1. Introduction
Pathogen contamination in drinking, recreational, and irrigation water results in numerous diseases and poses a serious health threat to human society.
E. coli and fecal coliforms in fresh water are considered to be closely associated with outbreaks of gastrointestinal illness and are thus used as typical indicator bacteria to evaluate the contamination level of fresh water [
1].
Attachment to sediment particles affects the transport and fate of bacteria in water and, in turn, has a major effect on the contamination level of water. Particles larger than a certain size (4~8 μm) are settleable under natural conditions [
2] and particle-associated bacteria (i.e., attached cells) thus have a higher settling rate than their free-floating counterparts. Once settled to the bottom, the bacteria can survive much longer than planktonic bacteria because of the hospitable environment [
3,
4]. The bacteria concentration in bottom sediments has been found to be 10–1000 times higher than that in the overlying water [
5]. Therefore, sediments can act as reservoirs of bacteria [
6] and when sediment particles are resuspended by, for example, storms, tides, or winds, attached bacteria can detach and move freely again in the water column. Studies have shown that in some cases the resuspension of sediments, rather than runoff from surrounding lands, results in elevated
E. coli concentration in water [
5].
Although the importance of bacterial attachment to particles is widely acknowledged, the exact portion of attached bacteria, i.e., the ratio of the number of attached bacteria to the total number in water, remains incompletely understood, as do the factors influencing this variable. In some cases, the portion was found to vary between 15% and 40% [
2,
7,
8], while in other cases it can exceed 90% [
9,
10]. Bacterial attachment is reported to be dependent on particle concentration; for example, George et al. found that the percentage of
E. coli associated with suspended particles larger than 5 μm increased proportionally with particle concentration [
11]. Bacterial attachment was also found to be significantly dependent on the size of particles. Most previous researchers have found that bacteria tend to attach to smaller particles; this finding is consistent with the explanation that, for a given mass, smaller solid particles have more surface area and can provide more attachment sites, resulting in more attached cells. Results have shown that bacterial attachment is more efficient on soil, silt and clay particles than on sand particles [
12]. Soupir et al. assumed any bacteria that could not pass through an 8-μm filter were attached and found that at least 60% of all attached
E. coli and enterococci were associated with particles smaller than 62 μm [
13]. Among particles of different sizes, more than 90% of
E. coli were attached to smaller particles (5–30 μm) [
14]. A similar study indicated that the number of attached cells per gram of sediment was negatively correlated with particle size in a size range from 4 μm to 75 μm [
15]. On the other hand, Oliver et al. investigated preferential attachment and found that
E. coli preferentially attached to soil particles within a size range of 16–32 μm [
16] by comparing the number of attached cells per unit surface area on particles of different sizes, but they did not explain the mechanism of this preferential attachment.
The linear partition model has been widely used to describe theoretically the relation between particle-associated and planktonic bacteria [
17,
18]:
where
Cs = the concentration of particle-associated bacteria (the number of bacteria associated with particles per unit water volume), colony-forming units (cfu)/L;
s = sediment concentration, g/L;
k = the partition coefficient, L/g;
Cw = the concentration of planktonic bacteria, cfu/L. This linear model works reasonably well in groundwater with relatively low bacteria concentrations [
19,
20] and has been verified in surface water [
19], although it is still an empirical formula and lacks a fundamental thermodynamic basis [
21,
22]. Equation (1) agrees with the aforementioned observations that the particle concentration has an impact on bacterial attachment, but it does not explain how factors such as particle size affect bacterial attachment, and the influencing factors of the partition coefficient
k is still not clear.
The abovementioned relationship between particle-associated and planktonic bacteria was observed using traditional methods. Typically, the attached and free cells are separated using filtration or centrifugation and then enumerated based on incubation [
23]. These methods, however, may involve great uncertainty and measurement error. With the development of microscopy, it may seem straightforward to directly observe the distribution of attached bacteria on particles. However, the number of bacteria is much fewer than the number of particles under ordinary conditions. For example, when the concentration of bacteria is 10
5 (cfu)/L and the suspended sediment concentration, with a density of 2.6 g/cm
3 and diameter of 30 μm, is 1 g/L, the number of sediment particles is 272 times the number of bacteria. This finding is in agreement with Goulder’s (1977) observation that in an estuary, there were many particles on which no bacteria were attached [
24]. Thus, very few particles have attached bacteria on their surface, which makes direct observation very difficult.
On the other hand, microfluidic devices have been shown to be able to make observations at high spatial resolution in controlled microenvironments [
25,
26,
27] and offer a powerful tool to study microbial motility, including bacterial attachment [
28]. Recent advances in bacterial imaging and tracking also enable the study of microbial activity to be performed at a single-cell level [
29,
30]. Microscopical features of substrates like surface topography of micrometer and nanometer scale were also found to influence bacterial attachment [
31]. The aim of this study is to gain an improved understanding of bacterial attachment to particles. The emphasis was placed on the impact of size on the attachment and to exclude the impact of other facts, we used regular spherical glass particles of different diameters in addition to natural sediment particles. The glass particles, similar to the natural sediment particles used, are mainly composed of SiO
2 and are similar in density to the natural particles. We placed particles of different sizes in flow cells, through which we continuously streamed an
E. coli suspension. Using microscopy, we chose a number of particles, estimated the number of bacteria coming into contact with them, and directly counted the number of bacteria that remained attached on them. From these data, the attachment probability, i.e., the number of bacteria attached to a particle versus the number ever approaching the particle, was calculated. Then, the relation between cell attachment probability and particle size was analyzed. The partition coefficient of the linear partition model was conceptualized based on the microscopic measurements and compared with the results from the literature.
2. Materials and Methods
2.1. Bacterial Strain and Bacterial Suspension
The strain of E. coli used in the experiments is American Type Culture Collection (ATCC) 8099 and was purchased from the China General Microbiological Culture Collection Center. Stock E. coli cultures were maintained on beef extract-peptone agar and stored at 4 °C between experiments.
The bacterial suspension used in the experiments was prepared by suspending ATCC 8099 cells in Phosphate-buffered saline (PBS) at OD600~0.1 at room temperature and the concentration was assessed following membrane filtration procedures (USEPA method 1603). Ten-micromolar PBS (pH = 7.4) was used throughout the experiments. Before each experiment, bacteria were first grown on nutrient broth (NB) agar plates at 37 °C overnight. Monoclonal colonies were isolated and inoculated into culturing tubes containing 5 mL NB medium and then grown in an incubator shaken at 150 rpm at 37 °C. After 7 h, 1 mL of E. coli culture, which reached the exponential growth phase (OD600 ≈ 1.0), was further diluted 20-fold in 100 mL NB medium and grown again in the same incubator for 15 h. Subsequently, cells were harvested in the stationary phase by centrifugation at 12,000 rpm for 2 min, after which they were washed three times with sterile PBS.
2.2. Particles
Two types of particles were used in this study: natural sediment particles (quartz sand) and monodispersed glass microspheres (Duke Standards).
The sediment was taken from the Yongding River in Beijing, China. Sediment particles within the size ranges of 8–16 μm, 16–31 μm, and 31–62 μm were separated out by repeated sedimentation based on Stokes’ law and were then used in experiments. The median diameters for the three groups are 12 μm, 23.5 μm, and 46.5 μm, respectively. Before use, the sediment particles were first cleaned by soaking in distilled water and hydrogen peroxide to remove any inorganic matter or microorganisms deposited on them. Then, the particles were dried in an oven at 45 °C for 10 h. The sediment particles have a specific gravity of 2.650 and are composed mainly of quartz (silica content > 90%), along with some feldspar, chlorite and illite. The size of the sediment particles was analyzed by a laser particle analyzer (HORIBA LA-920).
The glass microspheres used in this study are an ideal model for studying the isolated effect of particle size on bacterial attachment due to their uniform size and shape as well as their isotropic surface properties. Microspheres of three different diameters (10 μm, 20 μm, and 50 μm) were chosen, corresponding to the mean sizes of the sediment particles. The glass microspheres are made of borosilicate glass and are similar to the sediment particles in chemical composition (silica content > 80%) and specific gravity (2.550). The microspheres have a size polydispersity of no more than 10%.
2.3. Microfluidic Device and Microscope
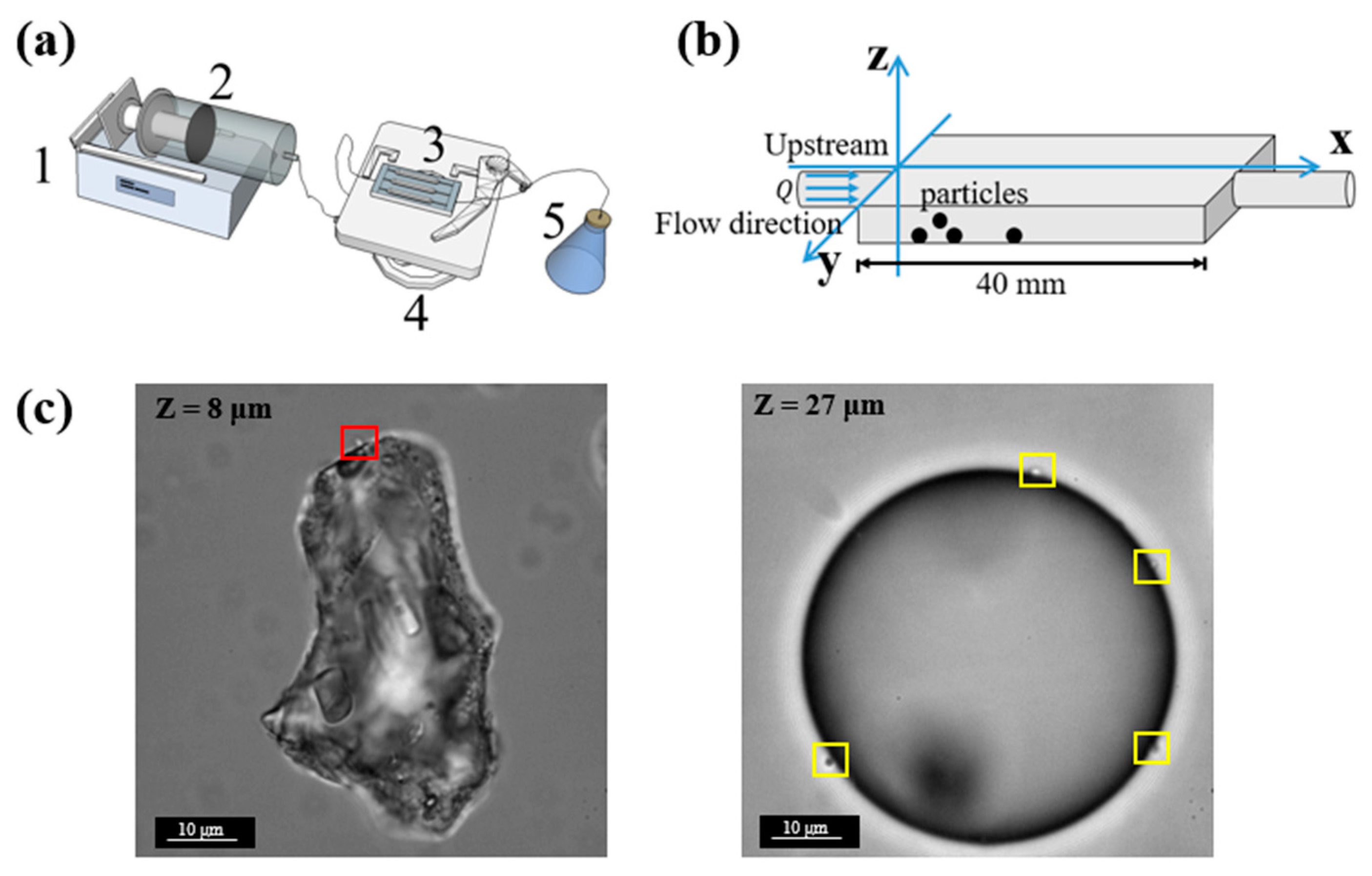
Flow cells (purchased from the Department of Biotechnology and Biomedicine, Technical University of Denmark) were used to observe bacterial attachment under a microscope. Each of the flow cells has three identical rectangle channels with dimensions of 40 mm × 4 mm × 1 mm (length × width × depth). The microfluidic system was prepared using a standard protocol [
32]. A syringe was connected to the inlet of a microchannel via silicone tubing (inner diameter: 0.8 mm), and the plunger was pushed by a syringe pump (Harvard Apparatus PHD Ultra 703006, Harvard Apparatus, MA, USA) (
Figure 1a).
Images and videos in the experiments were taken using an electron-multiplying charge-coupled device (EMCCD) camera (Andor iXon) on a Leica DMi8 inverted microscope. Due to the limited depth of focus of the optical microscope (with 100x objective, numerical aperture = 1.4), we could not obtain a full view of a particle in only one frame. Therefore, a z-scan was conducted. The z-scan was performed at a step size of 1 μm, a typical z-scan takes 5.5 s, 8.8 s, and 17.6 s for particles of 10 μm, 20 μm, and 50 μm, respectively. From the latter results, we can see that in each z-scan period, the attached bacteria can be considered stationary. The size of the field of view is 66.5 μm × 66.5 μm. Particles at different positions were picked. For each condition, at least 3 repeats were performed. To minimize errors, we manually checked each image from z-scan period and counted the number of attached cells.
2.4. Experimental Procedure
The attachment of bacteria to surfaces can be separated into three stages: (1) the approaching stage; (2) the initial attachment stage; and (3) attachment strengthening stage [
33]. In the approaching stage, the bacteria achieve proximity to a surface, which can be achieved by active motion or passive transport such as fluid flow and Brownian motion. When bacteria are sufficiently close to the surface, attachment can be initiated through further direct contact with the surface. The initial attachment is influenced by multiple factors, including the physical and chemical properties of the surface and the biological properties of the bacteria. After initial attachment, bacteria can strengthen their bond by producing adhesion molecules [
33].
Here, we define the adjacent area as the water region that is within 2 μm of the surface of the particle. When entering this adjacent area, bacteria are considered to have completed the approaching stage and could initialize attachment later.
The experiment contained two steps. In the first step, particles were placed in the channels before the flow-cell system was assembled and when the E. coli suspension flowed through the channels, cells attached to the particles were counted. In the second step, the number of cells that had ever approached a particle (entered its adjacent area) in the flow cell was estimated based on fluid mechanics. By comparing the number of attached cells and the number of approaching cells, the attachment probability was calculated and then analyzed. The attachment probability reflects the efficiency of initial attachment and attachment strengthening. Thus, the influence of sediment properties on bacterial attachment can be studied by analyzing the relationship between those properties and attachment probability. Moreover, the number of attached bacteria and the partition coefficient in real water can also be inferred through estimation of the approaching number combined with the attachment probability.
2.5. Counting of Attached Cells
Particles were placed in the channels before the flow-cell system was assembled. For each run, pure sterile PBS was first injected with a constant flow rate of 3.0 mL/h at 25 °C for 5 min until the particles settled to the bottom of the channel and remained immobile there. Then, the E. coli suspension was continuously injected into the flow-cell channel at a constant flow rate of 3.0 mL/h at 25 °C.
In this step, cells that remained attached to some of the particles were directly counted under the microscope. The observation fields were randomly chosen, while each field of view contained only one particle.
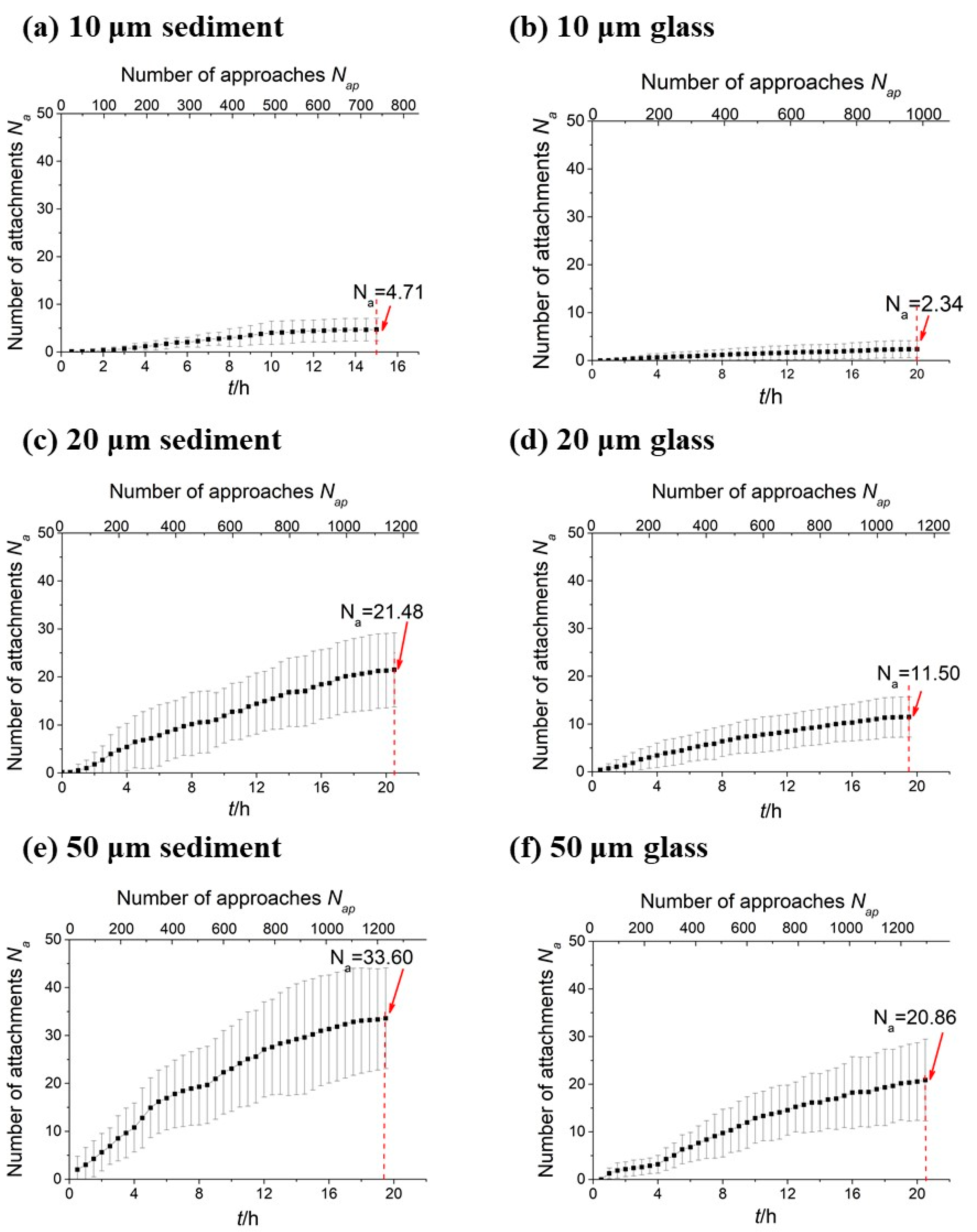
For each type of particle (sediment particles and glass microspheres of different sizes), 30 observation fields were picked randomly along the Y-axis (i.e., perpendicular to the flow direction). The data collection was performed every 0.5 h for 20 h. In each collection, a z-scan of the particles with a step size of 1 μm was conducted for each of the 30 observation fields in turn. Images obtained from the z-scans were carefully examined to count the number of attached
E. coli (
Figure 1c). The error due to misrecognition between bacteria and surface defects of the particles is estimated to be no more than 5%.
Microspheres are transparent; therefore, attached E. coli cells above the particle equator are still distinguishable when observed through an inverted microscope and the total number of cells attached to the whole surface can be counted. However, sediment particles are opaque; therefore, it is difficult to clearly see the attached cells above the particle equator. (Considering that a sediment particle is very often nonspherical, the equator of a sediment particle is defined here as the perimeter of the largest cross-section of the particle from the top/bottom view.) For this purpose, we first obtained a partial attachment number by counting the number of attached cells below the particle equator, then, with the assumption that the distribution of attached cells on the sediment particle is roughly uniform, we could estimate the total number of bacteria attached to the whole surface by multiplying the partial attachment number with a compensation factor defined by the ratio between the average diameter of sediment particles and the average height of the below-equator part of sediment particles, which is Na = fc Nlocal = DNlocal/H, where fc = the compensation factor; D = the median diameter of suspended sediment particles, μm; H = the height of the below-equator part of sediment particles that is used for counting E. coli, μm; Nlocal = the local attachment number of cells on the below-equator part of sediment particles. It is found that the number of attached cells on the front surface of the particles is roughly the same as that on the rear surface, which suggests that the assumption of a roughly uniform distribution of attached cells on particles is reasonable.
The average heights of the below-equator part of sediment particles were measured to be 9.48 μm, 12.50 μm and 27.78 μm for diameter groups of 8–16 μm, 16–31 μm, and 31–62 μm, respectively.
2.6. Estimating of Approaching Cells
Counting the number of cells contacting a single selected particle requires a very long time to obtain statistically meaningful counting results with the current setup, which makes this impractical. Therefore, the number of approaching cells was estimated based on fluid mechanics.
The
E. coli suspension was continuously injected into the flow-cell channel with a constant flow rate of 3.0 mL/h at 25 °C. At this flow rate, the velocity in the flow cell is approximately 208 μm/s, much greater than the swimming speed of
E. coli (~20 μm/s) [
34]. These conditions are similar to the case of typical surface waters, where the flow velocity is much greater and planktonic
E. coli can be considered to be carried by water flow while the active movement can be neglected [
34].
E. coli in the channel is pulled away by the flow, and because the Reynolds number is very low (<<1) and the flow follows Stokes flow.
During a time period of
t, we can estimate the number of cells that have approached a particle (entered the adjacent area)
Nap using Equation (2):
where
Φ1 = the flux of
E. coli at the adjacent area, count/(μm
2·s);
t = time period, s;
S = the projected area of the adjacent area perpendicular to the flow direction, which was a circular ring, μm
2;
u1 = the flow velocity at the adjacent area, μm/s;
l = distance between the particle surface and the boundary of adjacent area, μm
; R0 = the radius of particle, μm;
= the average concentration of planktonic bacteria, cfu/L.
Here, we use the average concentration of planktonic bacteria (
) to simplify the calculation. This assumption is reasonable because we selected many particles randomly and used the average cell concentration to obtain the overall number of approaching cells.
was measured by standard membrane filtration procedures. Furthermore, we used a direct counting method under a microscope to calculate
and obtained consistent results (
Supplementary Materials Figure S1). However, it is worth mentioning that
Cw may not be constant throughout the channel due to ‘shear trapping’. Some motile cells, such as
E. coli, tend to be depleted in the low-shear region and accumulate in the high-shear region due to the alignment of the swimming direction with fluid streamlines caused by shear forces [
35].
u1 can be expressed as Equation (3) according to the theoretical solution of Stokes flow:
where
U0 = average velocity, μm/s.
Thus,
Nap can be calculated with Equation (4):
where
Φp =
U0·
Cw, the flux of non-particle-associated
E. coli that is directly measured, count/(μm
2·s).
2.7. Attachment Probability Per Approach and Per Contact
We define attachment probability per approach as Equation (5):
where
Na is the number of cells that are ultimately attached to a particle, while
Nap is the number of cells that have ever approached the particle.
Furthermore, we introduce the attachment probability per contact,
pc. After approaching the particles, cells need to directly contact the surface to initiate attachment [
33]. However, not every cell-particle contact can lead to attachment.
E. coli in contact with particles can still escape from the near-surface regions of the particles. Tumbling, identified as rapid changes in swimming direction, is one of the influencing factors [
36]. Therefore,
pc reflects the influence of biological and chemical properties of cells and particles on attachment while excluding the effect of external hydrodynamic conditions.
pc can be calculated as the attachment probability per approach, dividing the number of contacts by the number per approach. In the microfluidic experiment, i.e., under laminar flow, the larger the size of the particle, the longer distance cells would move to pass through the particle and thus lead to higher contact frequency. The contact number per approach is assumed to be proportional to the particle size. Thus,
pc can be expressed as Equation (6):
4. Conclusions
In this work, bacterial attachment to sediment and glass particles of different sizes was directly observed. Our experiments separated the process of bacterial approach to particles from the process of initializing attachment after direct contact. The results showed that the particle size of sediments is the mainly influence on the number of cells that approach and directly contact the sediment particle. However, there is little difference in initializing attachment after direct contact when particle sizes changed.
In addition, the attachment probability per approach and per contact on sediment particles were found to be higher than those on glass microspheres with equivalent sizes under similar conditions. This difference in attachment is probably due to the different surface morphologies between sediment and glass particles. Work is underway to reveal the effect of sediment surface heterogeneity on E. coli attachment.
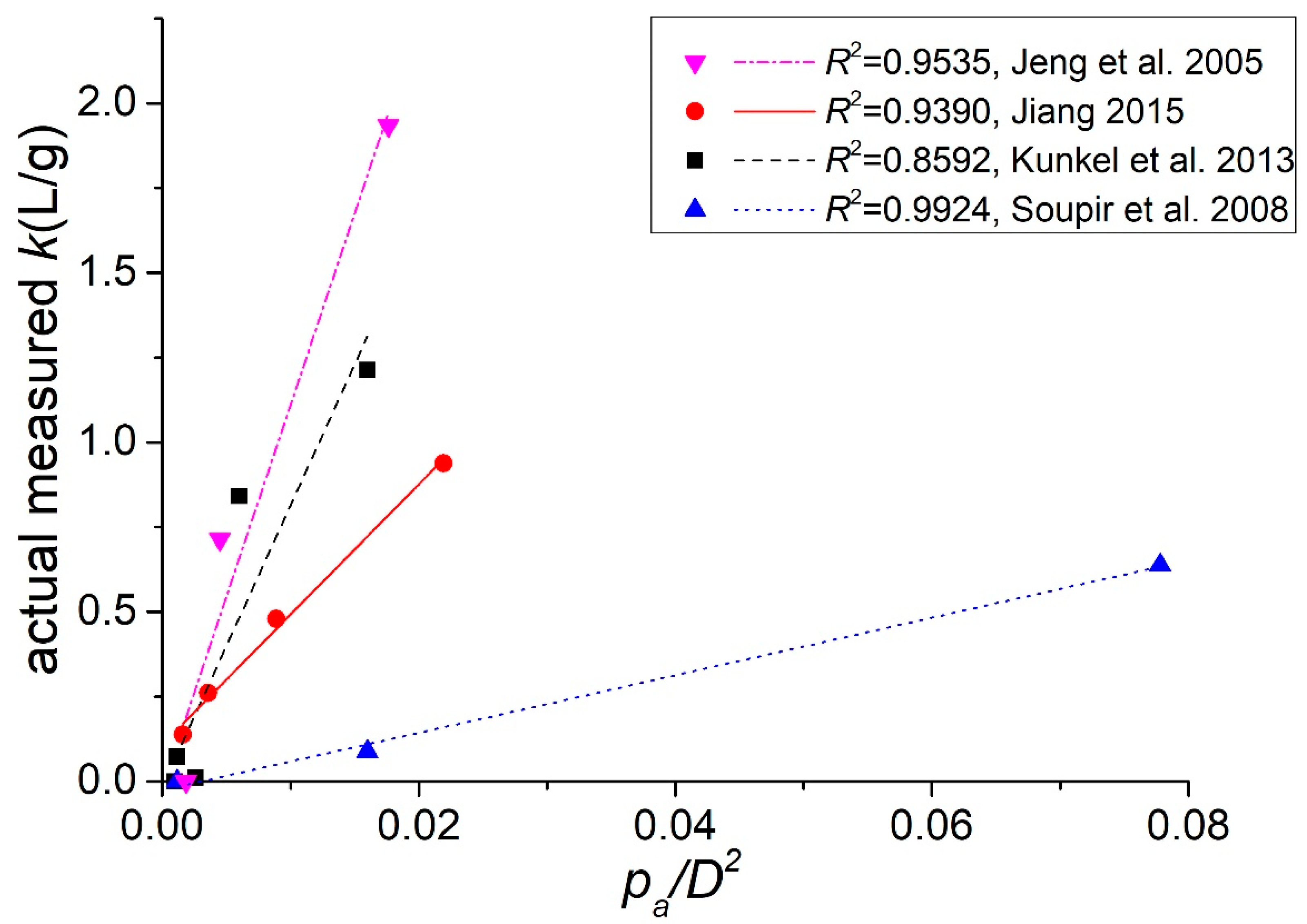
Based on the widely used linear partition model, we conceptualized the partition coefficient by incorporating the experimentally measured positive correlation between the attachment probability per approach and the particle size. The obtained expression of the partition coefficient k (Equation (9)) indicates a linear relation between k and the ratio pap/D2, which is quantitatively verified using data from the published literature. The work herein introduces a new way to study attachment and distribution of indicator bacteria to sediments at a single-cell resolution by combining bacterial tracking methods and microfluidic devices. Both physical and biochemical processes influence bacterial attachment to sediments. However, as shown in this work, we can separate and study the factors influencing the different stages of bacterial attachment individually. We hope this study will inspire additional work in the future to provide further details at the microscopic level for cell-particle interactions and to develop a comprehensive ecological model that could account for both attachment and detachment of E. coli on sediments, which will help to improve control over the contamination level of surface waters.