Abstract
The sustainable management of fisheries resources requires extensive knowledge of their reproductive biology, which is scarcely the case for marine invertebrates. Sea urchins are among the most intensively harvested invertebrates, since their gonads, or “roe”, constitute a highly appreciated gastronomic delicacy, causing a severe decline in natural populations worldwide. In the Mediterranean, the typical commercial echinoid species is Paracentrotus lividus; its biology, however, has not been adequately studied in the Aegean Sea. Within this context, the present study examined the reproductive biology of the edible sea urchin, P. lividus, in the Aegean Sea (Pagasitikos Gulf) over a two-year period. Adult specimens were randomly collected by SCUBA diving (3–5 m) at monthly intervals to determine the gonad-somatic index, fecundity, and gametogenesis through the morphological and histological examination of the gonads. An annual reproductive cycle was defined in both years with a clear spawning peak in early spring, conforming to previous reports from other Mediterranean populations. A discrete secondary spawning period was also detected in early winter, as the species undergoes a second gametogenic event. These results are indicative for the increased reproductive potential of P. lividus in its south distributional range, suggesting a relevant revision of the official fishing season of the species.
1. Introduction
The common sea urchin, Paracentrotus lividus (Lamarck, 1816), is a gregarious species, widely distributed on the sublittoral rocky shores and seagrass meadows of the Mediterranean and the eastern Atlantic coasts. It is commercially harvested for its gonads, or “roe”, which constitute a highly appreciated gastronomic delicacy of great economic profit [1]. As a result, a severe decline of P. lividus populations has been reported in many Mediterranean areas [2,3,4,5], leading to a gradual adoption of protective measures in some EU countries. In Greece, apart from some temporal restrictions on harvesting that were mostly addressed to amateurs, no official regulations had been established to promote the long-term sustainable exploitation of this resource, until very recently. Now, a 6-month fishing period (January, February, March, July, August, and December), a daily quota of 600 specimens/boat, and a minimum legal size of 5 cm test diameter have been established under the Presidential Degree 65/2014. The above regulations, however, have not been documented on relevant scientific data, as basic knowledge on the biology—reproductive biology, in particular—of P. lividus from the Greek territory, or even the SE Mediterranean, is missing.
Attempting to regulate sea urchin fisheries is challenging, due to the species being consumed when they are in a reproductive state [6]. Therefore, obtaining data on their reproductive biology is a prerequisite to establishing efficient management strategies [7,8].
Although the biology of P. lividus has been extensively studied almost throughout its distributional range [9] (and references therein), very little information is available for the eastern Mediterranean Sea. The existing information is limited to the detrimental effect of the increasing seawater temperature to the species population in the Levantine Sea [10], to its population biology [1] in the south Aegean Sea, and its genetic structure [11].
Moreover, as is the case for many invertebrate species, specific data on the reproductive biology of P. lividus are limited to a few geographical areas of the Mediterranean Sea, such as Spain [8,9,12,13], Italy [14], and Tunis [15], and of the European Atlantic coast [16]. Despite the above efforts, several issues on its reproductive processes remain unclear and the reported information is often contradictory [6,14]. This fact, in combination with the reported increase in the reproductive variability of P. lividus at various spatial scales, especially considering the Mediterranean populations [6], stresses the need to study its reproductive biology, covering areas, such as the Aegean Sea, where no such data exists.
Within this context, the present work aimed to study the reproductive biology of P. lividus in the central part of the Aegean Sea, as a representative area, for two successive years, to determine the annual cycle of gonadal growth and oogenesis, through the histological examination of the gonads as being the most reliable method for determining the reproductive cycle of the species [6].
2. Materials and Methods
2.1. Study Area
The study was conducted in the Pagasitikos Gulf, a semi-enclosed, shallow water (mean depth 69 m) basin [17] connected with the Aegean Sea through the narrow but deep Trikeri channel (Figure 1), which safeguards water renewal within the Gulf [18]. The water masses are cold and fairly mixed in winter (12.5 °C) and highly stratified in the warm season of the year (27.4 °C), following the seasonal pattern of atmospheric warming in the area. Despite the significant nutrient inputs, mainly derived from the agricultural sector and the rural effluents from the city of Volos, the Pagasitikos Gulf is characterized as meso-oligotrophic [17,18].
Figure 1.
Map of the study area indicating the location of the sampling stations (S1 and S2).
2.2. Field Sampling
Preliminary sampling across the coastline of Pagasitikos revealed the presence of P. lividus over the entire Gulf, on rocky substrates at depths ranging from 0.5 to 10 m. Two stations were, thus, randomly selected: S1 (39°18′19.3″ N, 22°56′5.3″ E) and S2 (39°18′25.1″ N, 23°5′50.9″ E), located at the north and northeast parts of the Gulf, respectively (Figure 1). In both stations, moderately inclined rock was the prevalent habitat type, surrounded by sandy bottoms, either unvegetated or covered by moderately dense seagrass meadows. Fleshy algae of the genus Cystoseira prevailed in S1, while coralline algae together with the brown alga Padina pavonica was found in S2. From each station, 35 specimens of over 4 cm in test diameter were randomly collected at monthly intervals, from December 2008 to November 2010, by a scientist using SCUBA diving equipment at approximately 3 to 5 m depth. Concurrently, the seawater temperature was recorded with an autographic conductivity temperature depth recorder, CTD (SeaBird, Bellevue, WA, USA).
2.3. Sample Processing
In the laboratory, each individual was measured for its test diameter using an electronic caliper (0.01 mm precision); it was drained for 5 min on filter paper and weighed for its total weight (tW) using an electronic scale (0.001 g precision). Then, it was dissected, and the five gonads were removed, measured for volume (Vg, 0.001 mL precision) by applying the liquid (seawater) displacement method [19], weighed (Wg, 0.001 g precision), and preserved in 70% ethanol. Weight measurements were used to estimate the wet gonad-somatic index (GSI) as tW/Wg %, which is the most applied index to assess the reproductive state of P. lividus [6,9,12]. The sex ratio (♂/♀) was also assessed by the macroscopic examination of the gonads or with a smear examination [15].
2.4. Histology
For the histological examination of the gonads, the haematoxylin–eosin staining procedure was applied on the middle portion (1 cm fragment) of one out of the five gonads from each sampled individual [12,15,16]. The gonad samples were sectioned in a microtome at 8 μm, and each section was set on a slide. The histological slides (at least 10 slides per gonad sample) were examined under light microscopy (10 × 100 magnification) to assess the gametogenic stages in both female and male urchins. The different developmental stages of gametogenesis were identified, according to a slightly modified Byrne’s classification scale, using a 5-level (I = spent, II = recovering, III = growing, IV = premature, V = mature) or a 4-level (I = spent, II = recovering, III = growing, IV = mature) scale for females and males, respectively (i.e., omitting the partly spawned stage of both sexes and the premature stage in males).
The histological slides of all female sea urchins that had been classified in the developmental stages III, IV, and V were observed under an Axiostar plus Carl Zeiss Light Microscopy (Carl Zeiss Ltd, Gottingen, Germany) (10 × 100 magnification) connected with a ProgRes C10 digital camera (Berlin, Germany), to estimate the diameter of the oocytes (Do, 0.001 μm precision). Several photographs were taken of each slide, coded, and subsequently processed through image analysis using the software ProgResCapturePro 2.1 (Berlin, Germany). The middle axis of each oocyte, representing Do, was measured using a calibrated scale in μm. Up to 100 oocytes per female were measured. These data were used to construct size frequencies and combined with the gonad volume data to estimate the per female fecundity, calculated as Fvo = Vg/Vvo + (Vpvo × p), where Vg is the gonad volume, Vvo is the volume of mature oocytes, Vpvo is the volume of premature oocytes, and p is the analogy of premature to mature oocytes in the 100 oocytes sample [20], and by assuming a spherical scheme of oocytes to apply the mathematical equation (4 × π × R3/3) that measures the volume of a sphere.
2.5. Statistical Analyses
Analysis of variance (general linear model, GLM ANOVA) was used to examine spatio-temporal differences in the GSI, Fvo, and Do values, e.g., among sampling stations (two-level fixed factor), months (12-level fixed factor), years (two-level random factor), and of their interactions [21]. Prior to the analyses, data were tested for normality with the Anderson–Darling test, while the homogeneity of variance was tested with Cohran’s test. The Fisher least significant differences, test was used for post-hoc comparisons when appropriate. ANOVAs were performed using the SPSS software package (IBM SPSS statistics version 19, Athens, Greece). Statistically significant deviations from a balanced sexual proportion 1:1 were assessed with a chi-square test.
3. Results
3.1. General Biometry and Sex Ratio
In total, 1680 P. lividus specimens were processed—840 from S1 and 840 from S2 (Table 1). The mean size was 50.94 ± 7.25 mm at S1 and 51.42 ± 6.39 mm at S2. The sex ratio was significantly biased in favor of females in both stations (S1: ♂/♀ = 0.87, x2 = 79.31 (p < 0.05) and S2: ♂/♀ = 0.82 x2 = 81.64 (p < 0.05)), being about 1:1.2 over the entire sea urchin population studied.

Table 1.
Number of Paracentrotus lividus female and male specimens per sampling month at the two sampling stations (S1 and S2) in the Pagasitikos Gulf.
3.2. Gonad-Somatic Index
The GSI ranged from 0.10 (January 2010) to 24.99 (March 2009) with a mean value of 4.13 ± 2.71 at S1 and 4.58 ± 2.43 at S2. The GSI showed non-significant differences between stations and years (p > 0.05), whereas the relevant seasonal differences were highly significant (ANOVA F = 7.47, p = 0.001). In both stations, increased GSI values were recorded from late winter to early spring, in March in particular (Figure 2). A secondary rise was also observed from summer to early autumn, which was more pronounced when examined for each year separately. The seasonal GSI trend was negatively correlated with the annual seawater temperature (Sρ = −0.52 and −0.48, for S1 and S2, respectively).
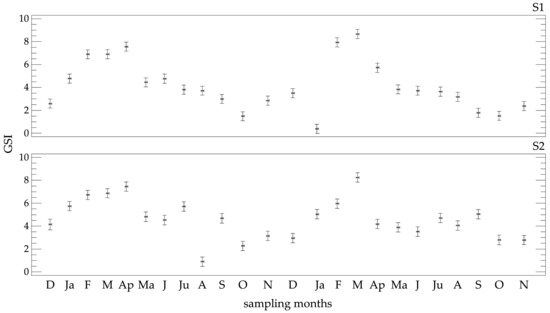
Figure 2.
Temporal trend of the mean gonad-somatic index (GSI) ± Fisher LSD of Paracentrotus lividus at the two sampling stations (S1 and S2) in the Pagasitikos Gulf.
3.3. Histology of Gonads
The pattern of testis growth was divided into four stages (Figure 3). Spent testes (Stage I) appear empty with only a few nutritive phagocytes, which will increase to form a meshwork in the subsequent recovering stage (Stage II). In the growing stage (Stage III), projections of spermatocytes are observable along the meshwork of nutritive phagocytes, which will grow to form spermatozoa and fill the mature testis (Stage IV).
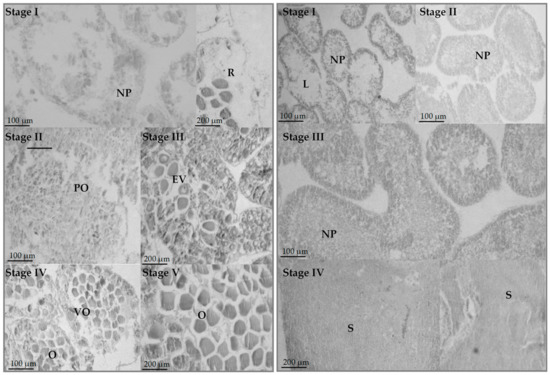
Figure 3.
Histology of ovaries (left) and testes (right) of Paracentrotus lividus. NP = nutritive phagocytes, R = relict oocytes, PO = previtellogenic oocytes, EV = early vitellogenic oocytes, VO = vitellogenic oocytes, O = oocytes, L = lumen, and S = spermatozoa.
The pattern of ovarian growth was divided into five stages (Figure 3). Spent ovaries (Stage I) appear empty with only a few relict oocytes, which will be reabsorbed under the action of nutritive phagocytes, observable in some ovaries. Recovering ovaries (Stage II) are characterized by the presence of nutritive phagocytes creating a meshwork, together with some small, previtellogenic oocytes. As the ovary passes through the growing stage (Stage III), early vitellogenic oocytes appear. By reaching the premature stage (Stage IV), they grow to form large oocytes that displace nutritive phagocytes from the center of the ova and start to accumulate in the lumen. In the mature stage, the ovaries are filled with closely packed and large ova. However, a few early primary oocytes may be observed in both IV and V stages, as well as large relict oocytes in Stages I, II, and III. Interestingly, 39 individuals were hermaphroditic (Figure 4), constituting 2.56% of the studied population. Hermaphroditic urchins were detected at both stations, without any evidence of temporality.
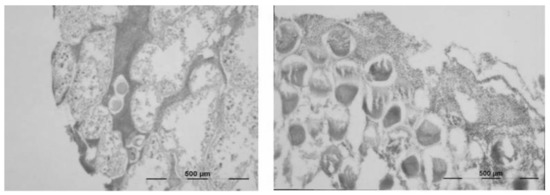
Figure 4.
Hermaphroditic Paracentrotus lividus individuals—female gonads with sperm (left) and male gonads with oocytes (right).
The relative frequencies of the maturity stages in female and male urchins were made for each station separately over the two annual cycles of the study (Figure 5 and Figure 6), despite the lack of significant differences in the GSI. The histograms showed a synchronized reproductive pattern between sexes. A clear annual reproductive cycle was evident in both years, with a spawning peak in late winter to early spring (February, March, and April); a secondary spawning period was also detected in late autumn–early winter (October and November). Spent or recovering gonads were observed in all sampling months, but in the summer months the entire population was in these “immature” stages. Although small differences between stations and years were observable, these differences had a limited impact on the overall annual reproductive pattern of the species.
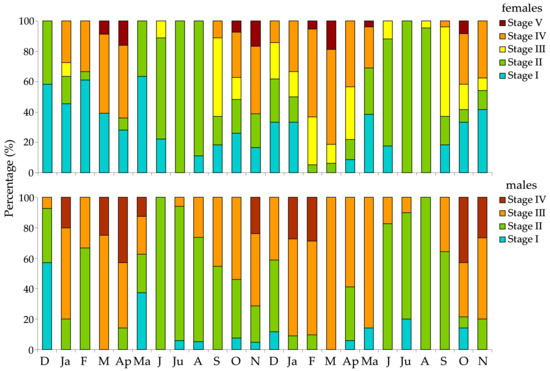
Figure 5.
Relative frequency of gonad maturity stages of female and male Paracentrotus lividus individuals at the S1 station in the Pagasitikos Gulf, per month, from December 2008 to November 2010.
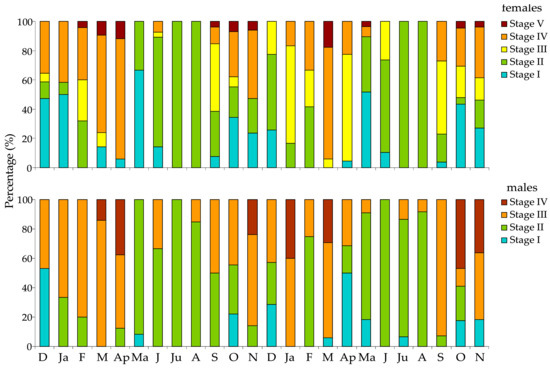
Figure 6.
Relative frequency of gonad maturity stages of female and male Paracentrotus lividus individuals at the S2 station in the Pagasitikos Gulf, per month, from December 2008 to November 2010.
3.4. Female Fecundity
The per female fecundity ranged from 6.78 to 0.72 × 106, with a mean value of 3.34 ± 2.07 × 106 at S1 and 2.82 ± 16.8 × 106 at S2. Fecundity was slightly increased in S1; however, non-significant differences were observed in Fvo between the two stations and the years of the study (p > 0.05). On the contrary, significant seasonal differences were observed (F = 10.67, p = 0.001), with increased Fvo values from late winter to early spring, in autumn, and in May or June (Figure 7). However, the percentage of females that had oocytes (i.e., growing III, premature IV, or mature V developmental stages of oogenesis) was very low in the latter case (less than 10% of examined females), as most females were in the early oogenetic stages (not allowing fecundity estimations), which infers uncertainty in Fvo estimations.
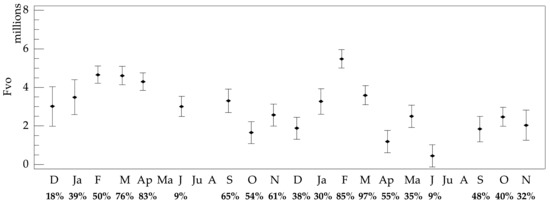
Figure 7.
Temporal trends of the mean per female fecundity, Fvo ± Fisher LSD, of Paracentrotus lividus in the Pagasitikos Gulf (data were pooled across both stations). The percentage of females having oocytes (oogenetic stages III, IV, and V) and, therefore, used to estimate fecundity is marked below each sampling month.
3.5. Oocyte Size
The oocyte diameter (Do) ranged from 14.12 to 121.95 μm, with a mean of 65.86 ± 16.52 and 67.96 ± 16.92 at S1 and S2, respectively. Non-significant differences were observed in Do on both the spatial (between the two stations, p > 0.05) and temporal scales (between the two years of the study). On the contrary, significant seasonal differences were observed (F = 135.81, p = 0.001) with increased values in March, April, and May and very low values in January and February. A size frequency analysis of Do per month (Figure 8) generally agreed with the GSI and the cycle of oogenesis. However, some large oocytes were detected after spawning, as well as some small oocytes in the mature stages.
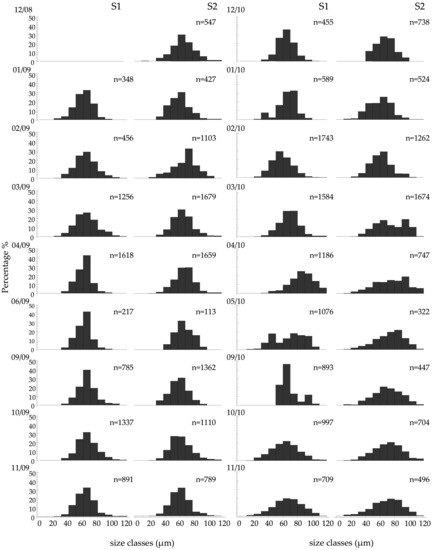
Figure 8.
Size frequency distribution of the oocyte diameter. Do of Paracentrotus lividus at the two sampling stations (S1 and S2) of the Pagasitikos Gulf, per sampling month. Only females in the oogenetic stages III, IV, and V were measured (up to 100 oocytes per female).
4. Discussion
Paracentrotus lividus is a gonochoristic species with external fertilization. Its size at sexual maturity is highly variable (2–4 cm in test diameter), depending on the geographical area and habitat, and is probably due to the different energetic investment in reproduction or growth [6,22,23,24]. The vast majority of studies select urchins larger than 3 cm to study reproduction, to ensure that all measured specimens are relevant. Accordingly, sea urchins of 4–5 cm were selected in the present study.
Literature data on the reproductive biology of P. lividus are diverse and often contradictory, as the species seems to follow different patterns in accordance with the geographical location and prominent environmental parameters, such as temperature, habitat, depth, and food availability [6,13,16]. In the Mediterranean Sea, the species typically has a main spawning episode usually in spring, driven by phytoplankton density [8,12,15,22], and a secondary less intense event in autumn [1,6,25]. However, winter events or prolonged spawning periods up to early summer have also been proposed, based on GSI or histological data [3,14,25,26]. Ovaries in mature stages are usually present from late winter to early spring [9,12,14], although mature gonads may be present all year round [14]. The latter pattern is also common in male urchins, which may have sperm all year round [12] or in specific months [14]. In the present study, spent or recovering gonads were observed in all sampling months, but in summer the entire population was in this state. It is widely accepted that P. lividus may have successive spawning events in the Mediterranean [6], (and references therein). These extensions in the main annual reproductive episode have been related with latitude, thermal regime, food availability, and hydrodynamics [6,27]. Food availability, fleshy macroalgae in particular, has been ascertained as the main driving factor of gonad maturation in P. lividus [26].
The results of the present study, based both on the GSI and histology, report a clear major spawning event in spring (March and April) and a second event in autumn (October and November), which is, however, less severe. This pattern conforms to the only available relevant data (GSI data) from eastern Mediterranean P. lividus populations, namely from Astypalaia Island in the south Aegean Sea [1]. However, it is still under question as to whether these second spawning episodes actually result from a new gametogenic cycle or from a short inactive period in the gametogenic cycle due to increased temperatures—vitellogenesis is inhibited at high temperatures (over 22 °C) [9,16]. The histological examination of the gonads together with the oocytes’ size frequency data provide some evidence for a second gametogenic event, as the gonads were in the recovery stage in the summer months (July and August) in the studied population. Therefore, the existence of a discrete second spawning period may be inferred, suggesting also an increased reproductive potential of P. lividus, especially over its southern distributional range.
Large oocytes (>90 μm) were present in most sampling months when females in maturing stages were sampled. These oocytes are indicative either of a spawning event, as is the case in the spring and autumn months, or they represent unspawned oocytes (relicts), as previously reported for the species [16]. Small, previtellogenic oocytes (<30 μm) and early vitellogenic oocytes (<50 μm) were also present throughout the year. Small oocytes have been ascertained as the most abundant in P. lividus ovaries, even in mature ones, as well as in other sea urchin species [16].
Incidences of hermaphroditism are common in echinoids, although they usually constitute a small portion of the population. Specimens with ovotestes that have predominately female gonads have been reported for P. lividus [16]. In the present study, ovotestes were observed, as well as testes having oocytes, supporting a mosaic structure of the gonad, i.e., a gonad producing both spermatozoa and oocytes, whereas the presence of both testes and ovaries in the same individual has also been reported [28].
The results of the present study show that the harvesting period of P. lividus in Greece, coincides either with the growing and spawning stage of the gonadal development (winter to spring) or the recovery stage (summer). Accordingly, sea urchins are mainly fished when nutritive material is stored in the gonads and before being converted into gametes. However, in March, the P. lividus population is in an advanced stage of sexual maturity (spawning stage) and ready to release mature oocytes and spermatozoa. On the other hand, in both summer months, when the urchin is fished, its gonads are undersized, nutritive material is just starting to accumulate, and they contain relict oocytes under the process of phagocytosis. It is, therefore, obvious that fisheries have a severe impact on the species’ reproductive output, and so, the fishing season should be revised, especially by considering the inclusion of March and the limited gonad product in summer. Regulating sea urchin fisheries is imperative, especially under the synergistic negative effect of global warming on P. lividus natural stocks [10].
Author Contributions
Conceptualization, D.V.; Formal analysis, C.A. and K.K.; Methodology, D.V.; Software, C.A. and K.K.; Supervision, D.V.; Writing—original draft, C.A.; Writing—review & editing, D.V., C.A. and K.K.
Funding
This research received no external funding.
Acknowledgments
The authors would like to thank Dr Alexios Lolas for his help during the sampling.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Antoniadou, C.; Vafidis, D. Population structure and morphometric relationships of Paracentrotus lividus (Echinodermata: Echinoidae) in the south Aegean Sea. Cah. Biol. Mar. 2009, 50, 293–301. [Google Scholar]
- Andrew, N.L.; Agatsuma, Y.; Ballesteros, E.; Bazhin, A.G.; Greaser, E.P.; Barnes, D.K.A.; Botsford, L.W.; Bradbury, A.; Campbell, A.; Dixon, J.D.; et al. Status and management of the world sea urchin fisheries. Oceanogr. Mar. Biol. 2002, 40, 343–425. [Google Scholar] [CrossRef]
- Guidetti, P.; Terlizzi, A.; Boero, F. Effects of the edible sea urchin Paracentrotus lividus fishery along the Apulian rocky coast (SE Italy, Mediterranean Sea). Fish. Res. 2004, 66, 287–297. [Google Scholar] [CrossRef]
- Pais, A.; Serra, S.; Meloni, G.; Saba, S.; Ceccherelli, G. Harvesting effects on Paracentrotus lividus population structure: A case study from northwestern Sardinia, Italy, before and after the fishing season. J. Coast. Res. 2012, 28, 570–575. [Google Scholar] [CrossRef]
- Bertocci, I.; Domínguez, R.; Machado, I.; Freitas, C.; Domínguez Godino, J.; Sousa-Pinto, I.; Gonçalves, M.; Gaspar, M. Multiple effects of harvesting on populations of the purple sea urchin Paracentrotus lividus in north Portugal. Fish. Res. 2014, 150, 60–65. [Google Scholar] [CrossRef]
- Ourens, R.; Fermamdez, L.; Freire, J. Geographic, population, and seasonal patterns in the reproductive parameters of the sea urchin Paracentrotus lividus. Mar. Biol. 2011, 158, 793–804. [Google Scholar] [CrossRef]
- Perry, I.R.; Walters, C.J.; Boutillier, J.A. A framework for providing scientific advice for the management of new and developing invertebrate fisheries. Rev. Fish Biol. Fish. 1999, 9, 125–150. [Google Scholar] [CrossRef]
- De la Uz, S.; Carrasco, F.; Rodriguez, C. Temporal variability of spawning in the sea urchin Paracentrotus lividus from northern Spain. Reg. Stud. Mar. Sci. 2018, 23, 2–7. [Google Scholar] [CrossRef]
- González-Irusta, J.M.; Goni de Cerio, F.; Canteras, J.C. Reproductive cycle of the sea urchin Paracentrotus lividus in the Cantabrian Sea (northern Spain): Environmental effects. J. Mar. Biol. Assoc. UK 2009, 90, 699–709. [Google Scholar] [CrossRef]
- Yeruham, E.; Rilov, G.; Shpigel, M.; Abelson, A. Collapse of the echinoid Paracentrotus lividus populations in the eastern Mediterranean—Result of climate change? Sci. Rep. 2015, 5, 13479. [Google Scholar]
- Konstantinidis, I.; Gkafas, G.A.; Karamitros, G.; Lolas, A.; Antoniadou, C.; Vafidis, D.; Exadactylos, A. Population structure of two benthic species with different larval stages in the eastern Mediterranean Sea. J. Environ. Prot. Ecol. 2017, 18, 930–939. [Google Scholar]
- Sánchez-España, A.I.; Martinez-Pita, I.; Garcia, F.J. Gonadal growth and reproduction in the commercial sea urchin Paracentrotus lividus (Lamarck, 1816) (Echinodermata: Echinoidea) from southern Spain. Hydrobiologia 2004, 519, 61–72. [Google Scholar] [CrossRef]
- Spirlet, C.; Grosjean, P.; Jangoux, M. Reproductive cycle of the echinoid Paracentrotus lividus: An analysis by means of the maturity index. Invertebr. Reprod. Dev. 1998, 34, 69–81. [Google Scholar] [CrossRef]
- Tenuzzo, B.A.; Zaccarelli, N.; Dini, L. The reproductive cycle of the commercial sea urchin Paracentrotus lividus (Lamarck, 1816) (Echinodermata: Echinoidea) in the Ionian Sea. Ital. J. Zool. 2012, 79, 200–211. [Google Scholar] [CrossRef]
- Sellem, F.; Guillou, M. Reproductive biology of Paracentrotus lividus (Echinodermata: Echinoidea) in two contrasting habitats of northern Tunisia (south-east Mediterranean). J. Mar. Biol. Assoc. UK 2007, 87, 763–767. [Google Scholar] [CrossRef]
- Byrne, M. Annual reproductive cycles of the commercial sea urchin Paracentrotus lividus from an exposed intertidal and a sheltered subtidal habitat on the west coast of Ireland. Mar. Biol. 1990, 104, 275–289. [Google Scholar]
- Petihakis, G.; Triantafyllou, G.; Pollani, A.; Koliou, A.; Theodorou, A. Field data analysis and application of a complex water column biogeochemical model in different areas of a semi-enclosed basin: Towards the development of an ecosystem management tool. Mar. Environ. Res. 2005, 59, 493–518. [Google Scholar] [CrossRef]
- Korres, G.; Triantafyllou, G.; Petihakis, G.; Raitsos, D.E.; Hoteit, I.; Pollani, A.; Colella, S.; Tsiaras, K. A data assimilation tool for the Pagasitikos Gulf ecosystem dynamics: Methods and benefits. J. Mar. Syst. 2012, 94, 102–117. [Google Scholar] [CrossRef]
- Garrido, M.J.; Haroun, R.J.; Lessios, H.A. Annual reproductive periodicity of the sea urchin Diadema antillarum Philippi in the Canary Islands. Bull. Mar. Sci. 2000, 67, 989–996. [Google Scholar]
- Ramirez-Llodra, E. Fecundity and life history strategies in marine invertebrates. Adv. Mar. Biol. 2002, 43, 87–170. [Google Scholar] [CrossRef]
- Underwood, A.J. Experiments in Ecology. Their Logical Design and Interpretation Using Analysis of Variance, 2nd ed.; Cambridge University Press: Cambridge, UK, 1997; 524p. [Google Scholar]
- Lozano, J.; Galera, J.; López, S.; Turon, X.; Palacín, C.; Morera, G. Biological cycles and recruitment of Paracentrotus lividus (Echinodermata: Echinoidea) in two contrasting habitats. Mar. Ecol. Prog. Ser. 1995, 122, 179–191. [Google Scholar]
- Turon, X.; Giribet, G.; Lopez, S.; Palacin, C. Growth and population structure of Paracentrotus lividus (Echinodermata: Echinoidae) in two contrasting habitats. Mar. Ecol. Prog. Ser. 1995, 122, 193–204. [Google Scholar]
- Ouréns, R.; Fernandez, L.; Fernandez-Boan, M.; Naya, I.; Freire, J. Reproductive dynamics of the sea urchin Paracentrotus lividus on the Galicia coast (NW Spain): Effects of habitat and population density. Mar. Biol. 2013, 160, 2413–2423. [Google Scholar] [CrossRef]
- Guettaf, M.; San Martin, G.A.; Francour, P. Interpopulation variability of the reproductive cycle of Paracentrotus lividus (Echinodermata: Echinoidea) in the south-western Mediterranean. J. Mar. Biol. Assoc. UK 2000, 80, 899–907. [Google Scholar] [CrossRef]
- Murillo-Navarro, R.; Jiménez-Guirado, D. Relationships between algal food and gut and gonad conditions in the Mediterranean sea urchin Paracentrotus lividus (Lam.). Mediterr. Mar. Sci. 2012, 13, 227–238. [Google Scholar] [CrossRef][Green Version]
- Gianguzza, P.; Bonaviri, C.; Prato, E.; Fanelli, G.; Chiantore, M.; Privitera, D.; Luzzu, F.; Agnetta, D. Hydrodynamism and its influence on the reproductive condition of the edible sea urchin Paracentrotus lividus. Mar. Environ. Res. 2013, 85, 29–33. [Google Scholar] [CrossRef]
- Gago, J.; Range, F.; Luis, O. Growth, reproductive biology and habitat selection of the sea urchin Paracentrotus lividus in the coastal waters of Cascais, Portugal. In Echinoderm Research, 1st ed.; Feral, J.P., David, B., Eds.; Balkema: Lisse, The Netherlands, 2003; pp. 269–276. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).