Wetland Biomass and Productivity in Coastal Louisiana: Base Line Data (1976–2015) and Knowledge Gaps for the Development of Spatially Explicit Models for Ecosystem Restoration and Rehabilitation Initiatives
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
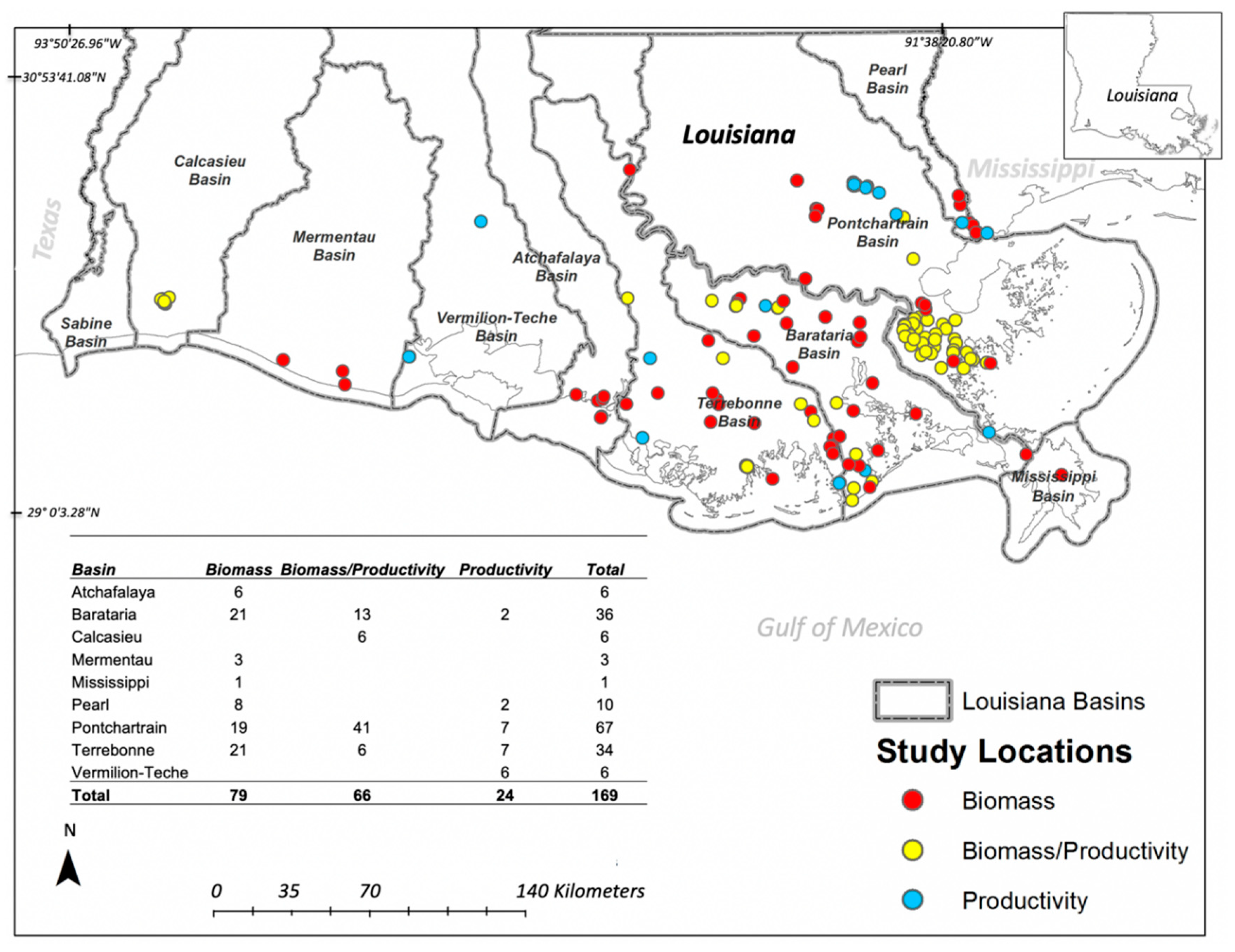
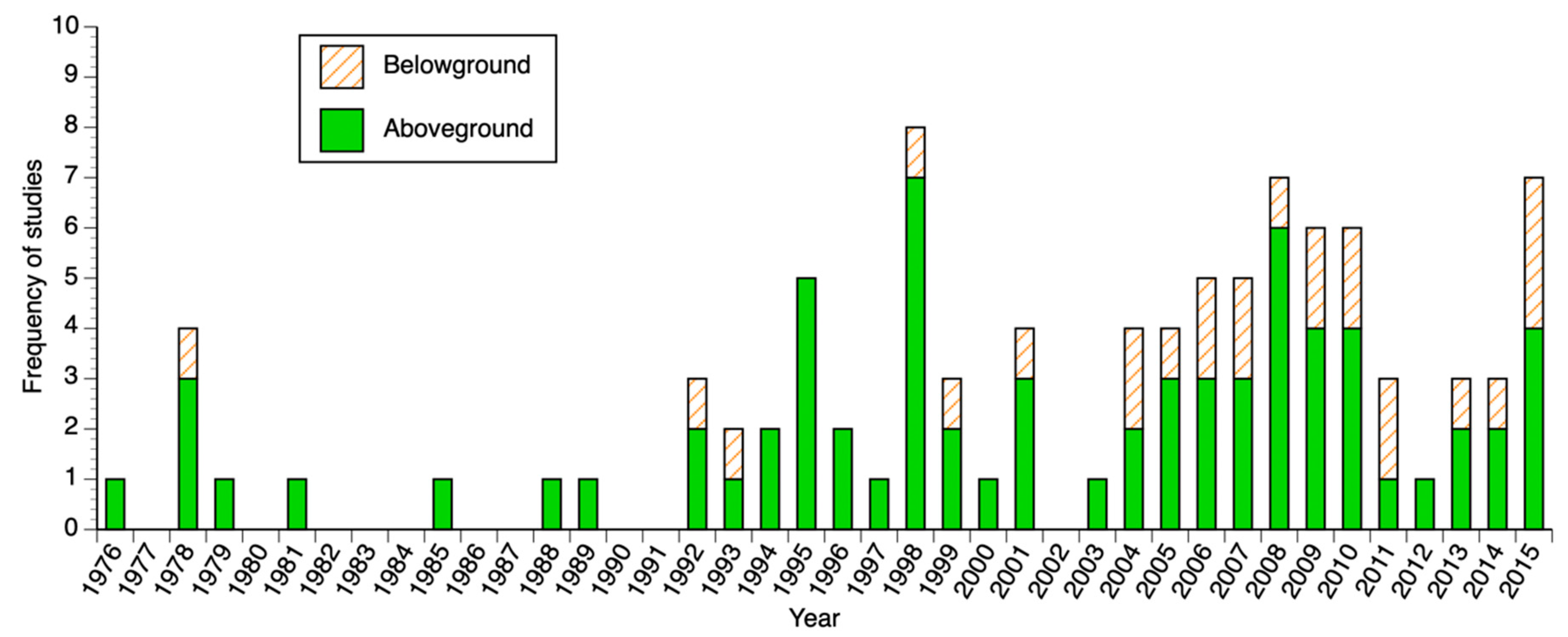
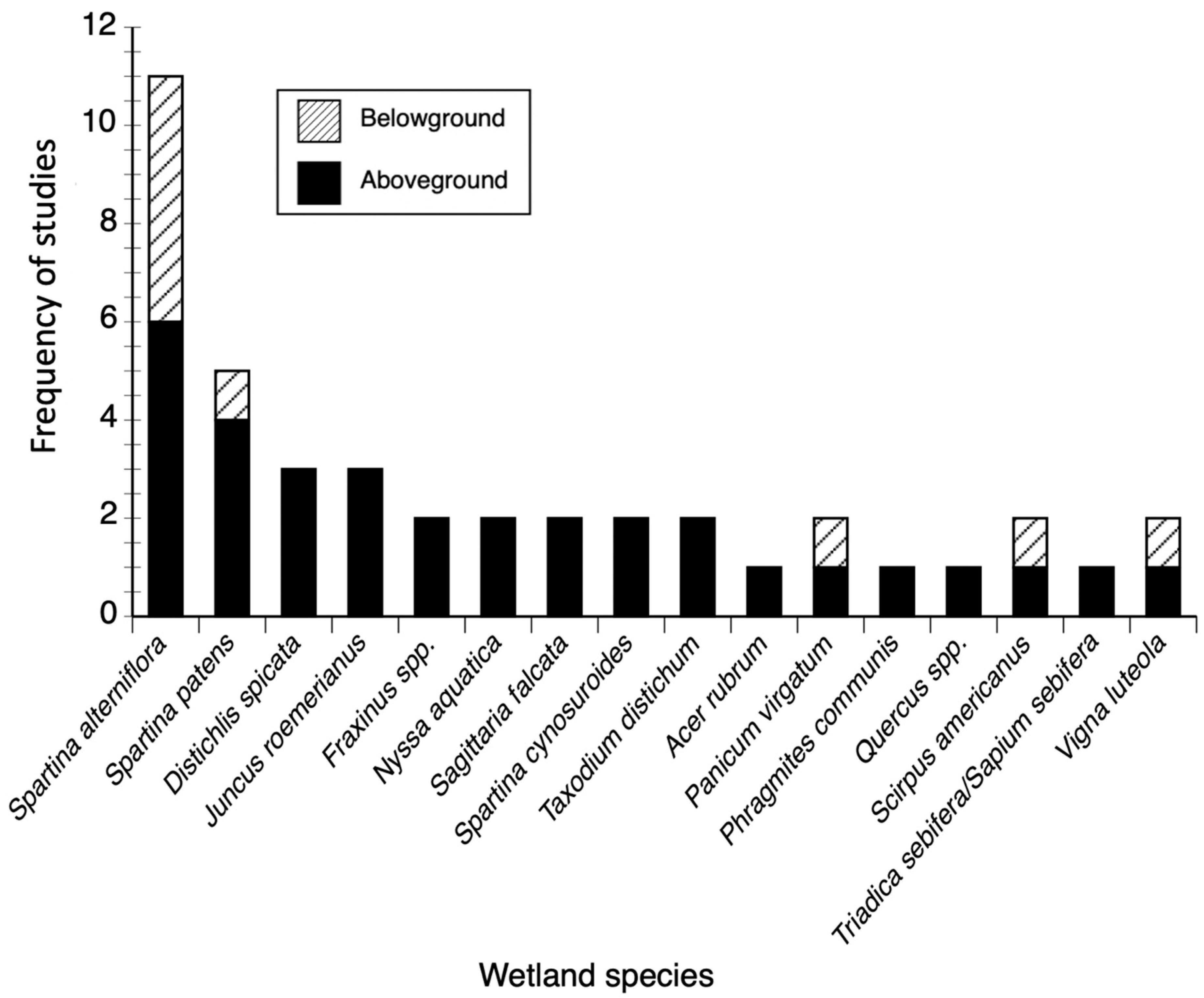
3.1. Biomass and Productivity Studies Frequency and Spatial Distribution
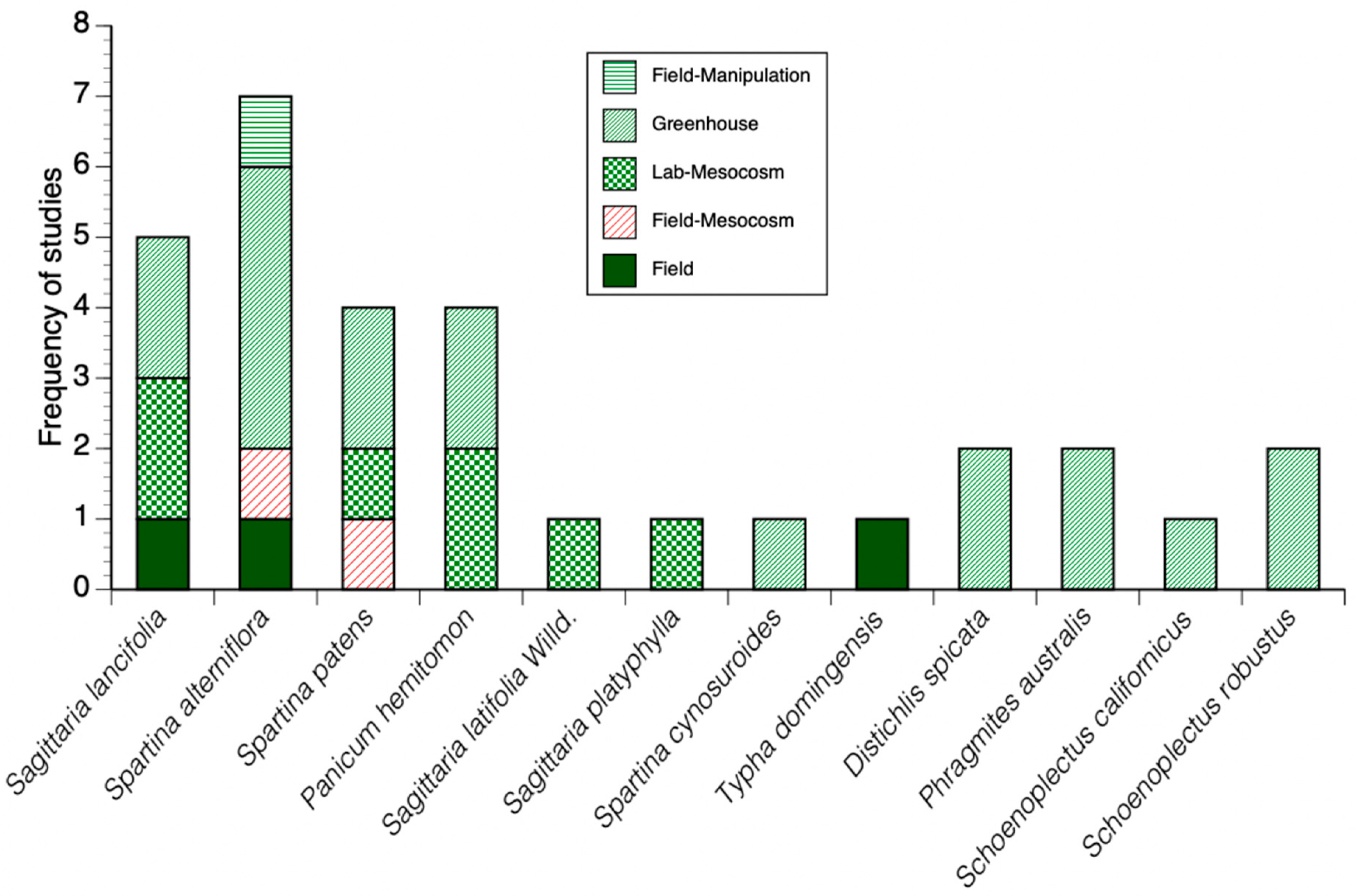
3.2. Biomass and Productivity Values
3.2.1. Spartina alterniflora
3.2.2. Spartina cynosuroides
3.2.3. Spartina patens
3.2.4. Sagittaria lancifolia (sy. S. falcata) and Sagittaria latifolia
3.2.5. Panicum hemitomon and Panicum virgatum
3.2.6. Distichlis spicata
3.2.7. Alternanthera philoxeroides (Mart.) griseb.
3.2.8. Polygonum punctatum
3.2.9. Schoenoplectus americanus (Syn.Scirpus americanus)
3.2.10. Leersia oryzoides
3.2.11. Vigna luteola
3.2.12. Juncus roemerianus
3.2.13. Lythrum lineare
3.2.14. Swamp (Taxodium distichum, Nyssa aquatica, Fraxinus spp)
3.3. Data Availability in Vegetation Modeling
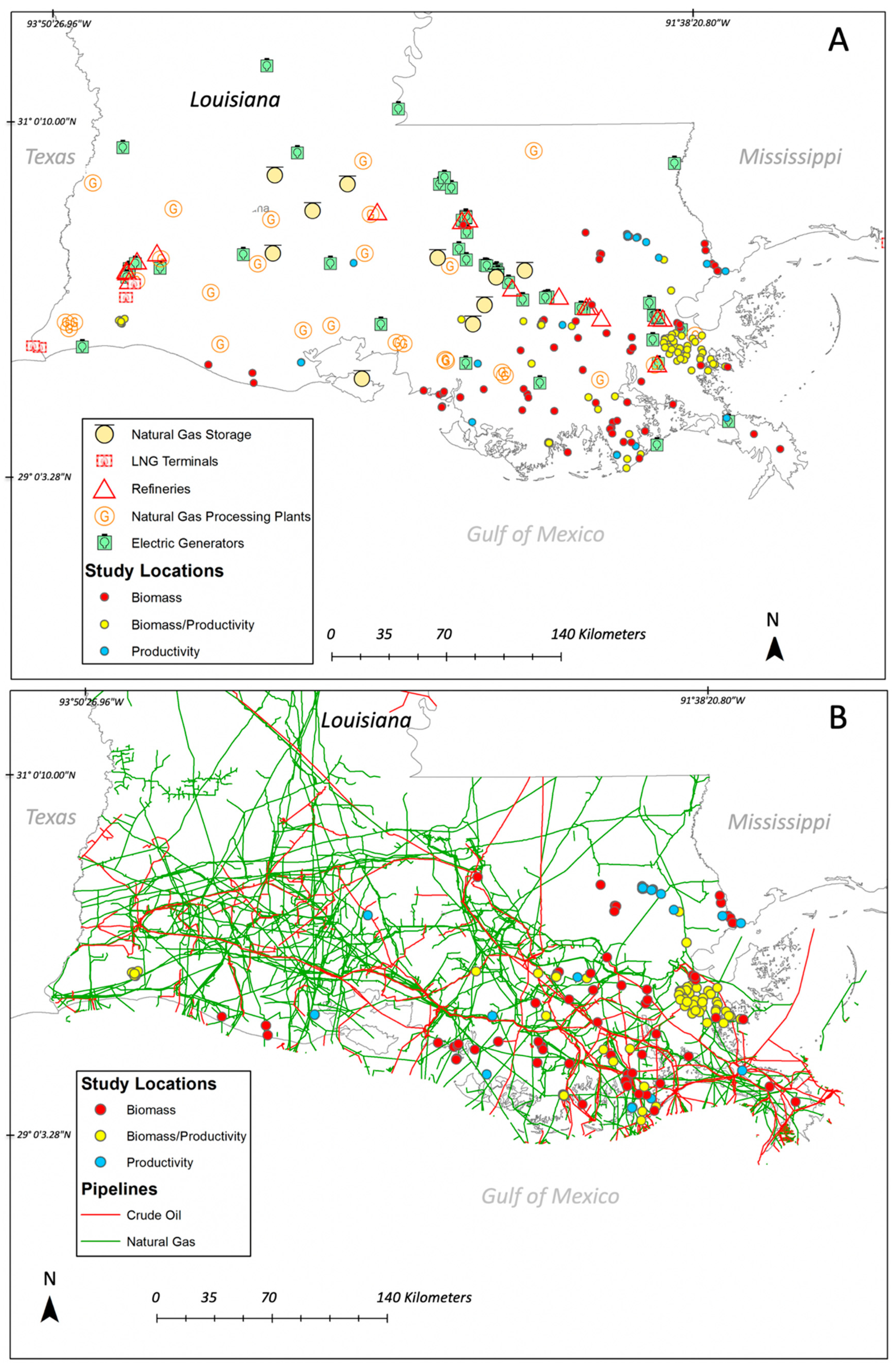
3.4. Basin-Level Vegetation Data Availability and Human Impacts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Davidson, N.C. How much wetland has the world lost? Long-term and recent trends in global wetland area. Mar. Freshw. Res. 2014, 65, 934–941. [Google Scholar] [CrossRef]
- Church, J.A.; White, N.J. A 20th century acceleration in global sea-level rise. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Nicholls, R.J. Coastal flooding and wetland loss in the 21st century: Changes under the SRES climate and socio-economic scenarios. Glob. Environ. Chang. 2004, 14, 69–86. [Google Scholar] [CrossRef]
- Hu, S.; Niu, Z.; Chen, Y.; Li, L.; Zhang, H. Global wetlands: Potential distribution, wetland loss, and status. Sci. Total Environ. 2017, 586, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.M.; Huh, O.K.; Braud, D., Jr. Wetland loss in world deltas. J. Coast. Res. 2008, 24, 1–14. [Google Scholar] [CrossRef]
- Day, J.W.; Barras, J.; Clairain, E.; Johnston, J.; Justic, D.; Kemp, G.P.; Ko, J.-Y.; Lane, R.; Mitsch, W.J.; Steyer, G.; et al. Implications of global climatic change and energy cost and availability for the restoration of the Mississippi delta. Ecol. Eng. 2005, 24, 253–265. [Google Scholar] [CrossRef]
- Williams, S.J.; Stone, G.W.; Burruss, A.E. A perspective on the Louisiana wetland loss and coastal erosion problem. J. Coast. Res. 1997, 13, 593–594. [Google Scholar]
- Barbier, E.B.; Georgiou, I.Y.; Enchelmeyer, B.; Reed, D.J. The Value of Wetlands in Protecting Southeast Louisiana from Hurricane Storm Surges. PLoS ONE 2013, 8, e58715. [Google Scholar] [CrossRef]
- LCWCRTF. Louisiana coastal wetlands restoration plan. In Coastal Wetlands Planning Protection and Restoration Act; US Army Corps of Enginners: New Orleans, LA, USA, 1993. [Google Scholar]
- Couvillion, B.R.; Beck, H.; Schoolmaster, D.; Fischer, M. Land Area Change in Coastal Louisiana 1932 to 2016; U.S. Geological Survey Reston: Reston, VA, USA, 2017; p. 16.
- Meckel, T.A.; Brink, U.S.T.; Williams, S.J. Current subsidence rates due to compaction of Holocene sediments in southern Louisiana. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Day, J.W. Restoration of wetlands in the Mississippi–Ohio–Missouri (MOM) River Basin: Experience and needed research. Ecol. Eng. 2006, 26, 55–69. [Google Scholar] [CrossRef]
- Peyronnin, N.; Green, M.; Richards, C.P.; Owens, A.; Reed, D.; Chamberlain, J.; Groves, D.G.; Rhinehart, W.K.; Belhadjali, K. Louisiana’s 2012 coastal master plan: Overview of a science-based and publicly informed decision-making process. J. Coast. Res. 2013, 1–15. [Google Scholar] [CrossRef]
- Evers, D.E.; Gosselink, J.G.; Sasser, C.E.; Hill, J.M. Wetland loss dynamics in southwestern Barataria basin, Louisiana (USA), 1945–1985. Wetl. Ecol. Manag. 1992, 2, 103–118. [Google Scholar] [CrossRef]
- Stone, G.W.; Grymes, J.M.; Dingler, J.R.; Pepper, D.A. Overview and significance of hurricanes on the Louisiana coast, USA. J. Coast. Res. 1997, 13, 656–669. [Google Scholar]
- McFalls, T.B.; Keddy, P.A.; Campbell, D.; Shaffer, G. Hurricanes, Floods, Levees, and Nutria: Vegetation Responses to Interacting Disturbance and Fertility Regimes with Implications for Coastal Wetland Restoration. J. Coast. Res. 2010, 265, 901–911. [Google Scholar] [CrossRef]
- Day, J.W.; Boesch, D.F.; Clairain, E.J.; Kemp, G.P.; Laska, S.B.; Mitsch, W.J.; Orth, K.; Mashriqui, H.; Reed, D.J.; Shabman, L.; et al. Restoration of the Mississippi Delta: Lessons from Hurricanes Katrina and Rita. Science 2007, 315, 1679–1684. [Google Scholar] [CrossRef]
- Liu, K.B.; Li, C.; Bianchette, T.A.; McCloskey, T.A.; Yao, Q.; Weeks, E. Storm deposition in a coastal backbarrier lake in Louisiana caused by hurricanes Gustav and Ike. J. Coast. Res. 2011, SI64, 1866–1870. [Google Scholar]
- Bianchette, T.A.; Liu, K.B.; Qiang, Y.; Lam, N.S.N. Wetland accretion rates along coastal Louisiana: Spatial and temporal variability in light of hurricane Isaac’s impacts. Water 2016, 8, 1. [Google Scholar] [CrossRef]
- Martin, J.F.; White, M.L.; Reyes, E.; Kemp, G.P.; Mashriqui, H.; Day, J.W. Evaluation of coastal management plans with a spatial model: Mississippi delta, Louisiana, USA. Environ. Manag. 2000, 26, 117–129. [Google Scholar] [CrossRef]
- Reyes, E.; White, M.L.; Martin, J.F.; Kemp, G.P.; Day, J.W.; Aravamuthan, V. Landscape modeling of coastal habitat change in the mississippi delta. Ecology 2000, 81, 2331–2349. [Google Scholar] [CrossRef]
- Reyes, E.; Georgiou, I.; Reed, D.; McCorquodale, A. Using models to evaluate the effects of barrier islands on estuarine hydrodynamics and habitats: A numerical experiment. J. Coast. Res. 2005, SI44, 176–185. [Google Scholar]
- Reyes, E.; Day, J.W.; White, M.L.; Yáñez-Arancibia, A. Ecological and resource management information transfer for Laguna de Terminos, Mexico: A computerized interface. Coast. Manag. 1993, 21, 37–51. [Google Scholar] [CrossRef]
- Habib, E.; Nuttle, W.K.; Rivera-Monroy, V.H.; Gautam, S.; Wang, J.; Meselhe, E.; Twilley, R.R. Assessing Effects of Data Limitations on Salinity Forecasting in Barataria Basin, Louisiana, with a Bayesian Analysis. J. Coast. Res. 2007, 233, 749–763. [Google Scholar] [CrossRef]
- Wang, H.; Steyer, G.D.; Couvillion, B.R.; Rybczyk, J.M.; Beck, H.J.; Sleavin, W.J.; Meselhe, E.A.; Allison, M.A.; Boustany, R.G.; Fischenich, C.J.; et al. Forecasting landscape effects of Mississippi River diversions on elevation and accretion in Louisiana deltaic wetlands under future environmental uncertainty scenarios. Estuar. Coast. Shelf Sci. 2014, 138, 57–68. [Google Scholar] [CrossRef]
- Meselhe, E.; McCorquodale, J.A.; Shelden, J.; Dortch, M.; Brown, T.S.; Elkan, P.; Rodrigue, M.D.; Schindler, J.K.; Wang, Z. Ecohydrology component of Louisiana’s 2012 coastal master plan: Mass-balance compartment model. J. Coast. Res. 2013, 67, 16–28. [Google Scholar] [CrossRef]
- Allison, M.A.; Meselhe, E.A. The use of large water and sediment diversions in the lower Mississippi River (Louisiana) for coastal restoration. J. Hydrol. 2010, 387, 346–360. [Google Scholar] [CrossRef]
- Twilley, R.R.; Couvillion, B.R.; Hossain, I.; Kaiser, C.; Owens, M.S.; Steyer, G.; Visser, J. Coastal Louisiana ecosystem assessment and restoration program: The role of ecosystem forecasting in evaluating restoration planning in the mississippi river deltaic plain. Trans. Am. Fish. Soc. 2008, 64, 29–46. [Google Scholar]
- Steyer, G.D.; Sasser, C.E.; Visser, J.M.; Swenson, E.M.; Nyman, J.A.; Raynie, R.C. A Proposed Coast-Wide Reference Monitoring System for Evaluating Wetland Restoration Trajectories in Louisiana. Coast. Monit. Through Partnersh. 2003, 81, 107–117. [Google Scholar]
- Morris, J.T.; Sundareshwar, P.V.; Nietch, C.T.; Kjerfve, B.; Cahoon, D.R. Responses of coastal wetlands to rising sea level. Ecology 2002, 83, 2869–2877. [Google Scholar] [CrossRef]
- Kirwan, M.L.; Guntenspergen, G.R. Influence of tidal range on the stability of coastal marshland. J. Geophys. Res. Space Phys. 2010, 115. [Google Scholar] [CrossRef]
- Visser, J.M.; Duke-Sylvester, S.M.; Carter, J.; Broussard, W.P. A computer model to forecast wetland vegetation changes resulting from restoration and protection in coastal Louisiana. J. Coast. Res. 2013, 67, 51–59. [Google Scholar] [CrossRef]
- Gough, L.; Grace, J.B.; Taylor, K.L. The Relationship between Species Richness and Community Biomass: The Importance of Environmental Variables. Oikos 1994, 70, 271–279. [Google Scholar] [CrossRef]
- Visser, J.M.; Duke-Sylvester, S.M. Lavegmod v2: Modeling coastal vegetation dynamics in response to proposed coastal restoration and protection projects in Louisiana, USA. Sustainability 2017, 9, 1625. [Google Scholar] [CrossRef]
- Fisher, R.; McDowell, N.; Purves, D.; Moorcroft, P.; Sitch, S.; Cox, P.; Huntingford, C.; Meir, P.; Woodward, F.I. Assessing uncertainties in a second-generation dynamic vegetation model caused by ecological scale limitations. New Phytol. 2010, 187, 666–681. [Google Scholar] [CrossRef] [PubMed]
- Habib, E.; Reed, D. Parametric uncertainty analysis of predictive models in Louisiana’s 2012 coastal master plan. J. Coast. Res. 2013, 67, 127–146. [Google Scholar] [CrossRef]
- Li, H.; Wu, B. Uncertainty analysis in ecological studies: An overview. In Scaling and Uncertainty Analysis in Ecology: Methods and Applications; Wu, J., Jones, K.B., Li, H., Loucks, O.L., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 45–66. [Google Scholar]
- Liang, W.; Lam, N.S.-N.; Qin, X.; Ju, W. A Two-level Agent-Based Model for Hurricane Evacuation in New Orleans. J. Homel. Secur. Emerg. Manag. 2015, 12, 407–435. [Google Scholar] [CrossRef]
- Visser, J.M.; Sasser, C.E.; Linscombe, R.G.; Chabreck, R.H. Marsh Vegetation Types of the Chenier Plain, Louisiana, USA. Estuaries 2000, 23, 318–327. [Google Scholar] [CrossRef]
- Parrondo, R.T.; Gosselink, J.G.; Hopkinson, C.S. Effects of Salinity and Drainage on the Growth of Three Salt Marsh Grasses. Int. J. Plant. Sci. 1978, 139, 102–107. [Google Scholar] [CrossRef]
- Hester, M.W.; Mendelssohn, I.A.; McKee, K.L. Species and population variation to salinity stress in Panicum hemitomon, Spartina patens, and Spartina alterniflora: Morphological and physiological constraints. Environ. Exp. Bot. 2001, 46, 277–297. [Google Scholar] [CrossRef]
- Broome, S.W.; Mendelssohn, I.A.; McKee, K.L. Relative growth of Spartina patens (Ait.) Muhl. andScirpus olneyi gray occurring in a mixed stand as affected by salinity and flooding depth. Wetlands 1995, 15, 20–30. [Google Scholar] [CrossRef]
- La Peyre, M.K.G.; Grace, J.B.; Hahn, E.; Mendelssohn, I.A. The Importance of Competition in Regulating Plant Species Abundance along a Salinity Gradient. Ecology 2001, 82, 62–69. [Google Scholar] [CrossRef]
- Merino, J.H.; Huval, D.; Nyman, A.J. Implication of nutrient and salinity interaction on the productivity of spartina patens. Wetl. Ecol. Manag. 2010, 18, 111–117. [Google Scholar] [CrossRef]
- Howard, R.J.; Mendelssohn, I.A. Salinity as a constraint on growth of oligohaline marsh macrophytes. I. Species variation in stress tolerance. Am. J. Bot. 1999, 86, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Howard, R.J.; Mendelssohn, I.A. Salinity as a constraint on growth of oligohaline marsh macrophytes. II. Salt pulses and recovery potential. Am. J. Bot. 1999, 86, 795–806. [Google Scholar] [CrossRef] [PubMed]
- Mayence, C.E.; Hester, M.W. Assessment of a Multi-Species Planting Approach for Restoring Thick-Mat Floating Marsh, Louisiana, U.S.A. Restor. Ecol. 2011, 19, 216–224. [Google Scholar] [CrossRef]
- Geho, E.M.; Campbell, D.; Keddy, P.A. Quantifying ecological filters: The relative impact of herbivory, neighbors, and sediment on an oligohaline marsh. Oikos 2007, 116, 1006–1016. [Google Scholar] [CrossRef][Green Version]
- Mayence, C.E.; Hester, M.W. Growth and allocation by a keystone wetland plant, Panicum hemitomon, and implications for managing and rehabilitating coastal freshwater marshes, Louisiana, USA. Wetl. Ecol. Manag. 2010, 18, 149–163. [Google Scholar] [CrossRef]
- Jeng, H.; Hong, Y.-J. Assessment of a Natural Wetland for Use in Wastewater Remediation. Environ. Monit. Assess. 2005, 111, 113–131. [Google Scholar] [CrossRef]
- McKee, K.L.; Cherry, J.A. Hurricane Katrina sediment slowed elevation loss in subsiding brackish marshes of the Mississippi River delta. Wetlands 2009, 29, 2–15. [Google Scholar] [CrossRef]
- U.S. Energy Information Administration. Energy Information Administration: Maps: Layer Information for Interactive State Maps. Available online: https://www.eia.gov/maps/map_data/NaturalGas_ProcessingPlants_US_EIA.zip (accessed on 1 December 2017).
- U.S. Energy Information Administration. Maps: Layer Information for Interactive State Maps. Available online: https://www.eia.gov/maps/map_data/NaturalGas_UndergroundStorage_US_EIA.zip (accessed on 1 December 2017).
- U.S. Energy Information Administration. Maps: Layer Information for Interactive State Maps. Available online: https://www.eia.gov/maps/map_data/Petroleum_Refineries_US_EIA.zip (accessed on 1 December 2017).
- U.S. Energy Information Administration. Maps: Layer Information for Interactive State Maps. Available online: https://www.eia.gov/maps/map_data/PowerPlants_US_EIA.zip (accessed on 1 December 2017).
- LSU Center for Energy Studies. Mapsearch: Louisiana Natural Gas and Petroleum Pipelines Baton Rouge, 70803. 2013. Available online: https://www.lsu.edu/ces/energydata/index.php (accessed on 1 December 2017).
- Dismukes, D.E.; Narra, S. Identifying the vulnerabilities of working coasts supporting critical energy infrastructure. Water 2016, 8, 8. [Google Scholar] [CrossRef]
- Wiegman, A.R.; Day, J.W.; D’Elia, C.F.; Rutherford, J.S.; Morris, J.T.; Roy, E.D.; Lane, R.R.; Dismukes, D.E.; Snyder, B.F. Modeling impacts of sea-level rise, oil price, and management strategy on the costs of sustaining Mississippi delta marshes with hydraulic dredging. Sci. Total Environ. 2018, 618, 1547–1559. [Google Scholar] [CrossRef]
- Hopkinson, C.S.; Gosselink, J.G.; Parrando, R.T. Aboveground production of seven marsh plant species in coastal Louisiana. Ecology 1978, 59, 760–769. [Google Scholar] [CrossRef]
- Kirby, C.J.; Gosselink, J.G. Primary production in a Louisiana gulf coast spartina-alterniflora marsh. Ecology 1976, 57, 1052–1059. [Google Scholar] [CrossRef]
- Conner, W.H.; Day, J.W. Productivity and Composition of a Baldcypress-Water Tupelo Site and a Bottomland Hardwood Site in a Louisiana Swamp. Am. J. Bot. 1976, 63, 1354–1364. [Google Scholar] [CrossRef]
- Delaune, R.; Buresh, R.; Patrick, W. Relationship of soil properties to standing crop biomass of Spartina alterniflora in a Louisiana marsh. Estuar. Coast. Mar. Sci. 1979, 8, 477–487. [Google Scholar] [CrossRef]
- Rivera-Monroy, V.H.; Delaune, R.D.; Owens, A.B.; Visser, J.; White, J.R.; Twilley, R.R.; Hernandez-Trejo, H.; Benitez, J.A. Removal of physical materials from systems: Loss of space, area, and habitats. In Treatise on Estuarine and Coastal Science; Wolanski, E., McLusky, D.S., Eds.; Academic Press: Waltham, MA, USA, 2011; Volume 8, pp. 185–215. [Google Scholar]
- Blum, M.D.; Roberts, H.H. The Mississippi Delta Region: Past, Present, and Future. Annu. Rev. Earth Planet. Sci. 2012, 40, 655–683. [Google Scholar] [CrossRef]
- Bevington, A.E.; Twilley, R.R. Bevington Island Edge Morphodynamics along a Chronosequence in a Prograding Deltaic Floodplain Wetland. J. Coast. Res. 2018, 344, 806–817. [Google Scholar] [CrossRef]
- Hiatt, M.; Castañeda-Moya, E.; Twilley, R.; Hodges, B.R.; Passalacqua, P. Channel-Island Connectivity Affects Water Exposure Time Distributions in a Coastal River Delta. Water Resour. Res. 2018, 54, 2212–2232. [Google Scholar] [CrossRef]
- Blum, M.D.; Roberts, H.H. Drowning of the Mississippi Delta due to insufficient sediment supply and global sea-level rise. Nat. Geosci. 2009, 2, 488–491. [Google Scholar] [CrossRef]
- Morton, R.A.; Bernier, J.C.; Barras, J.A. Evidence of regional subsidence and associated interior wetland loss induced by hydrocarbon production, Gulf Coast region, USA. Environ. Earth Sci. 2006, 50, 261–274. [Google Scholar] [CrossRef]
- Visser, J.M.; Sasser, C.E.; Chabreck, R.H.; Linscombe, R.G. Marsh Vegetation Types of the Mississippi River Deltaic Plain. Estuaries 1998, 21, 818–828. [Google Scholar] [CrossRef]
- Snedden, G.A.; Steyer, G.D. Predictive occurrence models for coastal wetland plant communities: Delineating hydrologic response surfaces with multinomial logistic regression. Estuar. Coast. Shelf Sci. 2013, 118, 11–23. [Google Scholar] [CrossRef]
- Snedden, G.A. Patterning emergent marsh vegetation assemblages in coastal Louisiana, USA, with unsupervised artificial neural networks. Appl. Veg. Sci. 2019, 22, 213–229. [Google Scholar] [CrossRef]
- Mendelssohn, I.A.; McKee, K.L. Spartina Alterniflora Die-Back in Louisiana: Time-Course Investigation of Soil Waterlogging Effects. J. Ecol. 1988, 76, 509–521. [Google Scholar] [CrossRef]
- Ramsey, E.; Rangoonwala, A.; Chi, Z.; Jones, C.E.; Bannister, T. Marsh Dieback, loss, and recovery mapped with satellite optical, airborne polarimetric radar, and field data. Remote Sens. Environ. 2014, 152, 364–374. [Google Scholar] [CrossRef]
- Turner, R.E.; Swenson, E.M.; Milan, C.S.; Lee, J.M.; Oswald, T.A. Below-ground biomass in healthy and impaired salt marshes. Ecol. Res. 2004, 19, 29–35. [Google Scholar] [CrossRef]
- Mendelssohn, I.A.; Kuhn, N.L. Sediment subsidy: Effects on soil–plant responses in a rapidly submerging coastal salt marsh. Ecol. Eng. 2003, 21, 115–128. [Google Scholar] [CrossRef]
- La Peyre, M.K.; Gossman, B.; Piazza, B.P. Short- and long-term response of deteriorating brackish marshes and open-water ponds to sediment enhancement by thin-layer dredge disposal. Estuaries Coasts 2009, 32, 390–402. [Google Scholar] [CrossRef]
- Snedden, G.A.; Cretini, K.; Patton, B. Inundation and salinity impacts to above- and belowground productivity in Spartina patens and Spartina alterniflora in the Mississippi River deltaic plain: Implications for using river diversions as restoration tools. Ecol. Eng. 2015, 81, 133–139. [Google Scholar] [CrossRef]
- Tong, C.; Baustian, J.J.; Graham, S.A.; Mendelssohn, I.A. Salt marsh restoration with sediment-slurry application: Effects on benthic macroinvertebrates and associated soil–plant variables. Ecol. Eng. 2013, 51, 151–160. [Google Scholar] [CrossRef]
- Reed, D.J.; Cahoon, D.R. The relationship between marsh surface-topography, hydroperiod, and growth of spartina-alterniflora in a deteriorating Louisiana salt-marsh. J. Coast. Res. 1992, 8, 77–87. [Google Scholar]
- Darby, F.A.; Turner, R.E. Below- and aboveground spartina alterniflora production in a Louisiana salt marsh. Estuaries Coasts 2008, 31, 223–231. [Google Scholar] [CrossRef]
- White, D.A.; Weiss, T.E.; Trapani, J.M.; Thien, L.B. Productivity and decomposition of the dominant salt marsh plants in Louisiana. Ecology 1978, 59, 751–759. [Google Scholar] [CrossRef]
- Edwards, K.R.; Mills, K.P. Aboveground and belowground productivity of Spartina alterniflora (Smooth Cordgrass) in natural and created Louisiana salt marshes. Estuaries 2005, 28, 252–265. [Google Scholar] [CrossRef]
- Lin, Q.; Mendelssohn, I.A. Impacts and Recovery of the Deepwater Horizon Oil Spill on Vegetation Structure and Function of Coastal Salt Marshes in the Northern Gulf of Mexico. Environ. Sci. Technol. 2012, 46, 3737–3743. [Google Scholar] [CrossRef] [PubMed]
- Day, J.; Lane, R.; Moerschbaecher, M.; Delaune, R.; Mendelssohn, I.; Baustian, J.; Twilley, R. Vegetation and Soil Dynamics of a Louisiana Estuary Receiving Pulsed Mississippi River Water Following Hurricane Katrina. Chesap. Sci. 2013, 36, 665–682. [Google Scholar] [CrossRef]
- Kaswadji, R.F.; Gosselink, J.G.; Turner, R.E. Estimation of primary produdction using five different methods in a spartina alterniflora salt marsh. Wetl. Ecol. Manag. 1990, 1, 57–64. [Google Scholar] [CrossRef]
- Stagg, C.L.; Mendelssohn, I.A. Restoring Ecological Function to a Submerged Salt Marsh. Restor. Ecol. 2010, 18, 10–17. [Google Scholar] [CrossRef]
- McKee, K.L.; Rooth, J.E. Where temperate meets tropical: Multi-factorial effects of elevated CO2, nitrogen enrichment, and competition on a mangrove-salt marsh community. Glob. Chang. Biol. 2008, 14, 971–984. [Google Scholar] [CrossRef]
- Nyman, J.A.; Walters, R.J.; Delaune, R.D.; Patrick, W.H. Marsh vertical accretion via vegetative growth. Estuar. Coast. Shelf Sci. 2006, 69, 370–380. [Google Scholar] [CrossRef]
- Hopkinson, C.S.; Gosselink, J.G.; Parrondo, R.T. Production of Coastal Louisiana Marsh Plants Calculated from Phenometric Techniques. Ecology 1980, 61, 1091–1098. [Google Scholar] [CrossRef]
- Ewing, K.; McKee, K.; Mendelssohn, I.; Hester, M. A comparison of indicators of sublethal salinity stress in the salt marsh grass, Spartina patens (Ait.) Muhl. Aquat. Bot. 1995, 52, 59–74. [Google Scholar] [CrossRef]
- Pezeshki, S.; Delaune, R. Effects of soil Hypoxia and salinity on gas exchange and growth of Spartina patens. Mar. Ecol. Prog. Ser. 1993, 96, 75–81. [Google Scholar] [CrossRef]
- Graham, S.A.; Mendelssohn, I.A. Functional assessment of differential sediment slurry applications in a deteriorating brackish marsh. Ecol. Eng. 2013, 51, 264–274. [Google Scholar] [CrossRef]
- Taylor, K.L.; Grace, J.B. The effects of vertebrate herbivory on plant community structure in the coastal marshes of the Pearl River, Louisiana, USA. Wetlands 1995, 15, 68–73. [Google Scholar] [CrossRef]
- Johnson, L.A.; Foote, A. Vertebrate herbivory in managed coastal wetlands: A manipulative experiment. Aquat. Bot. 1997, 59, 17–32. [Google Scholar] [CrossRef]
- Gough, L.; Grace, J.B. Herbivore effects on plant species density at varying productivity levels. Ecology 1998, 79, 1586–1594. [Google Scholar] [CrossRef]
- Gabrey, S.W.; Afton, A.D. Plant Community Composition and Biomass in Gulf Coast Chenier Plain Marshes: Responses to Winter Burning and Structural Marsh Management. Environ. Manag. 2001, 27, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Spalding, E.A.; Hester, M.W. Interactive effects of hydrology and salinity on oligohaline plant species productivity: Implications of relative sea-level rise. Chesap. Sci. 2007, 30, 214–225. [Google Scholar] [CrossRef]
- Cramer, G.W.; Day, J.W.; Conner, W.H. Productivity of Four Marsh Sites Surrounding Lake Pontchartrain, Louisiana. Am. Midl. Nat. 1981, 106, 65–72. [Google Scholar] [CrossRef]
- Slocum, M.G.; Mendelssohn, I.A. Effects of three stressors on vegetation in an oligohaline marsh. Freshw. Biol. 2008, 53, 1783–1796. [Google Scholar] [CrossRef]
- Webb, E.C.; Mendelssohn, I.A. Factors Affecting Vegetation Dieback of an Oligohaline Marsh in Coastal Louisiana: Field Manipulation of Salinity and Submergence. Am. J. Bot. 1996, 83, 1429–1434. [Google Scholar] [CrossRef]
- Nolfo-Clements, L.E. Habitat Selection by Nutria in a Freshwater Louisiana Marsh. Southeast. Nat. 2012, 11, 183–204. [Google Scholar] [CrossRef]
- Howard, R.J.; Mendelssohn, I.A. Structure and composition of oligohaline marsh plant communities exposed to salinity pulses. Aquat. Bot. 2000, 68, 143–164. [Google Scholar] [CrossRef]
- Lin, Q.; Mendelssohn, I.A. A comparative investigation of the effects of south Louisiana crude oil on the vegetation of fresh, brackish and salt marshes. Mar. Pollut. Bull. 1996, 32, 202–209. [Google Scholar] [CrossRef]
- Cahoon, D.R.; White, D.A.; Lynch, J.C. Sediment infilling and wetland formation dynamics in an active crevasse splay of the Mississippi River delta. Geomorphology 2011, 131, 57–68. [Google Scholar] [CrossRef]
- Graham, S.A.; Mendelssohn, I.A. Coastal wetland stability maintained through counterbalancing accretionary responses to chronic nutrient enrichment. Ecology 2014, 95, 3271–3283. [Google Scholar] [CrossRef]
- Brewer, J.S.; Grace, J.B. Plant community structure in an oligohaline tidal marsh. Vegetatio 1990, 90, 93–107. [Google Scholar] [CrossRef]
- Fuller, D.A.; Sasser, C.E.; Johnson, W.B.; Gosselink, J.G. The effects of herboviry on vegetation on islands in atchafalaya bay, Louisiana. Wetlands 1985, 4, 105–114. [Google Scholar] [CrossRef]
- White, D.A.; Simmons, M.J. Productivity of the marshes at the mouth of the Pearl River, Louisiana. Castanea 1988, 53, 215–224. [Google Scholar]
- Baldwin, A.H.; Mendelssohn, I.A. Response of two oligohaline marsh communities to lethal and nonlethal disturbance. Oecologia 1998, 116, 543–555. [Google Scholar] [CrossRef]
- Ford, M.A.; Grace, J.B. The interactive effects of fire and herbivory on a coastal marsh in Louisiana. Wetlands 1998, 18, 1–8. [Google Scholar] [CrossRef]
- Tobin, E.J.; Visser, J.M.; Peterson, J.K.; Leberg, P.L. Small-Mammal Occupancy in Freshwater Marshes of Mandalay National Wildlife Refuge, Louisiana. Southeast. Nat. 2014, 13, 463–474. [Google Scholar]
- Martin, S.B.; Shaffer, G.P. Sagittaria Biomass Partitioning Relative to Salinity, Hydrologic Regime, and Substrate Type: Implications for Plant Distribution Patterns in Coastal Louisiana, United States. J. Coast. Res. 2005, 211, 167–174. [Google Scholar] [CrossRef]
- Shaffer, G.P.; Day, J.W.; Hunter, R.G.; Lane, R.R.; Lundberg, C.J.; Wood, W.B.; Hillmann, E.R.; Day, J.N.; Strickland, E.; Kandalepas, D. System response, nutria herbivory, and vegetation recovery of a wetland receiving secondarily-treated effluent in coastal Louisiana. Ecol. Eng. 2015, 79, 120–131. [Google Scholar] [CrossRef]
- Sasser, C.E.; Visser, J.M.; Evers, D.E.; Gosselink, J.G. The role of environmental variables on interannual variation in species composition and biomass in a subtropical minerotrophic floating marsh. Can. J. Bot. 1995, 73, 413–424. [Google Scholar] [CrossRef]
- Evers, D.E.; Sasser, C.E.; Gosselink, J.G.; Fuller, D.A.; Visser, J.M. The Impact of Vertebrate Herbivores on Wetland Vegetation in Atchafalaya Bay, Louisiana. Estuaries 1998, 21, 1–13. [Google Scholar] [CrossRef]
- Carpenter, K.; Sasser, C.E.; Visser, J.M.; Delaune, R.D. Sediment input into a floating freshwater marsh: Effects on soil properties, buoyancy, and plant biomass. Wetlands 2007, 27, 1016–1024. [Google Scholar] [CrossRef]
- Visser, J.M.; Sasser, C.E. The effect of environmental factors on floating fresh marsh end-of-season biomass. Aquat. Bot. 2009, 91, 205–212. [Google Scholar] [CrossRef]
- Visser, J.M.; Sasser, C.E.; Cade, B.S. The effect of multiple stressors on salt marsh end-of-season biomass. Chesap. Sci. 2006, 29, 328–339. [Google Scholar] [CrossRef]
- Izdepski, C.W.; Day, J.W.; Sasser, C.E.; Fry, B. Early floating marsh establishment and growth dynamics in a nutrient amended wetland in the lower Mississippi delta. Wetlands 2009, 29, 1004–1013. [Google Scholar] [CrossRef]
- McKee, K.L.; Mendelssohn, I.A. Response of a freshwater marsh plant community to increased salinity and increased water level. Aquat. Bot. 1989, 34, 301–316. [Google Scholar] [CrossRef]
- Swarzenski, C.M.; Doyle, T.W.; Fry, B.; Hargis, T.G. Biogeochemical response of organic-rich freshwater marshes in the Louisiana delta plain to chronic river water influx. Biogeochemistry 2008, 90, 49–63. [Google Scholar] [CrossRef]
- Willis, J.M.; Hester, M.W. Interactive effects of salinity, flooding, and soil type on Panicum hemitomon. Wetlands 2004, 24, 43–50. [Google Scholar] [CrossRef]
- Visser, J.M.; Peterson, J.K. The Effects of Flooding Duration and Salinity on Three Common Upper Estuary Plants. Wetlands 2015, 35, 625–631. [Google Scholar] [CrossRef]
- Roberts, H.H.; Delaune, R.D.; White, J.R.; Li, C.; Sasser, C.E.; Braud, D.; Weeks, E.; Khalil, S. Floods and Cold Front Passages: Impacts on Coastal Marshes in a River Diversion Setting (Wax Lake Delta Area, Louisiana). J. Coast. Res. 2015, 315, 1057–1068. [Google Scholar] [CrossRef]
- Holm, G.O.; Sasser, C.E. The management and ecology of the wetland grass, maidencane. J. Aquat. Plant Manag. 2008, 46, 51–60. [Google Scholar]
- Howard, R.J.; Rafferty, P.S. Clonal variation in response to salinity and flooding stress in four marsh macrophytes of the northern gulf of Mexico, USA. Environ. Exp. Bot. 2006, 56, 301–313. [Google Scholar] [CrossRef]
- Howard, R.J. Intraspecific variation in growth of marsh macrophytes in response to salinity and soil type: Implications for wetland restoration. Estuaries Coasts 2010, 33, 127–138. [Google Scholar] [CrossRef]
- Julien, M.H.; Skarratt, B.; Maywald, G.F. Potential geographical-distribution of alligator weed and its biological-control by agasicles hygrophila. J. Aquat. Plant Manag. 1995, 33, 55–60. [Google Scholar]
- Baldwin, A.H.; Mendelssohn, I.A. Effects of salinity and water level on coastal marshes: An experimental test of disturbance as a catalyst for vegetation change. Aquat. Bot. 1998, 61, 255–268. [Google Scholar] [CrossRef]
- Baustian, J.; Mendelssohn, I.; Lin, Q.; Rapp, J. In Situ Burning Restores the Ecological Function and Structure of an Oil-Impacted Coastal Marsh. Environ. Manag. 2010, 46, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Gough, L.; Grace, J.B. Effects of flooding, salinity and herbivory on coastal plant communities, Louisiana, United States. Oecologia 1998, 117, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Visser, J.M.; Steyer, G.D.; Shaffer, G.P.; Höppner, S.S.; Hester, M.W.; Reyes, E.; Keddy, P.; Mendelssohn, I.A.; Sasser, C.E.; Swarzenski, C. Habitat Switching Module, Chapter 9; Universtity of Louisiana at Lafayette: Baton Rouge, LA, USA, 2003; p. 319. [Google Scholar]
- Allen, J.A.; Chambers, J.L.; McKinney, D. Intraspecific variation in the response of Taxodium distichum seedlings to salinity. For. Ecol. Manag. 1994, 70, 203–214. [Google Scholar] [CrossRef]
- Conner, W.H.; Duberstein, J.A.; Day, J.W.; Hutchinson, S. Impacts of Changing Hydrology and Hurricanes on Forest Structure and Growth Along a Flooding/Elevation Gradient in a South Louisiana Forested Wetland from 1986 to 2009. Wetlands 2014, 34, 803–814. [Google Scholar] [CrossRef]
- Day, J.W.; Westphal, A.; Pratt, R.; Hyfield, E.; Rybczyk, J.; Kemp, G.P.; Day, J.N.; Marx, B. Effects of long-term municipal effluent discharge on the nutrient dynamics, productivity, and benthic community structure of a tidal freshwater forested wetland in Louisiana. Ecol. Eng. 2006, 27, 242–257. [Google Scholar] [CrossRef]
- Day, J.; Ko, J.-Y.; Rybczyk, J.; Sabins, D.; Bean, R.; Berthelot, G.; Brantley, C.; Cardoch, L.; Conner, W.; Day, J.; et al. The use of wetlands in the Mississippi Delta for wastewater assimilation: A review. Ocean. Coast. Manag. 2004, 47, 671–691. [Google Scholar] [CrossRef]
- Conner, W.H.; Day, J.W.; Slater, W.R. Bottomland hardwood productivity: Case study in a rapidly subsiding, Louisiana, USA, watershed. Wetl. Ecol. Manag. 1993, 2, 189–197. [Google Scholar] [CrossRef]
- Brantley, C.G.; Day, J.W.; Lane, R.R.; Hyfield, E.; Day, J.N.; Ko, J.-Y. Primary production, nutrient dynamics, and accretion of a coastal freshwater forested wetland assimilation system in Louisiana. Ecol. Eng. 2008, 34, 7–22. [Google Scholar] [CrossRef]
- Hunter, R.G.; Day, J.W.; Lane, R.R.; Lindsey, J.; Day, J.N.; Hunter, M.G. Impacts of secondarily treated municipal effluent on a freshwater forested wetland after 60 years of discharge. Wetlands 2009, 29, 363–371. [Google Scholar] [CrossRef]
- Conner, W.H.; Gosselink, J.G.; Parrondo, R.T. Comparison of the vegetation of three Louisiana swamp sites with different flooding regimes. Am. J. Bot. 1981, 68, 320–331. [Google Scholar] [CrossRef]
- Conner, W.H.; Day, J.W. Water Level Variability and Litterfall Productivity of Forested Freshwater Wetlands in Louisiana. Am. Midl. Nat. 1992, 128, 237–245. [Google Scholar] [CrossRef]
- DeAngelis, D.L.; Mooij, W.M. Individual-Based Modeling of Ecological and Evolutionary Processes. Annu. Rev. Ecol. Evol. Syst. 2005, 36, 147–168. [Google Scholar] [CrossRef]
- Grimm, V.; Railsback, S.F. Individual-Based Modeling and Ecology; Princeton Universtiy Press: Princenton, NJ, USA, 2005. [Google Scholar]
- Crookston, N.L.; Dixon, G.E. The forest vegetation simulator: A review of its structure, content, and applications. Comput. Electron. Agric. 2005, 49, 60–80. [Google Scholar] [CrossRef]
- Kirwan, M.L.; Murray, A.B. A coupled geomorphic and ecological model of tidal marsh evolution. Proc. Natl. Acad. Sci. USA 2007, 104, 6118–6122. [Google Scholar] [CrossRef] [PubMed]
- Chabreck, R.H. Marsh Zones and Vegetative Types in the Louisiana Coastal Marshes. Ph.D. Thesis, Louisiana State University, Baton Rouge, LA, USA, May 1970. [Google Scholar]
- Allison, M.A.; Ramirez, M.T.; Meselhe, E.A. Diversion of Mississippi River Water Downstream of New Orleans, Louisiana, USA to Maximize Sediment Capture and Ameliorate Coastal Land Loss. Water Resour. Manag. 2014, 28, 4113–4126. [Google Scholar] [CrossRef]
- Stagg, C.L.; Schoolmaster, D.R.; Piazza, S.C.; Snedden, G.; Steyer, G.D.; Fischenich, C.J.; McComas, R.W. A landscape-scale assessment of above- and belowground primary production in coastal wetlands: Implications for climate change-induced community shifts. Estuaries Coasts 2017, 40, 856–879. [Google Scholar] [CrossRef]
- Wang, H.Q.; Steyer, G.D.; Couvillion, B.R.; Beck, H.J.; Rybczyk, J.M.; Rivera-Monroy, V.H.; Krauss, K.W.; Visser, J.M. Predicting landscape effects of mississippi river diversions on soil organic carbon sequestration. Ecosphere 2018, 8. [Google Scholar] [CrossRef]
- Turner, R.E. Wetland Loss in the Northern Gulf of Mexico: Multiple Working Hypotheses. Estuaries 1997, 20, 1–13. [Google Scholar] [CrossRef]
- Safford, T.G.; Ulrich, J.D.; Hamilton, L.C. Public perceptions of the response to the Deepwater Horizon oil spill: Personal experiences, information sources, and social context. J. Environ. Manag. 2012, 113, 31–39. [Google Scholar] [CrossRef]
- Scaife, W.W.; Turner, R.E.; Costanza, R. Coastal Louisiana recent land loss and canal impacts. Environ. Manag. 1983, 7, 433–442. [Google Scholar] [CrossRef]
- McClenachan, G.; Tweel, A.W.; Turner, R.E. Effects of oil on the rate and trajectory of Louisiana marsh shoreline erosion. Environ. Res. Lett. 2013, 8, 044030. [Google Scholar] [CrossRef]
- Perrons, R.K. Assessing the damage caused by Deepwater Horizon: Not just another Exxon Valdez. Mar. Pollut. Bull. 2013, 71, 20–22. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.-Y.; Day, J.W. A review of ecological impacts of oil and gas development on coastal ecosystems in the Mississippi Delta. Ocean. Coast. Manag. 2004, 47, 597–623. [Google Scholar] [CrossRef]
- Turner, R.E.; McClenachan, G.; Tweel, A.W. Islands in the oil: Quantifying salt marsh shoreline erosion after the Deepwater Horizon oiling. Mar. Pollut. Bull. 2016, 110, 316–323. [Google Scholar] [CrossRef]
- Couvillion, B.R.; Barras, J.A.; Steyer, G.D.; Sleavin, W.; Fischer, M.; Beck, H.; Trahan, N.; Griffin, B.; Heckman, D. Land Area Change in Coastal Louisiana from 1932 to 2010; U.S. Geological Survey: Reston, VA, USA, 2011; p. 12.
- Visser, J.M.; Sasser, C.E.; Chabreck, R.H.; Linscombe, R.G. Long-term vegetation change in Louisiana tidal marshes, 1968–1992. Wetlands 1999, 19, 168–175. [Google Scholar] [CrossRef]
- Barras, J.A.; Beville, S.; Britsch, D.; Hartley, S.; Hawes, S.; Johnston, J.; Kemp, P.; Kinler, Q.; Martucci, A.; Porthouse, J.; et al. Historical and Projected Coastal Louisiana Land Changes: 1978–2050; Usgs Open File Report 03-334; USGS: Baton Rouge, MS, USA, 2003.
- Britsch, L.D.; Dunbar, J.B. Land loss rates-Louisiana coastal plain. J. Coast. Res. 1993, 9, 324–338. [Google Scholar]
- Beyer, J.; Trannum, H.C.; Bakke, T.; Hodson, P.V.; Collier, T.K. Environmental effects of the Deepwater Horizon oil spill: A review. Mar. Pollut. Bull. 2016, 110, 28–51. [Google Scholar] [CrossRef]
- Mendelssohn, I.A.; Andersen, G.L.; Baltz, D.M.; Caffey, R.H.; Carman, K.R.; Fleeger, J.W.; Joye, S.B.; Lin, Q.; Maltby, E.; Overton, E.B.; et al. Oil Impacts on Coastal Wetlands: Implications for the Mississippi River Delta Ecosystem after the Deepwater Horizon Oil Spill. Bioscience 2012, 62, 562–574. [Google Scholar] [CrossRef]
- Joye, S.B.; Bracco, A.; Özgökmen, T.M.; Chanton, J.P.; Grosell, M.; Macdonald, I.R.; Cordes, E.E.; Montoya, J.P.; Passow, U. The Gulf of Mexico ecosystem, six years after the Macondo oil well blowout. Deep. Sea Res. Part II Top. Stud. Oceanogr. 2016, 129, 4–19. [Google Scholar] [CrossRef]
- Hester, M.W.; Willis, J.M.; Rouhani, S.; Steinhoff, M.A.; Baker, M.C. Impacts of the Deepwater Horizon oil spill on the salt marsh vegetation of Louisiana. Environ. Pollut. 2016, 216, 361–370. [Google Scholar] [CrossRef]
- CPRA. Louisiana’s Comprehensive master Plan for a Sustainable Coast, June 2017. Available online: http://coastal.la.gov/our-plan/2017-coastal-master-plan/ (accessed on 29 September 2019).
- Jankowski, K.L.; Tornqvist, T.E.; Fernandes, A.M. Vulnerability of Louisiana’s coastal wetlands to present-day rates of relative sea-level rise. Nat. Commun. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Klemas, V. Remote Sensing of Wetlands: Case Studies Comparing Practical Techniques. J. Coast. Res. 2011, 27, 418–427. [Google Scholar]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivera-Monroy, V.H.; Elliton, C.; Narra, S.; Meselhe, E.; Zhao, X.; White, E.; Sasser, C.E.; Visser, J.M.; Meng, X.; Wang, H.; et al. Wetland Biomass and Productivity in Coastal Louisiana: Base Line Data (1976–2015) and Knowledge Gaps for the Development of Spatially Explicit Models for Ecosystem Restoration and Rehabilitation Initiatives. Water 2019, 11, 2054. https://doi.org/10.3390/w11102054
Rivera-Monroy VH, Elliton C, Narra S, Meselhe E, Zhao X, White E, Sasser CE, Visser JM, Meng X, Wang H, et al. Wetland Biomass and Productivity in Coastal Louisiana: Base Line Data (1976–2015) and Knowledge Gaps for the Development of Spatially Explicit Models for Ecosystem Restoration and Rehabilitation Initiatives. Water. 2019; 11(10):2054. https://doi.org/10.3390/w11102054
Chicago/Turabian StyleRivera-Monroy, Victor H., Courtney Elliton, Siddhartha Narra, Ehab Meselhe, Xiaochen Zhao, Eric White, Charles E. Sasser, Jenneke M. Visser, Xuelian Meng, Hongqing Wang, and et al. 2019. "Wetland Biomass and Productivity in Coastal Louisiana: Base Line Data (1976–2015) and Knowledge Gaps for the Development of Spatially Explicit Models for Ecosystem Restoration and Rehabilitation Initiatives" Water 11, no. 10: 2054. https://doi.org/10.3390/w11102054
APA StyleRivera-Monroy, V. H., Elliton, C., Narra, S., Meselhe, E., Zhao, X., White, E., Sasser, C. E., Visser, J. M., Meng, X., Wang, H., Xue, Z., & Jaramillo, F. (2019). Wetland Biomass and Productivity in Coastal Louisiana: Base Line Data (1976–2015) and Knowledge Gaps for the Development of Spatially Explicit Models for Ecosystem Restoration and Rehabilitation Initiatives. Water, 11(10), 2054. https://doi.org/10.3390/w11102054






