Phosphorus Forms and Associated Properties along an Urban–Rural Gradient in Southern China
Abstract
:1. Introduction
2. Materials and Methods
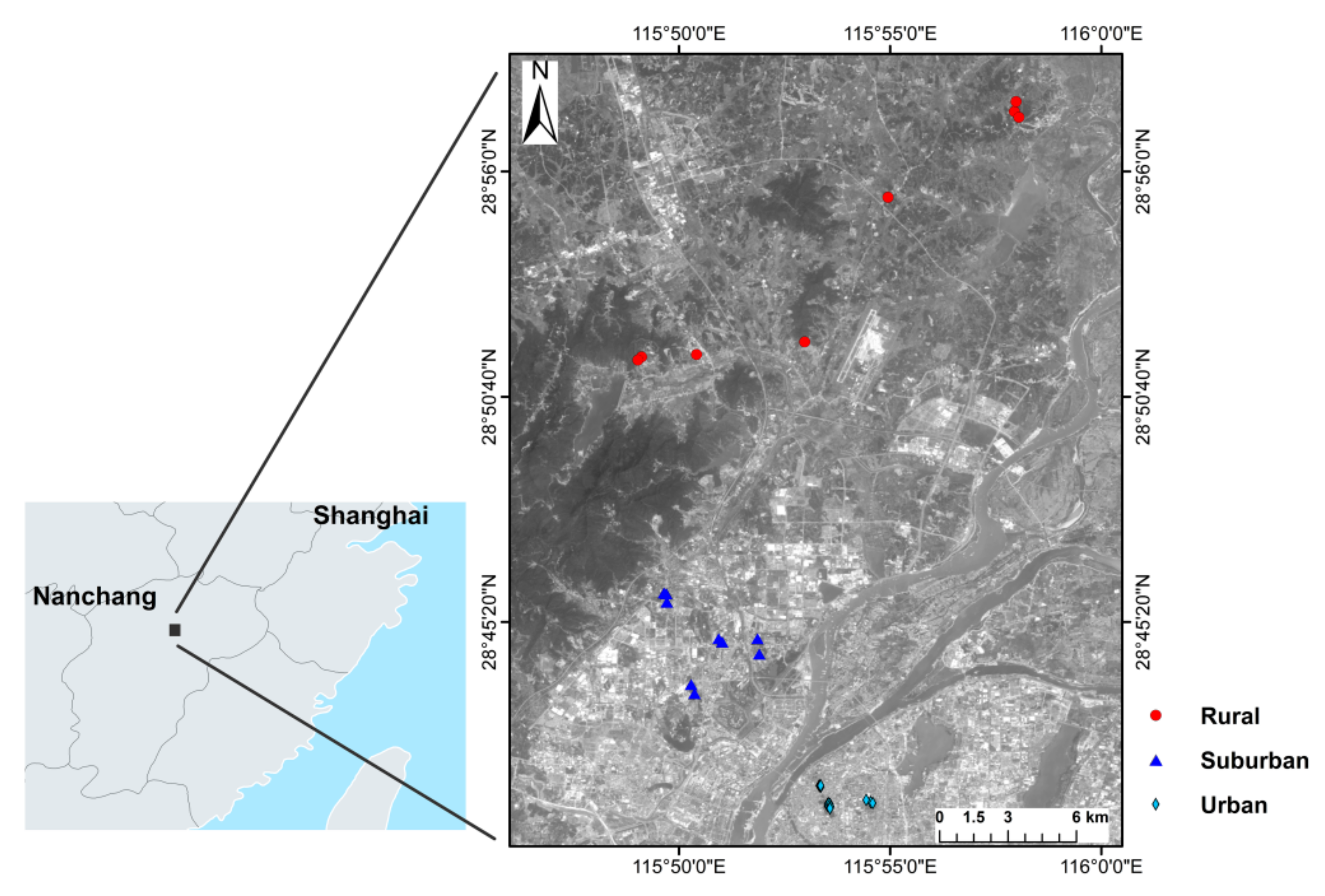
2.1. Studied Site and Soils
2.2. Soil Phosphorus Characterization
2.3. Statistical Analysis
3. Results
3.1. Soil P Fractions
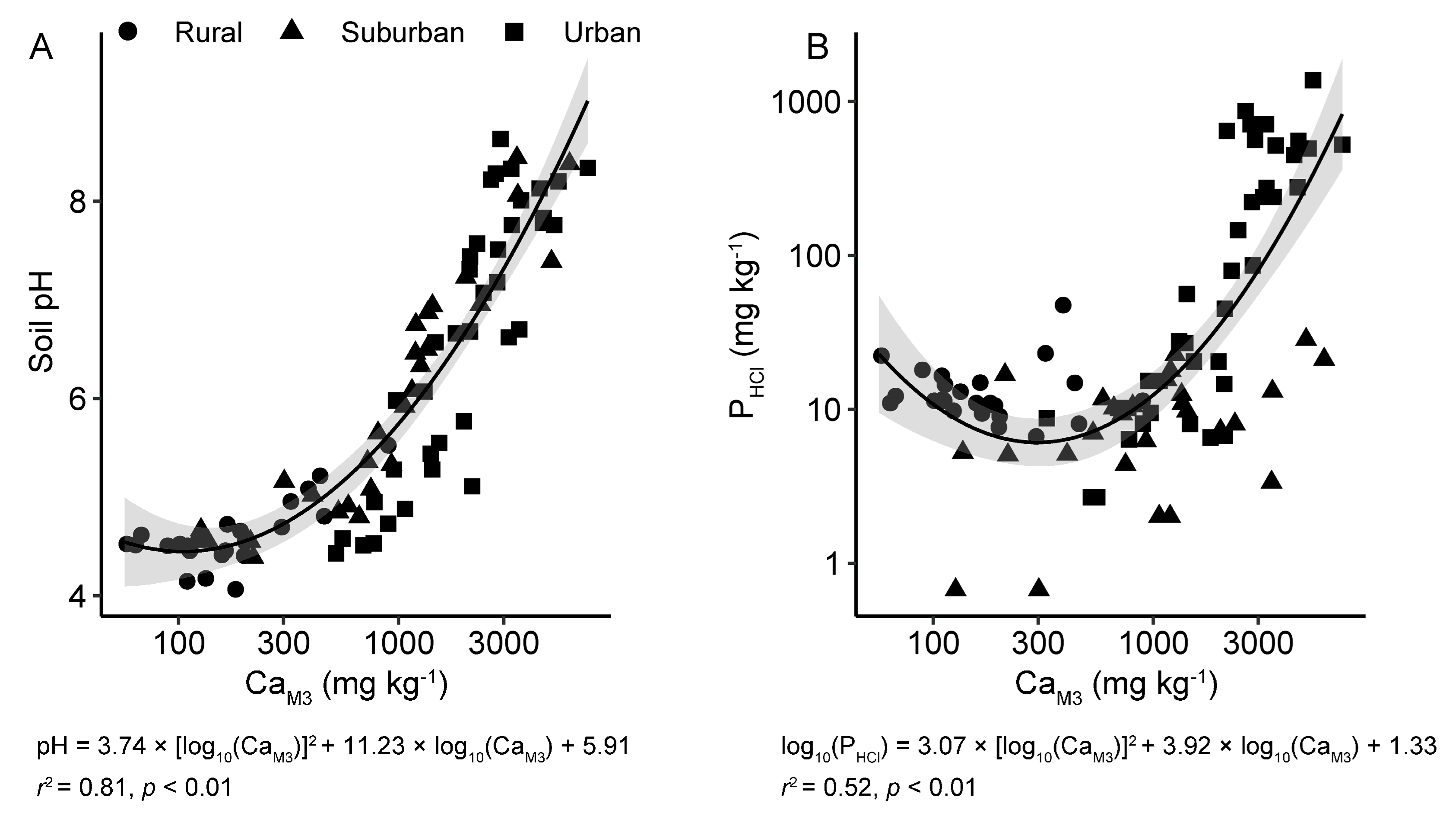
3.2. Soil Properties Associated with P Stabilization
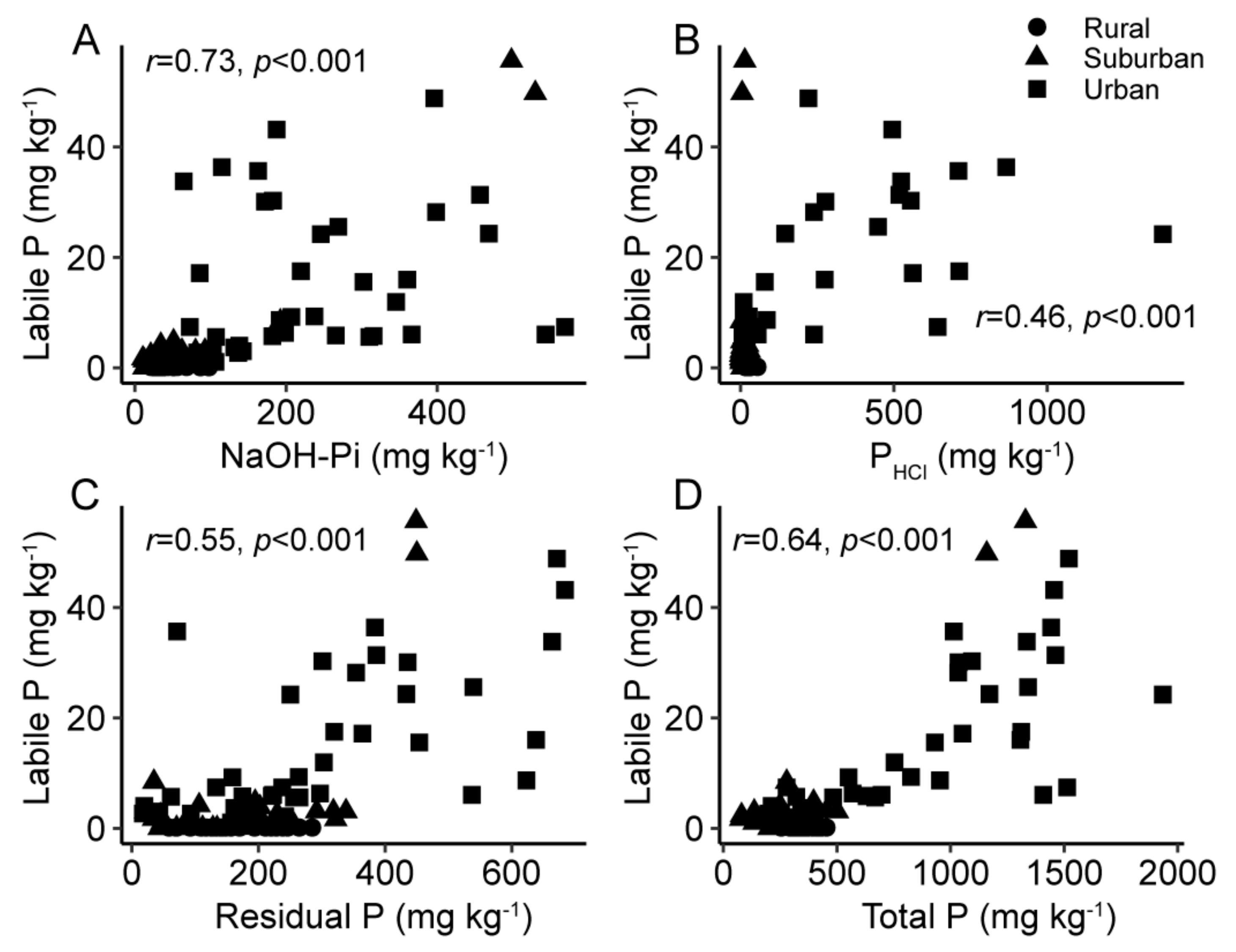
3.3. Relationships between P Fractions and P-Retentive Soil Properties
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- United Nations. World Urbanization Prospects: The 2014 Revision; UN: New York, NY, USA, 2015. [Google Scholar]
- Pan, X.Z.; Zhao, Q.G. Measurement of urbanization process and the paddy soil loss in Yixing city, China between 1949 and 2000. Catena 2007, 69, 65–73. [Google Scholar] [CrossRef]
- Scalenghe, R.; Marsan, F.A. The anthropogenic sealing of soils in urban areas. Landsc. Urban Plan. 2009, 90, 1–10. [Google Scholar] [CrossRef]
- Su, S.; Zhang, Q.; Zhang, Z.; Zhi, J.; Wu, J. Rural settlement expansion and paddy soil loss across an ex-urbanizing watershed in eastern coastal China during market transition. Reg. Environ. Chang. 2011, 11, 651–662. [Google Scholar] [CrossRef]
- Li, G.; Sun, G.X.; Ren, Y.; Luo, X.S.; Zhu, Y.G. Urban soil and human health: A review. Eur. J. Soil Sci. 2018, 69, 196–215. [Google Scholar]
- Pouyat, R.V.; Szlavecz, K.; Yesilonis, I.D.; Groffman, P.M.; Schwarz, K. Chemical, physical, and biological characteristics of urban soils. In Urban Ecosystem Ecology; Aitkenhead-Peterson, J., Volder, A., Eds.; ASA: Madison, WI, USA; CSSA: Madison, WI, USA; SSSA: Madison, WI, USA, 2010; pp. 119–152. [Google Scholar]
- Wei, Z.; Wu, S.; Zhou, S.; Lin, C. Installation of impervious surface in urban areas affects microbial biomass, activity (potential C mineralisation), and functional diversity of the fine earth. Soil Res. 2013, 51, 59–67. [Google Scholar] [CrossRef]
- Wilcke, W.; Müller, S.; Kanchanakool, N.; Zech, W. Urban soil contamination in Bangkok: Heavy metal and aluminium partitioning in topsoils. Geoderma 1998, 86, 211–228. [Google Scholar] [CrossRef]
- Manta, D.S.; Angelone, M.; Bellanca, A.; Neri, R.; Sprovieri, M. Heavy metals in urban soils: A case study from the city of Palermo (Sicily), Italy. Sci. Total Environ. 2002, 300, 229–243. [Google Scholar] [CrossRef]
- Wang, C.H.; Wu, S.H.; Zhou, S.L.; Wang, H.; Li, B.J.; Chen, H.; Na Yu, Y.; Shi, Y.X. Polycyclic aromatic hydrocarbons in soils from urban to rural areas in Nanjing: Concentration, source, spatial distribution, and potential human health risk. Sci. Total Environ. 2015, 527, 375–383. [Google Scholar]
- Zhang, G.L.; Burghardt, W.; Lu, Y.; Gong, Z.T. Phosphorus-enriched soils of urban and suburban Nanjing and their effect on groundwater phosphorus. J. Plant Nutr. Soil Sci. 2001, 164, 295–301. [Google Scholar]
- Daniel, T.C.; Sharpley, A.N.; Lemunyon, J.L. Agricultural Phosphorus and Eutrophication: A Symposium Overview. J. Environ. Qual. 1998, 27, 251–257. [Google Scholar] [CrossRef]
- Barbosa, F.; Bertol, I.; Luciano, R.; González, A.P. Phosphorus losses in water and sediments in runoff of the water erosion in oat and vetch crops seed in contour and downhill. Soil Tillage Res. 2009, 106, 22–28. [Google Scholar] [CrossRef]
- Kleinman, P.J.A.; Sharpley, A.N.; Saporito, L.S.; Buda, A.R.; Bryant, R.B. Application of manure to no-till soils: Phosphorus losses by sub-surface and surface pathways. Nutr. Cycl. Agroecosyst. 2009, 84, 215–227. [Google Scholar] [CrossRef]
- Zhang, M.K. Phosphorus Accumulation in Soils Along an Urban–Rural Land Use Gradient in Hangzhou, Southeast China. Commun. Soil Sci. Plant Anal. 2004, 35, 819–833. [Google Scholar] [CrossRef]
- Golyeva, A.; Zazovskaia, E.; Turova, I. Properties of ancient deeply transformed man-made soils (cultural layers) and their advances to classification by the example of Early Iron Age sites in Moscow Region. Catena 2016, 137, 605–610. [Google Scholar] [CrossRef]
- Hedley, M.J.; Stewart, J.W.B.; Chauhan, B.S. Changes in Inorganic and Organic Soil Phosphorus Fractions Induced by Cultivation Practices and by Laboratory Incubations1. Soil Sci. Soc. Am. J. 1982, 46, 970–976. [Google Scholar] [CrossRef]
- Reddy, K.R.; Wang, Y.; DeBusk, W.F.; Fisher, M.M.; Newman, S. Forms of Soil Phosphorus in Selected Hydrologic Units of the Florida Everglades. Soil Sci. Soc. Am. J. 1998, 62, 1134–1147. [Google Scholar] [CrossRef]
- Pierzynski, G.M.; McDowell, R.W.; Sims, T.J. Chemistry, Cycling, and Potential Movement of Inorganic Phosphorus in Soils. In Phosphorus: Agriculture and the Environment; Sims, T.J., Sharpley, A.N., Eds.; ASA: Madison, WI, USA; CSSA: Madison, WI, USA; SSSA: Madison, WI, USA, 2005; pp. 53–86. [Google Scholar]
- Chen, F.S.; Li, X.; Nagle, G.; Zhan, S.X. Topsoil phosphorus signature in five forest types along an urban-suburban-rural gradient in Nanchang, southern China. J. For. Res. 2010, 21, 39–44. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014, Update 2015, International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports No. 106; FAO: Rome, Italy, 2015. [Google Scholar]
- Irick, D.L.; Yuncong, C.L.; Harris, W.G.; Gu, B.; Ross, M.S.; Migliaccio, K.W.; Inglett, P.W.; Wright, A.L. Characteristics of Soil Phosphorus in Tree Island Hardwood Hammocks of the Southern Florida Everglades. Soil Sci. Soc. Am. J. 2013, 77, 1048–1056. [Google Scholar] [CrossRef]
- Olsen, S.L.; Sommers, L.E. Phosphorus. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA: Madison, WI, USA, 1982; pp. 403–427. [Google Scholar]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta. 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Motsara, M.R.; Roy, R.N. Guide to Laboratory Establishment for Plant Nutrient Analysis; Food and Agriculture Organization of the United Nations: Rome, Italy, 2008. [Google Scholar]
- Schoumans, O.F. Determination of the degree of phosphate saturation in noncalcareous soils. In Methods of Phosphorus Analysis for Soils, Sediments, Residuals, and Waters; Pierzynski, G.M., Ed.; Southern Cooperative Series Bulletin; North Carolina State University: Raleigh, NC, USA, 2000; Volume 369, pp. 31–34. [Google Scholar]
- Ziadi, N.; Tran, T.S. Mehlich 3-extractable elements. In Soil Sampling and Methods of Analysis, 2nd ed.; Carter, M.R., Gregorich, E.G., Eds.; CRC, Taylor & Francis Group: Boca Raton, FL, USA, 2007; pp. 81–88. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- De Mendiburu, F. Agricolae: Statistical Procedures for Agricultural Research; R Package Version 1.1; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; O’hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.M.; Wagner, H. Vegan: Community Ecology Package; R Package Version 2.3-0; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Xia, X.; Zhao, X.; Lai, Y. Levels and distribution of total nitrogen and total phosphorus in urban soils of Beijing, China. Environ. Earth Sci. 2013, 69, 1571–1577. [Google Scholar] [CrossRef]
- Meng, Y.; Cave, M.; Zhang, C. Spatial distribution patterns of phosphorus in top-soils of Greater London Authority area and their natural and anthropogenic factors. Appl. Geochem. 2018, 88, 213–220. [Google Scholar] [CrossRef]
- Yuan, D.G.; Zhang, G.L.; Gong, Z.T.; Burghardt, W. Variations of soil phosphorus accumulation in Nanjing, China as affected by urban development. J. Plant Nutr. Soil Sci. 2007, 170, 244–249. [Google Scholar] [CrossRef]
- Kalmykova, Y.; Harder, R.; Borgestedt, H.; Svanäng, I. Pathways and Management of Phosphorus in Urban Areas. J. Ind. Ecol. 2012, 16, 928–939. [Google Scholar] [CrossRef]
- Gong, Z.T. Chinese Soil Taxonomy: Theory Approaches and Application; China Science Press: Beijing, China, 1999; pp. 160–165. (In Chinese) [Google Scholar]
- Burghardt, W. Soils in urban and industrial environments. J. Plant Nutr. Soil Sci. 1994, 157, 205–214. [Google Scholar] [CrossRef]
- Jim, C.Y. Physical and chemical properties of a Hong Kong roadside soil in relation to urban tree growth. Urban Ecosyst. 1998, 2, 171–181. [Google Scholar] [CrossRef]
- Moore, J.W.; Schindler, D.E.; Scheuerell, M.D.; Smith, D.; Frodge, J. Lake Eutrophication at the Urban Fringe, Seattle Region, USA. AMBIO 2003, 32, 13–18. [Google Scholar] [CrossRef]
- Havlin, J.L.; Beaton, J.D.; Nelson, W.L.; Tisdale, S.L. Soil Fertility and Fertilizers: An Introduction to Nutrient Management, 7th ed.; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2005. [Google Scholar]
- Scalenghe, R.; Edwards, A.C.; Barberis, E.; Ajmone-Marsan, F. Release of phosphorus under reducing and simulated open drainage conditions from overfertilised soils. Chemosphere 2014, 95, 289–294. [Google Scholar] [CrossRef]


| PH2O | PKCl | NaOH-Pi | NaOH-Po | PHCl | Residual P | Total P | |
|---|---|---|---|---|---|---|---|
| Location | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| Depth | NS | NS | NS | 0.002 | NS | <0.001 | 0.02 |
| Location × Depth | NS | NS | NS | NS | NS | 0.03 | NS |
| Location | Depth | pH | SOC | Alox | Feox | CaM3 | MgM3 | Clay |
|---|---|---|---|---|---|---|---|---|
| g kg−1 | mg kg−1 | g kg−1 | ||||||
| Urban | 5 | 6.25 | 18.51 | 1001 | 2261 | 2035 | 160 | 205 |
| 15 | 6.95 | 11.96 | 1356 | 2594 | 2806 | 155 | 223 | |
| 30 | 6.72 | 11.25 | 1559 | 2946 | 2351 | 103 | 205 | |
| Suburban | 5 | 5.99 | 20.95 | 1256 | 2168 | 1732 | 116 | 157 |
| 15 | 6.02 | 10.23 | 1243 | 1995 | 1481 | 86 | 213 | |
| 30 | 6.06 | 7.15 | 1270 | 1873 | 1077 | 60 | 239 | |
| Rural | 5 | 4.65 | 12.30 | 1673 | 2248 | 279 | 28 | 240 |
| 15 | 4.60 | 8.11 | 1720 | 2324 | 174 | 16 | 250 | |
| 30 | 4.56 | 6.90 | 1761 | 2505 | 175 | 16 | 202 | |
| ANOVA | ||||||||
| Location | <0.001 | 0.009 | <0.001 | 0.02 | <0.001 | <0.001 | NS | |
| Depth | NS | <0.001 | NS | NS | NS | 0.003 | NS | |
| Location × Depth | NS | NS | NS | NS | NS | NS | NS | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, G.; Wu, J.; Zheng, X.; Zhou, R.; Wei, Z. Phosphorus Forms and Associated Properties along an Urban–Rural Gradient in Southern China. Water 2019, 11, 2504. https://doi.org/10.3390/w11122504
Qin G, Wu J, Zheng X, Zhou R, Wei Z. Phosphorus Forms and Associated Properties along an Urban–Rural Gradient in Southern China. Water. 2019; 11(12):2504. https://doi.org/10.3390/w11122504
Chicago/Turabian StyleQin, Guobing, Jianfu Wu, Xiaomei Zheng, Rongwei Zhou, and Zongqiang Wei. 2019. "Phosphorus Forms and Associated Properties along an Urban–Rural Gradient in Southern China" Water 11, no. 12: 2504. https://doi.org/10.3390/w11122504
APA StyleQin, G., Wu, J., Zheng, X., Zhou, R., & Wei, Z. (2019). Phosphorus Forms and Associated Properties along an Urban–Rural Gradient in Southern China. Water, 11(12), 2504. https://doi.org/10.3390/w11122504







