Diversity and Biotechnological Potential of Xylan-Degrading Microorganisms from Orange Juice Processing Waste
Abstract
:1. Introduction
2. Materials and Methods
2.1. Physicochemical Characterization of Orange Juice Processing Waste
2.2. Enrichment and Isolation of Xylan-Degraders
2.3. Determination of Endo-1,4-β-xylanase and 1,4-β-xylosidase Activities of Xylanolytic Isolates and Fermented Orange Juice Processing Waste
2.4. Molecular Characterization of Xylanolytic Bacteria Isolated from Orange Juice Processing Waste
2.5. Identification of Microbial Community Structure in Orange Juice Processing Waste by Illumina Sequencing
2.6. Estimation of Ecological Indicators and Statistical Analysis
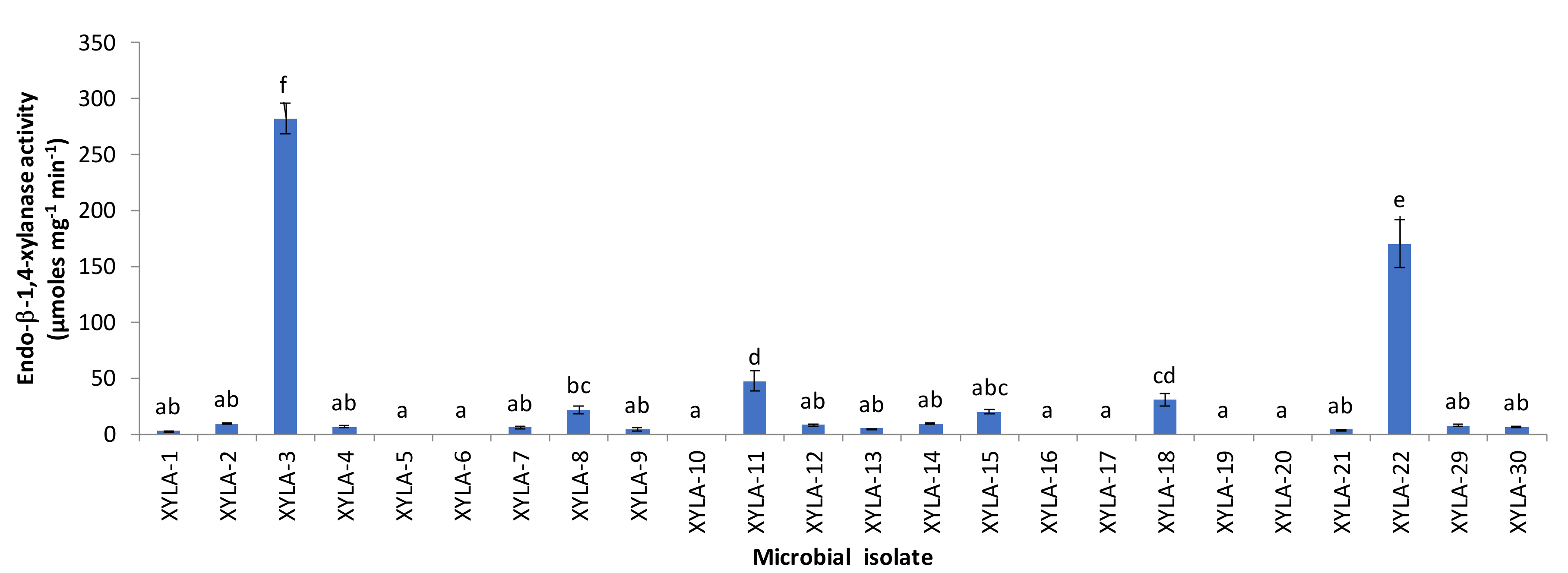
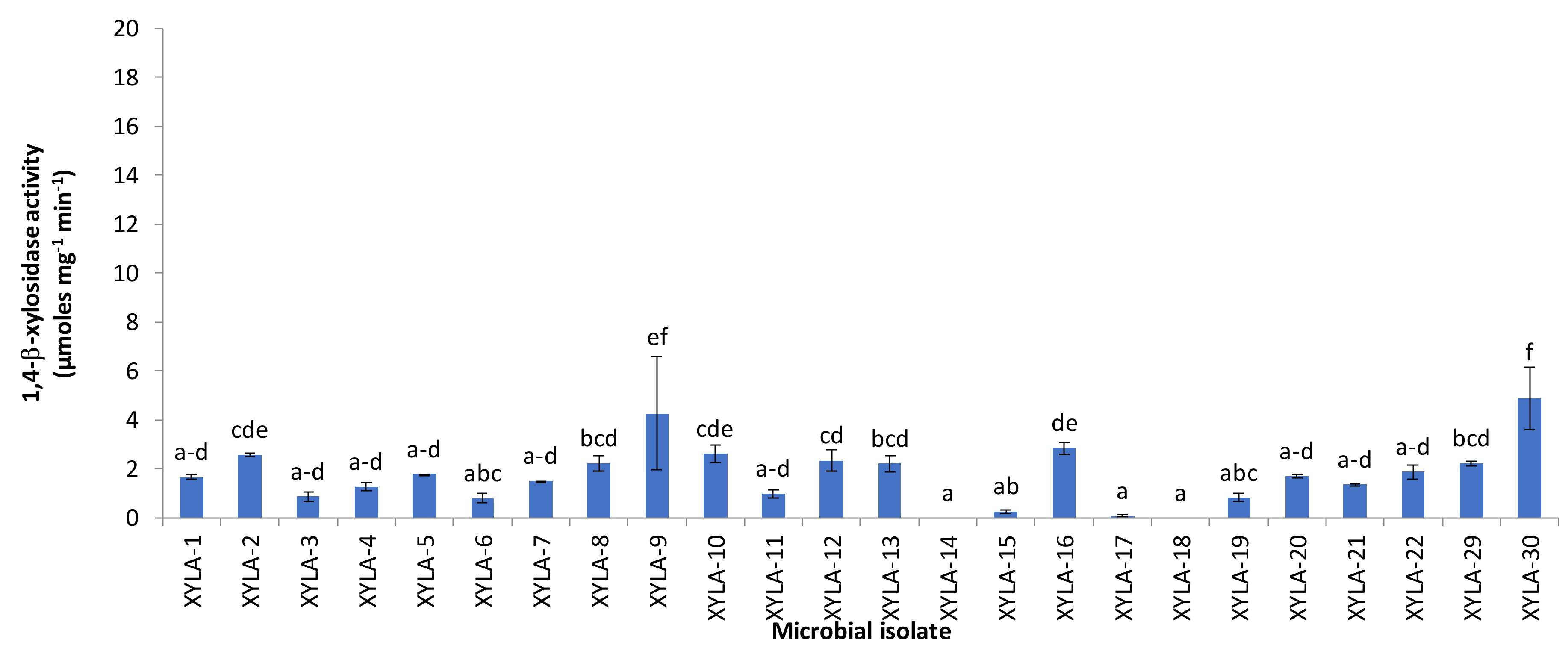
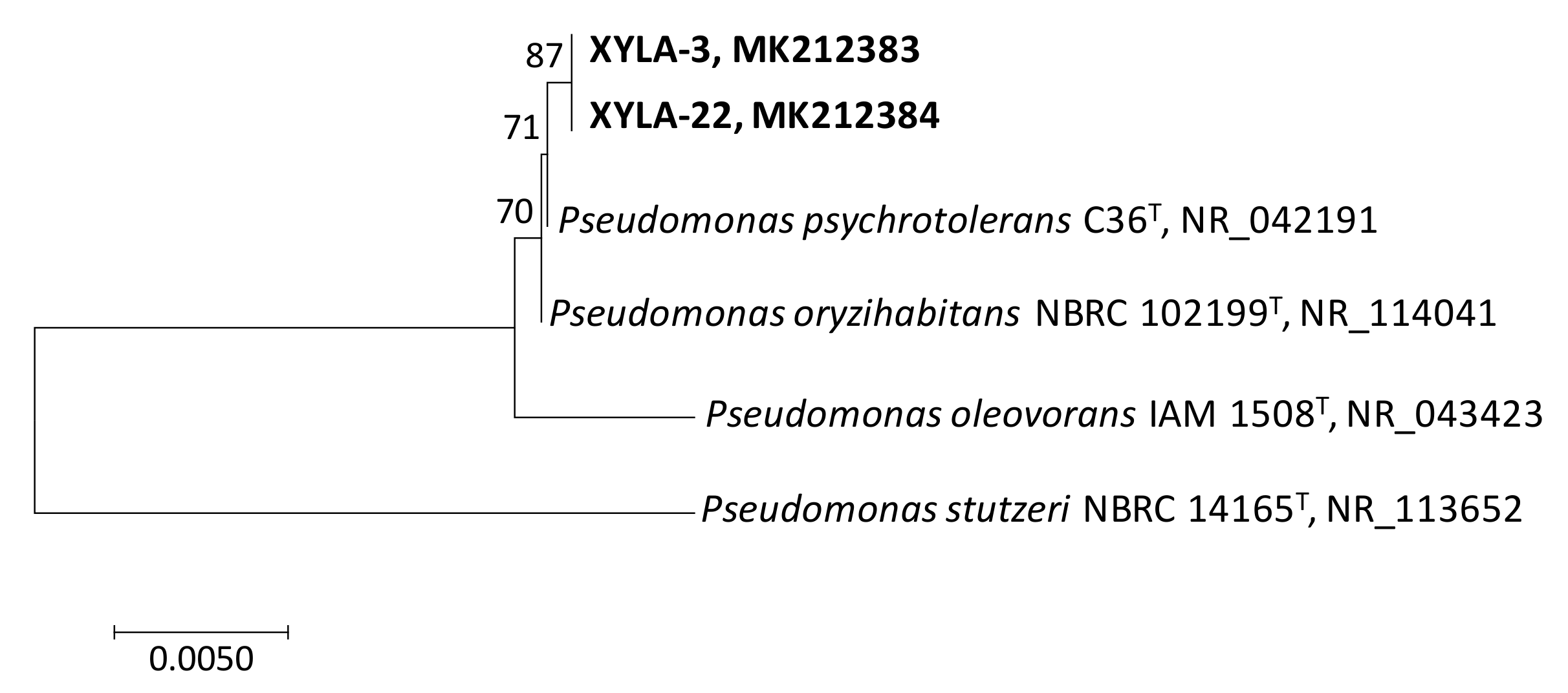
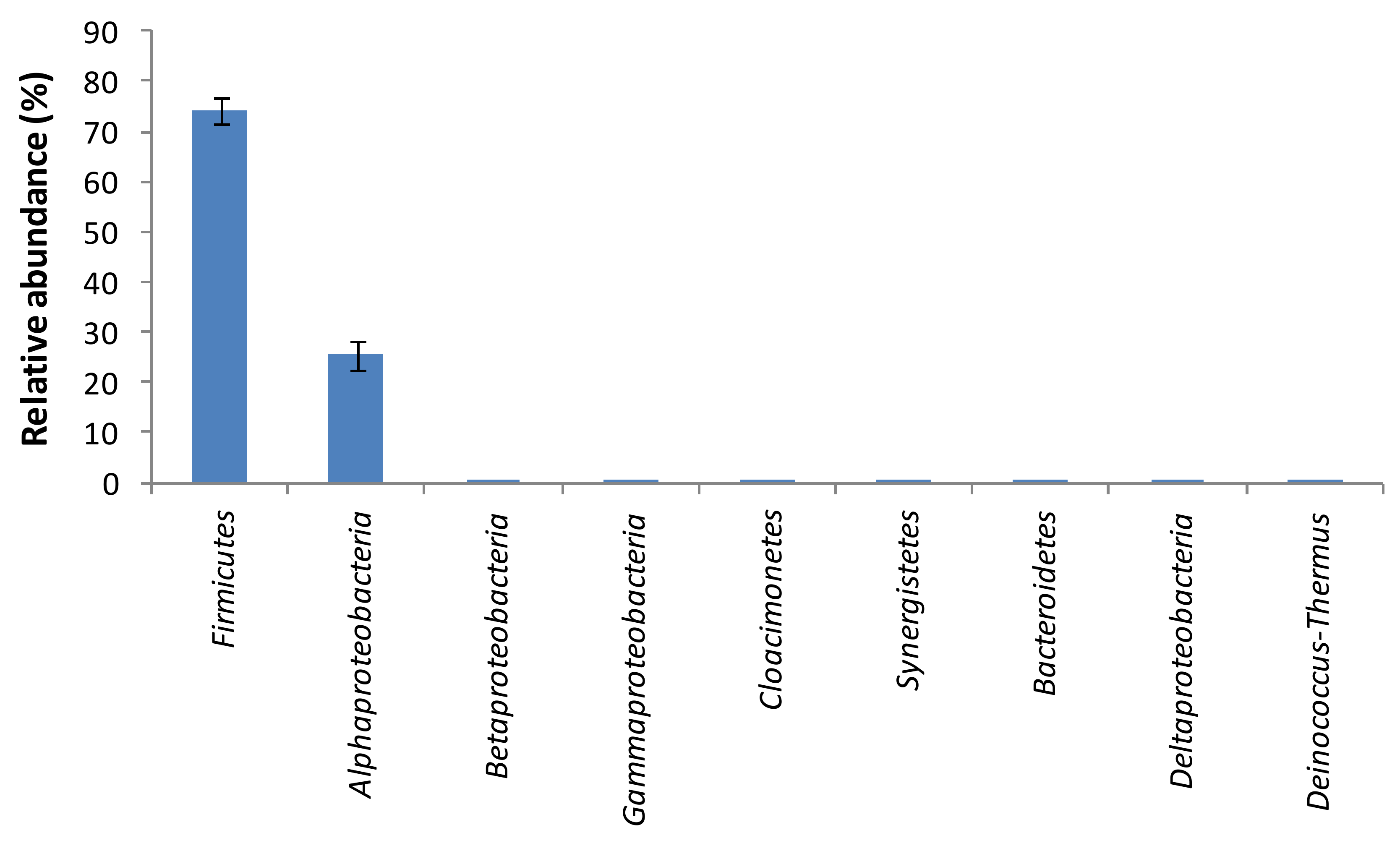
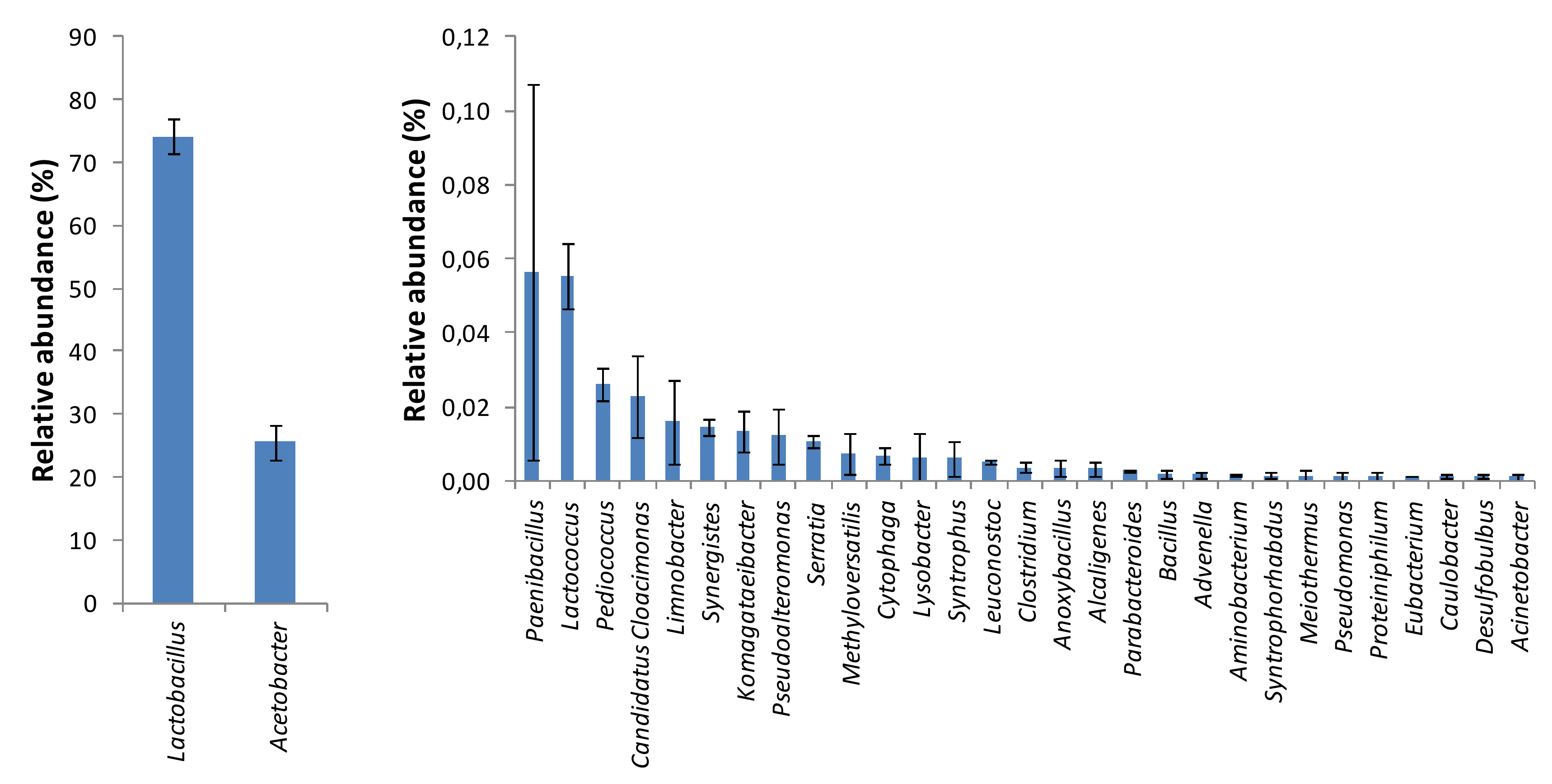
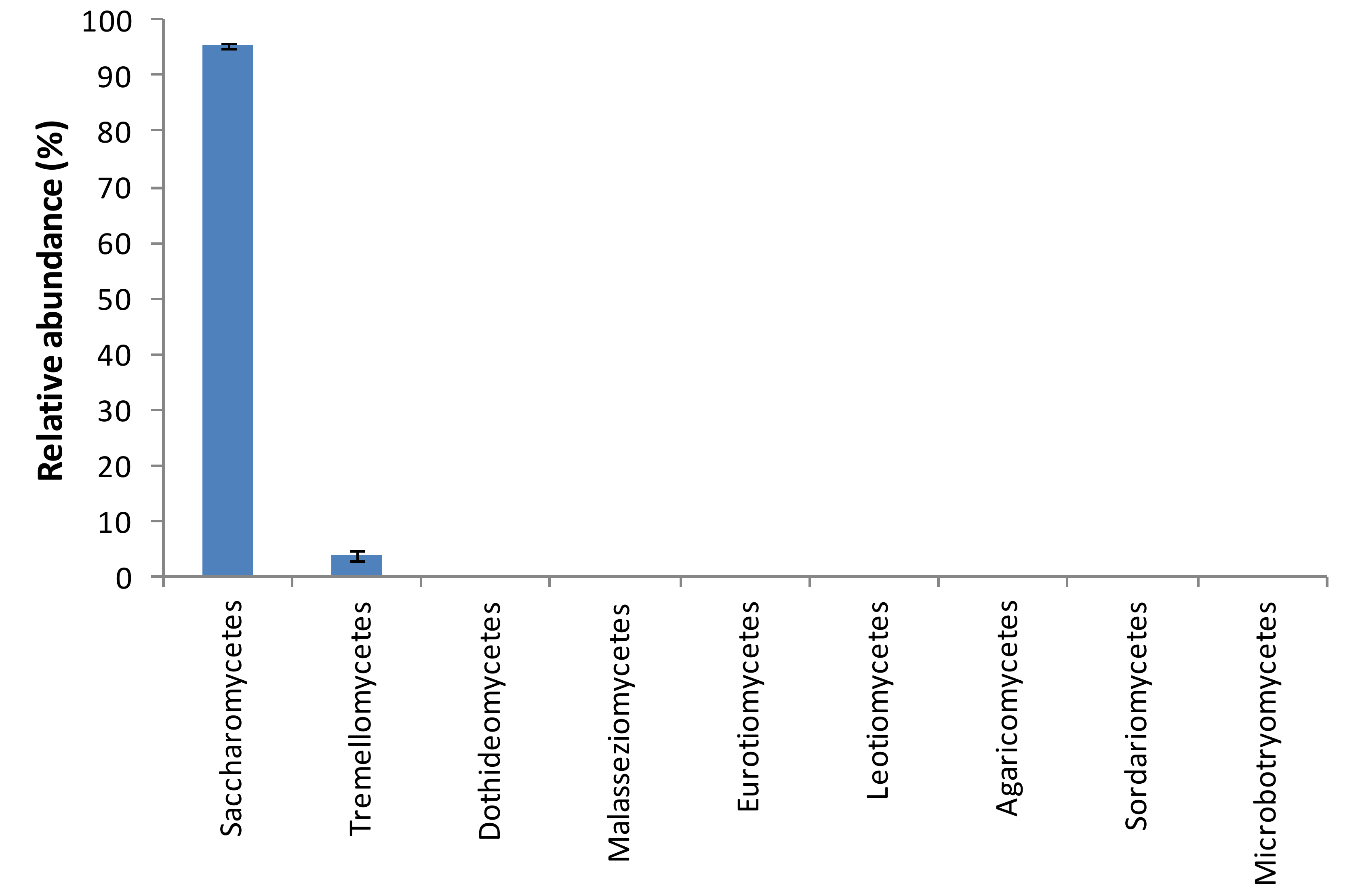
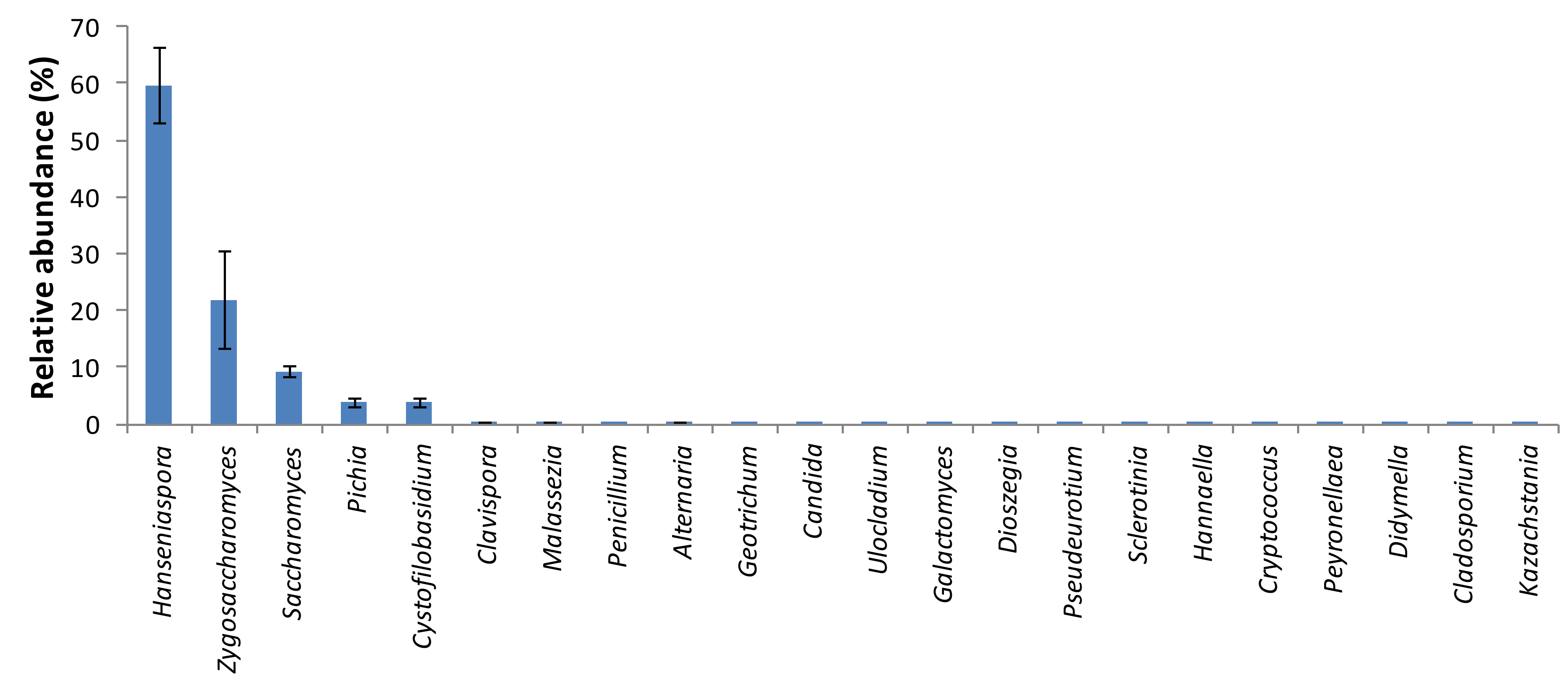
3. Results and Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAO. Citrus Fruit—Fresh and Processed, Statistical Bulletin, 2016; Food and Agriculture Organization of the United Nations: Rome, Italy, 2017. [Google Scholar]
- Dwivedi, P.; Singh, M.; Sehra, N.; Pandey, N.; Sangwan, R.S.; Mishra, B.B. Processing of wet Kinnow mandarin (Citrus reticulata) fruit waste into novel Brønsted acidic ionic liquids and their application in hydrolysis of sucrose. Bioresour. Technol. 2018, 250, 621–624. [Google Scholar] [CrossRef]
- de la Torre, I.; Ravelo, M.; Segarra, S.; Tortajada, M.; Santos, V.E.; Ladero, M. Study on the effects of several operational variables on the enzymatic batch saccharification of orange solid waste. Bioresour. Technol. 2017, 245, 906–915. [Google Scholar] [CrossRef]
- Ayala, J.R.; Montero, G.; Campbell, H.E.; García, C.; Coronado, M.A.; León, J.A.; Sagaste, C.A.; Pérez, L.J. Extraction and characterization of orange peel essential oil from Mexico and United States of America. J. Essent Oil Bear Plants 2017, 20, 897–914. [Google Scholar] [CrossRef]
- Barbosa, P.P.M.; Ruviaro, A.R.; Macedo, G.A. Comparison of different Brazilian citrus by-products as source of natural antioxidants. Food Sci. Biotechnol. 2018, 27, 1301–1309. [Google Scholar] [CrossRef]
- Irem, S.; Islam, E.; Mahmood Khan, Q.; Ul Haq, M.A.; Jamal Hashmat, A. Adsorption of arsenic from drinking water using natural orange waste: Kinetics and fluidized bed column studies. Water Sci. Technol. 2017, 17, 1149–1159. [Google Scholar] [CrossRef]
- John, I.; Muthukumar, K.; Arunagiri, A. A review on the potential of citrus waste for D-limonene, pectin, and bioethanol production. Int. J. Green Energy 2017, 14, 599–612. [Google Scholar] [CrossRef]
- Muscolo, A.; Settineri, G.; Papalia, T.; Attinà, E.; Basile, C.; Panuccio, M.R. Anaerobic co-digestion of recalcitrant agricultural wastes: Characterizing of biochemical parameters of digestate and its impacts on soil ecosystem. Sci. Total Environ. 2017, 586, 746–752. [Google Scholar] [CrossRef] [PubMed]
- Lotito, A.M.; De Sanctis, M.; Pastore, C.; Di Iaconi, C. Biomethanization of citrus waste: Effect of waste characteristics and of storage on treatability and evaluation of limonene degradation. J. Environ. Manag. 2018, 215, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, E.; Jones, K.D.; Taylor, K.E.; Hosseney, E.N.; Mills, P.L.; Escudero, J.M. Recombinant, E. coli cellulases, β-glucosidase, and polygalacturonase convert a citrus processing waste into biofuel precursors. ACS Sustain. Chem. Eng. 2018, 6, 7304–7312. [Google Scholar] [CrossRef]
- Patidar, M.K.; Nighojkar, S.; Kumar, A.; Nighojkar, A. Pectinolytic enzymes-solid state fermentation, assay methods and applications in fruit juice industries: A review. 3 Biotech. 2018, 8, 199. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, M.R.; Widmer, W.W.; Grohmann, K. Simultaneous saccharification and fermentation of citrus peel waste by Saccharomyces cerevisiae to produce ethanol. Process. Biochem. 2007, 42, 1614–1619. [Google Scholar] [CrossRef]
- Kumar, D.; Kumar, S.S.; Kumar, J.; Kumar, O.; Mishra, S.V.; Kumar, R.; Malyan, S.K. Xylanases and their industrial applications: A review. Biochem. Cell Arch. 2017, 17, 353–360. [Google Scholar]
- Biely, P.; Singh, S.; Puchart, V. Towards enzymatic breakdown of complex plant xylan structures: State of the art. Biotechnol. Adv. 2016, 34, 1260–1274. [Google Scholar] [CrossRef] [PubMed]
- Juturu, V.; Wu, J.C. Microbial exo-xylanases: A mini review. Appl. Biochem. Biotechnol. 2014, 174, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Tao, N.G.; Shi, W.Q.; Liu, Y.J.; Huang, S.R. Production of feed enzymes from citrus processing waste by solid-state fermentation with Eupenicillium javanicum. Int. J. Food Sci. Technol. 2011, 46, 1073–1079. [Google Scholar] [CrossRef]
- Li, P.J.; Xia, J.L.; Shan, Y.; Nie, Z.Y.; Wang, F.R. Effects of surfactants and microwave-assisted pretreatment of orange peel on extracellular enzymes production by Aspergillus japonicus PJ01. Appl. Biochem. Biotechnol. 2015, 176, 758–771. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J.M.; Mulvaney, C.S. Nitrogen total. In Methods of Soil Analysis, Part 2: Chemical and Microbiological Properties; Agronomy Monograph No. 9; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; American Society of Agronomy and Soil Science Society of America: Madison, WI, USA, 1982; pp. 595–624. ISBN 0-89118-072-9. [Google Scholar]
- Anderson, J.M.; Ingram, J.S.I. Microbial biomass. In Tropical Soil Biology and Fertility: A Handbook of Methods, 2nd ed.; Anderson, J.M., Ingram, J.S.I., Eds.; CAB International: Wallingford, UK, 1993; pp. 68–70. ISBN 0-85198-821-0. [Google Scholar]
- Lowe, L.E. Total and labile polysaccharide analysis of soils. In Soil Sampling and Methods of Analysis; Carter, M.R., Ed.; Canadian Society of Soil Science, Lewis Publishers: Boca Raton, FL, USA, 1993; pp. 373–376. ISBN 0-87371-861-5. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- Brink, R.H.; Dubach, P.; Lynch, D.L. Measurement of carbohydrates in soil hydrolyzates with anthrone. Soil Sci. 1960, 89, 157–166. [Google Scholar] [CrossRef]
- Remmas, N.; Melidis, P.; Voltsi, C.; Athanasiou, D.; Ntougias, S. Novel hydrolytic extremely halotolerant alkaliphiles from mature landfill leachate with key involvement in maturation process. J. Environ. Sci. Health A Tox Hazard. Subst. Environ. Eng. 2017, 52, 64–73. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Baldrian, P.; Valášková, V.; Merhautová, V.; Gabriel, J. Degradation of lignocellulose by Pleurotus ostreatus in the presence of copper, manganese, lead and zinc. Res. Microbiol 2005, 156, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Camassola, M.; Dillon, A.J.P. Effect of different pretreatment of sugar cane bagasse on cellulase and xylanases production by the mutant Penicillium echinulatum 9A02S1 grown in submerged culture. Biomed. Res. Int. 2014, 2014, 720740. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Huang, X.; Madan, A. CAP3: A DNA sequence assembly program. Genome Res. 1999, 9, 868–877. [Google Scholar] [CrossRef] [PubMed]
- Sievers, F.; Wilm, A.; Dineen, D.; Gibson, T.J.; Karplus, K.; Li, W.; Lopez, R.; McWilliam, H.; Remmert, M.; Söding, J.; et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 2011, 7, 539. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Jukes, T.H.; Cantor, C.R. Evolution of protein molecules. In Mammalian Protein Metabolism; Munro, H.N., Ed.; Academic Press: New York, NY, USA, 1969; pp. 21–132. ISBN 0125106017. [Google Scholar]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef]
- Cole, J.R.; Wang, Q.; Fish, J.A.; Chai, B.; McGarrell, D.M.; Sun, Y.; Brown, C.T.; Porras-Alfaro, A.; Kuske, C.R.; Tiedje, J.M. Ribosomal Database Project: Data and tools for high throughput rRNA analysis. Nucl. Acids Res. 2014, 42, D633–D642. [Google Scholar] [CrossRef]
- Masella, A.P.; Bartram, A.K.; Truszkowski, J.M.; Brown, D.G.; Neufeld, J.D. PANDAseq: Paired-end assembler for Illumina sequences. BMC Bioinform. 2012, 13, 31. [Google Scholar] [CrossRef]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef]
- Nawrocki, E.P.; Kolbe, D.L.; Eddy, S.R. Infernal 10: Inference of RNA alignments. Bioinformatics 2009, 25, 1335–1337. [Google Scholar] [CrossRef] [PubMed]
- Magurran, A.E. Ecological Diversity and Its Measurement, 1st ed.; Princeton University Press: Princeton, NJ, USA, 1988; ISBN 978-94-015-7358-0. [Google Scholar]
- Maki, M.L.; Idrees, A.; Leung, K.T.; Qin, W. Newly isolated and characterized bacteria with great application potential for decomposition of lignocellulosic biomass. J. Mol. Microbiol. Biotechnol. 2012, 22, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Schulte, M.F.; Tozakidis, I.E.; Jose, J. Autotransporter-based surface display of hemicellulases on Pseudomonas putida: Whole-cell biocatalysts for the degradation of biomass. ChemCatChem 2017, 9, 3955–3964. [Google Scholar] [CrossRef]
- Wang, S.L.; Yen, Y.H.; Shih, L.; Chang, A.C.; Chang, W.T.; Wu, W.C.; Chai, Y.D. Production of xylanases from rice bran by Streptomyces actuosus A-151. Enzyme Microb Technol. 2003, 33, 917–925. [Google Scholar] [CrossRef]
- Adigüzel, A.O.; Tunçer, M. Production, characterization and application of a xylanase from Streptomyces sp. AOA40 in fruit juice and bakery industries. Food Biotechnol. 2016, 30, 189–218. [Google Scholar] [CrossRef]
- Rosmine, E.; Sainjan, N.C.; Silvester, R.; Alikkunju, A.; Varghese, S.A. Statistical optimisation of xylanase production by estuarine Streptomyces sp. and its application in clarification of fruit juice. J. Gen. Eng. Biotechnol. 2017, 15, 393–401. [Google Scholar] [CrossRef]
- Bibi, Z.; Ansari, A.; Zohra, R.R.; Aman, A.; Qader, S.A.U. Production of xylan degrading endo-1,4-β-xylanase from thermophilic Geobacillus stearothermophilus KIBGE-IB29. J. Radiat. Res. Appl. Sci. 2014, 7, 478–485. [Google Scholar] [CrossRef]
- Kumar, L.; Nagar, S.; Mittal, A.; Garg, N.; Gupta, V.K. Immobilization of xylanase purified from Bacillus pumilus VLK-1 and its application in enrichment of orange and grape juices. J. Food Sci. Technol. 2014, 51, 1737–1749. [Google Scholar] [CrossRef]
- Collins, T.; Gerday, C.; Feller, G. Xylanases, xylanase families and extremophilic xylanases. FEMS Microbiol. Rev. 2005, 29, 3–23. [Google Scholar] [CrossRef]
- Tanaka, T.; Shimomura, Y.; Himejima, M.; Taniguchi, M.; Oi, S. Characterization of xylan-utilizing anaerobes from mesophilic and thermophilic methane sludge and their xylan degrading enzymes. Agric. Biol. Chem. 1986, 50, 2185–2192. [Google Scholar] [CrossRef]
- Murty, M.V.S.; Chandra, T.S. Expression of xylanase and cellulase enzymes in a newly isolated Clostridium sp. SAIV. Enzyme Microb. Technol. 1991, 13, 430–435. [Google Scholar] [CrossRef]
- Mamma, D.; Kourtoglou, E.; Christakopoulos, P. Fungal multienzyme production on industrial by-products of the citrus-processing industry. Bioresour. Technol. 2008, 99, 2373–2383. [Google Scholar] [CrossRef] [PubMed]
- Terrasan, C.R.F.; Temer, B.; Duarte, M.C.T.; Carmona, E.C. Production of xylanolytic enzymes by Penicillium janczewskii. Bioresour. Technol. 2010, 101, 4139–4143. [Google Scholar] [CrossRef] [PubMed]
- Facchini, F.D.A.; Vici, A.C.; Reis, V.R.A.; Jorge, J.A.; Terenzi, H.F.; Reis, R.A.; de Moraes, M.D.L.T. Production of fibrolytic enzymes by Aspergillus japonicus C03 using agro-industrial residues with potential application as additives in animal feed. Bioprocess. Biosyst. Eng. 2011, 34, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Rezzadori, K.; Benedetti, S.; Amante, E.R. Proposals for the residues recovery: Orange waste as raw material for new products. Food Bioprod. Process. 2012, 90, 606–614. [Google Scholar] [CrossRef]
- Aneja, K.R.; Dhiman, R.; Aggarwal, N.K.; Kumar, V.; Kaur, M. Microbes associated with freshly prepared juices of citrus and carrots. Int. J. Food Sci. Technol. 2014, 2014, 408085. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; Gliniewicz, K.; Gerritsen, A.T.; McDonald, A.G. Analysis of microbial community variation during the mixed culture fermentation of agricultural peel wastes to produce lactic acid. Bioresour. Technol. 2016, 208, 7–12. [Google Scholar] [CrossRef]
- El Sheikha, A.F.; Levin, R.; Xu, J. Molecular Techniques in Food Biology, 1st ed.; John Wiley & Sons Ltd.: New York, NY, USA, 2018; ISBN 9781119374633. [Google Scholar]
- Nelson, D.M.; Glawe, A.J.; Labeda, D.P.; Cann, I.K.; Mackie, R.I. Paenibacillus tundrae sp. nov. and Paenibacillus xylanexedens sp. nov., psychrotolerant, xylan-degrading bacteria from Alaskan tundra. Int. J. Syst. Evol. Microbiol. 2009, 59, 1708–1714. [Google Scholar] [CrossRef]
- Kitahara, M.; Takamine, F.; Imamura, T.; Benno, Y. Clostridium hiranonis sp. nov., a human intestinal bacterium with bile acid 7alpha-dehydroxylating activity. Int. J. Syst. Evol. Microbiol. 2001, 51, 39–44. [Google Scholar] [CrossRef]
- Belduz, A.O.; Dulger, S.; Demirbag, Z. Anoxybacillus gonensis sp. nov., a moderately thermophilic, xylose-utilizing, endospore-forming bacterium. Int. J. Syst. Evol. Microbiol. 2003, 53, 1315–1320. [Google Scholar] [CrossRef]
- Bouvet, P.J.M.; Grimont, P.A.D. Taxonomy of the genus Acinetobacter with the recognition of Acinetobacter baumannii sp. nov., Acinetobacter haemolyticus sp. nov., Acinetobacter johnsonii sp. nov., and Acinetobacter junii sp. nov. and emended descriptions of Acinetobacter calcoaceticus and Acinetobacter lwoffii. Int. J. Syst. Bacteriol. 1986, 36, 228–240. [Google Scholar] [CrossRef]
- Abraham, W.R.; Strompl, C.; Meyer, H.; Lindholst, S.; Moore, E.R.; Christ, R.; Vancanneyt, M.; Tindall, B.J.; Bennasar, A.; Smit, J.; et al. Phylogeny and polyphasic taxonomy of Caulobacter species. Proposal of Maricaulis gen. nov. with Maricaulis maris (Poindexter) comb. nov. as the type species, and emended description of the genera Brevundimonas and Caulobacter. Int. J. Syst. Bacteriol. 1999, 49, 1053–1073. [Google Scholar] [CrossRef] [PubMed]
- Arteau, M.; Labrie, S.; Roy, D. Terminal-restriction fragment length polymorphism and automated ribosomal intergenic spacer analysis profiling of fungal communities in Camembert cheese. Int Dairy J. 2010, 20, 545–554. [Google Scholar] [CrossRef]
- Vu, D.; Groenewald, M.; de Vries, M.; Gehrmann, T.; Stielow, B.; Eberhardt, U.; Al-Hatmi, A.; Groenewald, J.Z.; Cardinali, G.; Houbraken, J.; et al. Large-scale generation and analysis of filamentous fungal DNA barcodes boosts coverage for kingdom fungi and reveals thresholds for fungal species and higher taxon delimitation. Stud. Mycol. 2019, 92, 135–154. [Google Scholar] [CrossRef] [PubMed]
- Bensch, K.; Groenewald, J.Z.; Braun, U.; Dijksterhuis, J.; de Jesus Yanez-Morales, M.; Crous, P.W. Common but different: The expanding realm of Cladosporium. Stud. Mycol. 2015, 82, 23–74. [Google Scholar] [CrossRef]
- Crous, P.W.; Summerell, B.A.; Swart, L.; Denman, S.; Taylor, J.E.; Bezuidenhout, C.M.; Palm, M.E.; Marincowitz, S.; Groenewald, J.Z. Fungal pathogens of Proteaceae. Persoonia 2011, 27, 20–45. [Google Scholar] [CrossRef]
| Parameter | Mean ± SE (n = 3) |
|---|---|
| pH * | 4.00 ± 0.01 |
| EC (mS/cm) * | 1.60 ± 1.02 |
| Dry weight content (%) | 14.18 ± 0.90 |
| Total organic matter (% w/w) | 85.95 ± 1.78 |
| Carbon (% w/w) | 49.85 ± 1.03 |
| Ash content (% w/w) | 14.05 ± 1.78 |
| Total nitrogen (% w/w) | 1.21 ± 0.03 |
| Total soluble carbohydrates (% w/w) | 28.16 ± 1.38 |
| Anthrone-reactive C (% w/w) | 9.63 ± 0.99 |
| Total phosphorus (mg/g d.w.) | 2.38 ± 0.10 |
| Total soluble phenolics (mg/g d.w.) | 77.4 ± 4.0 |
| Ecological Indicator Index | Bacterial Community | Fungal Community |
|---|---|---|
| Chao’s richness index | 29.44 ± 0.89 | 43.67 ± 2.73 |
| Shannon diversity index | 0.59 ± 0.02 | 1.15 ± 0.04 |
| Pielou’s Evenness index | 0.18 ± 0.01 | 0.32 ± 0.01 |
| Xylanolytic Activity | Lysate | Bulk/Surface | p-Value |
|---|---|---|---|
| Endo-1,4-β-xylanase activity (U/g d.w.) | 5.21 ± 0.59 | 4.77 ± 0.11 | 0.509 |
| 1,4-β-xylosidase activity (U/g d.w.) | 3.41 ± 0.14 | 2.50 ± 0.13 | 0.009 |
| Taxon | 16S rRNA Gene Accession Number of the Closest Species | Closest Species | Identities | Reference |
| Potential xylan-degrading bacteria | ||||
| g__Paenibacillus | MF347934 | P. xylanexedens | 370/370 (100%) | [55] |
| g__Clostridium | NR_028611 | C. hiranonis | 350/396 (88.4%) | [56] |
| g__Anoxybacillus | CP012152 | A. gonensis | 392/396 (99.0%) | [57] |
| g__Acinetobacter | NR_113346 | A. lwoffii | 394/395 (99.7%) | [58] |
| g__Caulobacter | NR_074208 | C. segnis | 393/394 (99.7%) | [59] |
| Taxon | ITS-5.8S rRNA Gene Accession Number of the Closest Species | Closest Species | Identities | Reference |
| Potential xylan-degrading fungi | ||||
| g__Pichia | NR_130688 | P. fermentans | 173/174 (99.4%) | [60] |
| g__Penicillium | MH856375 | P. italicum | 267/267 (100%) | [61] |
| g__Cladosporium | NR_152286 | C. angustiherbarum | 250/250 (100%) | [62] |
| g__Aureobasidium | NR_156246 | A. leucospermi | 275/275 (100%) | [63] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zerva, I.; Remmas, N.; Ntougias, S. Diversity and Biotechnological Potential of Xylan-Degrading Microorganisms from Orange Juice Processing Waste. Water 2019, 11, 274. https://doi.org/10.3390/w11020274
Zerva I, Remmas N, Ntougias S. Diversity and Biotechnological Potential of Xylan-Degrading Microorganisms from Orange Juice Processing Waste. Water. 2019; 11(2):274. https://doi.org/10.3390/w11020274
Chicago/Turabian StyleZerva, Ioanna, Nikolaos Remmas, and Spyridon Ntougias. 2019. "Diversity and Biotechnological Potential of Xylan-Degrading Microorganisms from Orange Juice Processing Waste" Water 11, no. 2: 274. https://doi.org/10.3390/w11020274
APA StyleZerva, I., Remmas, N., & Ntougias, S. (2019). Diversity and Biotechnological Potential of Xylan-Degrading Microorganisms from Orange Juice Processing Waste. Water, 11(2), 274. https://doi.org/10.3390/w11020274







