Efficient Nitrogen Removal of Reject Water Generated from Anaerobic Digester Treating Sewage Sludge and Livestock Manure by Combining Anammox and Autotrophic Sulfur Denitrification Processes
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Status
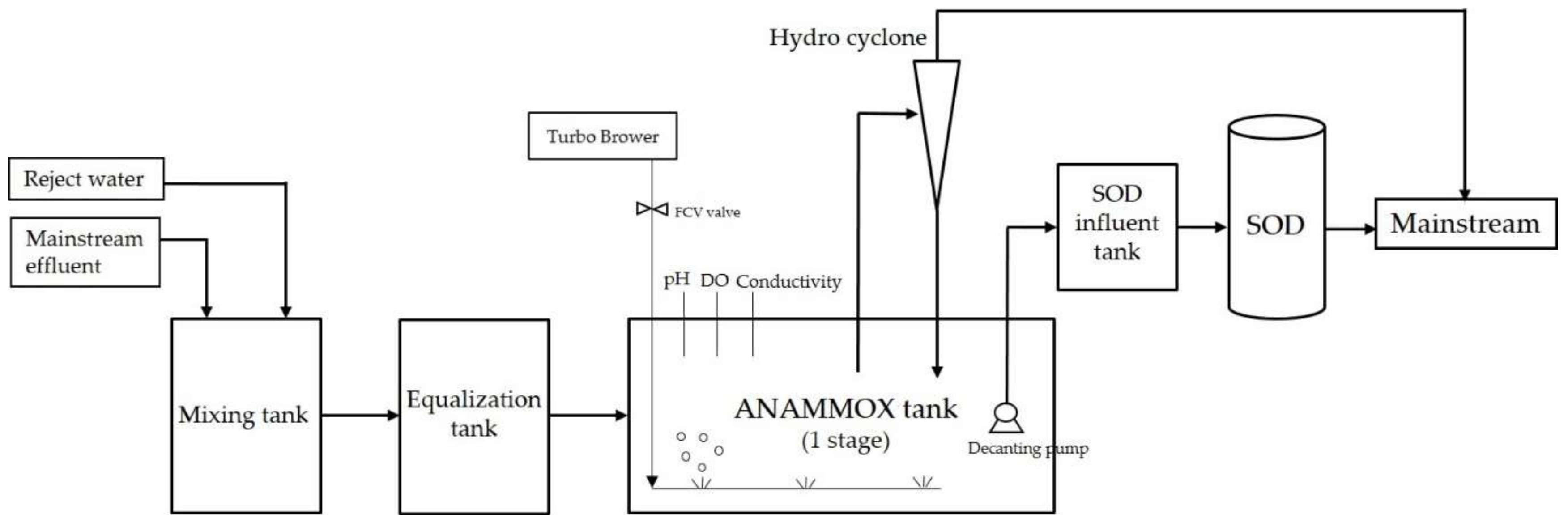
2.2. Reactor Setup and Operating Methods
2.3. Inoculation
2.4. Analytical Methods for Liquid Samples
2.5. Analytical Methods for Microbial Community Samples
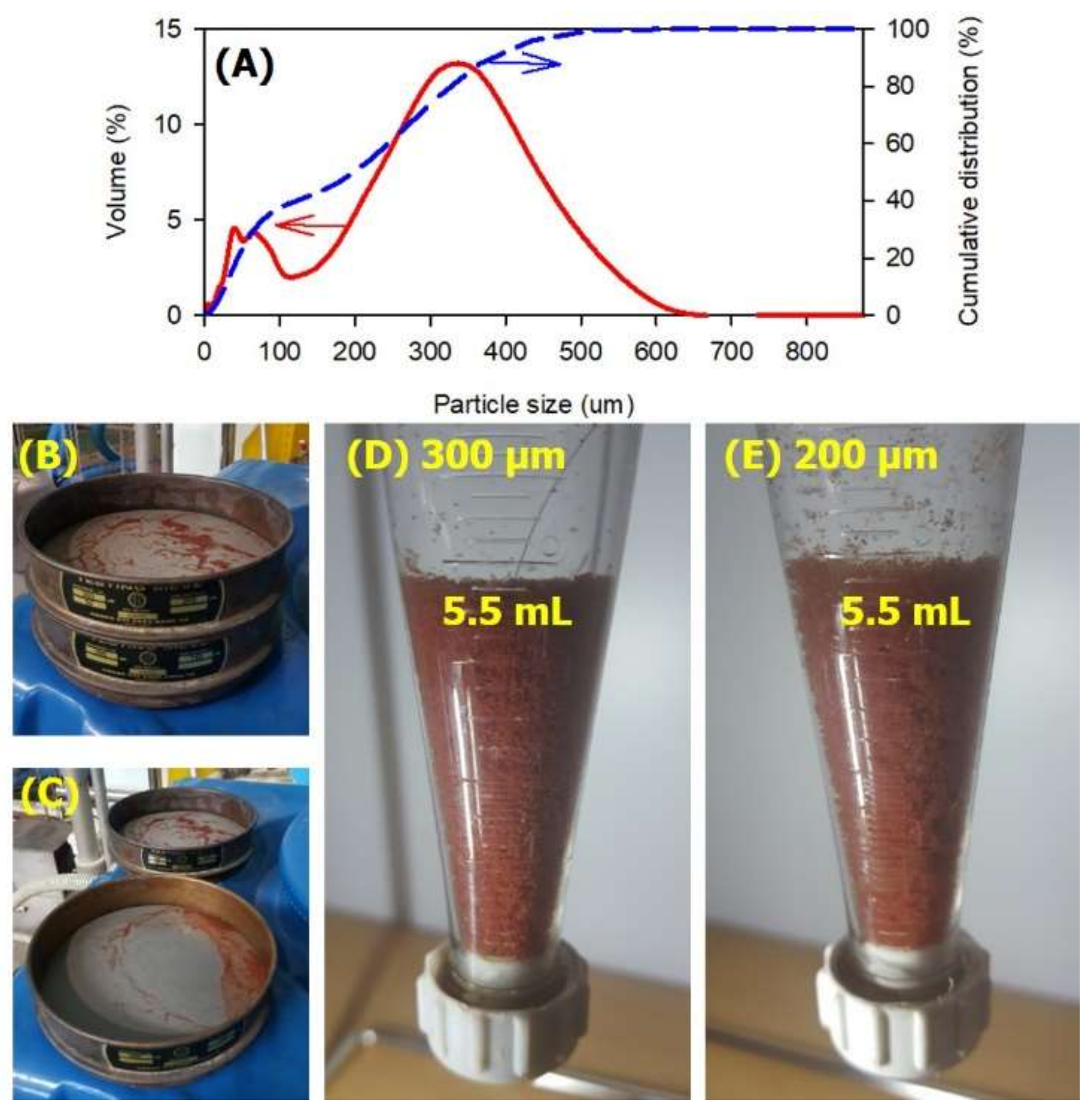
2.6. Particle Size and Morphological Analysis
3. Results and Discussion
3.1. Nitrogen Removal Performance of Anammox Process
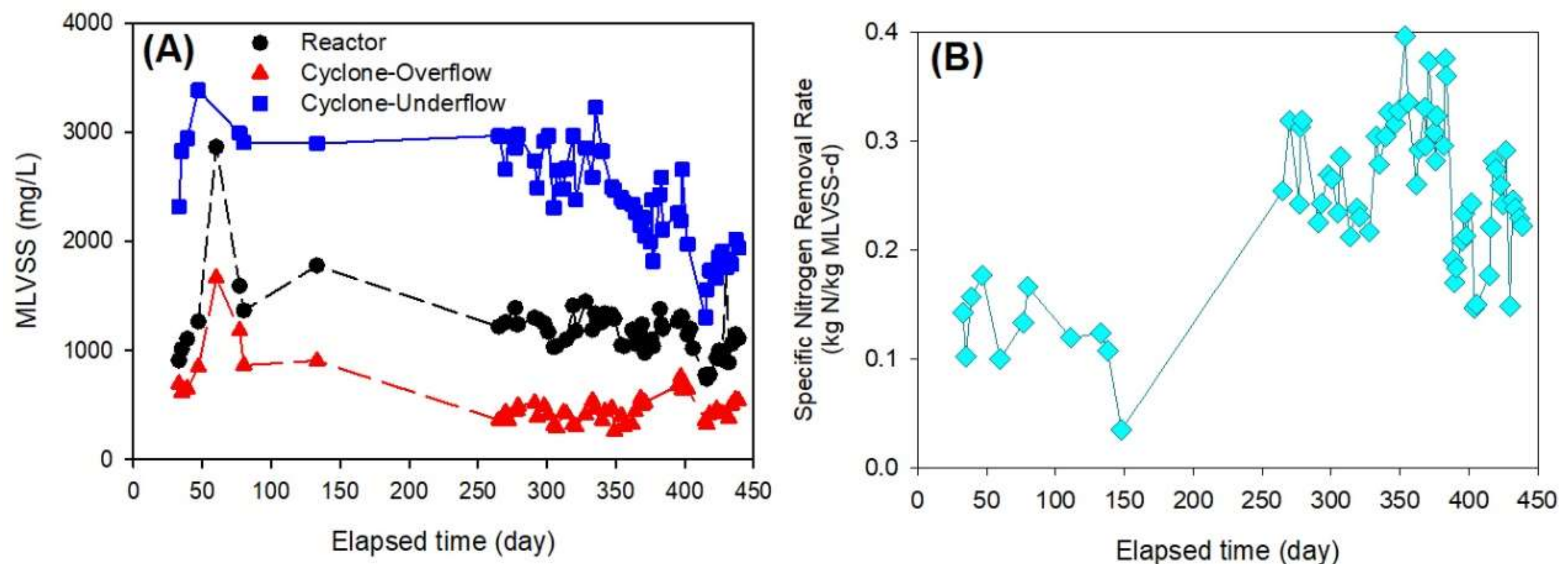
3.2. Anammox Process Problems and its Improvement
3.3. Feasibility and Application of Combined Anammox and the SOD Process
3.4. Nitrogen Removal Profile for Whole System
3.5. Changes in Microbial Communities
4. Conclusions
- The reject water was diluted with mainstream effluent and was used as the influent to the anammox reactor. The maximum TN removal efficiency of 80% was achieved for the anammox reactor under NLR of 0.45 kg-N/m3·d.
- As a result of decreasing dilution, influent conductivity and NLR values were increased to 7.8 mS/cm and 0.7 kg/m3·d, causing a rapid decrease in TN removal efficiency.
- The sludge concentration from the hydro-cyclone overflow was about 40 mg-MLVSS/L in which a small size of the anammox granules was detected. It was proven that an increase in MLVSS concentration in the anammox reactor was not easy under high influent conductivity and NLR.
- 97% of NO2−-N+NO3−-N generated from the anammox process could be removed successfully by the SOD reactor. A TN removal efficiency of 35% under poor annamox treatment could increase to 67% by applying the SOD reactor as a post treatment for the removal of NO3−-N.
- The dominant anammox bacteria in the anammox reactor was identified as Brocadia fulgida and 9.3% of the bacteria out of the total bacteria were anammox bacteria. Accompanied with the continuing operation, the population of anammox bacteria was increased by about 1.7 times compared to the initial inoculation stage.
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Li, D.; Kim, M.; Kim, H.; Choi, O.; Sang, B.-I.; Chiang, P.C.; Kim, H. Evaluation of relationship between biogas production and microbial communities in anaerobic co-digestion. Korean J. Chem. Eng. 2018, 35, 179–186. [Google Scholar] [CrossRef]
- Shen, Y.; Linville, J.L.; Urgun-Demirtas, M.; Mintz, M.M.; Snyder, S.W. An overview of biogas production and utilization at full-scale wastewater treatment plants (WWTPs) in the United States: Challenges and opportunities towards energy-neutral WWTPs. Renew. Sustain. Energy Rev. 2015, 50, 346–362. [Google Scholar] [CrossRef]
- Kartal, B.; Kuenen, J.G.; van Loosdrecht, M. Sewage treatment with anammox. Science 2010, 328, 702–703. [Google Scholar] [CrossRef] [PubMed]
- McCarty, P.L.; Bae, J.; Kim, J. Domestic wastewater treatment as a net energy producer–can this be achieved? Environ. Sci. Technol. 2011, 45, 7100–7106. [Google Scholar] [CrossRef] [PubMed]
- Schaubroeck, T.; De Clippeleir, H.; Weissenbacher, N.; Dewulf, J.; Boeckx, P.; Vlaeminck, S.E.; Wett, B. Environmental sustainability of an energy self-sufficient sewage treatment plant: improvements through DEMON and co-digestion. Water Res. 2015, 74, 166–179. [Google Scholar] [CrossRef] [PubMed]
- Siegrist, H.; Salzgeber, D.; Eugster, J.; Joss, A. Anammox brings WWTP closer to energy autarky due to increased biogas production and reduced aeration energy for N-removal. Water Sci. Technol. 2008, 57, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Van Loosdrecht, M.; Hao, X.; Jetten, M.; Abma, W. Use of Anammox in urban wastewater treatment. Water Sci. Technol. Water Supply 2004, 4, 87–94. [Google Scholar] [CrossRef]
- Lackner, S.; Gilbert, E.M.; Vlaeminck, S.E.; Joss, A.; Horn, H.; van Loosdrecht, M.C. Full-scale partial nitritation/anammox experiences—An application survey. Water Res. 2014, 55, 292–303. [Google Scholar] [CrossRef]
- Van der Star, W.R.; Abma, W.R.; Blommers, D.; Mulder, J.-W.; Tokutomi, T.; Strous, M.; Picioreanu, C.; van Loosdrecht, M.C. Startup of reactors for anoxic ammonium oxidation: experiences from the first full-scale anammox reactor in Rotterdam. Water Res. 2007, 41, 4149–4163. [Google Scholar] [CrossRef]
- Thuan, T.-H.; Jahng, D.-J.; Jung, J.-Y.; Kim, D.-J.; Kim, W.-K.; Park, Y.-J.; Kim, J.-E.; Ahn, D.-H. Anammox bacteria enrichment in upflow anaerobic sludge blanket (UASB) reactor. Biotechnol. Bioprocess Eng. 2004, 9, 345. [Google Scholar] [CrossRef]
- Dapena-Mora, A.; Fernandez, I.; Campos, J.; Mosquera-Corral, A.; Mendez, R.; Jetten, M. Evaluation of activity and inhibition effects on Anammox process by batch tests based on the nitrogen gas production. Enzyme Microb. Technol. 2007, 40, 859–865. [Google Scholar] [CrossRef]
- Fernández, I.; Dosta, J.; Fajardo, C.; Campos, J.; Mosquera-Corral, A.; Méndez, R. Short-and long-term effects of ammonium and nitrite on the Anammox process. J. Environ. Manag. 2012, 95, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Strous, M.; Kuenen, J.G.; Jetten, M.S. Key physiology of anaerobic ammonium oxidation. Appl. Environ. Microbiol. 1999, 65, 3248–3250. [Google Scholar] [PubMed]
- Scaglione, D.; Lotti, T.; Ficara, E.; Malpei, F. Inhibition on anammox bacteria upon exposure to digestates from biogas plants treating the organic fraction of municipal solid waste and the role of conductivity. Waste Manag. 2017, 61, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Strous, M.; Heijnen, J.; Kuenen, J.G.; Jetten, M. The sequencing batch reactor as a powerful tool for the study of slowly growing anaerobic ammonium-oxidizing microorganisms. Appl. Microbiol. Biotechnol. 1998, 50, 589–596. [Google Scholar] [CrossRef]
- Ahn, Y.H. Sustainable nitrogen elimination biotechnologies: A review. Process Biochem. 2006, 41, 1709–1721. [Google Scholar] [CrossRef]
- Oh, S.; Yoo, Y.; Young, J.; Kim, I. Effect of organics on sulfur-utilizing autotrophic denitrification under mixotrophic conditions. J. Biotechnol. 2001, 92, 1–8. [Google Scholar] [CrossRef]
- Kim, S.; Jung, H.; Kim, K.-S.; Kim, I.S. Treatment of high nitrate-containing wastewaters by sequential heterotrophic and autotrophic denitrification. J. Environ. Eng. 2004, 130, 1475–1480. [Google Scholar] [CrossRef]
- Liu, H.; Jiang, W.; Wan, D.; Qu, J. Study of a combined heterotrophic and sulfur autotrophic denitrification technology for removal of nitrate in water. J. Hazard. Mater. 2009, 169, 23–28. [Google Scholar] [CrossRef]
- Sahinkaya, E.; Dursun, N. Sulfur-oxidizing autotrophic and mixotrophic denitrification processes for drinking water treatment: Elimination of excess sulfate production and alkalinity requirement. Chemosphere 2012, 89, 144–149. [Google Scholar] [CrossRef]
- Du, R.; Peng, Y.; Cao, S.; Wang, S.; Wu, C. Advanced nitrogen removal from wastewater by combining anammox with partial denitrification. Bioresour. Technol. 2015, 179, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Du, R.; Niu, M.; Li, B.; Ren, N.; Peng, Y. Integrated anaerobic ammonium oxidization with partial denitrification process for advanced nitrogen removal from high-strength wastewater. Bioresour. Technol. 2016, 221, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Du, R.; Cao, S.; Li, B.; Niu, M.; Wang, S.; Peng, Y. Performance and microbial community analysis of a novel DEAMOX based on partial-denitrification and anammox treating ammonia and nitrate wastewaters. Water Res. 2017, 108, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Hu, H.-Y.; Shi, Z.-J.; Yang, C.-C.; Li, P.; Huang, M.; Ni, W.-M.; Shi, M.-L.; Jin, R.-C. Towards simultaneously removing nitrogen and sulfur by a novel process: Anammox and autotrophic desulfurization–denitrification (AADD). Chem. Eng. J. 2016, 297, 207–216. [Google Scholar] [CrossRef]
- Dasgupta, S.; Wu, S.; Goel, R. Coupling autotrophic denitrification with partial nitritation-anammox (PNA) for efficient total inorganic nitrogen removal. Bioresour. Technol. 2017, 243, 700–707. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Du, L.; Hou, Y.; Cheng, S.; Zhang, X.; Liu, B. Endogenous influences on anammox and sulfocompound-oxidizing autotrophic denitrification coupling system (A/SAD) and dynamic operating strategy. Bioresour. Technol. 2018, 264, 253–260. [Google Scholar] [CrossRef]
- Fernandez, I.; Mosquera-Corral, A.; Campos, J.; Mendez, R. Operation of an Anammox SBR in the presence of two broad-spectrum antibiotics. Process Biochem. 2009, 44, 494–498. [Google Scholar] [CrossRef]
- Egli, K.; Fanger, U.; Alvarez, P.J.; Siegrist, H.; van der Meer, J.R.; Zehnder, A.J. Enrichment and characterization of an anammox bacterium from a rotating biological contactor treating ammonium-rich leachate. Arch. Microbiol. 2001, 175, 198–207. [Google Scholar] [CrossRef]
- Van Hulle, S.W.; Vandeweyer, H.J.; Meesschaert, B.D.; Vanrolleghem, P.A.; Dejans, P.; Dumoulin, A. Engineering aspects and practical application of autotrophic nitrogen removal from nitrogen rich streams. Chem. Eng. J. 2010, 162, 1–20. [Google Scholar] [CrossRef]
- Cema, G.; Wiszniowski, J.; Żabczyński, S.; Zabłocka-Godlewska, E.; Raszka, A.; Surmacz-Górska, J. Biological nitrogen removal from landfill leachate by deammonification assisted by heterotrophic denitrification in a rotating biological contactor (RBC). Water Sci. Technol. 2007, 55, 35–42. [Google Scholar] [CrossRef]
- Dosta, J.; Fernandez, I.; Vazquez-Padin, J.; Mosquera-Corral, A.; Campos, J.; Mata-Alvarez, J.; Mendez, R. Short-and long-term effects of temperature on the Anammox process. J. Hazard. Mater. 2008, 154, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.-C.; Zheng, P.; Mahmood, Q.; Hu, B.-L. Osmotic stress on nitrification in an airlift bioreactor. J. Hazard. Mater. 2007, 146, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Carvajal-Arroyo, J.M.; Sun, W.; Sierra-Alvarez, R.; Field, J.A. Inhibition of anaerobic ammonium oxidizing (anammox) enrichment cultures by substrates, metabolites and common wastewater constituents. Chemosphere 2013, 91, 22–27. [Google Scholar] [CrossRef] [PubMed]




| Run | Elapsed Time (Day) | Reuse Water Flow Rate (m3/d) | Reject Water Flow Rate (m3/d) | Dilution Rate | Influent TN Concentration (mg/L) | Nitrogen Load (kg/d) |
|---|---|---|---|---|---|---|
| 1 | 0–90 | 95 ± 33 | 32 ± 14 | 4.0 | 411 ± 58 | 51 ± 27 |
| 2 | 91–105 | 121 ± 9 | 40 ± 3 | 4.0 | 411 ± 65 | 71 ± 7 |
| 3 | 106–151 | 136 ± 16 | 44 ± 8 | 4.1 | 436 ± 63 | 79 ± 12 |
| 4 | 152–236 | 127 ± 17 | 39 ± 7 | 4.3 | 482 ± 64 | 83 ± 16 |
| 5 | 237–326 | 141 ± 24 | 47 ± 8 | 4.0 | 549 ± 22 | 105 ± 16 |
| 6 | 327–390 | 154 ± 23 | 52 ± 10 | 4.0 | 542 ± 59 | 111 ± 17 |
| 7 | 391–411 | 112 ± 19 | 58 ± 9 | 2.9 | 661 ± 44 | 111 ± 21 |
| 8 | 412–440 | 160 ± 16 | 55 ± 5 | 3.9 | 463 ± 37 | 100 ± 6 |
| Elapsed Time (Day) | Sample Name | AOB | NOB | ANAMMOX | AOB+ ANAMMOX (%) |
|---|---|---|---|---|---|
| Nitrosomonas (%) | Nitrospira (%) | Brocadia (%) | |||
| 0 | Seeding sludge | 6.5 | - | - | - |
| 6 | IN | 2.6 | - | - | - |
| 86 | Over (Out) | 1.8 | - | - | 1.8 |
| Reactor | 1.7 | - | 5.4 | 7.1 | |
| Under (Recycle) | 1.2 | - | 12.4 | 13.6 | |
| 109 | Over (Out) | 6.5 | 1.3 | - | 6.5 |
| Reactor | 7.3 | - | 6.5 | 9.8 | |
| Under (Recycle) | 12.4 | - | 11.4 | 23.8 | |
| 215 | Over (Out) | 4.3 | 2.4 | - | 4.3 |
| Reactor | 2.9 | 2.2 | 7.5 | 10.4 | |
| Under (Recycle) | 3 | - | 3 | 6 | |
| 254 | Over (Out) | 1.8 | - | - | 1.8 |
| Reactor | 1.7 | - | 9.3 | 11.0 | |
| Under (Recycle) | 1.2 | - | 6.5 | 7.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, K.; Kim, H.; Kim, W.; Lee, J. Efficient Nitrogen Removal of Reject Water Generated from Anaerobic Digester Treating Sewage Sludge and Livestock Manure by Combining Anammox and Autotrophic Sulfur Denitrification Processes. Water 2019, 11, 204. https://doi.org/10.3390/w11020204
Kwon K, Kim H, Kim W, Lee J. Efficient Nitrogen Removal of Reject Water Generated from Anaerobic Digester Treating Sewage Sludge and Livestock Manure by Combining Anammox and Autotrophic Sulfur Denitrification Processes. Water. 2019; 11(2):204. https://doi.org/10.3390/w11020204
Chicago/Turabian StyleKwon, Kiwook, Hyosun Kim, Woojin Kim, and Junbae Lee. 2019. "Efficient Nitrogen Removal of Reject Water Generated from Anaerobic Digester Treating Sewage Sludge and Livestock Manure by Combining Anammox and Autotrophic Sulfur Denitrification Processes" Water 11, no. 2: 204. https://doi.org/10.3390/w11020204
APA StyleKwon, K., Kim, H., Kim, W., & Lee, J. (2019). Efficient Nitrogen Removal of Reject Water Generated from Anaerobic Digester Treating Sewage Sludge and Livestock Manure by Combining Anammox and Autotrophic Sulfur Denitrification Processes. Water, 11(2), 204. https://doi.org/10.3390/w11020204




