Halloysite Nanotubes as Adsorptive Material for Phosphate Removal from Aqueous Solution
Abstract
1. Introduction
2. Materials and Methods
2.1. Adsorbent Characterizations and Preparations
2.2. Classical Batch Experiments
2.3. Kinetic Experiments and Data Presentations
2.4. Kinetic Experiments and Data Presentations
3. Results and Discussions
3.1. Morphology and Characterization of HNTs
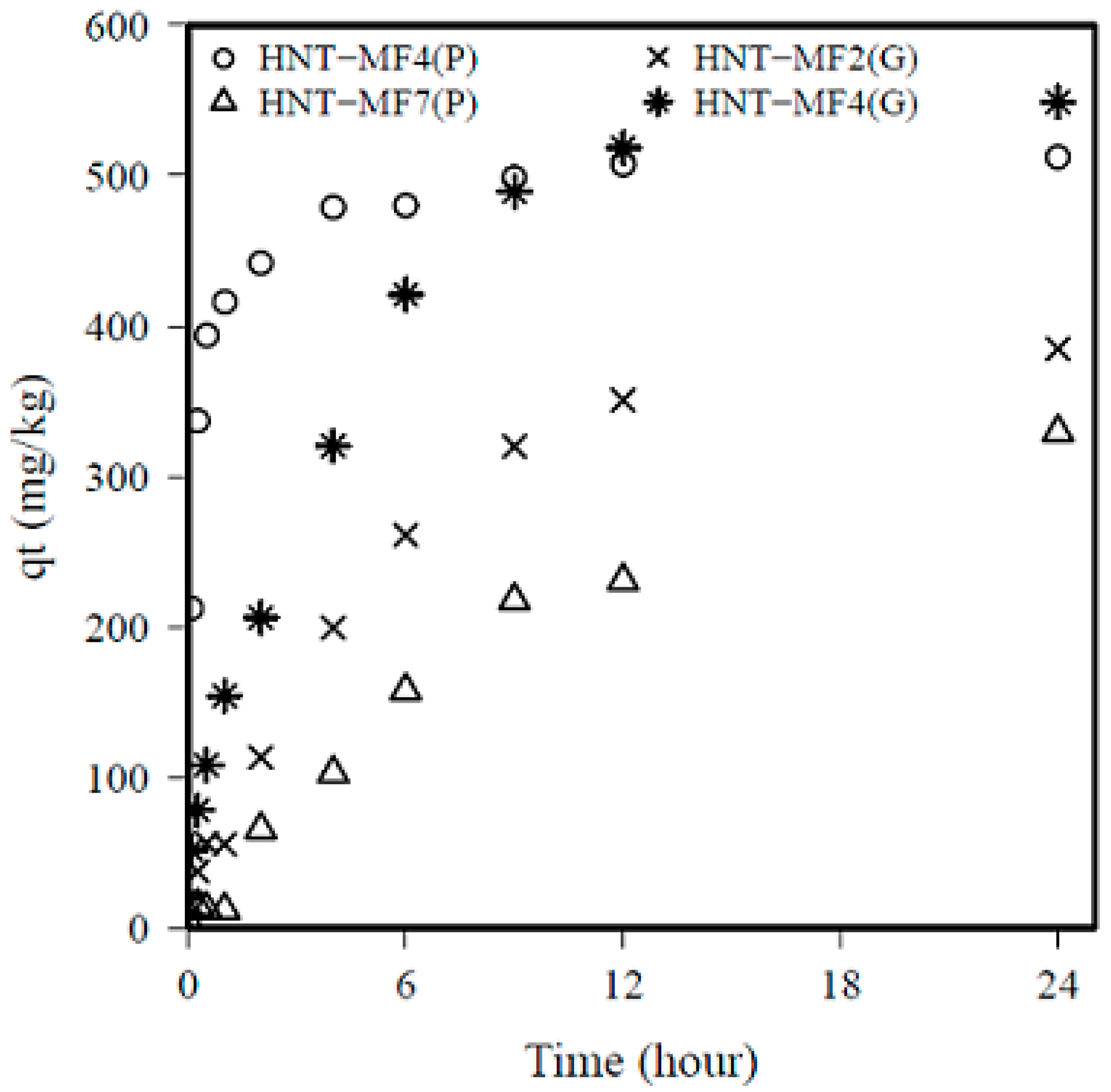
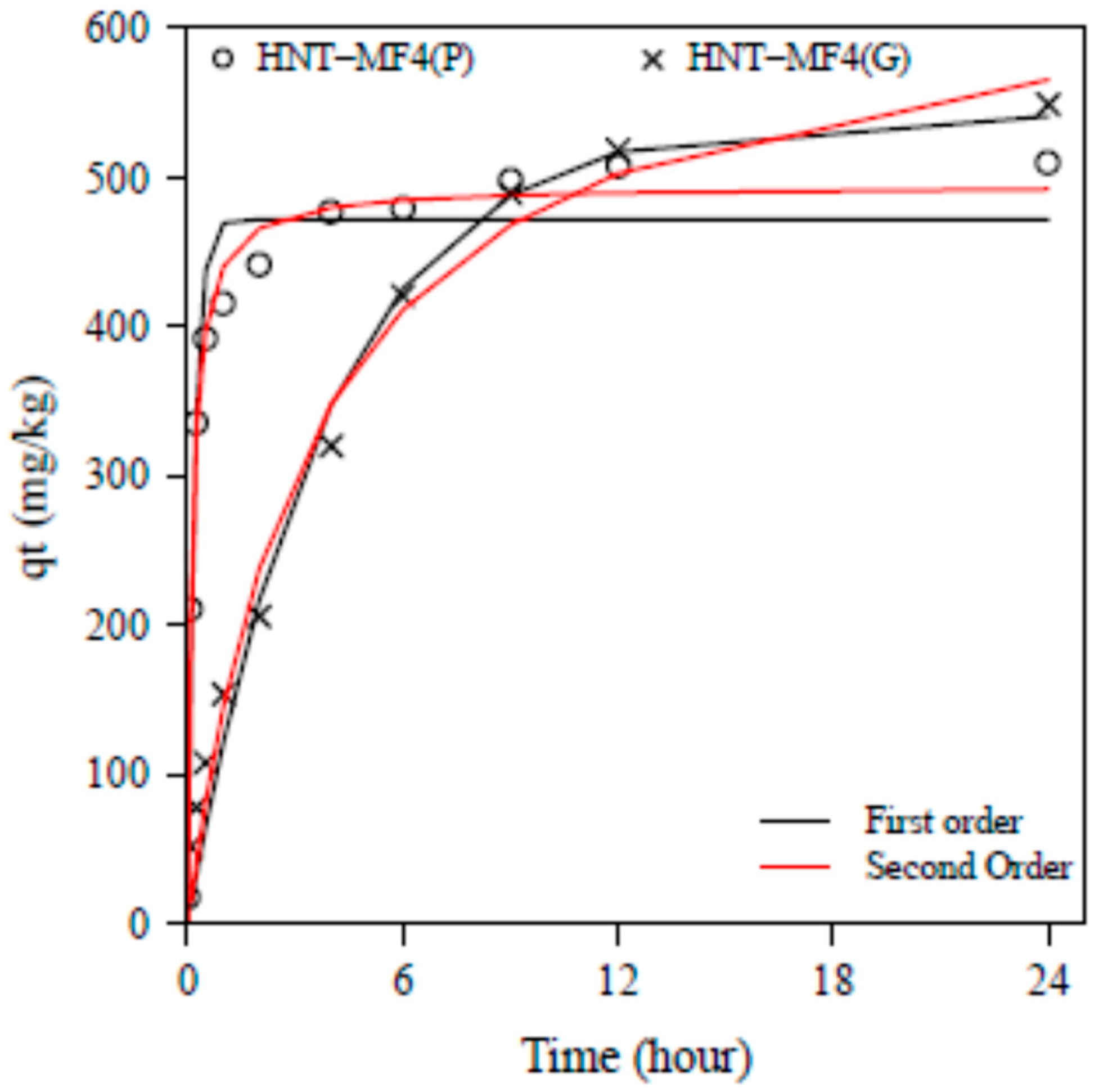
3.2. Phosphate Adsorption Kinetics
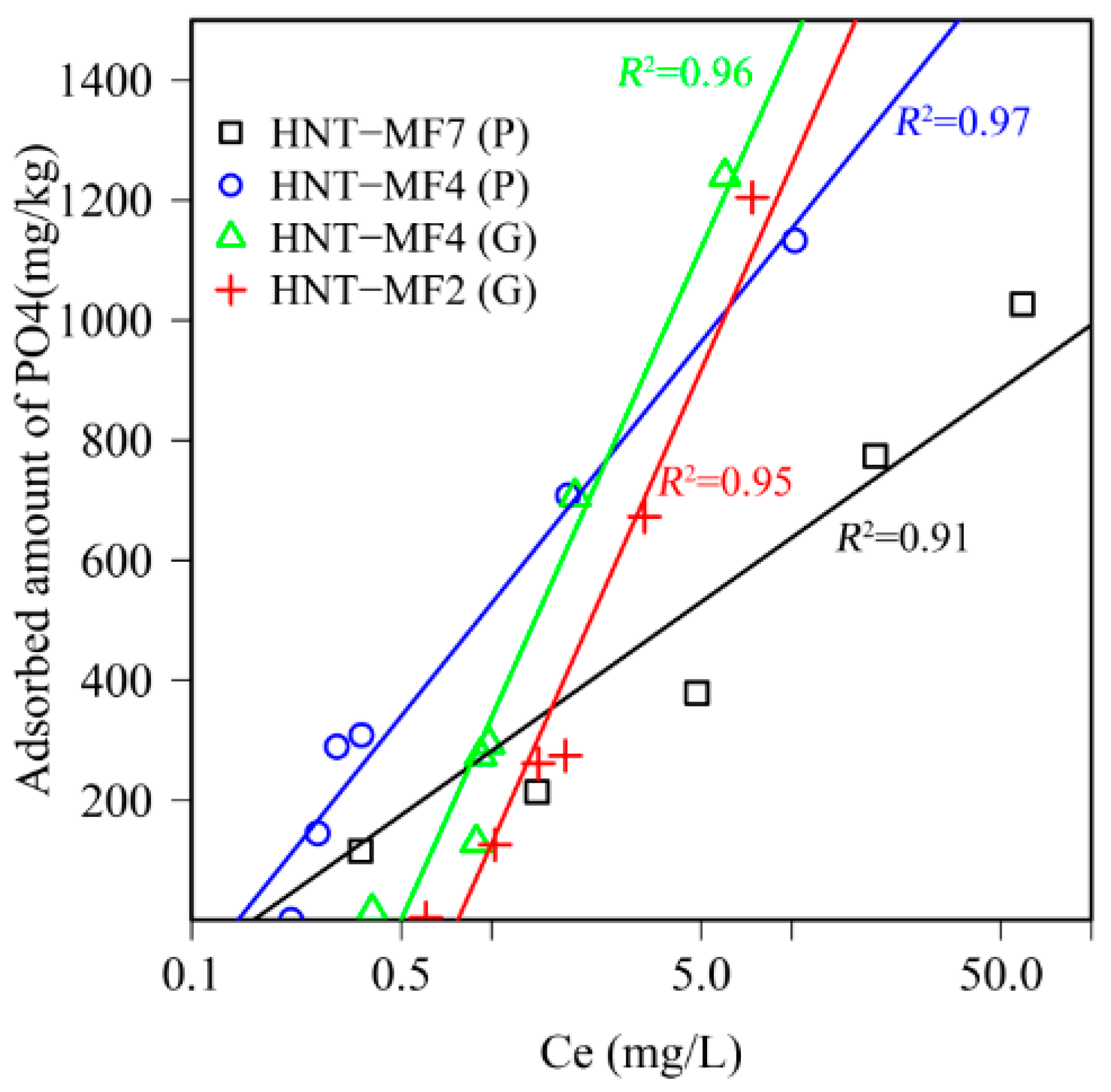
3.3. Adsorption Isotherms
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tabbara, H. Phosphorus loss to runoff water twenty-four hours after application of liquid swine manure or fertilizer. J. Environ. Qual. 2003, 32, 1044–1052. [Google Scholar] [CrossRef] [PubMed]
- Tarkalson, D.D.; Mikkelsen, R.L. Runoff Phosphorus Losses as Related to Phosphorus Source, Application Method, and Application Rate on a Piedmont Soil. J. Environ. Qual. 2004, 33, 1424–1430. [Google Scholar] [CrossRef] [PubMed]
- Daverede, I.C.; Kravchenko, A.N.; Hoeft, R.G.; Nafziger, E.D.; Bullock, D.G.; Warren, J.J.; Gonzini, L.C. Phosphorus Runoff from Incorporated and Surface-Applied Liquid Swine Manure and Phosphorus Fertilizer. J. Environ. Qual. 2004, 33, 1535–1544. [Google Scholar] [CrossRef]
- Hart, M.R.; Quin, B.F.; Nguyen, M.L. Phosphorus Runoff from Agricultural Land and Direct Fertilizer Effects. J. Environ. Qual. 2004, 33, 1954–1972. [Google Scholar] [CrossRef] [PubMed]
- Boesch, D.F.; Brinsfield, R.B.; Magnien, R.E. Chesapeake Bay eutrophication: Scientific understanding, ecosystem restoration, and challenges for agriculture. J. Environ. Qual. 2001, 30, 303–320. [Google Scholar] [CrossRef]
- Márquez-Pacheco, H.; Hansen, A.M.; Falcón-Rojas, A. Phosphorous control in a eutrophied reservoir. Environ. Sci. Pollut. Res. 2013, 20, 8446–8456. [Google Scholar] [CrossRef] [PubMed]
- Duranceau, S.J.; Biscardi, P.G.; Barnhill, D.K. Screening the toxicity of phosphorous-removal adsorbents using a bioluminescence inhibition test. Environ. Toxicol. 2016, 31, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Southam, D.C.; Lewis, T.W.; McFarlane, A.J.; Johnston, J.H. Amorphous calcium silicate as a biosorbent for phosphate. Curr. Appl. Phys. 2004, 4, 355–358. [Google Scholar] [CrossRef]
- Lewis, W.M.; Wurtsbaugh, W.A.; Paerl, H.W. Rationale for control of anthropogenic nitrogen and phosphorus to reduce eutrophication of inland waters. Environ. Sci. Technol. 2011, 45, 10300–10305. [Google Scholar] [CrossRef] [PubMed]
- Farmer, A.M. Reducing phosphate discharges: The role of the 1991 EC urban wastewater treatment directive. Water Sci. Technol. 2001, 44, 41–48. [Google Scholar] [CrossRef]
- Jones, E.V. Phosphorus in Environmental Technologies: Principles and Applications; IWA Publishing: London, UK, 2004; ISBN 9781843390015. [Google Scholar]
- Shilton, A.N.; Elmetri, I.; Drizo, A.; Pratt, S.; Haverkamp, R.G.; Bilby, S.C. Phosphorus removal by an “active” slag filter-a decade of full scale experience. Water Res. 2006, 40, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Gao, R.; Liu, M.; Yan, C.; Shan, A. Adsorption of modified halloysite nanotubes in vitro and the protective effect in rats exposed to zearalenone. Arch. Anim. Nutr. 2014, 68, 320–335. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Gao, B.; Inyang, M.; Zimmerman, A.R.; Cao, X.; Pullammanappallil, P.; Yang, L. Removal of phosphate from aqueous solution by biochar derived from anaerobically digested sugar beet tailings. J. Hazard. Mater. 2011, 190, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Carrott, P.J.M.; Ribeiro Carrott, M.M.L. Suhas Low-Cost adsorbents: Growing approach to wastewater treatmenta review. Crit. Rev. Environ. Sci. Technol. 2009, 39, 783–842. [Google Scholar] [CrossRef]
- Johansson Westholm, L. Substrates for phosphorus removal—Potential benefits for on-site wastewater treatment? Water Res. 2006, 40, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Hylander, L.D.; Kietlińska, A.; Renman, G.; Simán, G. Phosphorus retention in filter materials for wastewater treatment and its subsequent suitability for plant production. Bioresour. Technol. 2006, 97, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Cucarella, V.; Zaleski, T.; Mazurek, R.; Renman, G. Effect of reactive substrates used for the removal of phosphorus from wastewater on the fertility of acid soils. Bioresour. Technol. 2008, 99, 4308–4314. [Google Scholar] [CrossRef] [PubMed]
- Joussein, E.; Petit, S.; Churchman, J.; Theng, B.; Righi, D.; Delvaux, B. Halloysite clay minerals—A review. Clay Miner. 2005, 63, 383–426. [Google Scholar] [CrossRef]
- Prashantha Kumar, T.K.M.; Mandlimath, T.R.; Sangeetha, P.; Revathi, S.K.; Ashok Kumar, S.K. Nanoscale materials as sorbents for nitrate and phosphate removal from water. Environ. Chem. Lett. 2018, 16, 389–400. [Google Scholar] [CrossRef]
- Cataldo, S.; Lazzara, G.; Massaro, M.; Muratore, N.; Pettignano, A.; Riela, S. Functionalized halloysite nanotubes for enhanced removal of lead(II) ions from aqueous solutions. Appl. Clay Sci. 2018, 156, 87–95. [Google Scholar] [CrossRef]
- Baraka, A.M.; El-Tayieb, M.M.; Shafai, M.E.; Mohamed, N.Y. Sorptive Removal of Phosphate from Wastewater Using Activated Red Mud 1. Aust. J. Basic Appl. Sci. 2012, 6, 500–510. [Google Scholar]
- Liu, Q.; Guo, L.; Zhou, Y.; Dai, Y.; Feng, L.; Zhou, J.; Zhao, J.; Liu, J.; Qian, G. Phosphate adsorption on biogenetic calcium carbonate minerals: Effect of a crystalline phase. Desalin. Water Treat. 2012, 47, 78–85. [Google Scholar] [CrossRef]
- You, X.; Guaya, D.; Farran, A.; Valderrama, C.; Cortina, J.L. Phosphate removal from aqueous solution using a hybrid impregnated polymeric sorbent containing hydrated ferric oxide (HFO). J. Chem. Technol. Biotechnol. 2016, 91, 693–704. [Google Scholar] [CrossRef]
- Glocheux, Y.; Pasarín, M.M.; Albadarin, A.B.; Mangwandi, C.; Chazarenc, F.; Walker, G.M. Phosphorus adsorption onto an industrial acidified laterite by-product: Equilibrium and thermodynamic investigation. Asia-Pac. J. Chem. Eng. 2014, 9, 929–940. [Google Scholar] [CrossRef]
- Prochaska, C.A.; Zouboulis, A.I. Removal of phosphates by pilot vertical-flow constructed wetlands using a mixture of sand and dolomite as substrate. Ecol. Eng. 2006, 26, 293–303. [Google Scholar] [CrossRef]
- Ragheb, S.M. Phosphate removal from aqueous solution using slag and fly ash. HBRC J. 2013, 9, 270–275. [Google Scholar] [CrossRef]
- Barca, C.; Gérente, C.; Meyer, D.; Chazarenc, F.; Andrès, Y. Phosphate removal from synthetic and real wastewater using steel slags produced in Europe. Water Res. 2012, 46, 2376–2384. [Google Scholar] [CrossRef]
- Zhang, J.; Shen, Z.; Mei, Z.; Li, S.; Wang, W. Removal of phosphate by Fe-coordinated amino-functionalized 3D mesoporous silicates hybrid materials. J. Environ. Sci. 2011, 23, 199–205. [Google Scholar] [CrossRef]
- Eskandarpour, A.; Sassa, K.; Bando, Y.; Okido, M.; Asai, S. Magnetic Removal of Phosphate from Wastewater Using Schwertmannite. Mater. Trans. 2006, 47, 1832–1837. [Google Scholar] [CrossRef]
- Fetene, Y. Design Phosphate Removal Technology using Volcanic Rocks: Kinetics and Equilibrium Studies. Int. J. Eng. Res. Technol. 2015, 4, 155–162. [Google Scholar] [CrossRef]
- Mallikarjun, S.D.; Mise, S.R. A Batch Study of P Hosphate Adsorption Characteristics on Clay Soil. Int. J. Res. Eng. Technol. 2013, 1, 338–342. [Google Scholar]
- Dong, Y.; Liu, Z.; Chen, L. Removal of Zn(II) from aqueous solution by natural halloysite nanotubes. J. Radioanal. Nucl. Chem. 2012, 292, 435–443. [Google Scholar] [CrossRef]
- Cataldo, S.; Muratore, N.; Orecchio, S.; Pettignano, A. Enhancement of adsorption ability of calcium alginate gel beads towards Pd(II) ion. A kinetic and equilibrium study on hybrid Laponite and Montmorillonite-alginate gel beads. Appl. Clay Sci. 2015, 118, 162–170. [Google Scholar] [CrossRef]
- Matusik, J. Halloysite for Adsorption and Pollution Remediation. Dev. Clay Sci. 2016, 7, 606–627. [Google Scholar] [CrossRef]
- Yuan, P.; Tan, D.; Annabi-Bergaya, F. Properties and applications of halloysite nanotubes: Recent research advances and future prospects. Appl. Clay Sci. 2015, 112–113, 75–93. [Google Scholar] [CrossRef]
- Pasbakhsh, P.; Churchman, G.J.; Keeling, J.L. Characterisation of properties of various halloysites relevant to their use as nanotubes and microfibre fillers. Appl. Clay Sci. 2013, 74, 47–57. [Google Scholar] [CrossRef]
- Furumi, S.; Uchikoshi, T.; Shirahata, N.; Suzuki, T.S.; Sakka, Y. Aqueous dispersions of carbon nanotubes stabilized by zirconium acetate. Nanosci. Nanotechnol. 2009, 9, 662–665. [Google Scholar] [CrossRef]
- Singh, B.; Gilkes, R.J. An electron optical investigation of the alteration of kaolinite to halloysite. Clays Clay Miner. 1992, 40, 212–229. [Google Scholar] [CrossRef]
- Yuan, P.; Thill, A.; Bergaya, F. Nanosized Tubular Clay Minerals: Halloysite and Imogolite; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Zhang, S.; Shao, T. The Impacts of Aggregation and Surface Chemistry of Carbon Nanotubes on the Adsorption of Synthetic Organic Compounds. Environ. Sci. Technol. 2009, 43, 5719–5725. [Google Scholar] [CrossRef]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in nature waters. Anal. Chem. ACTA 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Panuccio, M.R.; Sorgonà, A.; Rizzo, M.; Cacco, G. Cadmium adsorption on vermiculite, zeolite and pumice: Batch experimental studies. J. Environ. Manag. 2009, 90, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.Q.; Cooper, C.; Ouki, S. Comparison of modified montmorillonite adsorbents Part I: Preparation, characterization and phenol adsorption. Chemosphere 2002, 47, 711–716. [Google Scholar] [CrossRef]
- Alemayehu, E.; Lennartz, B. Adsorptive removal of nickel from water using volcanic rocks. Appl. Geochem. 2010, 25, 1595–1602. [Google Scholar] [CrossRef]
- Loganathan, P.; Vigneswaren, S.; Kandasamy, J.; Bolan, N.S. Removal and recovery of phosphate from water using sorption. Environ. Sci. Technol. 2014, 44, 847–907. [Google Scholar] [CrossRef]

| Adsorbent | Al2O3 (%) | Fe2O3 (%) | Specific Surface Area (m2 g−1) | Morphology | ||||
|---|---|---|---|---|---|---|---|---|
| Length (µm) | Diameter (nm) | Aspect Ratio | Grain Size | |||||
| Inner | Outer | |||||||
| HNT-MF2 | 25.0 | 23.0 | 35 | 0.3–1.0 | 25 | 120 | 8 | <2 µm |
| HNT-MF4 | 37.5 | 0.4 | 25 | 0.6–1.2 | 20 | 100 | 10 | <7 µm |
| HNT-MF7 | 35.9 | 1.3 | 25 | 0.5–3.5 | 15 | 60 | 30 | <7 µm |
| Model | Adsorbent Halloysite Nanotubes | ||||
|---|---|---|---|---|---|
| HNT-MF2 (G) | HNT-MF4 (P) | HNT-MF7 (P) | HNT-MF4 (G) | ||
| Pseudo-first order | qe experiment | 384.5 | 511.6 | 329.5 | 549.0 |
| qe model | 369.9 | 472.1 | 328.5 | 541.1 | |
| K1 | 0.19 | 5.25 | 0.087 | 0.256 | |
| RMSE | 440.1 | 1459 | 143.9 | 1077 | |
| R2 | 0.98 | 0.87 | 0.99 | 0.99 | |
| Pseudo-second order | qe model | 384.6 | 492.3 | 331.9 | 565.6 |
| K2 | 0.0004 | 0.016 | 0.0001 | 0.0004 | |
| RMSE | 260.1 | 251.8 | 136.4 | 589.6 | |
| R2 | 0.98 | 0.97 | 0.99 | 0.98 | |
| Adsorbent | Freundlich | Langmuir | ||||||
|---|---|---|---|---|---|---|---|---|
| RMSE | RMSE | |||||||
| HNT-MF2(G) | 380 | 0.934 | 112 | 0.982 | 0.918 | 184 | 438 | 0.978 |
| HNT-MF4(P) | 334 | 0.848 | 203 | 0.977 | 0.865 | 61.2 | 621 | 0.798 |
| HNF-MF4(G) | 415 | 0.968 | 153 | 0.983 | 0.937 | 204 | 215 | 0.966 |
| HNT-MF7(P) | 207 | 0.404 | 311 | 0.964 | 0.564 | 59.4 | 976 | 0.727 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saki, H.; Alemayehu, E.; Schomburg, J.; Lennartz, B. Halloysite Nanotubes as Adsorptive Material for Phosphate Removal from Aqueous Solution. Water 2019, 11, 203. https://doi.org/10.3390/w11020203
Saki H, Alemayehu E, Schomburg J, Lennartz B. Halloysite Nanotubes as Adsorptive Material for Phosphate Removal from Aqueous Solution. Water. 2019; 11(2):203. https://doi.org/10.3390/w11020203
Chicago/Turabian StyleSaki, Hermin, Esayas Alemayehu, Joachim Schomburg, and Bernd Lennartz. 2019. "Halloysite Nanotubes as Adsorptive Material for Phosphate Removal from Aqueous Solution" Water 11, no. 2: 203. https://doi.org/10.3390/w11020203
APA StyleSaki, H., Alemayehu, E., Schomburg, J., & Lennartz, B. (2019). Halloysite Nanotubes as Adsorptive Material for Phosphate Removal from Aqueous Solution. Water, 11(2), 203. https://doi.org/10.3390/w11020203





