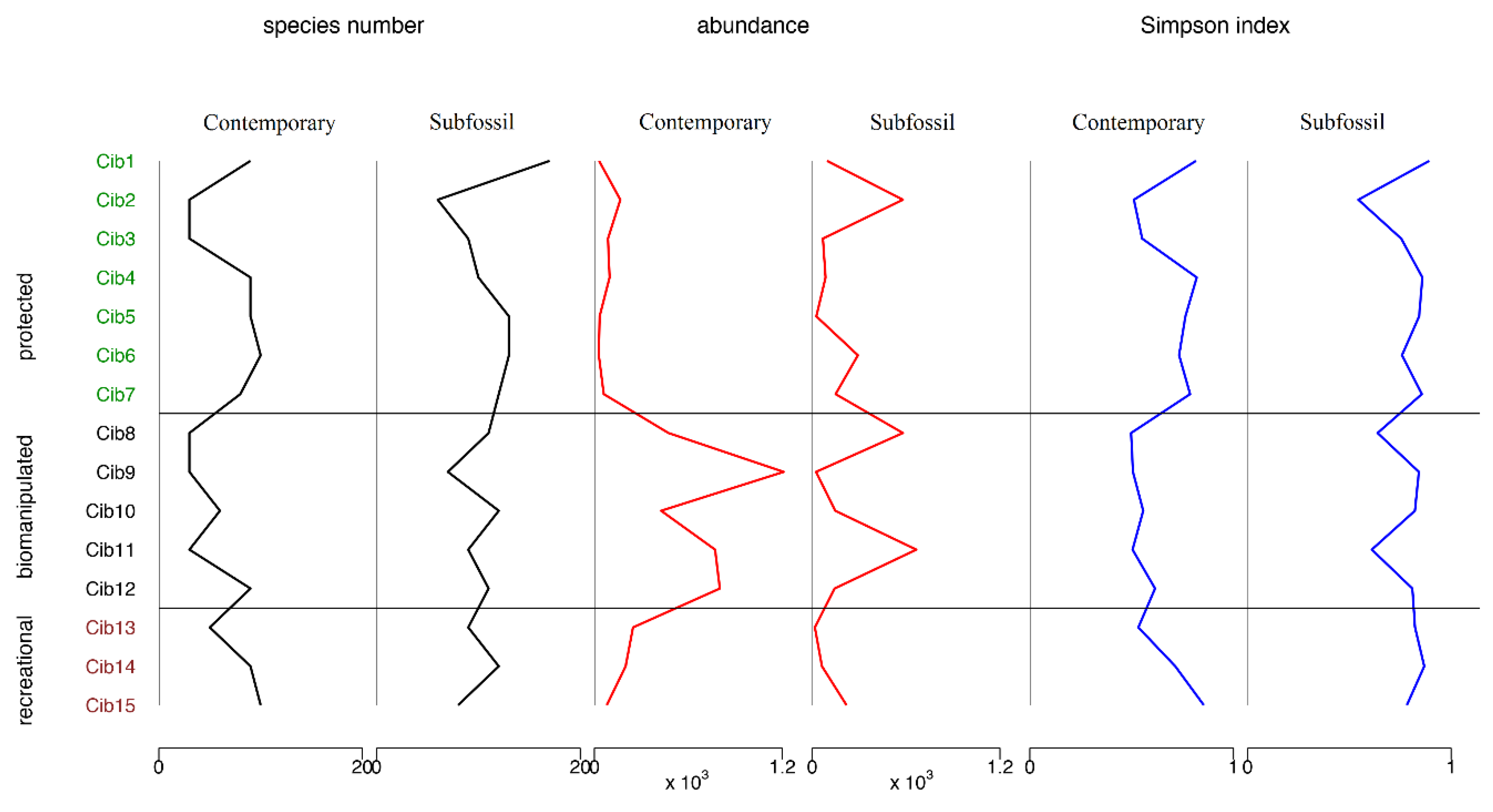
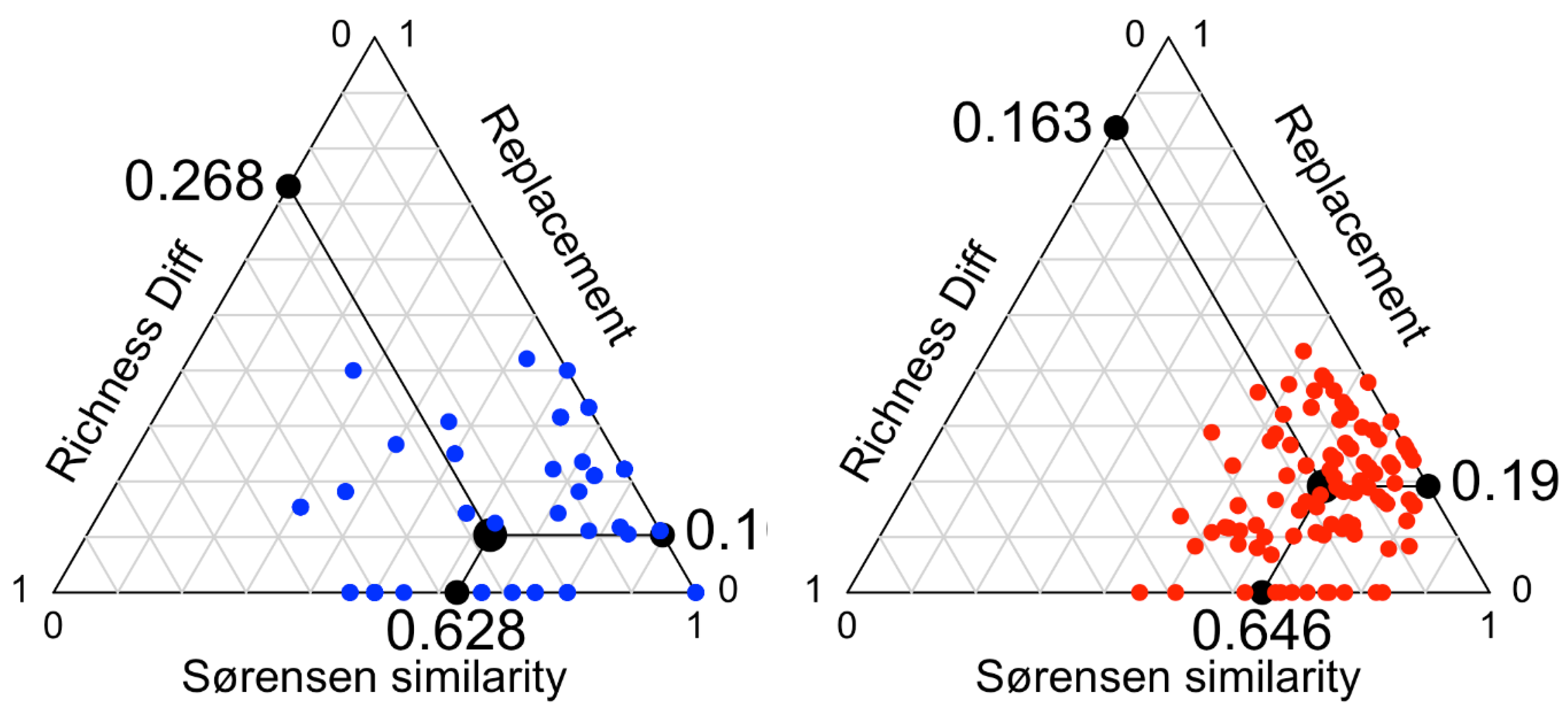
3.2. Contemporary vs. Subfossil Cladocera Communities
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Limburg, K.E.; Waldman, J.R. Dramatic declines in North Atlantic diadromous fishes. BioScience 2009, 59, 955–965. [Google Scholar] [CrossRef]
- Strayer, D.L.; Dudgeon, D. Freshwater biodiversity conservation: Recent progress and future challenges. J. N. Am. Benthol. Soc. 2010, 29, 344–358. [Google Scholar] [CrossRef]
- Vörösmarty, C.J.; McIntyre, P.B.; Gessner, M.O.; Dudgeon, D.; Prusevich, A.; Green, P.; Glidden, S.; Bunn, S.E.; Sullivan, C.A.; Liermann, C.R.; et al. Global threats to human water security and river biodiversity. Nature 2010, 467, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Hoof, R.C.; Peterson, W.T. Copepod biodiversity as an indicator of changes in ocean and climate conditions of the northern California current ecosystem. Limnol. Oceanogr. 2006, 51, 2607–2620. [Google Scholar] [CrossRef]
- Shurin, J.B.; Clasen, J.L.; Greig, H.S.; Kratina, P.; Thompson, P.L. Warming shifts top-down and bottom-up control of pond food web structure and function. Philos. Trans. R. Soc. Lond. Biol. Sci. 2012, 367, 3008–3017. [Google Scholar] [CrossRef] [PubMed]
- Krause, A.E.; Frank, K.A.; Mason, D.M.; Ulanowicz, R.E.; Taylor, W.W. Compartments revealed in food-web structure. Nature 2003, 426, 282–285. [Google Scholar] [CrossRef]
- Sarkar, P.; Das, T. Wetland ecosystem services and its valuation with special reference to India—A review. In Biodiversity and Environmental Conservation; Upadhaya, K., Ed.; Discovery Publishing House Pvt.: New Delhi, India, 2016; pp. 59–76. [Google Scholar]
- Varga, K.; Dévai, G.; Tóthmérész, B. Land use history of a floodplain area during the last 200 years in the Upper-Tisza region (Hungary). Reg. Environ. Chang. 2013, 13, 1109–1118. [Google Scholar] [CrossRef]
- Carneiro, F.M.; Angelini, R.; Carvalho, A.R.; Bini, L.M. Synchrony among limnological variables in a lotic system (Meia Ponte River, Goiás, Brazil). Acta Limnol. Bras. 2009, 21, 287–292. [Google Scholar]
- Hoverman, J.T.; Johnson, P.T.J. Ponds and lakes: A journey through the life aquatic. Nat. Educ. Knowl. 2012, 3, 17. [Google Scholar]
- Pringle, C. What is hydrologic connectivity and why is it ecologically important? Hydrol. Process. 2003, 17, 2685–2689. [Google Scholar] [CrossRef]
- Stella, J.S.; Pratchett, M.S.; Hutchings, P.A.; Jones, G.P. Coral-associated invertebrates: Diversity, ecological importance and vulnerability to disturbance. In Oceanography and Marine Biology: An Annual Review, 1st ed.; Gibson, R.N., Atkinson, R.J.A., Gordon, J.D.M., Eds.; CRC Press: Boca Raton, FL, USA, 2011; Volume 49, pp. 43–109. [Google Scholar]
- Obolewski, K. Macrozoobenthos patterns along environmental gradients and hydrological connectivity of oxbow lakes. Ecol. Eng. 2011, 37, 796–805. [Google Scholar] [CrossRef]
- Grabowska, M.; Mazur-Marzec, H. Vertical distribution of cyanobacteria biomass and cyanotoxin production in the polymictic Siemianówka Dam Reservoir (eastern Poland). Arch. Pol. Fish. 2014, 22, 41–51. [Google Scholar] [CrossRef]
- Kurek, J.; Korosi, J.B.; Jeziorski, A.; Smol, J.P. Establishing reliable minimum count size for cladoceran subfossils sampled from lake sediments. J. Paleolimnol. 2010, 44, 603–612. [Google Scholar] [CrossRef]
- Chaparro, G.; Kandus, P.; O’Farrell, I. Effect of spatial heterogeneity on zooplankton diversity: A multi-scale habitat approximation in a floodplain lake. River Res. Appl. 2013, 31, 85–97. [Google Scholar] [CrossRef]
- Obolewski, K.; Glińska-Lewczuk, K.; Jarząb, N.; Burandt, P.; Kobus, S.; Kujawa, R.; Okruszko, T.; Grabowska, M.; Lew, S.; Goździejewska, A.; et al. Benthic invertebrates in floodplain lakes of a Polish river: Structure and biodiversity analyses in relation to hydrological conditions. Pol. J. Environ. Stud. 2014, 23, 1679–1689. [Google Scholar]
- da Silva, M.T.; Pereira, J.O.; Vieira, L.J.S.; Petry, A.C. Hydrological seasonality of the river affecting fish community structure of oxbow lakes: A limnological approach on the Amapá Lake, southwestern Amazon. Limnologica 2013, 43, 79–90. [Google Scholar] [CrossRef]
- Hunt, R.J.; Matveev, V.F. The effects of nutrients and zooplankton community structure on phytoplankton growth in a subtropical Australian reservoir: An enclosure study. Limnologica 2005, 35, 90–101. [Google Scholar] [CrossRef]
- O’Brien, W.J.; de Noyelles, F., Jr. Relationship between nutrient concentration, phytoplankton density, and zooplankton density in nutrient enriched experimental ponds. Hydrobiologia 1974, 44, 105–125. [Google Scholar] [CrossRef]
- Tibúrcio, V.G.; Arreieira, R.L.; Schwind, L.T.F.; Bonecker, C.C.; Lansac-Tôha, F.A. Effects of nutrients increase on the copepod community of a reservoir using cages. Acta Limnol. Bras. 2015, 27, 265–274. [Google Scholar] [CrossRef]
- Christoffersen, K.; Riemann, B.; Klysner, A.; Søndergaard, M. Potential role of fish predation and natural populations of zooplankton in structuring a plankton community in eutrophic lake water. Limnol. Oceanogr. 1993, 38, 561–573. [Google Scholar] [CrossRef]
- Amundsen, P.-A.; Lafferty, K.D.; Knudsen, R.; Primicerio, R.; Kristofferesen, R.; Klemetsen, A.; Kuris, A.M. New parasites and predators follow the introduction of two fish species to a subarctic lake: Implications for food-web structure and functioning. Oecologia 2013, 171, 993–1002. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, C.; Mazzeo, N.; Meerhoff, M.; Lacerot, G.; Clemente, J.M.; Scasso, F.; Kruk, C.; Goyenola, G.; García-Alonso, J.; Amsinck, S.L.; et al. High predation is of key importance for dominance of small-bodied zooplankton in warm shallow lakes: Evidence from lakes, fish exclosures and surface sediments. Hydrobiologia 2011, 667, 133–147. [Google Scholar] [CrossRef]
- Persson, L.; Byström, P.; Wahlström, E.; Westman, E. Trophic dynamics in a whole lake experiment: Size-structured interactions and recruitment variation. Oikos 2004, 106, 263–274. [Google Scholar] [CrossRef]
- Haberman, J. Dominant zooplankton species in Lake Peipsi. Estonian J. Ecol. 2000, 49, 34–51. [Google Scholar]
- Mehner, T.; Keeling, C.; Emmrich, M.; Holmgren, K.; Argillier, C.; Volta, P.; Winfield, I.J.; Brucet, S. Effects of fish predation on density and size spectra of prey fish communities in lakes. Can. J. Fish. Aquat. Sci. 2016, 73, 506–518. [Google Scholar] [CrossRef]
- Gliwicz, Z.M. Predation or food limitation: An ultimate reason for extinction of planktonic cladoceran species. Arch. Hydrobiol. Beih. Ergeb. Limnol. 1985, 21, 419–430. [Google Scholar]
- Jeppesen, E.; Jensen, J.P.; Søndergaard, M.; Lauridsen, T.; Landkildehus, F. Trophic structure, species richness and biodiversity in Danish lakes: Changes along a phosphorus gradient. Freshw. Biol. 2010, 45, 201–218. [Google Scholar] [CrossRef]
- Brooks, J.L.; Dodson, S.I. Predation, body size, and composition of plankton. Science 1965, 150, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Gliwicz, Z.M. Between hazards of starvation and risk of predation: The ecology of offshore animals. In Excellence in Ecology; Kinne, O., Ed.; International Ecology Institute: Oldendorf/Luhe, Germany, 2003; Volume 12, pp. 1–379. [Google Scholar]
- Boltizár, O.; Müller, T.; Csenki, Z.; Bakos, K.; Staszny, Á.; Hegyi, Á.; Kucska, B.; Kucharczyk, D.; Horváth, L. Predatory effect of Copepods on the larvae of some freshwater fish. Fundam. Appl. Limnol. 2017, 190, 349–356. [Google Scholar] [CrossRef]
- Frimpong, E.A.; Lochmann, S.E. Mortality of fish larvae exposed to varying concentrations of cyclopoid copepods. N. Am. J. Aquac. 2005, 67, 66–71. [Google Scholar] [CrossRef]
- Letcher, B.H.; Rice, J.A.; Crowder, L.B.; Rose, K.A. Variability in survival of larval fish: Disentangling components with a generalized individual-based model. Can. J. Fish. Aquat. Sci. 1996, 53, 787–801. [Google Scholar] [CrossRef]
- Spectiár, A. A Balaton halfaunája: A halállomány összetétele, az egyes halfajok életkörülményei és a halállomány korszerű hasznosításának feltételrendszere. Acta Biol. Debr. Oecol. Hung. 2010, 23, 7–185. [Google Scholar]
- Schindler, D.W. Two useful devices for vertical plankton and water sampling. J. Fish. Res. Board Can. 1969, 26, 1948–1955. [Google Scholar] [CrossRef]
- Glew, J.R. Miniature gravity corer for recovering short sediment cores. J. Paleolimnol. 1991, 5, 285–287. [Google Scholar] [CrossRef]
- Dean, W. Determination of carbonate and organic matter in calcareous sediments and sedimentary rocks by loss on ignition: Comparison with other methods. J. Sediment. Res. 1974, 44, 242–248. [Google Scholar] [CrossRef]
- Korhola, A.; Rautio, M. Cladocera and other branchiopod crustaceans. In Tracking Environmental Change Using Lake Sediments: Biological Techniques and Indicators; Smol, J.P., Birks, H.J.B., Last, W.M., Eds.; Kluwer Academic Publisher: Dordrecht, The Netherlands, 2001; Volume 2, pp. 1–38. [Google Scholar]
- Frey, D.G. The taxonomy and biogeography of the Cladocera. Hydrobiologia 1987, 145, 5–17. [Google Scholar] [CrossRef]
- Szeroczyńska, K.; Sarmaja-Korjonen, K. Atlas of Subfossil Cladocera from Central and Northern Europe; Friends of the Lower Vistula Society: Świecie, Poland, 2007; pp. 1–84. [Google Scholar]
- Gulyás, P.; Forró, L. Az Ágascsápú Rákok (Cladocera) Kishatározója, 2nd ed.; Környezetgazdálkodási Intézet: Budapest, Hungary, 1999; pp. 1–237. [Google Scholar]
- Dray, S.; Bauman, D.; Blanchet, G.; Borcard, D.; Clappe, S.; Guenard, G.; Jombart, T.; Larocque, G.; Legendre, P.; Madi, N.; et al. Adespatial: Multivariate Multiscale Spatial Analysis. 2018. Available online: http://CRAN.R-project.org/package=adespatial (accessed on 18 February 2019).
- Podani, J.; Schmera, D. A new conceptual and methodological framework for exploring and explaining pattern in presence-absence data. Oikos 2011, 120, 1625–1638. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: http://www.R-project.org/ (accessed on 3 March 2018).
- Hammer, Ø.; Harper, D.A.T.; Paul, D.R. Past: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Legendre, P.; Legendre, L. Numerical Ecology (Developments in Environmental Modelling), 2nd ed.; Elsevier Science BV.: Amsterdam, The Netherlands, 1998; pp. 1–853. [Google Scholar]
- Davidson, T.A.; Sayer, C.D.; Perrow, M.R.; Bramm, M.; Jeppesen, E. Are the controls of species composition similar for contemporary and sub-fossil cladoceran assemblages? A study of 39 shallow lakes of contrasting trophic status. J. Paleolimnol. 2007, 38, 117–134. [Google Scholar] [CrossRef]
- Kattel, G.R.; Battarbee, R.W.; Mackay, A.; Birks, H.J.B. Are cladoceran fossils in lake sediment samples a biased reflection of the communities from which they are derived? J. Paleolimnol. 2007, 38, 157–181. [Google Scholar] [CrossRef]
- Davidson, T.A.; Amsinck, S.L.; Bennike, O.; Christoffersen, K.S.; Landkildehus, F.; Lauridsen, T.L.; Jeppesen, E. Inferring a single variable from an assemblage with multiple controls: Getting into deep water with cladoceran lake-depth transfer functions. Hydrobiologia 2011, 676, 129–142. [Google Scholar] [CrossRef]
- Korhola, A.; Tikkanen, M.; Weckström, J. Quantification of Holocene lake-level changes in Finnish Lapland using a cladocera—Lake depth transfer model. J. Paleolimnol. 2005, 34, 175–190. [Google Scholar] [CrossRef]
- Szeroczyńska, K.; Zawisza, E. Subfossil faunal and floral remains (Cladocera, Pediastrum) in two northern Lobelia lakes in Finland. Knowl. Manag. Aquat. Ecosyst. 2011, 402, 9–23. [Google Scholar] [CrossRef]
- Tolotti, M.; Milan, M.; Szeroczyńska, K. Subfossil Cladocera as a powerful tool for paleoecological reconstruction. Adv. Oceanogr. Limnol. 2016, 7, 125–130. [Google Scholar] [CrossRef]
- Gliwicz, Z.M. Food thresholds and body size in cladocerans. Nature 1990, 343, 638–640. [Google Scholar] [CrossRef]
- Boersma, M.; van Densen, W.L.T.; Vijverberg, J. The effect of predation by smelt (Osmerus eperlanus) on Daphnia hyalina in a shallow eutrophic lake. Verh. Int. Ver. Limnol. 1991, 24, 2438–2442. [Google Scholar] [CrossRef]
- Sarvala, J.; Helminen, H.; Saarikari, V.; Salonen, S.; Vuorio, K. Relations between planktivorous fish abundance, zooplankton and phytoplankton in three lakes of differing productivity. In Eutrophication in Planktonic Ecosystems: Food Web Dynamics and Elemental Cycling. Developments in Hydrobiology; Tamminen, T., Kuosa, H., Eds.; Springer: Dordrecht, The Netherlands, 1998; Volume 127, pp. 81–95. [Google Scholar] [CrossRef]
- Korponai, J.; Mátyás, K.; Paulovits, G.; Tátrai, I.; Kovács, N. The effect of different fish communities on the cladocerans plankton assemblages of the Kis-Balaton Reservoir, Hungary. In Cladocera: The Biology of Model Organisms. Developments in Hydrobiology; Branceli, A., De Meester, L., Spaak, P., Eds.; Springer: Dordrecht, The Netherlands, 1997; Volume 126, pp. 211–221. [Google Scholar] [CrossRef]
- Jeppesen, E.; Christoffersen, K.; Landkildehus, F.; Lauridsen, T.; Amsinck, S.L.; Riget, F.; Søndergaard, M. Fish and crustaceans in northeast Greenland lakes with special emphasis on interactions between Arctic charr (Salvelinus alpinus), Lepidurus arcticus and benthic chydorids. Hydrobiologia 2001, 442, 329–337. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Amsinck, S.L.; Landkildehus, F.; Lauridsen, T.; Mitchell, S.F. Reconstruction the historical changes in Daphnia mean size and planktivorous fish abundance in lakes from the size of Daphnia ephippia in the sediment. J. Paleolimnol. 2002, 27, 133–143. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Lauridsen, T.L.; Amsinck, S.L.; Christoffersen, K.; Søndergaard, M.; Mitchell, S.F. Sub-fossils of cladocerans in the surface sediment of 135 lakes as proxies for community structure of zooplankton, fish abundance and lake temperature. Hydrobiologia 2003, 491, 321–330. [Google Scholar] [CrossRef]
- Amsinck, S.L.; Strzelczak, A.; Bjerring, R.; Landkildehus, F.; Lauridsen, T.L.; Christoffersen, K.; Jeppesen, E. Lake depth rather than fish planktivory determines cladoceran community structure in Faroese lakes—Evidence from contemporary data and sediments. Freshw. Biol. 2006, 51, 2124–2142. [Google Scholar] [CrossRef]
- Brendonck, L.; De Meester, L. Egg banks in freshwater zooplankton: Evolutionary and ecological archives in the sediment. Hydrobiologia 2003, 491, 65–84. [Google Scholar] [CrossRef]
- Vandekerkhove, J.; Declerck, S.; Brendonck, L.; Conde-Porcuna, J.M.; Jeppesen, E.; Johansson, L.S.; De Meester, L. Uncovering hidden species: Hatching diapausing eggs for the analysis of cladoceran species richness. Limnol. Oceanogr. 2005, 3, 399–407. [Google Scholar] [CrossRef]
- Vad, C.F.; Horváth, Z.; Kiss, K.T.; Ács, É. Microcrustacean (Cladocera, Copepoda) communities in artificial lakes in the region of the North Hungarian mountains, with special reference to the adventive species. Acta Zool. Acad. Sci. Hung. 2012, 58, 47–61. [Google Scholar]
- Tóth, A.; Horváth, Z.; Vad, C.F.; Zsuga, K.; Nagy, S.A.; Boros, E. Zooplankton of the European soda pans: Fauna and conservation of a unique habitat type. Int. Rev. Hydrobiol. 2014, 99, 255–276. [Google Scholar] [CrossRef]
- Brucet, S.; Boix, D.; Quintana, X.D.; Jensen, E.; Nathansen, L.W.; Trochine, C.; Meerhoff, M.; Gascón, S.; Jeppesen, E. Factors influencing zooplankton size structure at contrasting temperatures in coastal shallow lakes: Implications for effects of climate change. Limnol. Oceanogr. 2010, 55, 1697–1711. [Google Scholar] [CrossRef]
- Reissing, M.; Trochine, C.; Queimaliños, C.; Balseiro, E.; Modenutti, B. Impact of fish introduction on planktonic food webs in lakes of the Patagonian Plateau. Biol. Conserv. 2006, 132, 437–447. [Google Scholar] [CrossRef]
- Çakiroğlu, A.I.; Tavşanoğlu, Ü.N.; Levi, E.E.; Davidson, T.A.; Bucak, T.; Özen, A.; Akyildiz, G.K.; Jeppesen, E.; Beklioğlu, M. Relatedness between contemporary and subfossil cladoceran assemblages in Turkish lakes. J. Paleolimnol. 2014, 52, 367–383. [Google Scholar] [CrossRef]
- García-Girón, J.; Fernández-Aláez, C.; Fernández-Aláez, M.; Luis, B. Subfossil Cladocera from surface sediment reflect contemporary assemblages and their environmental controls in Iberian flatland ponds. Ecol. Indic. 2018, 87, 33–42. [Google Scholar] [CrossRef]
- Jeppesen, E.; Nõges, P.; Davidson, T.A.; Haberman, J.; Nõges, T.; Blank, K.; Lauridsen, T.L.; Søndergaard, M.; Sayer, C.; Laugaste, C.; et al. Zooplankton as indicators in lakes: A scientific-based plea for including zooplankton in the ecological quality assessment of lakes according to the European Water Framework Directive (WFD). Hydrobiologia 2011, 676, 279–297. [Google Scholar] [CrossRef]
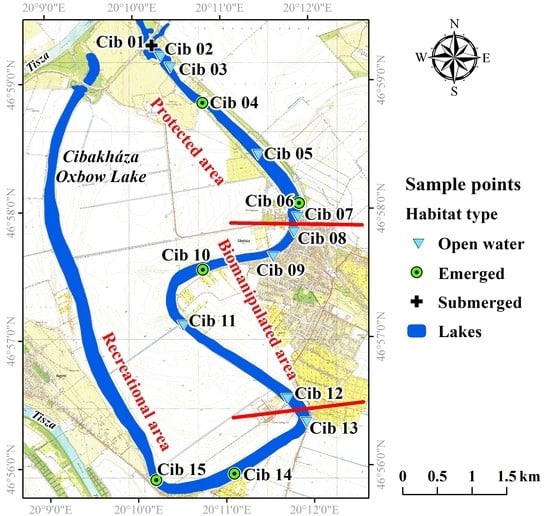
| Protected Area | Biomanipulated Area | Recreational Area | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Min. | Max. | Med. | Min. | Max. | Med. | Min. | Max. | Med. | |
| Conductivity (µS cm−1) | 398 | 788 | 424 | 709 | 1018 | 829 | 1142 | 1215 | 1180 |
| Depth (m) | 0.49 | 4.74 | 0.77 | 0.76 | 3.62 | 1.15 | 0.36 | 6.22 | 1.76 |
| DIN* (mg L−1) | 0.76 | 3.27 | 1.91 | 0.63 | 1.55 | 1.27 | 0.67 | 1.43 | 0.93 |
| Dissolved oxygen (mg L−1) | 1.27 | 9.84 | 4.36 | 7.1 | 10.7 | 8.09 | 3.62 | 9.25 | 6.84 |
| Open water surface (%) | 0 | 100 | 60 | 15 | 100 | 100 | 5 | 100 | 50 |
| Ortho-phosphate (mg L−1) | 0 | 0.60 | 0.05 | 0 | 0 | 0 | 0 | 0.29 | 0.01 |
| pH | 7.6 | 8.6 | 7.9 | 8.3 | 8.7 | 8.5 | 7.8 | 8.9 | 8.7 |
| SS** (mg L−1) | 0.6 | 9.6 | 1.8 | 1.8 | 2.9 | 2.3 | 1.8 | 3.1 | 2.7 |
| Transparency (m) | 0.39 | 0.77 | 0.59 | 0.46 | 0.64 | 0.54 | 0.36 | 0.73 | 0.73 |
| CaCO3 (%) | 1.67 | 10.51 | 7.07 | 2.15 | 16.12 | 6.25 | 2.36 | 6.87 | 5.79 |
| LOI*** (%) | 7.99 | 78.28 | 9.90 | 1.03 | 25.81 | 15.06 | 2.59 | 30.80 | 15.10 |
| Chlorophyll-a (mg m−3) | 2.17 | 22.18 | 7.07 | 7.98 | 11.09 | 7.07 | 5.65 | 13.65 | 13.44 |
| Submerged coverage (%) | 0 | 91 | 0 | 0 | 5 | 0 | 0 | 5 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berta, C.; Tóthmérész, B.; Wojewódka, M.; Augustyniuk, O.; Korponai, J.; Bertalan-Balázs, B.; Nagy, A.S.; Grigorszky, I.; Gyulai, I. Community Response of Cladocera to Trophic Stress by Biomanipulation in a Shallow Oxbow Lake. Water 2019, 11, 929. https://doi.org/10.3390/w11050929
Berta C, Tóthmérész B, Wojewódka M, Augustyniuk O, Korponai J, Bertalan-Balázs B, Nagy AS, Grigorszky I, Gyulai I. Community Response of Cladocera to Trophic Stress by Biomanipulation in a Shallow Oxbow Lake. Water. 2019; 11(5):929. https://doi.org/10.3390/w11050929
Chicago/Turabian StyleBerta, Csaba, Béla Tóthmérész, Marta Wojewódka, Olga Augustyniuk, János Korponai, Boglárka Bertalan-Balázs, Alex Sándor Nagy, István Grigorszky, and István Gyulai. 2019. "Community Response of Cladocera to Trophic Stress by Biomanipulation in a Shallow Oxbow Lake" Water 11, no. 5: 929. https://doi.org/10.3390/w11050929
APA StyleBerta, C., Tóthmérész, B., Wojewódka, M., Augustyniuk, O., Korponai, J., Bertalan-Balázs, B., Nagy, A. S., Grigorszky, I., & Gyulai, I. (2019). Community Response of Cladocera to Trophic Stress by Biomanipulation in a Shallow Oxbow Lake. Water, 11(5), 929. https://doi.org/10.3390/w11050929