Informally Vended Sachet Water: Handling Practices and Microbial Water Quality
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Ethical Approval
2.3. Sampling Procedure
2.4. Laboratory Analyses
2.5. Quality Control
2.6. Data Analyses
3. Results
3.1. Demographics
3.2. Microbiological Water Quality
- Conformity: <1 CFU/100 mL
- Low: 1–10 CFU/100 mL
- Intermediate: 11–100 CFU/100 mL; and
- High: >100 CFU/100 mL
3.3. Bivariate Analysis
3.4. Regression Analysis
3.5. Quality Control
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fisher, M.B.; Williams, A.R.; Jalloh, M.F.; Saquee, G.; Bain, R.E.S.; Bartram, J.K. Microbiological and chemical quality of packaged sachet water and household stored drinking water in freetown, Sierra Leone. PLoS ONE 2015, 10, e0131772. [Google Scholar] [CrossRef] [PubMed]
- Stoler, J.; Weeks, J.R.; Fink, G. Sachet drinking water in Ghana’s Accra—Tema metropolitan area: Past, present, and future. J. Water Sanit. Hyg. Dev. J. Int. Water Assoc. 2012, 4, 223–240. [Google Scholar] [CrossRef]
- Monney, I.; Buamah, R.; Odai, S.N.; Awuah, E.; Nyenje, P.M. Evaluating access to potable water and basic sanitation in Ghana’s largest urban slum community: Old fadama, Accra. J. Environ. Earth Sci. 2013, 3, 72–79. [Google Scholar]
- Yidana, N.; Osman, I.; Yidana, Z.; Nyeadi, J.D.; Atogenzoya, C.A. Assessing the compliance of sachet water producers to regulatory standards in Ghana: A study of the tamale metropolis. Int. J. Innol. Res. Dev. 2014, 3, 426–436. [Google Scholar]
- WHO and UNICEF. Progress on drinking water, sanitation and hygiene. In Joint Monitoring Programme; WHO and UNICEF: New York, NY, USA, 2017. [Google Scholar]
- World Health Organization. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011; 541p. [Google Scholar]
- Stoler, J.; Weeks, J.R.; Appiah Otoo, R. Drinking water in transition: A multilevel cross-sectional analysis of sachet water consumption in Accra. PLoS ONE 2013, 8, e67257. [Google Scholar] [CrossRef] [PubMed]
- MacArthur, R.L.; Darkwa, S. Production and vendor practices that compromise the quality of “sachet” water in the central region, Ghana. Int. J. Sci. Technol. Soc. 2013, 1, 64–70. [Google Scholar]
- Osei, A.S.; Newman, M.J.; Mingle, J.A.A.; Ayeh-Kumi, P.F.; Kwasi, M.O. Microbiological quality of packaged water sold in Accra, Ghana. Food Cont. 2013, 31, 172–175. [Google Scholar] [CrossRef]
- Oluwafemi, F.; Oluwole, M.E. Microbiological examination of sachet water due to a cholera outbreak in Ibadan, Nigeria. Open J. Med. Microbiol. 2012, 2, 115. [Google Scholar] [CrossRef]
- Wright, J.; Gundry, S.; Conroy, R. Household drinking water in developing countries: A systematic review of microbiological contamination between source and point-of-use. Trop. Med. Int. Health TM IH 2004, 9, 106–117. [Google Scholar] [CrossRef]
- Rane, S. Street vended food in developing world: Hazard analyses. Indian J. Microbiol. 2011, 51, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Igbeneghu, O.A.; Lamikanra, A. The bacteriological quality of different brands of bottled water available to consumers in Ile-Ife, south-western Nigeria. BMC Res. Notes 2014, 7, 859. [Google Scholar] [CrossRef] [PubMed]
- Stoler, J.; Tutu, R.A.; Ahmed, H.; Frimpong, L.A.; Bello, M. Sachet water quality and brand reputation in two low-income urban communities in greater Accra, Ghana. Am. J. Trop. Med. Hyg. 2014, 90, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.D.; Sreenivasan, N.; Lam, E.; Ayers, T.; Kargbo, D.; Dafae, F.; Jambai, A.; Alemu, W.; Kamara, A.; Islam, M.S.; et al. Cholera epidemic associated with consumption of unsafe drinking water and street-vended water—Eastern Freetown, Sierra Leone, 2012. Am. J. Trop. Med. Hyg. 2014, 90, 518–523. [Google Scholar] [CrossRef]
- Dzotsi, E.K.; Dongdem, A.Z.; Boateng, G.; Antwi, L. Surveillance of bacterial pathogens of diarrhoea in two selected sub metros within the Accra metropolis. Ghana Med. J. 2016, 49, 65–71. [Google Scholar] [CrossRef]
- Brown, J.; Cavill, S.; Cumming, O.; Jeandron, A. Water, sanitation, and hygiene in emergencies: Summary review and recommendations for further research. Waterlines 2012, 31, 11–29. [Google Scholar] [CrossRef]
- Beaumont, P. The Guardian. Available online: https://www.theguardian.com/global-development/2019/mar/27/cyclone-idai-first-cases-of-cholera-confirmed-beira-mozambique (accessed on 27 March 2019).
- Pickering, A.J.; Davis, J.; Walters, S.P.; Horak, H.M.; Keymer, D.P.; Mushi, D.; Strickfaden, R.; Chynoweth, J.S.; Liu, J.; Blum, A.; et al. Hands, water, and health: Fecal contamination in Tanzanian communities with improved, non-networked water supplies. Environ. Sci. Technol. 2010, 44, 3267–3272. [Google Scholar] [CrossRef] [PubMed]
- Pickering, A.J.; Julian, T.R.; Mamuya, S.; Boehm, A.B.; Davis, J. Bacterial hand contamination among Tanzanian mothers varies temporally and following household activities. Trop. Med. Int. Health 2011, 16, 233–239. [Google Scholar] [CrossRef]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; AlMazroa, M.A.; Amann, M.; Anderson, H.R.; Andrews, K.G.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Kumpel, E.; Nelson, K.L. Mechanisms affecting water quality in an intermittent piped water supply. Environ. Sci. Technol. 2014, 48, 2766–2775. [Google Scholar] [CrossRef]
- Overbo, A.; Williams, A.R.; Evans, B.; Hunter, P.R.; Bartram, J. On-plot drinking water supplies and health: A systematic review. Int. J. Hyg. Environ. Health 2016, 219, 317–330. [Google Scholar] [CrossRef]
- Brown, J.; Hien, V.T.; McMahan, L.; Jenkins, M.W.; Thie, L.; Liang, K.; Printy, E.; Sobsey, M.D. Relative benefits of on-plot water supply over other “improved” sources in rural Vietnam. Trop. Med. Int. Health 2013, 18, 65–74. [Google Scholar] [CrossRef]
- Copeland, C.C.; Beers, B.B.; Thompson, M.R.; Fitzgerald, R.P.; Barrett, L.J.; Sevilleja, J.E.; Alencar, S.; Lima, A.A.; Guerrant, R.L. Faecal contamination of drinking water in a Brazilian shanty town: Importance of household storage and new human faecal marker testing. J. Water Health 2009, 7, 324–331. [Google Scholar] [CrossRef]
- Sodha, S.V.; Menon, M.; Trivedi, K.; Ati, A.; Figueroa, M.E.; Ainslie, R.; Wannemuehler, K.; Quick, R. Microbiologic effectiveness of boiling and safe water storage in South Sulawesi, Indonesia. J. Water Health 2011, 9, 577–585. [Google Scholar] [CrossRef]
- Brick, T.; Primrose, B.; Chandrasekhar, R.; Roy, S.; Muliyil, J.; Kang, G. Water contamination in urban south India: Household storage practices and their implications for water safety and enteric infections. Int. J. Hyg. Environ. Health 2004, 207, 473–480. [Google Scholar] [CrossRef]
- Abrishami, S.H.; Tall, B.D.; Bruursema, T.J.; Epstein, P.S.; Shah, D.B. Bacterial adherence and viability on cutting board surfaces. J. Food Saf. 1994, 14, 153–172. [Google Scholar] [CrossRef]
- Ak, N.O.; Cliver, D.O.; Kaspar, C.W. Cutting boards of plastic and wood contaminated experimentally with bacteria. J. Food Prot. 1994, 57, 16–22. [Google Scholar] [CrossRef]
- Jessoe, K. Improved Source, Improved Quality? Estimating the Water Quality Gains from Groundwater Expansion in Rural India (Job Market Paper). 2009. Available online: https://www.semanticscholar.org/paper/Improved-Source-%2C-Improved-Quality-Estimating-the-(-Jessoe/39f533664f2feedae40cffb5d92356572d45e07a (accessed on 27 March 2019).
- Onjala, J.; Ndiritu, S.W.; Stage, J. Risk perception, choice of drinking water and water treatment: Evidence from Kenyan towns. J. Water Sanit. Hyg. Dev. 2014, 4, 268–280. [Google Scholar] [CrossRef]
| Sample Location | Sample Size | Description |
|---|---|---|
| Source water | 70 | Source of sachet water as provided by the vendor |
| Point of sale exterior | 76 | Sachet exterior surface was rinsed in sterile water after purchasing |
| Point of sale interior | 76 | Contents of the sachet |
| Variable | Sample Size | Value | |
|---|---|---|---|
| Male | 76 | 36% | |
| Mean (SD) age | 75 | 29 (10) | |
| Education level completed | 76 | ||
| None | 4% | ||
| Primary | 25% | ||
| Secondary | 62% | ||
| Tertiary | 9% | ||
| Responsible for | 76 | ||
| Fetching water | 87% | ||
| Treating water | 13% | ||
| Packaging water | 80% | ||
| Water source | 76 | ||
| Piped to house | 13% | ||
| Piped to yard | 42% | ||
| Piped to neighbor | 4% | ||
| Public tap | 3% | ||
| Public borehole | 37% | ||
| Shallow well | 1% | ||
| Mean (SD) time spent to collect (minutes round trip) | 47 | 69 (58) | |
| Mean (SD) number of trips per day | 55 | 1.8 (0.8) | |
| Water is stored with a lid | 76 | 80% | |
| Water is used directly without storage | 76 | 9% | |
| Water is removed with a cup (not tap) | 76 | 76% | |
| Mean (SD) price per 20 L of water (MK) | 76 | 200 (0) | |
| Water is treated | 76 | ||
| Boiling | 3% | ||
| Chlorine | 18% | ||
| Cloth | 3% | ||
| Sedimentation | 3% | ||
| No treatment | 70% | ||
| Do not know | 3% | ||
| Mean (SD) time spent packaging (minutes) | 71 | 55 (22) | |
| Mean (SD) sachets packaged per day | 76 | 92 (55) | |
| Water is packaged manually | 76 | 100% | |
| Sachets are open by blowing | 67 | 3% | |
| Bags are sealed by tying | 69 | 100% | |
| Sachets are refrigerated at home | 73 | 99% | |
| Sachets are sold from a cooler | 71 | 99% | |
| Coolers contain ice | 74 | 34% | |
| Mean (SD) price per sachet (MK) | 76 | 20 (0) | |
| Mean (SD) sachets sold per day | 76 | 81 (52) | |
| Source Water | Internal | External | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| E. coli | TC | E. coli | TC | E. coli | TC | |||||||
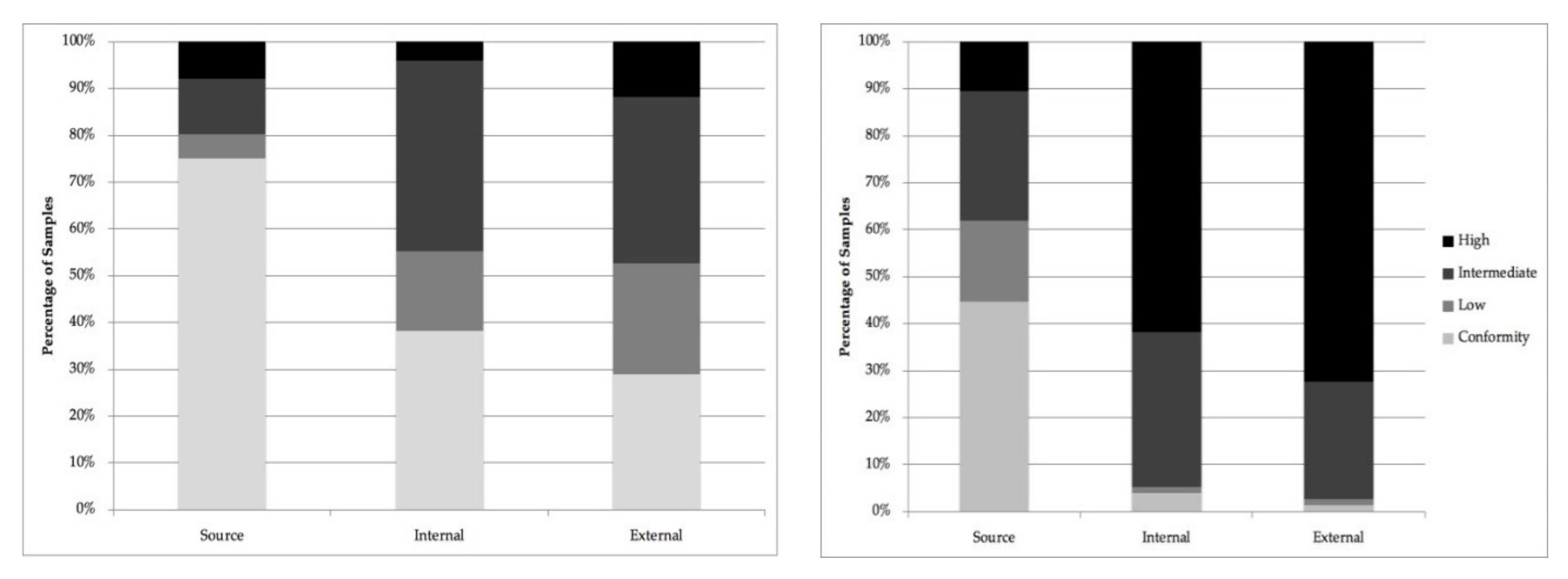
| Conformity * | 75% | (57) | 45% | (34) | 38% | (29) | 4% | (3) | 29% | (22) | 1% | (1) |
| Low | 5% | (4) | 17% | (13) | 17% | (13) | 1% | (1) | 24% | (18) | 1% | (1) |
| Intermediate | 12% | (9) | 28% | (21) | 41% | (31) | 33% | (25) | 36% | (27) | 25% | (19) |
| High | 8% | (6) | 11% | (8) | 4% | (3) | 62% | (47) | 12% | (9) | 72% | (55) |
| Geometric mean | 1.32 | 1.76 | 1.86 | 3.41 | 3.62 | 2.05 | ||||||
| [95% Conf. Interval] | [1.18–1.48] | [1.56–2.00] | [1.65–2.10] | [3.19–3.65] | [3.45–3.80] | [1.83–2.30] | ||||||
| Statistic | Source | Interior | Exterior | |||||
|---|---|---|---|---|---|---|---|---|
| EC | TC | EC | TC | EC | TC | |||
| N = 70 | N = 70 | N = 76 | N = 76 | N = 76 | N = 75 | |||
| Mean | 3 | 16 | 24 | 1000 | 44 | 1562 | ||
| (SD) | (7) | (28) | (38) | (2658) | (98) | (3240) | ||
| Median | 0.5 | 2 | 6 | 145 | 9 | 176 | ||
| Source | Wilcoxon | Z | 4.75 | 7.01 | 5.16 | 7.19 | ||
| p | <0.001 | <0.001 | <0.001 | <0.001 | ||||
| Mann Whitney | U | 1378 | 442 | 1115 | 243 | |||
| p | <0.001 | <0.001 | <0.001 | <0.001 | ||||
| r | 0.461 | 0.728 | 0.543 | 0.791 | ||||
| Spearman rank correlation | rs | 0.038 | 0.184 | −0.115 | 0.115 | |||
| p | 0.754 | 0.127 | 0.345 | 0.348 | ||||
| Interior | Wilcoxon | Z | 2.28 | 2.26 | ||||
| p | 0.023 | 0.024 | ||||||
| Mann Whitney | U | 2502 | 2313 | |||||
| p | 0.149 | 0.046 | ||||||
| r | 0.117 | 0.163 | ||||||
| Spearman’s rank-order correlation | rs | 0.460 | 0.333 | |||||
| p | <0.001 | 0.004 | ||||||
| Variable | log(TCinternal) | TCinternal | log(ECinternal) | ECinternal |
|---|---|---|---|---|
| log(TCsource water) | 0.251 | |||
| log(TCexternal) | 0.100 | |||
| TCsource | −3.600 | |||
| TCexternal | 0.029 | |||
| log(ECsource) | 0.084 | |||
| log(ECexternal) | 0.520 *** | |||
| ECsource | −0.237 | |||
| ECexternal | 0.137 *** | |||
| Gender | 0.272 | 588.816 | −0.489 | 2.365 |
| Does fetching | 0.797 | 3550 * | −0.608 | −18.784 |
| Does treating | 0.682 * | 2620 ** | 0.796 | 15.343 |
| Does packaging | −0.573 | −1354.672 | 1.574 * | 39.315 |
| Improved source | 0.128 | 140.975 | −0.972 ** | −11.316 |
| Water covered | 0.208 | 933.379 | 0.111 | −1.483 |
| Cup for water bucket | −0.095 | −1283.057 | −1.306 ** | −28.879 ** |
| Water treated | −0.362 | −1566.211 | −1.122 ** | −26.844 ** |
| Ice in cooler box | −0.295 | −169.121 | 0.481 | 12.861 |
| Constant | 2.213 ** | 3415.391 | 3.213 ** | 44.026 |
| N | 64 | 64 | 65 | 65 |
| R-squared | 0.228 | 0.267 | 0.445 | 0.338 |
| Adjusted R-squared | −0.013 | 0.038 | 0.276 | 0.135 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manjaya, D.; Tilley, E.; Marks, S.J. Informally Vended Sachet Water: Handling Practices and Microbial Water Quality. Water 2019, 11, 800. https://doi.org/10.3390/w11040800
Manjaya D, Tilley E, Marks SJ. Informally Vended Sachet Water: Handling Practices and Microbial Water Quality. Water. 2019; 11(4):800. https://doi.org/10.3390/w11040800
Chicago/Turabian StyleManjaya, David, Elizabeth Tilley, and Sara J. Marks. 2019. "Informally Vended Sachet Water: Handling Practices and Microbial Water Quality" Water 11, no. 4: 800. https://doi.org/10.3390/w11040800
APA StyleManjaya, D., Tilley, E., & Marks, S. J. (2019). Informally Vended Sachet Water: Handling Practices and Microbial Water Quality. Water, 11(4), 800. https://doi.org/10.3390/w11040800







