Optical Properties of Dissolved Organic Matter and Controlling Factors in Dianchi Lake Waters
Abstract
1. Introduction
2. Materials and Methods
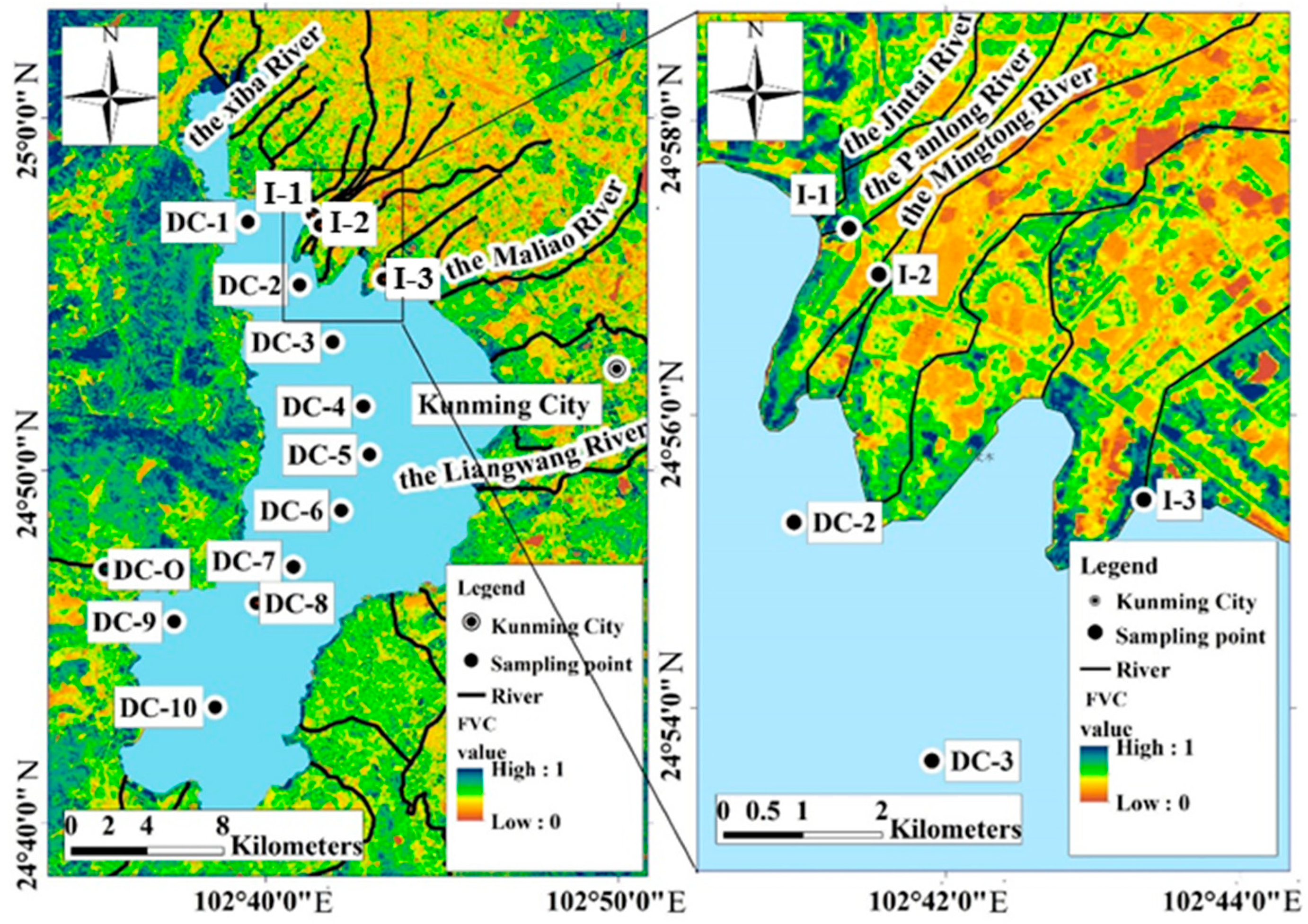
2.1. Study Area
2.2. Sample Collection and Field Measurements
2.3. Analyses of Inorganic and Organic Compounds in Water and Porewater
3. Results and Discussion
3.1. Characterization of Water Chemistry and DOC in the Dianchi Lake Waters
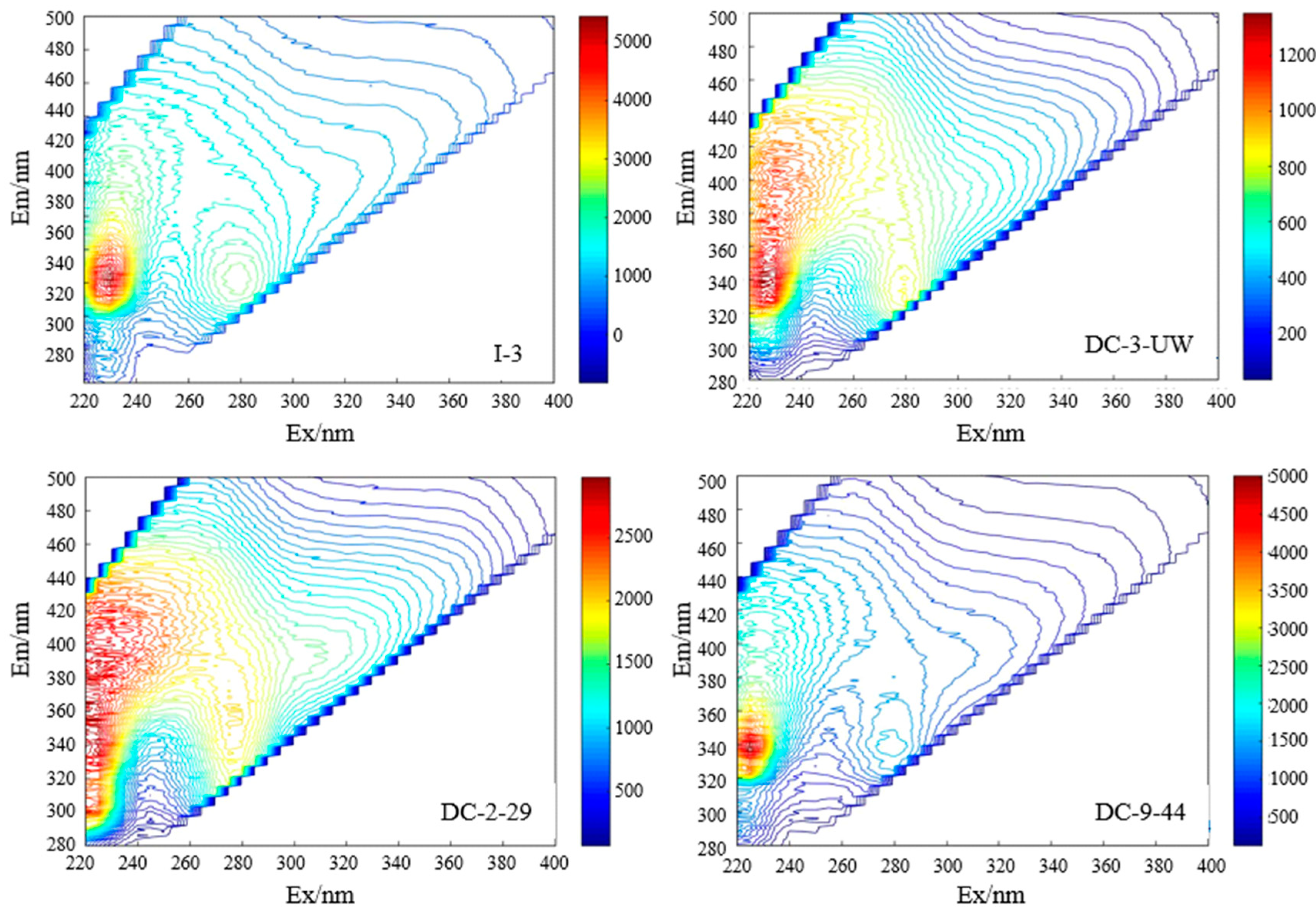
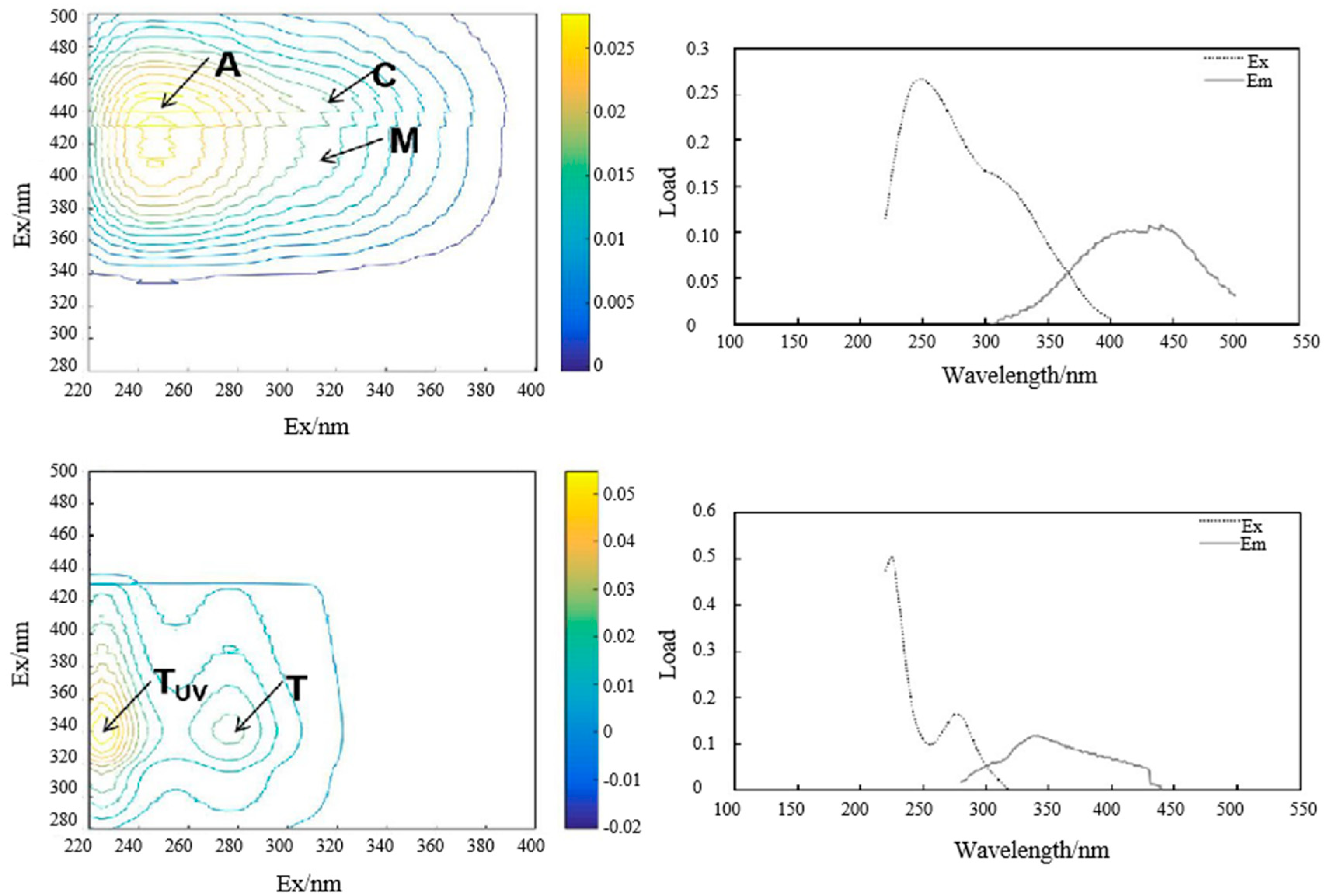
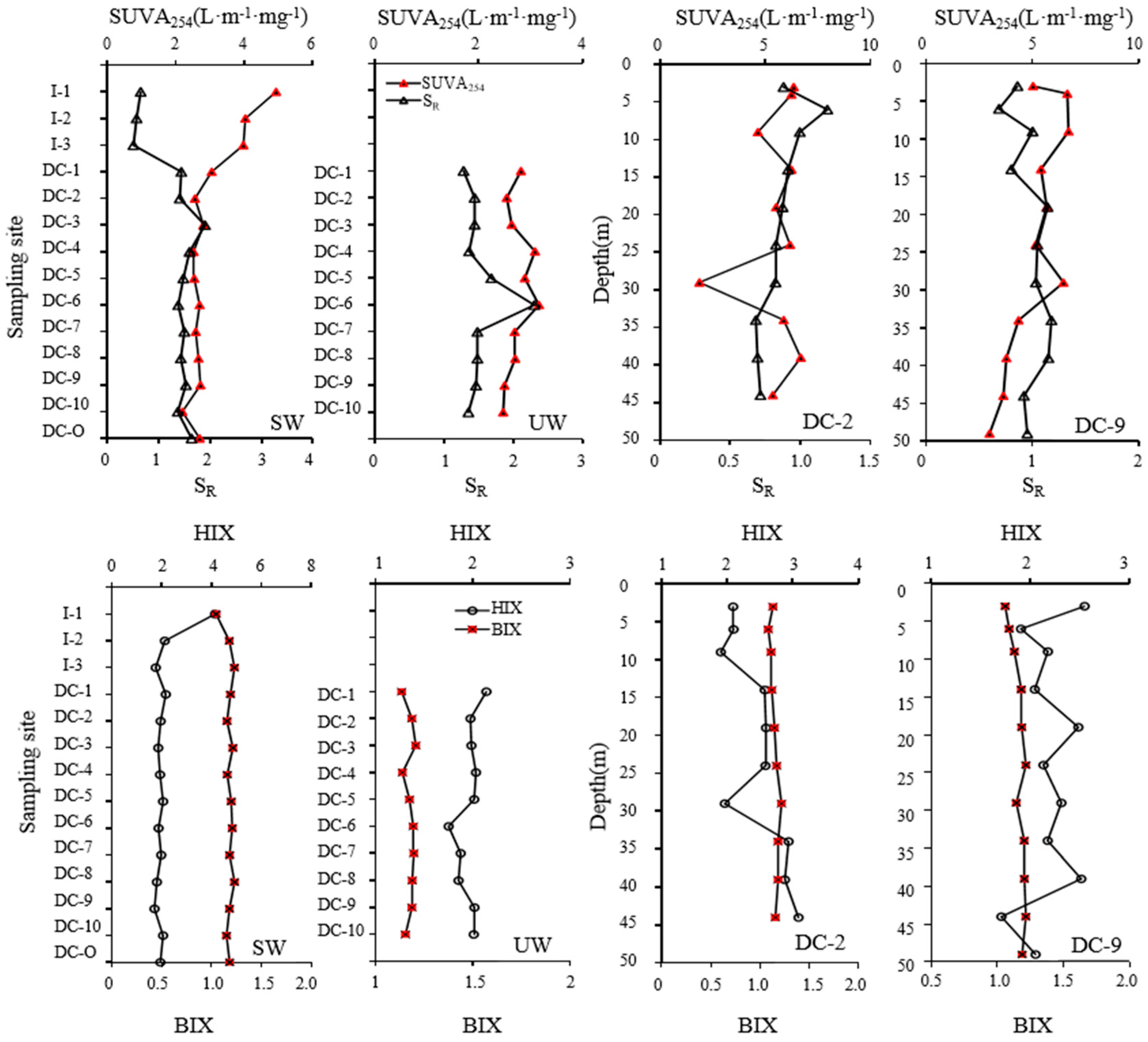
3.2. Sources of DOM Based on Spectral and Fluorescent Characteristics
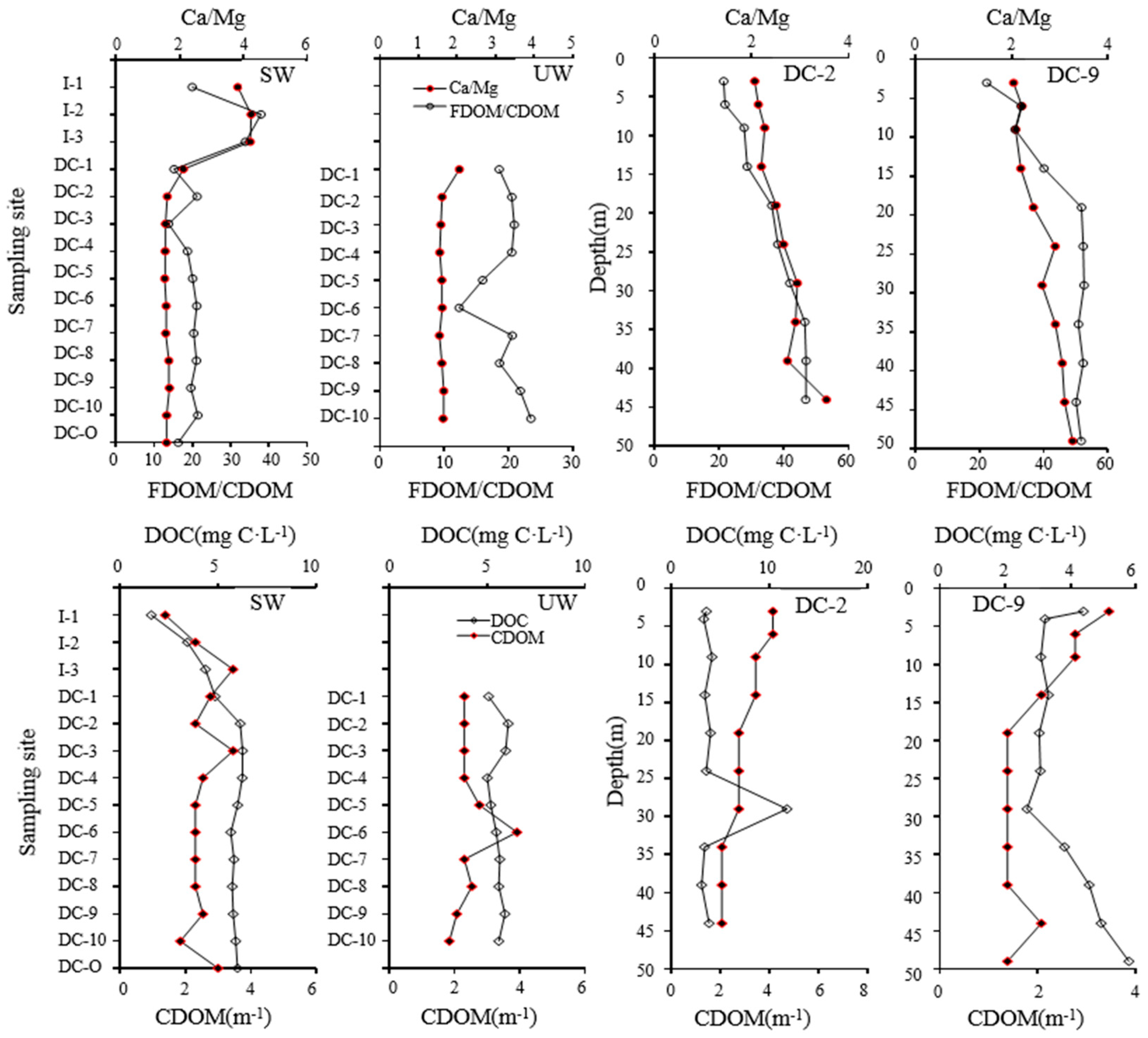
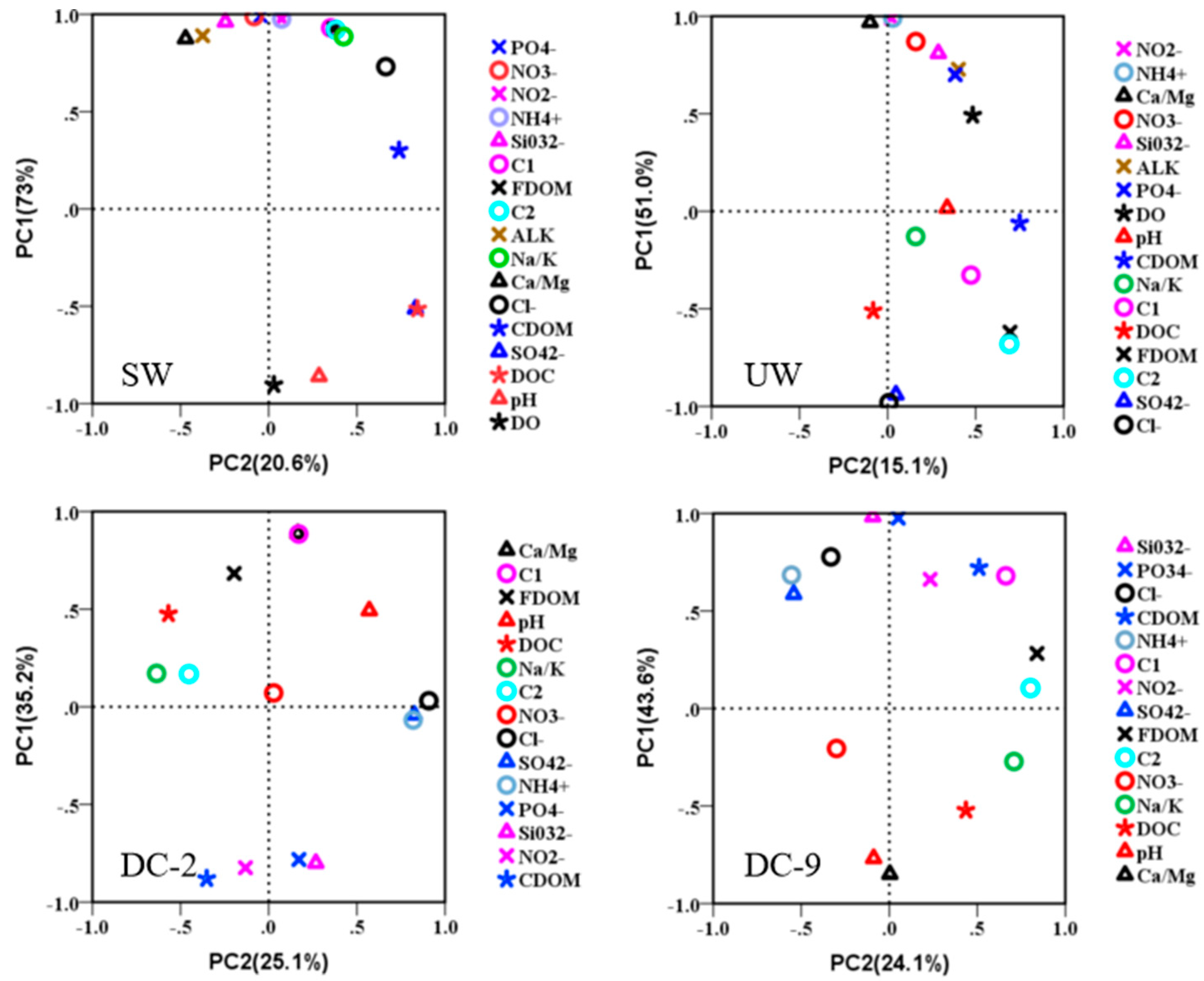
3.3. Analysis of Relationship between Water Chemistry and Characteristic of DOM in Dianchi Lake Waters
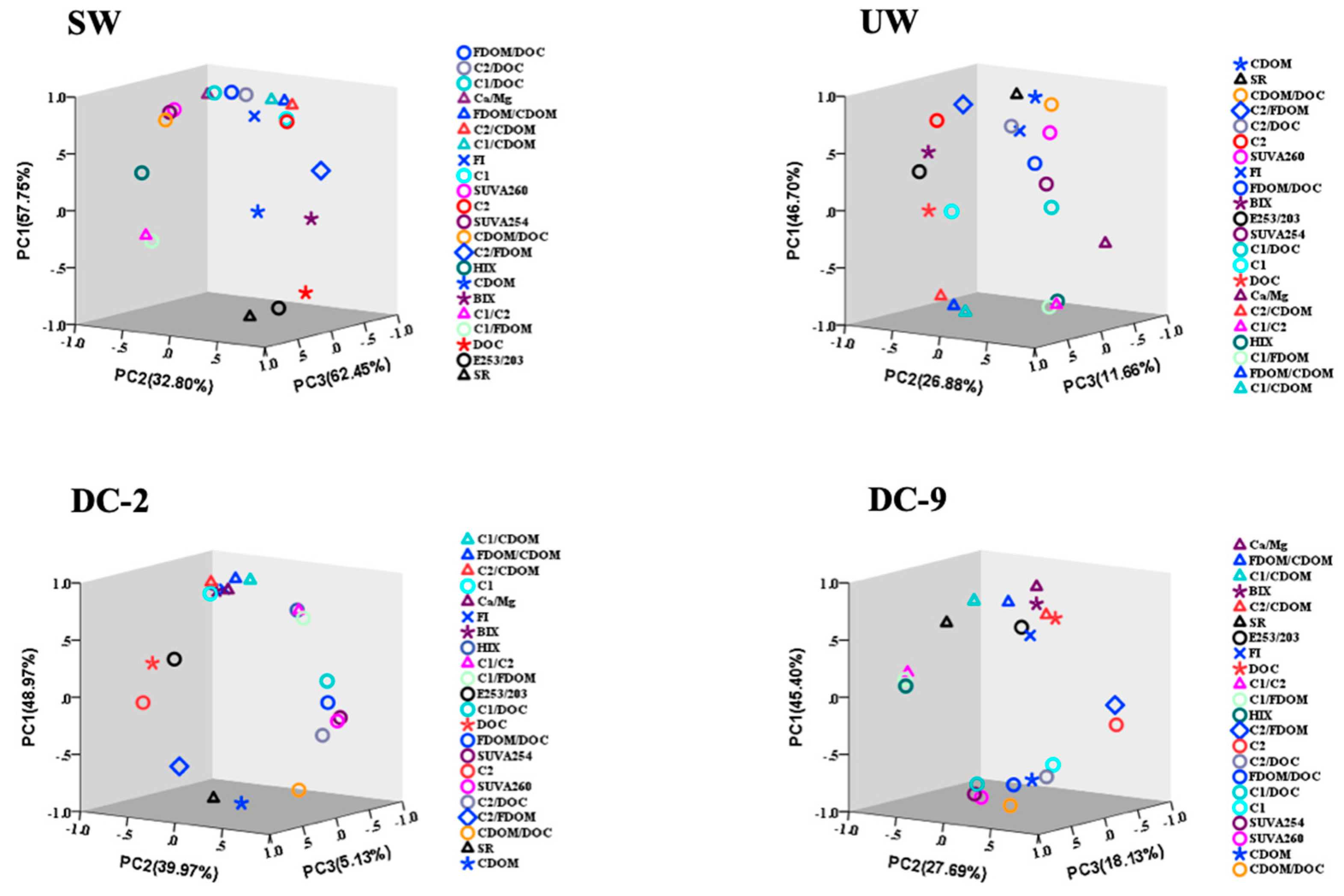
3.4. Optical Characterization of DOM for Tracing Moieties Conversion in Dianchi Lake Waters
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thomas, J.D. The role of dissolved organic matter, particularly free amino acids and humic substances, in freshwater ecosystems. Freshw. Biol. 1997, 38, 1–36. [Google Scholar] [CrossRef]
- Gao, Z.; Guéguen, C. Size distribution of absorbing and fluorescing DOM in Beaufort Sea, Canada Basin. Deep Sea Res. Part I Oceanogr. Res. Pap. 2017, 121, 30–37. [Google Scholar] [CrossRef]
- Chen, B.; Huang, W.; Ma, S.; Feng, M.; Liu, C.; Gu, X.; Chen, K. Characterization of Chromophoric Dissolved Organic Matter in the Littoral Zones of Eutrophic Lakes Taihu and Hongze during the Algal Bloom Season. Water 2018, 10, 861. [Google Scholar] [CrossRef]
- Kitis, M.; Karanfil, T.; Kilduff, J.E.; Wigton, A. The reactivity of natural organic matter to disinfection by-products formation and its relation to specific ultraviolet absorbance. Water Sci. Technol. 2001, 43, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Egeberg, P.K.; Alberts, J.J. Determination of hydrophobicity of NOM by RP-HPLC, and the effect of pH and ionic strength. Water Res. 2002, 36, 4997–5004. [Google Scholar] [CrossRef]
- Fellman, J.B.; Hood, E.; Spencer, R.G. Fluorescence spectroscopy opens new windows into dissolved organic matter dynamics in freshwater ecosystems: A review. Limnol. Oceanogr. 2010, 55, 2452–2462. [Google Scholar] [CrossRef]
- Li, S.; Zhang, J.; Mu, G.; Ju, H.; Wang, R.; Li, D.; Shabbir, A.H. Spatiotemporal Characterization of Chromophoric Dissolved Organic Matter (CDOM) and CDOM-DOC Relationships for Highly Polluted Rivers. Water 2016, 8, 399. [Google Scholar] [CrossRef]
- Manenti, S.; Todeschini, S.; Collivignarelli, M.C.; Abbà, A. Integrated RTD—CFD Hydrodynamic Analysis for Performance Assessment of Activated Sludge Reactors. Environ. Process. 2018, 5, 23–42. [Google Scholar] [CrossRef]
- Gu, B.; Schmitt, J.; Chen, Z.; Liang, L.; McCarthy, J.F. Adsorption and desorption of different organic matter fractions on iron oxide. Geochim. Cosmochim. Acta 1995, 59, 219–229. [Google Scholar] [CrossRef]
- Chin, Y.P.; Traina, S.J.; Swank, C.R.; Backhus, D. Abundance and properties of dissolved organic matter in pore waters of a freshwater wetland. Limnol. Oceanogr. 1998, 43, 1287–1296. [Google Scholar] [CrossRef]
- Coelho, C.; Heim, B.; Foerster, S.; Brosinsky, A.; Carlos de Araújo, J. In Situ and Satellite Observation of CDOM and Chlorophyll-a Dynamics in Small Water Surface Reservoirs in the Brazilian Semiarid Region. Water 2017, 9, 913. [Google Scholar] [CrossRef]
- Shang, Y.; Song, K.; Jacinthe, P.A.; Wen, Z.; Lyu, L.; Fang, C.; Liu, G. Characterization of CDOM in reservoirs and its linkage to trophic status assessment across China using spectroscopic analysis. J. Hydrol. 2019, 576, 1–11. [Google Scholar] [CrossRef]
- Thurman, E.M. Organic Geochemistry of Natural Waters. Springer Science & Business Media: Dordrecht, The Netherlands, 2012; Volume 2. [Google Scholar]
- Twardowski, M.S.; Boss, E.; Sullivan, J.M.; Donaghay, P.L. Modeling the spectral shape of absorption by chromophoric dissolved organic matter. Mar. Chem. 2004, 89, 69–88. [Google Scholar] [CrossRef]
- Burgos, W.D.; Pisutpaisal, N.; Tuntoolavest, M.; Chorover, J.; Unz, R.F. Biodegradation of 1-naphthol in the presence of humic acid. Environ. Eng. Sci. 2000, 17, 343–351. [Google Scholar] [CrossRef]
- Santos, L.; Santos, E.B.H.; Dias, J.M.; Cunha, A.; Almeida, A. Photochemical and microbial alterations of DOM spectroscopic properties in the estuarine system Ria de Aveiro. Photochem. Photobiol. Sci. 2014, 13, 1146–1159. [Google Scholar] [CrossRef] [PubMed]
- Helms, J.R.; Stubbins, A.; Ritchie, J.D.; Minor, E.C.; Kieber, D.J. Absorption spectral slopes and slope ratios as indicators of molecular weight, source and photobleaching of chromophoric dissolved organic matter. Limnol. Oceanogr. 2008, 53, 955–969. [Google Scholar] [CrossRef]
- Weishaar, J.L.; Aiken, G.R.; Bergamaschi, B.A.; Fram, M.S.; Fujii, R.; Mopper, K. Evaluation of specific ultraviolet absorbance as an indicator of the chemical composition and reactivity of dissolved organic carbon. Environ. Sci. Technol. 2003, 37, 4702–4708. [Google Scholar] [CrossRef] [PubMed]
- Mostofa, K.M.G.; Liu, C.Q.; Feng, X.B.; Yoshioka, T.; Vione, D.; Pan, X.L.; Wu, F.C. Complexation of Dissolved Organic Matter with Trace Metal Ions in Natural Waters. In Photobiogeochemistry of Organic Matter; Mostofa, K.M.G., Yoshioka, T., Mottaleb, M.A., Vione, D., Eds.; Springer: Heidelberg, Germany, 2013; pp. 769–849. [Google Scholar]
- Sun, H.W.; Yan, Q.S. Influence of Fenton oxidation on soil organic matter and its sorption and desorption of pyrene. J. Hazard. Mater. 2007, 144, 164–170. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Lee, J.H.; Hur, J. Anthropogenic signature of sediment organic matter probed by UV–Visible and fluorescence spectroscopy and the association with heavy metal enrichment. Chemosphere 2016, 150, 184–193. [Google Scholar] [CrossRef]
- Hou, G.; Song, L.; Liu, J.; Xiao, B.; Liu, Y. Modeling of cyanobacterial blooms in hypereutrophic Lake Dianchi, China. J. Freshw. Ecol. 2004, 19, 623–629. [Google Scholar] [CrossRef]
- Gong, Z.J.; Li, Y.L.; Shen, J.; Xie, P. Diatom community succession in the recent history of a eutrophic Yunnan Plateau lake, Lake Dianchi, in subtropical China. Limnology 2009, 10, 247–253. [Google Scholar] [CrossRef]
- Huang, B.; Wang, B.; Ren, D.; Jin, W.; Liu, J.; Peng, J.; Pan, X. Occurrence, removal and bioaccumulation of steroid estrogens in Dianchi Lake catchment, China. Environ. Int. 2013, 59, 262–273. [Google Scholar] [CrossRef] [PubMed]
- Stedmon, C.A.; Markager, S.; Bro, R. Tracing dissolved organic matter in aquatic environments using a new approach to fluorescence spectroscopy. Mar. Chem. 2003, 82, 239–254. [Google Scholar] [CrossRef]
- McKnight, D.M.; Boyer, E.W.; Westerhoff, P.K.; Doran, P.T.; Kulbe, T.; Andersen, D.T. Spectrofluorometric characterization of dissolved organic matter for indication of precursor organic material and aromaticity. Limnol. Oceanogr. 2001, 46, 38–48. [Google Scholar] [CrossRef]
- Yang, K.; Yu, Z.; Luo, Y.; Yang, Y.; Zhao, L.; Zhou, X. Spatial and temporal variations in the relationship between lake water surface temperatures and water quality-A case study of Dianchi Lake. Sci. Total Environ. 2018, 624, 859–871. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Li, S.L.; Liu, J.; Ding, H.; Sun, X.L.; Xu, S.; Wang, T.J.; Ellam, R.M.; Liu, C.Q. Climate variability controls on CO2 consumption fluxes and carbon dynamics for monsoonal rivers: Evidence from Xijiang river, Southwest China. J. Geophys. Res. Biogeosci. 2018, 123, 2553–2567. [Google Scholar] [CrossRef]
- Sundelin, B.; Eriksson, A.K. Mobility and bioavailability of trace metals in sulfidic coastal sediments. Environ. Toxicol. Chem. Int. J. 2001, 20, 748–756. [Google Scholar] [CrossRef]
- Zhu, W.Z.; Zhang, H.H.; Zhang, J.; Yang, G.P. Seasonal variation in chromophoric dissolved organic matter and relationships among fluorescent components, absorption coefficients and dissolved organic carbon in the Bohai Sea, the Yellow Sea and the East China Sea. J. Mar. Syst. 2018, 180, 9–23. [Google Scholar] [CrossRef]
- Huguet, A.; Vacher, L.; Relexans, S.; Saubusse, S.; Froidefond, J.M.; Parlanti, E. Properties of fluorescent dissolved organic matter in the Gironde Estuary. Org. Geochem. 2009, 40, 706–719. [Google Scholar] [CrossRef]
- Coble, P.G. Characterization of marine and terrestrial DOM in seawater using excitation-emission matrix spectroscopy. Mar. Chem. 1996, 51, 325–346. [Google Scholar] [CrossRef]
- He, X.S.; Xi, B.D.; Li, X.; Pan, H.W.; An, D.; Bai, S.G.; Cui, D.Y. Fluorescence excitation–emission matrix spectra coupled with parallel factor and regional integration analysis to characterize organic matter humification. Chemosphere 2013, 93, 2208–2215. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Guo, H.; Zheng, H.; Xiu, W.; He, W.; Ding, Q. Roles of different molecular weights of dissolved organic matter in arsenic enrichment in groundwater: Evidences from ultrafiltration and EEM-PARAFAC. Appl. Geochem. 2019, 104, 124–134. [Google Scholar] [CrossRef]
- Ishii, S.K.; Boyer, T.H. Behavior of reoccurring PARAFAC components in fluorescent dissolved organic matter in natural and engineered systems: A critical review. Environ. Sci. Technol. 2012, 46, 2006–2017. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, L.; Stedmon, C.A.; Kragh, T.; Markager, S.; Middelboe, M.; Søndergaard, M. Global trends in the fluorescence characteristics and distribution of marine dissolved organic matter. Mar. Chem. 2011, 126, 139–148. [Google Scholar] [CrossRef]
- Bridgeman, J.; Bieroza, M.; Baker, A. The application of fluorescence spectroscopy to organic matter characterization in drinking water treatment. Rev. Environ. Sci. Bio Technol. 2011, 10, 277–290. [Google Scholar] [CrossRef]
- Wu, F.C.; Tanoue, E. Tryptophan in the sediments of lakes from Southwestern China Plateau. Chem. Geol. 2002, 184, 139–149. [Google Scholar] [CrossRef]
- Azam, F.; Malfatti, F. Microbial structuring of marine ecosystems. Nat. Rev. Microbiol. 2007, 5, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; LeBoeuf, E.J.; Dai, S.; Gu, B. Fluorescence spectroscopic studies of natural organic matter fractions. Chemosphere 2003, 50, 639–647. [Google Scholar] [CrossRef]
- Koopmans, G.F.; Groenenberg, J.E. Effects of soil oven-drying on concentrations and speciation of trace metals and dissolved organic matter in soil solution extracts of sandy soils. Geoderma 2011, 161, 147–158. [Google Scholar] [CrossRef]
- Lu, Y.; Allen, H.E. Characterization of copper complexation with natural dissolved organic matter (DOM)—link to acidic moieties of DOM and competition by Ca and Mg. Water Res. 2002, 36, 5083–5101. [Google Scholar] [CrossRef]
- Playter, T.; Konhauser, K.; Owttrim, G.; Hodgson, C.; Warchola, T.; Mloszewska, A.M.; Gingras, M. Microbe-clay interactions as a mechanism for the preservation of organic matter and trace metal biosignatures in black shales. Chem. Geol. 2017, 459, 75–90. [Google Scholar] [CrossRef]
- Gur-Reznik, S.; Katz, I.; Dosoretz, C.G. Removal of dissolved organic matter by granular-activated carbon adsorption as a pretreatment to reverse osmosis of membrane bioreactor effluents. Water Res. 2008, 42, 1595–1605. [Google Scholar] [CrossRef] [PubMed]
| Station | DOC mg C·L−1 | FDOM QSU | CDOM m−1 | C1 | C2 | C1/FDOM | C1/CDOM QSU·m | C2/CDOM QSU·m | SUVA254 Lm−1 (mg C)−1 | SUVA260 Lm−1 (mg C)−1 | FI | HIX | BIX |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| I-1 | 1.59 | 27.47 | 1.38 | 16.17 | 11.30 | 0.59 | 11.72 | 8.18 | 4.92 | 4.63 | 1.87 | 4.14 | 1.04 |
| I-2 | 3.43 | 87.24 | 2.30 | 41.49 | 45.75 | 0.48 | 18.02 | 19.86 | 4.03 | 4.03 | 2.09 | 2.12 | 1.18 |
| I-3 | 4.35 | 116.61 | 3.45 | 50.92 | 65.69 | 0.44 | 14.76 | 19.04 | 3.97 | 3.71 | 2.07 | 1.76 | 1.23 |
| DC-O | 5.98 | 48.61 | 2.99 | 24.53 | 24.07 | 0.50 | 8.19 | 8.04 | 2.70 | 2.54 | 1.88 | 1.95 | 1.18 |
| SW | 5.25 | 53.12 | 2.48 | 26.47 | 26.65 | 0.51 | 10.79 | 10.63 | 3.00 | 2.83 | 1.91 | 2.10 | 1.18 |
| UW | 5.55 | 46.12 | 2.46 | 23.80 | 22.32 | 0.52 | 10.01 | 9.33 | 2.60 | 2.58 | 1.90 | 1.96 | 1.18 |
| DC-2 | 4.40 | 98.52 | 2.97 | 54.70 | 43.82 | 0.56 | 19.99 | 15.61 | 5.50 | 4.89 | 2.06 | 2.47 | 1.15 |
| DC-9 | 3.81 | 78.98 | 1.94 | 41.19 | 37.79 | 0.53 | 23.25 | 21.05 | 5.08 | 4.41 | 2.06 | 2.19 | 1.17 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, M.; Wu, F.; Yi, Y.; Han, Z.; Wang, Z. Optical Properties of Dissolved Organic Matter and Controlling Factors in Dianchi Lake Waters. Water 2019, 11, 1967. https://doi.org/10.3390/w11101967
Xiao M, Wu F, Yi Y, Han Z, Wang Z. Optical Properties of Dissolved Organic Matter and Controlling Factors in Dianchi Lake Waters. Water. 2019; 11(10):1967. https://doi.org/10.3390/w11101967
Chicago/Turabian StyleXiao, Min, Fengchang Wu, Yuanbi Yi, Zenglei Han, and Zhongliang Wang. 2019. "Optical Properties of Dissolved Organic Matter and Controlling Factors in Dianchi Lake Waters" Water 11, no. 10: 1967. https://doi.org/10.3390/w11101967
APA StyleXiao, M., Wu, F., Yi, Y., Han, Z., & Wang, Z. (2019). Optical Properties of Dissolved Organic Matter and Controlling Factors in Dianchi Lake Waters. Water, 11(10), 1967. https://doi.org/10.3390/w11101967




