Contrasting Effects of an Alien Worm on Benthic N Cycling in Muddy and Sandy Sediments
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Procedure and Sediment Characterization
2.2. Incubation Setup and Measurement of Benthic Fluxes
2.3. Statistical Analyses
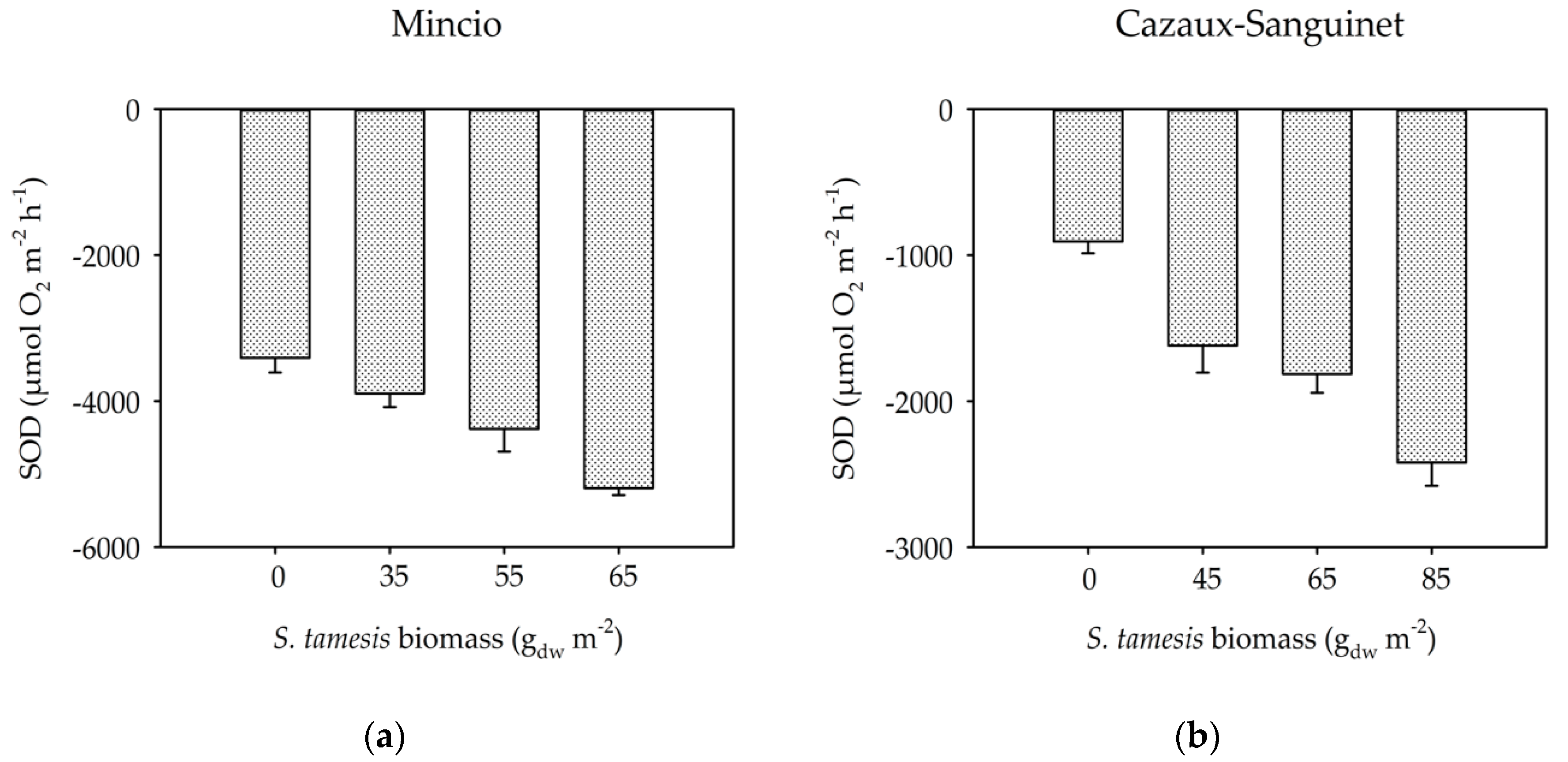
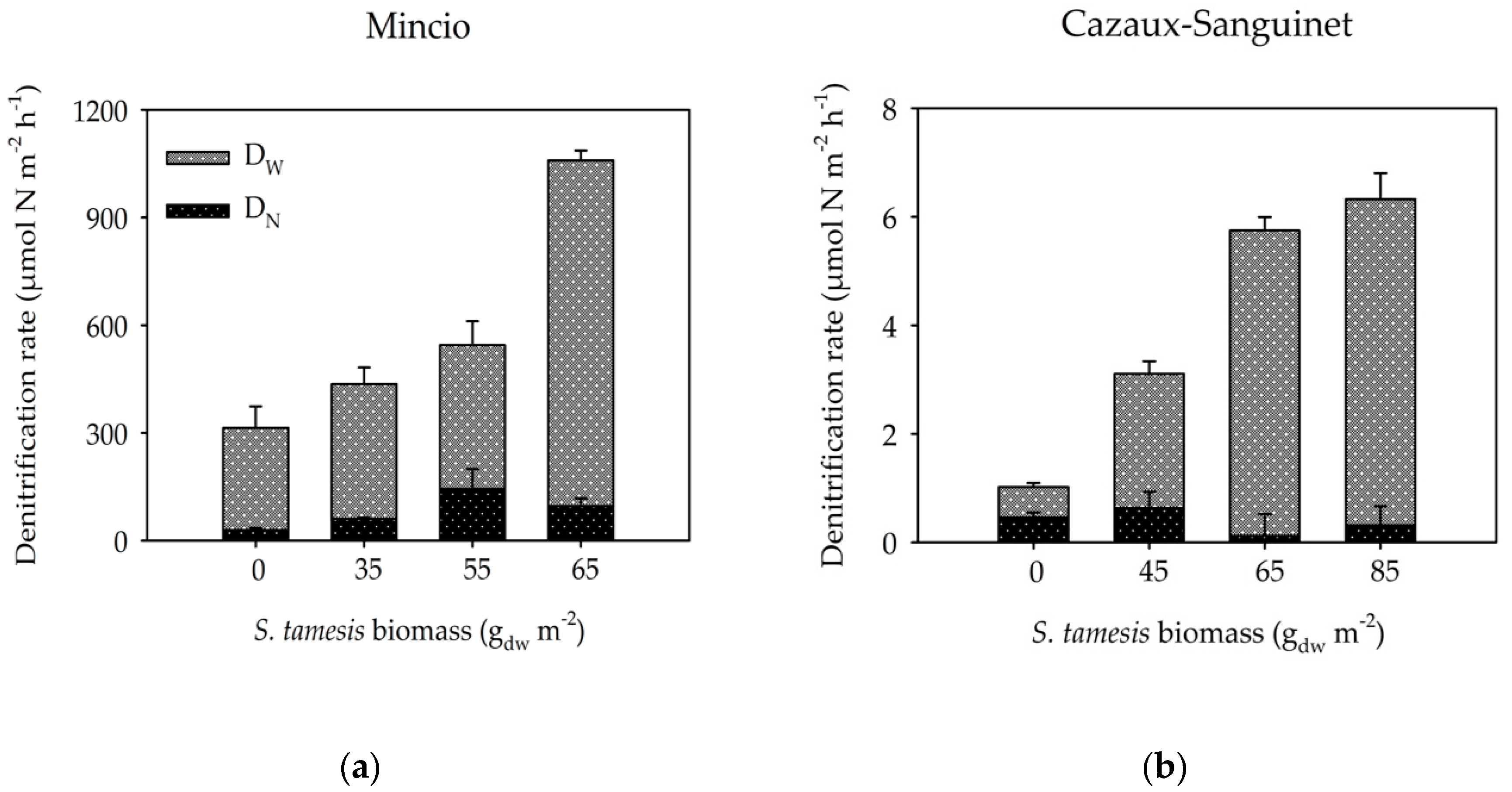
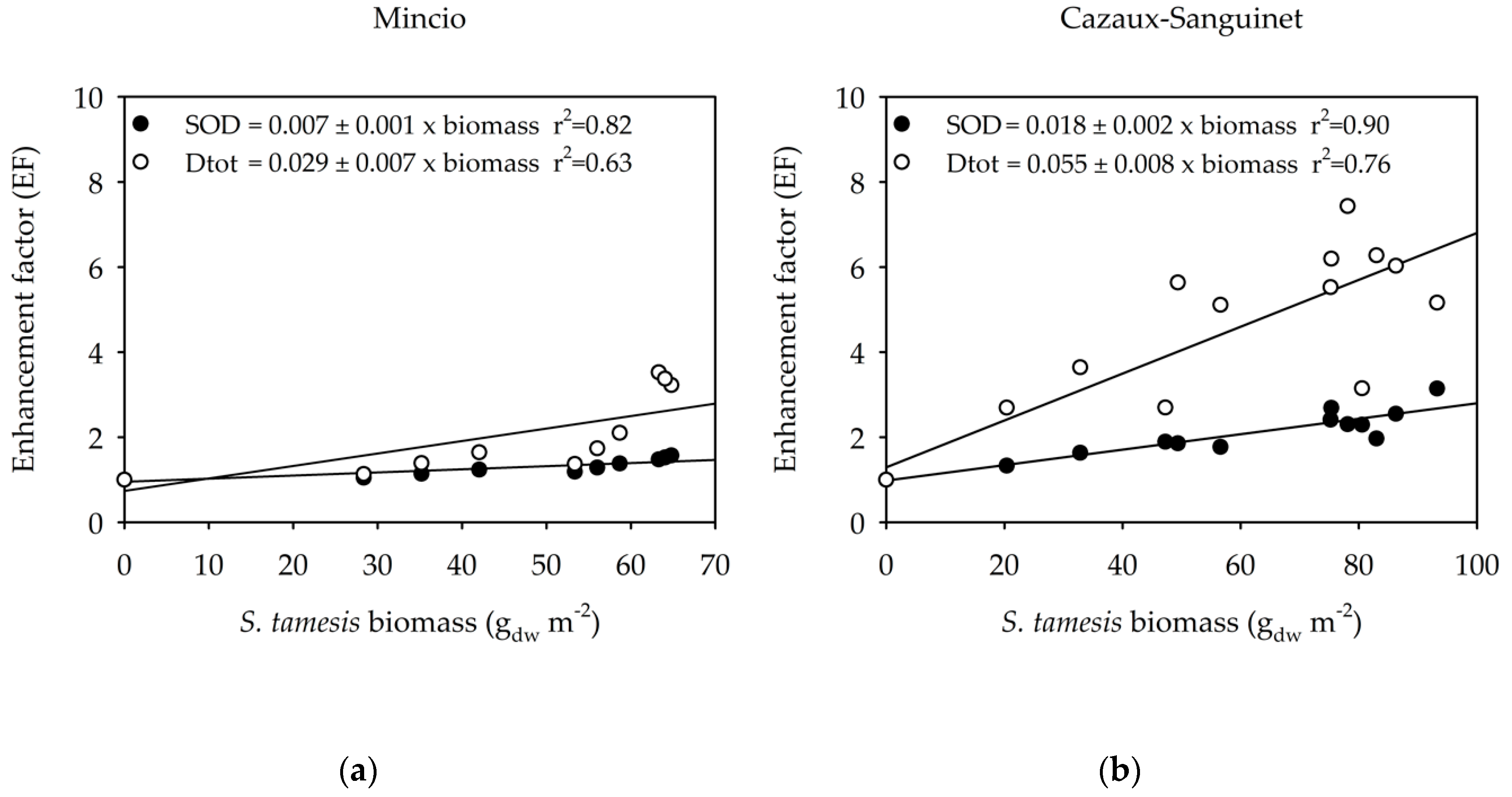
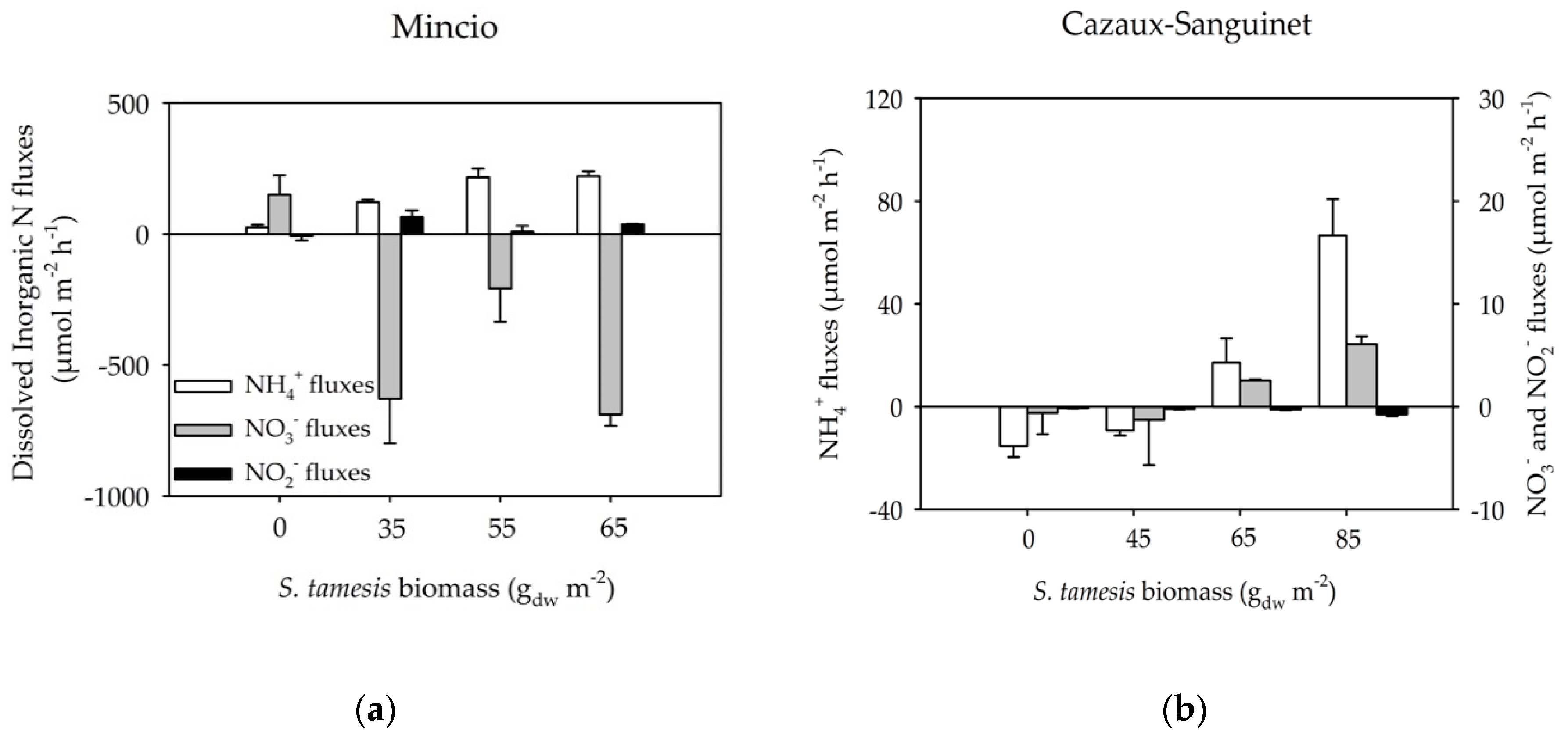
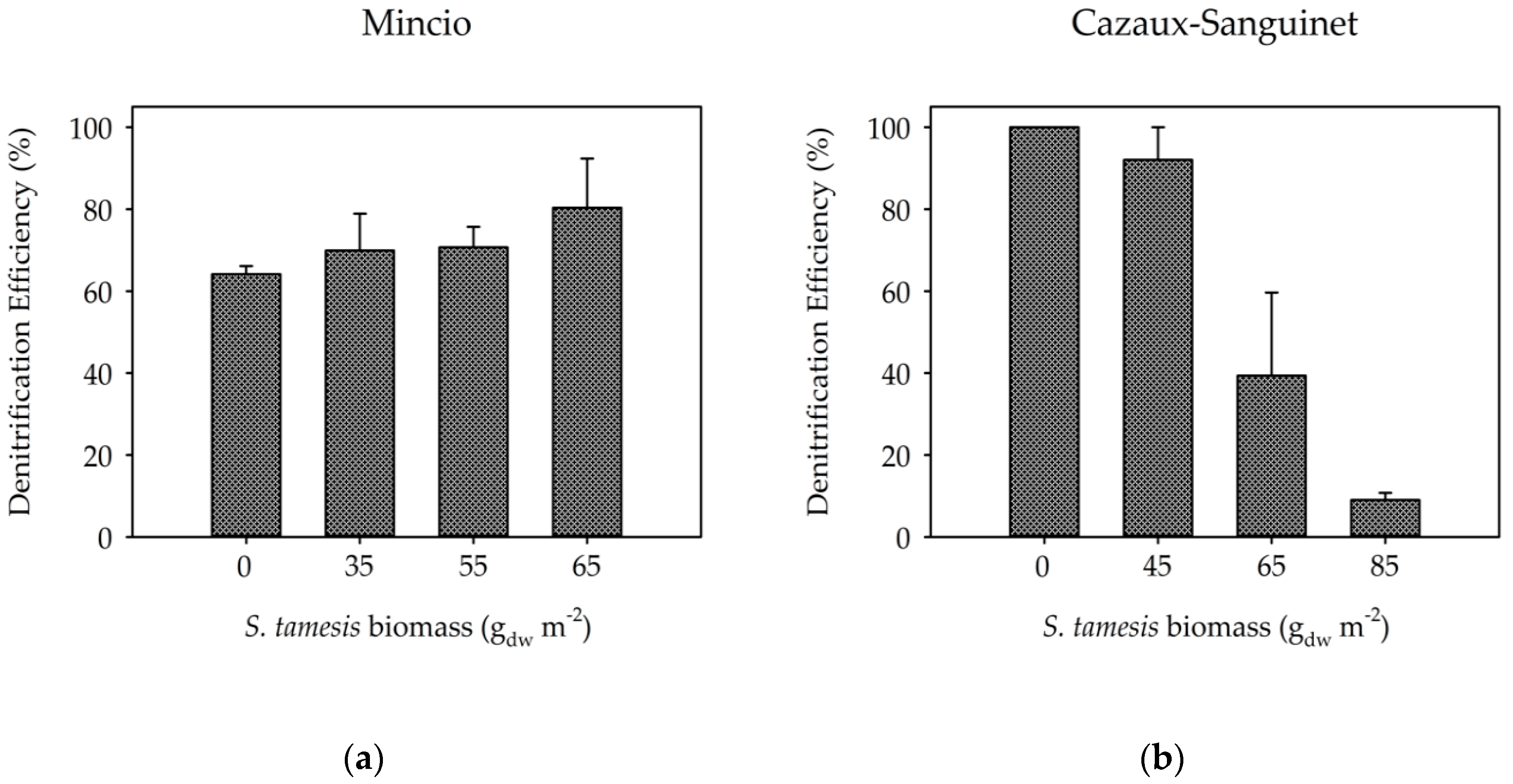
3. Results
3.1. Sedimentary Features
3.2. Benthic Fluxes Along Increasing Sparganophilus tamesis Biomass
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Norkko, J.; Reed, D.C.; Timmermann, K.; Norkko, A.; Gustafsson, B.G.; Bonsdorff, E.; Slomp, C.P.; Carstensen, J.; Conley, D.J. A welcome can of worms? Hypoxia mitigation by an invasive species. Glob. Chang. Biol. 2012, 18, 422–434. [Google Scholar] [CrossRef]
- Strayer, D.L. Alien species in fresh waters: Ecological effects, interactions with other stressors, and prospects for the future. Freshw. Biol. 2010, 55, 152–174. [Google Scholar] [CrossRef]
- Kristensen, E. Benthic fauna and biogeochemical processes in marine sediments: Microbial activities and fluxes. In Nitrogen Cycling in Coastal Marine Environments; Blackburn, T.H., Sorensen, J., Eds.; John Wiley & Sons Ltd.: Hoboken, NI, USA, 1988; pp. 275–299. [Google Scholar]
- Hietanen, S.; Laine, A.O.; Lukkari, K. The complex effects of the invasive polychaetes Marenzelleria spp. on benthic nutrient dynamics. J. Exp. Mar. Biol. Ecol. 2007, 352, 89–102. [Google Scholar] [CrossRef]
- Strayer, D.L. Effects of alien species on freshwater mollusks in North America. Freshw. Sci. 1999, 18, 74–98. [Google Scholar] [CrossRef]
- Crooks, J.A. Characterizing ecosystem-level consequences of biological invasions: The role of ecosystem engineers. Oikos 2002, 97, 153–166. [Google Scholar] [CrossRef]
- Strayer, D.L.; Caraco, N.F.; Cole, J.J.; Findlay, S.; Pace, M.L. Transformation of freshwater ecosystems by bivalves: A case study of zebra mussels in the Hudson River. Bioscience 1999, 49, 19–27. [Google Scholar] [CrossRef]
- Strayer, D.L. Twenty years of zebra mussels: Lessons from the mollusk that made headlines. Front. Ecol. Environ. 2009, 7, 135–141. [Google Scholar] [CrossRef]
- Ruginis, T.; Bartoli, M.; Petkuviene, J.; Zilius, M.; Lubiene, I.; Laini, A.; Razinkovas-Baziukas, A. Benthic respiration and stoichiometry of regenerated nutrients in lake sediments with Dreissena polymorpha. Aquat. Sci. 2014, 76, 405–417. [Google Scholar] [CrossRef]
- Sousa, R.; Novais, A.; Costa, R.; Strayer, D.L. Invasive bivalves in fresh waters: Impacts from individuals to ecosystems and possible control strategies. Hydrobiologia 2014, 735, 233–251. [Google Scholar] [CrossRef]
- Fahnenstiel, G.L.; Lang, G.A.; Nalepa, T.F.; Johengen, T.H. Effects of Zebra Mussel (Dreissena polymorpha) Colonization on Water Quality Parameters in Saginaw Bay, Lake Huron. J. Great Lakes Res. 1995, 21, 435–448. [Google Scholar] [CrossRef]
- Cha, Y.; Stow, C.A.; Bernhardt, E.S. Impacts of dreissenid mussel invasions on chlorophyll and total phosphorus in 25 lakes in the USA. Freshw. Biol. 2013, 58, 192–206. [Google Scholar] [CrossRef]
- Gardner, W.S.; Cavaletto, J.F.; Johengen, T.H.; Johnson, J.R.; Heath, R.T.; Cotner, J.B., Jr. Effects of the zebra mussel, Dreissena polymorpha, on community nitrogen dynamics in Saginaw Bay, Lake Huron. J. Gt. Lakes Res. 1995, 21, 529–544. [Google Scholar] [CrossRef]
- Caraco, N.F.; Cole, J.J.; Strayer, D.L. Top down control from the bottom: Regulation of eutrophication in a large river by benthic grazing. Limnol. Oceanogr. 2006, 51, 664–670. [Google Scholar] [CrossRef]
- Kristensen, E.; Hansen, T.; Delefosse, M.; Banta, G.T.; Quintana, C.O. Contrasting effects of the polychaetes Marenzelleria viridis and Nereis diversicolor on benthic metabolism and solute transport in sandy coastal sediment. Mar. Ecol. Prog. Ser. 2011, 425, 125–139. [Google Scholar] [CrossRef]
- Bonaglia, S.; Bartoli, M.; Gunnarsson, J.S.; Rahm, L.; Raymond, C.; Svensson, O.; Yekta, S.S.; Brüchert, V. Effect of reoxygenation and Marenzelleria spp. bioturbation on Baltic Sea sediment metabolism. Mar. Ecol. Prog. Ser. 2013, 482, 43–55. [Google Scholar] [CrossRef]
- Quintana, C.O.; Kristensen, E.; Valdemarsen, T. Impact of the invasive polychaete Marenzelleria viridis on the biogeochemistry of sandy marine sediments. Biogeochemistry 2013, 115, 95–109. [Google Scholar] [CrossRef]
- Kauppi, L.; Norkko, A.; Norkko, J. Large-scale species invasion into a low-diversity system: Spatial and temporal distribution of the invasive polychaetes Marenzelleria spp. in the Baltic Sea. Biol. Invasions 2015, 17, 2055–2074. [Google Scholar] [CrossRef]
- Benelli, S.; Bartoli, M.; Zilius, M.; Vybernaite-Lubiene, I.; Ruginis, T.; Petkuviene, J.; Fano, E.A. Microphytobenthos and chironomid larvae attenuate nutrient recycling in shallow-water sediments. Freshw. Biol. 2018, 63, 187–201. [Google Scholar] [CrossRef]
- Vanni, M.J. Nutrient cycling by animals in freshwater ecosystems. Annu. Rev. Ecol. Syst. 2002, 33, 341–370. [Google Scholar] [CrossRef]
- Rota, E.; Benelli, S.; Erséus, C.; Soors, J.; Bartoli, M. New data and hypotheses on the invasiveness, habitat selection, and ecological role of the limicolous earthworm Sparganophilus tamesis Benham, 1892. Arch. Hydrobiol. 2018, 192, 129–136. [Google Scholar] [CrossRef]
- Ribaudo, C.; Bertrin, V.; Jan, G.; Anschutz, P.; Abril, G. Benthic production, respiration and methane oxidation in Lobelia dortmanna lawns. Hydrobiologia 2017, 784, 21–34. [Google Scholar] [CrossRef]
- Dalsgaard, T.; Underwood, J.C.; Nedwell, D.B.; Sundbäck, K.; Rysgaard, S.; Miles, A.; Bartoli, M.; Dong, L.; Thornton, D.C.O.; Ottosen, L.D.M.; et al. Protocol Handbook for NICE-Nitrogen Cycling in Estuaries; National Environmental Research Institute: Silkeborg, Denmark, 2000. [Google Scholar]
- Golterman, H.L.; Clymo, R.S.; Ohnstad, M.A.M. Methods for Physical and Chemical Analysis of Fresh Waters, 2nd ed.; IBP Handbook; Blackwell Scientific: Oxford, UK, 1978; Volume 8. [Google Scholar]
- Bower, C.E.; Holm-Hansen, T. A salicylate–hypochlorite method for determining ammonia in seawater. Can. J. Fish. Aquat. Sci. 1980, 37, 794–798. [Google Scholar] [CrossRef]
- Nielsen, L.P. Denitrification in sediment determined from nitrogen isotope pairing. FEMS Microbiol. Lett. 1992, 86, 357–362. [Google Scholar] [CrossRef]
- Benelli, S.; Bartoli, M.; Zilius, M.; Vybernaite-Lubiene, I.; Ruginis, T.; Vaiciute, D.; Petkuviene, J.; Fano, E.A. Stoichiometry of regenerated nutrients differs between native and invasive freshwater mussels with implications for algal growth. Freshw. Biol. 2019. [Google Scholar] [CrossRef]
- Magri, M.; Benelli, S.; Bondavalli, C.; Bartoli, M.; Christian, R.R.; Bodini, A. Benthic N pathways in illuminated and bioturbated sediments studied with network analysis: Network analysis of benthic N processes. Limnol. Oceanogr. 2018, 63, S68–S84. [Google Scholar] [CrossRef]
- Marzocchi, U.; Benelli, S.; Larsen, M.; Bartoli, M.; Glud, R.N. Spatial heterogeneity and short-term oxygen dynamics in the rhizosphere of Vallisneria spiralis: Implications for nutrient cycling. Freshw. Biol. 2019. [Google Scholar] [CrossRef]
- Nizzoli, D.; Bartoli, M.; Cooper, M.; Welsh, D.T.; Underwood, G.J.C.; Viaroli, P. Implications for oxygen, nutrient fluxes and denitrification rates during the early stage of sediment colonisation by the polychaete Nereis spp. in four estuaries. Estuar. Coast. Shelf Sci. 2007, 75, 125–134. [Google Scholar] [CrossRef]
- Bartoli, M.; Longhi, D.; Nizzoli, D.; Como, S.; Magni, P.; Viaroli, P. Short term effects of hypoxia and bioturbation on solute fluxes, denitrification and buffering capacity in a shallow dystrophic pond. J. Exp. Mar. Biol. Ecol. 2009, 381, 105–113. [Google Scholar] [CrossRef]
- Pinardi, M.; Bartoli, M.; Longhi, D.; Marzocchi, U.; Laini, A.; Ribaudo, C.; Viaroli, P. Benthic metabolism and denitrification in a river reach: A comparison between vegetated and bare sediments. J. Limnol. 2009, 68, 133–145. [Google Scholar] [CrossRef]
- Pelegri, S.P.; Nielsen, L.P.; Blackburn, T.H. Denitrification in estuarine sediment stimulated by the irrigation activity of the amphipod Corophium volutator. Mar. Ecol. Prog. Ser. 1994, 105, 285–290. [Google Scholar] [CrossRef]
- Aller, R.C. Benthic fauna and biogeochemical processes in marine sediments: The role of burrow structures. In Nitrogen Cycling in Coastal Marine Environments; Blackburn, T.H., Sorensen, J., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 1988; pp. 301–338. [Google Scholar]
- Pelegrí, S.P.; Blackburn, T.H. Effect of Bioturbation by Nereis sp., Mya Arenaria and Cerastoderma sp. on nitrification and denitrification in estuarine sediments. Ophelia 1995, 42, 289–299. [Google Scholar] [CrossRef]
- Nielsen, O.; Gribsholt, B.; Kristensen, E.; Revsbech, N. Microscale distribution of oxygen and nitrate in sediment inhabited by Nereis diversicolor: Spatial patterns and estimated reaction rates. Aquat. Microb. Ecol. 2004, 34, 23–32. [Google Scholar] [CrossRef]
- Kristensen, E.; Penha-Lopes, G.; Delefosse, M.; Valdemarsen, T.; Quintana, C.O.; Banta, G.T. What is bioturbation? The need for a precise definition for fauna in aquatic sciences. Mar. Ecol. Prog. Ser. 2012, 446, 285–302. [Google Scholar] [CrossRef]
- Kristensen, E.; Delefosse, M.; Quintana, C.O.; Flindt, M.R.; Valdemarsen, T. Influence of benthic macrofauna community shifts on ecosystem functioning in shallow estuaries. Front. Mar. Sci. 2014, 1, 41. [Google Scholar] [CrossRef]
- Jensen, K.; Jensen, M.; Kristensen, E. Nitrification and denitrification in Wadden Sea sediments (Königshafen, Island of Sylt, Germany) as measured by nitrogen isotope pairing and isotope dilution. Aquat. Microb. Ecol. 1996, 11, 181–191. [Google Scholar] [CrossRef]
- Svensson, J.M. Influence of Chironomus plumosus larvae on ammonium flux and denitrification (measured by the acetylene blockage-and the isotope pairing-technique) in eutrophic lake sediment. Hydrobiologia 1997, 346, 157–168. [Google Scholar] [CrossRef]
- Karlson, K.; Hulth, S.; Ringdahl, K.; Rosenberg, R. Experimental recolonisation of Baltic Sea reduced sediments: Survival of benthic macrofauna and effects on nutrient cycling. Mar. Ecol. Prog. Ser. 2005, 294, 35–49. [Google Scholar] [CrossRef]
- Na, T.; Gribsholt, B.; Galaktionov, O.S.; Lee, T.; Meysman, F.J.R. Influence of advective bio-irrigation on carbon and nitrogen cycling in sandy sediments. J. Mar. Res. 2008, 66, 691–722. [Google Scholar] [CrossRef]
- Risgaard-Petersen, N. Coupled nitrification-denitrification in autotrophic and heterotrophic estuarine sediments: On the influence of benthic microalgae. Limnol. Oceanogr. 2003, 48, 93–105. [Google Scholar] [CrossRef]
- Richards, F.A. Anoxic basins and fjords. Chemical Oceanography; Ryley, J.P., Skirrow, G., Eds.; Academic Press: London, UK, 1965; pp. 611–645. [Google Scholar]
- Sand-Jensen, K.; Borum, J.; Binzer, T. Oxygen stress and reduced growth of Lobelia dortmanna in sandy lake sediments subject to organic enrichment. Freshw. Biol. 2005, 50, 1034–1048. [Google Scholar] [CrossRef]
- Schuurkes, J.A.A.R.; Kok, C.J.; Den Hartog, C. Ammonium and nitrate uptake by aquatic plants from poorly buffered and acidified waters. Aquat. Bot. 1986, 24, 131–146. [Google Scholar] [CrossRef]
| Mincio | Cazaux-Sanguinet | |
|---|---|---|
| NH4+ (µM) | 2.0 ± 0.2 | 3.6 ± 0.1 |
| NO3− (µM) | 160.7 ± 5.6 | 1.5 ± 0.1 |
| NO2− (µM) | 3.7 ± 0.3 | 0.4 ± 0.1 |
| Sediment typology | Muddy | Sandy |
| Density (g cm−3) | 1.34 ± 0.08 | 1.83 ± 0.09 |
| Porosity | 0.77 ± 0.04 | 0.36 ± 0.05 |
| Organic matter content (%) | 9.20 ± 0.70 | 0.11 ± 0.03 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benelli, S.; Bartoli, M.; Ribaudo, C.; Fano, E.A. Contrasting Effects of an Alien Worm on Benthic N Cycling in Muddy and Sandy Sediments. Water 2019, 11, 465. https://doi.org/10.3390/w11030465
Benelli S, Bartoli M, Ribaudo C, Fano EA. Contrasting Effects of an Alien Worm on Benthic N Cycling in Muddy and Sandy Sediments. Water. 2019; 11(3):465. https://doi.org/10.3390/w11030465
Chicago/Turabian StyleBenelli, Sara, Marco Bartoli, Cristina Ribaudo, and Elisa Anna Fano. 2019. "Contrasting Effects of an Alien Worm on Benthic N Cycling in Muddy and Sandy Sediments" Water 11, no. 3: 465. https://doi.org/10.3390/w11030465
APA StyleBenelli, S., Bartoli, M., Ribaudo, C., & Fano, E. A. (2019). Contrasting Effects of an Alien Worm on Benthic N Cycling in Muddy and Sandy Sediments. Water, 11(3), 465. https://doi.org/10.3390/w11030465







