Variations in Illicit Compound Discharged from Treated Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Program
2.2. Analytical Techniques
2.3. Calculation of Total Discharge and Mass Amounts
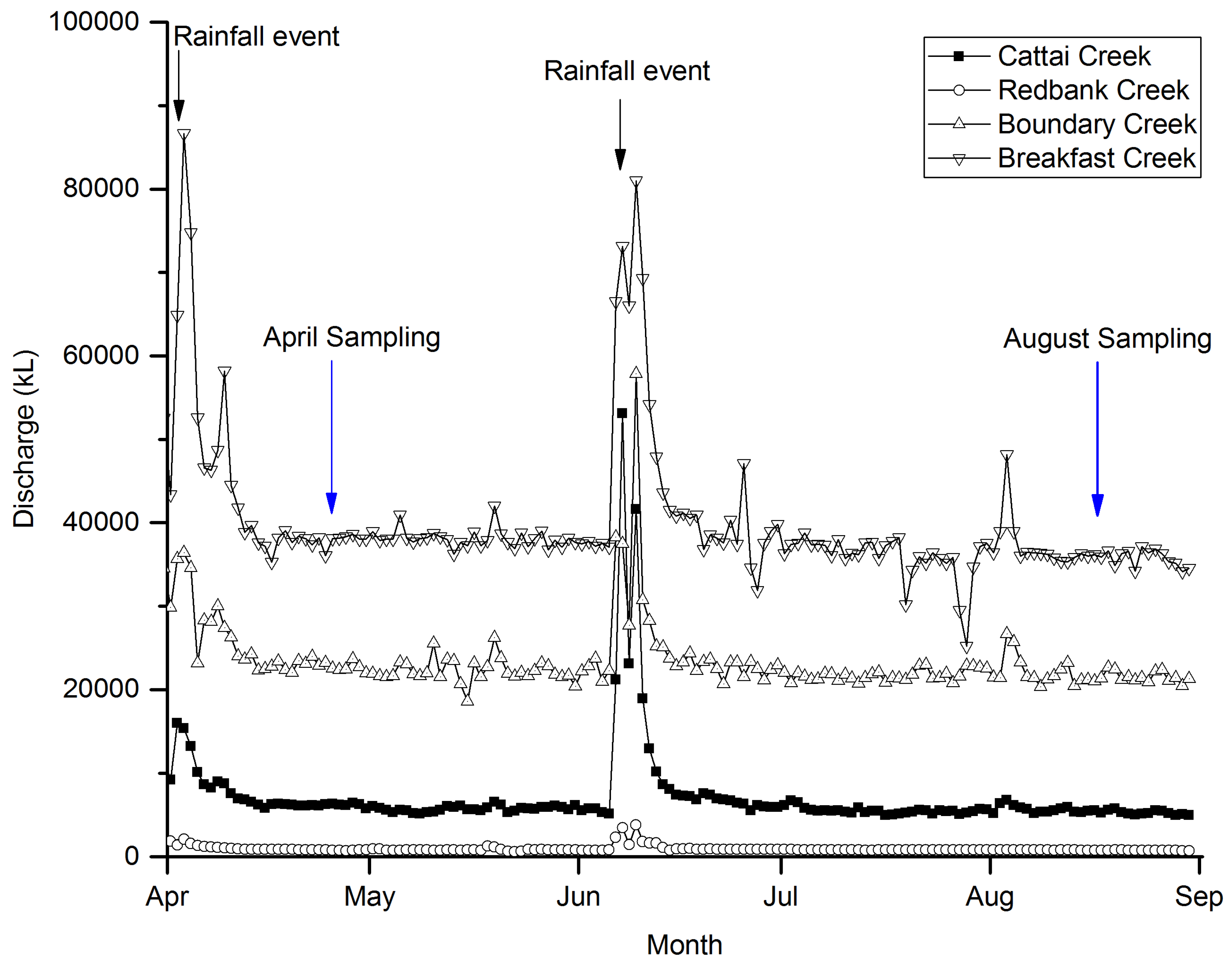
3. Results
3.1. Detection of Illicit Compounds
3.2. Discharge Quantities
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Compound | % Recovery ± SD at 10 ng/L | LOD (ng/L) | LOQ (ng/L) |
|---|---|---|---|
| Ephedrine | 93 ± 3.1 | 0.10 | 0.31 |
| Pseudoephedrine | 103 ± 2.8 | 0.08 | 0.25 |
| Amphetamine | 95 ± 3.4 | 0.10 | 0.33 |
| MDA | 101 ± 5.6 | 0.35 | 1.01 |
| Methamphetamine | 107 ± 3.2 | 0.18 | 0.8 |
| MDMA | 101 ± 2.4 | 0.10 | 0.33 |
| Cocaine | 100 ± 3.8 | 0.02 | 0.07 |
| Benzoylecognine | 101 ± 2.7 | 0.06 | 0.18 |
References
- Rosi-Marshall, E.; Snow, D.; Bartelt-Hunt, S.; Paspalof, A.; Tank, J. A review of ecological effects and environmental fate of illicit drugs in aquatic ecosystems. J. Hazard. Mater. 2015, 282, 18–25. [Google Scholar] [CrossRef]
- Kasprzyk-Hordern, B.; Dinsdale, R.M.; Guwy, A.J. The occurrence of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs in surface water in South Wales, UK. Water Res. 2008, 42, 3498–3518. [Google Scholar] [CrossRef]
- Ort, C.; Lawrence, M.G.; Rieckermann, J.; Joss, A. Sampling for pharmaceuticals and personal care products (PPCPs) and illicit drugs in wastewater systems: Are your conclusions valid? A critical review. Environ. Sci. Technol. 2010, 44, 6024–6035. [Google Scholar] [CrossRef]
- Metcalfe, C.; Tindale, K.; Li, H.; Rodayan, A.; Yargeau, V. Illicit drugs in Canadian municipal wastewater and estimates of community drug use. Environ. Pollut. 2010, 158, 3179–3185. [Google Scholar] [CrossRef]
- van Nuijs, A.L.; Castiglioni, S.; Tarcomnicu, I.; Postigo, C.; Lopez de Alda, M.; Neels, H.; Zuccato, E.; Barcelo, D.; Covaci, A. Illicit drug consumption estimations derived from wastewater analysis: A critical review. Sci. Total Environ. 2011, 409, 3564–3577. [Google Scholar] [CrossRef]
- Yadav, M.K.; Short, M.D.; Aryal, R.; Gerber, C.; van den Akker, B.; Saint, C.P. Occurrence of illicit drugs in water and wastewater and their removal during wastewater treatment. Water Res. 2017, 124, 713–727. [Google Scholar] [CrossRef]
- Zuccato, E.; Castiglioni, S.; Bagnati, R.; Chiabrando, C.; Grassi, P.; Fanelli, R. Illicit drugs, a novel group of environmental contaminants. Water Res. 2008, 42, 961–968. [Google Scholar] [CrossRef]
- Baker, D.R.; Kasprzyk-Hordern, B. Spatial and temporal occurrence of pharmaceuticals and illicit drugs in the aqueous environment and during wastewater treatment: New developments. Sci. Total Environ. 2013, 454–455, 442–456. [Google Scholar] [CrossRef]
- Scott, P.D.; Bartkow, M.; Blockwell, S.J.; Coleman, H.M.; Khan, S.J.; Lim, R.; McDonald, J.A.; Nice, H.; Nugegoda, D.; Pettigrove, V.; et al. A national survey of trace organic contaminants in Australian rivers. J. Environ. Qual. 2014, 43, 1702–1712. [Google Scholar] [CrossRef]
- Pal, R.; Megharaj, M.; Kirkbride, K.P.; Naidu, R. Illicit drugs and the environment—A review. Sci. Total Environ. 2013, 463–464, 1079–1092. [Google Scholar] [CrossRef]
- Australian Criminal Intelligence Commission. National Wastewater Drug Monitoring Program Report No. 3; Australian Criminal Intelligence Commission, 2017. Available online: https://acic.govcms.gov.au/ (accessed on 21 May 2019).
- Australian Criminal Intelligence Commission. National Wastewater Drug Monitoring Program Report No. 5; Australian Criminal Intelligence Commission, 2018. Available online: https://acic.govcms.gov.au/ (accessed on 21 May 2019).
- Wilkins, C.; Lai, F.Y.; O’Brien, J.; Thai, P.; Mueller, J. Comparing methamphetamine, MDMA, cocaine, codeine and methadone use between the Auckland region and four Australian states using wastewater-based epidemiology (WBE). N. Z. Med. J. 2018, 131, 12–20. [Google Scholar]
- Bones, J.; Thomas, K.V.; Paull, B. Using environmental analytical data to estimate levels of community consumption of illicit drugs and abused pharmaceuticals. J. Environ. Monit. 2007, 9, 701–707. [Google Scholar] [CrossRef]
- Castiglioni, S.; Zuccato, E.; Crisci, E.; Chiabrando, C.; Fanelli, R.; Bagnati, R. Identification and measurement of illicit drugs and their metabolites in urban wastewater by liquid chromatography-tandem mass spectrometry. Anal. Chem. 2006, 78, 8421–8429. [Google Scholar] [CrossRef]
- Kasprzyk-Hordern, B.; Dinsdale, R.M.; Guwy, A.J. The removal of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs during wastewater treatment and its impact on the quality of receiving waters. Water Res. 2009, 43, 363–380. [Google Scholar] [CrossRef]
- Jones-Lepp, T.L.; Alvarez, D.A.; Petty, J.D.; Huckins, J.N. Polar organic chemical integrative sampling and liquid chromatography-electrospray/ion-trap mass spectrometry for assessing selected prescription and illicit drugs in treated sewage effluents. Arch. Environ. Contam. Toxicol. 2004, 47, 427–439. [Google Scholar] [CrossRef]
- Loganathan, B.; Phillips, M.; Mowery, H.; Jones-Lepp, T.L. Contamination profiles and mass loadings of macrolide antibiotics and illicit drugs from a small urban wastewater treatment plant. Chemosphere 2009, 75, 70–77. [Google Scholar] [CrossRef]
- Rodayan, A.; Afana, S.; Segura, P.A.; Sultana, T.; Metcalfe, C.D.; Yargeau, V. Linking drugs of abuse in wastewater to contamination of surface and drinking water. Environ. Toxicol. Chem. 2016, 35, 843–849. [Google Scholar] [CrossRef]
- dos Santos Barbosa Ortega, A.; Maranho, L.A.; Nobre, C.R.; Moreno, B.B.; Guimarães, R.S.; Lebre, D.T.; de Souza Abessa, D.M.; Ribeiro, D.A.; Pereira, C.D.S. Detoxification, oxidative stress, and cytogenotoxicity of crack cocaine in the brown mussel Perna perna. Environ. Sci. Pollut. Res. 2018. [Google Scholar] [CrossRef]
- Gay, F.; Maddaloni, M.; Valiante, S.; Laforgia, V.; Capaldo, A. Endocrine Disruption in the European Eel, Anguilla anguilla, Exposed to an Environmental Cocaine Concentration. Water Air Soil Pollut. 2013, 224, 1579. [Google Scholar] [CrossRef]
- Gay, F.; Ferrandino, I.; Monaco, A.; Cerulo, M.; Capasso, G.; Capaldo, A. Histological and hormonal changes in the European eel (Anguilla anguilla) after exposure to environmental cocaine concentration. J. Fish Dis. 2016, 39, 295–308. [Google Scholar] [CrossRef]
- Capaldo, A.; Gay, F.; Lepretti, M.; Paolella, G.; Martucciello, S.; Lionetti, L.; Caputo, I.; Laforgia, V. Effects of environmental cocaine concentrations on the skeletal muscle of the European eel (Anguilla anguilla). Sci. Total Environ. 2018, 640–641, 862–873. [Google Scholar] [CrossRef]
- Boles, T.; Wells, M. Analysis of amphetamine and methamphetamine as emerging pollutants in wastewater and wastewater-impacted streams. J. Chromatogr. A 2010, 1217, 2561–2568. [Google Scholar] [CrossRef]
- Sydney Wastewater Treatment Plants. Available online: http://www.sydneywater.com.au/SW/water-the-environment/how-we-manage-sydney-s-water/wastewater-network/wastewater-treatment-plants/index.htm (accessed on 22 May 2018).
- Zuccato, E.; Chiabrando, C.; Castiglioni, S.; Calamari, D.; Bagnati, R.; Schiarea, S.; Fanelli, R. Cocaine in surface waters: A new evidence-based tool to monitor community drug abuse. Environ. Health 2005, 4, 14. [Google Scholar] [CrossRef]
- Thomas, K.V.; Bijlsma, L.; Castiglioni, S.; Covaci, A.; Emke, E.; Grabic, R.; Hernández, F.; Karolak, S.; Kasprzyk-Hordern, B.; Lindberg, R.H.; et al. Comparing illicit drug use in 19 European cities through sewage analysis. Sci. Total Environ. 2012, 432, 432–439. [Google Scholar] [CrossRef]
- Huerta-Fontela, M.; Galceran, M.T.; Ventura, F. Stimulatory Drugs of Abuse in Surface Waters and Their Removal in a Conventional Drinking Water Treatment Plant. Environ. Sci. Technol. 2008, 42, 6809–6816. [Google Scholar] [CrossRef]
- van Nuijs, A.L.; Pecceu, B.; Theunis, L.; Dubois, N.; Charlier, C.; Jorens, P.G.; Bervoets, L.; Blust, R.; Neels, H.; Covaci, A. Cocaine and metabolites in waste and surface water across Belgium. Environ. Pollut. 2009, 157, 123–129. [Google Scholar] [CrossRef]
- Bueno, M.M.; Uclés, S.; Hernando, M.; Fernández-Alba, A. Development of a solvent-free method for the simultaneous identification/quantification of drugs of abuse and their metabolites in environmental water by LC-MS/MS. Talanta 2011, 85, 157–166. [Google Scholar] [CrossRef]
- González-Mariño, I.; Quintana, J.; Rodríguez, I.; Cela, R. Determination of drugs of abuse in water by solid-phase extraction, derivatisation and gas chromatography-ion trap-tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 1748–1760. [Google Scholar] [CrossRef]
- Huerta-Fontela, M.; Galceran, M.; Martin-Alonso, J.; Ventura, F. Occurrence of psychoactive stimulatory drugs in wastewaters in north-eastern Spain. Sci. Total Environ. 2008, 397, 31–40. [Google Scholar] [CrossRef]
- Postigo, C.; de Alda, M.J.; Barceló, D. Fully automated determination in the low nanogram per liter level of different classes of drugs of abuse in sewage water by on-line solid-phase extraction-liquid chromatography-electrospray-tandem mass spectrometry. Anal. Chem. 2008, 80, 3123–3134. [Google Scholar] [CrossRef]
- Vazquez-Roig, P.; Andreu, V.; Blasco, C.; Picó, Y. SPE and LC-MS/MS determination of 14 illicit drugs in surface waters from the Natural Park of L’Albufera (València, Spain). Anal. Bioanal. Chem. 2010, 397, 2851–2864. [Google Scholar] [CrossRef]
- European Monitoring Centre for Drugs and Drug Addiction. Assessing Illicit Drugs in Wastewater: Advances in Wastewater-Based Drug Epidemiology; Insights 22; Publications Office of the European Union: Luxembourg, Luxembourg, 2016. [Google Scholar] [CrossRef]

| Redbank Creek | Boundary Creek | Breakfast Creek | Cattai Creek | |||||
|---|---|---|---|---|---|---|---|---|
| April | August | April | August | April | August | April | August | |
| Ephedrine | 1.78 ± 0.11 | 2.21 ± 0.27 | 0.43 ± 0.04 | 1.16 ± 0.15 | 0.43 ± 0.01 | bdl | 1.72 ± 0.06 | 1.93 ± 0.28 |
| Pseudoephedrine | 11.33 ± 0.07 | 13.82 ± 1.80 | 2.96 ± 0.19 | 6.56 ± 1.71 | 7.46 ± 0.18 | 10.50 ± 0.01 | 7.88 ± 0.21 | 18.63 ± 2.53 |
| Amphetamine | 0.05 ± 0.01 | 0.17 ± 0.01 | 0.03 ± 0.00 | 0.56 ± 0.43 | 0.04 ± 0.00 | 0.08 ± 0.01 | 0.10 ± 0.01 | 0.24 ± 0.07 |
| MDA | 0.21 ± 0.01 | 1.51 ± 0.16 | 0.06 ± 0.00 | 0.60 ± 0.39 | bdl | 0.03 ± 0.00 | 0.97 ± 0.00 | 0.58 ± 0.07 |
| Methamphetamine | 15.46 ± 0.23 | 25.25 ± 2.83 | 3.09 ± 0.03 | 7.55 ± 2.27 | 4.40 ± 0.15 | 10.27 ± 0.22 | 3.89 ± 0.10 | 7.84 ± 1.02 |
| MDMA | 1.13 ± 0.01 | 1.51 ± 0.16 | 0.43 ± 0.00 | 0.60 ± 0.39 | 0.22 ± 0.00 | 0.03 ± 0.00 | 1.99 ± 0.05 | 0.58 ± 0.07 |
| Cocaine | 0.63 ± 0.00 | 2.99 ± 0.51 | 0.07 ± 0.00 | 0.68 ± 0.40 | 0.13 ± 0.01 | 0.75 ± 0.02 | 0.75 ± 0.03 | 0.91 ± 0.10 |
| Benzoylecgonine | 4.28 ± 0.02 | 21.57 ± 1.96 | 1.11 ± 0.02 | 3.29 ± 0.65 | 1.61 ± 0.05 | 5.94 ± 0.10 | 18.87 ± 0.40 | 17.58 ± 1.92 |
| COC:BEN | 0.15 | 0.14 | 0.06 | 0.21 | 0.08 | 0.13 | 0.04 | 0.05 |
| Redbank Creek | Boundary Creek | Breakfast Creek | Cattai Creek | |||||
|---|---|---|---|---|---|---|---|---|
| April | August | April | August | April | August | April | August | |
| Ephedrine | 1.95 | 1.77 | 11.44 | 27.12 | 19.78 | 0 | 13.93 | 11.19 |
| Pseudoephedrine | 12.46 | 11.06 | 78.74 | 153.4 | 343.2 | 409.5 | 63.82 | 108.1 |
| Amphetamine | 0.06 | 0.14 | 0.80 | 13.09 | 1.84 | 3.12 | 0.81 | 1.39 |
| MDA | 0.23 | 1.21 | 1.59 | 14.03 | 0 | 1.17 | 7.86 | 3.36 |
| Methamphetamine | 17.00 | 20.20 | 82.19 | 176.52 | 202.4 | 400.5 | 31.51 | 45.47 |
| MDMA | 1.24 | 1.21 | 11.44 | 14.03 | 10.12 | 1.17 | 16.12 | 3.36 |
| Cocaine | 0.69 | 2.39 | 1.86 | 15.89 | 5.98 | 29.25 | 6.08 | 5.28 |
| Benzoylecgonine | 4.71 | 17.26 | 29.53 | 76.92 | 74.06 | 231.6 | 71.85 | 101.96 |
| Total | 38.36 | 55.22 | 217.6 | 490.9 | 657.3 | 1076 | 211.9 | 280.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paciuszkiewicz, K.; Ryan, M.; Wright, I.A.; Reynolds, J.K. Variations in Illicit Compound Discharged from Treated Wastewater. Water 2019, 11, 1071. https://doi.org/10.3390/w11051071
Paciuszkiewicz K, Ryan M, Wright IA, Reynolds JK. Variations in Illicit Compound Discharged from Treated Wastewater. Water. 2019; 11(5):1071. https://doi.org/10.3390/w11051071
Chicago/Turabian StylePaciuszkiewicz, Kasjan, Michelle Ryan, Ian A. Wright, and Jason K. Reynolds. 2019. "Variations in Illicit Compound Discharged from Treated Wastewater" Water 11, no. 5: 1071. https://doi.org/10.3390/w11051071
APA StylePaciuszkiewicz, K., Ryan, M., Wright, I. A., & Reynolds, J. K. (2019). Variations in Illicit Compound Discharged from Treated Wastewater. Water, 11(5), 1071. https://doi.org/10.3390/w11051071







