1. Introduction
Microorganisms play important roles in freshwater ecosystems [
1]. Some bacteria are decomposers, breaking down organic matter into nitrate, phosphate, and other essential nutrients [
1]. The diversity and abundance of microorganisms in lakes can drastically change the water quality of a freshwater system [
2]. Cyanobacteria obtain their energy from photosynthesis and provide an important link in the food chain as they introduce oxygen and energy from the sun’s light to freshwater systems [
1].
Freshwater lakes are an important source of microbial ice nucleators—microorganisms that facilitate the heterogeneous formation of ice at temperatures above −8 °C [
3]. Pietsch et al. [
3] collected strains of the bacterium
Pseudomonas syringae from the surface of a freshwater lake in Virginia at different locations, and reported significantly more ice nucleation active (INA or ice+) strains in the fall and the winter compared to the spring. Ice-nucleating strains of
P. syringae may aerosolize at greater rates than those that are not ice nucleators [
4], and there could be a potential role for these ice nucleating strains of bacteria in precipitation processes [
5,
6]. The presence of biological particles has been reported in clouds and could impact precipitation at concentrations high enough to catalyze heterogeneous ice nucleation [
7,
8].
Water samples have traditionally been collected using devices such as Nansen bottles [
9], Van Dorn samplers [
10], or syringes [
11]. These devices are usually actuated using a messenger dropped from the surface on the line, a bottom-induced trigger, or a powered servo (for large sampling cassettes). Recently, there has been an increasing interest in the development and use of unmanned robotic systems for collecting and characterizing samples in aquatic environments. Powers et al. [
12] developed an unmanned surface vehicle (USV) to collect microorganisms at two different depths in a freshwater lake. This same USV platform was modified to collect microorganisms in aerosols above freshwater and saltwater environments [
13].
New technologies with unmanned aerial systems (UASs, or drones) may assist in collecting water samples in remote areas, hard-to-reach environments, and in settings where hazardous agents may be present in the environment. Ore et al. [
14] developed an autonomous drone platform for collecting water samples in aquatic environments. This system used a pump, a short hose, and three 25 mL vials that were filled by a rotating nozzle at the end of the hose. Koparan et al. [
15] designed a small 3D-printed Van Dorn sampler for collecting water samples with a drone. This device was triggered by a messenger that was dropped down a cable from the drone. One of the limitations of both of these systems is the potential for cross-sample contamination, since the same device was used for collecting multiple samples. Consequently, new drone water sampling systems are needed to collect water samples without the potential for contamination across samples. Such drone water-sampling systems would be useful for collecting microorganisms that could be causal agents of disease.
In this manuscript, we report the development of a system to collect water samples from the surface of lakes for microbiological analyses using a 3D-printed sampling device tethered to a drone (DOWSE, DrOne Water Sampling SystEm). The sampling device was designed around a sterile 50 mL conical tube, enabling the collection and characterization of microbes without the potential for cross-contamination across samples. This sampling device is an extension of the traditional methods for water sampling (e.g., Nansen and Van Dorn samplers), without the need for an actuated mechanism to collect samples at the water surface [
15]. We hypothesized that (1) concentrations of culturable microorganisms in freshwater lakes would vary with distance from the shore and (2) the frequency of ice-nucleating strains of
Pseudomonas spp. would vary across different freshwater lakes. Our first hypothesis was based on the observation by Powers et al. [
12] that there were significantly different concentrations of bacteria at different locations in Claytor Lake, Virginia. Our second hypothesis was based on the observations of Pietsch et al. [
3] that the frequency of ice nucleating microbes varied with season, suggesting a temperature-dependent association with this phenotype (i.e., lakes with different temperatures could have different percentages of ice nucleating microorganisms). These hypotheses need to be tested to inform appropriate sampling strategies for microbes at the water surface, and to better understand the diversity and function of ice nucleating microorganisms in aquatic environments. To test these hypotheses, we used the DOWSE to collect surface water samples at different distances from the shore (1, 25, and 50 m) at eight different freshwater lakes in Austria in June 2018. Water samples were filtered, and microorganisms were cultured on two different media types (TSA and KBC). To our knowledge, this is the first report of a drone water sampling system for studying microorganisms in aquatic habitats. Such a system has the potential to be used in a variety of hard-to-reach aquatic environments, such as high-altitude alpine lakes and ponds.
2. Materials and Methods
2.1. DOWSE: DrOne Water Sampling SystEm
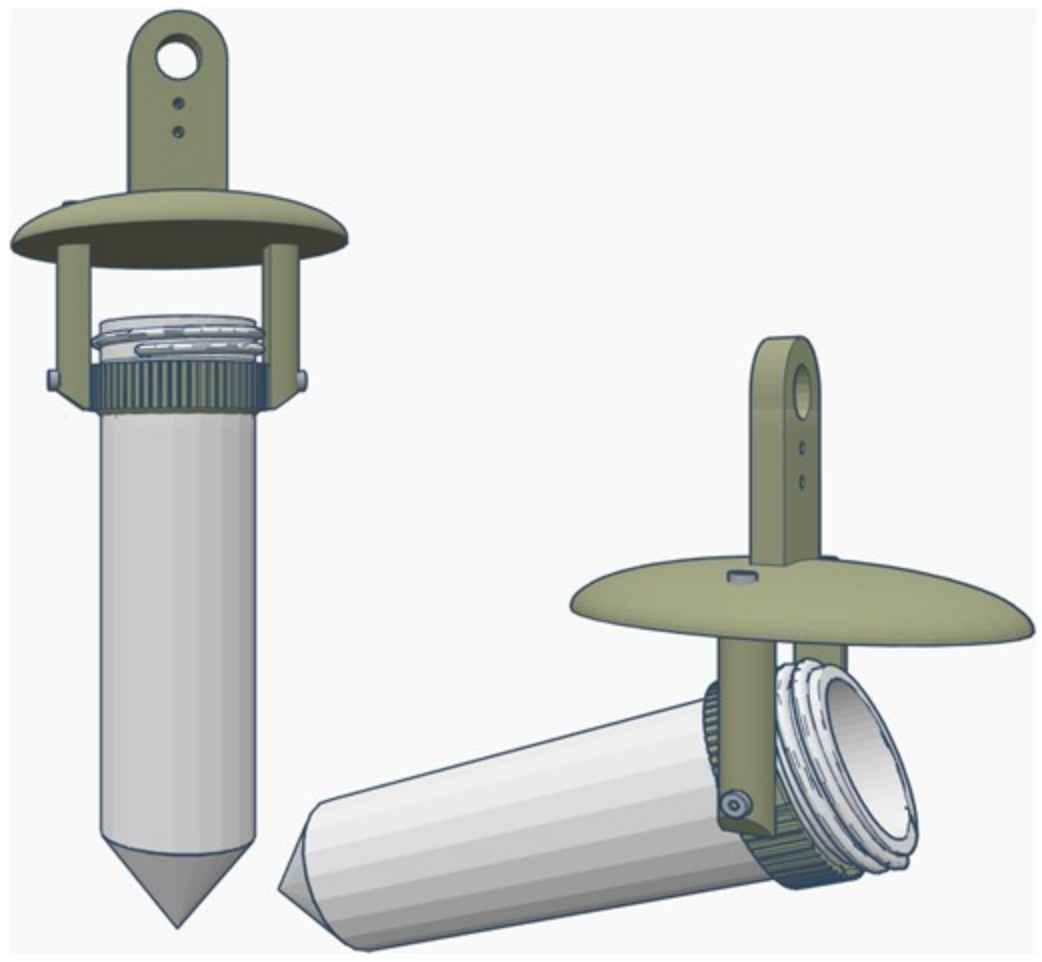
The sampler was tethered to the center of a carbon fiber beam between two 3D-printed mounts attached the landing gear of the drone. The tether between the drone and the sampling device consisted of a fluorescent orange nylon string and was 4.5 m long (
Figure 2a), and a carabiner was used to quickly attach and remove the water sampler following a sampling mission (
Figure 2b). The DOWSE was used to collect samples at three locations 25 m from the nearest shore, and three locations 50 m from the nearest shore. A fishing rod tied to a fluorescent nylon string 2 m in length was used to collect water samples 1 m from the nearest shore with the same 3D-printed device that was tethered to the drone.
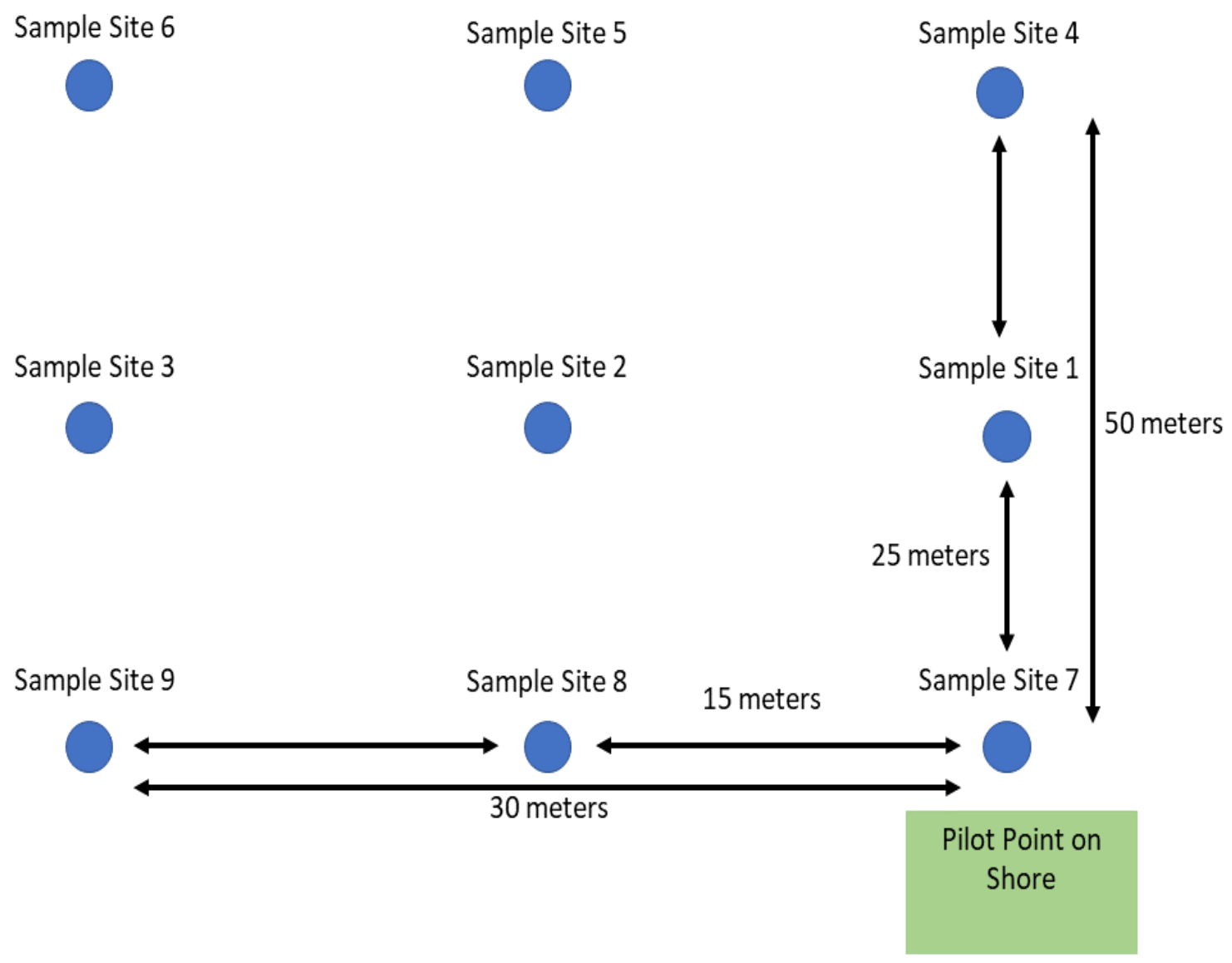
The drone was a Phantom 4 quadcopter (DJI, Shenzhen, China) powered by a 5870 mAh Lithium-ion polymer battery. This battery provided a safe, operational flight time of about 20 min. The Waypoint Intelligent Flight mode in the DJI GO 4 app was used to establish a grid of defined GPS waypoints equidistant from one another in a X, Y Cartesian plane. Three waypoints were 50 m from the nearest shore, three waypoints were 25 m from the nearest shore, and three waypoints were 1 m from the nearest shore (
Figure 3).
After the GPS waypoints were established, the sampling device was loaded with a sterile 50 mL conical tube, and the drone was flown manually to each waypoint location to collect samples. Once the drone reached the sampling location, it descended to the surface of the water and allowed the sampling device to rest on the surface for a few seconds. During this time, the sampling device filled with water. An overhead image was then taken of the sampling device in the water (
Figure 2c), which provided a precise record of the location and sampling time embedded in the Exif file of each of the images. Following each collection, the sample was returned to the shore and the 50 mL tube (now containing a sample of water) was switched out with a new tube while the drone was still in flight (
Figure 2b). The drone then flew to another waypoint to collect another sample, until all targeted collections had been completed (
Figure 2a).
2.2. Lake and Sampling Site Selection
Eight different freshwater lakes were sampled in Austria including Altausseersee (ALT), Grundlsee (GRU), Toplitzsee (TOP), Vorderer Gosausee (GOS), Gosaulacke (GOL), Hinterer Gosausee (HIN), Wörthersee (WOR), and Ossiachersee (OSS) (
Table 1). Sites for sampling each lake were selected based on the availability of a safe and obstruction-free landing zone for the drone. Lakes were sampled 1, 25, and 50 m from the shore line. Personnel and equipment were located at the water’s edge, or on available dock space, except for Ossiachersee (OSS) where operations were carried out in the water about 8 m from the shore line. In order to pilot the drone, clearance was needed in front of the pilot point location area (
Figure 3) with a minimum of 5 m of clearance on either side. The drone required space to hover above the pilot location in order for samples to be returned and allow for new collection tubes to be inserted into the sampler. These sites were also chosen to be minimally intrusive to the residents and others in the area by selecting sites that were away from other people at the time of sampling. Permissions to sample the lakes in June 2018 as part of this project were granted by the Austrian Federal Forests AG, DI Martin Heinz Stürmer, 3 April 2018.
2.3. Drone Bathymetry System
After all water samples were collected in a lake, a wireless bathymetry sensor (the Deeper ProPlus, Deeper, Vilnius, Lithuania) was tethered to the drone to map the temperature and depth from the shore line to 50 m from the shore (doing this after the water sampling minimized the mixing of water across the sampling domain, since the sensor was pulled through the water by the drone). The string length from the drone was 1.64 m to a line connection point with a 32 g weight, followed by a secondary connection, 1.20 m from the connection point to the bathymetry sensor. The weight was used to stabilize the line during windy conditions, and to facilitate a more controlled drag through the water. The bathymetry sensor was dragged through the water by the drone to a 50 m sampling position in the lake while continuously recording depth, GPS coordinates, and temperature of the water at one second intervals. The sensor was set to onshore GPS mode.
2.4. Filtering of Samples
Lake samples were processed on the same day that they were collected. Each 100 mL sample was poured into a single use, sterile Microfunnel 0.2 μm Gidded disposable Pall filter funnel (VWR International, Radnor, PA, USA, #28143-542). The lid was closed, and each unit was fitted onto a 500 mL filtration flask. A water jet pump (VWR International, Radnor, PA, USA, #181-9100) was used to vacuum filter each sample through the 0.22 μm filter. The filter assembly was dismantled, and the filter was recovered and transferred to a sterile 15 mL conical tube for shipping. The conical tubes with filters were separated into bags representing the lake they were collected in and stored at 4 °C until all samples were collected. All lake samples were shipped together in a cooler with ice packs.
2.5. Culturing of Microbes
Filters were stored at 4 °C for up to two weeks. The filters were taken from refrigerated storage and placed into sterile 100 mL glass bottles with 4 mL deionized (DI) water and a sterile stir bar. The bottles were set on a stir plate and the suspension was stirred for 10 min to lift microbes from the filter into the 4 mL resuspension water, resulting in a 25× concentration of collected lake sample. Aliquots of 1 mL of this water were serially diluted in 9 mL tubes, resulting in a 2.5× concentration, 0.25 dilution, and 0.025 dilution. Microbes from each sample site location were plated once at two different concentrations, on both TSA, and KBC agar plates. Aliquots of 200 μL of the 25× and 2.5× concentration were spread onto KBC media with the antibiotic cephalexin and cycloheximide, a eukaryotic protein synthesis inhibitor used to stop fungal growth. The KBC medium is semi-selective for bacteria in the genus
Pseudomonas [
16]. Aliquots of 200 μL of each remaining dilution (0.25 and 0.025) were spread on plates of Tryptone Soya Agar (TSA) media containing cycloheximide. Negative controls were plated with 200 μL of sterile water.
2.6. Quantification of Microbes from Plates
Pictures were taken of the TSA 0.025 dilution plates and of the 2.5× concentration KBC plates. These pictures were uploaded and analyzed using OpenCFU (Quentin Geissmann, Berlin, Germany, Version 3.8.11) an open source software for counting colonies. OpenCFU was utilized to count one half of the plate, and counts were modified manually to correct for false positive and false negative counts. The final corrected plate count value was used to calculate CFU/mL for each filter location, for TSA, and for KBC.
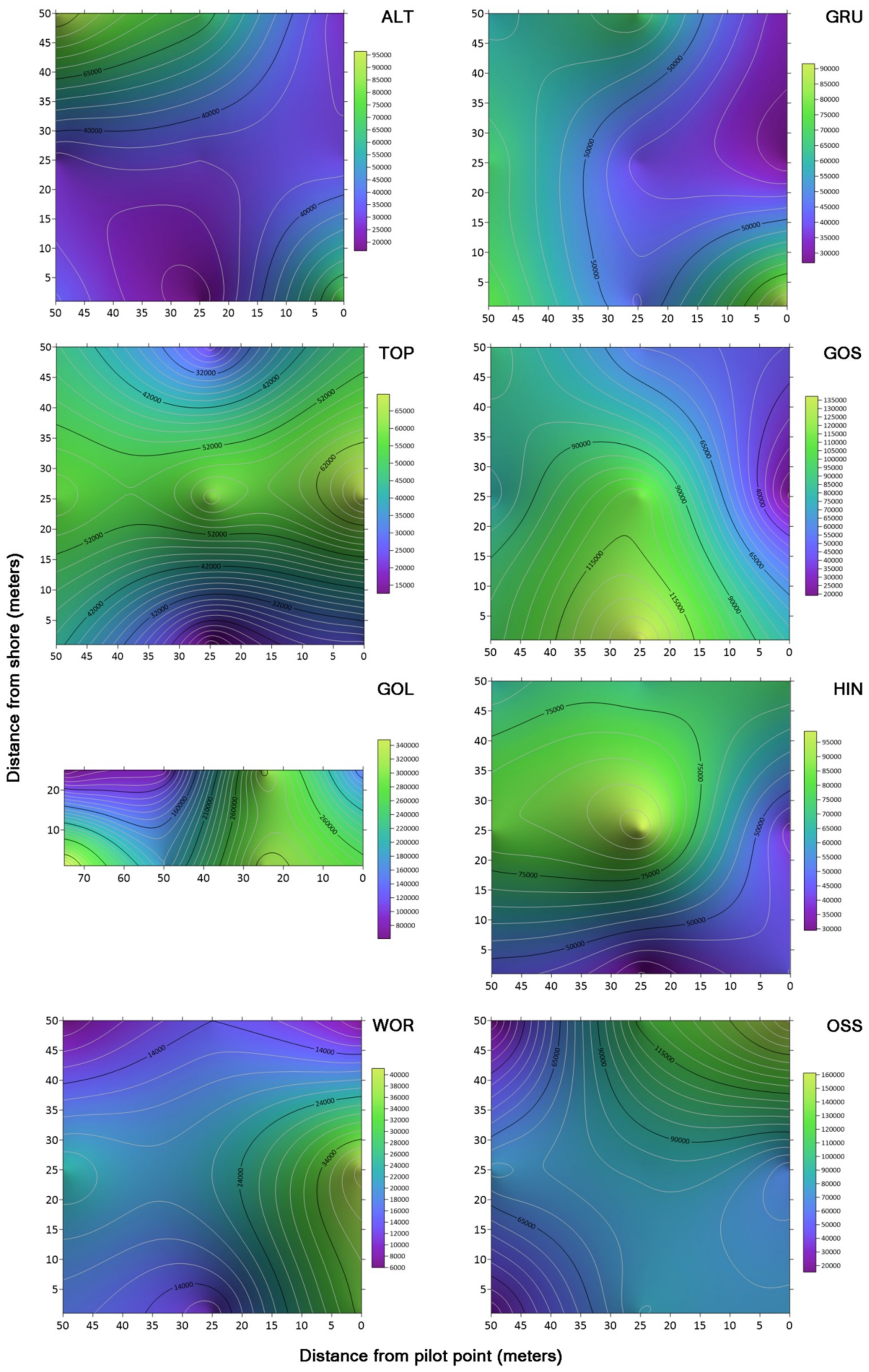
2.7. Contour Maps of Microbe Concentrations
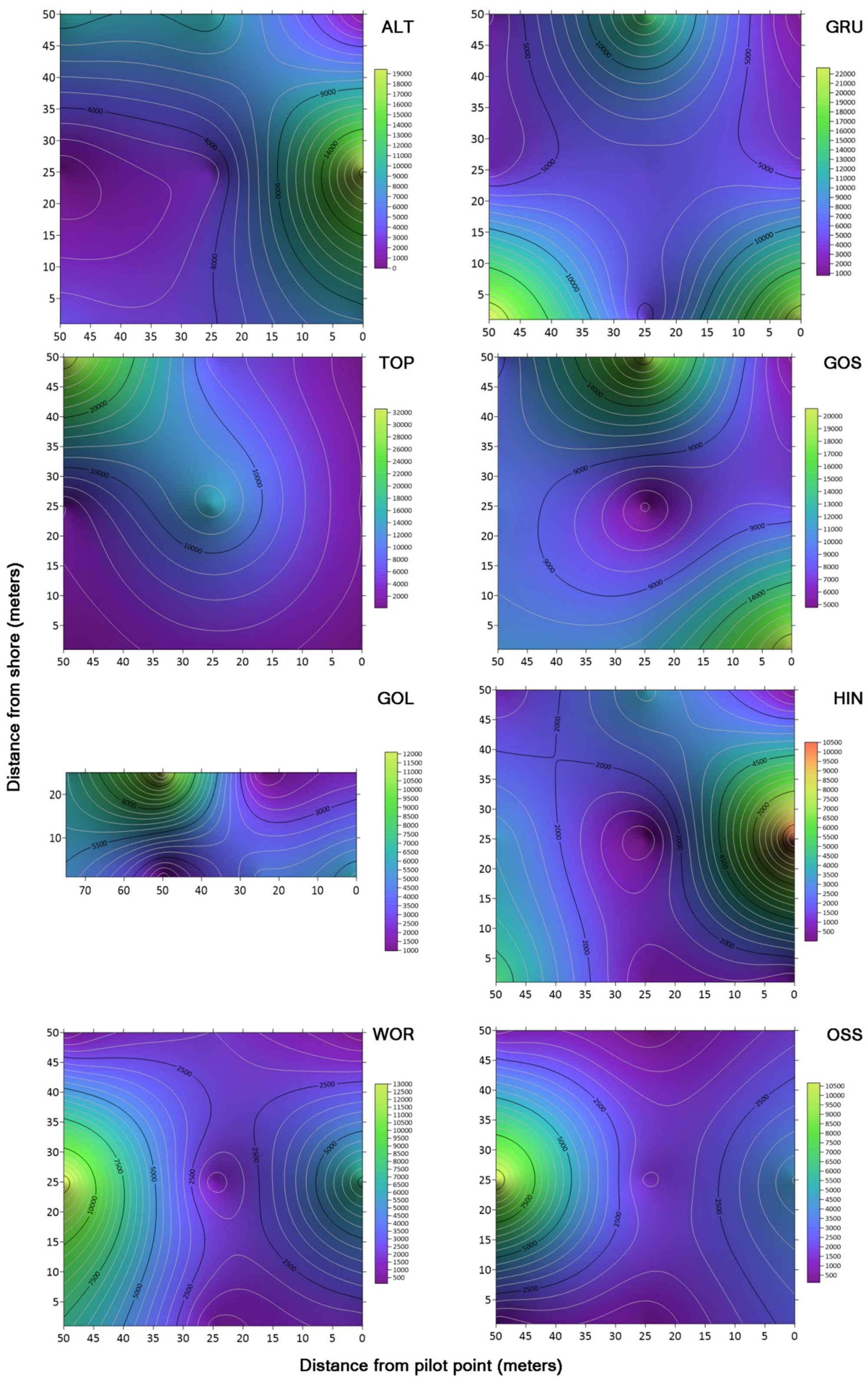
To visualize the distribution of microbes across each of the lakes studied, contour maps were created using the CFU/mL data collected from both sets of media using Surfer 15 software (Golden Software, Golden, CO, USA). Concentration data for TSA and KBC was plotted on an X, Y Cartesian plane with distance in each dimension measured from the designated pilot start point (
Figure 3). This grid file was then used to create a contour map with a color relief overlay.
2.8. Ice Nucleation Assays
A series of ice nucleation assays (INA) were conducted with a subset of colonies from each of the lakes. Eight colonies were selected at random from each plate of KBC from each of the eight sampling locations. A maximum of 72 colonies from each lake were screened for the ice+ phenotype. These colonies were picked with a sterile wooden tooth pick and deposited into 140 μL of sterile DI water in a 96-well plate. When growth from a low dilution plate was absent, colonies from the higher dilution plate of the same media type were picked. The 96-well assay plates were stored at 4 °C overnight, and then pulse-vortexed for 30 s and incubated at 4 °C for an additional hour. A multichannel pipet was used to transfer duplicate rows of 12 μL drops onto a PARAFILM® M (Sigma, St. Louis, MO, USA, P6543) float assembly in a Cryo bath circulator Lauda Alpha RA 12 (LCKD 4908) cooling bath (LAUDA-Brinkmann, LP, Delran, NJ 08075, USA) with ethylene glycol coolant fluid (Air gas RAD64000246). The circulator was set to −6 °C, and the temperature dropped first to −7 °C, then −8 °C with one-minute incubation intervals at each temperature and a final incubation time of 10 min at −8 °C.
2.9. Statistical Analyses
Statistical analyses were performed in JMP 14 (SAS, Cary, NC, USA) to test our hypotheses that (1) concentrations of culturable microorganisms in freshwater lakes would vary with distance from the shore and (2) the frequency of ice-nucleating strains of Pseudomonas spp. would vary across different freshwater lakes. An analysis of variance (ANOVA) was used to test for significant differences in concentrations of culturable bacteria between the three sampling distances across all of the lakes, and when controlling for the lake studied. Gosaulacke (GOL) was excluded from these tests, since this lake was not wide enough to allow the collection of samples 50 m from each shore. Tukey’s Honest Significant Difference (HSD) was used to test for differences in concentrations of culturable microbes at each of the sampling locations within a lake. ANOVA was used to test for significant differences in concentrations of culturable microbes at different sampling distances across all of the lakes, when controlling for lake depth and temperature. ANOVA and Tukey’s HSD were used to test for differences in the frequency of ice+ bacteria at different distances from the shore in each of the lakes. ANOVA was also used to test for differences in the altitude of the sample sites and the total percent of colonies that were ice nucleation active at −8 °C.
4. Discussion
New tools and technology are needed to study microorganisms in freshwater environments. Little is known about spatial distribution and ice nucleation activity (INA) of microorganisms in freshwater lakes. We developed a system to collect water samples from the surface of lakes using a 3D-printed sampling device tethered to a drone (DOWSE, DrOne Water Sampling SystEm). The DOWSE was used to collect surface water samples at different distances from the shore (1, 25, and 50 m) at eight different freshwater lakes in Austria in June 2018. Water samples were filtered, and microorganisms were cultured on two different media types (TSA and KBC). Our work extends the drone water sampling concept developed by Ore et al. [
14] and Koparan et al. [
15]. To our knowledge, this is the first report of a drone water sampling system for studying microorganisms in aquatic habitats.
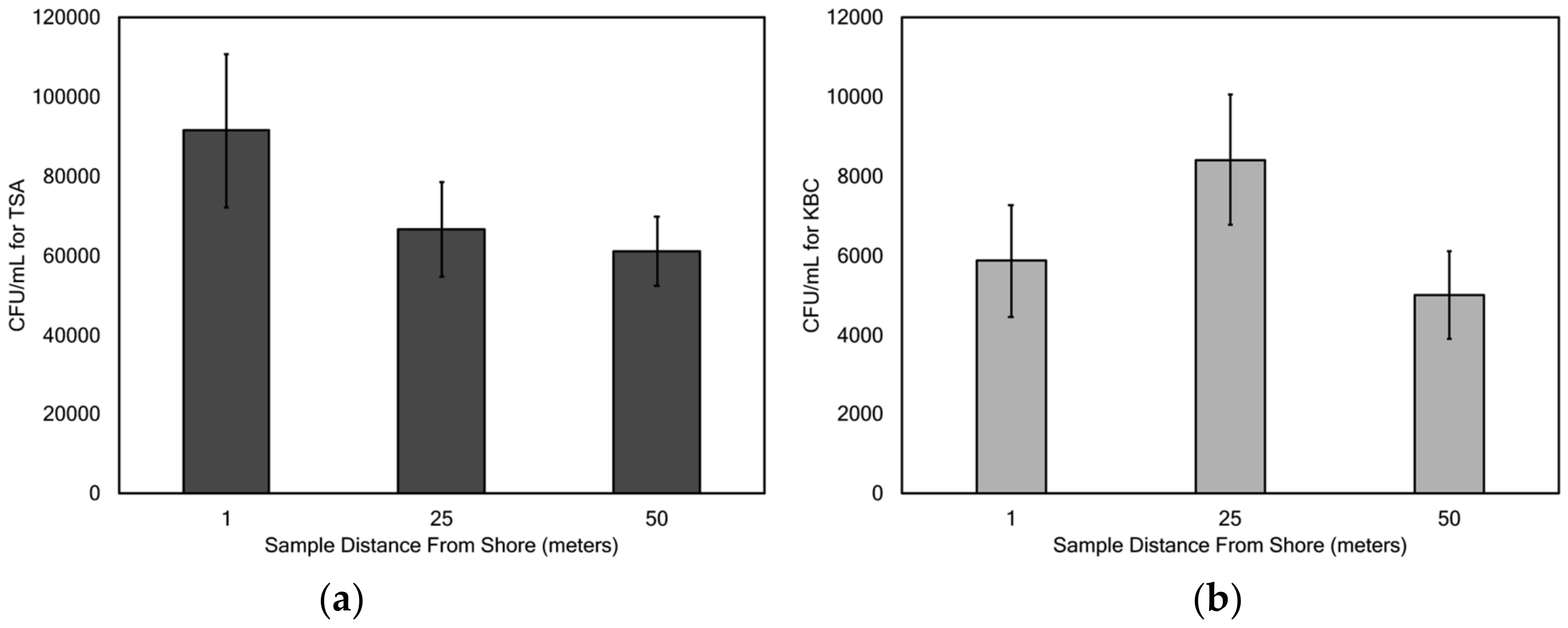
Mean concentrations of microbes cultured on TSA ranged from 19,800 (Wörthersee) to 215,300 (Gosaulacke) CFU/mL, and mean concentrations of microbes cultured on KBC ranged from 2590 (Ossiachersee) to 10,969 (Vorderer Gosausee) CFU/mL. These ranges in concentration are much lower than those reported in Claytor Lake in southwest Virginia, USA [
3]. The mean concentration of bacteria from the Claytor Lake surface samples between 2 May 2016 and 7 June 2016 on TSA was 5.20 × 10
8 CFU/mL, and on KBC media was 1.27 × 10
7 CFU/mL. The larger concentrations of bacteria measured in Claytor Lake could reflect differences in the collection sites. Claytor Lake is a reservoir without glacial inputs, and with a comparatively larger watershed for nutrients to be collected before running into the lake [
17]. The alpine lakes sampled in this study have relatively small watersheds, and likely have much a more limited supply of the nutrients required for bacterial growth [
17]. Another potential explanation for the lower concentrations in the Austrian lakes could be the amount of time from sample collection to plating; due to the remote nature of the study sites and the need to transport the samples to another country, the sample filters were plated on culture media within about two weeks. It is possible that there could have been a loss in viability during storage, potentially underestimating the actual culturable diversity in the lakes. However, since all of the samples were processed following similar storage times, any potential loss of viability would have likely been similar for all of the samples collected in this study.
There was no significant difference in sampling distance from the shore for concentrations of bacteria cultured on TSA and KBC, with the exception of lake Toplitzsee. With this sole exception, these results suggest that either the general bacterial and
Pseudomonas populations are randomly distributed around the surface waters of the lakes, or that there is some other factor that influences this distribution that was not tested for in this study. There may be some factor found in Toplitzsee lake that is not found in the other lakes studied that results in the difference between the 1 and 50 m distance from shore. Cole et al. [
18] found that bacterial concentrations are generally consistent in all depths of stratified lakes’ epilimnion. Powers et al. [
12] came to a similar conclusion when comparing concentrations of bacteria collected at 5 and 50 cm below the surface of the water at Claytor Lake, Virginia, USA. Pietsch et al. [
3] found that there was no significant difference between the surface water and water sampled at depths down to 9 m in bacterial concentration in Claytor Lake. Thus, we hypothesize that similar distributions of bacteria are likely to be found at different depths below the surface of the Austrian lakes, but this was not tested in the current study.
Concentrations of bacteria cultured on TSA varied significantly with lake depth and within the range of temperatures measured (17.7–25.0 °C), but concentrations of bacteria cultured on KBC did not vary significantly with depth and temperature. Thus, the depth of the lake may have an impact on the concentrations of many of the culturable bacteria at the lake surface, but not for
Pseudomonas-like bacteria. Pietsch et al. [
3] reported a similar range in surface water temperatures when sampling the surface water of Claytor Lake in southwest Virginia, USA between the May and June sampling dates. However, the temperature ranges from their study did not include distance from shore or depth of measurement. Alvarez et al. [
19] generated a bathymetric map of Finn Creek Site 21 Reservoir in McCain County, Central Oklahoma, USA using a small unmanned aerial system (sUAS), and attached a floating data collector similar to the system used in our study. The coupling of aerial drone and bathymetric data collectors provides a quick and accurate means to map lakes [
19]. Perhaps unmanned systems could be tuned to predict concentrations of bacteria in lakes, provided parameters such as temperature and depth can be measured remotely.
Contour maps of concentrations of microbes across the drone sampling domain revealed areas of high concentrations (hot spots) in some of the lakes. However, there were no repeating patterns in the concentrations of bacteria between lakes that suggested a trend between the lakes in relation to the distance samples were taken from the shore. Schmale et al. [
20] showed both random and aggregated patterns of spore deposition in corn canopies over 2 years. The sampling grids used to generate their contour maps contained 12 to 16 discrete sample sites. This would suggest that additional data sets with expanded sampling locations from a designated origin in two dimensions could provide for a more robust analysis. An expanded collection of specific environmental variables could allow for a more complex analysis, such as a four-factor principal components analaysis (PCA) study that could elucidate trends in spatial and temporal variables and provide a determination ranking among collected data sets to show specific environmental factors that have a higher influence on observed environmental variance. In a four component PCA study with multiple environmental categories, Iizuka et al. [
21] showed which environmental components contributed to the variance of the sample set. It should also be noted that contour maps of concentrations of bacteria could be influenced by currents in the lake, including water inputs near the sampling grid. Wind pushing and mixing the water in the lakes could influence the concentrations of bacteria on the surface of the lakes inside the sampling grid [
18]. If the wind can transfer enough energy to the water, the composition of bacteria at the surface may change due to mixing of the water and nutrients. The effect of this vertical mixing could alter the concentration of surface waters as bacteria compete for these nutrients [
22]. The wind could also aerosolize the bacteria and even transport them among lakes [
19,
20,
21]. One limitation of our study is that we did not collect any water quality data from our samples (e.g., pH, nutrients, chlorophyll, dissolved organic carbon, and conductivity), which could help explain some of the observed differences within and among the lakes sampled during our campaign. Future work should consider obtaining these environmental data, which will also likely need to include an expansion of the sampling device to obtain larger sample volumes during collection.
The percentage of ice+ microbes cultured on KBC (selective for bacteria in the genus
Pseudomonas) was significantly different across all the lakes sampled, and ranged from 0% (0/64) (Wörthersee) to 58% (42/72) (Vorderer Gosausee), with a mean of 28% (153/544) for the entire sample set. Samples that showed an ice+ phenotype by freezing in the droplet assay at −8 °C support previous literature that biogenic ice nucleators influence water to freeze at temperatures warmer than anthropogenic ice nucleators [
23,
24]. Over 90% (actual/numbers here) of colonies tested froze after loading the drops onto the float assembly at the pre-cooled temperature of −6 °C. This consistent frequency of freezing from colonies that were picked from KBC plates suggests that the glacial lake systems in this study contain ice+ bacteria that belong to the genus
Pseudomoas. This average is much higher than would be expected, based on the findings of Pietsch et al. [
3], where the mean ice+ percentage in samples collected in spring and early summer was 0.49%. These authors found that the time of year had a significant influence on the percentage of ice+ strains that were found. As the percentage of ice+ strains was so much higher in the Austrian lakes sampled, there are likely to be a suite of factors that makes it more advantageous for these strains to be present later in the year in the lakes studied in Austria. Altitude of the lakes sampled did not have a significant impact on the percentage of ice+ bacteria collected at the different lakes sampled. Although there was a positive trend between elevation above sea level and percent of colonies that were ice+, this was not significant. If more alpine lakes were sampled, perhaps the additional data points would strengthen these observations. GRU (43% ice+) is downstream relative to TOP (18% ice+), and had a higher percentage of ice+ bacteria, suggesting that in the months immediately following glacial melt, ice+ bacteria flow down and accumulate in lower altitudes. This trend held true for the higher altitude lake, HIN (32% ice+) relative to the lower altitude lake, GOS (58%). For lakes in close proximity that share the same source of glacial run-off, the lower altitude lakes had a higher percentage of ice-nucleating bacteria. Lake size, depth, and/or surface area could be a significant factor in the percent of bacteria that exhibit the ice+ phenotype throughout the year and warrants further study. Altitude might also play a role in the source sink relationship for ice nucleation activity, particularly in the context of our connected high-altitude lakes sampled on 7 June and 8 June 2018. The highest altitude site in a cluster of three lakes is referred to as the “source” lake, while the lowest altitude lake is referred to as the sink lake. We observed an increase in ice nucleation activity in the “sink” lake during the month of June 2018. Interestingly, the “source” lake located in the highest altitude, showed a higher percentage of ice nucleation activity than the mid-altitude, or “flow-thru” lake. Samples were taken during active thawing. The lowest percentage of ice nucleators in the “flow-thru” lakes, could indicate that those lakes are providing transport during the active thaw cycle.
Our drone water sampling system was designed to inform appropriate sampling strategies for microbes at the water surface, and to better understand the diversity and function of ice nucleating microorganisms in aquatic environments. Our system has the potential to be used in a variety of hard-to-reach aquatic environments, such as high-altitude alpine lakes and ponds. The DOWSE sampler could be used to identify and track potential toxins, and contribute to efforts aimed at finding causes of these harmful blooms [
25,
26,
27]. This system allows water sampling near the shore line without any major disturbances or mixing of the sample area. Future work aims to elucidate the structure and function of entire microbial assemblages within and among the Austrian lakes; diversity analyses of culturable plate growth on the non-selection TSA media could be determined by 16S sequence analysis [
28]. Additional analyses of the abundance of ice+ bacteria in surrounding bodies of water, such as glacial streams and rivers, could also be conducted to determine sources and reservoirs of ice nucleating microorganisms.