Physiological Profiling and Functional Diversity of Groundwater Microbial Communities in a Municipal Solid Waste Landfill Area
Abstract
1. Introduction
2. Materials and Methods
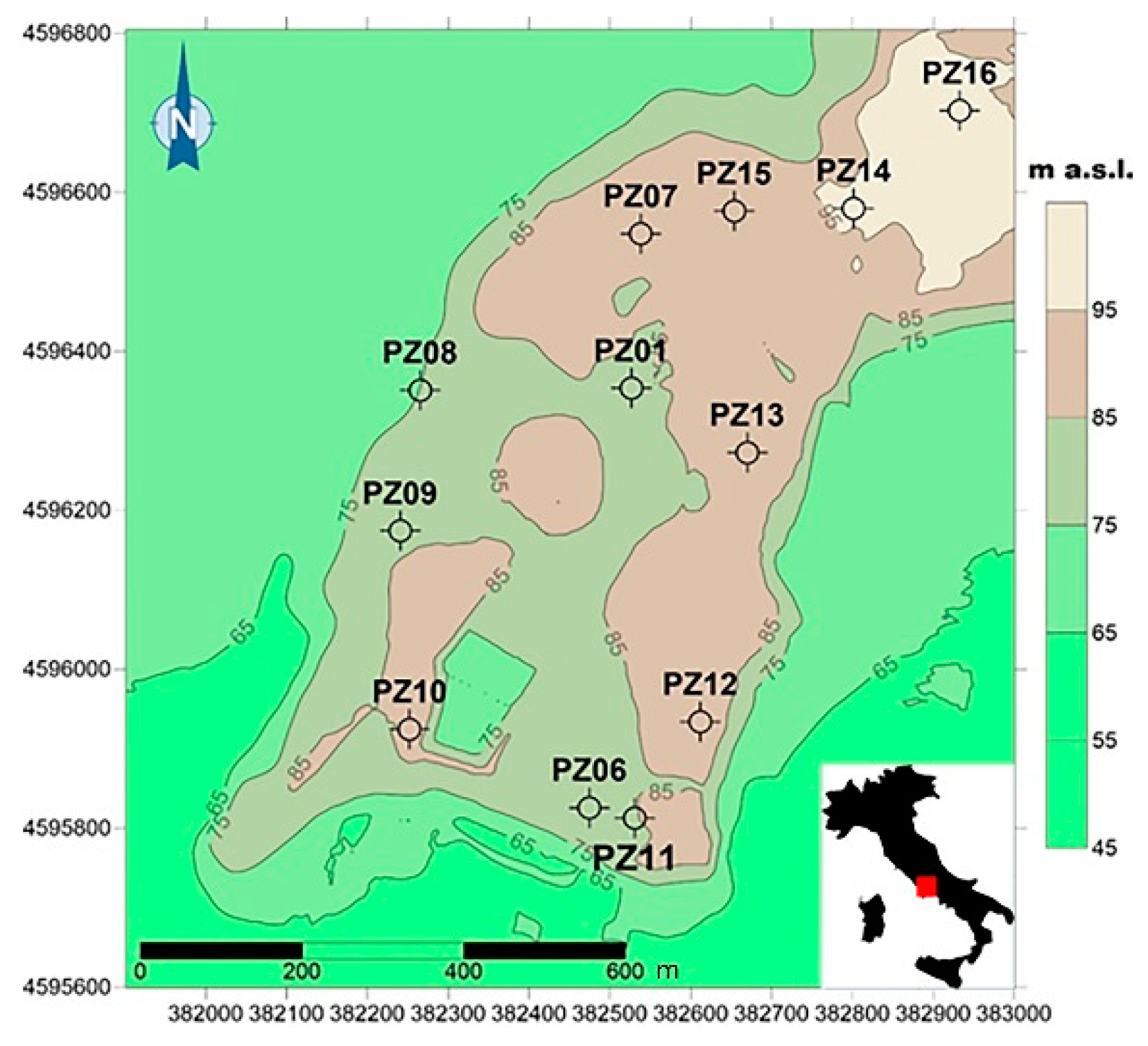
2.1. Field Sampling and Major Water Characteristics
2.2. Microbial Community Characterization by Flow Cytometry
2.3. Metabolic Potential, Functional Diversity and Community Level Physiological Profile
- K = the asymptote/carrying capacity
- μ = exponential rate of AWCD change
- t = time (h)
- s = the time when y = K/2
- H = −∑pi (ln pi)
- pi = the ratio of the activity of a particular substrate to the sums of activities of all substrates
- S = number of positive wells in the microplates
2.4. Statistical Analysis
3. Results
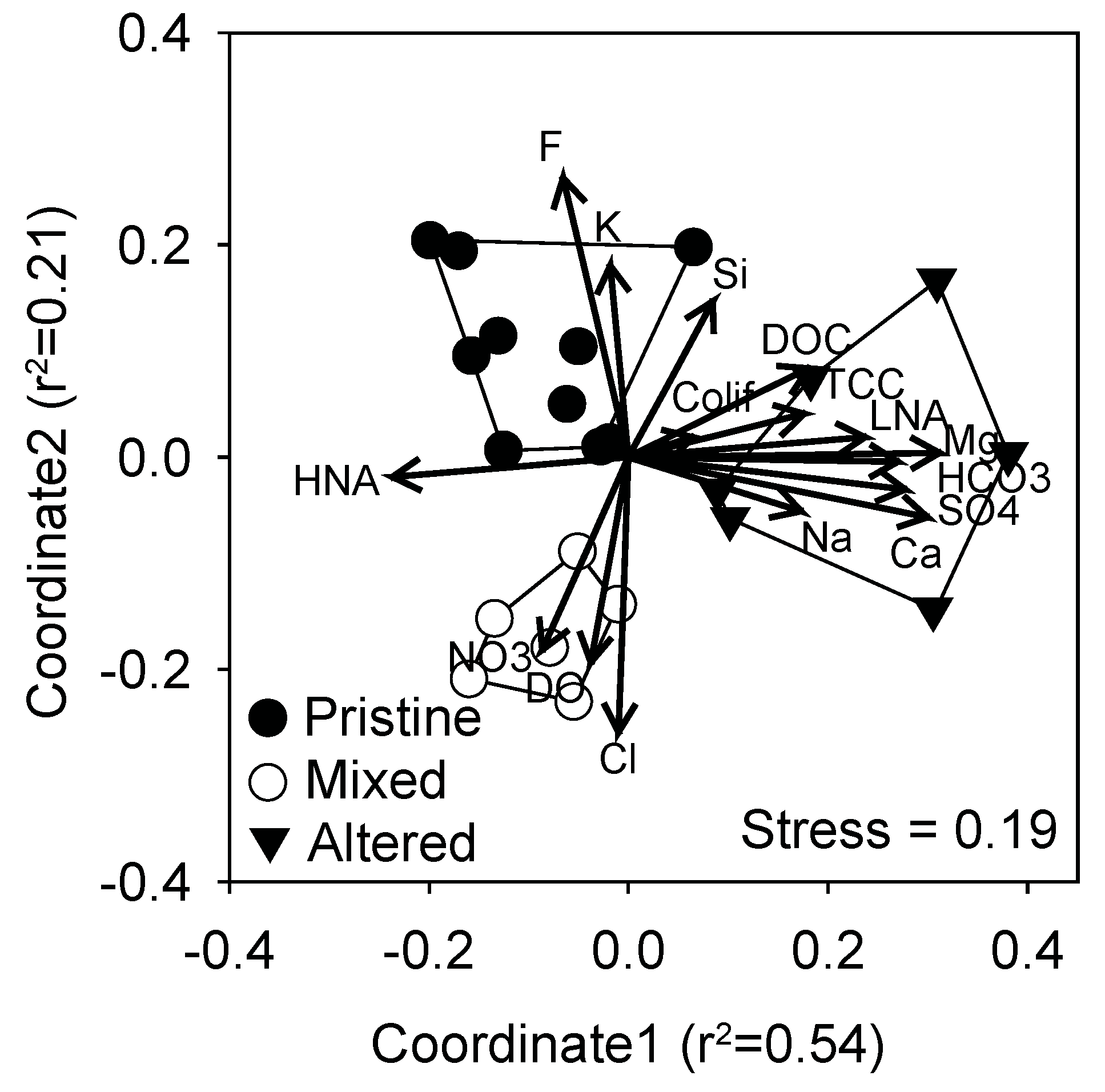
3.1. Groundwater Physical, Chemical and Microbial Characteristics
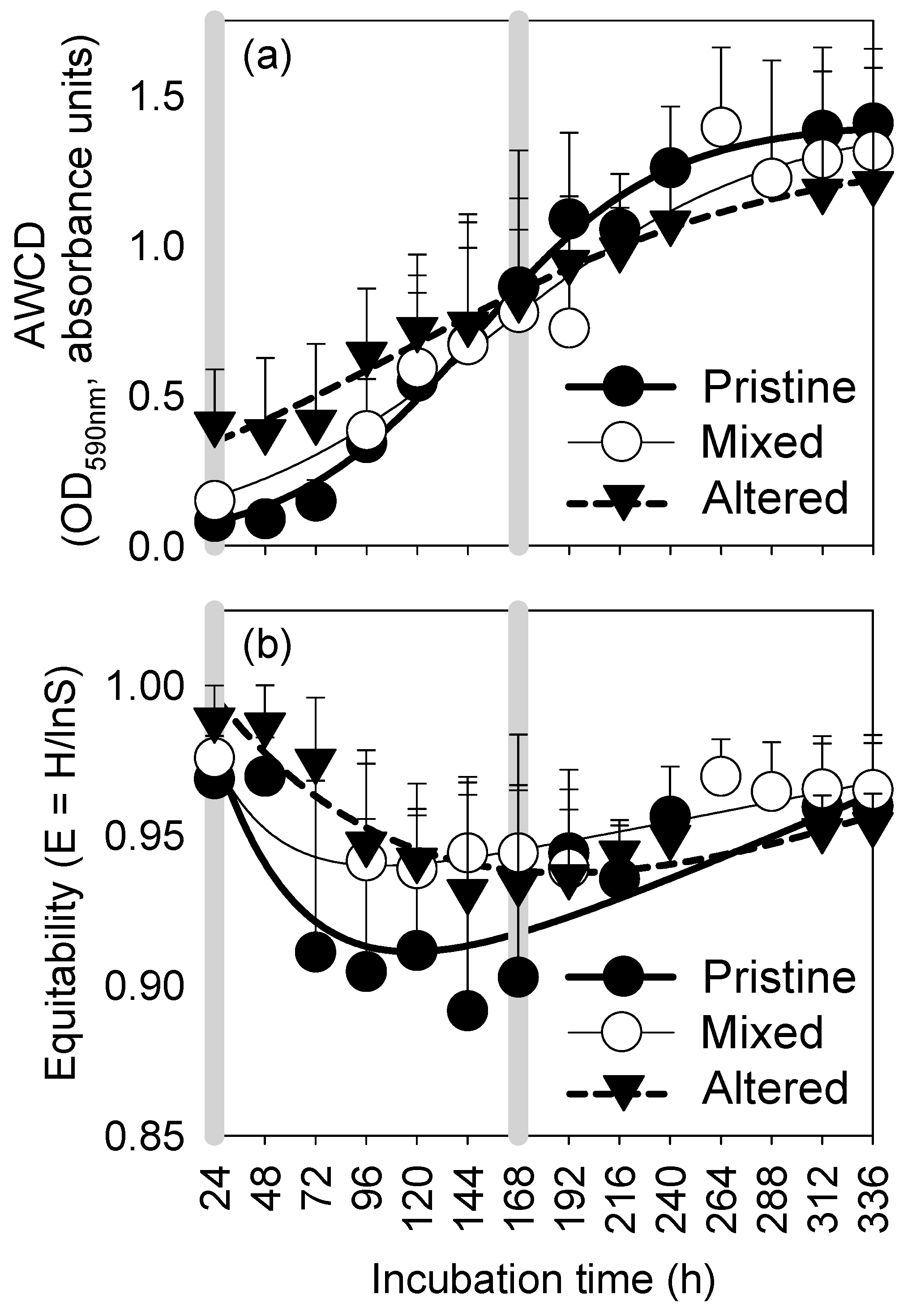
3.2. Kinetic Profiles of Metabolic Potential and Functional Diversity
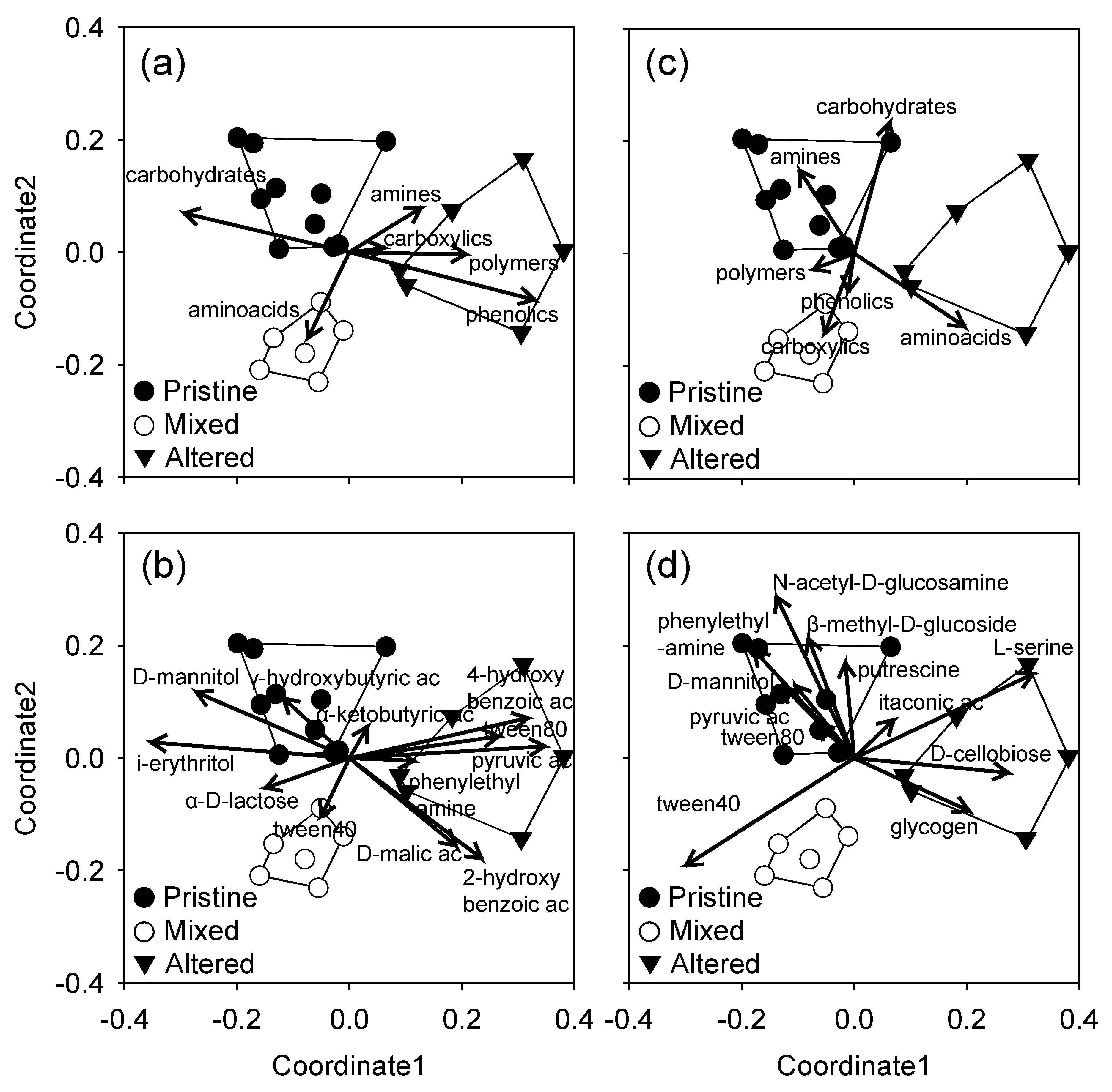
3.3. Community Level Physiological Profiles
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Downing, J.A.; Prairie, Y.T.; Cole, J.J.; Duarte, C.M.; Tranvik, L.J.; Striegl, R.G.; McDowell, W.H.; Kortelainen, P.; Caraco, N.F.; Melack, J.M.; et al. The global abundance and size distribution of lakes, ponds, and impoundments. Limnol. Oceanogr. 2006, 51, 2388–2397. [Google Scholar] [CrossRef]
- Pham, H.V.; Torresan, S.; Critto, A.; Marcomini, A. Alteration of freshwater ecosystem services under global change–A review focusing on the Po River basin Italy and the Red River basin Vietnam. Sci. Total Environ. 2019, 652, 1347–1365. [Google Scholar] [CrossRef] [PubMed]
- Mpelasoka, F.; Hennessy, K.; Jones, R.; Bates, B. Comparison of suitable drought indices for climate change impacts assessment over Australia towards resource management. Int. J. Climatol. 2008, 28, 1283–1292. [Google Scholar] [CrossRef]
- Kløve, B.; Ala-Aho, P.; Bertrand, G.; Gurdak, J.J.; Kupfersberger, H.; Kværner, J.; Muotka, T.; Mykrä, H.; Preda, E.; Rossi, P.; et al. Climate change impacts on groundwater and dependent ecosystems. J. Hydrol. 2014, 518, 250–266. [Google Scholar] [CrossRef]
- Gozlan, R.E.; Karimov, B.K.; Zadereev, E.; Kuznetsova, D.; Brucet, S. Status, trends, and future dynamics of freshwater ecosystems in Europe and Central Asia. Inland Waters 2019, 9, 78–94. [Google Scholar] [CrossRef]
- EU Directive on the protection of groundwater against pollution and deterioration. European Parliament and Council Directive 2006 2006/118/EC of 12 December 2006. Off. J. Eur. Union 2006, 372, 19–31.
- Griebler, C.; Stein, H.; Kellermann, C.; Berkhoff, S.; Brielmann, H.; Schmidt, S.; Selesi, D.; Steube, C.; Fuchs, A.; Hahn, H.J. Ecological assessment of groundwater ecosystems-Vision or illusion? Ecol. Eng. 2010, 36, 1174–1190. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Galassi, D.M.P. Agricultural impact on Mediterranean alluvial aquifers: Do groundwater communities respond? Fundam. Appl. Limnol. Arch. Hydrobiol. 2013, 182, 271–282. [Google Scholar] [CrossRef]
- Griebler, C.; Avramov, M. Groundwater ecosystem services: A review. Freshw. Sci. 2015, 34, 355–367. [Google Scholar] [CrossRef]
- Griebler, C.; Lueders, T. Microbial biodiversity in groundwater ecosystems. Freshw. Biol. 2009, 54, 649–677. [Google Scholar] [CrossRef]
- Moser, D.P.; Fredrickson, J.K.; Geist, D.R.; Arntzen, E.V.; Peacock, A.D.; Li, S.-M.W.; Spadoni, T.; McKinley, J.P. Biogeochemical Processes and Microbial Characteristics across Groundwater−Surface Water Boundaries of the Hanford Reach of the Columbia River. Environ. Sci. Technol. 2003, 37, 5127–5134. [Google Scholar] [CrossRef]
- Yagi, J.M.; Neuhauser, E.F.; Ripp, J.A.; Mauro, D.M.; Madsen, E.L. Subsurface ecosystem resilience: Long-term attenuation of subsurface contaminants supports a dynamic microbial community. ISME J. 2010, 4, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Röling, W.F.M.; van Breukelen, B.M.; Braster, M.; Lin, B.; van Verseveld, H.W. Relationships between Microbial Community Structure and Hydrochemistry in a Landfill Leachate-Polluted Aquifer. Appl. Environ. Microbiol. 2001, 67, 4619–4629. [Google Scholar] [CrossRef] [PubMed]
- D’Angeli, I.M.; Serrazanetti, D.I.; Montanari, C.; Vannini, L.; Gardini, F.; De Waele, J. Geochemistry and microbial diversity of cave waters in the gypsum karst aquifers of Emilia Romagna region, Italy. Sci. Total Environ. 2017, 598, 538–552. [Google Scholar] [CrossRef] [PubMed]
- Amalfitano, S.; Del Bon, A.; Zoppini, A.; Ghergo, S.; Fazi, S.; Parrone, D.; Casella, P.; Stano, F.; Preziosi, E. Groundwater geochemistry and microbial community structure in the aquifer transition from volcanic to alluvial areas. Water Res. 2014, 65, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Röling, W.F.M.; Van Breukelen, B.M.; Braster, M.; Goeltom, M.T.; Groen, J.; Van Verseveld, H.W. Analysis of microbial communities in a landfill leachate polluted aquifer using a new method for anaerobic physiological profiling and 16S rDNA based fingerprinting. Microb. Ecol. 2000, 40, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Castañeda, S.S.; Sucgang, R.J.; Almoneda, R.V.; Mendoza, N.D.S.; David, C.P.C. Environmental isotopes and major ions for tracing leachate contamination from a municipal landfill in Metro Manila, Philippines. J. Environ. Radioact. 2012, 110, 30–37. [Google Scholar] [CrossRef] [PubMed]
- EU Directive on Landfill of Waste. European Union Council Directive 1999/31/EC issued 26 April 1999. Off. J. Eur. Communities 1999, 182, 228–246.
- Smith, R.J.; Jeffries, T.C.; Roudnew, B.; Fitch, A.J.; Seymour, J.R.; Delpin, M.W.; Newton, K.; Brown, M.H.; Mitchell, J.G. Metagenomic comparison of microbial communities inhabiting confined and unconfined aquifer ecosystems. Environ. Microbiol. 2012, 14, 240–253. [Google Scholar] [CrossRef]
- Janniche, G.S.; Spliid, H.; Albrechtsen, H.-J. Microbial Community-Level Physiological Profiles (CLPP) and herbicide mineralization potential in groundwater affected by agricultural land use. J. Contam. Hydrol. 2012, 140–141, 45–55. [Google Scholar] [CrossRef]
- Preziosi, E.; Frollini, E.; Zoppini, A.; Ghergo, S.; Melita, M.; Parrone, D.; Rossi, D.; Amalfitano, S. Disentangling natural and anthropogenic impacts on groundwater by hydrogeochemical, isotopic and microbiological data: Hints from a municipal solid waste landfill. Waste Manag. 2019, 84, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Barba, C.; Folch, A.; Gaju, N.; Sanchez-Vila, X.; Carrasquilla, M.; Grau-Martínez, A.; Martínez-Alonso, M. Microbial community changes induced by Managed Aquifer Recharge activities: Linking hydrogeological and biological processes. Hydrol. Earth Syst. Sci. 2019, 23, 139–154. [Google Scholar] [CrossRef]
- Salo, M.J. Anthropogenically Driven Changes to Shallow Groundwater in Southeastern Wisconsin and Its Effects on the Aquifer Microbial Communities. ProQuest Diss. Theses 2019, 1–178. [Google Scholar]
- Song, Y.; Mao, G.; Gao, G.; Bartlam, M.; Wang, Y. Structural and Functional Changes of Groundwater Bacterial Community During Temperature and pH Disturbances. Microb. Ecol. 2019, 78, 428–445. [Google Scholar] [CrossRef] [PubMed]
- Frollini, E.; Rossi, D.; Rainaldi, M.; Parrone, D.; Ghergo, S.; Preziosi, E. A proposal for groundwater sampling guidelines: Application to a case study in southern Latium. Rend. Online Soc. Geol. Ital. 2019, 47, 46–51. [Google Scholar] [CrossRef]
- Eaton, A.D.; American Public Health Association; Water Environment Federation; American Water Works Associations. Standard Methods for the Examination of Water and Wastewater, 21st ed.; APHA-AWWA-WEF: Washington, DC, USA, 2005. [Google Scholar]
- Amalfitano, S.; Fazi, S.; Ejarque, E.; Freixa, A.; Romaní, A.M.; Butturini, A. Deconvolution model to resolve cytometric microbial community patterns in flowing waters. Cytom. Part A 2018, 93, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Insam, H.; Goberna, M. Molecular Microbial Ecology Manual; Kowalchuk, G.A., de Bruijn, F.J., Head, I.M., Akkermans, A.D., van Elsas, J.D., Eds.; Springer: Dordrecht, The Netherlands, 2004; ISBN 978-1-4020-2176-3. [Google Scholar]
- Garland, J.L.; Mills, A.L. Classification and Characterization of Heterotrophic Microbial Communities on the Basis of Patterns of Community-Level Sole-Carbon-Source Utilization. Appl. Environ. Microbiol. 1991, 57, 2351–2359. [Google Scholar]
- Salomo, S.; Münch, C.; Röske, I. Evaluation of the metabolic diversity of microbial communities in four different filter layers of a constructed wetland with vertical flow by BiologTM analysis. Water Res. 2009, 43, 4569–4578. [Google Scholar] [CrossRef] [PubMed]
- Weber, K.P.; Gehder, M.; Legge, R.L. Assessment of changes in the microbial community of constructed wetland mesocosms in response to acid mine drainage exposure. Water Res. 2008, 42, 180–188. [Google Scholar] [CrossRef]
- Garland, J.L. Analytical approaches to the characterization of samples of microbial communities using patterns of potential C source utilization. Soil Biol. Biochem. 1996, 28, 213–221. [Google Scholar] [CrossRef]
- Konopka, A.; Oliver, L.; Turco, R.F., Jr. The Use of Carbon Substrate Utilization Patterns in Environmental and Ecological Microbiology. Microb. Ecol. 1998, 35, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Insam, H. A New Set of Substrates Proposed for Community Characterization in Environmental Samples. In Microbial Communities; Insam, H., Rangger, A., Eds.; Springer: Berlin/Heidelberg, Germany, 1997; pp. 259–260. [Google Scholar] [CrossRef]
- Guckert, J.B.; Carr, G.J.; Johnson, T.D.; Hamm, B.G.; Davidson, D.H.; Kumagai, Y. Community analysis by Biolog: Curve integration for statistical analysis of activated sludge microbial habitats. J. Microbiol. Methods 1996, 27, 183–197. [Google Scholar] [CrossRef]
- Clarke, K.R.; Green, R.H. Statistical design and analysis for a biological effects study. Mar. Ecol. Prog. Ser. 1988, 46, 213–226. [Google Scholar] [CrossRef]
- Tiquia, S.M. Metabolic diversity of the heterotrophic microorganisms and potential link to pollution of the Rouge River. Environ. Pollut. 2010, 158, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Taş, N.; Brandt, B.W.; Braster, M.; van Breukelen, B.M.; Röling, W.F.M. Subsurface landfill leachate contamination affects microbial metabolic potential and gene expression in the Banisveld aquifer. FEMS Microbiol. Ecol. 2018, 94, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Preston-Mafham, J.; Boddy, L.; Randerson, P.F. Analysis of microbial community functional diversity using sole-carbon-source utilisation profiles-A critique. FEMS Microbiol. Ecol. 2002, 42, 1–14. [Google Scholar]
- Christian, B.W.; Lind, O.T. Multiple carbon substrate utilization by bacteria at the sediment–water interface: Seasonal patterns in a stratified eutrophic reservoir. Hydrobiologia 2007, 586, 43–56. [Google Scholar] [CrossRef]
- Kothawala, D.N.; Stedmon, C.A.; Müller, R.A.; Weyhenmeyer, G.A.; Köhler, S.J.; Tranvik, L.J. Controls of dissolved organic matter quality: Evidence from a large-scale boreal lake survey. Glob. Chang. Biol. 2014, 20, 1101–1114. [Google Scholar] [CrossRef]
- Logue, J.B.; Stedmon, C.A.; Kellerman, A.M.; Nielsen, N.J.; Andersson, A.F.; Laudon, H.; Lindström, E.S.; Kritzberg, E.S. Experimental insights into the importance of aquatic bacterial community composition to the degradation of dissolved organic matter. ISME J. 2016, 10, 533–545. [Google Scholar] [CrossRef]
- Griebler, C.; Malard, F.; Lefébure, T. Current developments in groundwater ecology—from biodiversity to ecosystem function and services. Curr. Opin. Biotechnol. 2014, 27, 159–167. [Google Scholar] [CrossRef]
- Shen, Y.; Chapelle, F.H.; Strom, E.W.; Benner, R. Origins and bioavailability of dissolved organic matter in groundwater. Biogeochemistry 2015, 122, 61–78. [Google Scholar] [CrossRef]
- Meckenstock, R.U.; Elsner, M.; Griebler, C.; Lueders, T.; Stumpp, C.; Aamand, J.; Agathos, S.N.; Albrechtsen, H.-J.; Bastiaens, L.; Bjerg, P.L.; et al. Biodegradation: Updating the Concepts of Control for Microbial Cleanup in Contaminated Aquifers. Environ. Sci. Technol. 2015, 49, 7073–7081. [Google Scholar] [CrossRef] [PubMed]
- Pretty, J.L.; Hildrew, A.G.; Trimmer, M. Nutrient dynamics in relation to surface–subsurface hydrological exchange in a groundwater fed chalk stream. J. Hydrol. 2006, 330, 84–100. [Google Scholar] [CrossRef]
- Hofmann, R.; Griebler, C. DOM and bacterial growth efficiency in oligotrophic groundwater: Absence of priming and co-limitation by organic carbon and phosphorus. Aquat. Microb. Ecol. 2018, 81, 55–71. [Google Scholar] [CrossRef]
- Gounot, A.-M. Microbial oxidation and reduction of manganese: Consequences in groundwater and applications. FEMS Microbiol. Rev. 1994, 14, 339–349. [Google Scholar] [CrossRef]
- Hemme, C.L.; Tu, Q.; Shi, Z.; Qin, Y.; Gao, W.; Deng, Y.; Nostrand, J.D.; van Wu, L.; He, Z.; Chain, P.S.G.; et al. Comparative metagenomics reveals impact of contaminants on groundwater microbiomes. Front. Microbiol. 2015, 6, 1205. [Google Scholar] [CrossRef]
- Zoppini, A.; Puddu, A.; Fazi, S.; Rosati, M.; Sist, P. Extracellular enzyme activity and dynamics of bacterial community in mucilaginous aggregates of the northern Adriatic Sea. Sci. Total Environ. 2005, 353, 270–286. [Google Scholar] [CrossRef]
- Arnosti, C.; Bell, C.; Moorhead, D.L.; Sinsabaugh, R.L.; Steen, A.D.; Stromberger, M.; Wallenstein, M.; Weintraub, M.N. Extracellular enzymes in terrestrial, freshwater, and marine environments: Perspectives on system variability and common research needs. Biogeochemistry 2014, 117, 5–21. [Google Scholar] [CrossRef]
- Keith, S.C.; Arnosti, C. Extracellular enzyme activity in a river-bay-shelf transect: Variations in polysaccharide hydrolysis rates with substrate and size class. Aquat. Microb. Ecol. 2001, 24, 243–253. [Google Scholar] [CrossRef]
- Rosenstock, B.; Simon, M. Consumption of Dissolved Amino Acids and Carbohydrates by Limnetic Bacterioplankton According to Molecular Weight Fractions and Proportions Bound to Humic Matter. Microb. Ecol. 2003, 45, 433–443. [Google Scholar] [CrossRef]
- Arnosti, C. Patterns of Microbially Driven Carbon Cycling in the Ocean: Links between Extracellular Enzymes and Microbial Communities. Adv. Oceanogr. 2014, 2014, 1–12. [Google Scholar] [CrossRef]
- Oest, A.; Alsaffar, A.; Fenner, M.; Azzopardi, D.; Tiquia-Arashiro, S.M. Patterns of change in metabolic capabilities of sediment microbial communities in river and lake ecosystems. Int. J. Microbiol. 2018, 2018, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Hollibaugh, J.T.; Azam, F. Microbial degradation of dissolved proteins in seawater1. Limnol. Oceanogr. 1983, 28, 1104–1116. [Google Scholar] [CrossRef]
- Cho, J.-C.; Kim, S.-J. Increase in Bacterial Community Diversity in Subsurface Aquifers Receiving Livestock Wastewater Input. Appl. Environ. Microbiol. 2000, 66, 956–965. [Google Scholar] [CrossRef][Green Version]
- Feris, K.P.; Hristova, K.; Gebreyesus, B.; Mackay, D.; Scow, K.M. A Shallow BTEX and MTBE Contaminated Aquifer Supports a Diverse Microbial Community. Microb. Ecol. 2004, 48, 589–600. [Google Scholar] [CrossRef]
- Anantharaman, K.; Brown, C.T.; Hug, L.A.; Sharon, I.; Castelle, C.J.; Probst, A.J.; Thomas, B.C.; Singh, A.; Wilkins, M.J.; Karaoz, U.; et al. Thousands of microbial genomes shed light on interconnected biogeochemical processes in an aquifer system. Nat. Commun. 2016, 7, 13219. [Google Scholar] [CrossRef]
- Gözdereliler, E. Groundwater Bacteria: Diversity, Activity and Physiology of Pesticide Degradation at Low Concentrations. PhD thesis, Technical University of Denmark, Lyngby, Denmark, 2012. [Google Scholar]
- Proctor, C.R.; Besmer, M.D.; Langenegger, T.; Beck, K.; Walser, J.-C.; Ackermann, M.; Bürgmann, H.; Hammes, F. Phylogenetic clustering of small low nucleic acid-content bacteria across diverse freshwater ecosystems. ISME J. 2018, 12, 1344–1359. [Google Scholar] [CrossRef]
- Obernosterer, I.; Benner, R. Competition between biological and photochemical processes in the mineralization of dissolved organic carbon. Limnol. Oceanogr. 2004, 49, 117–124. [Google Scholar] [CrossRef]
- Sala, M.M.; Estrada, M. Seasonal changes in the functional diversity of bacterioplankton in contrasting coastal environments of the NW Mediterranean. Aquat. Microb. Ecol. 2006, 44, 1–9. [Google Scholar] [CrossRef]
- Ding, H.; Sun, M.-Y. Biochemical degradation of algal fatty acids in oxic and anoxic sediment–seawater interface systems: Effects of structural association and relative roles of aerobic and anaerobic bacteria. Mar. Chem. 2005, 93, 1–19. [Google Scholar] [CrossRef]
- Bertilsson, S.; Tranvik, L.J. Photochemical transformation of dissolved organic matter in lakes. Limnol. Oceanogr. 2000, 45, 753–762. [Google Scholar] [CrossRef]
- Preiss, J.; Romeo, T. Molecular Biology and Regulatory Aspects of Glycogen Biosynthesis in Bacteria; Cohn, W.E., Moldave, K.B.T.-P., Eds.; Academic Press: New York, NY, USA, 1994; Volume 47, pp. 299–329. [Google Scholar]
- Henrissat, B.; Deleury, E.; Coutinho, P.M. Glycogen metabolism loss: A common marker of parasitic behaviour in bacteria? Trends Genet. 2002, 18, 437–440. [Google Scholar] [CrossRef]
| Parameter | Unit | Pristine | Mixed | Altered |
|---|---|---|---|---|
| EC | μS/cm | 766.4 a | 915.6 | 1452.7 |
| ORP | mV | −119.3 a | 74.3 b | −40.8 c |
| T | °C | 17.8 | 19.1 | 17.3 |
| pH | 7.2 a | 7.0 b | 6.7 c | |
| DO | mg/L | 0.2 a | 3.1 b | 0.4 a |
| NO3 | mg/L | 0.9 a | 18.2 b | 0.8 a |
| SO4 | mg/L | 11.7 a | 45.9 b | 232.5 c |
| K | mg/L | 10.5 a | 2.1 b | 5.1 ab |
| DOC | mg/L | 2.2 a | 0.8 b | 2.0 a |
| F | mg/L | 0.4 a | 0.2 b | 0.2 ab |
| Cl | mg/L | 7.9 a | 19.5 b | 11.3 a |
| Si | mg/L | 16.4 a | 7.5 b | 19.7 a |
| Mg | mg/L | 23.1 b | 22.0 b | 45.7 a |
| Ca | mg/L | 94.1 b | 135.4 c | 238.3 a |
| Na | mg/L | 9.9 | 8.2 | 8.8 |
| HCO3 | mg/L | 419.8 b | 424.5 b | 632.4 a |
| Parameter | Unit | Pristine | Mixed | Altered |
|---|---|---|---|---|
| Total Cells Count | 105 cells/mL | 4.4 | 3.1 | 6.6 |
| HNA Cells | % of TCC | 52.8 b | 52.6 b | 27.1 a |
| LNA Cells | % of TCC | 41.6 b | 46.4 b | 65.5 a |
| Total Coliforms | MPN/100 mL | 157.8 | 103.0 | 292.6 |
| E. coli | MPN/100 mL | <1 | <1 | 3.3 |
| Parameter | Unit | Pristine | Mixed | Altered |
|---|---|---|---|---|
| K | OD590nm | 1.42 | 1.41 | 1.33 |
| s | h | 157.1 | 147 | 117.1 |
| μ | h−1 | 0.029 | 0.023 | 0.021 |
| r2 | 0.98 | 0.99 | 0.99 |
| 24 h | |||||||||||||||||||||||
| Classes of Substrates | Substrates | Pristine | Mixed | Altered | |||||||||||||||||||
| Carbohydrates | β-methyl-D-glucoside | 4.7 | 6.4 | 4.8 | 4.9 | 4.6 | 5.1 | 5.3 | 3.8 | 5.6 | 4.4 | 4.8 | 5.6 | 3.3 | 3.4 | 3.6 | 4.8 | 3.3 | 3.1 | 4.9 | 3.5 | 3.5 | 3.5 |
| D-xylose | 4.1 | 1.5 | 3.2 | 2.9 | 4.0 | 1.6 | 0.0 | 5.7 | 1.5 | 3.0 | 3.6 | 3.6 | 1.9 | 3.7 | 4.5 | 3.4 | 3.9 | 3.1 | 3.6 | 3.7 | 3.3 | 3.6 | |
| i-erythritol | 7.0 | 4.2 | 5.2 | 5.7 | 6.3 | 6.6 | 8.1 | 5.6 | 7.0 | 4.6 | 5.8 | 5.3 | 6.7 | 4.9 | 6.0 | 5.9 | 4.1 | 3.7 | 4.5 | 4.0 | 3.7 | 6.5 | |
| D-mannitol | 5.5 | 7.2 | 5.1 | 5.5 | 5.0 | 5.2 | 7.1 | 4.4 | 8.6 | 4.9 | 5.6 | 6.8 | 5.7 | 3.6 | 5.3 | 5.3 | 3.9 | 2.9 | 4.1 | 3.4 | 3.6 | 5.2 | |
| N-acetyl-D-glucosamine | 4.9 | 6.9 | 5.5 | 5.4 | 5.0 | 4.4 | 7.3 | 4.0 | 6.1 | 6.2 | 5.3 | 4.6 | 3.8 | 4.5 | 4.3 | 4.1 | 3.8 | 2.9 | 3.3 | 3.6 | 3.8 | 5.0 | |
| D-cellobiose | 5.0 | 2.9 | 5.1 | 5.6 | 6.6 | 3.7 | 6.2 | 4.8 | 5.9 | 5.3 | 5.5 | 3.2 | 1.7 | 4.6 | 5.0 | 5.7 | 3.5 | 4.7 | 4.2 | 3.6 | 3.7 | 4.7 | |
| α-D-lactose | 6.0 | 3.3 | 5.0 | 6.1 | 5.2 | 5.5 | 1.7 | 4.7 | 6.7 | 5.5 | 5.2 | 7.7 | 1.8 | 4.1 | 5.2 | 6.2 | 3.2 | 5.0 | 4.0 | 2.9 | 4.1 | 4.4 | |
| glucose-1-phosphate | 2.7 | 1.9 | 1.5 | 2.7 | 1.9 | 0.0 | 7.3 | 2.3 | 0.0 | 2.0 | 2.5 | 1.1 | 2.9 | 2.5 | 1.4 | 1.8 | 2.5 | 2.5 | 2.0 | 2.2 | 3.0 | 1.6 | |
| D,L-α-glycerol phosphate | 2.0 | 2.8 | 0.0 | 0.6 | 3.1 | 0.0 | 0.0 | 0.9 | 2.8 | 1.1 | 1.5 | 2.0 | 2.7 | 1.8 | 1.9 | 1.7 | 2.5 | 1.6 | 2.1 | 2.5 | 2.9 | 0.5 | |
| Polymers | tween40 | 5.7 | 5.1 | 5.7 | 4.1 | 6.7 | 3.0 | 3.3 | 4.5 | 4.6 | 7.6 | 4.6 | 4.0 | 5.3 | 4.5 | 7.8 | 4.2 | 4.4 | 5.7 | 3.6 | 3.3 | 4.0 | 6.4 |
| tween80 | 0.0 | 2.4 | 3.4 | 3.6 | 0.0 | 2.4 | 0.1 | 2.6 | 0.0 | 1.6 | 0.0 | 0.0 | 1.7 | 3.1 | 2.9 | 3.4 | 3.7 | 3.3 | 3.0 | 3.3 | 3.8 | 3.3 | |
| α-cyclodextrin | 2.3 | 1.7 | 2.3 | 2.3 | 1.7 | 1.1 | 3.1 | 2.4 | 2.7 | 2.4 | 2.0 | 2.5 | 3.7 | 2.2 | 1.3 | 3.0 | 2.3 | 2.8 | 3.1 | 3.6 | 2.9 | 1.7 | |
| glycogen | 5.0 | 6.3 | 3.8 | 2.6 | 2.8 | 3.8 | 4.4 | 3.6 | 4.2 | 4.4 | 3.9 | 5.5 | 2.7 | 2.8 | 3.3 | 3.9 | 3.0 | 3.4 | 3.0 | 3.3 | 3.7 | 3.9 | |
| Carboxylic Acids | D-galactonic acid γ-lactone | 4.5 | 2.5 | 4.6 | 3.0 | 3.7 | 3.9 | 4.3 | 2.9 | 2.4 | 3.4 | 4.3 | 2.8 | 4.4 | 2.6 | 4.2 | 3.5 | 3.7 | 3.8 | 3.4 | 3.5 | 2.5 | 4.3 |
| D-galacturonic acid | 4.6 | 2.8 | 0.7 | 2.4 | 2.8 | 2.8 | 2.2 | 3.1 | 2.8 | 0.0 | 2.6 | 3.8 | 3.5 | 3.1 | 3.0 | 3.5 | 1.9 | 2.9 | 3.2 | 3.0 | 3.1 | 1.9 | |
| γ-hydroxybutyric acid | 4.1 | 1.8 | 4.3 | 5.3 | 3.9 | 11 | 1.0 | 2.7 | 9.3 | 7.6 | 5.0 | 2.6 | 2.7 | 3.8 | 2.7 | 3.8 | 4.1 | 3.9 | 2.6 | 3.7 | 3.9 | 5.1 | |
| D-glucosaminic acid | 4.3 | 2.7 | 3.1 | 4.2 | 3.8 | 3.2 | 7.0 | 3.2 | 5.8 | 4.4 | 4.1 | 4.1 | 4.5 | 3.3 | 3.7 | 3.6 | 2.5 | 2.4 | 3.1 | 3.1 | 3.2 | 3.0 | |
| itaconic acid | 2.6 | 2.0 | 2.9 | 2.6 | 2.7 | 2.1 | 2.7 | 2.4 | 3.2 | 2.2 | 2.6 | 3.4 | 1.9 | 2.2 | 2.0 | 2.2 | 3.5 | 3.1 | 2.6 | 2.8 | 2.4 | 2.4 | |
| α-ketobutyric acid | 0.0 | 2.7 | 3.7 | 3.0 | 0.0 | 5.9 | 0.8 | 3.2 | 0.0 | 2.6 | 4.6 | 0.0 | 2.3 | 2.9 | 1.5 | 1.2 | 2.0 | 1.5 | 2.1 | 2.8 | 3.1 | 2.7 | |
| D-malic acid | 0.0 | 2.1 | 2.8 | 1.6 | 3.0 | 0.0 | 0.5 | 2.3 | 1.7 | 1.5 | 0.0 | 3.4 | 3.7 | 3.5 | 3.3 | 1.8 | 2.6 | 3.8 | 3.0 | 3.4 | 3.6 | 1.8 | |
| pyruvic acid methyl ester | 0.0 | 1.7 | 2.9 | 0.0 | 0.0 | 0.0 | 0.0 | 0.7 | 0.0 | 0.8 | 0.0 | 0.0 | 2.8 | 1.0 | 0.5 | 0.0 | 2.6 | 2.2 | 2.0 | 3.4 | 3.6 | 0.4 | |
| Phenolic Compounds | 2-hydroxy benzoic acid | 0.0 | 1.9 | 2.1 | 0.0 | 2.2 | 0.0 | 0.0 | 2.2 | 0.0 | 0.0 | 0.0 | 2.2 | 3.3 | 3.5 | 3.5 | 5.0 | 3.2 | 2.5 | 4.4 | 3.3 | 3.4 | 1.7 |
| 4-hydroxy benzoic acid | 0.0 | 1.5 | 3.2 | 1.0 | 0.0 | 1.2 | 0.2 | 3.6 | 0.0 | 1.4 | 0.0 | 0.0 | 1.2 | 1.6 | 0.8 | 0.0 | 3.2 | 1.9 | 2.2 | 2.9 | 2.7 | 2.8 | |
| Aminoacids | L-arginine | 4.1 | 3.6 | 3.0 | 4.1 | 3.8 | 4.4 | 6.4 | 3.7 | 2.9 | 5.9 | 3.6 | 4.6 | 4.1 | 3.2 | 3.9 | 3.7 | 2.8 | 3.7 | 3.5 | 3.5 | 3.3 | 3.9 |
| L-asparagine | 3.8 | 3.8 | 2.8 | 3.0 | 3.4 | 3.6 | 3.1 | 2.7 | 1.2 | 2.8 | 3.5 | 3.7 | 4.6 | 4.1 | 3.1 | 2.5 | 3.8 | 4.2 | 3.7 | 3.1 | 3.0 | 3.5 | |
| L-phenylalanine | 3.0 | 2.3 | 1.6 | 2.3 | 2.0 | 2.4 | 1.4 | 2.8 | 1.9 | 1.8 | 2.4 | 2.0 | 2.9 | 2.5 | 1.5 | 1.4 | 3.3 | 2.1 | 2.0 | 2.9 | 1.3 | 2.0 | |
| L-serine | 3.4 | 3.8 | 2.3 | 3.1 | 3.5 | 3.9 | 5.0 | 2.7 | 2.6 | 2.5 | 3.6 | 4.2 | 3.5 | 4.2 | 3.1 | 3.3 | 3.4 | 3.6 | 3.2 | 3.3 | 3.3 | 2.9 | |
| L-threonine | 3.8 | 2.8 | 4.7 | 4.3 | 4.2 | 4.0 | 4.3 | 3.9 | 3.7 | 4.6 | 4.1 | 1.5 | 3.9 | 4.2 | 3.7 | 4.3 | 4.1 | 4.1 | 3.5 | 3.7 | 3.6 | 4.2 | |
| glycyl-L-glutamic acid | 3.6 | 2.6 | 1.8 | 2.8 | 2.2 | 0.0 | 1.5 | 2.3 | 2.6 | 2.4 | 3.0 | 3.3 | 3.1 | 3.4 | 2.1 | 2.9 | 2.3 | 3.2 | 3.0 | 2.7 | 2.3 | 2.0 | |
| Amines | phenylethyl-amine | 0.0 | 2.7 | 0.0 | 2.5 | 2.3 | 5.7 | 1.2 | 3.9 | 2.3 | 0.9 | 3.3 | 3.0 | 2.1 | 2.9 | 2.2 | 1.2 | 3.8 | 3.0 | 4.2 | 3.1 | 2.7 | 2.5 |
| putrescine | 3.2 | 4.0 | 2.7 | 2.8 | 3.4 | 3.8 | 3.6 | 2.6 | 2.1 | 2.3 | 2.8 | 3.5 | 1.5 | 2.2 | 2.7 | 2.7 | 3.1 | 3.4 | 2.9 | 3.0 | 3.1 | 2.6 | |
| 168 h | |||||||||||||||||||||||
| Classes of substrates | Substrates | Pristine | Mixed | Altered | |||||||||||||||||||
| Carbohydrates | β-methyl-D-glucoside | 4.8 | 4.4 | 8.8 | 7.6 | 1.3 | 2.6 | 5.8 | 2.1 | 11 | 5.8 | 3.4 | 5.7 | 3.8 | 2.8 | 1.5 | 5.6 | 4.8 | 5.5 | 4.5 | 5.3 | 2.6 | 8.2 |
| D-xylose | 3.7 | 3.8 | 0.3 | 0.5 | 3.4 | 0.3 | 0.0 | 1.9 | 0.1 | 0.2 | 1.2 | 0.6 | 0.9 | 1.3 | 0.9 | 1.5 | 1.2 | 1.2 | 2.7 | 1.8 | 2.3 | 0.7 | |
| i-erythritol | 1.7 | 1.5 | 1.2 | 1.7 | 1.4 | 1.1 | 2.9 | 2.5 | 0.9 | 2.4 | 2.7 | 2.4 | 1.7 | 2.9 | 2.1 | 4.2 | 2.7 | 1.6 | 3.4 | 2.5 | 2.8 | 2.9 | |
| D-mannitol | 5.0 | 5.1 | 8.2 | 9.2 | 2.1 | 3.1 | 7.6 | 3.7 | 5.3 | 10 | 6.7 | 3.4 | 6.0 | 5.6 | 6.1 | 5.3 | 5.6 | 5.3 | 3.7 | 4.7 | 5.9 | 5.7 | |
| N-acetyl-D-glucosamine | 4.0 | 4.9 | 6.1 | 8.1 | 3.0 | 6.8 | 7.4 | 7.4 | 1.4 | 6.0 | 4.3 | 7.0 | 5.1 | 4.9 | 4.2 | 4.0 | 5.0 | 5.4 | 3.0 | 3.0 | 7.2 | 5.3 | |
| D-cellobiose | 5.7 | 4.7 | 5.5 | 3.1 | 4.6 | 2.0 | 3.6 | 3.2 | 5.0 | 7.4 | 4.2 | 2.2 | 3.0 | 4.0 | 2.3 | 4.9 | 4.9 | 7.5 | 4.6 | 6.2 | 2.6 | 6.2 | |
| α-D-lactose | 4.3 | 3.8 | 2.1 | 2.9 | 3.0 | 6.9 | 2.2 | 5.0 | 1.9 | 4.2 | 3.8 | 2.5 | 1.8 | 2.4 | 1.8 | 4.9 | 2.3 | 2.5 | 3.5 | 3.0 | 2.9 | 2.1 | |
| glucose-1-phosphate | 2.2 | 2.9 | 2.1 | 4.6 | 0.4 | 3.7 | 4.0 | 2.3 | 0.3 | 2.0 | 2.1 | 0.5 | 2.8 | 2.4 | 0.8 | 1.6 | 3.3 | 2.8 | 1.6 | 3.8 | 4.5 | 3.8 | |
| D,L-α-glycerol phosphate | 1.0 | 2.0 | 0.3 | 1.6 | 1.7 | 1.3 | 1.2 | 1.7 | 1.4 | 0.7 | 1.0 | 1.1 | 1.4 | 1.2 | 1.6 | 1.6 | 2.4 | 1.3 | 2.1 | 2.2 | 2.0 | 1.4 | |
| Polymers | tween40 | 3.2 | 3.7 | 3.9 | 5.8 | 2.9 | 6.0 | 4.0 | 4.1 | 7.3 | 4.0 | 4.9 | 8.3 | 3.0 | 4.9 | 7.6 | 6.1 | 3.7 | 5.1 | 2.7 | 4.0 | 4.7 | 3.3 |
| tween80 | 4.4 | 3.8 | 4.1 | 2.4 | 0.0 | 8.7 | 6.7 | 1.6 | 7.4 | 2.5 | 4.4 | 5.9 | 4.1 | 4.5 | 8.6 | 2.8 | 5.1 | 3.1 | 3.4 | 4.8 | 5.1 | 4.6 | |
| α-cyclodextrin | 4.5 | 3.6 | 2.3 | 1.5 | 1.0 | 3.7 | 3.2 | 4.1 | 5.4 | 2.6 | 3.2 | 3.8 | 3.9 | 3.8 | 3.2 | 1.8 | 1.0 | 2.1 | 3.2 | 2.7 | 3.2 | 2.5 | |
| glycogen | 4.7 | 4.3 | 11 | 3.2 | 0.4 | 1.9 | 2.4 | 2.8 | 1.4 | 7.1 | 3.0 | 4.5 | 4.7 | 3.0 | 3.8 | 3.6 | 4.8 | 6.8 | 2.6 | 4.2 | 2.6 | 10 | |
| Carboxylic Acids | D-galactonic acid γ-lactone | 1.9 | 2.9 | 1.0 | 1.3 | 2.3 | 1.0 | 2.6 | 0.8 | 0.5 | 6.1 | 2.8 | 3.2 | 2.9 | 3.7 | 3.1 | 5.3 | 3.5 | 1.5 | 3.3 | 2.0 | 1.9 | 2.0 |
| D-galacturonic acid | 3.4 | 3.0 | 6.4 | 2.6 | 8.2 | 1.5 | 3.0 | 3.4 | 5.0 | 0.3 | 1.6 | 2.9 | 3.6 | 3.2 | 2.6 | 2.5 | 2.8 | 1.4 | 2.6 | 2.0 | 2.2 | 3.9 | |
| γ-hydroxybutyric acid | 0.8 | 1.1 | 0.5 | 1.5 | 3.1 | 1.2 | 0.4 | 0.6 | 1.4 | 1.1 | 3.1 | 0.5 | 0.6 | 1.7 | 1.0 | 2.9 | 2.3 | 1.2 | 2.2 | 1.6 | 2.4 | 1.9 | |
| D-glucosaminic acid | 2.3 | 2.6 | 0.7 | 2.1 | 3.3 | 2.9 | 1.7 | 1.0 | 1.4 | 3.1 | 3.3 | 4.0 | 4.7 | 4.3 | 6.3 | 4.4 | 1.9 | 1.7 | 2.3 | 2.2 | 2.2 | 2.9 | |
| itaconic acid | 4.1 | 3.9 | 6.5 | 4.2 | 5.3 | 6.3 | 2.0 | 6.3 | 1.9 | 3.3 | 3.9 | 3.2 | 4.4 | 3.9 | 3.8 | 4.9 | 4.5 | 5.1 | 9.1 | 3.5 | 2.0 | 3.8 | |
| α-ketobutyric acid | 1.9 | 3.0 | 0.8 | 1.6 | 0.1 | 3.4 | 0.3 | 1.4 | 0.1 | 0.6 | 5.8 | 1.3 | 1.5 | 1.7 | 0.8 | 0.6 | 0.9 | 0.6 | 1.3 | 1.7 | 2.0 | 0.4 | |
| D-malic acid | 1.8 | 1.5 | 0.6 | 0.9 | 1.9 | 0.7 | 0.6 | 1.0 | 0.6 | 2.4 | 1.0 | 1.6 | 5.3 | 4.5 | 3.4 | 1.2 | 2.3 | 1.9 | 2.8 | 2.7 | 2.7 | 0.7 | |
| pyruvic acid methyl ester | 4.3 | 3.0 | 3.9 | 2.9 | 9.7 | 9.9 | 3.9 | 5.5 | 4.1 | 3.0 | 5.1 | 5.6 | 3.5 | 2.9 | 4.0 | 2.8 | 4.5 | 4.9 | 5.5 | 4.3 | 4.3 | 4.7 | |
| Phenolic Compounds | 2-hydroxy benzoic acid | 1.0 | 1.0 | 0.2 | 0.0 | 0.5 | 0.0 | 4.0 | 0.4 | 0.0 | 0.1 | 0.6 | 0.8 | 2.0 | 2.1 | 0.8 | 3.6 | 1.6 | 0.9 | 2.9 | 2.1 | 2.2 | 0.5 |
| 4-hydroxy benzoic acid | 3.3 | 4.4 | 2.1 | 3.0 | 6.8 | 4.4 | 5.4 | 6.4 | 0.9 | 4.7 | 3.8 | 3.9 | 5.3 | 3.4 | 4.4 | 1.9 | 4.1 | 6.1 | 2.0 | 3.8 | 1.7 | 4.9 | |
| Aminoacids | L-arginine | 5.1 | 2.9 | 1.4 | 3.4 | 1.6 | 1.8 | 2.1 | 3.9 | 0.3 | 1.4 | 3.8 | 4.2 | 2.6 | 3.7 | 3.8 | 5.5 | 3.6 | 3.9 | 3.4 | 4.6 | 2.9 | 1.9 |
| L-asparagine | 4.2 | 4.2 | 5.3 | 2.4 | 3.5 | 1.6 | 5.4 | 6.8 | 3.9 | 4.4 | 4.4 | 6.8 | 4.6 | 4.5 | 3.6 | 2.3 | 5.6 | 3.5 | 3.7 | 4.7 | 3.5 | 4.2 | |
| L-phenylalanine | 3.6 | 3.2 | 1.8 | 4.8 | 1.6 | 2.6 | 1.6 | 2.3 | 2.1 | 1.3 | 2.1 | 1.5 | 2.6 | 1.6 | 1.3 | 1.6 | 1.9 | 1.6 | 1.2 | 2.7 | 1.3 | 1.5 | |
| L-serine | 3.6 | 4.1 | 5.7 | 2.0 | 3.2 | 1.6 | 3.0 | 2.3 | 0.6 | 8.0 | 2.7 | 2.9 | 3.6 | 4.2 | 1.9 | 2.9 | 3.9 | 4.3 | 3.7 | 3.6 | 6.9 | 1.1 | |
| L-threonine | 1.5 | 2.0 | 1.3 | 2.3 | 3.7 | 0.9 | 2.0 | 2.9 | 0.6 | 0.9 | 2.3 | 2.1 | 2.1 | 2.6 | 1.4 | 2.9 | 2.6 | 2.2 | 3.2 | 2.4 | 2.6 | 2.0 | |
| glycyl-L-glutamic acid | 1.6 | 1.9 | 1.8 | 3.9 | 1.6 | 1.1 | 0.7 | 1.4 | 1.5 | 0.5 | 2.0 | 1.6 | 2.1 | 2.2 | 2.2 | 1.6 | 1.6 | 3.0 | 2.4 | 2.2 | 1.8 | 1.1 | |
| Amines | phenylethyl-amine | 3.8 | 2.8 | 0.1 | 2.2 | 1.3 | 7.4 | 6.1 | 5.2 | 0.3 | 0.2 | 2.5 | 2.3 | 3.8 | 2.7 | 4.6 | 0.4 | 3.3 | 1.1 | 3.6 | 1.8 | 1.8 | 0.7 |
| putrescine | 2.9 | 4.1 | 4.1 | 6.8 | 0.9 | 3.6 | 4.2 | 5.9 | 8.2 | 3.7 | 4.4 | 4.2 | 2.8 | 3.5 | 6.4 | 4.9 | 2.3 | 5.2 | 4.0 | 3.9 | 6.9 | 4.7 | |
| <1 | 1–2 | 2–3 | 3–4 | 4–5 | 5–6 | 6–7 | 7–8 | 8–9 | 9–10 | >10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melita, M.; Amalfitano, S.; Preziosi, E.; Ghergo, S.; Frollini, E.; Parrone, D.; Zoppini, A. Physiological Profiling and Functional Diversity of Groundwater Microbial Communities in a Municipal Solid Waste Landfill Area. Water 2019, 11, 2624. https://doi.org/10.3390/w11122624
Melita M, Amalfitano S, Preziosi E, Ghergo S, Frollini E, Parrone D, Zoppini A. Physiological Profiling and Functional Diversity of Groundwater Microbial Communities in a Municipal Solid Waste Landfill Area. Water. 2019; 11(12):2624. https://doi.org/10.3390/w11122624
Chicago/Turabian StyleMelita, Marco, Stefano Amalfitano, Elisabetta Preziosi, Stefano Ghergo, Eleonora Frollini, Daniele Parrone, and Annamaria Zoppini. 2019. "Physiological Profiling and Functional Diversity of Groundwater Microbial Communities in a Municipal Solid Waste Landfill Area" Water 11, no. 12: 2624. https://doi.org/10.3390/w11122624
APA StyleMelita, M., Amalfitano, S., Preziosi, E., Ghergo, S., Frollini, E., Parrone, D., & Zoppini, A. (2019). Physiological Profiling and Functional Diversity of Groundwater Microbial Communities in a Municipal Solid Waste Landfill Area. Water, 11(12), 2624. https://doi.org/10.3390/w11122624







