1. Introduction
Global change is resulting in more variable environmental conditions in both terrestrial and aquatic ecosystems [
1,
2,
3], which may in turn be altering the variability of biotic communities [
4,
5,
6]. Here we define variability as biomass variability, that is, as the temporal variability of biomass in an ecological community that is summed across all taxa present [
7,
8], and compositional variability as the relative change in the composition of taxa within that community [
9,
10,
11]. Understanding the variability of ecological communities is critical to predicting how ecosystems will respond to environmental change [
12].
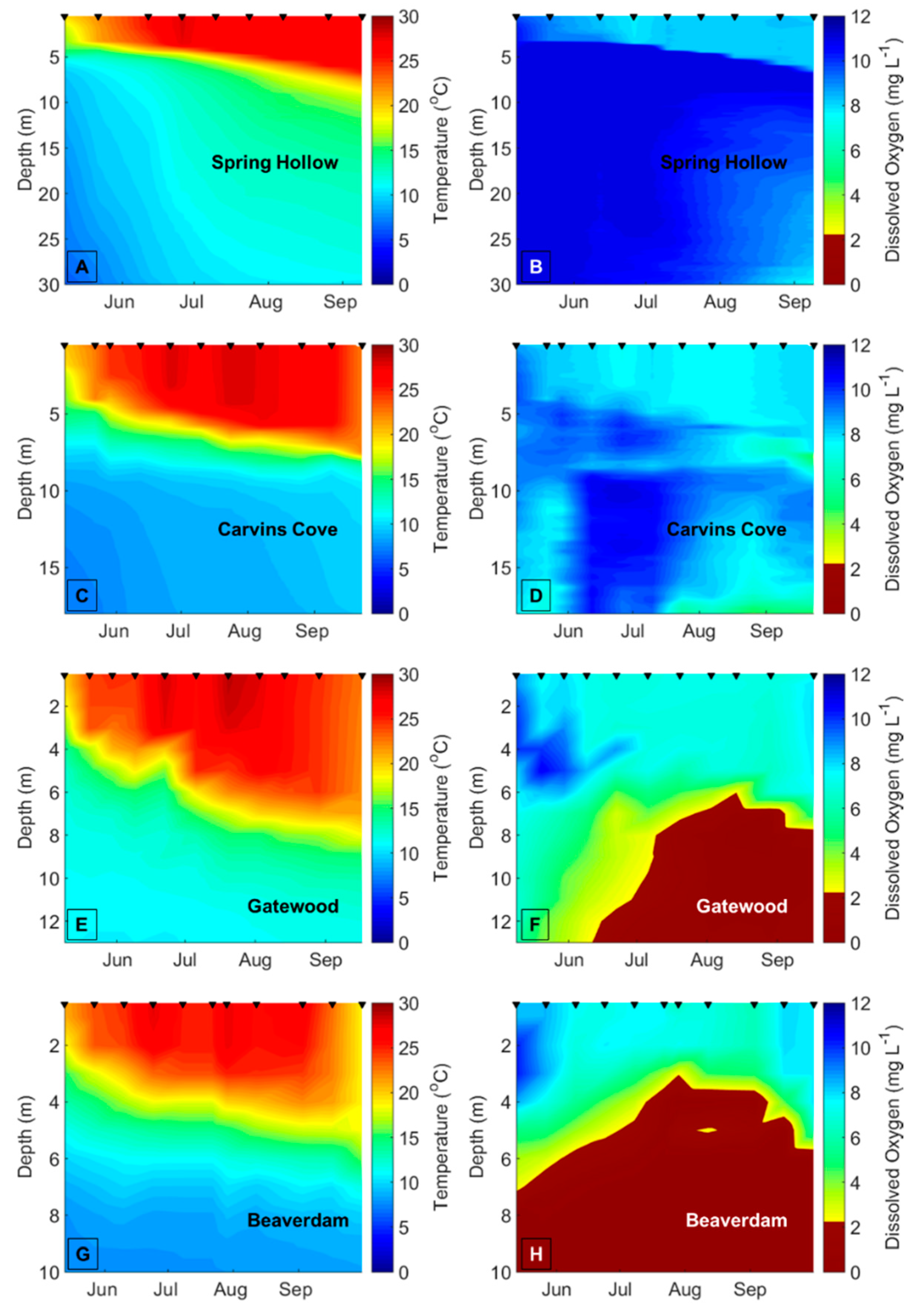
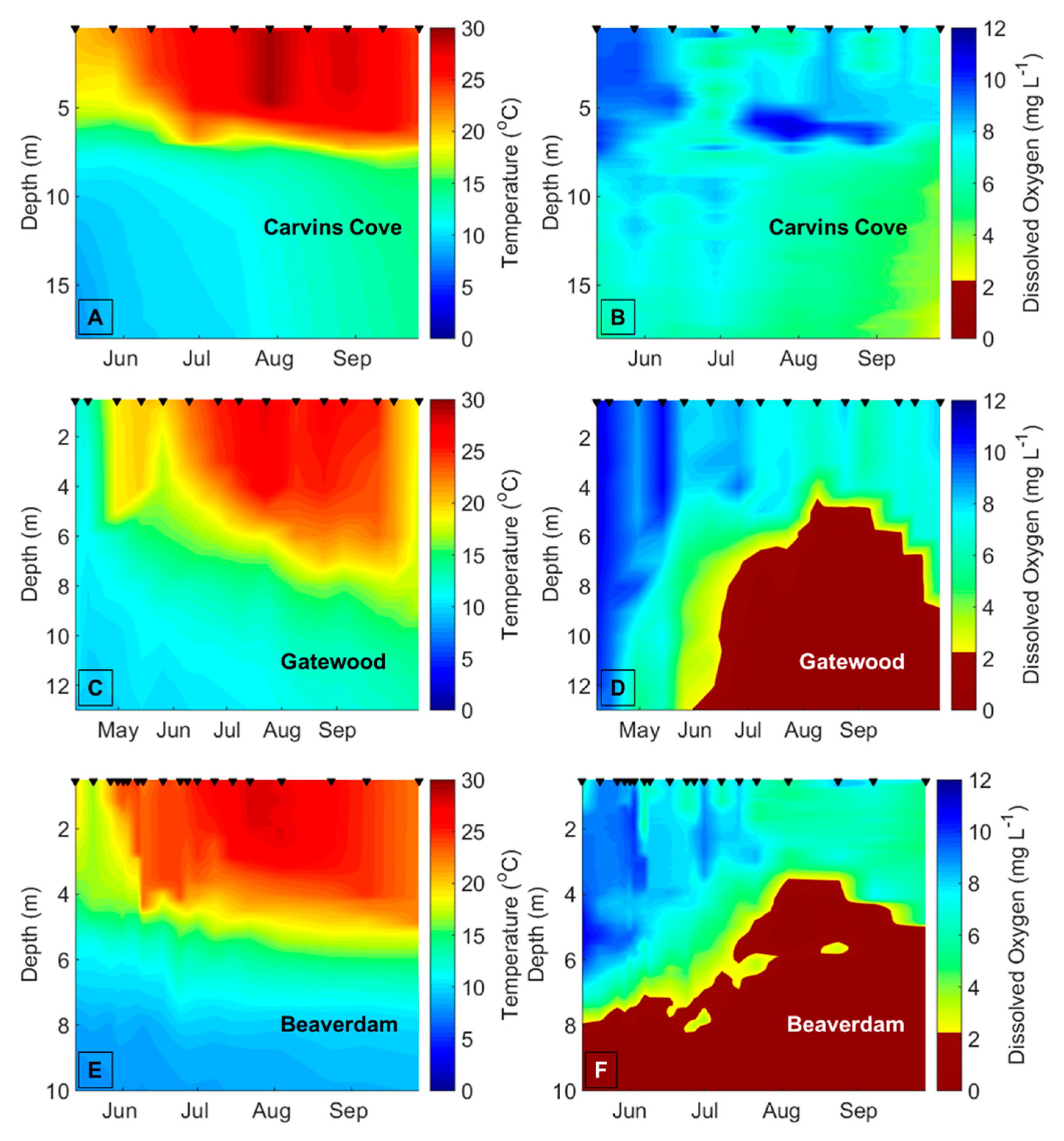
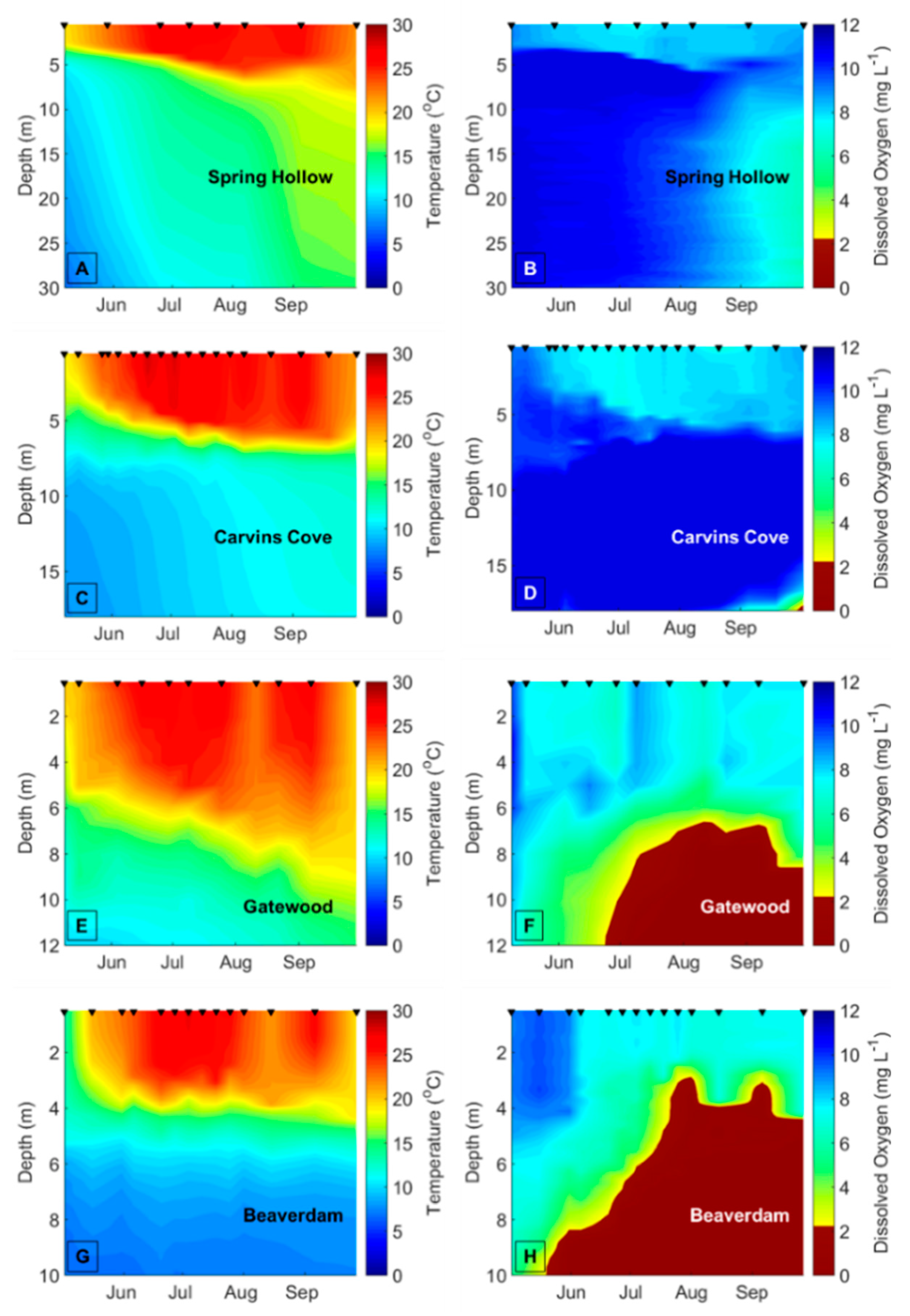
Decreasing dissolved oxygen (DO) concentrations below 2 mg L
−1 in the bottom waters of lakes and reservoirs (hypolimnetic hypoxia) may disrupt biotic interactions and change the variability of freshwater ecological communities [
13,
14]. Hypolimnetic hypoxia is increasing in many lakes and reservoirs due to climate change, as the strength of thermal stratification increases due to warmer surface water (epilimnion) temperatures, thereby decreasing the exchange of oxygen between the epilimnion and hypolimnion [
15,
16,
17]. Higher nutrient concentrations due to eutrophication from land use change are also driving a decrease in DO concentrations in the hypolimnion of many waterbodies [
18,
19,
20].
The variability of crustacean (copepod + cladoceran) zooplankton communities may be sensitive to hypolimnetic hypoxia, because zooplankton require DO for respiration and metabolism [
21,
22], and are more sensitive to lower DO concentrations than some other aquatic animals, such as fish [
23]. Zooplankton play a crucial role in regulating water quality and food webs in aquatic ecosystems because of their position in the food chain: zooplankton eat phytoplankton at the base of the food web, and are in turn consumed by fish [
24,
25]. Subsequently, understanding the effects of hypoxia on zooplankton community variability is important because alterations to zooplankton communities can have escalating effects up and down the food chain.
Hypoxia may decrease the biomass variability of zooplankton taxa that typically have a greater prevalence later in the season when hypoxia would generally occur in temperate, thermally-stratified waterbodies [
26], thereby decreasing the biomass variability of the crustacean zooplankton community. Generally, larger cladoceran taxa (i.e., daphnids) dominate the zooplankton community in the early summer months, whereas copepods and smaller cladocera have greater dominance (defined as the proportion of a taxon’s biomass within the total crustacean zooplankton community’s biomass) in later summer months [
26,
27]. Cladocera, like
Daphnia spp., focus most of their energy into reproduction, and subsequently grow quickly from benthic dormant stages in the spring months, and mature quickly after emerging into the water column. Copepods grow slower, molt multiple times before adulthood, and put more energy into energy storage before reproduction [
28,
29]. Hypoxia may inhibit the growth of small cladocera and copepods in the late summer, resulting in more consistent levels of biomass, and thereby lower biomass variability.
The life history characteristics of different taxa within the cladoceran and copepod communities may result in differential responses to hypolimnetic hypoxia, resulting in a decrease in compositional variability [
30]. Functional trait and size differences between different crustacean zooplankton taxa could underlie their hypoxia-tolerance: e.g., there may be differences in their hemoglobin-carrying capacity [
31,
32] or adaptations which allow them to be present longer in the oxic epilimnion during the daytime [
33,
34,
35]. Therefore, hypoxia may increase the presence and dominance of certain zooplankton taxa in the zooplankton community that are less sensitive to hypoxic conditions, thereby decreasing zooplankton compositional variability under hypoxic conditions vs. oxic conditions.
We collected zooplankton and other environmental variables in four reservoirs during the thermally-stratified period across three years to assess how hypolimnetic DO concentrations alter crustacean zooplankton biomass variability and compositional variability. Two of the reservoirs remained fully oxic during the thermally-stratified period, whereas the other two reservoirs developed seasonal hypolimnetic hypoxia. We predicted that that there would be no difference in the biomass variability or compositional variability of the zooplankton communities between the early and late stratified periods in the oxic reservoirs. In the hypoxic reservoirs, however, we predicted that there would be lower biomass variability and compositional variability of zooplankton communities in the late stratified period, compared to the early stratified period.
3. Data Analyses
Our goal was to examine how the presence or absence of hypolimnetic hypoxia affected the variability of the zooplankton community throughout the summer stratified season, while also accounting for the changes in the zooplankton community that naturally occur from early to late summer due to seasonal succession [
26,
45,
46]. To conduct these comparisons, we followed the precedent of prior studies, which divided the summer stratified season into different periods [
26,
47]. We used the terms “early” and “late” stratified periods to denote approximately the first and second half of the monitoring period of each reservoir, not the entire stratified period, which represented April–October. We then compared the variability of the zooplankton community between these early and late summer stratified periods in reservoirs that exhibited late summer oxic vs. hypoxic hypolimnetic conditions.
Our main response variables were the biomass variability of crustacean, copepod and cladoceran zooplankton, and the compositional variability of crustacean zooplankton biomass. Biomass variability was measured as the coefficient of variation (CV) of total crustacean, cladoceran and copepod zooplankton biomass [
8,
48]. Compositional variability for the crustacean zooplankton communities was calculated from quantifying biomass for each taxon on each sampling day and then through measuring multivariate dispersion by Euclidean distance using the betadisper function in the
vegan package in R [
49,
50,
51,
52]. We calculated compositional variability of the zooplankton communities using the Bray-Curtis index to assess differences in taxa dominance in the zooplankton community [
53]. We did not conduct compositional variability analyses using the Jaccard index, which is based on taxa presence/absence comparisons, because of the low number of zooplankton genera present in the reservoirs.
We first performed Kruskal-Wallis analyses to test the effect of sampling year (2014, 2015, and 2016) on the biomass variability and compositional variability of the zooplankton response variables to confirm the independence of zooplankton communities among years. We chose Kruskal-Wallis tests for these analyses because of the unbalanced sample sizes of reservoirs among years, as no zooplankton community data were collected in SHR in 2016.
To demarcate the early vs. late stratified periods each year in BVR and GWR, the two reservoirs that exhibited summer hypolimnetic hypoxia, we first determined the depth of the thermocline on each sampling date using rLakeAnalyzer [
54], and then calculated the mean hypolimnetic DO concentrations on the sampling dates from the CTD or YSI DO profiles. We used the mean hypolimnetic DO concentrations to identify the timing of the oxic (DO > 2 mg L
−1) and hypoxic (DO ≤ 2 mg L
−1) periods within each summer (
Table A1). The hypolimnion in GWR and BVR became hypoxic between late June and mid-July every year, except in BVR in 2015, when the hypolimnion became hypoxic prior to the beginning of the sampling season (14 May), so it was impossible to demarcate the onset of hypoxia in the BVR that year. We assigned 20 July as the breakpoint between the early and late stratified periods in CCR and SHR, the two reservoirs that never exhibited hypoxia, across all years because both BVR and GWR always exhibited hypolimnetic hypoxia after this date every year, and this delineation evenly divided the number of sampling dates between the early and late stratified periods. To ensure that our results were not confounded by the delineation of the early vs. late stratified periods, we reran all analyses with different breakpoints between the early and late stratified period that ranged from ± 1 week of the oxic/hypoxic boundaries (
Table A2 and
Table A3).
We then calculated the biomass variability of crustacean, copepod and cladoceran zooplankton measured on sample days within the early stratified vs. late stratified periods for each reservoir and year. To account for different numbers of sampling dates in the early vs. late stratified periods, we randomly sampled the minimum number of observations (n = 3) during the shortest early or late period 1000 times (mean
n of observations within a period = 5.2 ± 2.1, 1 SD) [
55,
56]. We calculated the mean CV from the 1000 samples to ensure that the biomass variability estimates were comparable between early and late stratified periods. We similarly repeated the bootstrapping to calculate the compositional variability from the minimum number of observations during a stratified period. Only the compositional variability of the aggregated crustacean zooplankton community (not individual copepod or cladoceran groups) was calculated, since there were only two copepod genera in the reservoirs.
Wilcoxon signed rank tests were performed to compare changes in the biomass variability and compositional variability of the reservoirs’ zooplankton communities between the early and late stratified periods. We grouped the reservoirs by whether they exhibited summer hypolimnetic hypoxia (i.e., oxic CCR and SHR vs. hypoxic BVR and GWR) and performed the two-tailed tests separately for the biomass variability of total crustacean, copepod, and cladoceran zooplankton and the compositional variability of crustacean zooplankton biomass in the oxic vs. hypoxic reservoirs. Wilcoxon signed rank tests were required because there was not a seasonally oxic period of the sampling season for BVR in 2015, and no samples were analyzed from SHR in 2016, creating an unbalanced design.
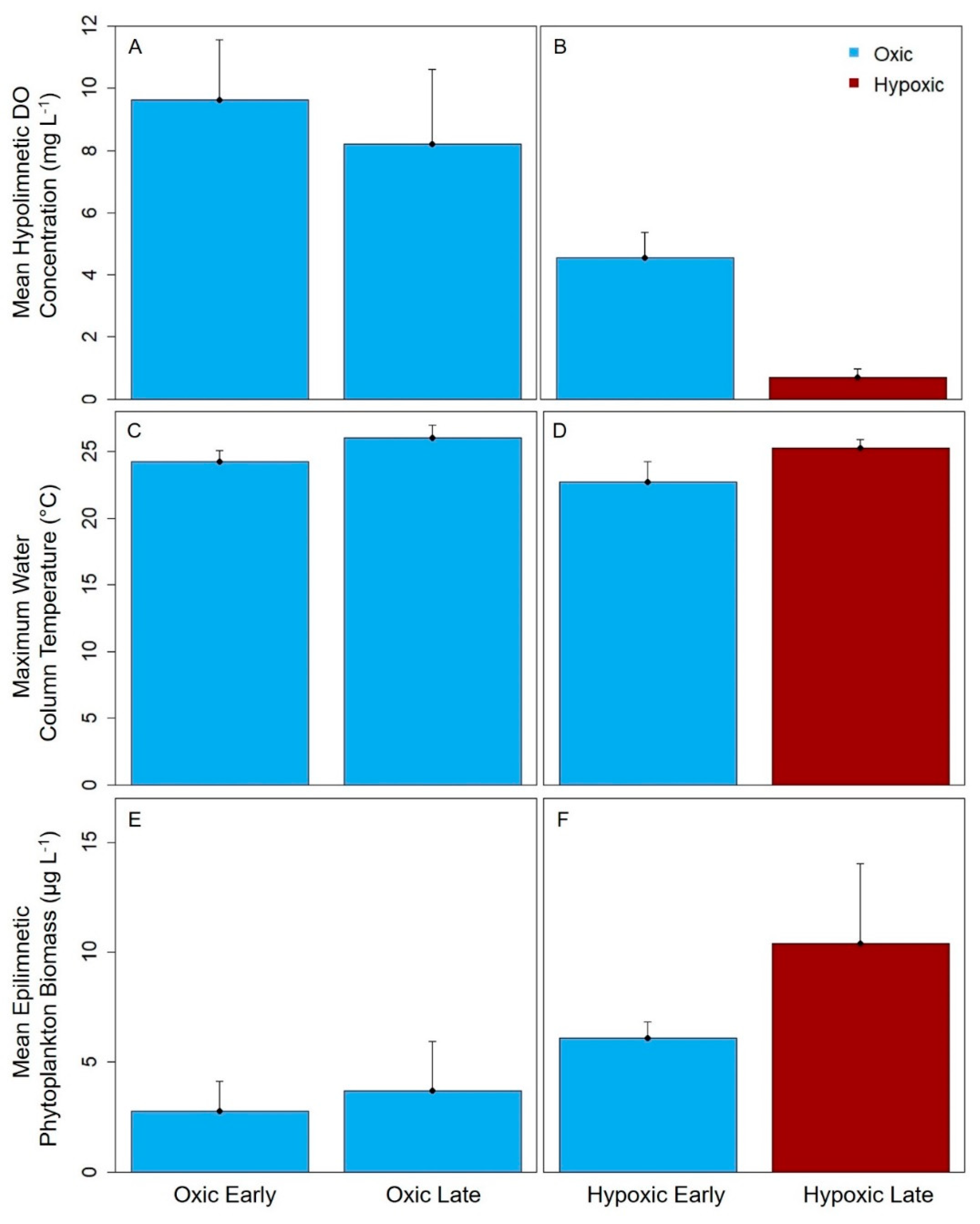
Finally, we analyzed the differences in mean hypolimnetic DO concentration, water column temperature, mean epilimnetic phytoplankton biomass, Secchi disk depth, TN concentration and TP concentration between the early vs. late stratified periods with Wilcoxon signed rank tests. These analyses were performed to assess which environmental variables other than hypolimnetic DO concentrations changed between the periods.
All analyses were performed in R v.3.5.1 (R Core Development Team, 2018). Given the small number of reservoirs in our analyses, we interpreted statistical significance at α = 0.10.
5. Discussion
Global change is predicted to create more variable environmental conditions [
1,
2,
3], which can alter community interactions and the variability of ecological communities [
4,
5,
6]. Contrary to our predictions, we found that hypolimnetic hypoxia was associated with an increased biomass variability and compositional variability of crustacean zooplankton communities in reservoirs. Our data suggest that the variability of zooplankton communities may increase with hypolimnetic hypoxia in temperate inland waterbodies.
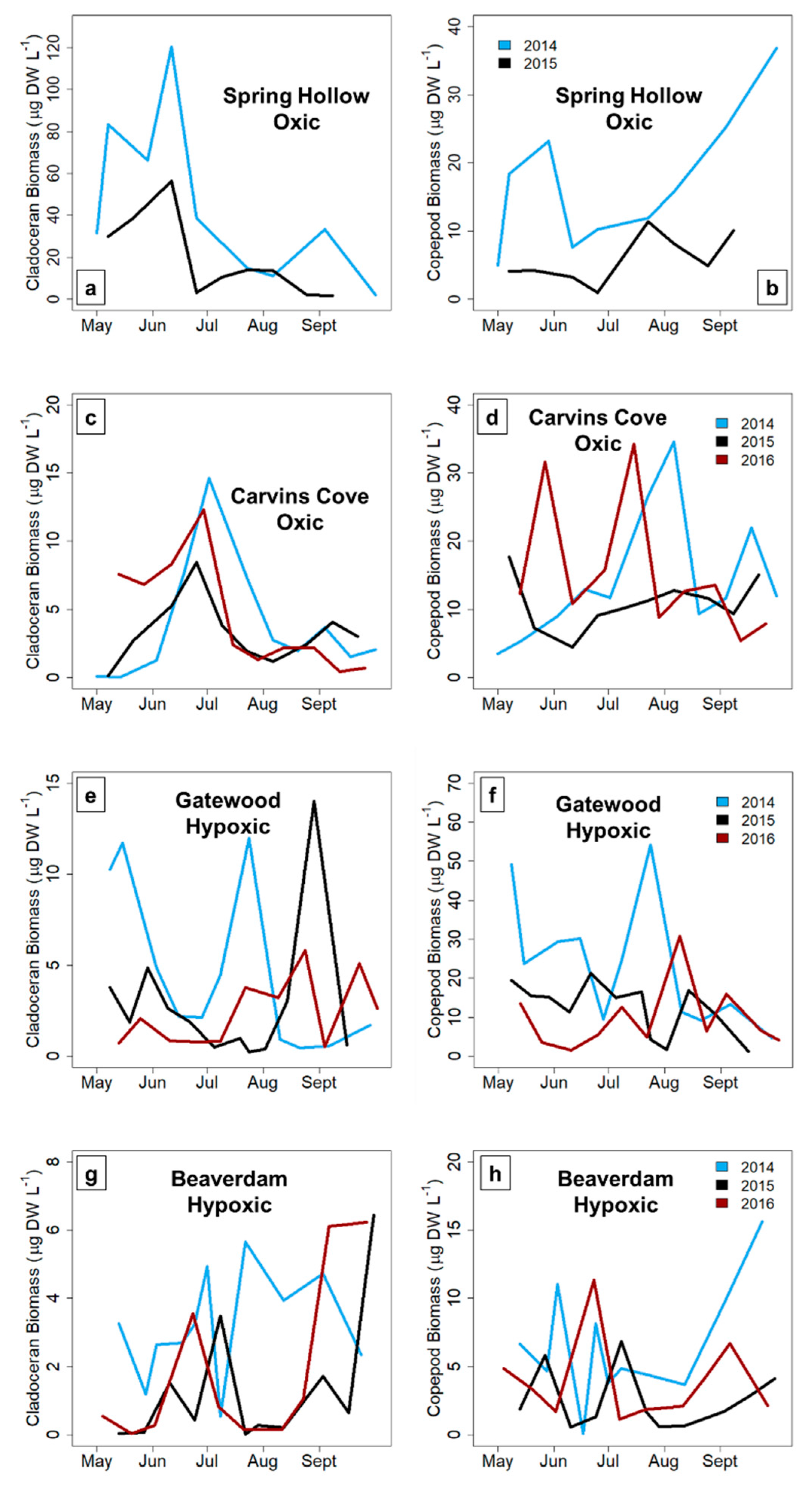
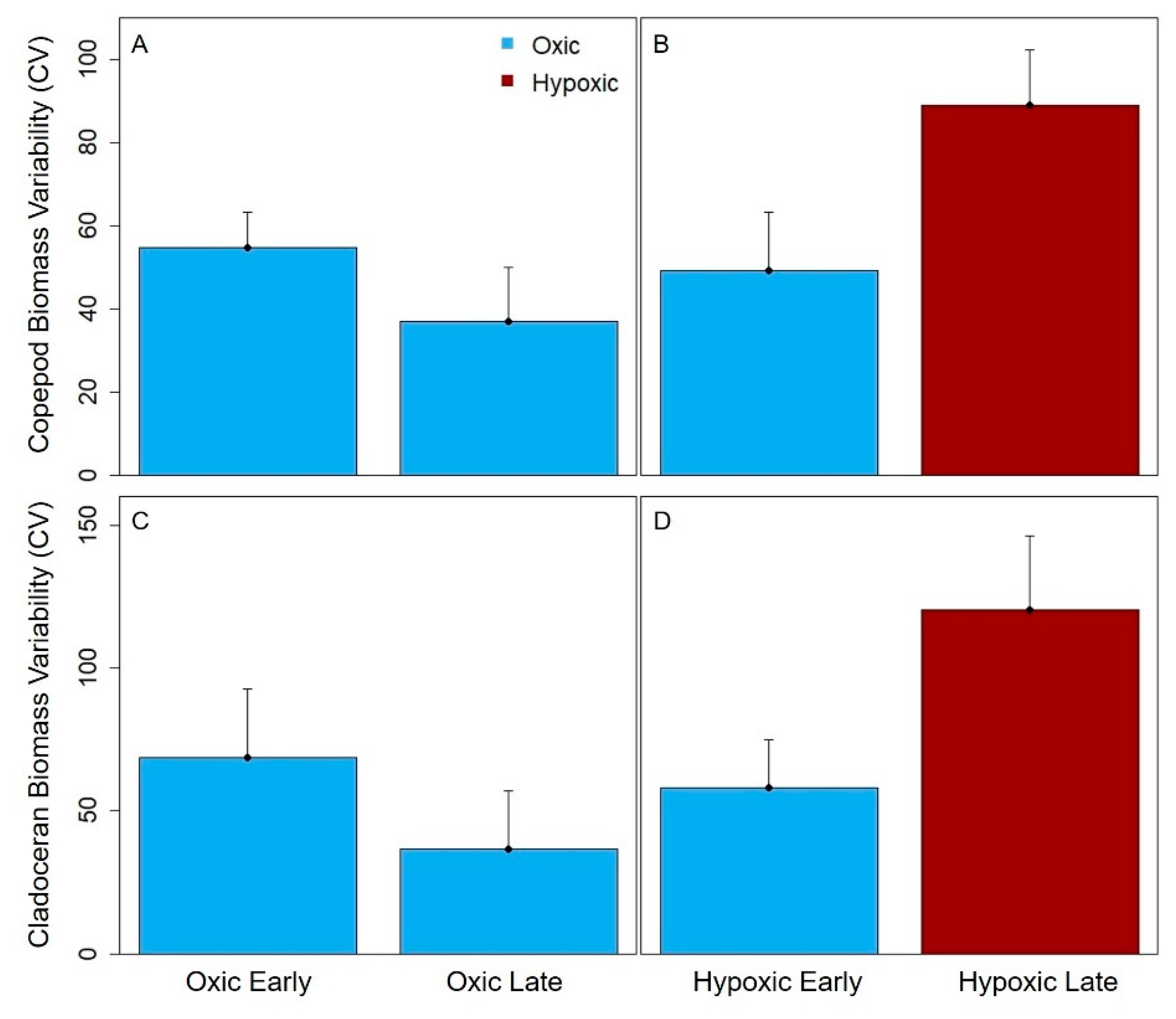
Total crustacean, cladoceran and copepod zooplankton biomass variability significantly increased from the early to late stratified period in the hypoxic reservoirs, but did not significantly change in the seasonally oxic reservoirs in the late stratified period (except for the copepods, which exhibited a decrease in biomass variability). The biomass variability increase in the hypoxic reservoirs was largely driven by copepods, which are generally the dominant group of zooplankton present in temperate waterbodies in the late stratified period [
26,
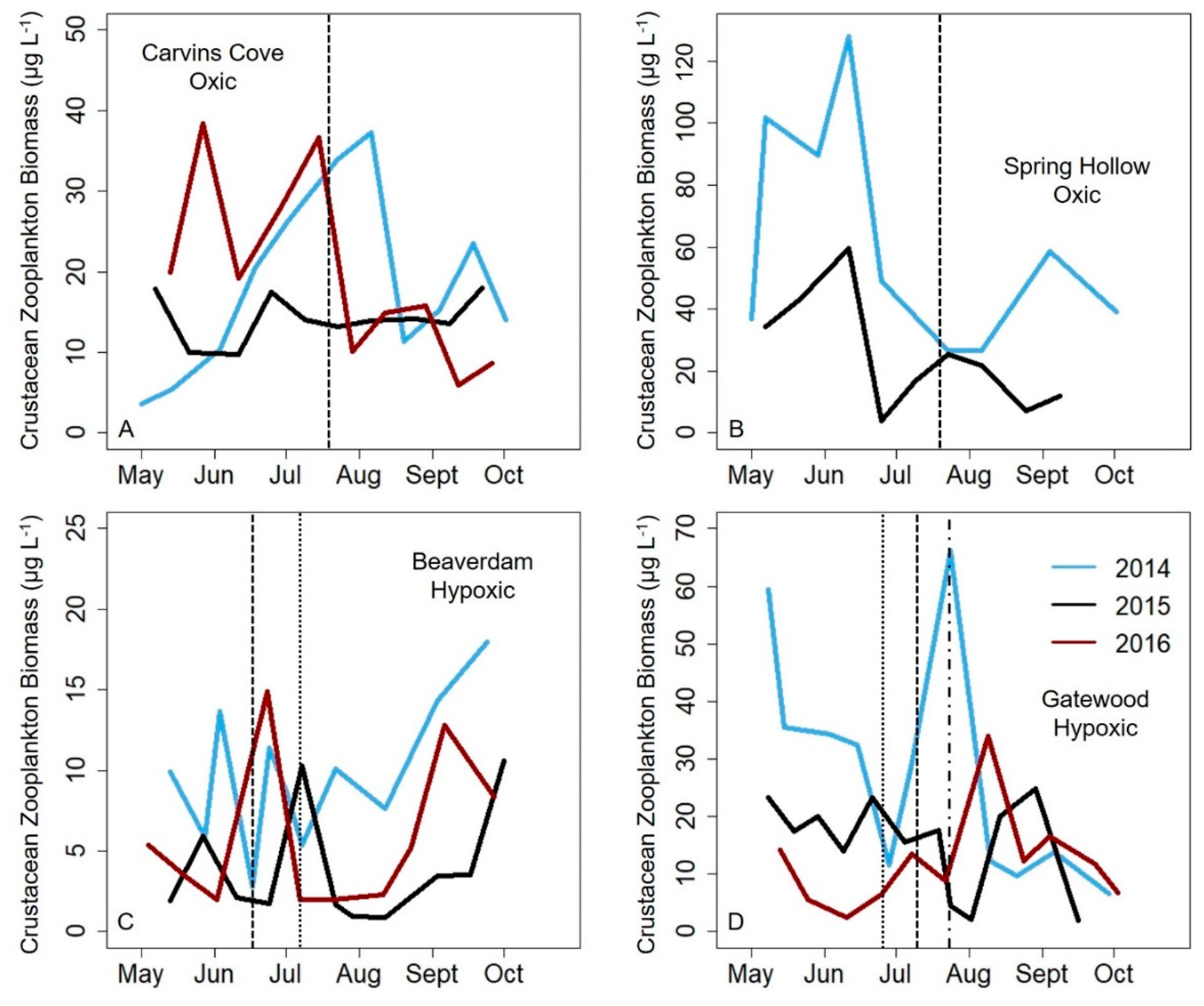
27]. In almost all reservoirs and years, a zooplankton maximum was present in the early stratified period in each reservoir (except the GWR, but it is possible that we missed the spring zooplankton maximum because the first sampling date during the three study years often exhibited higher biomass). In contrast, although a second, smaller zooplankton biomass maximum occurred in the late stratified period in the oxic CCR and SHR, this peak did not consistently occur in the hypoxic BVR and GWR reservoirs, driving the biomass variability difference. The magnitude of the biomass maximum in the BVR and GWR in the late stratified season varied by ~9× in both reservoirs among years, in comparison to an ~3× and ~2× difference in biomass among years for CCR and SHR, respectively. Therefore, our data suggest that hypolimnetic hypoxia may increase the biomass variability of zooplankton communities, resulting in a larger deviation from the typical pattern of zooplankton seasonal succession that occurs in the late stratified period in more oxic systems.
One possible mechanism for increased biomass variability under hypoxic conditions is due to diel vertical migration (DVM) of crustacean zooplankton. DVM is a ubiquitous zooplankton behavior, in which a majority of the population migrates to the dark hypolimnion during the day to escape predation from fish and damaging ultraviolet (UV) radiation [
33,
34,
57]. At nighttime, a large portion of the zooplankton population migrates back to the epilimnion, where they are closer to phytoplankton as a food resource, with a decreased risk of being eaten, or ending up damaged by UV radiation. However, because zooplankton also need oxygen for respiration [
20,
21], they may be forced out of the safety of the hypolimnion during the daytime and instead remain in the epilimnion, trading oxic stress for increased risk of predation or UV radiation damage [
35]. Subsequently, the increase in the biomass variability of the zooplankton community during hypolimnetic hypoxic periods may be related to sporadic pulses of zooplankton taxa that suddenly increase due to emergence from sediment resting stages, and then shortly thereafter exhibit population crashes due to fish predation in the epilimnion or the deleterious effects of UV radiation. Additional data are needed to support these hypotheses, which were not possible to examine within our study scope.
In the hypoxic BVR and GWR, cyclopoid copepods primarily drove the pattern of increased biomass variability in the late stratified period. Because copepods have longer generation times than cladocera and are generally conspicuous for fish in well-lit waters due to their more opaque coloration, copepods are more likely to conduct DVM, and hence may be sensitive to hypolimnetic hypoxia [
28,
58], resulting in greater variability in their biomass during hypoxic conditions. In contrast, late season copepods in the oxic reservoirs did not exhibit as many fluctuations in their biomass, resulting in a decrease in their biomass variability throughout the stratified period (
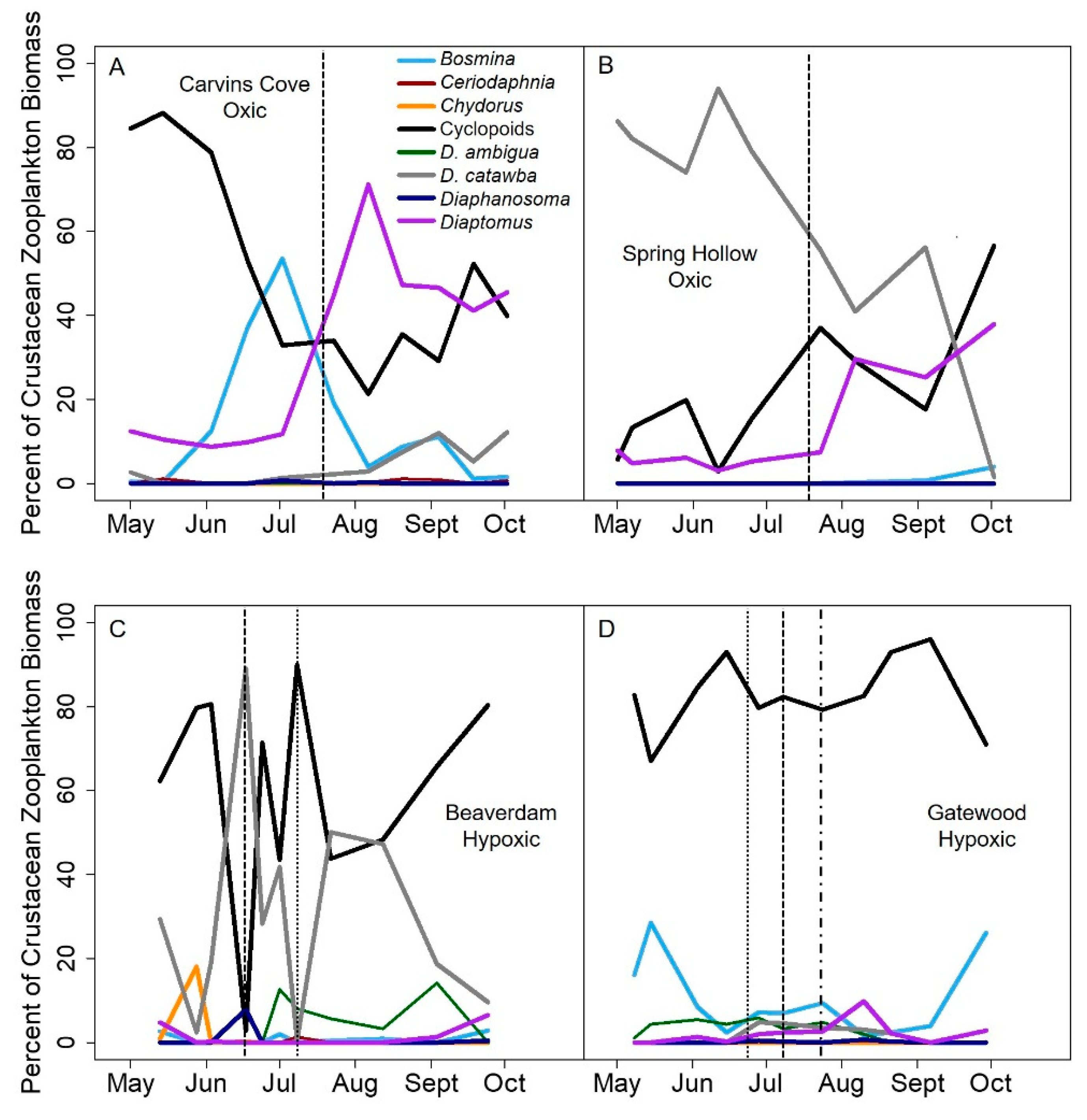
Figure 5A). While cladocera are expected to be less sensitive to hypoxia than copepods because they are more transparent and overall much smaller [
26,
59], thereby allowing them some release from visual predators (i.e., fish), we observed that the biomass variability of both taxa increased with the onset of hypoxia. Thus, our results suggest that hypoxia may affect the entire crustacean zooplankton community, though future studies are needed to directly assess the effects of hypoxia on individual zooplankton at a finer taxonomic level.
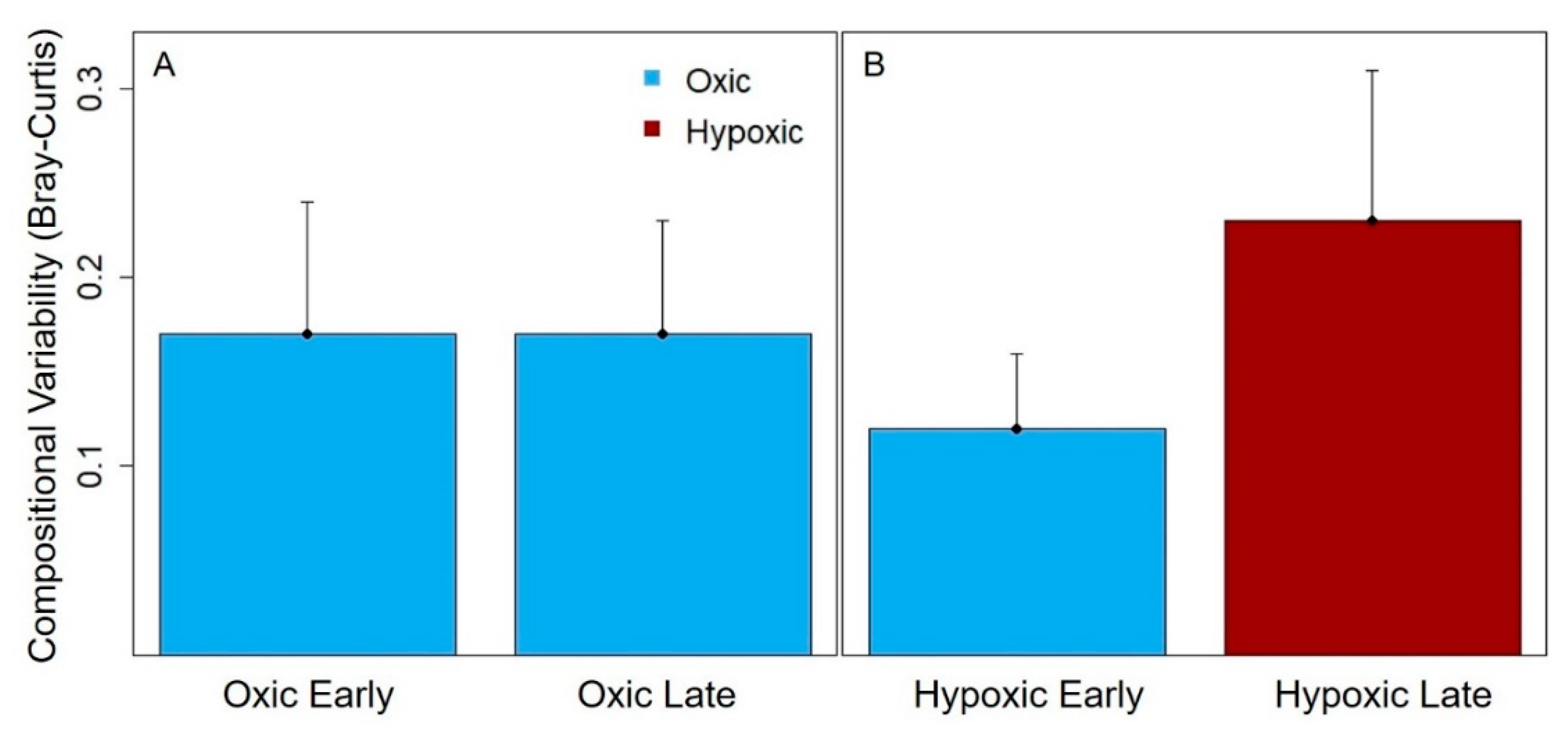
Counter to our predictions, we found that hypolimnetic hypoxia also increased the compositional variability for the dominance in crustacean zooplankton communities. The seasonally hypoxic reservoirs were more likely to be dominated by one or just a few taxa, primarily cyclopoid copepods and smaller cladocera, than the oxic reservoirs in the late stratified period (
Figure 4). Following the hypothesis above, possibly the substantial day-to-day variability in the biomass of those dominant taxa drove the greater compositional variability in the hypoxic reservoirs than oxic reservoirs in the late stratified period. However, we were not able to test the specific mechanisms underlying the compositional variability differences between hypoxic and oxic reservoirs.
Hypolimnetic hypoxia was likely the main driver of the observed changes in the biomass variability and compositional variability of the crustacean zooplankton community. The mean hypolimnetic DO concentration decreased by about 7× in the late vs. early stratified period in the seasonally hypoxic reservoirs. In contrast, the mean hypolimnetic DO changed only slightly in the early vs. late stratified periods of the seasonally oxic reservoirs, which is likely why there were fewer differences in the variability of the zooplankton communities in these reservoirs. Changes in the other environmental variables between the early and late stratified periods are likely related to DO concentrations. For example, the significant increases in TN concentrations and phytoplankton biomass between the early and late stratified periods in the BVR and GWR may be related to the release of nutrients from the sediments to the water column that occurs under hypoxic conditions [
60,
61,
62] and the subsequent stimulation of primary productivity.
Because crustacean zooplankton feed on phytoplankton [
24,
42,
63], increased phytoplankton biomass in the hypoxic reservoirs throughout the summer may have also amplified the biomass variability and compositional zooplankton variability differences between reservoirs. An increase in the Secchi disk depth and decrease in TP concentrations also existed in the seasonally oxic reservoirs from the early to late stratified period (but not in the hypoxic reservoirs), which may have also played a factor in the variability differences. Altogether, our data support [
64]’s paradox of enrichment, in that greater food availability (phytoplankton biomass) results in greater variability of the predator (zooplankton) populations. While we are unable to model phytoplankton –zooplankton populations given the coarse temporal resolution of our sampling relative to phytoplankton generation times, our study adds field survey data to previous microcosm [
65] and paleolimnological [
12] studies that observed similar variability patterns.
A limitation of our work was that we were unable to account for some other environmental factors that may also affect the variability in zooplankton communities. For example, there may have been differences in fish grazing rates [
66,
67], disease transmission [
68], diel horizontal migration [
69,
70], or interactions among multiple variables that may have also affected zooplankton community variability. Also, the demarcation of the early vs. late stratified period inherently created non-independence between zooplankton samples and the other environmental variables across the season that we were not able to fully account for. Finally, the patterns of biomass variability and compositional variability were dependent upon the taxa present in our study systems; the results might be different in other lakes and reservoirs with different zooplankton assemblages.
Our work suggests that increases in hypolimnetic hypoxia intensity and duration may alter the biomass variability and compositional variability of zooplankton, thereby altering the seasonal succession dynamics of the zooplankton communities. The alteration of zooplankton communities could also lead to changes in the populations in other trophic levels (phytoplankton and fish), which has implications for water quality. Therefore, it is critically important to better understand the variability of ecological communities in waterbodies, and what the resulting consequences may be for ecosystem functioning and processes.