Electrospun Nylon 6,6/ZIF-8 Nanofiber Membrane for Produced Water Filtration
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of ZIF-8
2.3. Electrospinning of Nylon 6,6/ZIF-8 NFM
2.4. Membrane and Additive Characterization
2.5. Permeability Analysis
2.6. Rejection Analysis
3. Results and Discussion
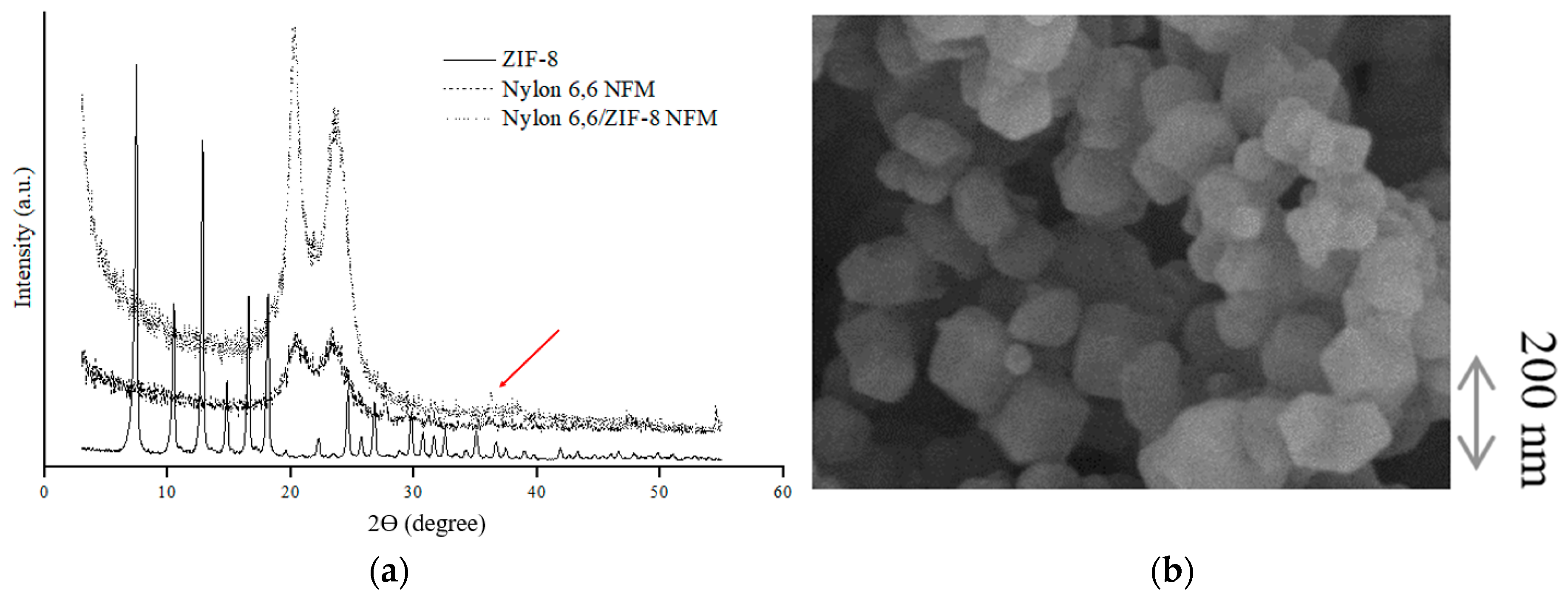
3.1. ZIF-8 Characterization
3.2. Membrane Crystallization
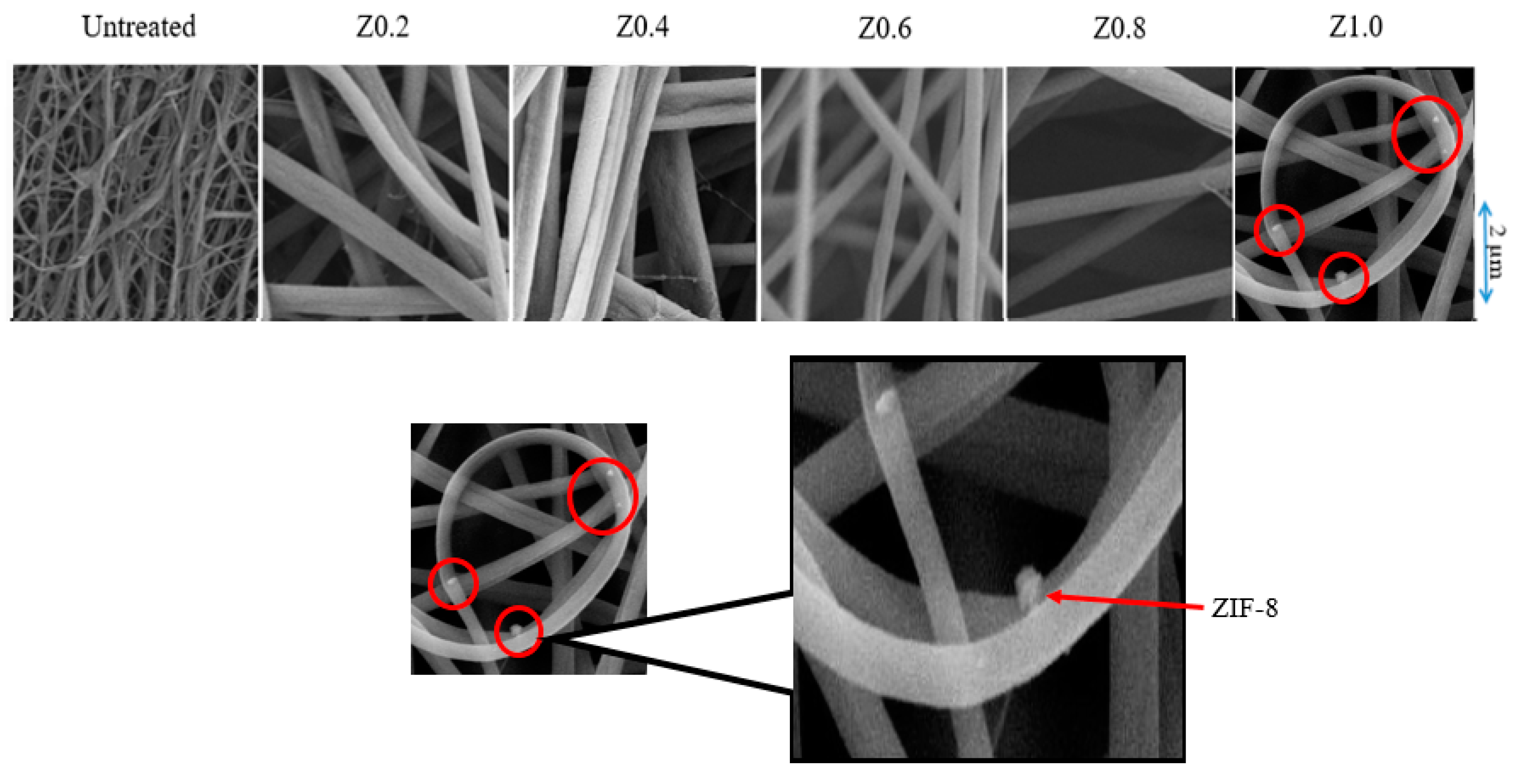
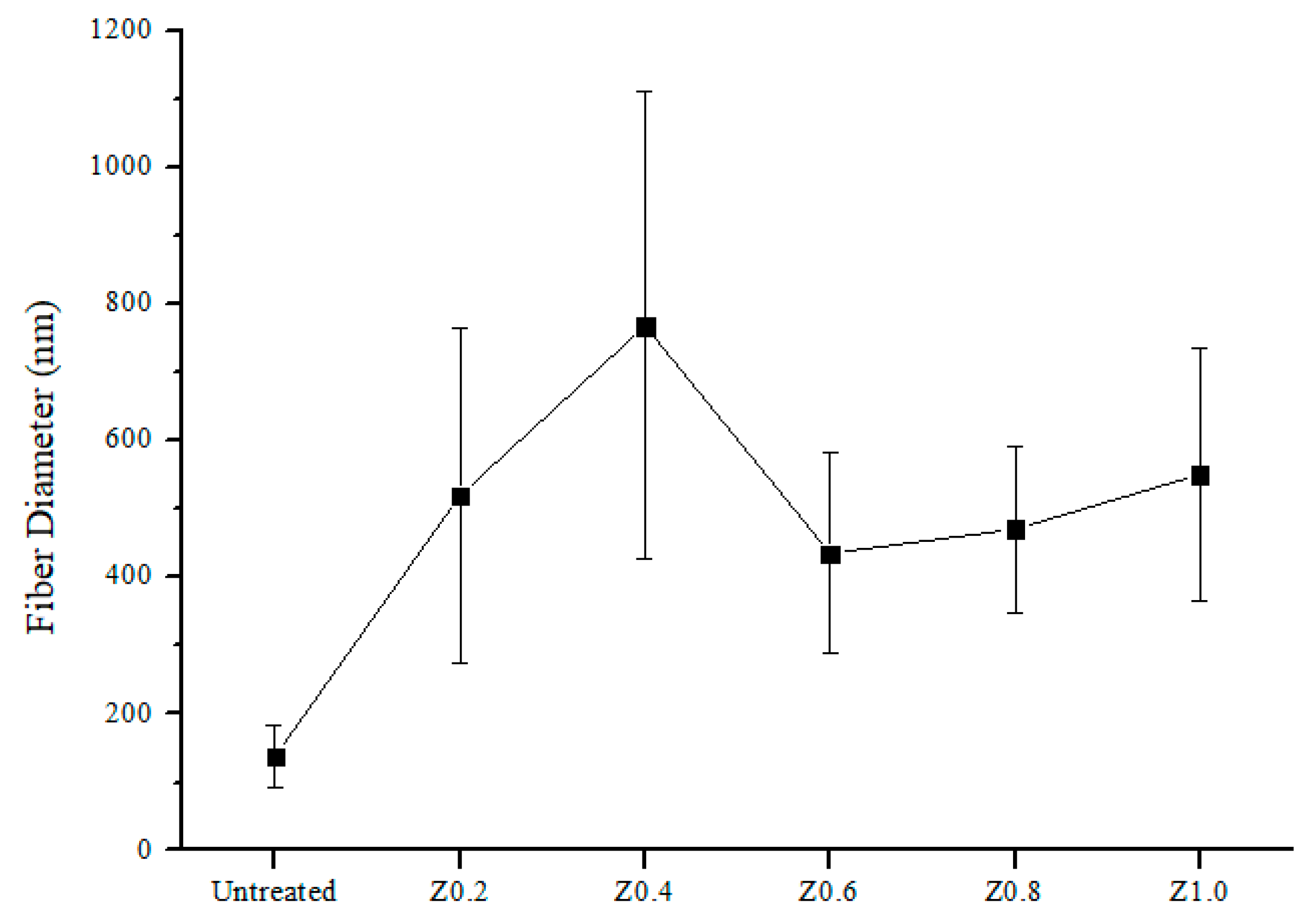
3.3. Surface Morphology and Fiber Diameter of Nylon 6,6/ZIF-8 NFM
3.4. Membrane Porosity and Surface Roughness
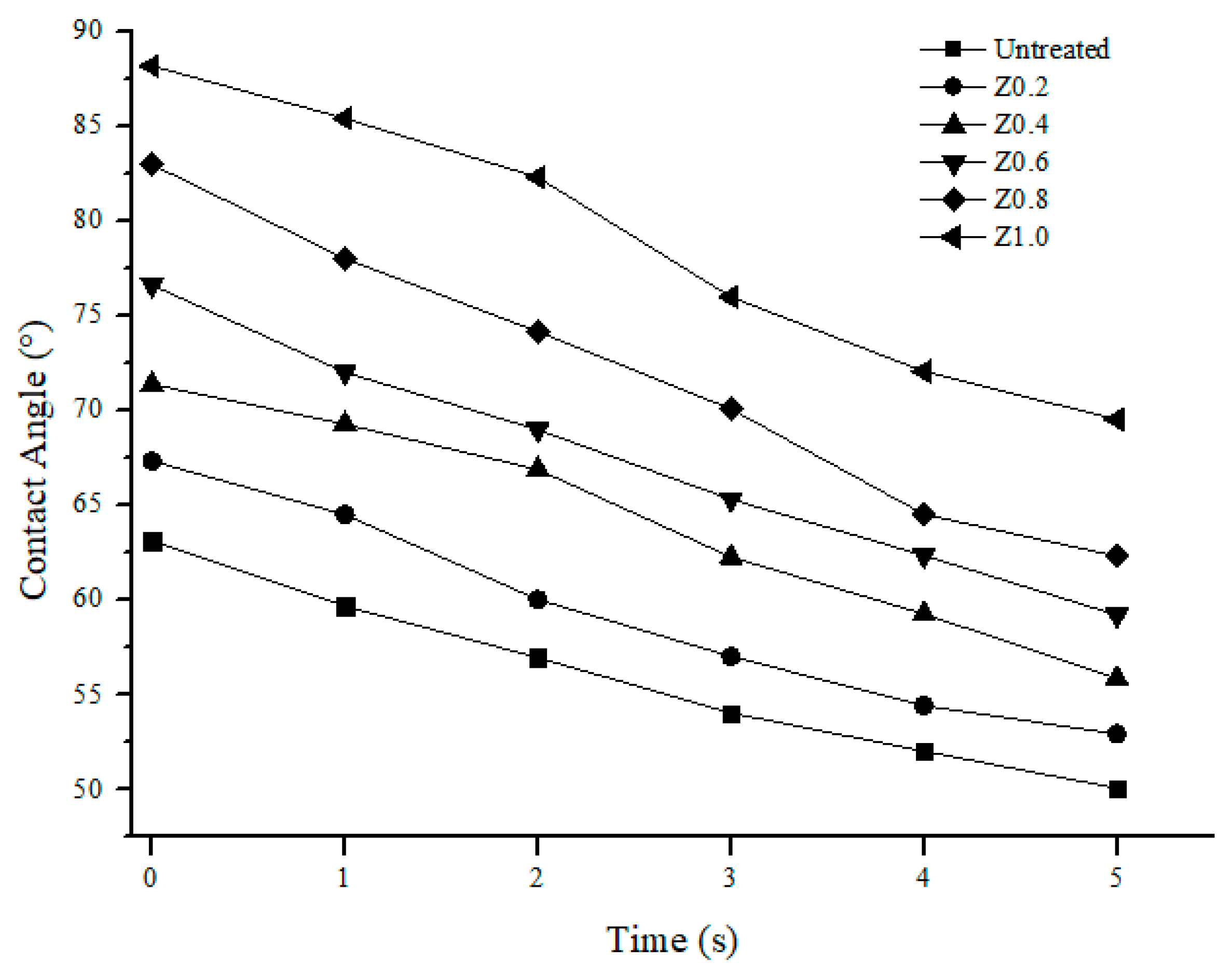
3.5. Contact Angle
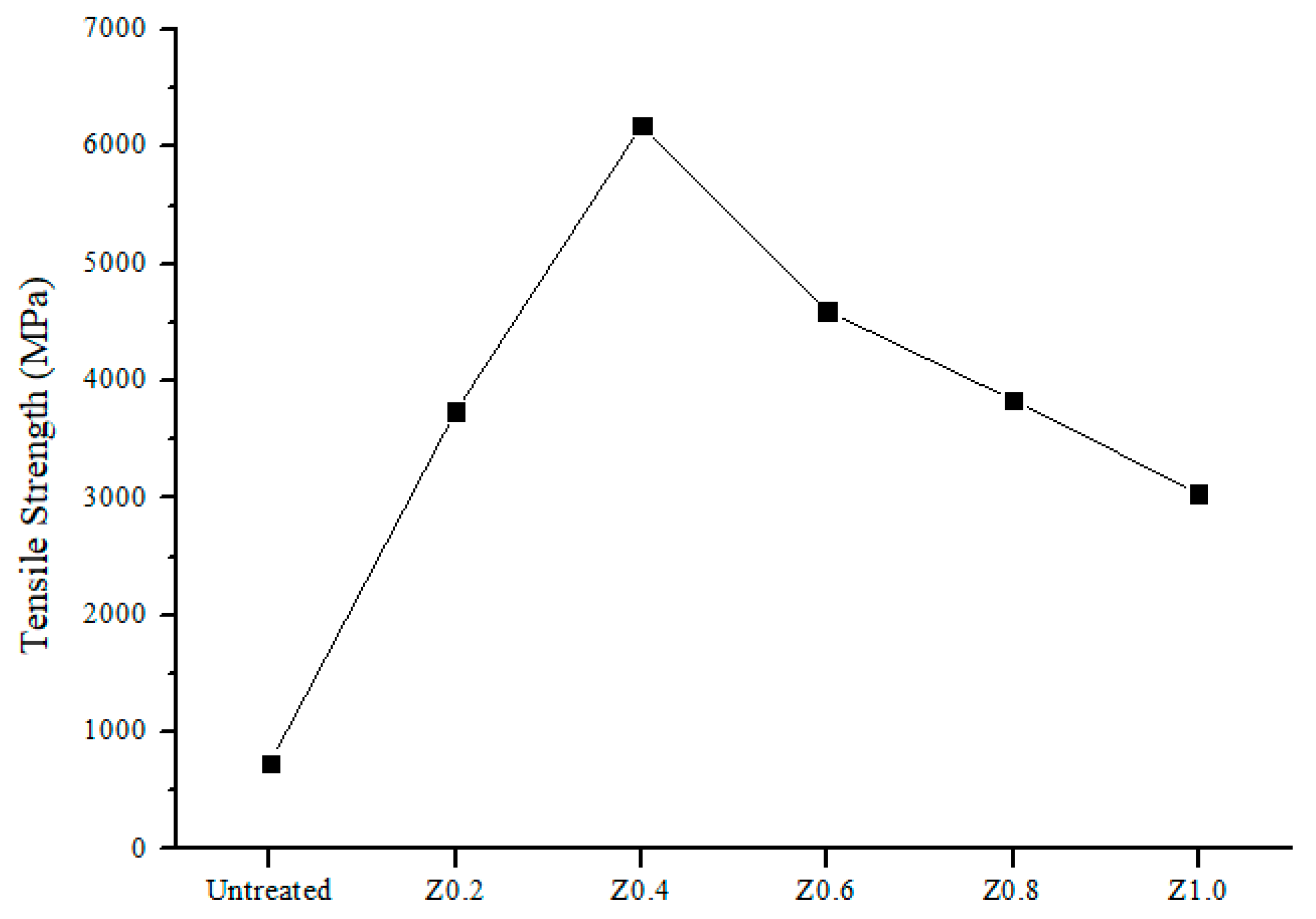
3.6. Mechanical Properties of Nylon 6,6/ZIF-8 NFM
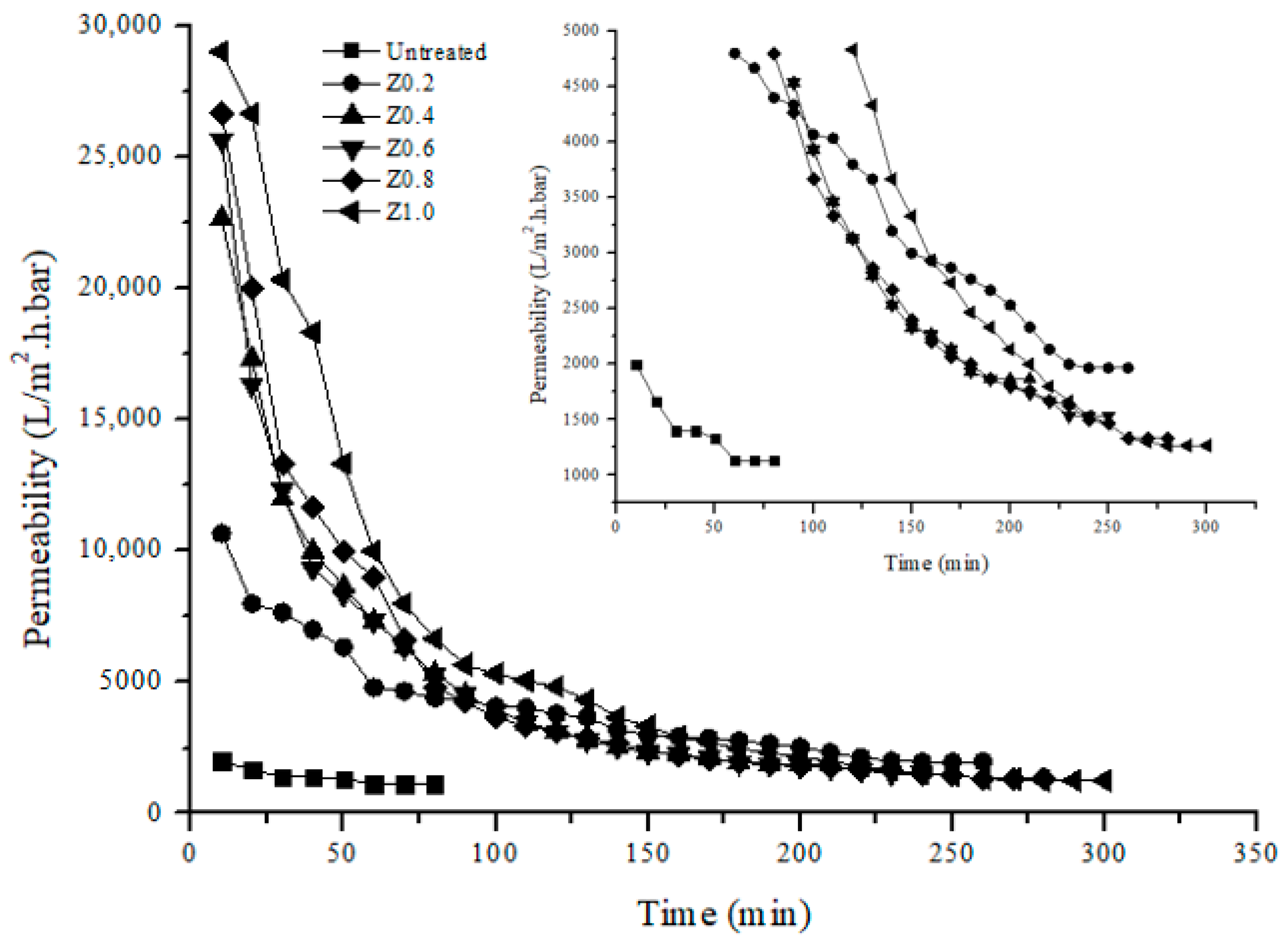
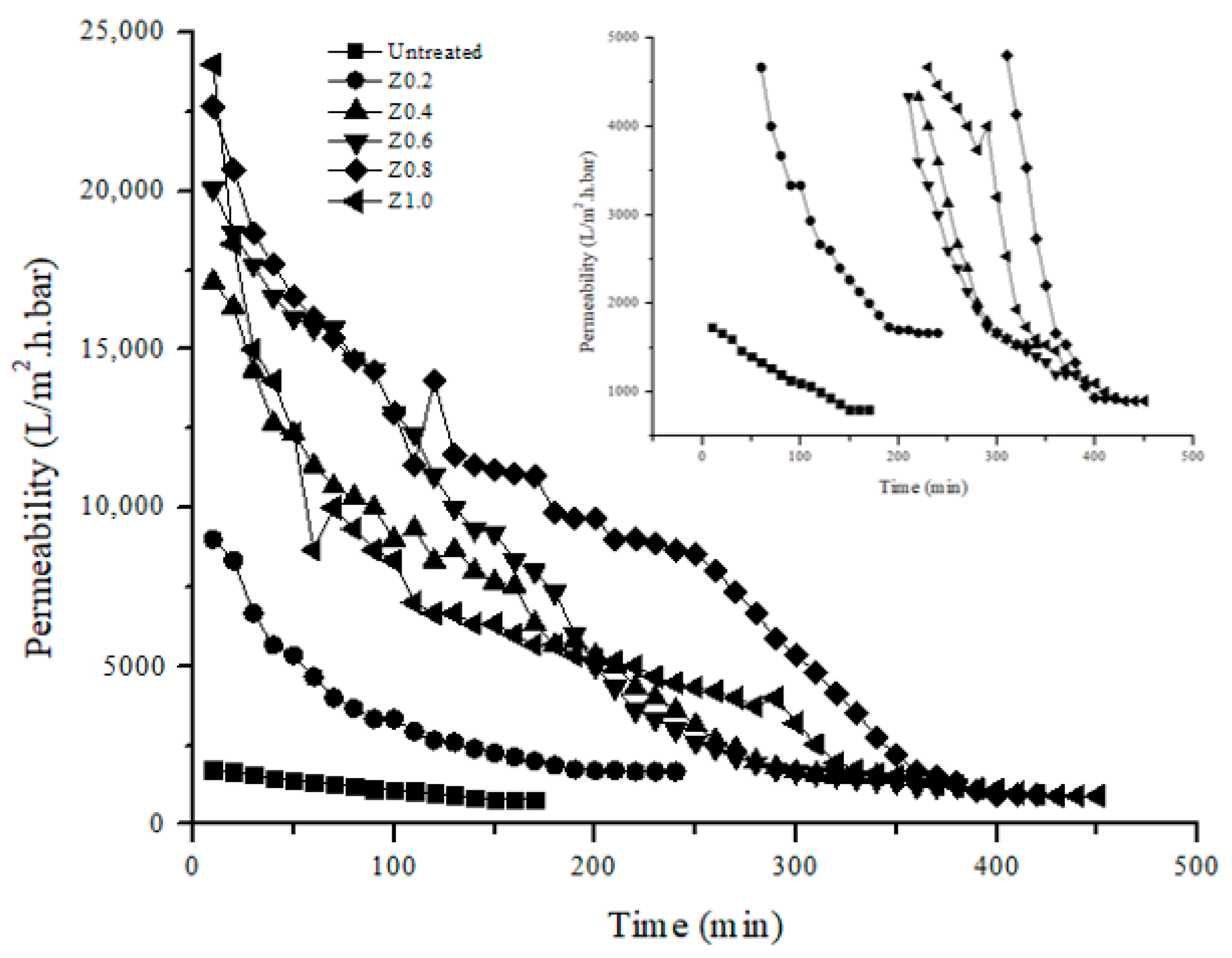
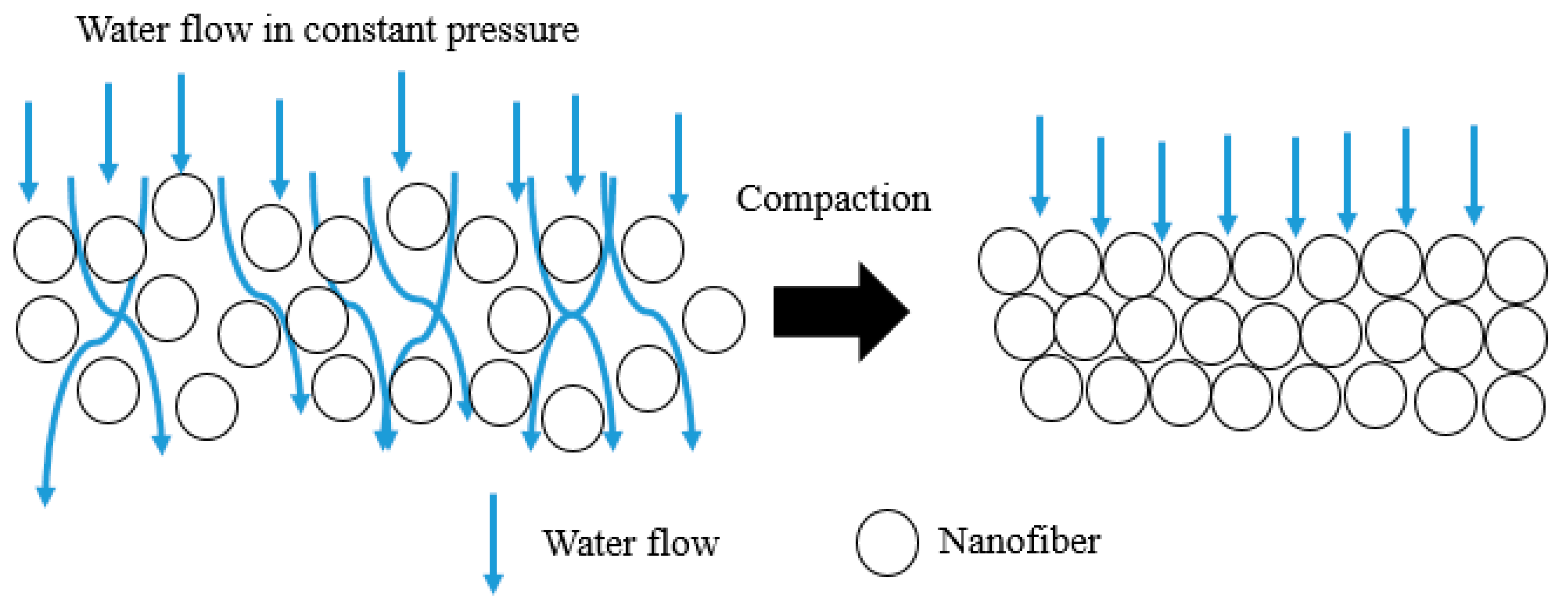
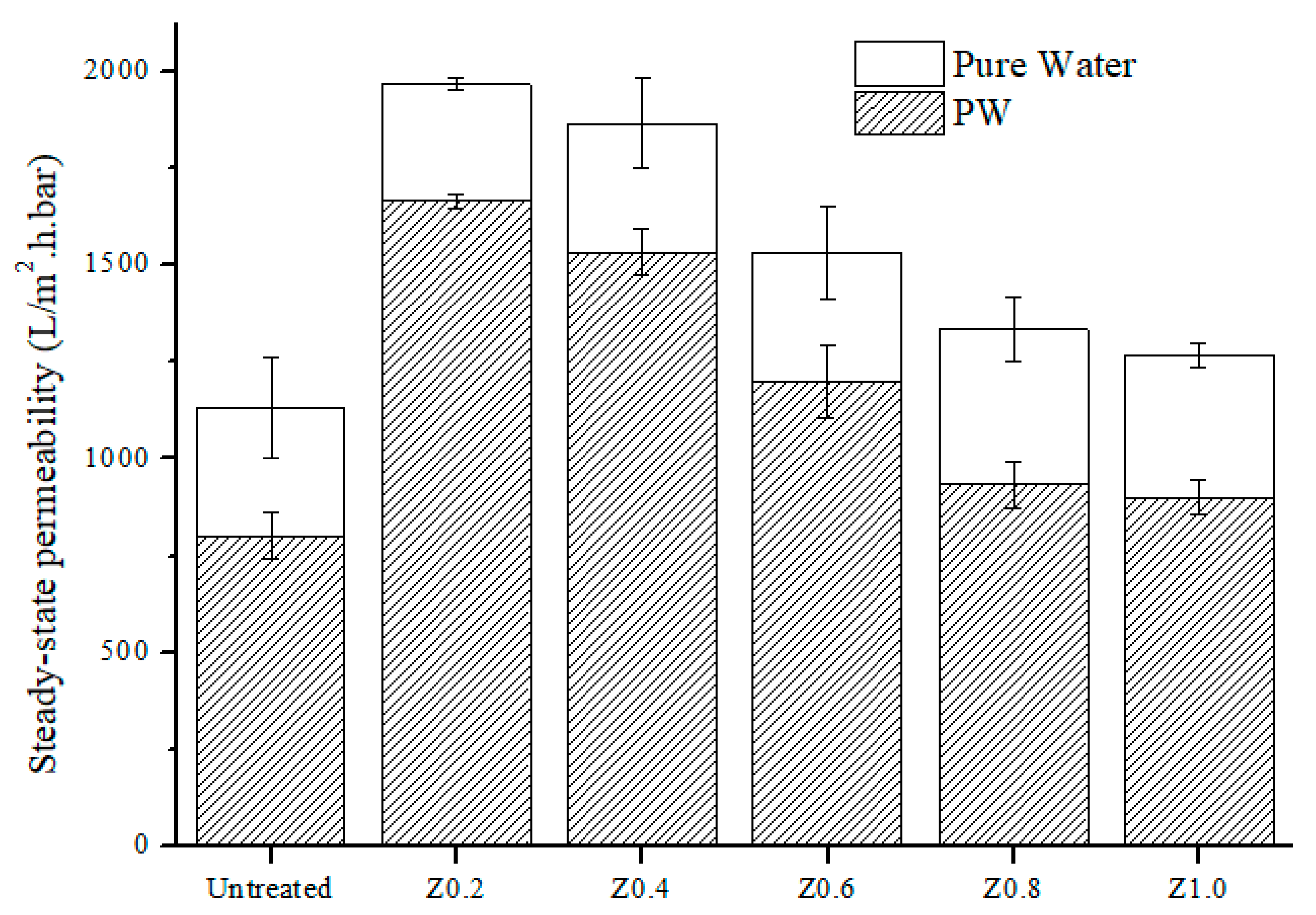
3.7. Permeability Analysis
3.8. Rejection Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bakke, T.; Klungsøyr, J.; Sanni, S. Environmental impacts of produced water and drilling waste discharges from the Norwegian offshore petroleum industry. Mar. Environ. Res. 2013, 92, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Soto, L.V.; Botello, A.; Licea, S.; Lizárraga-Partida, M.; Yáñez-Arancibia, A. The environmental legacy of the Ixtoc-I oil spill in Campeche Sound, southwestern Gulf of Mexico. Front. Mar. Sci. 2014, 1, 1–9. [Google Scholar] [CrossRef]
- Zhang, C.; Li, P.; Cao, B. Electrospun Microfibrous Membranes Based on PIM-1/POSS with High Oil Wettability for Separation of Oil–Water Mixtures and Cleanup of Oil Soluble Contaminants. Ind. Eng. Chem. Res. 2015, 54, 8772–8781. [Google Scholar] [CrossRef]
- Su, Y.; Zhao, Q.; Liu, J.; Zhao, J.; Li, Y.; Jiang, Z. Improved oil/water emulsion separation performance of PVC/CPVC blend ultrafiltration membranes by fluorination treatment. Desalin. Water Treat. 2015, 55, 304–314. [Google Scholar] [CrossRef]
- Ibrahim, N.A.; Wirzal, M.D.H.; Nordin, N.A.H.; Halim, N.S.A. Development of Polyvinylidene fluoride (PVDF)-ZIF-8 Membrane for Wastewater Treatment. IOP Conf. Ser. Earth Environ. Sci. 2018, 140, 012021. [Google Scholar] [CrossRef]
- Benito, A.; Garcia, G.; Gonzalez-Olmos, R. Fouling reduction by UV-based pretreatment in hollow fiber ultrafiltration membrane for urban wastewater reuse. J. Membr. Sci. 2017, 536, 141–147. [Google Scholar] [CrossRef]
- Eliseus, A.; Bilad, M.R.; Nordin, N.A.H.M.; Putra, Z.A.; Wirzal, M.D.H. Tilted membrane panel: A new module concept to maximize the impact of air bubbles for membrane fouling control in microalgae harvesting. Bioresour. Technol. 2017, 241, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Jepsen, K.L.; Bram, M.V.; Pedersen, S.; Yang, Z. Membrane Fouling for Produced Water Treatment: A Review Study from a Process Control Perspective. Water 2018, 10, 847. [Google Scholar] [CrossRef]
- Bilad, M.R.; Westbroek, P.; Vankelecom, I.F.J. Assessment and optimization of electrospun nanofiber-membranes in a membrane bioreactor (MBR). J. Membr. Sci. 2011, 380, 181–191. [Google Scholar] [CrossRef]
- Jiříček, T.; Komárek, M.; Lederer, T. Polyurethane Nanofiber Membranes for Waste Water Treatment by Membrane Distillation. J. Nanotechnol. 2017, 2017. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Elbahri, M. An Amphiphilic, Graphitic Buckypaper Capturing Enzyme Biomolecules from Water. Water 2019, 11, 2. [Google Scholar] [CrossRef]
- Liu, C.; Li, X.; Liu, T.; Liu, Z.; Li, N.; Zhang, Y.; Xiao, C.; Feng, X. Microporous CA/PVDF membranes based on electrospun nanofibers with controlled crosslinking induced by solvent vapor. J. Membr. Sci. 2016, 512, 1–12. [Google Scholar] [CrossRef]
- Ahmadi, A.; Qanati, O.; Seyed Dorraji, M.S.; Rasoulifard, M.H.; Vatanpour, V. Investigation of antifouling performance a novel nanofibrous S-PVDF/PVDF and S-PVDF/PVDF/GO membranes against negatively charged oily foulants. J. Membr. Sci. 2017, 536, 86–97. [Google Scholar] [CrossRef]
- Huang, L.; Manickam, S.S.; McCutcheon, J.R. Increasing strength of electrospun nanofiber membranes for water filtration using solvent vapor. J. Membr. Sci. 2013, 436, 213–220. [Google Scholar] [CrossRef]
- Islam, M.S.; McCutcheon, J.R.; Rahaman, M.S. A high flux polyvinyl acetate-coated electrospun nylon 6/SiO2 composite microfiltration membrane for the separation of oil-in-water emulsion with improved antifouling performance. J. Membr. Sci. 2017, 537, 297–309. [Google Scholar] [CrossRef]
- Naseeb, N.; Mohammed, A.A.; Laoui, T.; Khan, Z. A Novel PAN-GO-SiO2 Hybrid Membrane for Separating Oil and Water from Emulsified Mixture. Materials 2019, 12, 212. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wang, Z.; Zhang, J. Zeolite imidazolate framework hybrid nanofiltration (NF) membranes with enhanced permselectivity for dye removal. J. Membr. Sci. 2017, 532, 76–86. [Google Scholar] [CrossRef]
- Basu, S.; Balakrishnan, M. Polyamide thin film composite membranes containing ZIF-8 for the separation of pharmaceutical compounds from aqueous streams. Sep. Purif. Technol. 2017, 179, 118–125. [Google Scholar] [CrossRef]
- Nordin, N.A.H.M.; Racha, S.M.; Matsuura, T.; Misdan, N.; Sani, N.A.A.; Ismail, A.F.; Mustafa, A. Facile modification of ZIF-8 mixed matrix membrane for CO2/CH4 separation: Synthesis and preparation. RSC Adv. 2015, 5, 43110–43120. [Google Scholar] [CrossRef]
- Nordin, N.A.H.M.; Ismail, A.F.; Mustafa, A. Synthesis and Preparation of Asymmetric PSf/ZIF-8 Mixed Matrix Membrane for CO2/CH4 Separation. J. Teknologi 2014, 69, 9. [Google Scholar] [CrossRef]
- Nordin, N.a.H.M.; Ismail, A.F.; Mustafa, A.; Murali, R.S.; Matsuura, T. The impact of ZIF-8 particle size and heat treatment on CO2/CH4 separation using asymmetric mixed matrix membrane. RSC Adv. 2014, 4, 52530–52541. [Google Scholar] [CrossRef]
- Hadi, A.; Karimi-Sabet, J.; Dastbaz, A. Parametric study on the mixed solvent synthesis of ZIF-8 nano- and micro-particles for CO adsorption: A response surface study. Front. Chem. Sci. Eng. 2019, 1–16. [Google Scholar] [CrossRef]
- Dai, X.; Cao, Y.; Shi, X.; Wang, X. The PLA/ZIF-8 Nanocomposite Membranes: The Diameter and Surface Roughness Adjustment by ZIF-8 Nanoparticles, High Wettability, Improved Mechanical Property, and Efficient Oil/Water Separation. Adv. Mater. Interfaces 2016, 3, 1–6. [Google Scholar] [CrossRef]
- Bilad, M.R.; Azizo, A.S.; Wirzal, M.D.H.; Jia Jia, L.; Putra, Z.A.; Nordin, N.A.H.M.; Mavukkandy, M.O.; Jasni, M.J.F.; Yusoff, A.R.M. Tackling membrane fouling in microalgae filtration using nylon 6,6 nanofiber membrane. J. Environ. Manag. 2018, 223, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Jian, M.; Liu, B.; Zhang, G.; Liu, R.; Zhang, X. Adsorptive removal of arsenic from aqueous solution by zeolitic imidazolate framework-8 (ZIF-8) nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2015, 465, 67–76. [Google Scholar] [CrossRef]
- Schejn, A.; Balan, L.; Falk, V.; Aranda, L.; Medjahdi, G.; Schneider, R. Controlling ZIF-8 nano- and microcrystal formation and reactivity through zinc salt variations. CrystEngComm 2014, 16, 4493–4500. [Google Scholar] [CrossRef]
- Araujo, E.; Leite, A.M.D.; Medeiros, V.D.N.; Paz, R.A.; Lira, H.D.L. Comparative Study of Membranes Obtained from PA6 and PA66/National Clay Nanocomposites. Adv. Nanocomposite Technol. 2011. [Google Scholar] [CrossRef][Green Version]
- Zhong, Z.; Cao, Q.; Wang, X.; Wu, N.; Wang, Y. PVC–PMMA composite electrospun membranes as polymer electrolytes for polymer lithium-ion batteries. Ionics 2012, 18, 47–53. [Google Scholar] [CrossRef]
- Rnjak-Kovacina, J.; Weiss, A.S. Increasing the pore size of electrospun scaffolds. Tissue Eng. Part B Rev. 2011, 17, 365–372. [Google Scholar] [CrossRef]
- Širc, J.; Hobzová, R.; Kostina, N.; Munzarová, M.; Juklíčková, M.; Lhotka, M.; Kubinová, Š.; Zajícová, A.; Michálek, J. Morphological Characterization of Nanofibers: Methods and Application in Practice. J. Nanomater. 2012. [Google Scholar] [CrossRef]
- Pillay, V.; Dott, C.; Choonara, Y.E.; Tyagi, C.; Tomar, L.; Kumar, P.; du Toit, L.C.; Ndesendo, V.M.K. A Review of the Effect of Processing Variables on the Fabrication of Electrospun Nanofibers for Drug Delivery Applications. J. Nanomater. 2013. [Google Scholar] [CrossRef]
- Ortiz, A.U.; Freitas, A.P.; Boutin, A.; Fuchs, A.H.; Coudert, F.-X. What makes zeolitic imidazolateframeworks hydrophobic or hydrophilic? The impact of geometry and functionalization on water adsorption. Phys. Chem. Chem. Phys. 2014, 16, 9940–9949. [Google Scholar] [CrossRef] [PubMed]
- Nosonovsky, M.; Bhushan, B. Wetting of rough three-dimensional superhydrophobic surfaces. Microsyst. Technol. 2006, 12, 273–281. [Google Scholar] [CrossRef]
- Reshmi, C.R.; Suja, P.S.; Juraij, A.; Athiyanathil, S. Fabrication of superhydrophobic polycaprolactone/beeswax electrospun membranes for high-efficiency oil/water separation. RSC Adv. 2017, 7, 2092–2102. [Google Scholar]
- Gholami, M.; Mohammadi, T.; Mosleh, S.; Hemmati, M. CO2/CH4 separation using mixed matrix membrane-based polyurethane incorporated with ZIF-8 nanoparticles. Chem. Pap. 2017, 71, 1839–1853. [Google Scholar] [CrossRef]
- Fu, S.-Y.; Feng, X.-Q.; Lauke, B.; Mai, Y.-W. Effects of particle size, particle/matrix interface adhesion and particle loading on mechanical properties of particulate–polymer composites. Compos. Part B Eng. 2008, 39, 933–961. [Google Scholar] [CrossRef]
- Metın, D.; Tihminlioğlu, F.; Balköse, D.; Ülkü, S. The effect of interfacial interactions on the mechanical properties of polypropylene/natural zeolite composites. Compos. Part A Appl. Sci. Manuf. 2004, 35, 23–32. [Google Scholar] [CrossRef]
- Wang, Y.; Górecki, R.P.; Stamate, E.; Norrman, K.; Aili, D.; Zuo, M.; Guo, W.; Hélix-Nielsen, C.; Zhang, W. Preparation of super-hydrophilic polyphenylsulfone nanofiber membranes for water treatment. RSC Adv. 2018, 9, 278–286. [Google Scholar] [CrossRef]
- Lubasova, D.; Mullerova, J.; Netravali, A.N. Water-resistant plant protein-based nanofiber membranes. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]


| ZIF-8 Loading (%) | Sample Name | Thickness (mm) | Porosity (%) | Mean Pore Size (µm) | Surface Roughness (nm) |
|---|---|---|---|---|---|
| 0.00 | Untreated | 0.22 ± 0.08 | 70.00 ± 0.50 | 0.20 | 231.10 |
| 0.20 | Z0.2 | 0.19 ± 0.02 | 72.00 ± 1.50 | 0.86 | 270.00 |
| 0.40 | Z0.4 | 0.22 ± 0.05 | 76.00 ± 1.00 | 2.56 | 384.00 |
| 0.60 | Z0.6 | 0.20 ± 0.04 | 78.00 ± 0.50 | 3.45 | 569.00 |
| 0.80 | Z0.8 | 0.19 ± 0.03 | 78.00 ± 2.00 | 3.65 | 681.00 |
| 1.00 | Z1.0 | 0.18 ± 0.03 | 79.00 ± 1.00 | 2.51 | 758.00 |
| Sample Name | Turbidity (NTU) | TOC (ppm) | Oil Conc. (ppm) | Rejection of Oil (%) |
|---|---|---|---|---|
| PW (Feed) | 33.50 | 583.00 | 88.43 | - |
| Untreated | 1.88 | 572.60 | 4.93 | 94.40 |
| Z0.2 | 0.67 | 573.80 | 9.18 | 89.00 |
| Z0.4 | 0.67 | 570.80 | 10.68 | 88.00 |
| Z0.6 | 0.65 | 572.00 | 12.43 | 86.00 |
| Z0.8 | 0.61 | 569.10 | 14.43 | 84.00 |
| Z1.0 | 0.56 | 575.80 | 16.18 | 82.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd Halim, N.S.; Wirzal, M.D.H.; Bilad, M.R.; Md Nordin, N.A.H.; Adi Putra, Z.; Mohd Yusoff, A.R.; Narkkun, T.; Faungnawakij, K. Electrospun Nylon 6,6/ZIF-8 Nanofiber Membrane for Produced Water Filtration. Water 2019, 11, 2111. https://doi.org/10.3390/w11102111
Abd Halim NS, Wirzal MDH, Bilad MR, Md Nordin NAH, Adi Putra Z, Mohd Yusoff AR, Narkkun T, Faungnawakij K. Electrospun Nylon 6,6/ZIF-8 Nanofiber Membrane for Produced Water Filtration. Water. 2019; 11(10):2111. https://doi.org/10.3390/w11102111
Chicago/Turabian StyleAbd Halim, Nur Syakinah, Mohd Dzul Hakim Wirzal, Muhammad Roil Bilad, Nik Abdul Hadi Md Nordin, Zulfan Adi Putra, Abdull Rahim Mohd Yusoff, Thanitporn Narkkun, and Kajornsak Faungnawakij. 2019. "Electrospun Nylon 6,6/ZIF-8 Nanofiber Membrane for Produced Water Filtration" Water 11, no. 10: 2111. https://doi.org/10.3390/w11102111
APA StyleAbd Halim, N. S., Wirzal, M. D. H., Bilad, M. R., Md Nordin, N. A. H., Adi Putra, Z., Mohd Yusoff, A. R., Narkkun, T., & Faungnawakij, K. (2019). Electrospun Nylon 6,6/ZIF-8 Nanofiber Membrane for Produced Water Filtration. Water, 11(10), 2111. https://doi.org/10.3390/w11102111






