Methane Emissions Driven by Adding a Gradient of Ethanol as Carbon Source in Integrated Vertical-Flow Constructed Wetlands
Abstract
1. Introduction
2. Material and Methods
2.1. Study Area
2.2. Integrated Vertical-Flow Constructed Wetland
2.3. Experimental Design and Operation
2.4. Carbon Utilization Analysis
2.5. Analytical Methods
2.6. Statistical Analysis
3. Results
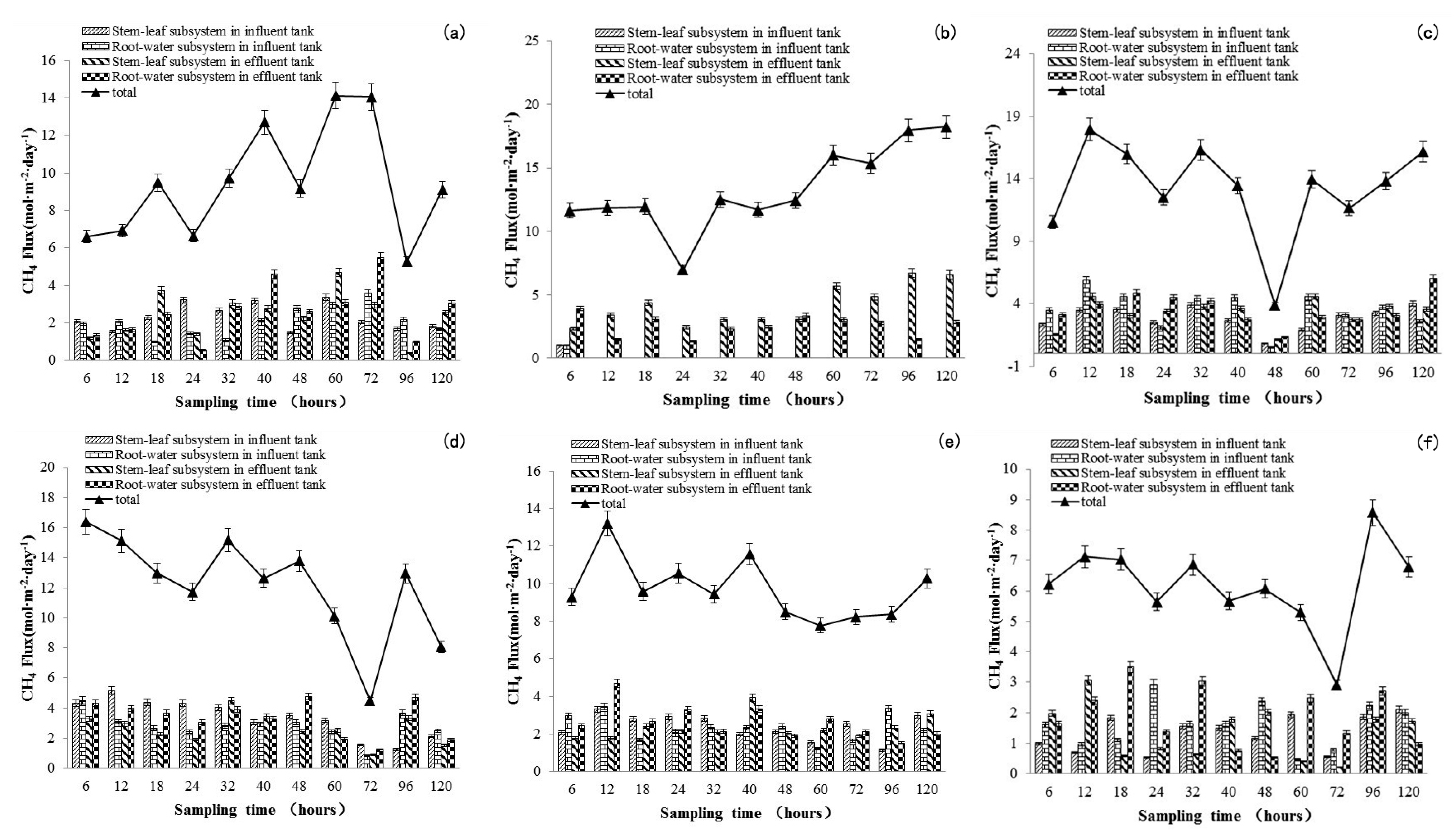
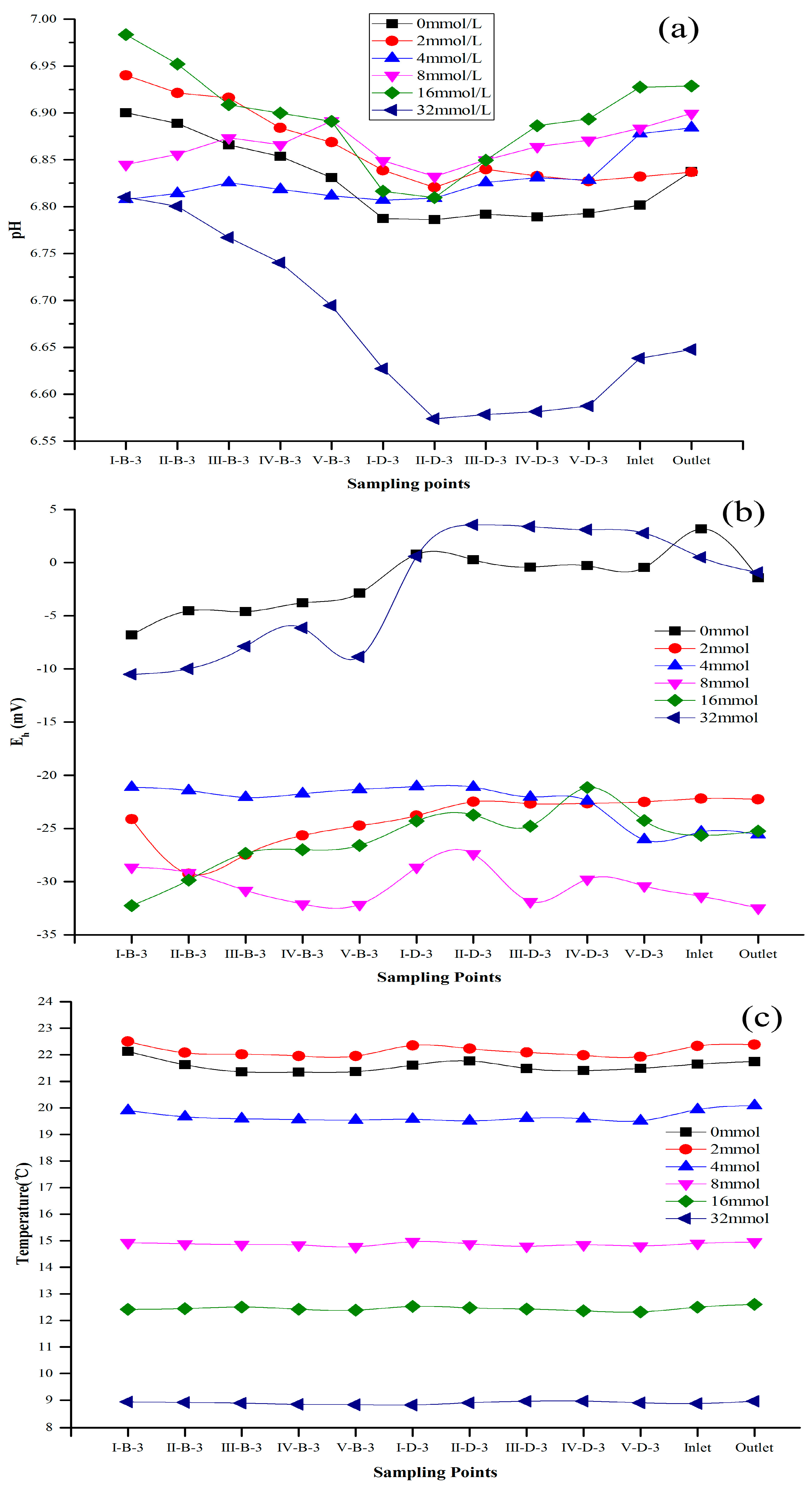
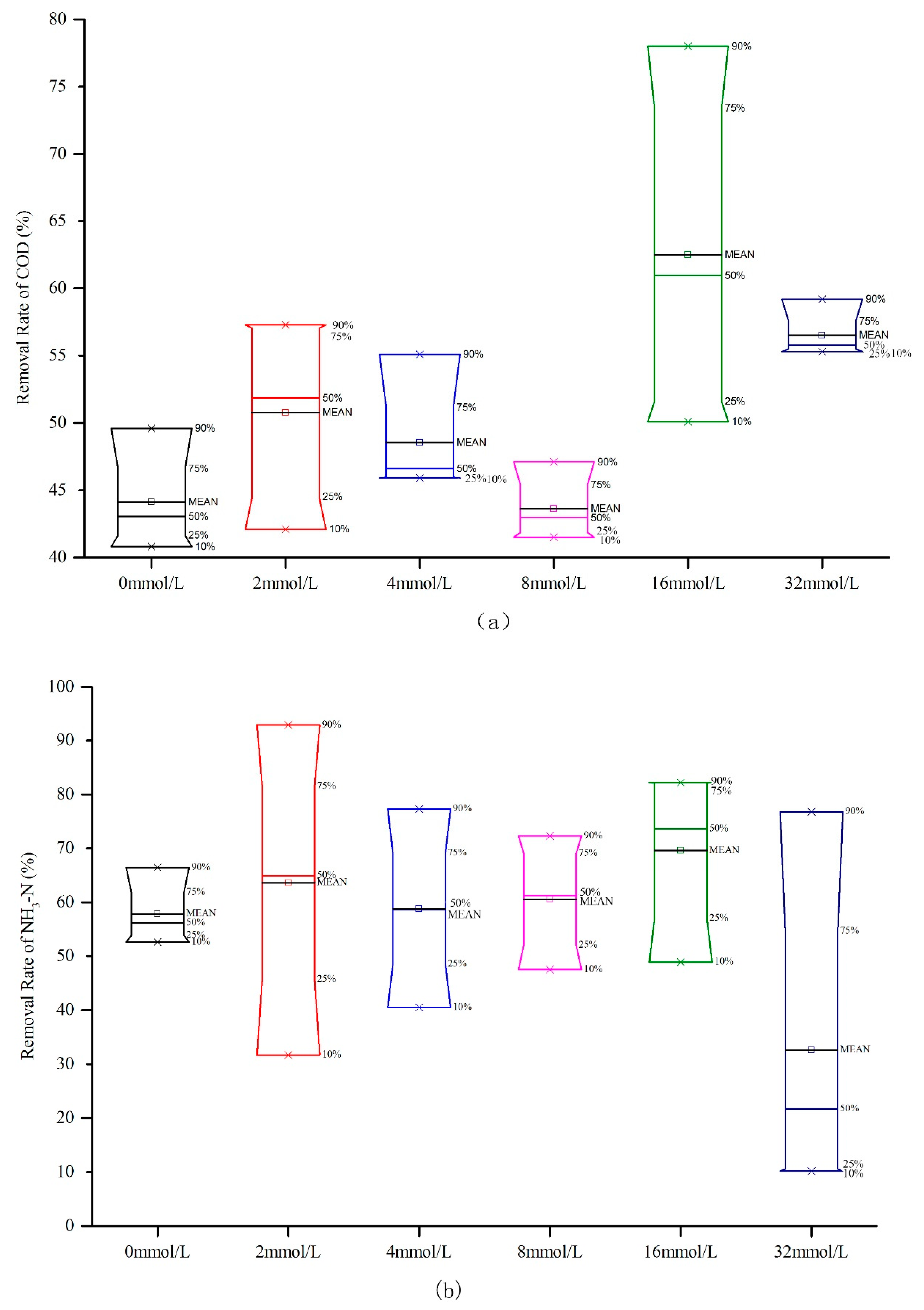
3.1. Methane Emissions and Effects on Water Quality Driven by Ethanol Addition in the IVCW
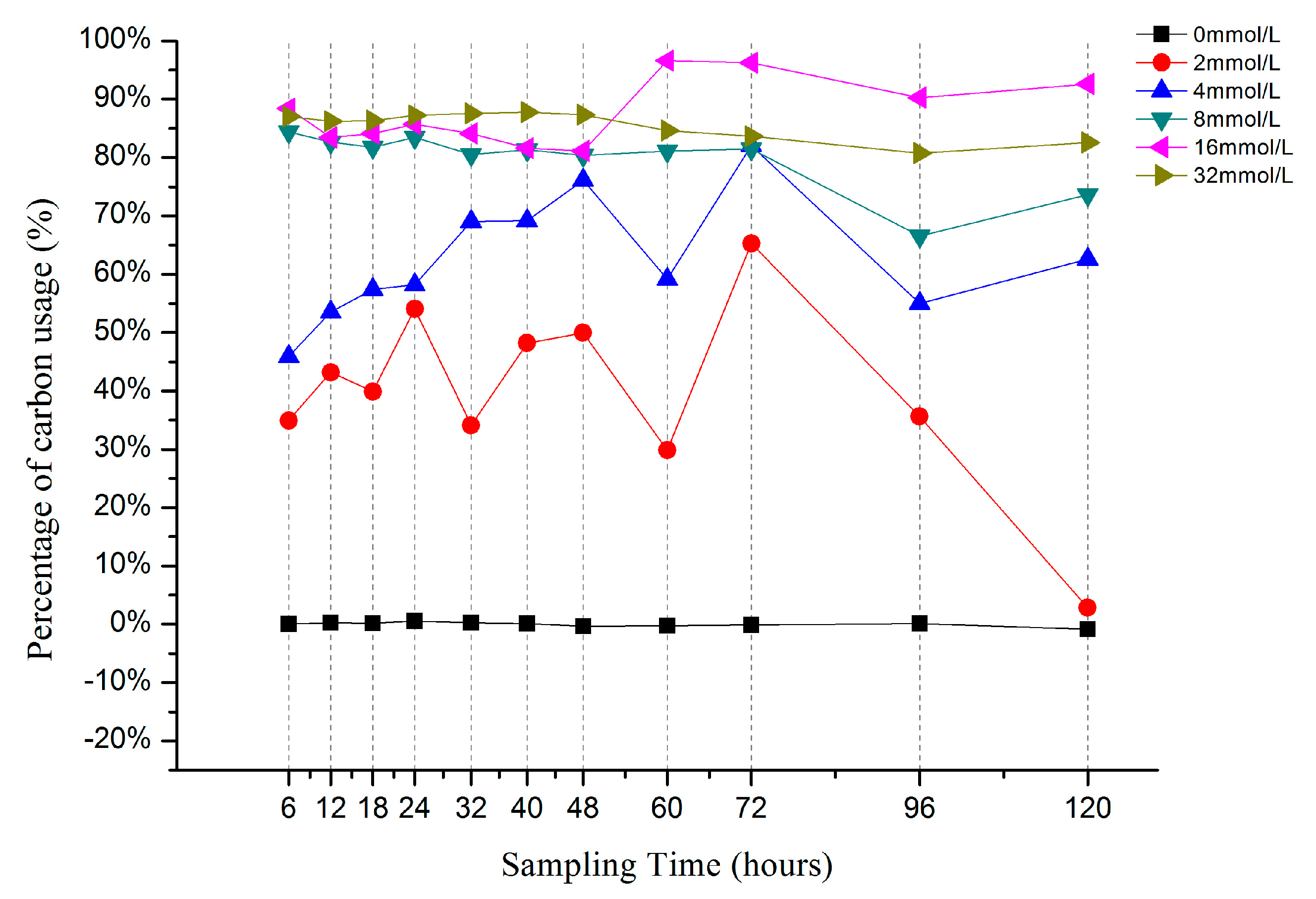
3.2. Carbon utilization of Different Ethanol Concentrations in the IVCW
3.3. CH4-C Emission
4. Discussion
4.1. Methane Emission Flux Effects Driven by Ethanol and Water Quality
4.2. Methane Emission Impacted on C/N Ratio in IVCW
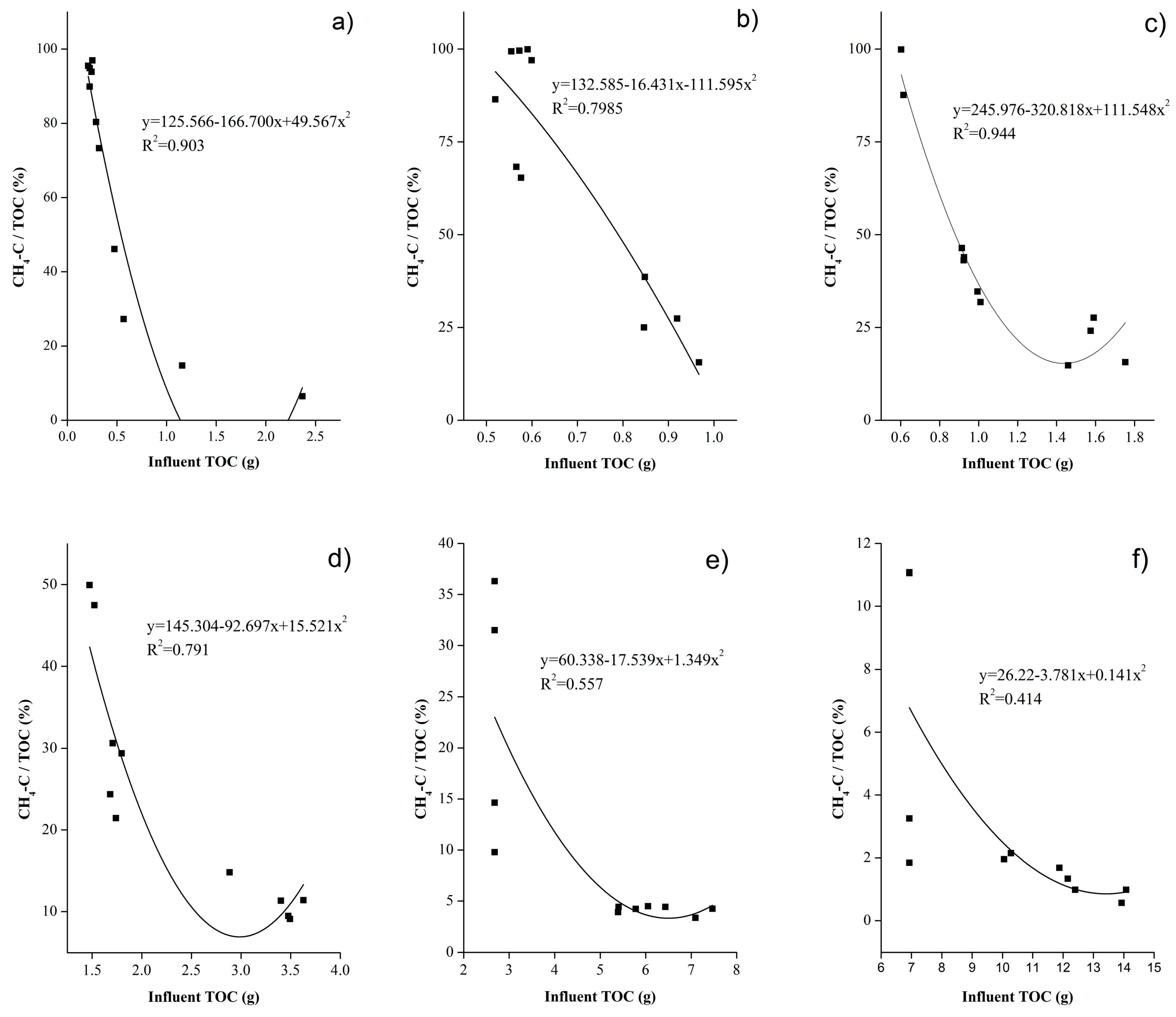
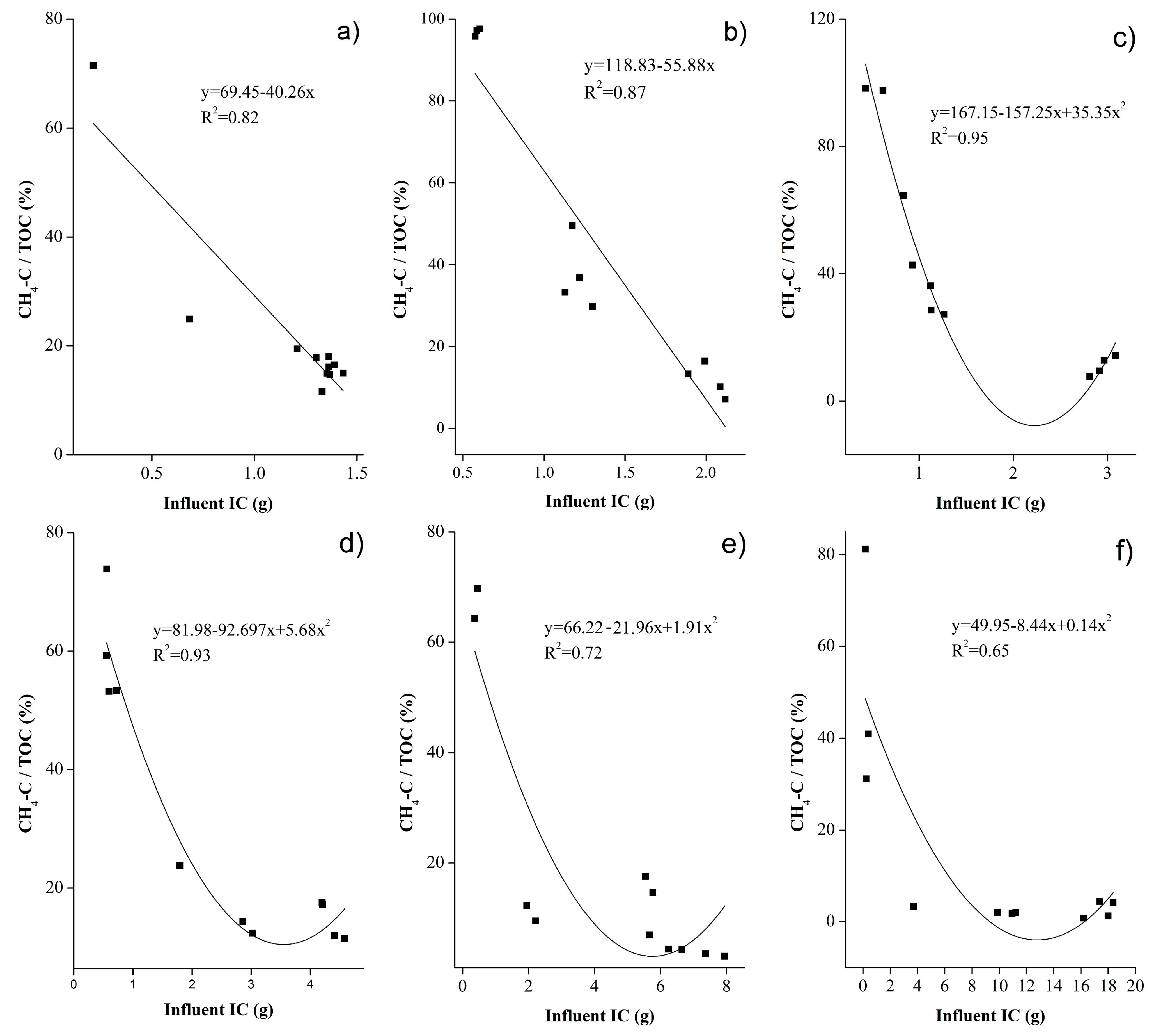
4.3. Analysis of CH4-C/TOC and CH4-C/IC
4.4. Carbon Cycle in IVCW
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mitsch, W.J.; Zhang, L.; Waletzko, E.; Bernal, B. Validation of the ecosystem services of created wetlands: Two decades of plant succession, nutrient retention, and carbon sequestration in experimental riverine marshes. Ecol. Eng. 2014, 72, 11–24. [Google Scholar] [CrossRef]
- Ockenden, M.C.; Deasy, C.; Quinton, J.N.; Bailey, A.P.; Surridge, B.; Stoate, C. Evaluation of field wetlands for mitigation of diffuse pollution from agriculture: Sediment retention, cost and effectiveness. Environ. Sci. Policy 2012, 24, 110–119. [Google Scholar] [CrossRef]
- Fu, G.; Yu, T.; Ning, K.; Guo, Z.; Wong, M. Effects of nitrogen removal microbes and partial nitrification-denitrification in the integrated vertical-flow constructed wetland. Ecol. Eng. 2016, 95, 83–89. [Google Scholar] [CrossRef]
- Peng, L.; Hua, Y.; Cai, J.; Zhao, J.; Zhou, W.; Zhu, D. Effects of plants and temperature on nitrogen removal and microbiology in a pilot-scale integrated vertical-flow wetland treating primary domestic wastewater. Ecol. Eng. 2014, 64, 285–290. [Google Scholar] [CrossRef]
- de la Varga, D.; Ruiz, I.; Álvarez, J.A.; Soto, M. Methane and carbon dioxide emissions from constructed wetlands receiving anaerobically pretreated sewage. Sci. Total Environ. 2015, 538, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Reinman, S.L. Intergovernmental Panel on Climate Change (IPCC). Ref. Rev. 2012, 26, 41–42. [Google Scholar]
- Pearce, W.; Holmberg, K.; Hellsten, I.; Nerlich, B. Climate change on Twitter: topics, communities and conversations about the 2013 IPCC Working Group 1 report. PLoS ONE 2014, 9, e94785. [Google Scholar] [CrossRef]
- Tan, Z. Air Pollution and Greenhouse Gases-From Basic Concepts to Engineering Applications for Air Emission Control; Springer Science Business Media: Singapore, 2014; 418p, ISBN 978-981-287-212-8. [Google Scholar]
- Li, X.; Mitsch, W.J. Methane emissions from created and restored freshwater and brackish marshes in southwest Florida, USA. Ecol. Eng. 2016, 91, 529–536. [Google Scholar] [CrossRef]
- IPCC. 2013 Supplement to the 2006 IPCC Guidelines for National Greenhouse Gas Inventories: Wetlands. In Methodological Guidance on Lands with Wet and Drained Soils, and Constructed Wetlands for Wastewater Treatment; Takahiko, H., Thelma, K., Kiyoto, T., Nalin, S., Baasansuren, J., Maya, F., Tiffany, T., Eds.; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Bloom, A.A.; Palmer, P.I.; Fraser, A.; Reay, D.S.; Frankenberg, C. Large-Scale Controls of Methanogenesis Inferred from Methane and Gravity Spaceborne Data. Science 2010, 327, 322. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Bernal, B.; Nahlik, A.M.; Mander, Ü.; Zhang, L.; Anderson, C.J.; Jørgensen, S.E.; Brix, H. Wetlands, carbon, and climate change. Landscape Ecol. 2012, 28, 583–597. [Google Scholar] [CrossRef]
- Niu, C.; He, Z.; Ge, Y.; Chang, J.; Lu, Z. Effect of plant species richness on methane fluxes and associated microbial processes in wetland microcosms. Ecol. Eng. 2015, 84, 250–259. [Google Scholar] [CrossRef]
- Liu, D.Y.; Ding, W.X. Progress on Spatial Variation of Methanogens and Their Influencing Factors in Natural Wetlands. Sci. Geog. Sin. 2011, 31, 136–142. [Google Scholar]
- Pangala, S.R.; Reay, D.S.; Heal, K.V. Mitigation of methane emissions from constructed farm wetlands. Chemosphere 2010, 78, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Inamori, R.; Kong, H.; Xu, K.; Inamori, Y.; Kondo, T.; Zhang, J. Influence of plant species and wastewater strength on constructed wetland methane emissions and associated microbial populations. Ecol. Eng. 2008, 32, 22–29. [Google Scholar] [CrossRef]
- Lu, S.; Hu, H.; Sun, Y.; Yang, J. Effect of carbon source on the denitrification in constructed wetlands. J Environ. Sci. (China) 2009, 21, 1036–1043. [Google Scholar] [CrossRef]
- Zhao, L.; Zhu, W. Nitrogen removal mechanism in constructed wetland used for treating polluted river water with lower ratio of carbon to nitrogen. Acta Scientiae Circumstantiae 2006, 26, 1821–1827. [Google Scholar]
- Ragab, M.; Mohamed, S.; Mehana, T.; Aldag, R. Denitrification and nitrogen immobilization as affected by organic matter and different forms of nitrogen added to an anaerobic water-sediment system. Biol. Fertil. Soils 1994, 17, 219–224. [Google Scholar] [CrossRef]
- Tang, J.; Wang, X.C.; Hu, Y.; Ngo, H.H.; Li, Y.; Zhang, Y. Applying fermentation liquid of food waste as carbon source to a pilot-scale anoxic/oxic-membrane bioreactor for enhancing nitrogen removal: Microbial communities and membrane fouling behaviour. Bioresour. Technol. 2017, 236, 164–173. [Google Scholar] [CrossRef]
- Xing, L.; Yang, S.; Yin, Q.; Xie, S.; Strong, P.J.; Wu, G. Effects of carbon source on methanogenic activities and pathways incorporating metagenomic analysis of microbial community. Bioresour. Technol. 2017, 244, 982–988. [Google Scholar] [CrossRef]
- Li, S.Y. Effect of organic carbon resource on specific methanogenic activity of anaerobic microorganism. J. Anhui Agric. Sci. Chin. 2009, 37, 8331–8333. [Google Scholar]
- She, L.H.; He, F.; Xu, D.; Lin, J.D.; Wu, Z.B. Nitrogen removal under the condition of carbon source supplement in integrated vertical-flow constructed wetland. Environ. Sci. Chin. 2009, 30, 3300. [Google Scholar]
- Yan, N.; Jin, X.B.; Zang, J.Q. A Compasrison between the processes of denitrification with glucose and methanol as carbon sourse. J. Shanghai Teachers Univ. Chin. 2002, 3, 14–16. [Google Scholar]
- Caselles-Osorio, A.; García, J. Performance of experimental horizontal subsurface flow constructed wetlands fed with dissolved or particulate organic matter. Water Res. 2006, 40, 3603–3611. [Google Scholar] [CrossRef] [PubMed]
- Vymazal, J. Removal of nutrients in various types of constructed wetlands. Sci. Total Environ. 2007, 380, 48–65. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.C.; Cheng, Z.; Li, Y.; Tang, J. Effect of fermentation liquid from food waste as a carbon source for enhancing denitrification in wastewater treatment. Chemosphere 2015, 144, 689–696. [Google Scholar] [CrossRef]
- Kuba, T.; van Loosdrecht, M.C.M.; Heijnen, J.J. Phosphorus and nitrogen removal with minimal COD requirement by integration of denitrifying dephosphatation and nitrification in a two-sludge system. Water Res. 1996, 30, 1702–1710. [Google Scholar] [CrossRef]
- Peng, Y.; Ma, B. Review of biological nitrogen removal enhancement technologies and processes under low C/N ratio. Acta Scientiae Circumstantiae Chin. 2009, 29, 225–230. [Google Scholar]
- Fan, J.; Wang, W.; Bo, Z.; Guo, Y.; Ngo, H.H.; Guo, W.; Jian, Z.; Wu, H. Nitrogen removal in intermittently aerated vertical flow constructed wetlands: Impact of influent COD/N ratios. Bioresour. Technol. 2013, 143, 461–466. [Google Scholar] [CrossRef]
- Lee, N.M.; Welander, T. The effect of different carbon sources on respiratory denitrification in biological wastewater treatment. J. Ferment. Bioeng. 1996, 82, 277–285. [Google Scholar] [CrossRef]
- Mielcarek, A.; Rodziewicz, J.; Janczukowicz, W.; Thornton, A. The feasibility of citric acid as external carbon source for biological phosphorus removal in a sequencing batch biofilm reactor (SBBR). Biochem. Eng. J. 2015, 93, 102–107. [Google Scholar] [CrossRef]
- Wang, W.; Ding, Y.; Wang, Y.; Song, X.; Ambrose, R.F.; Ullman, J.L. Intensified nitrogen removal in immobilized nitrifier enhanced constructed wetlands with external carbon addition. Bioresour. Technol. 2016, 218, 1261–1265. [Google Scholar] [CrossRef]
- Wen, Y.; Chen, Y.; Zheng, N.; Yang, D.; Zhou, Q. Effects of plant biomass on nitrate removal and transformation of carbon sources in subsurface-flow constructed wetlands. Bioresour. Technol. 2010, 101, 7286–7292. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Nava, Y.; Marañón, E.; Soons, J.; Castrillón, L. Denitrification of high nitrate concentration wastewater using alternative carbon sources. J. Hazard. Mater. 2010, 173, 682–688. [Google Scholar] [CrossRef]
- Tian, Q.; Wang, Q.; Zhu, Y.; Li, F.; Zhuang, L.; Yang, B. Enhanced primary sludge sonication by heat insulation to reclaim carbon source for biological phosphorous removal. Ultrason. Sonochem. 2017, 34, 123–129. [Google Scholar] [CrossRef]
- Chowdhury, T.R.; Dick, R.P. Ecology of aerobic methanotrophs in controlling methane fluxes from wetlands. Appl. Soil Ecol. 2013, 65, 8–22. [Google Scholar] [CrossRef]
- Tanner, C.C.; Adams, D.D.; Downes, M.T. Methane Emissions from Constructed Wetlands Treating Agricultural Wastewaters. J. Environ. Qual. 1997, 26, 1056–1062. [Google Scholar] [CrossRef]
- Lal, R. Soil Carbon Sequestration Impacts on Global Climate Change and Food Security. Science 2004, 304, 1623. [Google Scholar] [CrossRef]
- Nugroho, S.G.; Lumbanraja, J.; Suprapto, H.S.; Haraguchi, H.; Kimura, M. Three-year measurement of methane emission from an Indonesian paddy field. Plant Soil 1996, 181, 287–293. [Google Scholar] [CrossRef]
- Mander, Ü.; Dotro, G.; Ebie, Y.; Towprayoon, S.; Chiemchaisri, C.; Nogueira, S.F.; Jamsranjav, B.; Kasak, K.; Truu, J.; Tournebize, J.; Mitsch, W.J. Greenhouse gas emission in constructed wetlands for wastewater treatment: A review. Ecol. Eng. 2014, 66, 19–35. [Google Scholar] [CrossRef]
- Mata-Alvarez, J.; Macé, S.; Llabrés, P. Anaerobic digestion of organic solid wastes. An overview of research achievements and perspectives. Bioresour. Technol. 2000, 74, 3–16. [Google Scholar] [CrossRef]
- Zhang, Q.; Sun, R.; Jiang, G.; Xu, Z.; Liu, S. Carbon and energy flux from a Phragmites australis wetland in Zhangye oasis-desert area, China. Agr. Forest Meteorol. 2016, 230, 45–57. [Google Scholar] [CrossRef]
- Ye, R.; Espe, M.B.; Linquist, B.; Parikh, S.J.; Doane, T.A.; Horwath, W.R. A soil carbon proxy to predict CH4 and N2O emissions from rewetted agricultural peatlands. Agric. Ecosyst. Environ. 2016, 220, 64–75. [Google Scholar] [CrossRef]
- Kuroda, K.; Chosei, T.; Nakahara, N.; Hatamoto, M.; Wakabayashi, T.; Kawai, T.; Araki, N.; Syutsubo, K.; Yamaguchi, T. High organic loading treatment for industrial molasses wastewater and microbial community shifts corresponding to system development. Bioresour. Technol. 2015, 196, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wu, S.; Chen, L.; Dong, R. How substrate influences nitrogen transformations in tidal flow constructed wetlands treating high ammonium wastewater? Ecol. Eng. 2014, 73, 478–486. [Google Scholar] [CrossRef]
- Song, X.; Qin, L.; Yan, D. Nutrient Removal by Hybrid Subsurface Flow Constructed Wetlands for High Concentration Ammonia Nitrogen Wastewater. Procedia Environ. Sci. 2010, 2, 1461–1468. [Google Scholar]
- Guisasola, A.; De, H.D.; Keller, J.; Yuan, Z. Methane formation in sewer systems. Water Res. 2008, 42, 1421–1430. [Google Scholar] [CrossRef]
- Jeong, T.Y.; Chung, H.K.; Yeom, S.H.; Choi, S.S. Analysis of methane production inhibition for treatment of sewage sludge containing sulfate using an anaerobic continuous degradation process. Korean J. Chem. Eng. 2009, 26, 1319–1322. [Google Scholar] [CrossRef]
- Gómez, M.A.; González-López, J.; Hontoria-Garc A, E. Influence of carbon source on nitrate removal of contaminated groundwater in a denitrifying submerged filter. J. Hazard. Mater. 2000, 80, 69–80. [Google Scholar] [CrossRef]
- Nyberg, U.; Andersson, B.; Aspegren, H. Long-term experiences with external carbon sources for nitrogen removal. Water Sci. Technol. 1996, 33, 109–116. [Google Scholar] [CrossRef]
- Michael, S. Economic Feasibility of Ethanol Production from Sugar Crops. Louisiana Agriculture. 2007. Available online: https://www.lsuagcenter.com/portals/communications/publications/agmag/archive/2007/winter/the-economic-feasibility-of-ethanol-production-from-sugar-crops (accessed on 14 November 2018).
- Philippsen, A.; Wild, P.; Rowe, A. Energy input, carbon intensity and cost for ethanol produced from farmed seaweed. Renew. Sustainable Energy Rev. 2014, 38, 609–623. [Google Scholar] [CrossRef]
- Magyar, M.; Da Costa Sousa, L.; Jayanthi, S.; Balan, V. Pie waste-A component of food waste and a renewable substrate for producing ethanol. Waste Manage. 2017, 62, 247–254. [Google Scholar] [CrossRef]
- Bao, Q.P.C.E. Electric field induced growth of well aligned carbon nanotubes from ethanol flames. Nanotechnology 2006, 17, 1016–1021. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Porder, S.; Houlton, B.Z.; Chadwick, O.A. Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen-phosphorus interactions. Ecol. Appl. 2010, 20, 5–15. [Google Scholar] [CrossRef]
- Luo, H.; Liu, X.; Zhang, K.; Li, M.; Huang, B.; Zhang, F.; Anderson, B.C.; Fan, L.; Chen, F.; Fu, S. Driven Mechanism and Emission Control on Methane Greenhouse Emission during Rural Sewage Non-point Source Pollution Treatment by Vertical-Flow Constructed Wetlands; Chinese Science Press: Beijing, China, 2015. (in Chinese) [Google Scholar]
- Yan, C.; Zhang, H.; Li, B.; Wang, D.; Zhao, Y.; Zheng, Z. Effects of influent C/N ratios on CO2 and CH4 emissions from vertical subsurface flow constructed wetlands treating synthetic municipal wastewater. J. Hazard. Mater. 2012, 203, 188–194. [Google Scholar] [CrossRef]
- Picek, T.; Čížková, H.; Dušek, J. Greenhouse gas emissions from a constructed wetland-Plants as important sources of carbon. Ecol. Eng. 2007, 31, 98–106. [Google Scholar] [CrossRef]
- IPCC. Working Group I: The Scientific Basis. In IPCC: Climate Change 2001 (TAR); Houghton, J.T., Ding, Y., Griggs, D.J., Noguer, M., van der Linden, P.J., Dai, X., Maskell, K., Johnson, C.A., Eds.; Cambridge University Press: Cambridge, UK, 2001. [Google Scholar]
- Zimov, S.A.; Voropaev, Y.V.; Semiletov, I.P.; Davidov, S.P.; Prosiannikov, S.F.; Chapin, F.S.; Chapin, M.C.; Trumbore, S.; Tyler, S. North Siberian lakes: A methane source fueled by Pleistocene carbon. Science 1997, 277, 800–802. [Google Scholar] [CrossRef]
- Chin, K.J.; Conrad, R. Intermediary metabolism in methanogenic paddy soil and the influence of temperature. Fems Microbiol. Ecol. 1995, 18, 85–102. [Google Scholar] [CrossRef]
- Corbella, C.; Puigagut, J. Effect of primary treatment and organic loading on methane emissions from horizontal subsurface flow constructed wetlands treating urban wastewater. Ecol. Eng. 2014, 80, 79–84. [Google Scholar] [CrossRef]
- Elsgaard, L.; Olsen, A.B.; Petersen, S.O. Temperature response of methane production in liquid manures and co-digestates. Sci. Total Environ. 2016, 539, 78–84. [Google Scholar] [CrossRef]
- Evgheni, E.; Sa, J.; Cecilia, S.; Sven, S.R.; Mikael, P.; H Kan, J.N. Nitrous oxide and methane emissions from food waste composting at different temperatures. Waste Manage. 2015, 46, 113–119. [Google Scholar]
- Khalil, M.A.K.; Rasmussen, R.A.; Wang, M.X.; Ren, L. Methane emissions from rice fields in China. Environ. Sci. Technol. 2002, 25, 979–981. [Google Scholar] [CrossRef]
- Yagi, K.; Minami, K. Effect of organic matter application on methane emission from some Japanese paddy fields. Soil Sci. Plant Nutr. 1990, 36, 599–610. [Google Scholar] [CrossRef]
- Johnson-Beebout, S.E.; Angeles, O.R.; Alberto, M.C.R.; Buresh, R.J. Simultaneous minimization of nitrous oxide and methane emission from rice paddy soils is improbable due to redox potential changes with depth in a greenhouse experiment without plants. Geoderma 2009, 149, 45–53. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, H.; Ye, C.; Chen, X.; Xie, B.; Huang, C.; Zhang, J.; Xu, M. Effects of plant species on soil microbial processes and CH4 emission from constructed wetlands. Environ. Pollut. 2013, 174, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A.E.; Gustavsson, A.M.; Öquist, M.G.; Svensson, B.H. Methane emissions from a constructed wetland treating wastewater--seasonal and spatial distribution and dependence on edaphic factors. Water Res. 2004, 38, 3960–3970. [Google Scholar] [CrossRef] [PubMed]
- Uggetti, E.; García, J.; Lind, S.E.; Martikainen, P.J.; Ferrer, I. Quantification of greenhouse gas emissions from sludge treatment wetlands. Water Res. 2012, 46, 1755–1762. [Google Scholar] [CrossRef]
- Nouchi, I.; Hosono, T.; Aoki, K.; Minami, K. Seasonal variation in methane flux from rice paddies associated with methane concentration in soil water, rice biomass and temperature, and its modelling. Plant Soil 1994, 161, 195–208. [Google Scholar] [CrossRef]
- Zhu, N.; An, P.; Krishnakumar, B.; Zhao, L.; Sun, L.; Mizuochi, M.; Inamori, Y. Effect of plant harvest on methane emission from two constructed wetlands designed for the treatment of wastewater. J. Environ. Manage. 2007, 85, 936–943. [Google Scholar] [CrossRef]
- Meng, W.; Zhanlei, W.U.; Wang, Z. Control factors and critical conditions between carbon sinking and sourcing of wetland ecosystem. Ecol. Environ. Sci. 2011, 20, 1359–1366. [Google Scholar]
- He, Y.; Tao, W.; Wang, Z.; Shayya, W. Effects of pH and seasonal temperature variation on simultaneous partial nitrification and anammox in free-water surface wetlands. J. Environ. Manage. 2012, 110, 103–109. [Google Scholar] [CrossRef]
- Plüg, B.D.; Cibati, A.; Trois, C. The use of organic wastes at different degrees of maturity as carbon sources for denitrification of landfill leachate. Waste Manage. 2015, 46, 373–379. [Google Scholar] [CrossRef]
- Tsui, L.; Krapac, I.G.; Roy, W.R. The feasibility of applying immature yard-waste compost to remove nitrate from agricultural drainage effluents: A preliminary assessment. J. Hazard. Mater. 2007, 144, 585–589. [Google Scholar] [CrossRef]
- Tang, Y.; Shigematsu, T.; Morimura, S.; Kida, K. Dynamics of the microbial community during continuous methane fermentation in continuously stirred tank reactors. J. Biosci. Bioeng. 2015, 119, 375–383. [Google Scholar] [CrossRef]
- Wang, Z.P.; Delaune, R.D.; Patrick, W.H.; Masscheleyn, P.H. Soil Redox and pH Effects on Methane Production in a Flooded Rice Soil. Soil Sci. Soc. Am. J. 1993, 57, 382–385. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, A.; Ren, N. Effect of carbon sources on sulfidogenic bacterial communities during the starting-up of acidogenic sulfate-reducing bioreactors. Bioresource Technol. 2010, 101, 2952–2959. [Google Scholar] [CrossRef]
- Bais, H.P.; Weir, T.L.; Perry, L.G.; Gilroy, S.; Vivanco, J.M. The Role of Root Exudates in Rhizosphere Interactions with Plants and Other Organisms. Annu. Rev. Plant Biol. 2006, 57, 233–266. [Google Scholar] [CrossRef]
- Ström, L.; Mastepanov, M.; Christensen, T. Species-specific Effects of Vascular Plants on Carbon Turnover and Methane Emissions from Wetlands. Biogeochemistry 2005, 75, 65–82. [Google Scholar] [CrossRef]
- Bodelier, P.L.; Roslev, P.; Henckel, T.; Frenzel, P. Stimulation by ammonium-based fertilizers of methane oxidation in soil around rice roots. Nature 2000, 403, 421–424. [Google Scholar] [CrossRef]
- Siljanen, H.M.; Saari, A.; Bodrossy, L.; Martikainen, P.J. Effects of Nitrogen Load on the Function and Diversity of Methanotrophs in the Littoral Wetland of a Boreal Lake. Front Microbiol. 2012, 3, 39. [Google Scholar] [CrossRef]
- Nurliyana, M.Y.; H’Ng, P.S.; Rasmina, H.; Kalsom, M.S.U.; Chin, K.L.; Lee, S.H.; Lum, W.C.; Khoo, G.D. Effect of C/N ratio in methane productivity and biodegradability during facultative co-digestion of palm oil mill effluent and empty fruit bunch. Ind. Crop. Prod. 2015, 76, 409–415. [Google Scholar] [CrossRef]
- Wang, X.; Yang, G.; Feng, Y.; Ren, G.; Han, X. Optimizing feeding composition and carbon—nitrogen ratios for improved methane yield during anaerobic co-digestion of dairy, chicken manure and wheat straw. Bioresource Technol. 2012, 120, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Grønlund, A.; Sveistrup, T.E.; Søvik, A.K.; Rasse, D.P.; Kløve, B. Degradation of cultivated peat soils in northern norway based on field scale CO2, N2O and CH4 emission measurements. Arch. Agron. Soil Sci. 2006, 52, 149–159. [Google Scholar] [CrossRef]
- Rodríguez-Murillo, J.C.; Almendros, G.; Knicker, H. Wetland soil organic matter composition in a Mediterranean semiarid wetland (Las Tablas de Daimiel, Central Spain): Insight into different carbon sequestration pathways. Org. Geochem. 2011, 42, 762–773. [Google Scholar] [CrossRef]
- UNESCO. The United Nations World Water Development Report 2017, Wastewater: The Untapped Resource; United Nations World Water Assessment Programme; United Nation Educational, Scientific and Cultural Organization: Paris, France, 2017. [Google Scholar]
- Vanderzaag, A.C.; Gordon, R.J.; Burton, D.L.; Jamieson, R.C.; Stratton, G.W. Greenhouse gas emissions from surface flow and subsurface flow constructed wetlands treating dairy wastewater. J. Environ. Qual. 2010, 39, 460–471. [Google Scholar] [CrossRef]
- Mander, U.; Teiter, S.; Augustin, J. Emission of greenhouse gases from constructed wetlands for wastewater treatment and from riparian buffer zones. Water Sci. Technol. 2005, 52, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, K.; Fan, L.; Luo, H.; Jiang, M.; Anderson, B.C.; Li, M.; Huang, B.; Yu, L.; He, G.; Wang, J.; Pu, A. Intermittent micro-aeration control of methane emissions from an integrated vertical-flow constructed wetland during agricultural domestic wastewater treatment. Environ. Sci. Pollut. R. 2018, 25, 24426–24444. [Google Scholar] [CrossRef] [PubMed]
- Pan, T.; Zhu, X.; Ye, Y. Estimate of life-cycle greenhouse gas emissions from a vertical subsurface flow constructed wetland and conventional wastewater treatment plants: A case study in China. Ecol. Eng. 2011, 37, 248–254. [Google Scholar] [CrossRef]
- Fuchs, V.J.; Mihelcic, J.R.; Gierke, J.S. Life cycle assessment of vertical and horizontal flow constructed wetlands for wastewater treatment considering nitrogen and carbon greenhouse gas emissions. Water Res. 2011, 45, 2073–2081. [Google Scholar] [CrossRef] [PubMed]
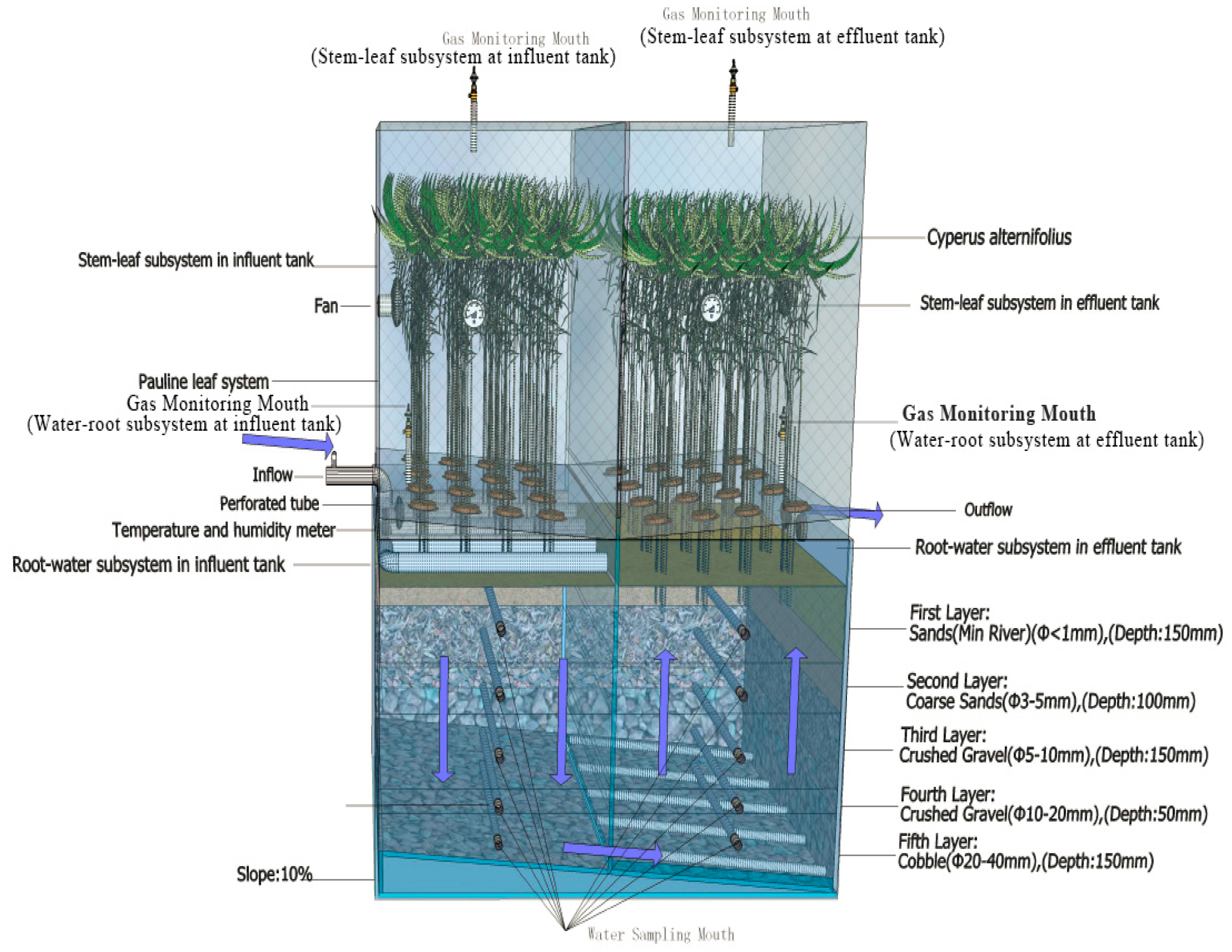
| IVCW | Group B (Influent Tank) | Group D (Effluent Tank) | The Distance from the Sampling Spots to Bottom of the Tank | Position |
|---|---|---|---|---|
| Plane I | I-B3 | I-D3 | 525 mm | The middle position of first layer |
| Plane II | II-B3 | II-D3 | 400 mm | The middle position of second layer |
| Plane III | III-B3 | III-D3 | 300 mm | The middle position of third layer |
| Plane IV | IV-B3 | IV-D3 | 200 mm | The middle position of fourth layer |
| Plane V | V-B3 | V-D3 | 75 mm | The middle position of fifth layer |
| Methane Fluxes (g CH4 m−2 day−1) | Ethanol Concentrations | |||||
|---|---|---|---|---|---|---|
| 0 mmol/L (Blank Experiment) | 2 mmol/L | 4 mmol/L | 8 mmol/L | 16 mmol/L | 32 mmol/L | |
| Total methane flux in IVCW | 101.5 ± 0.07 | 146.49 ± 0.08 | 146.19 ± 0.08 | 133.44 ± 0.09 | 106.88 ± 0.04 | 69.19 ± 0.05 |
| Stem-leaf subsystem in influent tank | 25.07 ± 0.05 | 36.94 ± 0.07 | 31.47 ± 0.06 | 36.83 ± 0.08 | 26.32 ± 0.04 | 14.69 ± 0.04 |
| Root-water subsystem in influent tank | 22.96 ± 0.09 | 36.14 ± 0.08 | 39.55 ± 0.09 | 30.91 ± 0.06 | 25.84 ± 0.04 | 17.81 ± 0.05 |
| Stem-leaf subsystem in effluent tank | 26.70 ± 0.08 | 45.42 ± 0.10 | 35.74 ± 0.07 | 29.02 ± 0.06 | 25.78 ± 0.04 | 14.976 ± 0.06 |
| Root-water subsystem in effluent tank | 28.72 ± 0.09 | 27.95 ± 0.05 | 39.44 ± 0.08 | 36.66 ± 0.08 | 28.95 ± 0.06 | 20.72 ± 0.06 |
| Added methane flux of control experiment, compared with blank experiment | - | 43.04 ± 0.25 (42%) | 42.74 ± 0.31 (42%) | 29.98 ± 0.37 (29%) | 3.42 ± 0.24 (33%) | −35.26 ± 0.27 (−35%) |
| Parameters | Ethanol Concentrations | |||||
|---|---|---|---|---|---|---|
| 0 mmol/L (Blank Experiment) | 2 mmol/L | 4 mmol/L | 8 mmol/L | 16 mmol/L | 32 mmol/L | |
| Inflow of TC (mg/L) | 0.6 | 0.7 | 1.3 | 1.9 | 3.8 | 6.8 |
| Outflow of TC (mg/L) | 0.6 | 0.3 | 9.3 | 5.8 | 2.6 | 8.0 |
| Inflow of TN (mg/L) | 4.2 | 2.5 | 2.4 | 2.0 | 2.2 | 3.5 |
| Outflow of TN (mg/L) | 1.6 | 1.8 | 2.5 | 1.7 | 1.7 | 1.0 |
| Inflow of C/N ratios (%) | 15.6 | 28.8 | 56.1 | 97.8 | 177.9 | 193.8 |
| Outflow of C/N ratios (%) | 36.9 | 192.1 | 374.1 | 339.2 | 152.2 | 801.1 |
| Ethanol Concentrations | Average CH4-C/TOC (%) | Average IC (g) | Average TC (g) | Average CH4-C/IC (%) | Average CH4-C/TC (%) |
|---|---|---|---|---|---|
| 0 mmol/L | 65 | 1.18 | 1.76 | 22 | 12 |
| 2 mmol/L | 66 | 1.33 | 2.02 | 44 | 26 |
| 4 mmol/L | 43 | 1.65 | 2.77 | 40 | 20 |
| 8 mmol/L | 24 | 2.5 | 4.94 | 32 | 9 |
| 16 mmol/L | 11 | 4.56 | 9.5 | 19 | 4 |
| 32 mmol/L | 3 | 9.68 | 19.91 | 16 | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Wang, J.; Fu, X.; Luo, H.; Anderson, B.C.; Zhang, K.; Li, M.; Huang, B.; Fan, L.; Yu, L.; et al. Methane Emissions Driven by Adding a Gradient of Ethanol as Carbon Source in Integrated Vertical-Flow Constructed Wetlands. Water 2019, 11, 1086. https://doi.org/10.3390/w11051086
Liu X, Wang J, Fu X, Luo H, Anderson BC, Zhang K, Li M, Huang B, Fan L, Yu L, et al. Methane Emissions Driven by Adding a Gradient of Ethanol as Carbon Source in Integrated Vertical-Flow Constructed Wetlands. Water. 2019; 11(5):1086. https://doi.org/10.3390/w11051086
Chicago/Turabian StyleLiu, Xiaoling, Jingting Wang, Xiaoying Fu, Hongbing Luo, Bruce C. Anderson, Ke Zhang, Mei Li, Bo Huang, Liangqian Fan, Lijuan Yu, and et al. 2019. "Methane Emissions Driven by Adding a Gradient of Ethanol as Carbon Source in Integrated Vertical-Flow Constructed Wetlands" Water 11, no. 5: 1086. https://doi.org/10.3390/w11051086
APA StyleLiu, X., Wang, J., Fu, X., Luo, H., Anderson, B. C., Zhang, K., Li, M., Huang, B., Fan, L., Yu, L., He, G., Zou, L., Fu, S., Hu, L., & Jiang, M. (2019). Methane Emissions Driven by Adding a Gradient of Ethanol as Carbon Source in Integrated Vertical-Flow Constructed Wetlands. Water, 11(5), 1086. https://doi.org/10.3390/w11051086





