Assessment of Recommendation for the Containment and Disinfection of Human Excreta in Cholera Treatment Centers
Abstract
1. Introduction
2. Materials and Methods
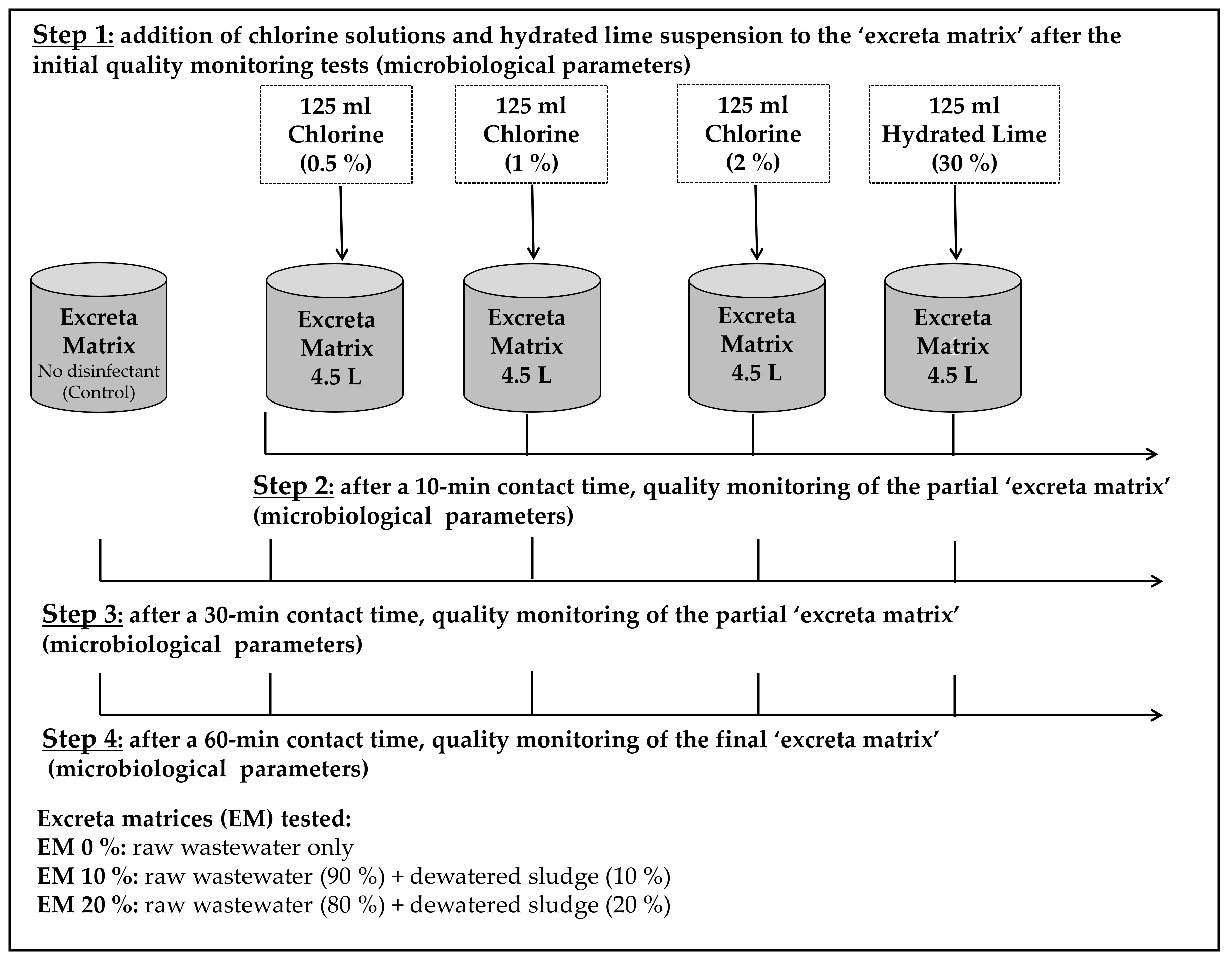
2.1. Production of Excreta Matrices and Disinfectant Solutions
2.2. Experimental Setup
- A smaller volume of disinfectant (125 mL) was used to treat the excreta matrices (4.5 L), in this case, 2.8% (v:v) instead of 10% (450 mL), the reason for this was to follow more closely the specific recommendations for cholera treatment centres [12].
- Contact time (Ct) was tested at 10, 30, and 60 min (not just 10 and 30 min).
- In this study, hydrated lime was only tested at the 30% concentration (not at 10% or 20%), this was because previous research [34] showed that it presented the highest efficacy to contain and treat excreta matrices.
- Only NADCC chlorine compound was used to produce chlorine solutions, the reason was that in the previous study [34], there was no statistical difference in treatment efficacy when compared to the other compounds (HTH and bleach (NaOCl)).
- Only somatic coliphages (not F-specific coliphages and/or phages of Bacteroides fragilis GB-124) were used as viral indicators when testing treatment efficacy, this was because somatic coliphages were the most resistant [34].
2.3. Statistical Analyses
3. Results
3.1. Overall Efficacy with Respect to Each Approach
3.2. Efficacy of Each Approach with Respect to Contact Time (Ct)
3.3. Efficacy of Each Disinfectant with Respect to Excreta Matrix (EM)
3.4. Fitted Line Plots with Regression Equations, Representing Log Reduction of Microorganisms, according to Chlorine Concentrations
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- US CDC. Cholera-General Information; Centres for Disease Control and Prevention: Druid Hills, GA, USA, 2018. Available online: https://www.cdc.gov/cholera/general/index.html (accessed on 6 October 2018).
- Lipp, E.K.; Huq, A.; Colwell, R.R. Effects of Global Climate on Infectious Disease: The Cholera Model. Clin. Microbiol. Rev. 2002, 15, 757–770. [Google Scholar] [CrossRef]
- UNICEF. 2013 Cholera Tool Kit. Available online: https://www.unicef.org/cholera/Cholera-Toolkit-2013.pdf (accessed on 5 October 2018).
- Alexakis, L.C. Cholera-Rice water stools. Pan Afr. Med. J. 2017, 26, 147–147. [Google Scholar] [CrossRef]
- Levine, M.M.; Kaper, J.B.; Herrington, D.; Ketley, J.; Losonsky, G.; Tacket, C.O.; Tall, B.; Cryz, S. Safety, immunogenicity, and efficacy of recombinant live oral cholera vaccines, CVD 103 and CVD 103-HgR. Lancet 1988, 2, 467–470. [Google Scholar] [CrossRef]
- Feachem, R.G. Environmental aspects of cholera epidemiology. III. Transmission and control. Trop. Dis. Bull. 1982, 79, 1–47. [Google Scholar] [PubMed]
- Mosley, W.H.; Ahmad, S.; Benenson, A.S.; Ahmed, A. The relationship of vibriocidal antibody titre to susceptibility to cholera in family contacts of cholera patients. Bull. World Health Organ. 1968, 38, 777–785. [Google Scholar]
- Schmid-Hempel, P.; Frank, S.A. Pathogenesis, virulence, and infective dose. PLoS Pathog. 2007, 3, 1372–1373. [Google Scholar] [CrossRef] [PubMed]
- Sterk, E. Filovirus Haemorrhagic Fever Guideline; Médecins Sans Frontières (MSF): 2008. Available online: https://www.oregonpa.org/resources/2014CME/Speaker%20Presentations/Ebola%20Guideline%20-%20Crawford.pdf (accessed on 6 March 2018).
- World Health Organization. Interim Infection Prevention and Control Guidance for Care of Patients with Suspected or Confirmed Filovirus Hemorrhagic Fever in Health-Care Settings, with Focus on Ebola; WHO: Geneva, Switzerland, 2014; Available online: http://www.euro.who.int/__data/assets/pdf_file/0005/268772/Interim-Infection-Prevention-and-Control-Guidance-for-Care-of-Patients-with-Suspected-or-Confirmed-Filovirus-Haemorrhagic-Fever-in-Health-Care-Settings,-with-Focus-on-Ebola-Eng.pdf (accessed on 7 March 2018).
- CDC. Interim Recommendations for Cleaning Houses Safely in West Africa Ebola-Affected Areas after Persons with Symptoms of Ebola are transferred to Ebola Treatment Units or Community Care Centers; Centers for Disease Control and Prevention: Druid Hills, GA, USA, 2015; Available online: http://www.cdc.gov/vhf/ebola/hcp/cleaning-houses-safely-in-west-africa.html (accessed on 8 March 2018).
- MSF. Cholera Guidelines; Médecins Sans Frontières: 2004. Available online: https://www.unicef.org/cholera/Chapter_2_basics/04_MSF_cholera_guidelines.pdf (accessed on 7 March 2018).
- Gray, N.F. Chapter 12—Pathogens and Their Removal. In Water Technology, 3rd ed.; Butterworth-Heinemann: Oxford, UK, 2010; pp. 360–400. [Google Scholar]
- Rutala, W.A.; Weber, D.J. Disinfection and sterilization in health care facilities: What clinicians need to know? Clin. Infect. Dis. 2004, 39, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Branz, A.; Matthew, L.M.; Lehmann, L.; Bastable, A.; Ali, S.I.; Kadir, K.; Yates, T.; Bloom, D.; Lantagne, D. Chlorination of drinking water in emergencies: A review of knowledge to develop recommendations for implementation and research needed. Waterlines 2017, 36. [Google Scholar] [CrossRef]
- Donnermair, M.M.; Blatchley, E.R. Disinfection efficacy of organic chloramines. Water Res. 2003, 37, 1557–1570. [Google Scholar] [CrossRef]
- Lechevallier, M.W.; Evans, T.M.; Seidler, R.J. Effect of turbidity on chlorination efficiency and bacterial persistence in drinking water. Appl. Environ. Microbiol. 1981, 42, 159–167. [Google Scholar]
- Trajano Gomes da Silva, D.; Dias, E.; Ebdon, J.; Taylor, H. Limitations of chlorine disinfection of human excreta: Implications for Ebola disease control. In Proceedings of the 39th WEDC International Conference, Kumasi, Ghana, 11–15 July 2016; Available online: http://wedc.lboro.ac.uk/resources/conference/39/Trajano-2446.pdf (accessed on 15 March 2018).
- World Health Organization. Report: Ebola Virus Disease (EVD) Key Questions and Answers Concerning Water, Sanitation and Hygiene; WHO: Geneva, Switzerland, 2014; Available online: http://apps.who.int/iris/bitstream/10665/137181/1/WHO_EVD_WSH_14_eng.pdf?ua=1&ua=1 (accessed on 8 March 2018).
- Grabow, W.O.K.; Grabow, N.A.; Burger, J.S. The bactericidal effect of lime flocculation/flotation as a primary unit process in a multiple system for the advanced purification of sewage works effluent. Water Res. 1969, 3, 943–953. [Google Scholar] [CrossRef]
- Grabow, W.O.K.; Middendorff, I.G.; Basson, N.C. Role of lime treatment in the removal of bacteria, enteric viruses, and coliphages in a wastewater reclamation plant. Appl. Environ. Microbiol. 1978, 35, 663–669. [Google Scholar] [PubMed]
- Semerjian, L.; Ayoub, G.M. High-pH magnesium coagulation-flocculation in wastewater treatment. Adv. Environ. Res. 2003, 7, 389–403. [Google Scholar] [CrossRef]
- Gambrill, M.P.; Mara, D.D.; Oragui, J.I.; Silva, S.A. Wastewater treatment for effluent reuse—Lime-induced removal of excreted pathogens. Water Sci. Technol. 1989, 21, 79–84. [Google Scholar] [CrossRef]
- Taylor, H.D.; Gambrill, M.P.; Mara, D.D. Lime Treatment of Municipal Wastewater. Research Monographs in Tropical Public Health Engineering; No. 3; The University of Leeds: Leeds, UK, 1994. [Google Scholar]
- Taylor, H.D.; Gambrill, M.P.; Mara, D.D.; Silva, S.A. Upgrading a low-cost physicochemical wastewater treatment plant to solve operational problems. Water Sci. Technol. 1994, 29, 247–254. [Google Scholar] [CrossRef]
- Hasegawa, T.; Kurosey, Y.; Tanaka, A. Advanced treatment technique for swine wastewater using two agents: Thermally polymerized amorphous silica and hydrated lime for color and phosphorus removal and sulfur for nitrogen removal. Anim. Sci. J. 2017, 88, 1636–1643. [Google Scholar] [CrossRef]
- Morrison, S.M.; Martin, K.L.; Humble, D.E. Lime Disinfection of Sewage Bacteria at Low Temperature; Report, No EPA-660/2-73-017; U.S. Environmental Protection Agency: Washington, DC, USA, 1973. [Google Scholar]
- Yamashita, T.; Aketo, T.; Minowa, N.; Sugimoto, K.; Yokoyama, H.; Ogino, A.; Yasuo Tanaka, Y. Simultaneous removal of colour, phosphorus and disinfection from treated wastewater using an agent synthesized from amorphous silica and hydrated lime. Environ. Technol. 2013, 34, 1017–1025. [Google Scholar] [CrossRef]
- Polprasert, C.; Valencia, L.G. The inactivation of faecal coliforms and ascaris ova in faeces by lime. Water Res. 1981, 15, 31–36. [Google Scholar] [CrossRef]
- Mignotte, B. The Effect Liming on the Microbiological Quality of Urban Sludge. Water Sci. Technol. 2001, 43, 195–200. [Google Scholar] [CrossRef]
- Bina, B.; Movahedian, H.; Kord, I. The Effect of Lime Stabilization on the Microbiological Quality of Sewage Sludge. J. Environ. Health Sci. Eng. 2004, 1, 34–38. [Google Scholar]
- Sozzi, E.; Fabre, K.; Fesselet, J.-F.; Ebdon, J.E.; Taylor, H. Minimizing the risk of disease transmission in emergency settings: Novel in situ physico-chemical disinfection of pathogen-laden hospital wastewaters. PLoS Neglect. Trop. Dis. 2015. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.; Malambo, D.H.; Gonzalez Perez, M.R.; Nobela, H.N.; Pooter, L.; Spit, J.; Hooijmans, C.M.; de Vossenberg Jv Greya, W.; Thole, B.; et al. Lactic Acid Fermentation, Urea and Lime Addition: Promising Faecal Sludge Sanitizing Methods for Emergency Sanitation. Int. J. Environ. Res. Public Health 2015, 12, 13871–13885. [Google Scholar] [CrossRef] [PubMed]
- Trajano Gomes da Silva, D.; Dias, E.; Ebdon, J.; Taylor, H. Assessment of recommended approaches for containment and safe handling of human excreta in emergency settings. PLoS ONE 2018, 13, e0201344. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. ISO 9308-1:2000. Water Quality. Detection and Enumeration of Escherichia coli and Coliform bacteria Part 1: Membrane Filtration Method; International Organization for Standardization: Geneva, Switzerland, 2000. [Google Scholar]
- Anonymous. ISO 7899-2:2000. Water Quality: Detection and Enumeration of Intestinal Enterococci Part 2: Membrane Filtration Method; International Organization for Standardization: Geneva, Switzerland, 2000. [Google Scholar]
- Anonymous. ISO 10705-2. Water Quality—Detection and Enumeration of Bacteriophages—Part 2: Enumeration of Somatic Coliphages; International Organization for Standardization: Geneva, Switzerland, 2001. [Google Scholar]
- Grabow, W.O.K.; Coubrough, P. Practical direct plaque-assay for coliphages in 100-ml samples of drinking-water. Appl. Environ. Microbiol. 1986, 52, 430–433. [Google Scholar] [PubMed]
- Helsel, D.R. More Than Obvious: Better Methods for Interpreting Non-Detect Data. Environ. Sci. Technol. 2005, 39, 419A–423A. [Google Scholar] [CrossRef]
- Shuval, H.I.; Cohen, J.; Kolodney, R. Regrowth of coliforms and fecal coliforms in chlorinated wastewater effluent. Water Res. 1973, 7, 537–546. [Google Scholar] [CrossRef]
- Iqbal, Q.; Lubeck-Schricker, M.; Wells, E.; Wolfe, M.K.; Lantagne, D. Shelf-Life of Chlorine Solutions Recommended in Ebola Virus Disease Response. PLoS ONE 2016, 11, e0156136. [Google Scholar] [CrossRef]
- Wells, E.; Wolfe, M.K.; Murray, A.; Lantagne, D. Accuracy, Precision, Ease-of-Use, and Cost of Methods to Test Ebola-Relevant Chlorine Solutions. PLoS ONE 2016, 11, e0152442. [Google Scholar] [CrossRef]
- Pickup, J. Human Health Aspects of Halogenated Organic By-Products from Use of Active Chlorine. Euro Chlor Science Dossier 15. 2017. Available online: http://www.eurochlor.org/media/109463/science_dossier_hhdbp_2016_final.pdf (accessed on 6 March 2018).
- Villanueva, C.M.; Gracia-Lavedan, E.; Bosetti, C.; Righi, E.; Molina, A.J.; Martín, V.; Boldo, E.; Aragones, N.; Perez-Gomez, B.; Pollan, M.; et al. Colorectal Cancer and Long-Term Exposure to Trihalomethanes in Drinking Water: A Multicenter Case-Control Study in Spain and Italy. Environ. Health Perspect. 2017, 125, 56–65. [Google Scholar] [CrossRef]
- U.S. Geological Survey. Lime. Mineral Commodity Summaries, January 2018. Available online: https://minerals.usgs.gov/minerals/pubs/commodity/lime/mcs-2018-lime.pdf (accessed on 14 January 2019).
- Lutz, C.; Erken, M.; Noorian, P.; Sun, S.; McDougald, D. Environmental reservoirs and mechanisms of persistence of Vibrio cholerae. Front. Microbiol. 2013, 4, 375. [Google Scholar] [CrossRef]
- Muñoz Ruiz, C.; Collazo Ponce, A.; Alvarado, F.J. Bactericidal Effect of Hydrated Lime in Aqueous Solution. Boletin de la Oficina Sanitaria Panamericana 1995, 118, 302–306. [Google Scholar] [PubMed]
- Kovacikova, G.; Skorupski, K.A. Vibrio cholerae LysR homolog, AphB, cooperates with AphA at the tcpPH promoter to activate expression of the ToxR virulence cascade. J. Bacteriol. 1999, 181, 4250–4256. [Google Scholar] [PubMed]
- Mccarthy, S.A.; Miller, A. Effect of Three Biocides on Latin American and Gulf Coast Strains of Toxigenic Vibrio cholerae O1. J. Food Protect. 1994, 57, 865–869. [Google Scholar] [CrossRef]
- Monney, I.; Awuah, E. Sanitizing Fecal Sludge for Reuse Using Wood Ash as an Additive. Recycling 2016, 1, 14. [Google Scholar] [CrossRef]
- Bonetta, S.; Pignata, C.; Lorenzi, E.; De Ceglia, M.; Meucci, L.; Bonetta, S.; Gilli, G.; Carraro, E. Peracetic Acid (PAA) Disinfection: Inactivation of Microbial Indicators and Pathogenic Bacteria in a Municipal Wastewater Plant. Water 2017, 9, 427. [Google Scholar] [CrossRef]
- Amabilis-Sosa, L.E.; Vázquez-López, M.; García Rojas, J.L.; Roé-Sosa, A.; Moeller-Chávez, G.E. Efficient Bacteria Inactivation by Ultrasound in Municipal Wastewater. Environment 2018, 5, 47. [Google Scholar] [CrossRef]
- Zimmermann, K. Microwave Technologies: An Emerging Tool for Inactivation of Biohazardous Material in Developing Countries. Recycling 2018, 3, 34. [Google Scholar] [CrossRef]
- Gensch, R.; Jennings, A.; Renggli, S.; Reymond, P. Compendium of Sanitation Technologies in Emergencies, 1st ed.; German WASH Network; Swiss Federal Institute of Aquatic Science and Technology; Global WASH Cluster; Sustainable Sanitation Alliance: Duebendorf, Switzerland, 2018. [Google Scholar]

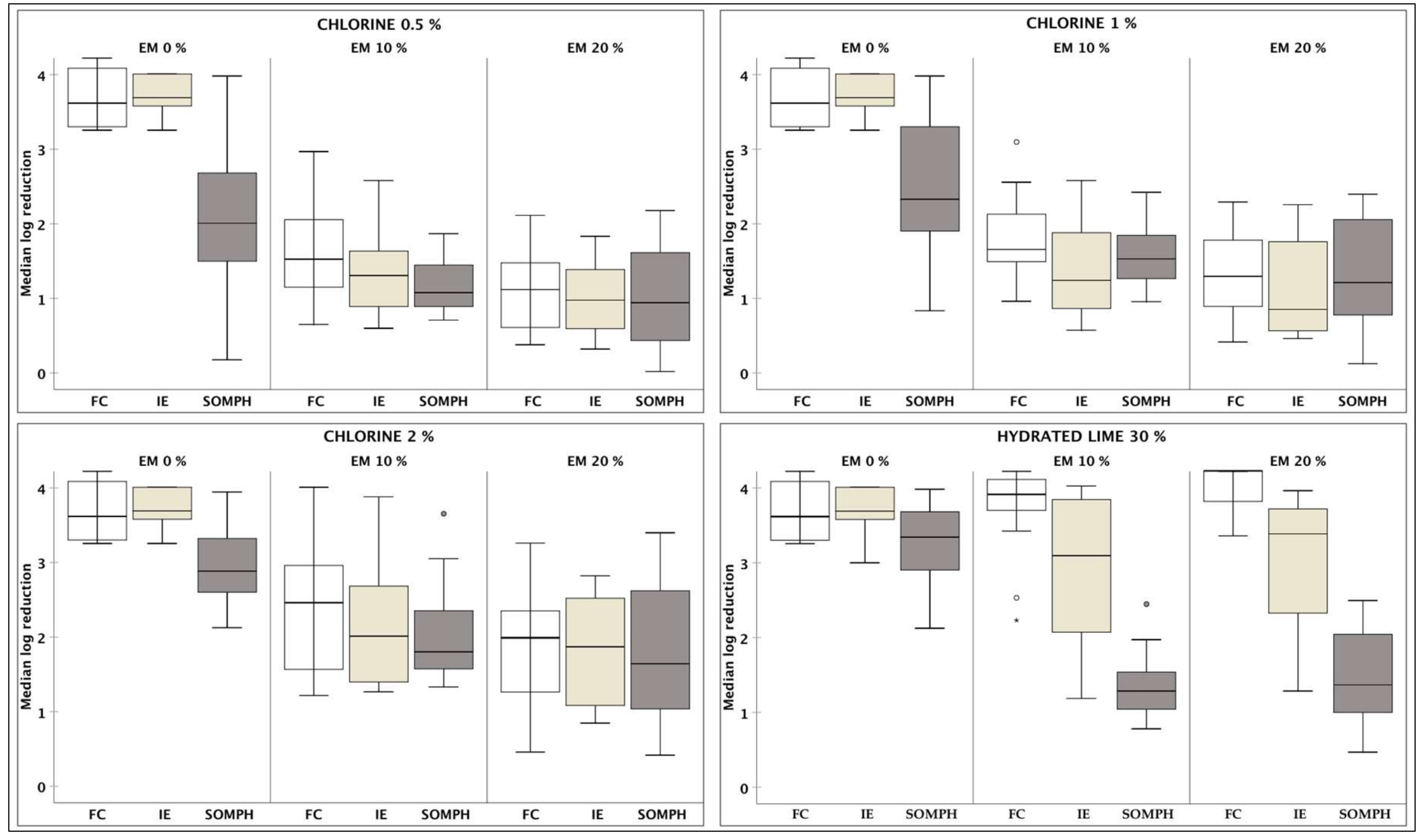
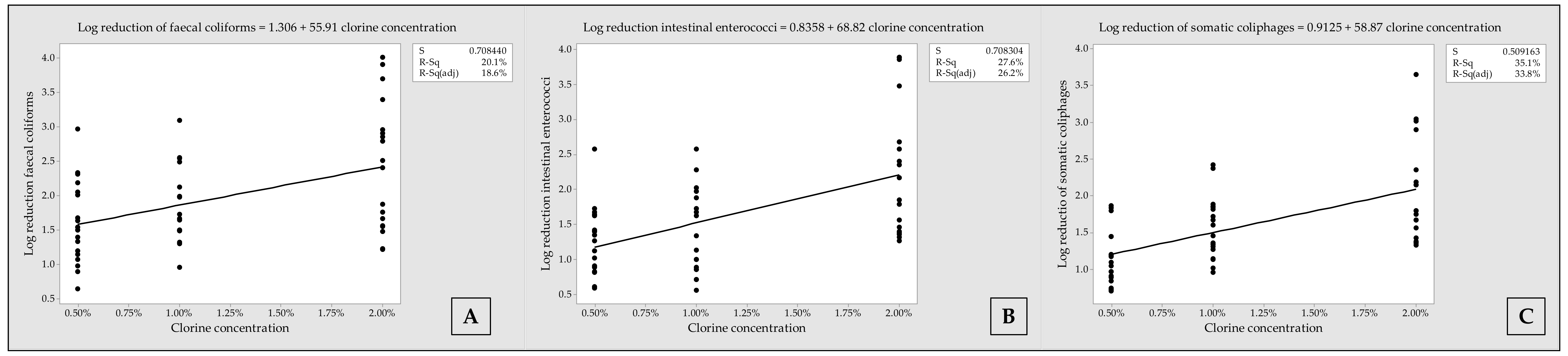
| Approach | Contact Time | Excreta Matrix | FC | IE | SOMPH |
|---|---|---|---|---|---|
| Chlorine 0.5% | 10 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.08) | 1.52 (0.18–2.68) |
| 10% | 1.57 (1.15–2.97) | 1.38 (0.62–2.58) | 1.09 (0.73–1.87) | ||
| 20% | 1.37 (0.47–2.11) | .98 (0.59–1.76) | 1.01 (0.28–2.18) | ||
| 30 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.08) | 1.92 (0.58–3.38) | |
| 10% | 1.48 (0.98–2.34) | 1.31 (0.83–1.65) | 1.09 (0.71–1.84) | ||
| 20% | 1.12 (0.38–1.97) | 0.99 (0.32–1.83) | 0.83 (0.06–1.98) | ||
| 60 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.01) | 2.26 (1.87–3.98) | |
| 10% | 1.51 (0.65–2.32) | 1.21 (0.60–1.73) | 1.08 (0.75–1.80) | ||
| 20% | 0.86 (0.45–4.28) | 0.93 (0.39–1.67) | 0.98 (0.02–3.98) | ||
| Chlorine 1% | 10 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.01) | 2.31 (0.83–3.38) |
| 10% | 1.58 (1.49–2.56) | 1.34 (0.57–2.28) | 1.70 (1.02–2.37) | ||
| 20% | 1.41 (0.78–2.29) | 0.85 (0.54–1.76) | 1.21 (0.61–2.19) | ||
| 30 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.01) | 2.11 (1.33–3.98) | |
| 10% | 1.74 (1.31–3.10) | 1.17 (.86–2.58) | 1.53 (1.14–2.42) | ||
| 20% | 1.33 (0.55–1.78) | 1.17 (0.46–2.11) | 1.27 (0.34–2.26) | ||
| 60 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.01) | 2.47 (1.76–3.68) | |
| 10% | 1.69 (0.96–2.49) | 1.44 (0.72–2.03) | 1.35 (0.96–1.89) | ||
| 20% | 1.17 (0.42–2.05) | 0.96 (0.49–2.26) | 1.40 (0.12–2.40) | ||
| Chlorine 2% | 10 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.01) | 2.83 (2.20 – 3.94) |
| 10% | 1.99 (1.22–2.91) | 1.59 (1.32–2.58) | 1.80 (1.57–2.90) | ||
| 20% | 1.77 (1.00- 3.26) | 1.87 (1.08–2.61) | 1.57 (1.04–2.80) | ||
| 30 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.01) | 3.31 (2.12–3.94) | |
| 10% | 2.95 (1.56–4.01) | 3.08 (1.38–3.88) | 1.95 (1.36–3.05) | ||
| 20% | 2.17 (0.46–4.34) | 1.91 (0.97–4.01) | 1.74 (0.53–3.10) | ||
| 60 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.01) | 2.72 (2.40–3.34) | |
| 10% | 2.37 (1.48–3.70) | 1.87 (1.27–3.88) | 1.91 (1.33–3.65) | ||
| 20% | 1.97 (0.69–4.34) | 1.73 (0.85–2.82) | 1.90 (0.42–3.40) | ||
| Hydrated lime 30% | 10 min | 0% | 3.62 (3.26–4.28) | 3.68 (3.00–4.01) | 3.11 (2.12–3.94) |
| 10% | 3.96 (3.70–4.32) | 2.86 (2.07–3.88) | 1.40 (0.78–1.60) | ||
| 20% | 4.27 (3.36–4.82) | 3.39 (1.29–3.72) | 1.45 (0.74–2.49) | ||
| 30 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.01) | 3.34 (2.90–3.94) | |
| 10% | 3.81 (2.23–4.32) | 2.92 (1.82–4.03) | 1.22 (0.85–2.45) | ||
| 20% | 4.30 (3.82–4.82) | 3.37 (1.78–3.72) | 1.18 (0.64–3.94) | ||
| 60 min | 0% | 3.62 (3.26–4.28) | 3.69 (3.26–4.01) | 3.48 (2.20–3.98) | |
| 10% | 3.81 (2.53–4.32) | 3.21 (1.19–3.88) | 1.16 (0.81–1.97) | ||
| 20% | 4.08 (3.68–4.82) | 3.04 (1.40–3.96) | 1.51 (0.47–2.10) |
| Indicator Organism | Chlorine 0.5% | Hydrated Lime 30% | ||||
|---|---|---|---|---|---|---|
| Previous Study * | This Study ** | Difference | Previous Study * | This Study ** | Difference | |
| FC | 2.78 | 1.66 | −1.12 | 4.41 | 3.93 | −0.48 |
| IE | 1.92 | 1.41 | −0.51 | 3.94 | 3.50 | −0.44 |
| SOMPH | 3.24 | 1.28 | −1.96 | 1.80 | 1.67 | −0.13 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trajano Gomes da Silva, D.; Ives, K.; Fesselet, J.-F.; Ebdon, J.; Taylor, H. Assessment of Recommendation for the Containment and Disinfection of Human Excreta in Cholera Treatment Centers. Water 2019, 11, 188. https://doi.org/10.3390/w11020188
Trajano Gomes da Silva D, Ives K, Fesselet J-F, Ebdon J, Taylor H. Assessment of Recommendation for the Containment and Disinfection of Human Excreta in Cholera Treatment Centers. Water. 2019; 11(2):188. https://doi.org/10.3390/w11020188
Chicago/Turabian StyleTrajano Gomes da Silva, Diogo, Kevin Ives, Jean-Francois Fesselet, James Ebdon, and Huw Taylor. 2019. "Assessment of Recommendation for the Containment and Disinfection of Human Excreta in Cholera Treatment Centers" Water 11, no. 2: 188. https://doi.org/10.3390/w11020188
APA StyleTrajano Gomes da Silva, D., Ives, K., Fesselet, J.-F., Ebdon, J., & Taylor, H. (2019). Assessment of Recommendation for the Containment and Disinfection of Human Excreta in Cholera Treatment Centers. Water, 11(2), 188. https://doi.org/10.3390/w11020188





