Release of Ag/ZnO Nanomaterials and Associated Risks of a Novel Water Sterilization Technology
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of Sterilization Materials
2.2. Methodology for Assessing Release of Nanomaterials and Dissolved Metals
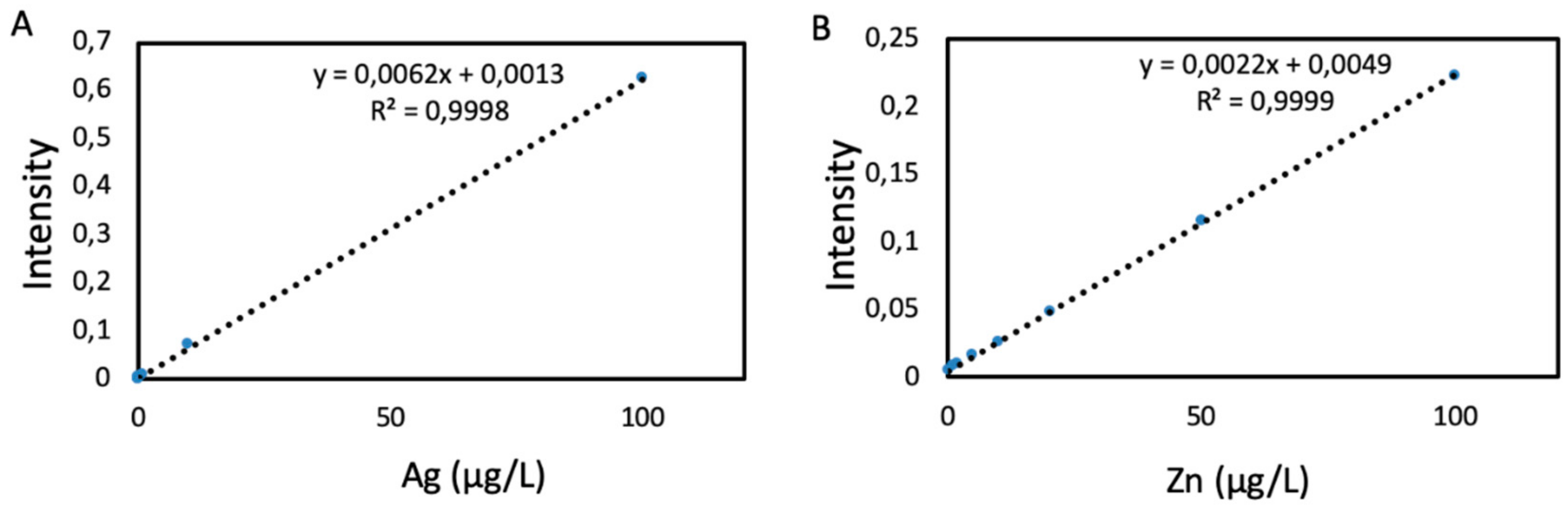
2.3. Characterization of Released Nanomaterials and Dissolved Metals
2.4. Statistical Analysis
3. Results and Discussion
3.1. Characterization of the Sterilization Carbon Cloth
3.2. Media Impact on Metal Releases from Sterilization Carbon Cloth Containing Ag/ZnO Nanomaterials
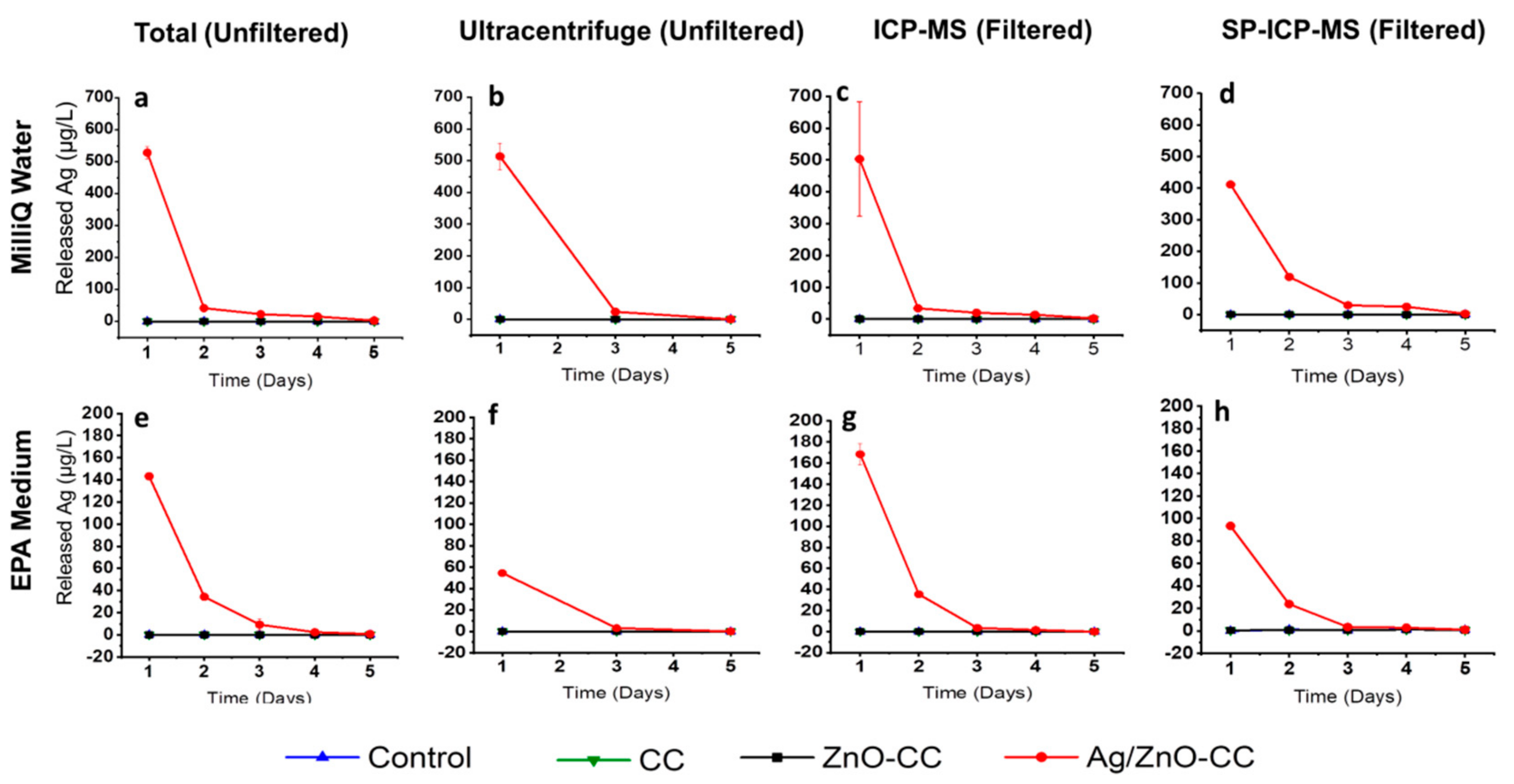
3.2.1. Silver Release
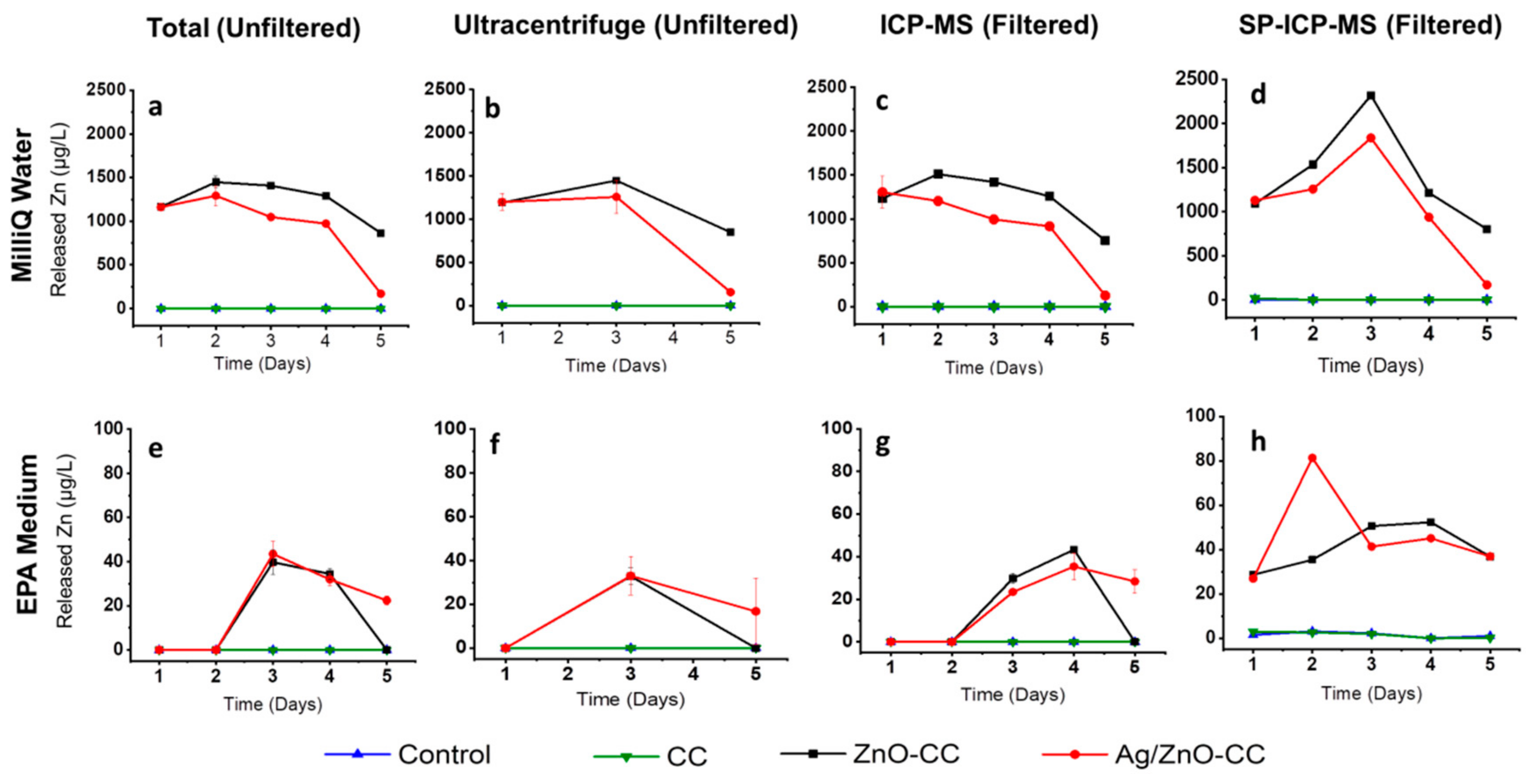
3.2.2. Zinc Release
3.3. Indicative Risk Evaluation of Released Ag and Zn from Sterilization Carbon Cloth Containing Ag/ZnO Nanomaterials to Environment and Human Health
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| TENG | Wave-driven triboelectric nanogenerator |
| CFU | Colony forming units |
| ZnO-NW | ZnO-nanowires |
| AgNPs | Silver nanoparticles |
| EF | Electrical field |
| ROS | Reactive oxygen species |
| SEM | Scanning Electron Microscope |
| TEM | Transmission Electron Microscopy |
| EDX | Energy Dispersive X-Ray Analysis (EDX) |
| ICP-MS | Inductively Coupled Plasma Mass Spectrometry |
| SP-ICP-MS | Single Particle Inductively Coupled Plasma Mass Spectrometry |
| CC | Carbon cloth (CC) |
| ZnO-CC | Carbon cloth containing ZnO-NW |
| Ag/ZnO-CC | Carbon cloth containing AgNPs and ZnO-NW |
| NOM | Natural organic matters |
References
- Alvarez, P.J.; Chan, C.K.; Elimelech, M.; Halas, N.J.; Villagrán, D. Emerging opportunities for nanotechnology to enhance water security. Nat. Nanotechnol. 2018, 13, 634. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Feng, J.; Fan, M.; Pi, Y.; Hu, L.; Han, X.; Liu, M.; Sun, J.; Sun, J. Recent developments in heterogeneous photocatalytic water treatment using visible light-responsive photocatalysts: A review. Rsc Adv. 2015, 5, 14610–14630. [Google Scholar] [CrossRef]
- Kumar, S.G.; Devi, L.G. Review on modified TiO2 photocatalysis under UV/visible light: Selected results and related mechanisms on interfacial charge carrier transfer dynamics. J. Phys. Chem. A 2011, 115, 13211–13241. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yan, T.; Liu, P.; Chen, G.; Shi, L.; Zhang, J.; Zhong, Q.; Zhang, D. In situ creating interconnected pores across 3D graphene architectures and their application as high performance electrodes for flow-through deionization capacitors. J. Mater. Chem. A 2016, 4, 4908–4919. [Google Scholar] [CrossRef]
- Yang, Y.; Hoffmann, M.R. Synthesis and stabilization of blue-black TiO2 nanotube arrays for electrochemical oxidant generation and wastewater treatment. Environ. Sci. Technol. 2016, 50, 11888–11894. [Google Scholar] [CrossRef]
- Liu, S.; Wang, Y.; Zhou, X.; Han, W.; Li, J.; Sun, X.; Shen, J.; Wang, L. Improved degradation of the aqueous flutriafol using a nanostructure macroporous PbO2 as reactive electrochemical membrane. Electrochim. Acta 2017, 253, 357–367. [Google Scholar] [CrossRef]
- Radjenovic, J.; Sedlak, D.L. Challenges and opportunities for electrochemical processes as next-generation technologies for the treatment of contaminated water. Environ. Sci. Technol. 2015, 49, 11292–11302. [Google Scholar] [CrossRef]
- Savage, N.; Diallo, M.S. Nanomaterials and water purification: Opportunities and challenges. J. Nanopart. Res. 2005, 7, 331–342. [Google Scholar] [CrossRef]
- Awual, M.R. A novel facial composite adsorbent for enhanced copper (II) detection and removal from wastewater. Chem. Eng. J. 2015, 266, 368–375. [Google Scholar] [CrossRef]
- Shahat, A.; Hassan, H.M.; Azzazy, H.M.; El-Sharkawy, E.A.; Abdou, H.M.; Awual, M.R. Novel hierarchical composite adsorbent for selective lead (II) ions capturing from wastewater samples. Chem. Eng. J. 2018, 332, 377–386. [Google Scholar] [CrossRef]
- Karn, B.; Masciangioli, T.; Zhan, W.X.; Colvin, V.; Alivisatos, P. Nanotechnology and the Environment: Applications and Implications; Oxford University Press: Oxford, MS, USA, 2005. [Google Scholar]
- Awual, M.R.; Yaita, T.; Shiwaku, H. Design a novel optical adsorbent for simultaneous ultra-trace cerium (III) detection, sorption and recovery. Chem. Eng. J. 2013, 228, 327–335. [Google Scholar] [CrossRef]
- Awual, M.R.; Hasan, M.M.; Znad, H. Organic-inorganic based nano-conjugate adsorbent for selective palladium (II) detection, separation and recovery. Chem. Eng. J. 2015, 259, 611–619. [Google Scholar] [CrossRef]
- Awual, M.R.; Hasan, M.M. Colorimetric detection and removal of copper (II) ions from wastewater samples using tailor-made composite adsorbent. Sens. Actuators B Chem. 2015, 206, 692–700. [Google Scholar] [CrossRef]
- Awual, M.R.; Eldesoky, G.E.; Yaita, T.; Naushad, M.; Shiwaku, H.; AlOthman, Z.A.; Suzuki, S. Schiff based ligand containing nano-composite adsorbent for optical copper (II) ions removal from aqueous solutions. Chem. Eng. J. 2015, 279, 639–647. [Google Scholar] [CrossRef]
- Awual, M.R. Assessing of lead (III) capturing from contaminated wastewater using ligand doped conjugate adsorbent. Chem. Eng. J. 2016, 289, 65–73. [Google Scholar] [CrossRef]
- Awual, M.R.; Khraisheh, M.; Alharthi, N.H.; Luqman, M.; Islam, A.; Karim, M.R.; Rahman, M.M.; Khaleque, M.A. Efficient detection and adsorption of cadmium (II) ions using innovative nano-composite materials. Chem. Eng. J. 2018, 343, 118–127. [Google Scholar] [CrossRef]
- Awual, M.R. Innovative composite material for efficient and highly selective Pb (II) ion capturing from wastewater. J. Mol. Liq. 2019, 284, 502–510. [Google Scholar] [CrossRef]
- Awual, M.R.; Asiri, A.M.; Rahman, M.M.; Alharthi, N.H. Assessment of enhanced nitrite removal and monitoring using ligand modified stable conjugate materials. Chem. Eng. J. 2019, 363, 64–72. [Google Scholar] [CrossRef]
- Shahat, A.; Hassan, H.M.; Azzazy, H.M.; Hosni, M.; Awual, M.R. Novel nano-conjugate materials for effective arsenic (V) and phosphate capturing in aqueous media. Chem. Eng. J. 2018, 331, 54–63. [Google Scholar] [CrossRef]
- Yavuz, C.T.; Mayo, J.T.; William, W.Y.; Prakash, A.; Falkner, J.C.; Yean, S.; Cong, L.; Shipley, H.J.; Kan, A.; Tomson, M.; et al. Low-field magnetic separation of monodisperse Fe3O4 nanocrystals. Science 2006, 314, 964–967. [Google Scholar] [CrossRef]
- Wickson, F.; Hartmann, N.B.; Hjorth, R.; Hansen, S.F.; Wynne, B.; Baun, A. Balancing scientific tensions. Nat. Nanotechnol. 2014, 9, 870. [Google Scholar] [CrossRef] [PubMed]
- Cupi, D.; Hartmann, N.B.; Baun, A. Influence of pH and media composition on suspension stability of silver, zinc oxide, and titanium dioxide nanoparticles and immobilization of Daphnia magna under guideline testing conditions. Ecotoxicol. Environ. Saf. 2016, 127, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, R.; Skjolding, L.M.; Sørensen, S.N.; Baun, A. Regulatory adequacy of aquatic ecotoxicity testing of nanomaterials. NanoImpact 2017, 8, 28–37. [Google Scholar] [CrossRef]
- Pang, C.; Hristozov, D.; Zabeo, A.; Pizzol, L.; Tsang, M.P.; Sayre, P.; Marcomini, A. Probabilistic approach for assessing infants’ health risks due to ingestion of nanoscale silver released from consumer products. Environ. Int. 2017, 99, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Baun, A.; Sayre, P.; Steinhäuser, K.G.; Rose, J. Regulatory relevant and reliable methods and data for determining the environmental fate of manufactured nanomaterials. NanoImpact 2017, 8, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Docter, D.; Westmeier, D.; Markiewicz, M.; Stolte, S.; Knauer, S.K.; Stauber, R.H. The nanoparticle biomolecule corona: Lessons learned–challenge accepted? Chem. Soc. Rev. 2015, 44, 6094–6121. [Google Scholar] [CrossRef]
- Feliu, N.; Docter, D.; Heine, M.; del Pino, P.; Ashraf, S.; Kolosnjaj-Tabi, J.; Macchiarini, P.; Nielsen, P.; Alloyeau, D.; Gazeau, F.; et al. In vivo degeneration and the fate of inorganic nanoparticles. Chem. Soc. Rev. 2016, 45, 2440–2457. [Google Scholar] [CrossRef] [Green Version]
- Setyawati, M.I.; Tay, C.Y.; Docter, D.; Stauber, R.H.; Leong, D.T. Understanding and exploiting nanoparticles’ intimacy with the blood vessel and blood. Chem. Soc. Rev. 2015, 44, 8174–8199. [Google Scholar] [CrossRef]
- Hansen, S.F.; Heggelund, L.R.; Besora, P.R.; Mackevica, A.; Boldrin, A.; Baun, A. Nanoproducts—What is actually available to European consumers? Environ. Sci. Nano 2016, 3, 169–180. [Google Scholar] [CrossRef]
- Siemer, S.; Westmeier, D.; Barz, M.; Eckrich, J.; Wünsch, D.; Seckert, C.; Thyssen, C.; Schilling, O.; Hasenberg, M.; Pang, C.; et al. Biomolecule-corona formation confers resistance of bacteria to nanoparticle. Biomaterials 2019, 192, 551–559. [Google Scholar] [CrossRef]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.Z.; Kim, T.N.; Kim, J.O. A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Gottschalk, F.; Nowack, B. The release of engineered nanomaterials to the environment. J. Environ. Monit. 2011, 13, 1145–1155. [Google Scholar] [CrossRef] [PubMed]
- Mackevica, A.; Foss Hansen, S. Release of nanomaterials from solid nanocomposites and consumer exposure assessment—A forward-looking review. Nanotoxicology 2016, 10, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Feng, H.; Yan, L.; Yu, M.; Ouyang, H.; Li, H.; Jiang, W.; Jin, Y.; Zhu, G.; Li, Z.; et al. A self-powered sterilization system with both instant and sustainable anti-bacterial ability. Nano Energy 2017, 36, 241–249. [Google Scholar] [CrossRef]
- Miao, A.J.; Luo, Z.; Chen, C.S.; Chin, W.C.; Santschi, P.H.; Quigg, A. Intracellular uptake: A possible mechanism for silver engineered nanoparticle toxicity to a freshwater alga Ochromonas danica. PLoS ONE 2010, 5, 15196. [Google Scholar] [CrossRef] [PubMed]
- Ivask, A.; ElBadawy, A.; Kaweeteerawat, C.; Boren, D.; Fischer, H.; Ji, Z.; Chang, C.H.; Liu, R.; Tolaymat, T.; Telesca, D.; et al. Toxicity mechanisms in Escherichia coli vary for silver nanoparticles and differ from ionic silver. ACS Nano 2013, 8, 374–386. [Google Scholar] [CrossRef]
- Zeng, X.; McCarthy, D.T.; Deletic, A.; Zhang, X. Silver/reduced graphene oxide hydrogel as novel bactericidal filter for point-of-use water disinfection. Adv. Funct. Mater. 2015, 25, 4344–4351. [Google Scholar] [CrossRef]
- Loo, S.L.; Krantz, W.B.; Fane, A.G.; Gao, Y.; Lim, T.T.; Hu, X. Bactericidal mechanisms revealed for rapid water disinfection by superabsorbent cryogels decorated with silver nanoparticles. Environ. Sci. Technol. 2015, 49, 2310–2318. [Google Scholar] [CrossRef]
- Pang, C.; Neubauer, N.; Boyles, M.; Brown, D.; Kanase, N.; Hristozov, D.; Fernandes, T.; Stone, V.; Wohlleben, W.; Marcomini, A. Releases from transparent blue automobile coatings containing nanoscale copper phthalocyanine and their effects on J774 A1 macrophages. NanoImpact 2017, 7, 75–83. [Google Scholar] [CrossRef]
- Spielman-Sun, E.; Zaikova, T.; Dankovich, T.; Yun, J.; Ryan, M.; Hutchison, J.E.; Lowry, G.V. Effect of silver concentration and chemical transformations on release and antibacterial efficacy in silver-containing textiles. NanoImpact 2018, 11, 51–57. [Google Scholar] [CrossRef]
- Ivask, A.; Kurvet, I.; Kasemets, K.; Blinova, I.; Aruoja, V.; Suppi, S.; Vija, H.; Käkinen, A.; Titma, T.; Heinlaan, M.; et al. Size-dependent toxicity of silver nanoparticles to bacteria, yeast, algae, crustaceans and mammalian cells in vitro. PLoS ONE 2014, 9, 102108. [Google Scholar] [CrossRef] [PubMed]
- Bondarenko, O.; Juganson, K.; Ivask, A.; Kasemets, K.; Mortimer, M.; Kahru, A. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: A critical review. Arch. Toxicol. 2013, 87, 1181–1200. [Google Scholar] [CrossRef] [PubMed]
- Kjølholt, J.; Gottschalk, F.; Brinch, A.; Lützhøft, H.C.H.; Hartmann, N.B.; Nowack, B.; Baun, A. Environmental Assessment of Nanomaterial Use in Denmark: Final Report; Danish Environmental Protection Agency: Copenhagen, Denmark, 15 September 2015. [Google Scholar]
- ECHA (The European Chemicals Agency). 2012. Available online: https://echa.europa.eu/registration-dossier/-/registered-dossier/16155/6/2/1 (accessed on 5 April 2019).
- OECD (The Organisation for Economic Co-Operation and Development). Example for the OECD Workshop on Metals Specificities in Environmental Hazard Assessment Paris, September 7–8, 2011. Available online: http://www.oecd.org/chemicalsafety/risk-assessment/48720427.pdf (accessed on 4 May 2018).
- Krishnaraj, C.; Jagan, E.G.; Rajasekar, S.; Selvakumar, P.; Kalaichelvan, P.T.; Mohan, N.J.C.S.B.B. Synthesis of silver nanoparticles using Acalypha indica leaf extracts and its antibacterial activity against water borne pathogens. Colloids Surf. B Biointerfaces 2010, 76, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Navarro, E.; Piccapietra, F.; Wagner, B.; Marconi, F.; Kaegi, R.; Odzak, N.; Sigg, L.; Behra, R. Toxicity of silver nanoparticles to Chlamydomonas reinhardtii. Environ. Sci. Technol. 2008, 42, 8959–8964. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organization). a. Silver in Drinking-Water-Background Document for Development of WHO Guidelines for Drinking-Water Quality. Available online: https://www.who.int/water_sanitation_health/dwq/chemicals/silver.pdf (accessed on 2 March 2019).
- WHO (World Health Organization). b. Zinc in Drinking-Water-Background Document for Development of WHO Guidelines for Drinking-Water Quality. WHO/SDE/WSH/03.04/17. Available online: https://www.who.int/water_sanitation_health/dwq/chemicals/zinc.pdf (accessed on 31 October 2019).
- ECHA (The European Chemicals Agency). Guidance on Information Requirements and Chemical Safety Assessment. 2016. Available online: https://echa.europa.eu/documents/10162/13632/information_requirements_r16_en.pdf (accessed on 5 May 2018).
- U.S. EPA. (U.S. Environmental Protection Agency). Office of Pesticide Programs, Decision Document: Conditional Registration of HeiQ AGS-20 as a Materials Preservative in Textiles, at iv-v (Dec. 1, 2011). 2011; Available online: https://caat.jhsph.edu/programs/workshops/HeiQ%20Proposed%20Registration%20Decision%20Document%208-12-2010.pdf (accessed on 6 April 2014).


| Metals | Zn (mg) | Ag (mg) | ||||||
|---|---|---|---|---|---|---|---|---|
| CC Type | CC | ZnO-CC | Ag/ZnO-CC | Ag/ZnO-CC | ||||
| Medium | MilliQ | EPA | MilliQ | EPA | MilliQ | EPA | MilliQ | EPA |
| Remaining on CC (5 days) | 0.03 | 0.06 | 18.56 | 20.52 | 27.19 | 30.67 | 0.50 | 0.39 |
| Total release (medium) 1 | < 0.06 | < 0.06 | 1.90 | 0.12 | 1.54 | 0.28 | 0.20 | 0.06 |
| Adsorption on tubes | - | - | 0.04 | 0.98 | 0.15 | 0.25 | 0.015 | 0.003 |
| Recovery based on mass balance 2 | - | - | −3.2% | +3.2% | +11.8% | +20.5% | −55.8% | −71.6% |
| Washing Methods | Media | MilliQ Media | EPA Media | ||
|---|---|---|---|---|---|
| Metals | Zn (µg/L) | Ag (µg/L) | Zn (µg/L) | Ag (µg/L) | |
| MilliQ (6th day) | Control | < 20 | < 1 | < 20 | < 1 |
| CC | < 20 | < 1 | < 20 | < 1 | |
| ZnO-CC | < 20 | < 1 | < 20 | < 1 | |
| Ag/ZnO-CC | < 20 | < 1 | 70.5 ± 4.16 | 1.11 ± 0.018 | |
| 10% HNO3 (7th day) | Control | < 20 | < 1 | <1 | < 1 |
| CC | < 20 | < 1 | 23.52 ± 3.78 | < 1 | |
| ZnO-CC | 142.52 ± 5.65 | < 1 | 327.3 ± 11.5 | < 1 | |
| Ag/ZnO-CC | 496.06 ± 120.6 | 50.37 ± 2.69 | 754.53 ± 18.75 | < 1 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pang, C.; Mackevica, A.; Tian, J.; Feng, H.; Li, Z.; Baun, A. Release of Ag/ZnO Nanomaterials and Associated Risks of a Novel Water Sterilization Technology. Water 2019, 11, 2276. https://doi.org/10.3390/w11112276
Pang C, Mackevica A, Tian J, Feng H, Li Z, Baun A. Release of Ag/ZnO Nanomaterials and Associated Risks of a Novel Water Sterilization Technology. Water. 2019; 11(11):2276. https://doi.org/10.3390/w11112276
Chicago/Turabian StylePang, Chengfang, Aiga Mackevica, Jingjing Tian, Hongqing Feng, Zhou Li, and Anders Baun. 2019. "Release of Ag/ZnO Nanomaterials and Associated Risks of a Novel Water Sterilization Technology" Water 11, no. 11: 2276. https://doi.org/10.3390/w11112276







