Effects of a Groundwater Heat Pump on Thermophilic Bacteria Activity
Abstract
:1. Introduction
2. Study Area and Methods
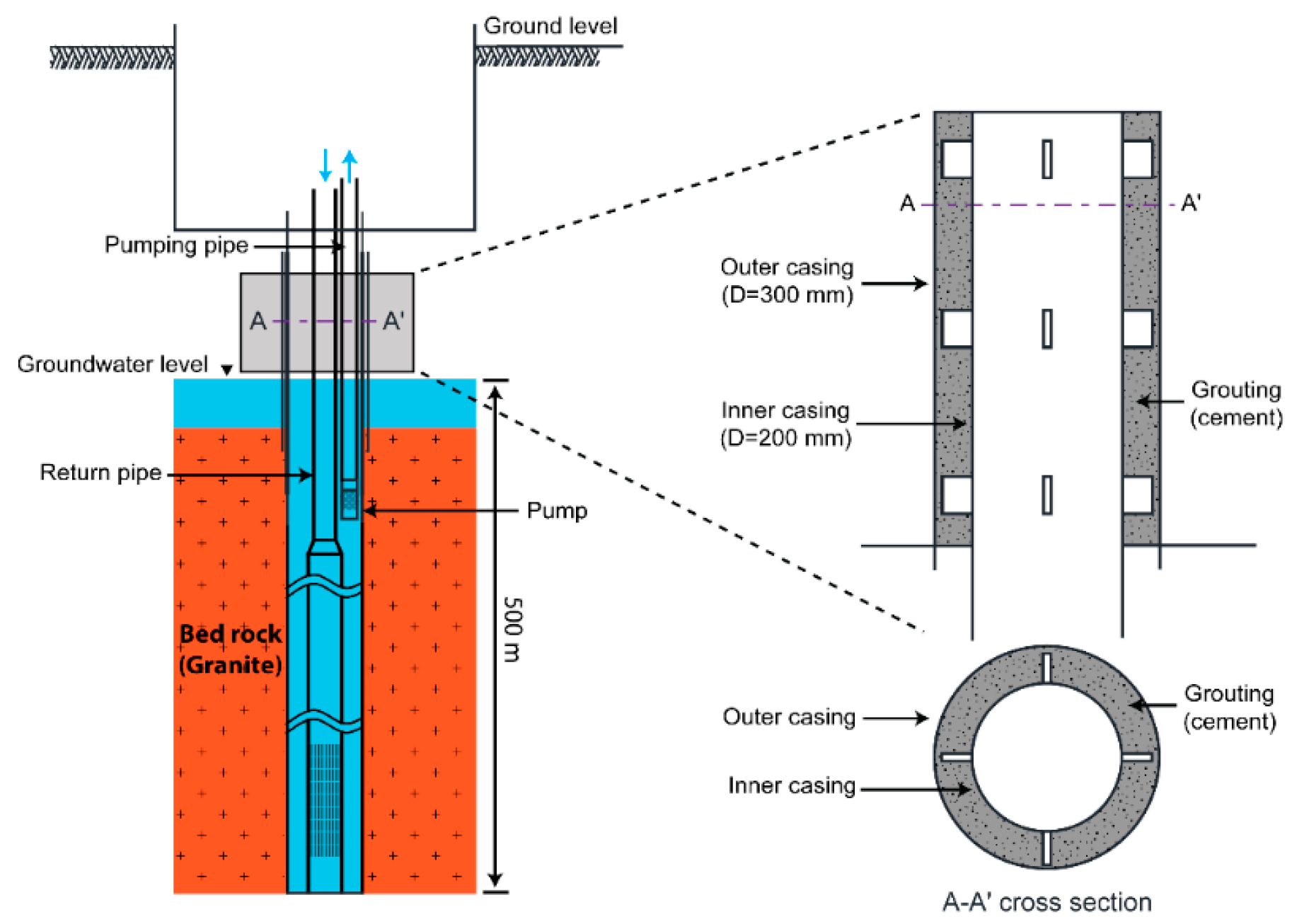
2.1. Study Area
2.2. Physicochemical Analysis
2.3. Microbial Isolation and Culture
2.4. Bacterial Identification
3. Results and Discussion
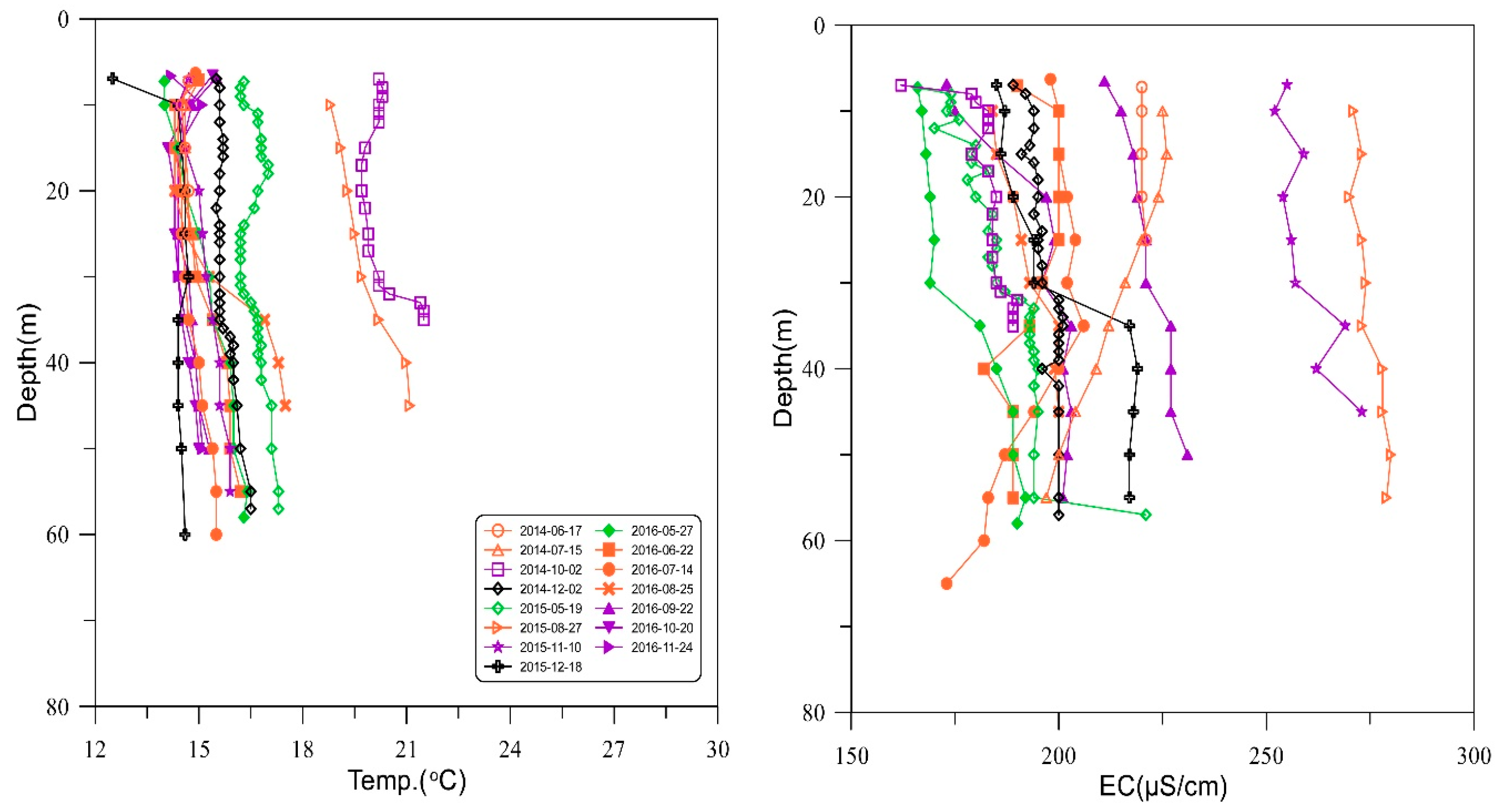
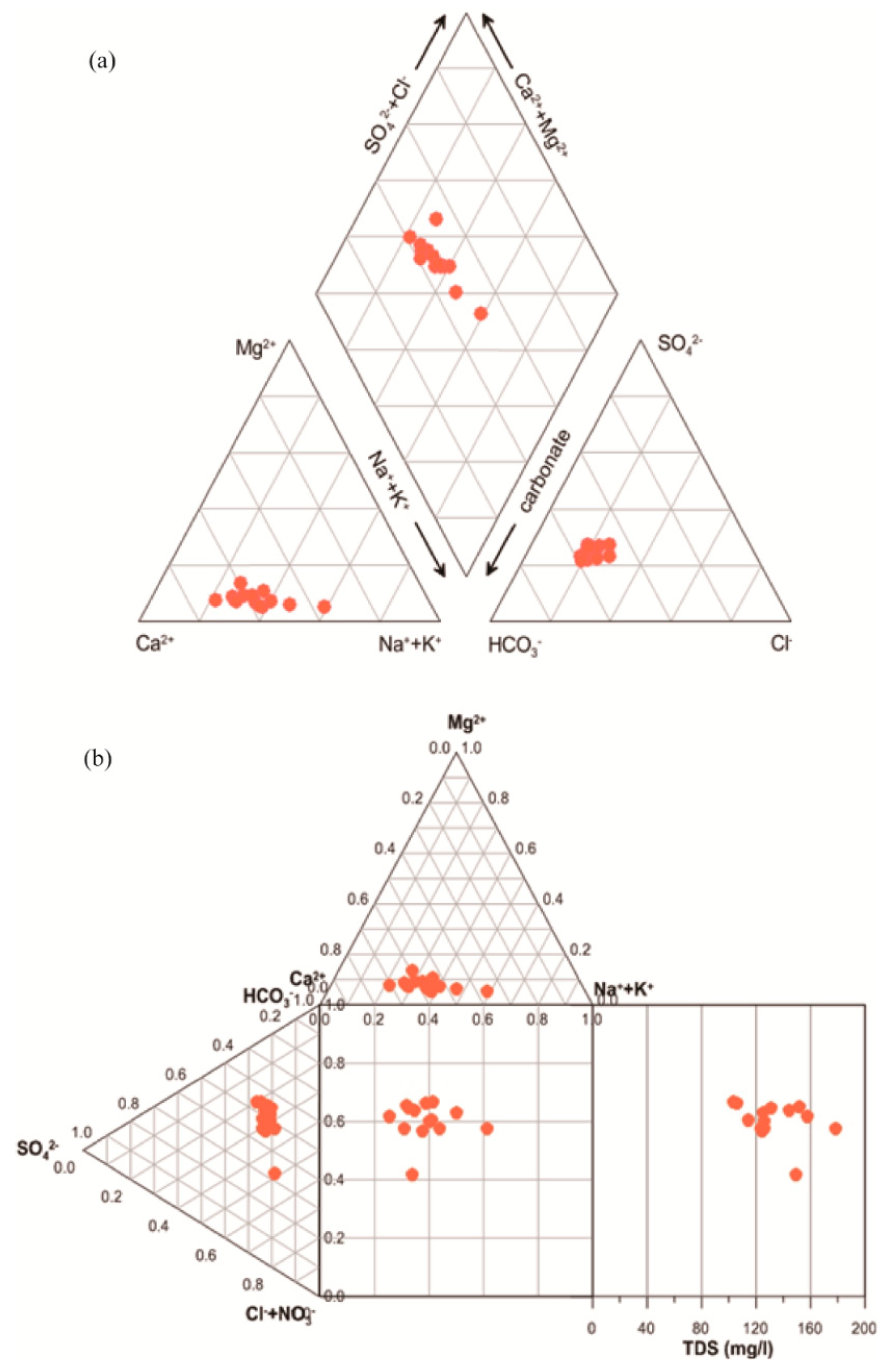
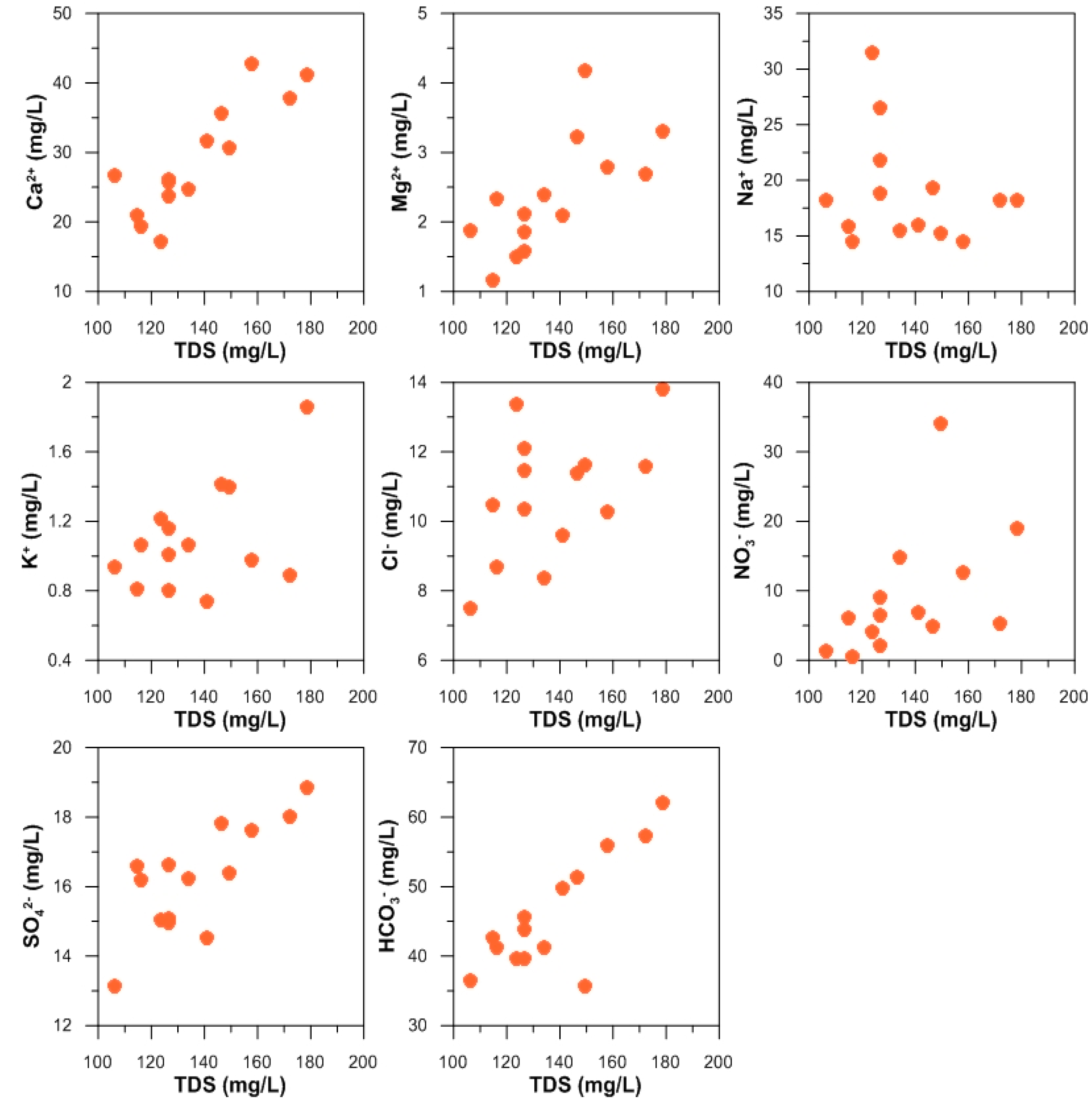
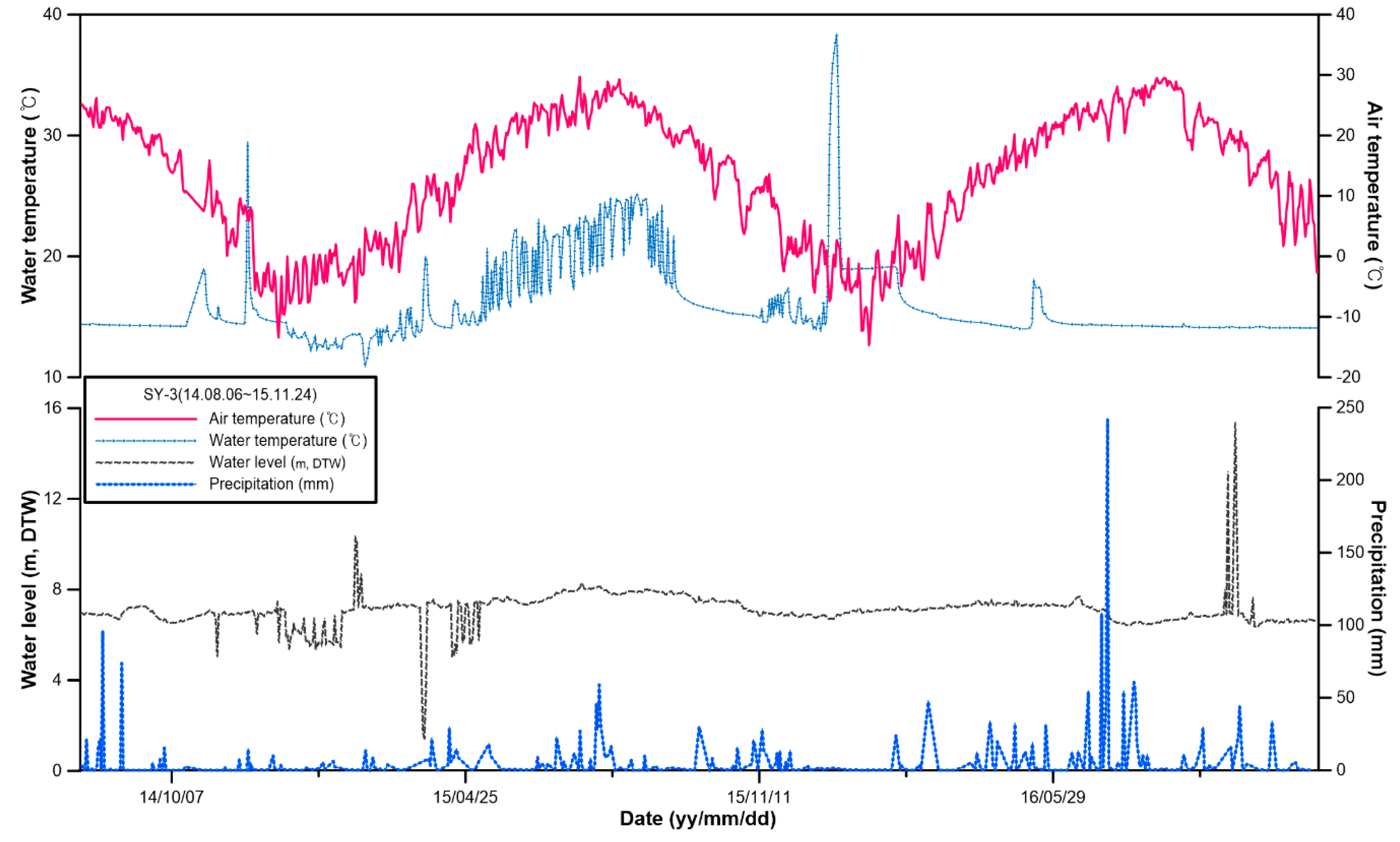
3.1. Physicochemical Composition
3.2. Culturable Bacteria Based on GWHP Operation
3.3. Identified Bacteria
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Boyle, G. Renewable Energy; Oxford University Press: Oxford, UK, 2004. [Google Scholar]
- Quaschning, V.V. Renewable Energy and Climate Change; Wiley: Leicester, UK, 2010. [Google Scholar]
- Lee, J.Y. Current status of ground source heat pumps in Korea. Renew. Sustain. Energy Rev. 2009, 13, 1560–1568. [Google Scholar] [CrossRef]
- Hahn, J.; Han, H.; Hahn, C.; Kim, H.S.; Jeon, J.S. Design guidelines of geothermal heat pump system using standing column well. Econ. Environ. Geol. 2006, 39, 607–613. [Google Scholar]
- Snijders, A.L.; Drijver, B.C. Open-loop heat pump and thermal energy storage systems. In Advances in Ground-Source Heat Pump Systems; Woodhead Publishing: Dexford, UK, 2016; pp. 247–268. [Google Scholar]
- Kim, H.; Mok, J.K.; Park, Y.; Kaown, D.; Lee, K.K. Composition of Groundwater Bacterial Communities before and after Air Surging in a Groundwater Heat Pump System According to a Pyrosequencing Assay. Water 2017, 9, 891. [Google Scholar] [CrossRef]
- Park, Y.; Mok, J.K.; Jang, B.J.; Lee, J.Y.; Park, Y.C. Influence of closed loop ground source heat pumps on groundwater: A case study. J. Geol. Soc. Korea 2015, 51, 243–251. (In Korean) [Google Scholar] [CrossRef]
- Casasso, A.; Sethi, R. Assessment and Minimization of Potential Environmental Impacts of Ground Source Heat Pump (GSHP) Systems. Water 2019, 11, 1573. [Google Scholar] [CrossRef]
- García-Gil, A.; Gasco-Cavero, S.; Garrido, E.; Mejías, M.; Epting, J.; Navarro-Elipe, M.; Alejandre, C.; Sevilla-Alcaine, E. Decreased waterborne pathogenic bacteria in an urban aquifer related to intense shallow geothermal exploitation. Sci. Total Environ. 2018, 633, 765–775. [Google Scholar] [CrossRef]
- García-Gil, A.; Epting, J.; Garrido, E.; Vázquez-Suñé, E.; Lázaro, J.M.; Sánchez Navarro, J.Á.; Huggenberger, P.; Calvo, M.Á.M. A city scale study on the effects of intensive groundwater heat pump systems on heavy metal contents in groundwater. Sci. Total Environ. 2016, 572, 1047–1058. [Google Scholar] [CrossRef]
- Bucci, A.; Prevot, A.B.; Buoso, S.; De Luca, D.A.; Lasagna, M.; Malandrino, M.; Maurino, V. Impacts of borehole heat exchangers (BHEs) on groundwater quality: The role of heat-carrier fluid and borehole grouting. Environ. Earth Sci. 2018, 77, 175. [Google Scholar] [CrossRef]
- Klotzbücher, T.; Kappler, A.; Straub, K.L.; Haderlein, S.B. Biodegradability and groundwater pollutant potential of organic anti-freeze liquids used in borehole heat exchangers. Geothermics 2007, 36, 348–361. [Google Scholar] [CrossRef]
- Bonte, M.; Röling, W.F.M.; Zaura, E.; Van Der Wielen, P.W.J.J.; Stuyfzand, P.J.; Van Breukelen, B.M. Impacts of shallow geothermal energy production on redox processes and microbial communities. Environ. Sci. Technol. 2013, 47, 14476–14484. [Google Scholar] [CrossRef]
- Griebler, C.; Brielmann, H.; Haberer, C.M.; Kaschuba, S.; Kellermann, C.; Stumpp, C.; Hegler, F.; Kuntz, D.; Walker-Hertkorn, S.; Lueders, T. Potential impacts of geothermal energy use and storage of heat on groundwater quality, biodiversity, and ecosystem processes. Environ. Earth Sci. 2016, 75, 1391. [Google Scholar] [CrossRef]
- Rafferty, K.D. Water Chemistry Issues in Geothermal Heat Pump Systems. Ashrae Trans. 2004, 110, 550. [Google Scholar]
- Sanner, B.; Karytsas, C.; Mendrinos, D.; Rybach, L. Current status of ground source heat pumps and underground thermal energy storage in Europe. Geothermics 2003, 32, 579–588. [Google Scholar] [CrossRef]
- Lund, J.; Sanner, B.; Rybach, L.; Curtis, R.; Hellström, G. Geothermal (ground-source) heat pumps: A world overview. Geo-Heat Cent. Bull. 2004, 25, 1–10. [Google Scholar]
- Gao, Q.; Li, M.; Yu, M.; Spitler, J.D.; Yan, Y.Y. Review of development from GSHP to UTES in China and other countries. Renew. Sustain. Energy Rev. 2009, 13, 1383–1394. [Google Scholar] [CrossRef]
- Abesser, C. Open-Loop Ground Source Heat Pumps and the Groundwater Systems: A Literature Review of Current Application, Regulations and Problems; Energy Geoscience Programme Open Report OR/10/045; British Geological Survey: Nottingham, UK, 2010. [Google Scholar]
- Bonte, M. Impacts of Shallow Geothermal Energy on Groundwater Quality—A Hydrochemical and Geomicrobial Study on the Effects of Ground Source Heat Pumps and Aquifer Thermal Energy Storage. Ph.D. Thesis, VU University Amsterdam, Amsterdam, The Netherlands, 2013. [Google Scholar]
- Antics, M.; Bertani, R.; Sanner, B. Summary of EGC 2016 Country Update Reports on Geothermal Energy in Europe. In Proceedings of the European Geothermal Congress 2016, Strasbourg, France, 19–24 September 2016; pp. 1–16. [Google Scholar]
- Park, Y.; Kim, N.; Lee, J.Y. Geochemical properties of groundwater affected by open loop geothermal heat pump systems in Korea. Geosci. J. 2015, 19, 515–526. [Google Scholar] [CrossRef]
- York, K.P.; Jahangir, Z.M.G.S.; Solomon, T.; Stafford, L. Effects of a large scale geothermal heat pump installation on aquifer microbiota. In Proceedings of the 2nd Stockton International Geothermal Conference, Stockton, NJ, USA, 16–17 March 1998; p. 8. [Google Scholar]
- Jo, Y.J.; Lee, J.Y.; Lim, S.Y.; Hong, G.P. A review on potential effects of installation and operation of ground source heat pumps on soil and groundwater environment. J. Soil Groundw. Environ. 2009, 14, 22–31. [Google Scholar]
- Jo, Y.J.; Lee, J.Y.; Kim, C.G.; Han, J.S. Effects of grouts and temperature change on microorganisms in geothermal heat pump. J. Soil Groundw. Environ. 2009, 14, 10–14. [Google Scholar]
- Saner, D.; Juraske, R.; Kübert, M.; Blum, P.; Hellweg, S.; Bayer, P. Is it only CO2 that matters? A life cycle perspective on shallow geothermal systems. Renew. Sustain. Energy Rev. 2010, 14, 1798–1813. [Google Scholar] [CrossRef]
- Bayer, P.; Saner, D.; Bolay, S.; Rybach, L.; Blum, P. Greenhouse gas emission savings of ground source heat pump systems in Europe: A review. Renew. Sustain. Energy Rev. 2012, 16, 1256–1267. [Google Scholar] [CrossRef]
- Rivoire, M.; Casasso, A.; Piga, B.; Sethi, R. Assessment of Energetic, Economic and Environmental Performance of Ground-Coupled Heat Pumps. Energies 2018, 11, 1941. [Google Scholar] [CrossRef]
- Houben, G.; Treskatis, C. Water Well Rehabilitation and Reconstruction; McGraw-Hill: New York, NY, USA, 2007. [Google Scholar]
- Smith, S.A.; Comeskey, A.E. Sustainable Wells: Maintenance, Problem Prevention, and Rehabilitation; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- van Beek, C.K. Cause and Prevention of Clogging of Wells Abstracting Groundwater from Unconsolidated Aquifers; IWA Publishing: London, UK, 2011. [Google Scholar]
- Burté, L.; Cravotta, C.; Bethencourt, L.; Farasin, J.; Pédrot, M.; Dufresne, A.; Gérard, M.-F.; Baranger, C.; Le Borgne, T.; Aquilina, L. Kinetic study on clogging of a geothermal pumping well triggered by mixing-induced biogeochemical reactions. Environ. Sci. Technol. 2019, 53, 10. [Google Scholar] [CrossRef] [PubMed]
- Mirza, M.M.Q. The Ganges Water Diversion: Environmental Effects and Implications; Springer: Dordrecht, The Netherlands, 2004; pp. 93–95. [Google Scholar]
- Chadha, D.K. A proposed new diagram for geochemical classification of natural waters and interpretation of chemical data. Hydrogeol. J. 1999, 7, 431–439. [Google Scholar] [CrossRef]
- Schneider, E.A.G.; García-Gil, A.; Vázquez-Suñè, E.; Sánchez-Navarro, J.Á. Geochemical impacts of groundwater heat pump systems in an urban alluvial aquifer with evaporitic bedrock. Sci. Total Environ. 2016, 544, 354–368. [Google Scholar] [CrossRef] [PubMed]
- Belduz, A.O.; Dulger, S.; Demirbag, Z. Anoxybacillus gonensis sp. nov., a moderately thermophilic, xylose-utilizing, endospore-forming bacterium. Int. J. Syst. Evol. Microbiol. 2003, 53, 1315–1320. [Google Scholar] [CrossRef]
- Pikuta, E.; Cleland, D.; Tang, J. Aerobic growth of Anoxybacillus pushchinoensis K1T: Emended descriptions of A. pushchinoensis and the genus Anoxybacillus. Int. J. Syst. Evol. Microbiol. 2003, 53, 1561–1562. [Google Scholar] [CrossRef]
- Filippidou, S.; Jaussi, M.; Junier, T.; Wunderlin, T.; Jeanneret, N.; Palmieri, F.; Palmieri, I.; Roussel-Delif, L.; Vieth-Hillebrand, A.; Vetter, A.; et al. Anoxybacillus geothermalis sp. nov., a facultatively anaerobic, endospore-forming bacterium isolated from mineral deposits in a geothermal station. Int. J. Syst. Evol. Microbiol. 2016, 66, 2944–2951. [Google Scholar] [CrossRef]
- Dai, J.; Liu, Y.; Lei, Y.; Gao, Y.; Han, F.; Xiao, Y.; Peng, H. A new subspecies of Anoxybacillus flavithermus ssp. yunnanensis ssp. nov. with very high ethanol tolerance. FEMS Microbiol. Lett. 2011, 320, 72–78. [Google Scholar]
- Schäffer, C.; Franck, W.L.; Scheberl, A.; Kosma, P.; McDermott, T.R.; Messner, P. Classification of isolates from locations in Austria and Yellowstone National Park as Geobacillus tepidamans sp. nov. Int. J. Syst. Evol. Microbiol. 2004, 54, 2361–2368. [Google Scholar] [CrossRef]
- Coorevits, A.; Dinsdale, A.E.; Halket, G.; Lebbe, L.; De Vos, P.; Van Landschoot, A.; Logan, N.A. Taxonomic revision of the genus Geobacillus: Emendation of Geobacillus, G. stearothermophilus, G. jurassicus, G. toebii, G. thermodenitrificans and G. thermoglucosidans (nom. corrig., formerly ‘thermoglucosidasius’); transfer of Bacillus thermantarcticus to the genus as G. thermantarcticus comb. nov.; proposal of Caldibacillus debilis gen. nov., comb. nov.; transfer of G. tepidamans to Anoxybacillus as A. tepidamans comb. nov.; and proposal of Anoxybacillus caldiproteolyticus sp. nov. Int. J. Syst. Evol. Microbiol. 2012, 62, 1470–1485. [Google Scholar]
- Zhang, J.; Wang, J.; Fang, C.; Song, F.; Xin, Y.; Qu, L.; Ding, K. Bacillus oceanisediminis sp. nov., isolated from marine sediment. Int. J. Syst. Evol. Microbiol. 2010, 60, 2924–2929. [Google Scholar] [CrossRef] [PubMed]
- Kolari, M.; Nuutinen, J.; Salkinoja-Salonen, M.S. Mechanisms of biofilm formation in paper machine by Bacillus species: The role of Deinococcus geothermalis. J. Ind. Microbiol. Biotechnol. 2001, 27, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Kojima, H.; Fukui, M. Proposal of Effusibacillus lacus gen. nov., sp. nov., and reclassification of Alicyclobacillus pohliae as Effusibacillus pohliae comb. nov. and Alicyclobacillus consociatus as Effusibacillus consociatus comb. nov. Int. J. Syst. Evol. Microbiol. 2014, 64, 2770–2774. [Google Scholar] [CrossRef] [PubMed]
- Heyndrickx, M.; Vandemeulebroecke, K.; Scheldeman, P.; Kersters, K.; De Vos, P.; Logan, N.A.; Aziz, A.M.; Ali, N.; Berkeley, R.C.W. A polyphasic reassessment of the genus Paenibacillus, reclassification of Bacillus lautus (Nakamura 1984) as Paenibacillus lautus comb. nov. and of Bacillus peoriae (Montefusco et al. 1993) as Paenibacillus peoriae comb. nov., and emended descriptions of P. lautus and of P. peoriae. Int. J. Syst. Evol. Microbiol. 1996, 46, 988–1003. [Google Scholar]
- Yu, T.T.; Zhou, E.M.; Yin, Y.R.; Yao, J.C.; Ming, H.; Dong, L.; Nie, G.X.; Li, W.J. Vulcaniibacterium tengchongense gen. nov., sp. nov. isolated from a geothermally heated soil sample, and reclassification of Lysobacter thermophilus Wei et al. 2012 as Vulcaniibacterium thermophilum comb. nov. Antonie van Leeuwenhoek 2013, 104, 369–376. [Google Scholar] [CrossRef]
| # | Sample | Date | CFU/L |
|---|---|---|---|
| 1 | SY-3 | 13 August 2014 | 1.6 × 109 |
| 2 | 28 October 2014 | 7.0 × 107 | |
| 3 | 2 December 2014 | 9.0 × 107 | |
| 4 | 9 November 2015 | 2.9 × 108 | |
| 5 | 27 May 2016 | 7.4 × 108 | |
| 6 | 25 August 2016 | 1.0 × 108 | |
| 7 | 24 November 2016 | 1.0 × 107 |
| Sample | Isolate | Strain | Hit | Similarity (%) | |
|---|---|---|---|---|---|
| SY-3 | 13 August 2014 | Major | 1M | Vulcaniibacterium thermophilum | 99.9 |
| Specific | 11 | Anoxybacillus tepidamans | 98.8 | ||
| 28 October 2014 | Major | 2M | Anoxybacillus tepidamans | 98.8 | |
| Specific | 21 | Effusibacillus pohliae | 99.2 | ||
| 2 December 2014 | Major | 3M | Anoxybacillus tepidamans | 98.8 | |
| Specific | 31 | Anoxybacillus tepidamans | 98.9 | ||
| 9 November 2015 | Major | 4M | Deinococcus geothermalis | 98.1 | |
| Specific | 41 | Anoxybacillus tepidamans | 98.6 | ||
| 27 May 2016 | Major | 5M | Effusibacillus pohliae | 99.3 | |
| Specific | 51 | Not determined | - | ||
| 25 August 2016 | Major | 6M | Anoxybacillus tepidamans | 98.7 | |
| Specific | 61 | Anoxybacillus tepidamans | 98.8 | ||
| 24 November 2016 | Major | 7M | Effusibacillus pohliae | 99.2 | |
| Specific | 71 | Bacillus oceanisediminis | 99.4 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Lee, J.-Y. Effects of a Groundwater Heat Pump on Thermophilic Bacteria Activity. Water 2019, 11, 2084. https://doi.org/10.3390/w11102084
Kim H, Lee J-Y. Effects of a Groundwater Heat Pump on Thermophilic Bacteria Activity. Water. 2019; 11(10):2084. https://doi.org/10.3390/w11102084
Chicago/Turabian StyleKim, Heejung, and Jin-Yong Lee. 2019. "Effects of a Groundwater Heat Pump on Thermophilic Bacteria Activity" Water 11, no. 10: 2084. https://doi.org/10.3390/w11102084
APA StyleKim, H., & Lee, J.-Y. (2019). Effects of a Groundwater Heat Pump on Thermophilic Bacteria Activity. Water, 11(10), 2084. https://doi.org/10.3390/w11102084






