1. Introduction
The oceans play a key role in the current climate change scenario. From a physical point of view, they have stored more than 90% of the heat absorbed by the Earth since the mid twentieth century [
1] and deep water formation processes are capable of transferring large amounts of CO
2 to the deep ocean because of the higher solubility of gases in cold winter waters [
2]. From a biological point of view, wind-driven upwelling processes, mixing of the upper water column caused by winter stormy activity, and deep water convection inject nutrients to the photic layer increasing the primary production. A fraction of this new production eventually reaches the deep waters being transformed into CO
2 and nutrients by the action of nitrifying bacteria. In this way, the so called solubility and biological pumps contribute to the sequestration of CO
2 [
3] absorbing about 30% of the human CO
2 emissions during past decades [
4,
5].
The warming of the upper layer of the ocean would increase its thermal stratification reducing the efficiency of the winter mixing and deep convection processes, decreasing the ventilation of the deep layers, the productivity of the sea and the sequestration of CO
2 [
6,
7,
8]. Therefore, it is expected that the oxygen and nutrient distributions and the first steps of the food webs, that is, the phyto and zooplanktonic communities, will be affected by the warming of the oceans. Any change in the phyto and zooplankton communities would finally have an impact on the rest of the marine ecosystems through bottom-up trophic cascade effects [
9].
The Mediterranean Sea is not an exception within the scenario depicted above [
10,
11]. On the contrary, it has been suggested that because of its reduced dimensions and its location, between three continents, with an increasing pressure from the touristic, agriculture and industrial sector, it could be especially vulnerable to climate change as well as to other anthropogenic stressors [
12].
Detecting lasting changes in marine ecosystems requires the long-term monitoring of variables that could be considered as indicators of the environmental state of the sea. Then, average seasonal cycles can be calculated and long term changes for such cycles could be detected. Nevertheless, long-term monitoring programs in the Mediterranean Sea are scarce and data availability for physical, chemical and biological variables is uneven. Temperature and salinity data for the twentieth century are suitable for the detection of long-term trends in the intermediate and deep Mediterranean waters, but are not appropriate for the analysis of the upper layer where the high frequency variability is stronger than that of deeper levels and can mask such long-term changes [
13,
14,
15]. The deployment of autonomous devices operating both in real time or delayed mode, such as profilers within the MedArgo program [
16], or moored conductivity-temperature-depthmonitors (CTDs) in the Hydrochanges program [
17], and the compilation of hydrographic data in several platforms such as Copernicus [
18] or SeaDataNet [
19] have helped to improve the availability of temperature and salinity data for the twenty-first century. Nevertheless, the study of most of the biochemical variables as well as the abundance and taxonomic composition of the phyto and zooplankton communities are based on in situ sampling by means of oceanographic campaigns.
Unfortunately, biological long-term time series in the Mediterranean Sea are scarce. In the particular case of the Western Mediterranean, very few biogeochemical monitoring programs do exist. To our knowledge, some of these few monitoring stations are the LTER (Long Term Ecological Research) Mare Chiara station, in the Bay of Naples [
20,
21,
22], the Dyfamed station, in the Ligurian Sea [
23,
24], the Blanes Bay Microbial Observatory in the Blanes Bay (Catalan Sea coastal waters, [
25]), the PHYTOCLY station in Calvi Bay (Corsica, [
11,
26]) and the zooplankton time series at the Station B at Villefranche [
27,
28]. The time series obtained at these sites have allowed us to improve our understanding of marine ecosystems. They have provided detailed descriptions of the seasonal cycles and trends of temperature and salinity, the dissolved oxygen and nutrient dynamics as well as the changes in the abundances and taxonomic composition of the phyto and zooplankton assemblages [
20,
21,
22,
25,
27]. When dealing with more extended areas, our knowledge about such seasonal dynamics usually comes from the analysis of data gathered in research projects covering almost one seasonal cycle. Typically, the comparison of data from two or at most four oceanographic cruises, covering the different seasons, has allowed us to infer the main traits of biochemical and planktonic cycles in areas such as the Catalan and Balearic Seas [
29,
30,
31] the Alboran Sea [
32,
33], or the whole Mediterranean [
34].
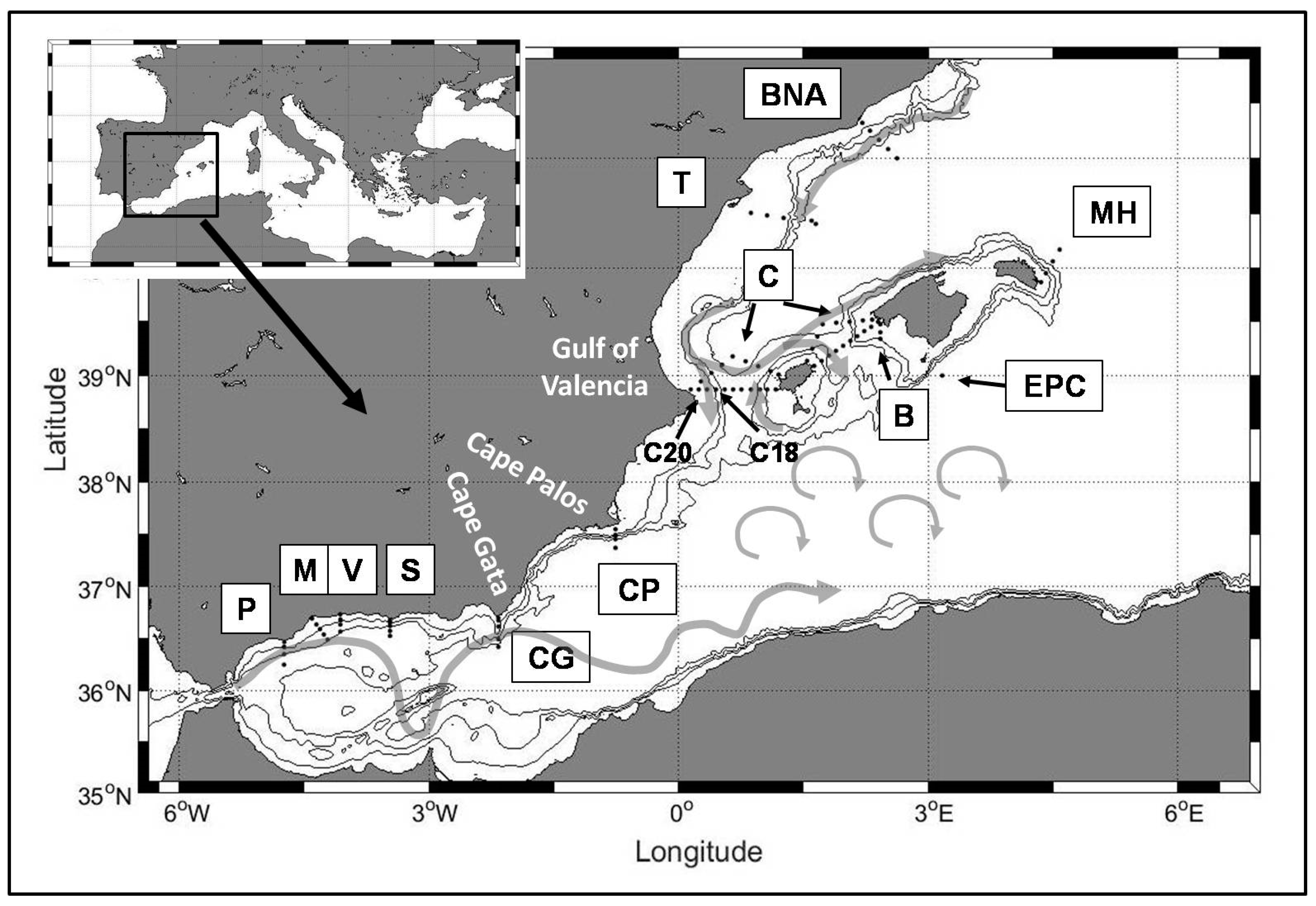
The Instituto Español de Oceanografía (IEO, Spanish Institute for Oceanography) initiated multidisciplinary monitoring programs in 1992, 1994 and 1996 in the areas of Malaga Bay, at the northwestern sector of the Alboran Sea (ECOMALAGA project), to the south of Cape Palos (ECOMURCIA project), to the south of Mallorca Island (ECOBALEARES project) and in the Ibiza and Mallorca Channels (CIRBAL project, Circulación Regional en las aguas de las Islas BAleares). These programs have been unified and extended to the Catalan Sea in 2007 under the umbrella of the new monitoring program RADMED, Series Temporales de Datos Oceanográficos en el Mediterráneo (
Figure 1 [
35]). Previous works have shown the capability of RADMED for providing a description of the seasonal dynamics of the physical properties of the water masses around the Spanish continental shelf and slope and to detect long term trends in the intermediate and deep waters of the Western Mediterranean [
13,
15]. Previous works have grouped the RADMED time series by seasons and estimated the mean values corresponding to each season for the concentrations of dissolved oxygen, nutrients and chlorophyll [
36]. Besides this description of the average seasonal cycles, these authors analyzed the existence of decadal changes since the mid-1990s.
The goal of the present work is to provide baseline data to support water quality and aquatic living resource characterization of the Spanish Mediterranean ecosystem. The datasets can further support development and validation of ecological and management-decision models of the ecosystem.For this purpose, phyto and zooplankton distributions were analyzed as follows:
- (i)
Seasonal cycles were analyzed for the micro-phytoplankton (>20 µm) abundances on the Spanish continental shelf from Malaga, in the westernmost sector of the Alboran Sea, to Barcelona (Catalan Sea), including the Balearic Islands.
- (ii)
The average cycles were estimated for the abundances of the nano (2–20 µm) and pico-eukaryote and pico-prokaryote (0.2–2 µm) phytoplankton on the continental shelf and slope.
- (iii)
Decadal changes in the abundances of micro-phytoplanktonic groups and in the meso-zooplanktonic biomass were studied at the stations where the time series extension made it possible.
- (iv)
The seasonal cycles of meso-zooplanktonic (>250 µm) biomass andabundances of broad meso-zooplanktonic groups were obtained.
In some cases, the time series analyzed will be more than 20 years long. In these cases the mean values which characterize the average seasonal cycles and the standard deviations and ranges of variability (minimum and maximum values recorded) can be considered as robust statistics. For the case of such long time series, decadal changes can also be estimated fitting a straight line by means of least squares fit. At other stations, only a complete seasonal cycle (one data set per season) is available, and results should be considered with caution. In these latter cases the results are presented for the completeness of the work and are discussed in light of previous works also dealing with four cruises distributed along one single year.
Section 2 is a detailed description of the sampling and analysis strategy within the RADMED project. The main results are in
Section 3 and a discussion and conclusions are presented in
Section 4.
2. Materials and Methods
The RADMED project is a monitoring program funded by the IEO covering the Spanish Mediterranean coast and Balearic waters (
Figure 1). As mentioned above, this program was launched in 2007 unifying and extending previous monitoring programs initiated in 1992, 1994 and 1996. The sampling stations are distributed in transects perpendicular to the coast, covering the continental shelf and slope and in some cases including some deep stations (>2000 m). Stations are named by a letter corresponding to each transect and a number increasing from the coast to the open sea. In the Alboran Sea, the westernmost transects are Cape Pino (P in
Figure 1), Málaga (M) and Vélez (V). For instance, the closest station to the coast in Cape Pino transect is named as P1, and the most offshore station is P4. Sacratif transect extends from Cape Sacratif in the central part of the Alboran Sea, and Cape Gata transect (CG) is on its eastern limit. Those transects extending from the eastern Spanish coast are Cape Palos (CP), Tarragona (T) and Barcelona (BNA). Two more transects are located in the Balearic Islands, one of them to the south of Mallorca Island (B) and another one extending in a northeast direction from Mahón, in Menorca Island (MH). Apart from these transects, there are 37 oceanographic stations forming two triangles covering the Balearic channels: the Ibiza Channel, between the Spanish Peninsula and Ibiza Island, and the Mallorca Channel, between Ibiza and Mallorca. These stations are labeled as C. Finally, a deep station (>2200 m) is located to the south of Cabrera Island (EPC). All the stations are visited every three months, that is, once per season, when vessel availability and weather conditions make it possible.
CTD (conductivity-temperature-depth) profiles were obtained in all the stations. The hydrographic sampling was done using CTDs, mainly the model SBE (Sea-Bird Scientific) 911 installed in a carousel water sampler SBE 32. Some profiles were obtained using a CTD of the model SBE 25. CTDs were equipped with a dissolved oxygen (DO) SBE 43 sensor. DO and conductivity sensors were calibrated using water samples from selected stations and depths. Water samples for the determination of nutrients and chlorophyll-
a were taken at all the stations. Water samples for nutrient determinations were taken at 0, 10, 20, 50, 75, 100, 200, 300, 500, 700, 1000 m and sea bottom for deep stations while sampling was limited to the station depth for the shallower ones. Samples for the determination of chlorophyll concentrations were limited to the upper 100 m of the water column. As already commented, 37 oceanographic stations cover the Ibiza and Mallorca Channels. Such stations are distributed forming two triangles. Stations C20 and C18 within the Ibiza Channel triangle (see
Figure 1), were chosen as representative of the peninsular continental shelf and slope conditions respectively (see
Figure 1). Details concerning nutrients and chlorophyll sampling can be found in previous works [
35,
36].
Additionally to the standard sampling at each station (temperature, salinity, Dissolved Oxygen, chlorophyll-
a, and nutrient concentrations), water samples for the determination of the micro-phytoplankton abundance were taken at the second station of each transect, e.g., P2, M2, V2, etc., located on the continental shelf. Samples for the determination of nano and picoeukaryote plankton abundance and prokaryote pico-phytoplankton abundance (
Prochlorococcus and
Synechococcus cyanobacteria) were taken at the second (continental shelf) and fourth (continental slope) stations of each transect (e.g., P2, P4, M2, M4, etc.). Water samples for micro-phytoplankton studies were obtained with 5L Niskin bottles from the sea surface to 100 m at the same discrete depths as chlorophyll and nutrient samples. Micro-phytoplankton samples were preserved in 150 mL bottles and fixed with 2 mL of acid Lugol solution and stored in darkness at ambient temperature until they were analyzed. Cell counts were performed using an inverted microscope after sedimentation of variable volumes of seawater (25–100 mL), depending on cell concentration [
37]. First, cells from subsamples were counted over the whole bottom of the sedimentation chamber scanned at 50× magnification. Secondly, on two transects of the whole bottom area of the sedimentation chamber at 100× magnification, and finally, on one transect of the whole bottom area of the sedimentation chamber at 200× magnification. Determinations were done to genera level in most of the cases and to species level when possible. Nevertheless, considering the long-term objective of this project and that this task has a great time and personnel demand, time series were finally constructed for main groups. Diatoms, dinoflagellates, small flagellates and ciliates are well preserved within Lugol solutions. By contrast, coccolithophorids suffer a rapid dissolution. Good preservation of coccolithophorid cells has been obtained with very weak formaldehyde solutions buffered with hexamethylenetetramine [
38,
39]. This means having to store twice the volume of samples and double the examination work, which cannot be achieved due to the lack of space and qualified personnel during the campaigns and at the laboratory. For these reasons, the final time series in RADMED project were those of abundances of diatoms, dinoflagellates and small flagellates.
Pico and nano-phytoplankton samples were preserved in 5 mL cryotubes fixed with 200 µL of 50% glutaraldehyde solution and frozen at −80 °C until they were analyzed using a FACSCalibur (Becton Dickinson) flow cytometer. The pico-phytoplankton (both eukaryote and prokaryote) and nano-phytoeukaryote groups were described and enumerated according to their specific auto-fluorescence properties and side scatter differences [
40].
Zooplankton samples were obtained at the second and fourth stations of each transect. Oblique hauls were carried out between the bottom (for stations <100 m depth) or 100 m depth (for stations >100 m depth) and the sea surface at a vessel speed of two knots with a Bongo-20 Plankton net fitted with 250 µm to sample meso-zooplankton. A general Oceanics flowmeter was fitted to the net to estimate the distance traveled during the oblique haul. Then the volume of filtered water was calculated using the net diameter and distance.
Immediately after collection, the zooplankton samples were split in two subsamples for biomass and taxonomic analysis with a Folsom plankton splitter. Subsamples for taxonomic studies were preserved with 4% neutralized formaldehyde buffered with hexamethylenetetramine. Representative aliquots for taxonomic analysis were analyzed and the organisms identified to the level of the main taxonomic groups: copepods, appendicularians, cladocerans, doliolids, chaetognaths, siphonofores, ostracods and scyphozoans. Abundance was calculated as individuals/m
3. Subsamples for biomass were frozen (−20 °C) for subsequent estimation of the biomass as dry weight [
41,
42]. Samples for biomass measurements were concentrated on Whatman GF/C filters. The mass of the filters had been previously recorded. These filters were then dried in a drying oven at 60 °C for about 24 h and stored in a desiccator for approximately 1 h. Then biomass values were estimated by subtracting the initial mass of the filter from the final mass. Biomass was calculated as mg/m
3.
Statistical Analyses
The main objective of the RADMED project is to estimate basic statistics such as average seasonal values, ranges of variability and long-term trends of variablesthat could be used as indicators of possible natural and/or anthropogenic alterations, and help to a better understanding of the physical and biochemical dynamics of the Mediterranean waters. Such variables should satisfy two main requirements. The first one is that they should provide some insight about the environmental state of the Spanish Mediterranean waters, and second, that they should be easily monitored with a long-term perspective, maximizing data acquisition while optimizing the economic efficiency of the monitoring program. The present work analyzes all the time series collected for micro-phytoplankton, nano and picoeukaryote cell abundances and prokaryote pico-phytoplankton cells (Synechococcus and Prochlorococcus cyanobacteria) from the sea surface to 100 m depth. For each depth, mean values were estimated for each season. Standard deviations and the minimum and maximum values recorded were also obtained for characterizing the time variability associated to each variable and season. Once the seasonal cycle was estimated, it was subtracted to the original data and time series of residuals or deviation were obtained. Linear trends were estimated by least squares fits and confidence intervals at the 5% significance level (95% confidence level) were calculated using a t-student distribution for the slope after checking for the normality of the residuals. The same analyses, mean values, standard deviation and range of variability were accomplished for the abundances of the main meso-zooplanktonic groups and biomass. In this case it was not possible to determine the vertical distribution of abundances and biomasses as the data proceeded from oblique hauls from 100 m to the sea surface.
A monitoring program such as RADMED faces different difficulties such as weather conditions which make the sampling impossible in some cases, instrument failures, or simply the lack of vessel availability. In other cases there are difficulties to have the needed personnel for the analysis of a huge number of samples. Besides this, the RADMED program was initiated in 2007, unifying some previous more local monitoring projects. Therefore, data previous to, and after 2007 have frequent gaps and different temporal coverage depending on the station considered.
Table 1 shows this coverage for each variable and station. Graphical representations of the seasonal cycles for phytoplankton, zooplankton and prokaryote pico-phytoplankton are presented when at least one seasonal cycle is available. Nevertheless, detailed tables presenting robust statistics that could be considered as the most likely seasonal dynamics will be presented only when the data availability allow such calculations. Linear trends for the detection of possible long-term changes will only be presented for those stations initiated in the 1990s.
4. Discussion and Conclusions
According to the general picture usually accepted for the Mediterranean, it is an oligotrophic sea [
44,
45] where regenerated production prevails over the new one [
24,
46]. Intense cold and dry winds during winter produce the mixing of the upper water column supplying nutrients to the photic layer and producing a homogeneous vertical distribution of nutrients and phytoplankton abundance [
47,
48]. Our long-term seasonal patterns show that diatom abundances increase during winter or spring and this phytoplankton bloom is followed by a zooplankton bloom in spring-summer (highest zooplankton values in
Figure 12 are observed in summer), which would agree with the increase of zooplanktonic abundances during summer previously reported [
49]. In winter, maximum phytoplankton abundances can occur in the surface layer. As the spring advances, a seasonal thermo cline is developed, inhibiting the nutrient supply to the photic layer and nutrients are depleted by phytoplankton uptake [
33] which in turn is grazed by zooplankton. During this stratified period, from mid-spring to late autumn, the phytoplankton is dominated by the picoplanktonic cells [
50]. A deep chlorophyll maximum is developed and the depth of this maximum, which usually corresponds to the highest phytoplankton abundances, deepens from spring to autumn [
36]. The zooplankton abundance also experiences a succession of groups. Copepods are the most abundant group in winter and spring, while cladocerans experience an important increase in summer becoming, in some cases, the most important group for the summer season.
Nevertheless, many works have shown that this general patternmay undergoimportant modifications as several mechanisms can fertilize the upper layer of the sea, altering the oligotrophy of the Mediterranean waters and creating mesotrophic conditions [
48,
51]. Other works have shown that differentiated regions can be established in the Mediterranean Sea according to the concentrations of chlorophyll at the sea surface and along the water column,and the timing of the phytoplanktonic bloom [
52,
53]. Some of these differences have been evidenced within the RADMED area by means of the use of long time series of phyto and zooplankton data. One of the mechanisms that can inject nutrients to the photic layer, increasing the primary production and reducing the oligotrophy of the Mediterranean waters, is the existence of frontal systems and eddies [
51,
54,
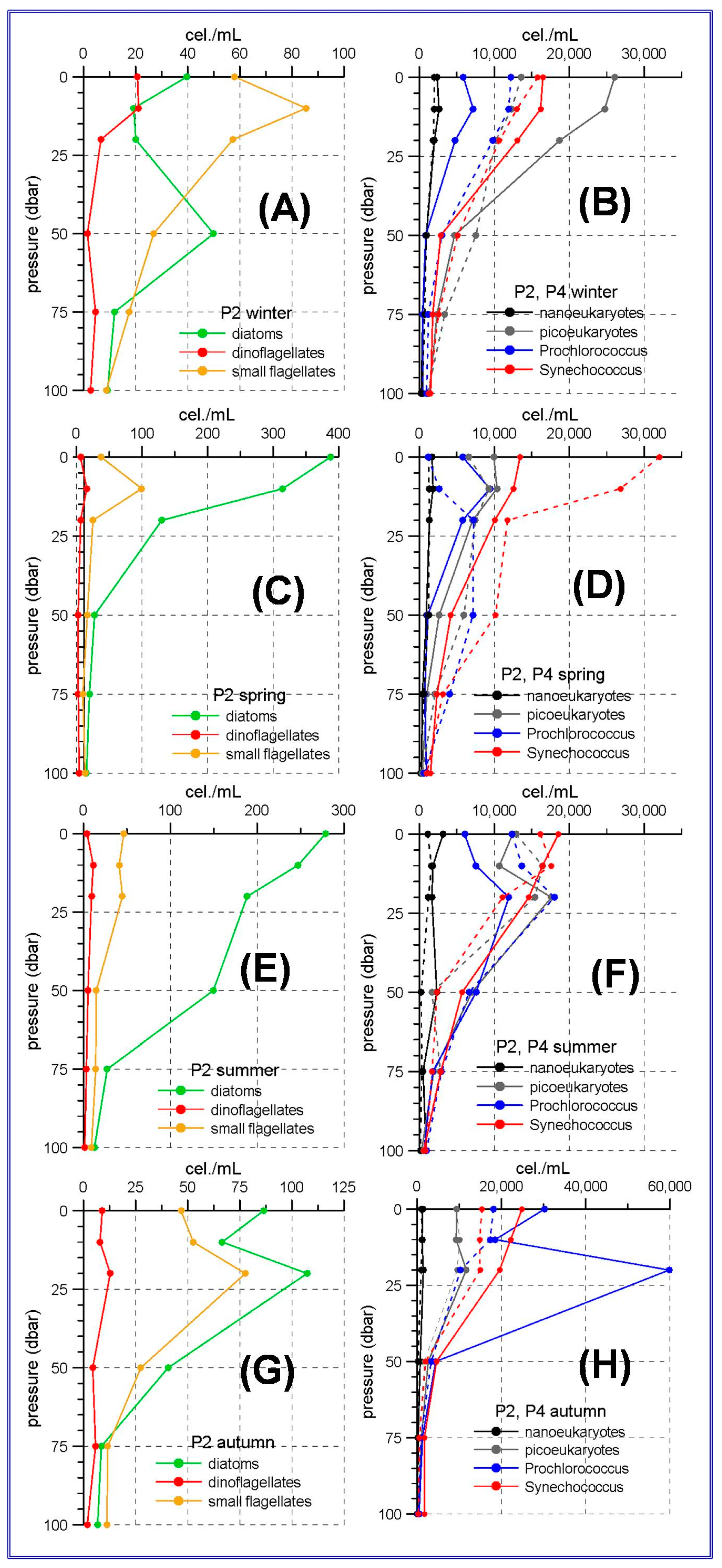
55]. The results of the present work show that diatoms are the most abundant group in the phytoplanktonic community in the westernmost stations of the Alboran Sea, not only in winter or spring, but throughout the whole year. From spring to autumn the maximum diatom abundances are observed at the sea surface or at the upper 20 m. It is also remarkable that summer abundances in the station P2 (
Figure 2E) are higher than those corresponding to the winter or spring abundances in other regions of the RADMED area. As the highest abundances in the Western Alboran Sea occur in spring, this suggests that one of the causes for this phytoplankton bloom is the winter mixing, as in other areas of the Mediterranean Sea, and the spring prevalence of westerly winds which can induce wind-driven upwelling events in the northern coast of the Alboran Sea [
56]. The persistence of high abundances of diatoms for most of the year suggests that other fertilizing mechanisms exist in this part of the Mediterranean. First, it has been hypothesized that the strong mixing in the Gibraltar Strait is a mechanism that acts throughout the year supplying nutrients to the upper layer of the sea [
57,
58]. These nutrient-rich waters would favor the phytoplankton growth at the northeastern side of the Strait and at the northwestern Alboran Sea. The high biomass produced by this mechanism would be advected by the Atlantic jet around the Western Alboran Gyre [
59,
60] affecting the transect P in the RADMED network. Besides this, two cyclonic circulation areas have frequently been described in the area between Gibraltar and Punta de Calaburras (to the west of transect P), and in front of Malaga Bay, affecting transects M and V, [
54,
61,
62]. All these circulation traits do not seem to depend on the season and constitute permanent mechanisms for the fertilization of the photic layer, acting along the whole year. They would explain the high phytoplanktonic biomass in this sector of the Spanish Mediterranean with a weak seasonality if compared to other regions monitored in the RADMED project.
Concerning the nano and picoeukaryotes and the prokaryote pico-phytoplankton in the Alboran Sea, it is observed that
Synechococcus abundances are maxima at the sea surface, decreasing with depth (
Figure 2B,D,H). The only exception is the continental slope station P4 in summer, when the maximum abundances occur at 10 m depth. This behavior has frequently been observed, indicating the preference of this group for well illuminated waters and its adaptation to nutrient-poor waters [
23,
63,
64]. By contrast,
Prochlorococcus usually develop a deep maximum reflecting a better adaptation to low light intensities [
63,
64] permitting
Prochlorococcus to take advantage, growing on deep nutriclines in stratified oligotrophic waters, not accessible for
Synechococcus. Nevertheless, the observed
Prochlorococcus maxima at 10 m depth in winter and spring and at 20 m in summer and autumn, suggest the existence of a shallow nutricline and a high nutrient availability in the uppermost layerof this part of the Alboran Sea, even during the stratified period.
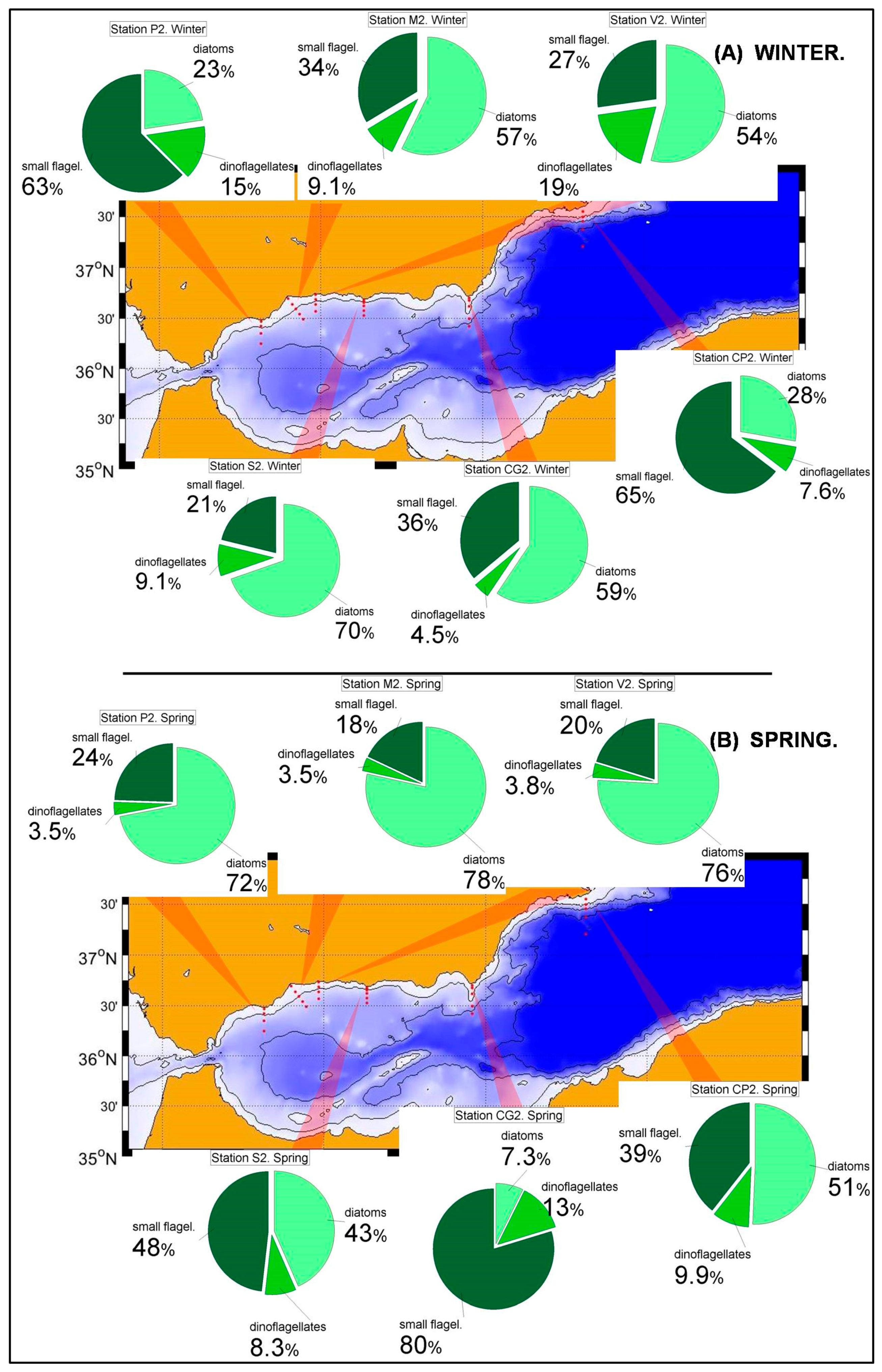
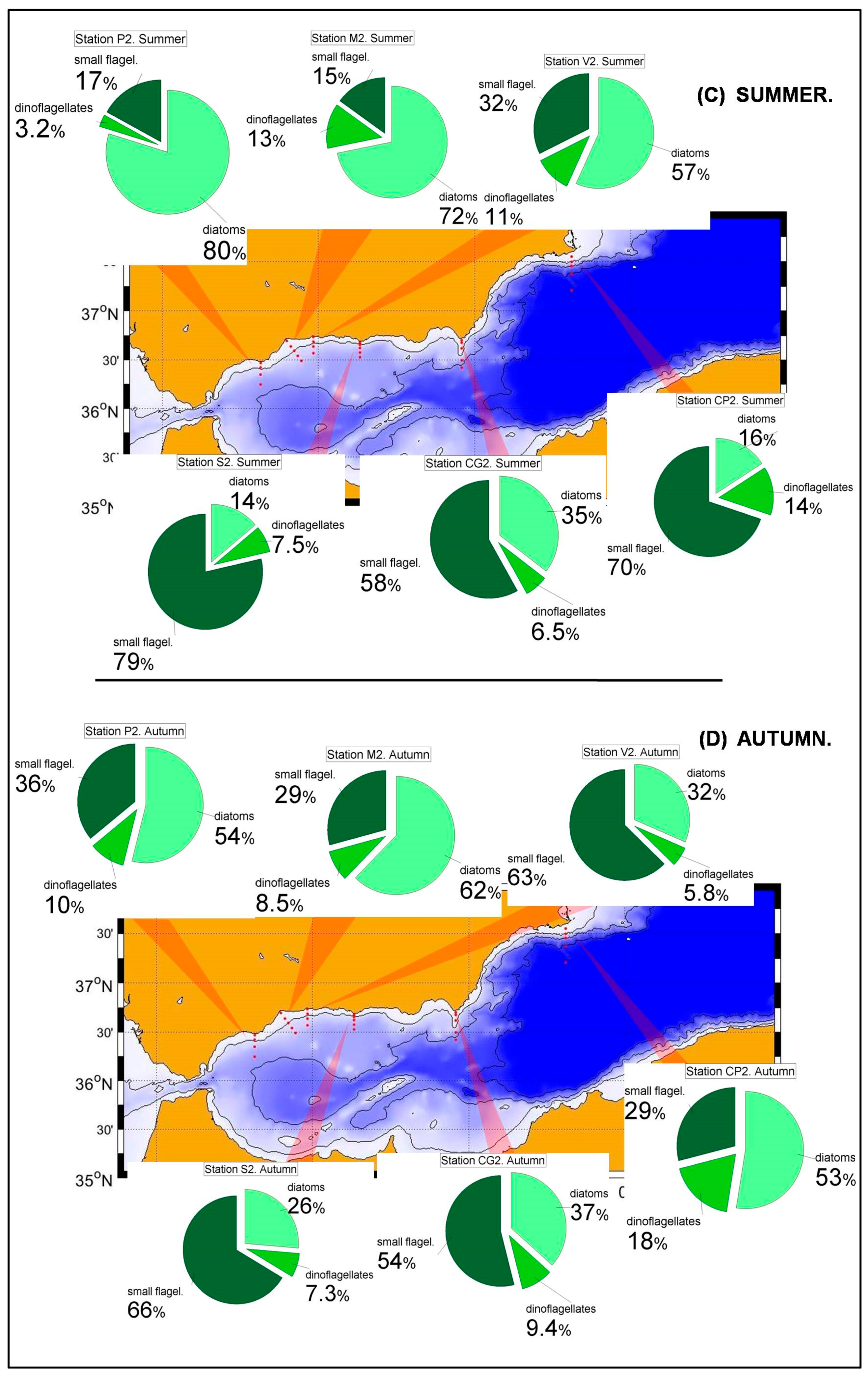
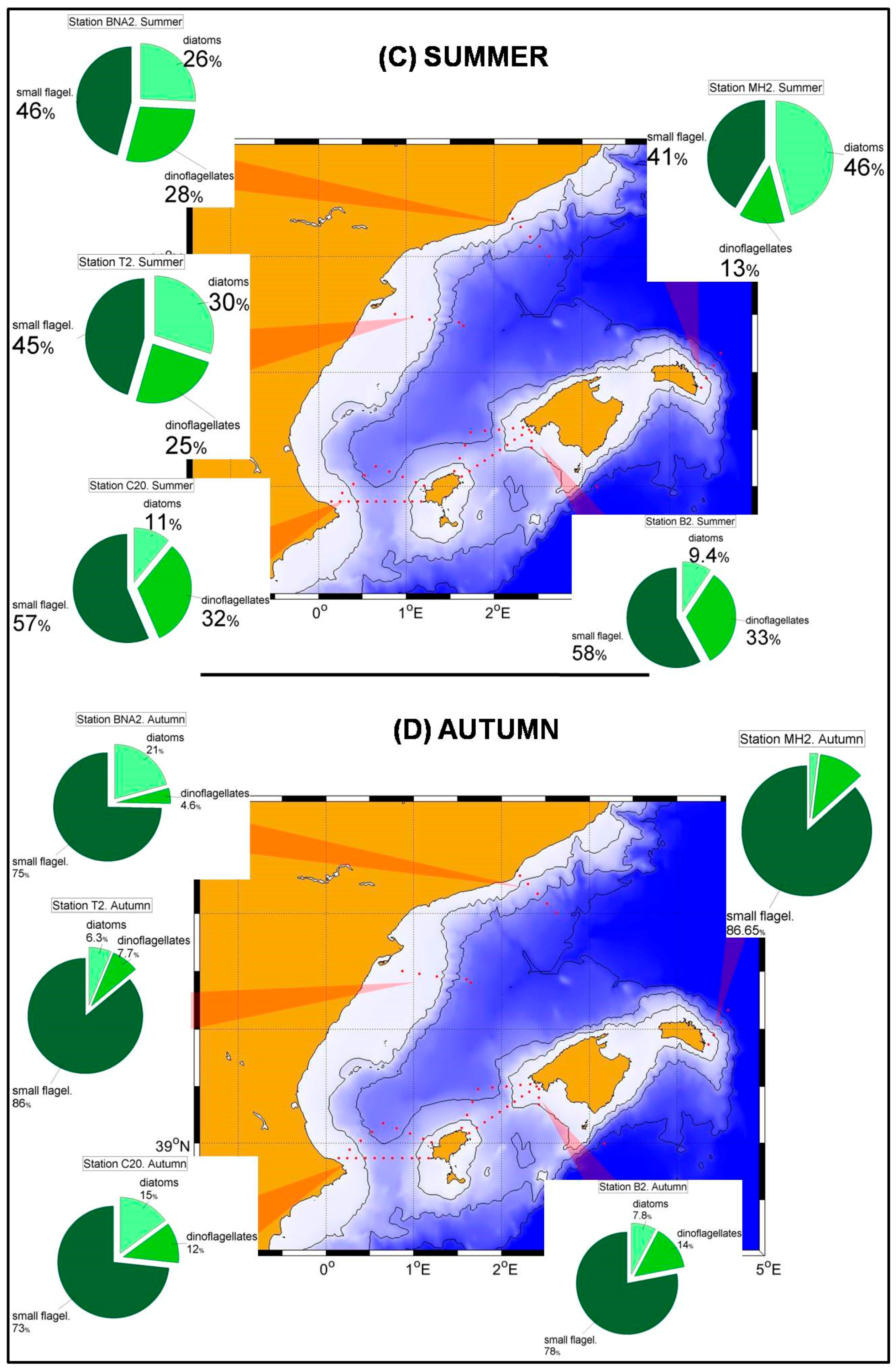
Nutrient and chlorophyll distributions show the existence of a southwest-northeast gradient in the trophic conditions of the Spanish Mediterranean waters [
36]. This is reflected in the phytoplanktonic distributions within the RADMED area. The prevalence of diatoms decreases eastwards within the Alboran Sea and to the north of Cape Gata, while the small flagellates are the most abundant group to the East of Cape Sacratiffor most of the year. Diatoms were the most abundant group in the Sacratif and Cape Gata transects only in winter, reflecting that the winter mixing is the main fertilizing mechanism in this area and the existence of a strong seasonality.
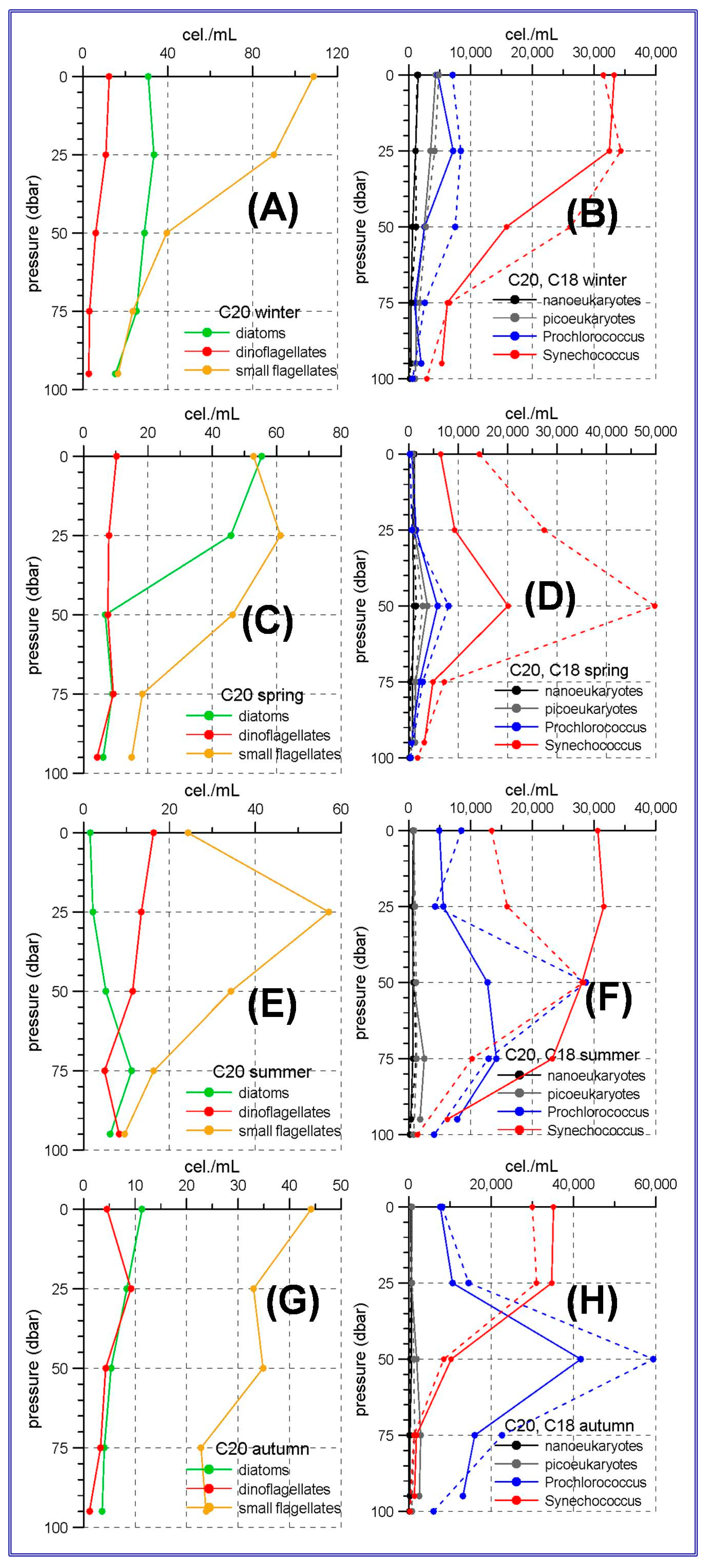
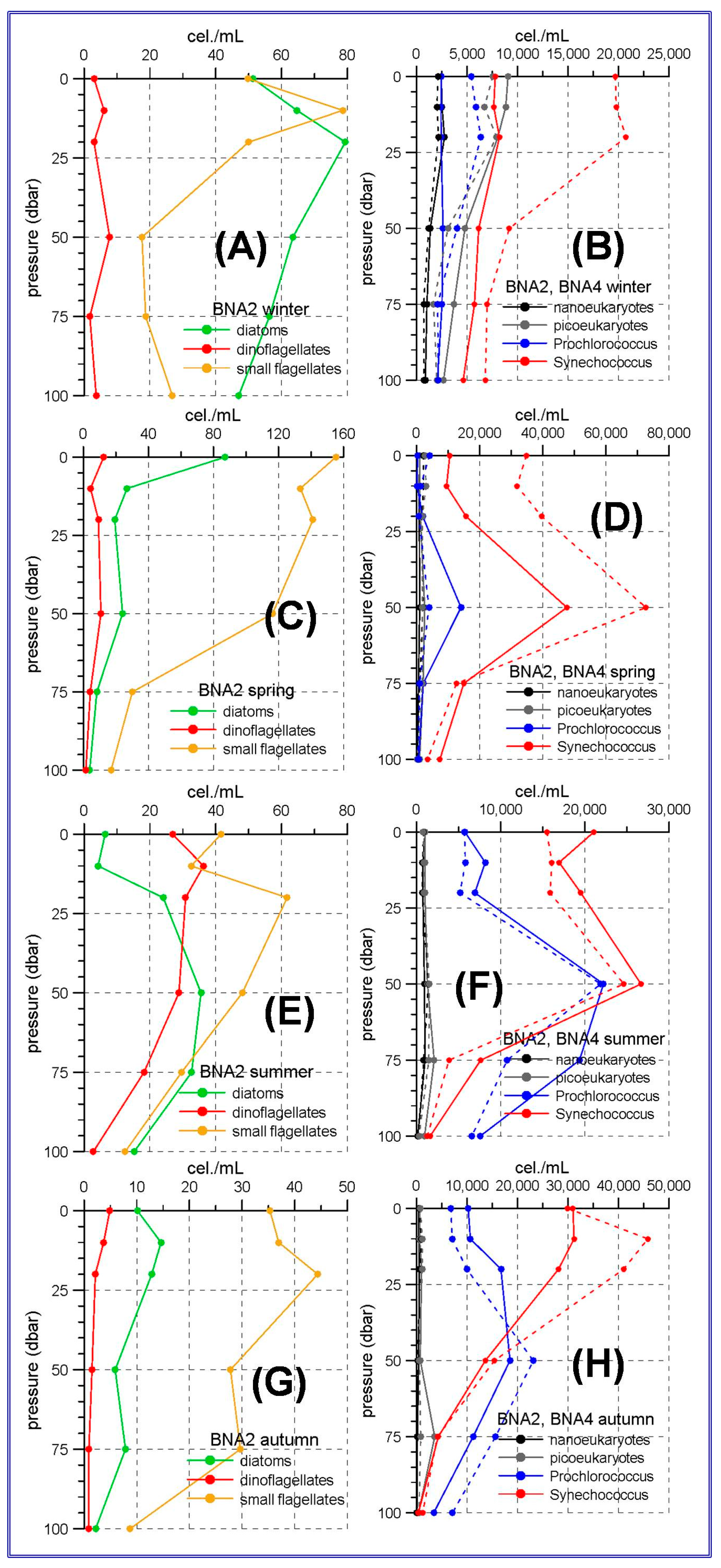
The aforementioned trophic gradient is accentuated to the north. The peninsular side of the Ibiza Channel and the transect to the south of Mallorca Island showed much lower micro-phytoplankton abundances (
Figure 3 and
Figure 4A,C,E,G) than those observed in the Alboran Sea. The small flagellates group was the most abundant in both sites. In the station C20, within the Ibiza Channel, the diatoms experienced an increase in spring when this group became the most abundant one only at the sea surface.
Synechococcus showed maximum abundances at the sea surface or at the upper 25 m with the only exception of spring at stations C20 and C18 and summer at station C18 (
Figure 3D,F), when the maxima were located at 50 m depth.
Prochlorococcus showed deep maxima in all the cases, being these maxima at 25 m during winter, and deepening to 50 and even 75 m during spring, summer and autumn.
Figure 3 and
Figure 4 show that nano and picoeukaryotes abundances (≈10
3 cel./mL) are an order of magnitude lower than those corresponding to the prokaryote group (≈10
4 cel./mL). This is another difference with the Alboran Sea where the picoeukaryotes have abundances comparable to those of the prokaryotes for some stations and seasons (
Figure 2D,F,H).
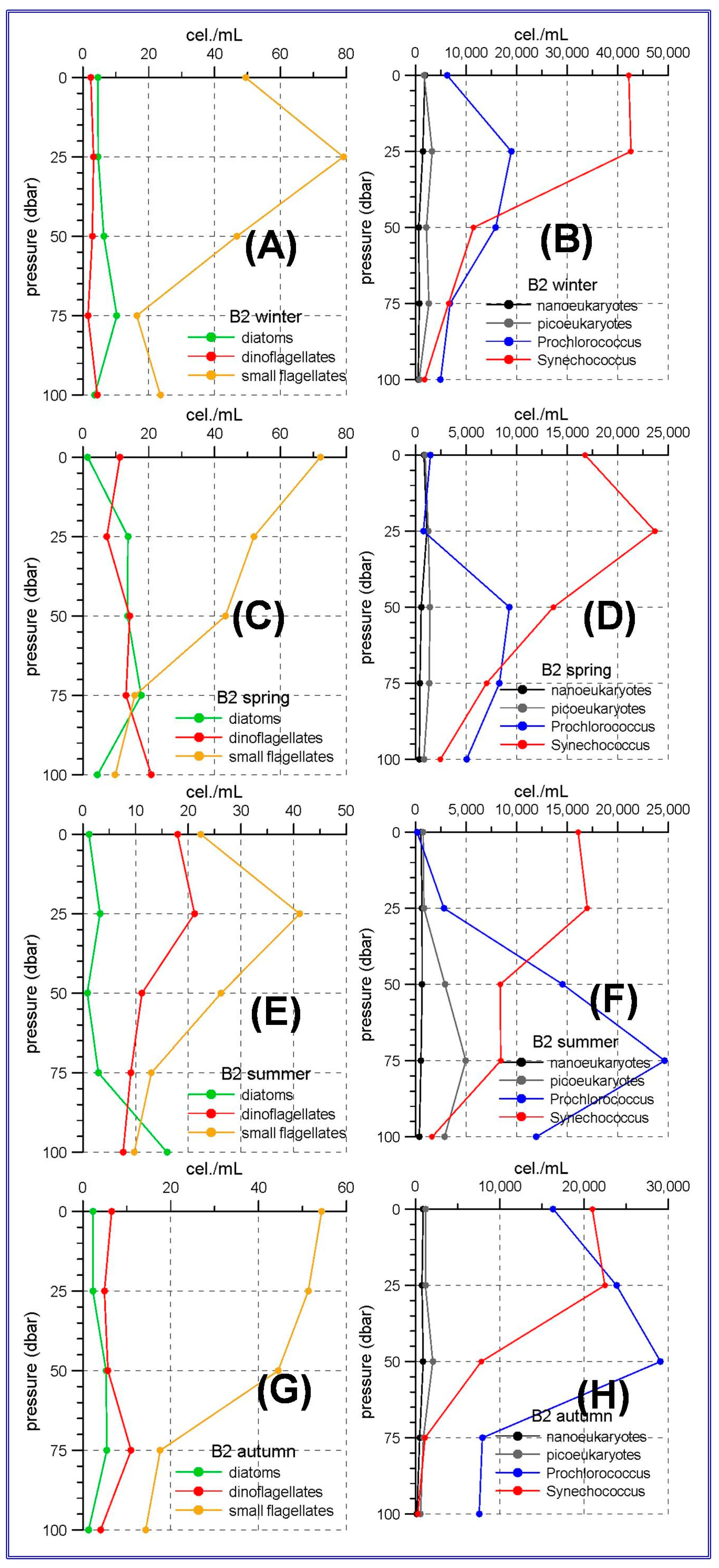
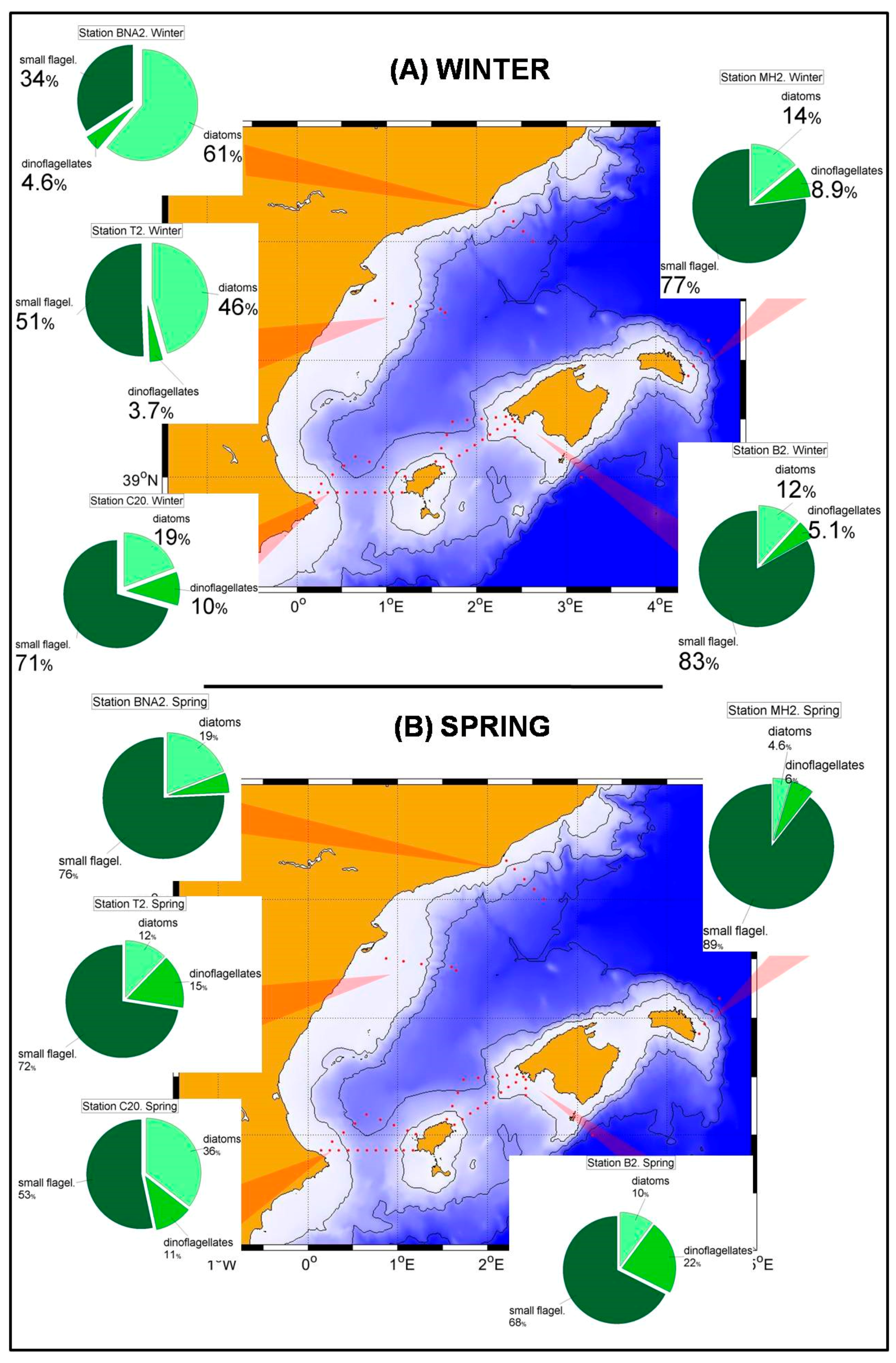
In the northern sector of the RADMED area the conditions are similar to those observed in the Balearic Sea, although some differences can be outlined. Diatoms became the most abundant group in the micro-phytoplankton community in winter (
Figure 5A), being the maximum values at 20 m depth. Although small flagellates were the main group in spring, high diatom abundances were observed at the sea surface (>80 cel./mL), and a deep maximum was developed between 50 and 75 m in summer (
Figure 5B,C). If vertically integrated micro-phytoplankton abundances were considered, the dominance of diatoms was observed in the Barcelona transect during winter, and high diatom abundances were observed at Tarragona transect, also located in the Catalan Sea and to the south of the Barcelona transect. In all the other stations and seasons, the small flagellates were the most abundant group, with the only exception of autumn in station MH2 (northeast of Menorca Island). These patterns described above suggest that the main fertilizing mechanism easts of Cape of Gata is the winter mixing which is responsible for the increase of microplankton abundances. Nevertheless, there are also important differences within this area. It is well known that the Gulf of Lions and adjacent waters experience very strong vertical convection and a violent mixing of the water column in late winter [
65]. These processes inject large amounts of nutrients to the photic layer enhancing primary production at late winter–early spring [
47,
48]. The intensity of winter winds and convection decreases southwards from the Gulf of Lions towards the Catalan Sea. This seems to be the cause for the higher winter micro-phytoplankton abundances at the Barcelona transect, where diatoms became the main group, and also of the importance of this group in Tarragona transect. As pointed out by [
29] nanopankton seems to be less variable. Thus, the main differences between the different geographical areas are associated to the microplankton size fraction. Using satellite data, [
52] classified the NW Mediterranean as a bloom site, that is, an area where an intense phytoplanktonic bloom occurs after severe winter mixing. The long-term time series of the RAMED project provides statistically robust data of the whole photic layer confirming that the Barcelona transect and, to a lesser extent the Tarragona one, belong to this category established by the cited authors.
The results from the statistical analysis presented in this work suggest that there are three main areas in the Spanish Mediterranean waters.First, the most productive waters located in the western part of the Alboran Sea where primary production and the presence of large cells such as diatoms are influenced by winter mixing, but also by other fertilizing processes linked to the dynamics of the Strait of Gibraltar [
57,
58] and the cyclonic circulation and frontal areas [
32,
33,
66,
67]. The consequence is a high phytoplanktonic biomass throughout the year. These high biomass values are also located at the very surface waters, indicating the nutrient supply to surface or sub-surface depths. Second, the poorest waters are those located to the south of the Balearic Islands and Cape Palos where small flagellates dominate during all the seasons. Third, the Catalan Sea, where the described oligotrophy could be partially “relaxed” by strong wind episodes, being winter mixing stronger than at the Balearic Sea.
Figure S1 in Supplementary Materials offers a schematic description of these traits. In order to show more clearly the trophic gradient between the different RADMED regions, this figure is completed with nutrient and chlorophyll data from [
36].
It is also important to mention that another factor able to fertilize the upper part of the water column within the Catalan-Balearic Sea is the frontal zone associated to the northern current flowing southward along the Catalan continental slope and then turning to the east at the northern continental slope of the Balearic Islands (Balearic current, [
68]). The only stations in the RADMED project that could be affected by this circulation feature are those at the transects BNA, T and MH. It is interesting to note that the station MH2 also showed a diatomdominance in summer (
Figure 7C). Finally, dinoflagellates showed low abundances throughout the year with a summer increase in the stations of the Catalan-Balearic Sea in summer. This behavior had already been described and attributed to the ability of this group to adapt to low nutrient conditions because of the mixotrophy and mobility of numerous species within this group [
69] allowing the migration between the lower limit of the photic layer and a deeper nutricline in summer. This summer increment in the relative importance of dinoflagellates is not observed in the Alboran Sea. We hypothesize that once again this is the result of the higher nutrient availability throughout the year where diatoms take advantage over dinoflagellates.
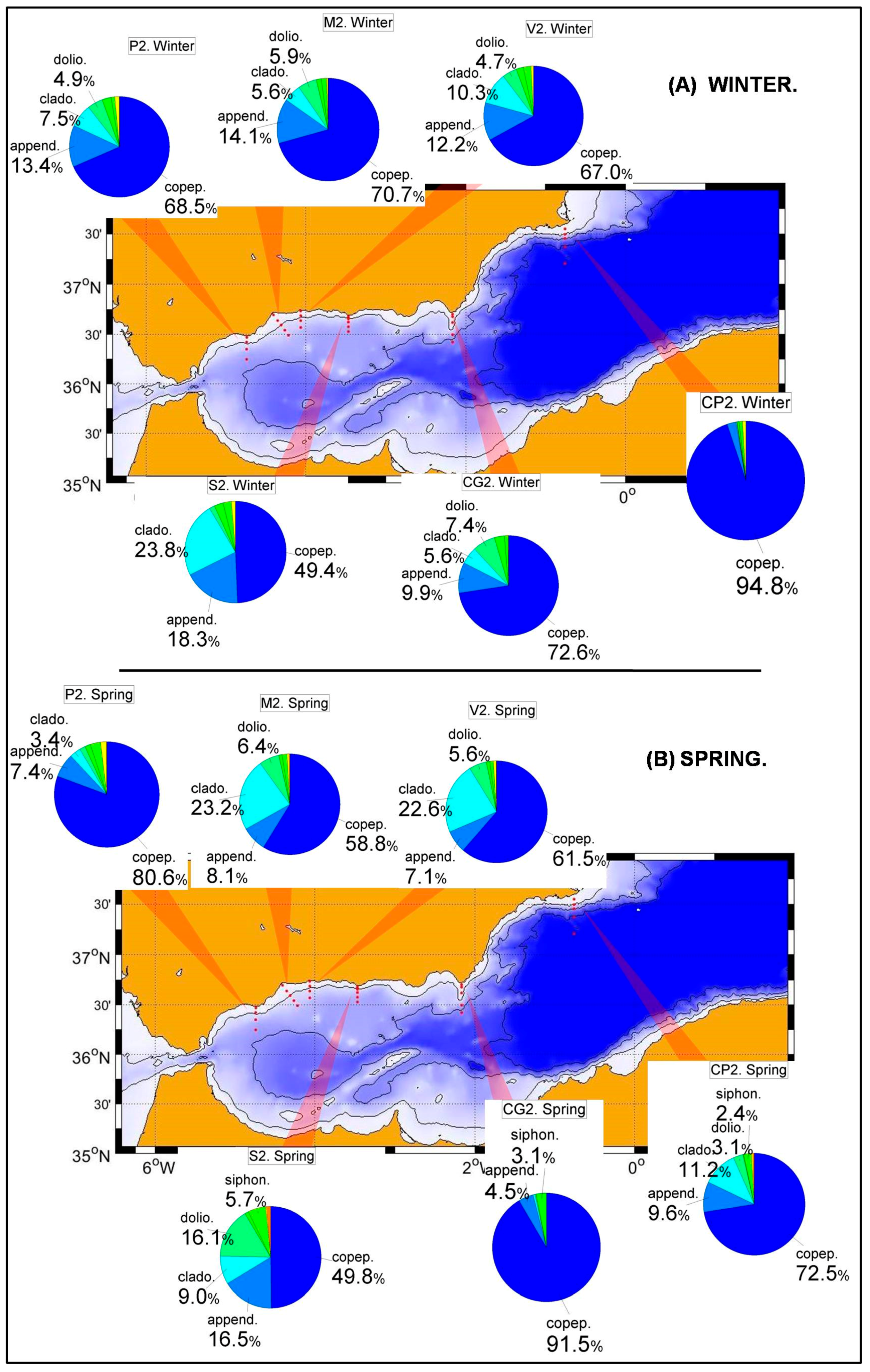
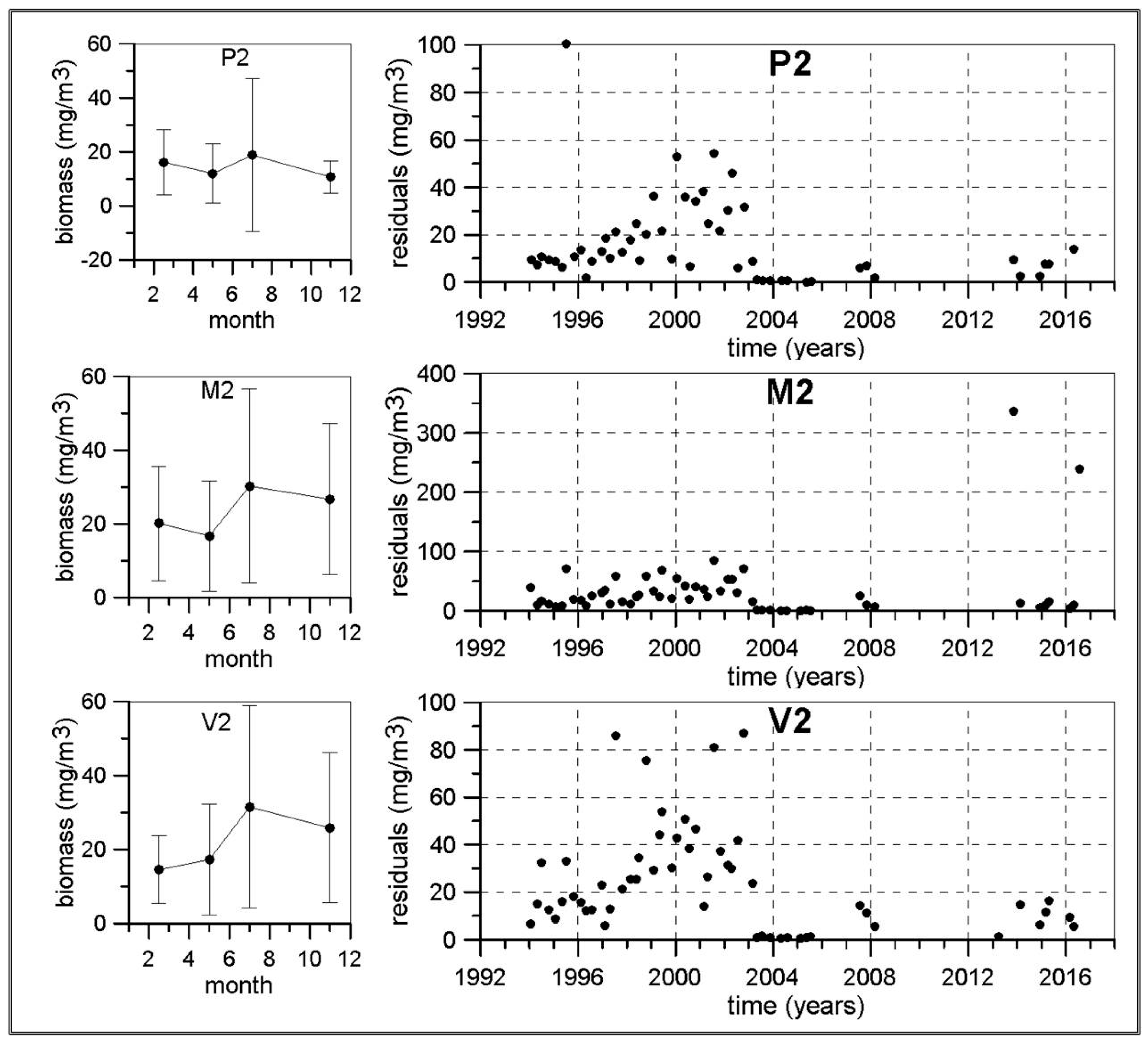
The length of some of the meso-zooplankton time series are greater than 20 years as this sampling was initiated in the early 1990s in the continental shelf of P, M and V transects. The other transects and the continental slope stations were included from 2007. Nevertheless, frequent problems in zooplankton sampling and the lack of personnel have not allowed us to construct long time series for meso-zooplankton abundances. Some of the few exceptions are the P2, M2 and V2 stations around Málaga Bay, where data from 1992 to 2000 from the previous monitoring program ECOMÁLAGA are available [
70,
71]. These data have been merged with abundances from years 2007, 2008 and winter 2009. The resulting statistics show that copepods are the main group in winter and spring in the whole Alboran Sea and Cape Palos. Notice that Sacratif, Cape Gataand Cape Palostransects were initiated in 2007 and because of the already mentioned problems, only one or two values are available for each season. In these cases the seasonal cycles obtained have been included for the completeness of the figures and they could be compared to other works dealing with just one single seasonal cycle, but they should be taken with caution within the frame of the present work.
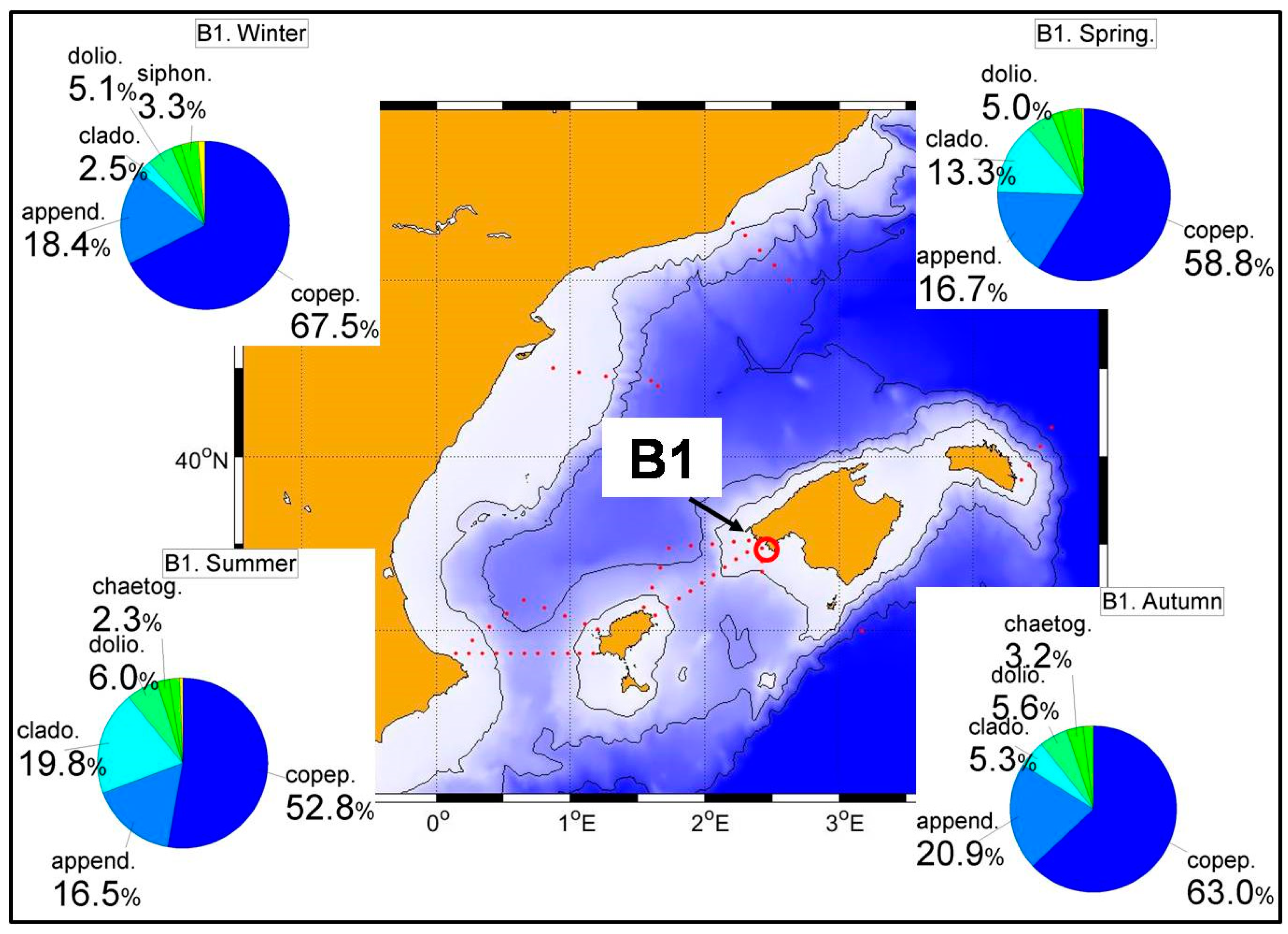
Figure 10 and
Figure 11 show the dominance of copepods throughout most of the year with an important increase of cladocerans, and to a lesser extent, appendicularians in summer and autumn. Although the corresponding figures have not been shown and despite the low number of available samples, this behavior is common to all transects in the RADMED area. This succession of zooplanktonic groups coincides with previous works [
21,
22,
55,
71,
72,
73] and could be linked to the different feeding preferences. Copepods can select prey and tend to feed on large size particles from the micro-phytoplankton [
74]. On the contrary, cladocerans are filters and mainly feed on pico and nanoplankton. Appendicularians would feed on the same size range than cladocerans [
74,
75]. Nevertheless, for the case of station P2 and other stations within the Alboran Sea, it cannot be established a clear decrement of large cells (diatoms) in summer, nor an increase of pico and nanoplankton that would justify the hypothesis mentioned above. In the case of the Balearic Sea, (
Figure 3 and
Figure 4), and the Catalan Sea (
Figure 5), the micro-phytoplankton abundance diminished during the summer season, supporting the meso-zooplankton groups succession associated to the change in prey availability.
Considering the total number of meso-zooplankton individuals and biomass (dry weight), there are also important differences from the south to the north. In the Alboran Sea the maximum values of total individuals are observed in summer with 1217 ind./m
3 at station P2 (
Table S9) or 2172 ind./m
3 at the station V2 (
Table S11). Biomass also reached a maximum in summer with 18.8, 30.2 and 31.5 mg/m
3 in P2, M2 and V2 respectively. By contrast, according to [
43] (
Table S12), the highest values of both abundances and biomass in station B1 were observed in spring with values of 1082 ind./m
3 and 6.4 mg/m
3. These figures simply reflect the lower productivity of the Balearic Sea which is transferred to the secondary producers.
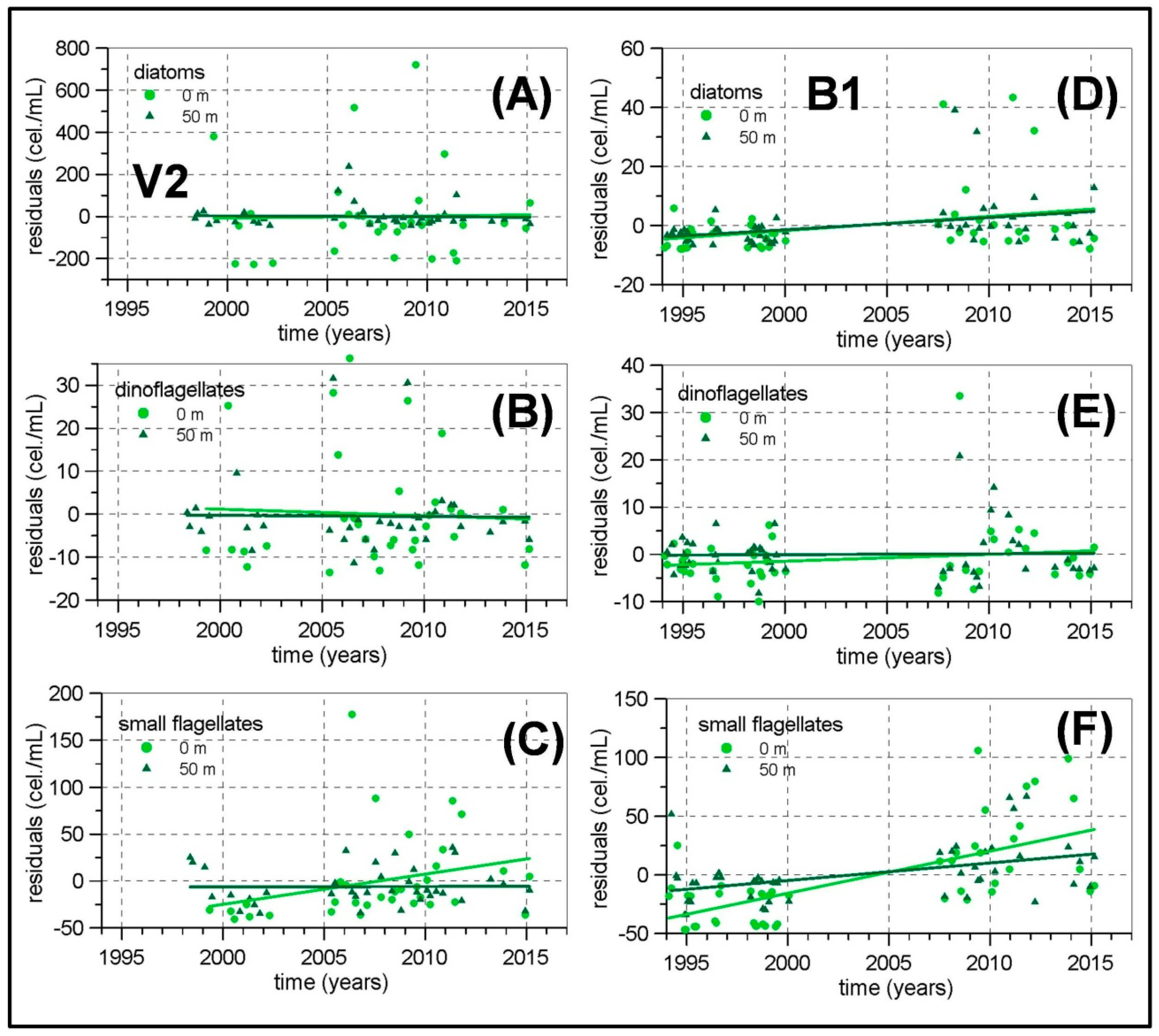
Concerning the possible existence of long-term changes, the higher temperatures already observed in the Western Mediterranean [
46] and in the RADMED area [
15,
36] could increase the thermal stratification of the water column decreasing the efficiency of winter mixing and the nutrient supply to the photic layer. This could lead to a negative trend in planktonic abundances and changes in the community structure with a higher dominance of small-sized cells which are expected to have lower nutrient requirements [
76,
77,
78]. The micro-phytoplankton and meso-zooplankton biomass time series in the Málaga Bay area (transects P, M and V) are the longest and more complete ones from the RADMED area. These time series extend over a 20 year-long period. If the strong inter-annual and decadal variability present in planktonic time series is considered, it should be concluded that 20 years is a short period of time, even more if the frequent data gaps are taken into account. For instance, [
11] observed a negative trend for the chlorophyll-
a concentrations at the station B at Villefranche from 1979 to 1998. This long-term change disappeared when the series was extended to 2005 [
26]. All these reasons make us consider the present results with caution. Nevertheless, some negative trends have been estimated for the diatom abundances in the northwestern Alboran Sea and also for the meso-zooplanktonic biomass, which could be a signal of increasing stratification, less vertical nutrient supply, and decreasing micro-phytoplankton and zooplankton biomass. In summary, it can be concluded that the general oligotrophy of the Mediterranean waters is modulated in the Spanish Mediterranean by several mechanisms. In the Alboran Sea, the strong tidal mixing in the nearby Strait of Gibraltar and the existence of quasi-permanent frontal structures and cyclonic circulation cells are responsible for a high productivity which in turn produces the dominance of large phytoplanktonic cells (diatoms) throughout the whole year. Maximum values for such abundances are observed in the upper part of the water column indicating the availability of nutrients in the surface and sub-surface layers. The meso-zooplankton is dominated by copepods with an important increment of cladocerans and appendicularians in summer. The diatom abundances in this area are the highest of the whole RADMED area with values higher than 400 cel./mL. Zooplanktonic biomass also shows the highest values reaching more than 30 mg/m
3 in the summer season. The dominance of the diatom group disappears towards the eastern sector of the Alboran Sea, where small flagellates are the most abundant group. This trend continues and is accentuated to the north. The only exception is the winter season in the Barcelona and Tarragona transects which are affected by strong winter mixing. These processes would relax the oligotrophy of waters in the Catalan-Balearic Sea. Nevertheless, this effect decreases southward affecting in a much weaker way to the Ibiza Channel and Mallorca waters which seem to be the most oligotrophic ones in the RADMED area. The picoplanktonic fraction is the most abundant one in the phytoplanktonic community, with abundances of the order of 10
4 cel./mL, being followed by the nanoplanktonic fraction, which abundances are one order of magnitude lower (10
3 cel./mL). The general trend shows larger abundances for
Synechococcus at the upper 20 m of the water column, while
Prochlorococcus tend to develop a deep maximum from 25 m to 75 m. Nevertheless, this behavior shows frequent exceptions. Maximum values for
Synechococcus abundances are at the sea surface and the deep
Prochlorococcus maximum is at shallower waters in winter in most of the stations, when the nutrient supply to the photic layer is higher, and also in the Alboran Sea throughout the whole year. The zooplanktonic biomass in the Balearic Sea is lower than that observed in the Alboran Sea, reflecting the oligotrophy of these waters. Finally, the available time series do not allow us to obtain long-term trends for phytoplankton abundances and zooplanktonic biomass in most of the stations. Nevertheless, the best sampled area in the Málaga Bay, suggests a possible decrease of the large fraction of the phytoplankton (diatoms) and of the meso-zooplanktonic biomass. Such important questions should be followed in the future and evidence the importance of preserving monitoring programs and time series in the Mediterranean Sea and the world ocean.