Zeolite as a Potential Medium for Ammonium Recovery and Second Cheese Whey Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Artificial Wastewater and Secondary Cheese Whey
2.2. Zeolite Characterization
2.3. Zeolite Characterization
2.4. Ammonium Adsorption Kinetics and Isotherms
2.4.1. Ammonium Adsorption Kinetics
2.4.2. Ammonium Isothermal Curves
2.5. Adsorption and Desorption Tests in Laboratory-Scale Columns Using AWW and SCW
2.6. Analytical Methods
3. Results and Discussion
3.1. Adsorption Batch Tests
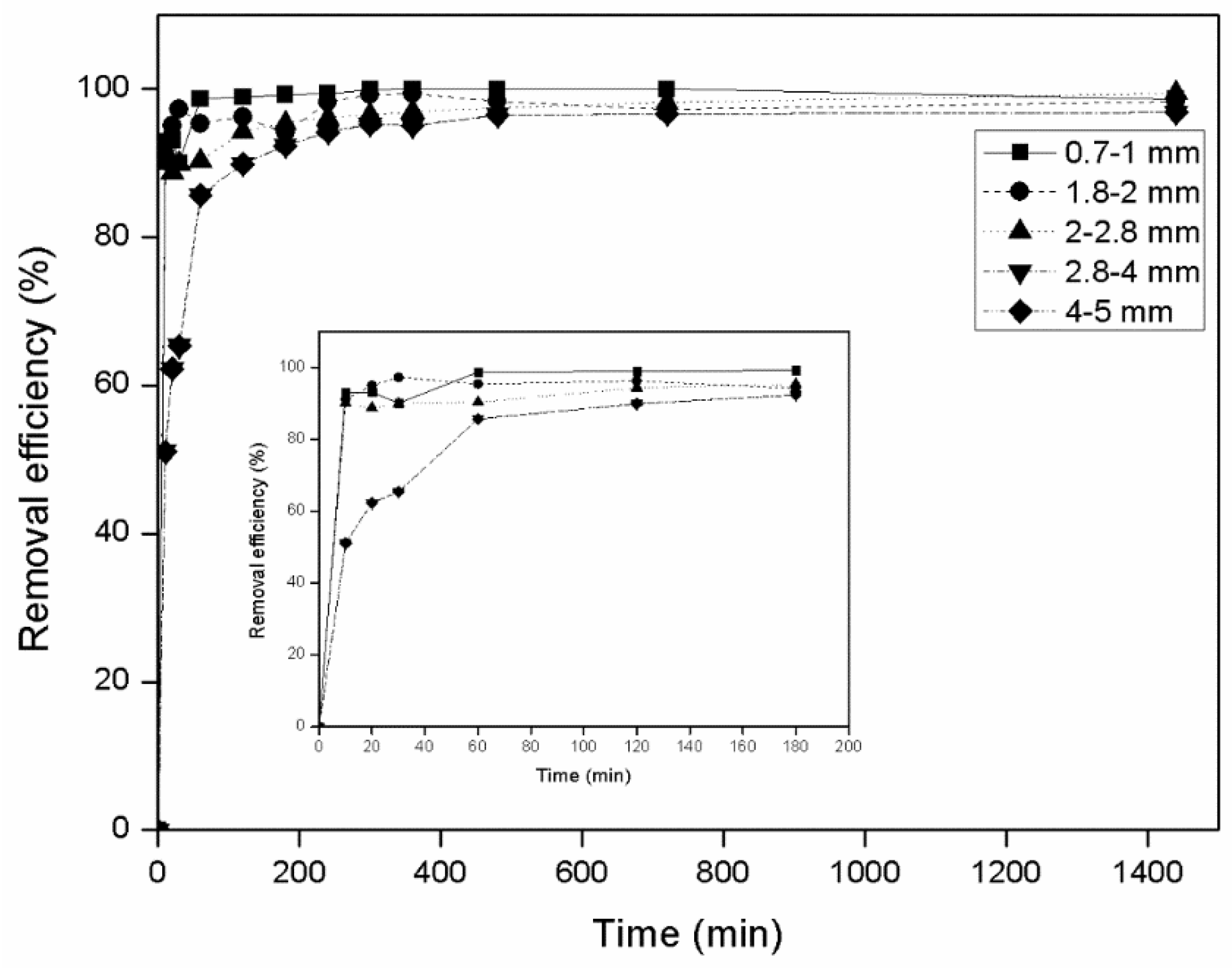
3.1.1. Effect of Particle Size
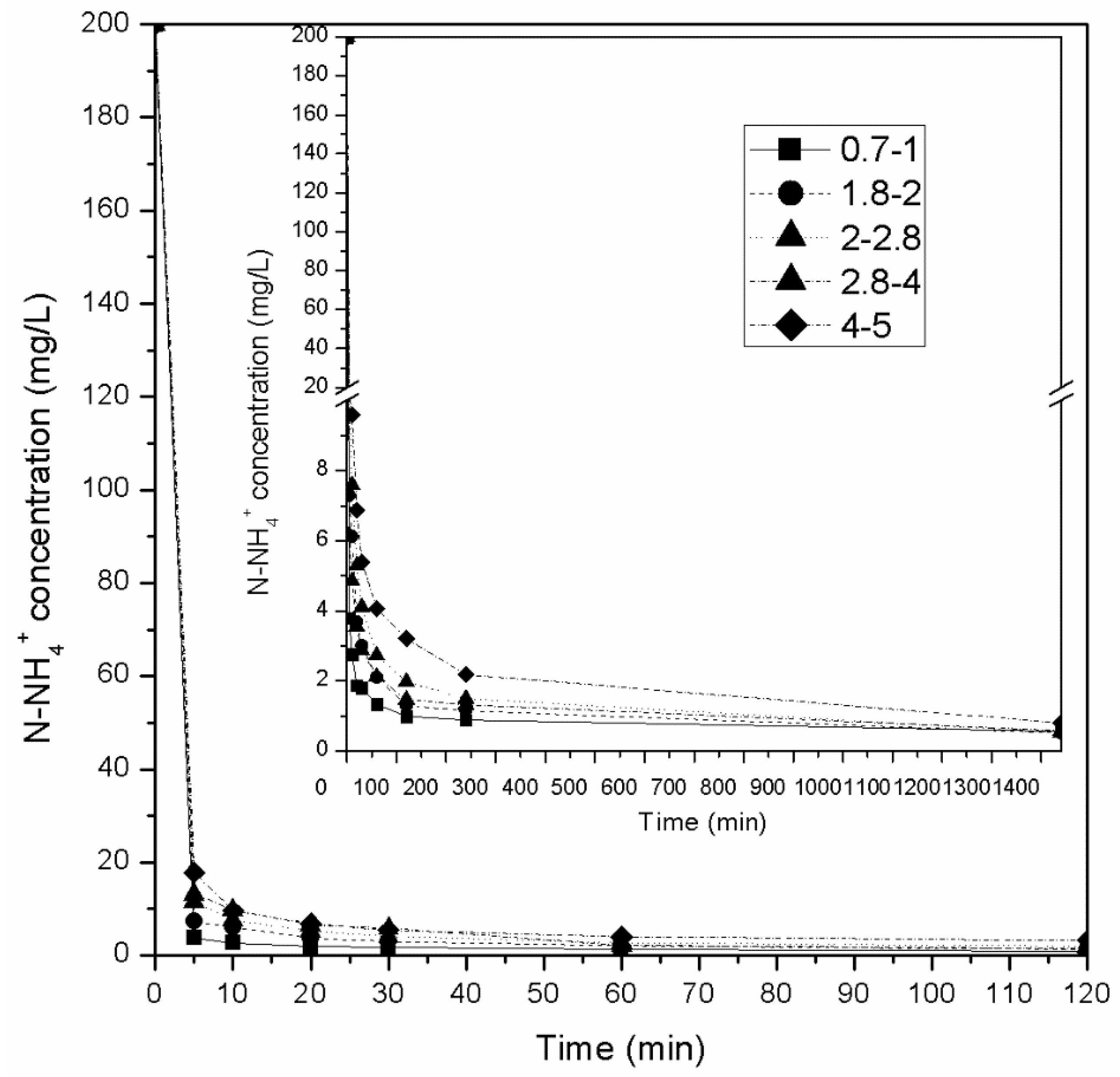
3.1.2. Effect of Contact Time
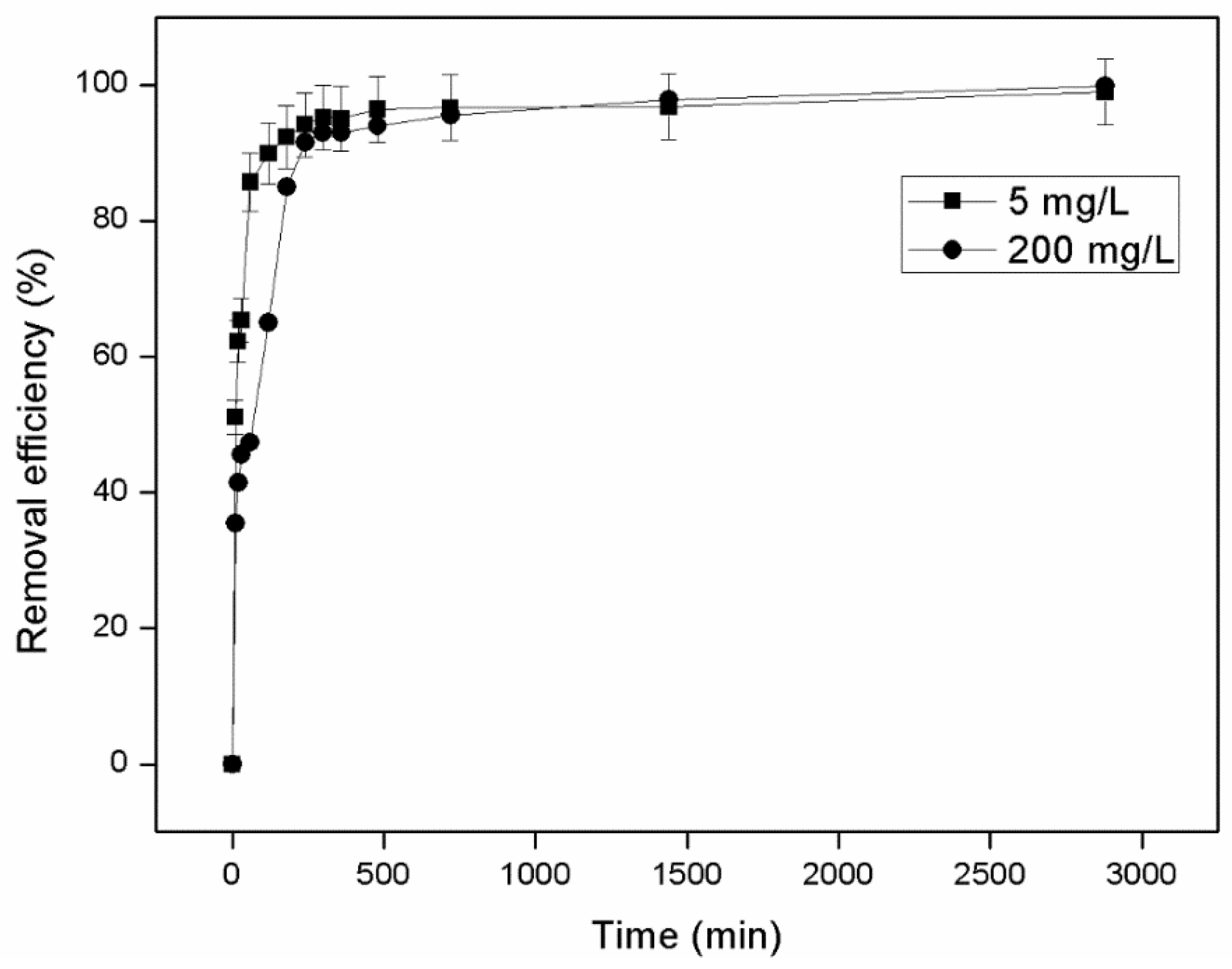
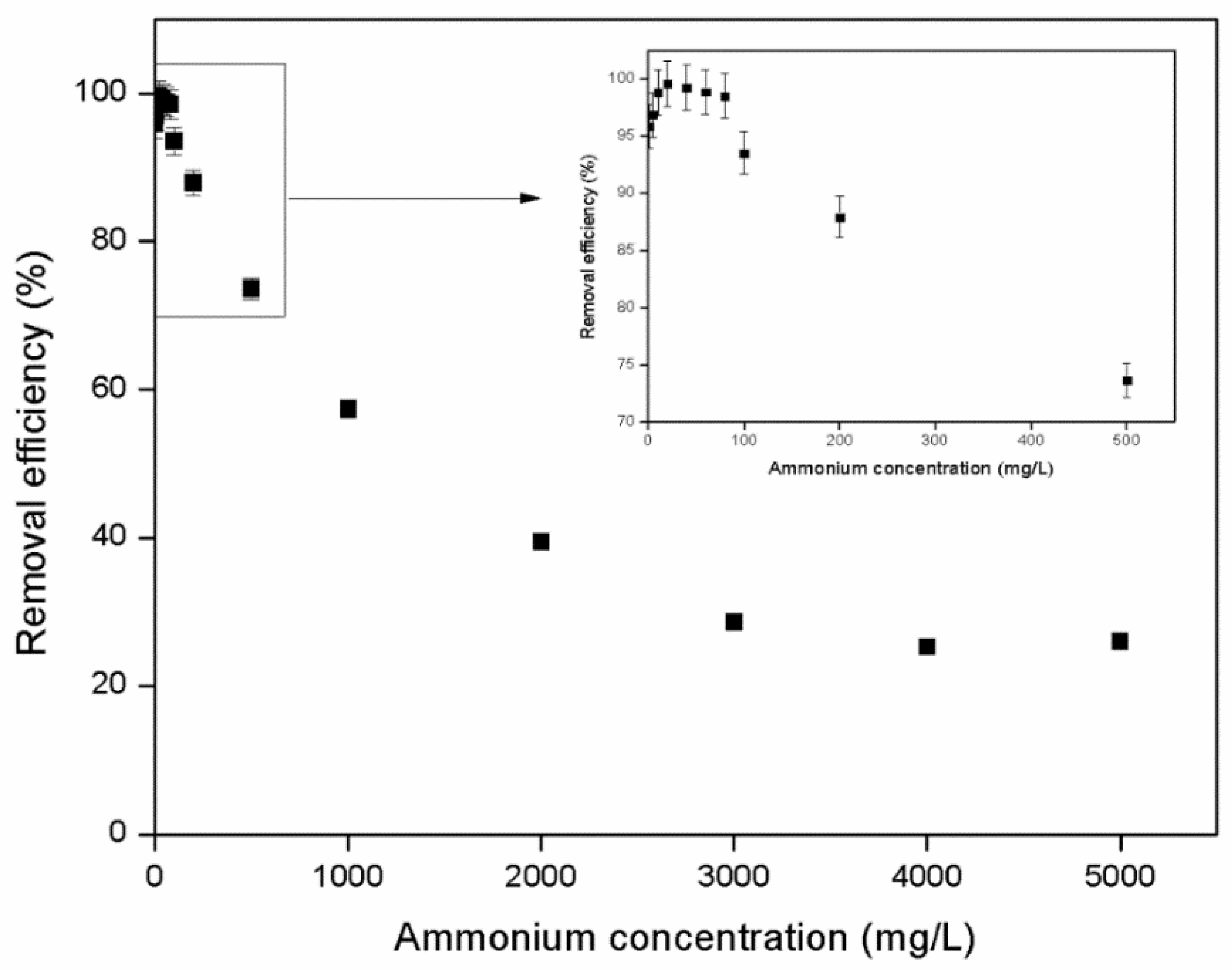
3.1.3. Effect of Initial Concentration
3.1.4. Kinetic Models
3.1.5. Equilibrium Isotherms
3.2. Desorption Batch Tests
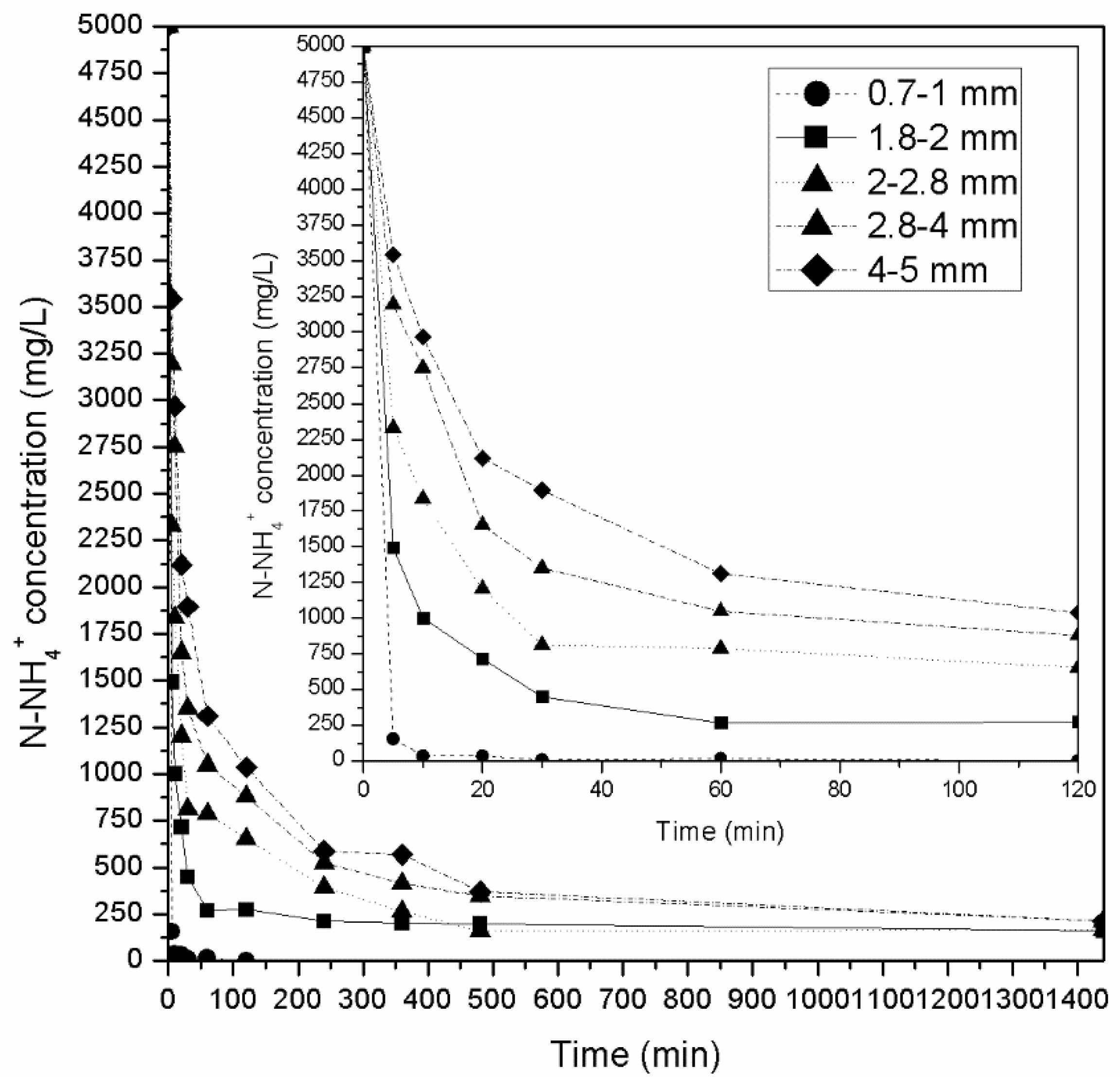
3.3. Adsorption and Desorption Experiments on Laboratory Scale Column Using AWW
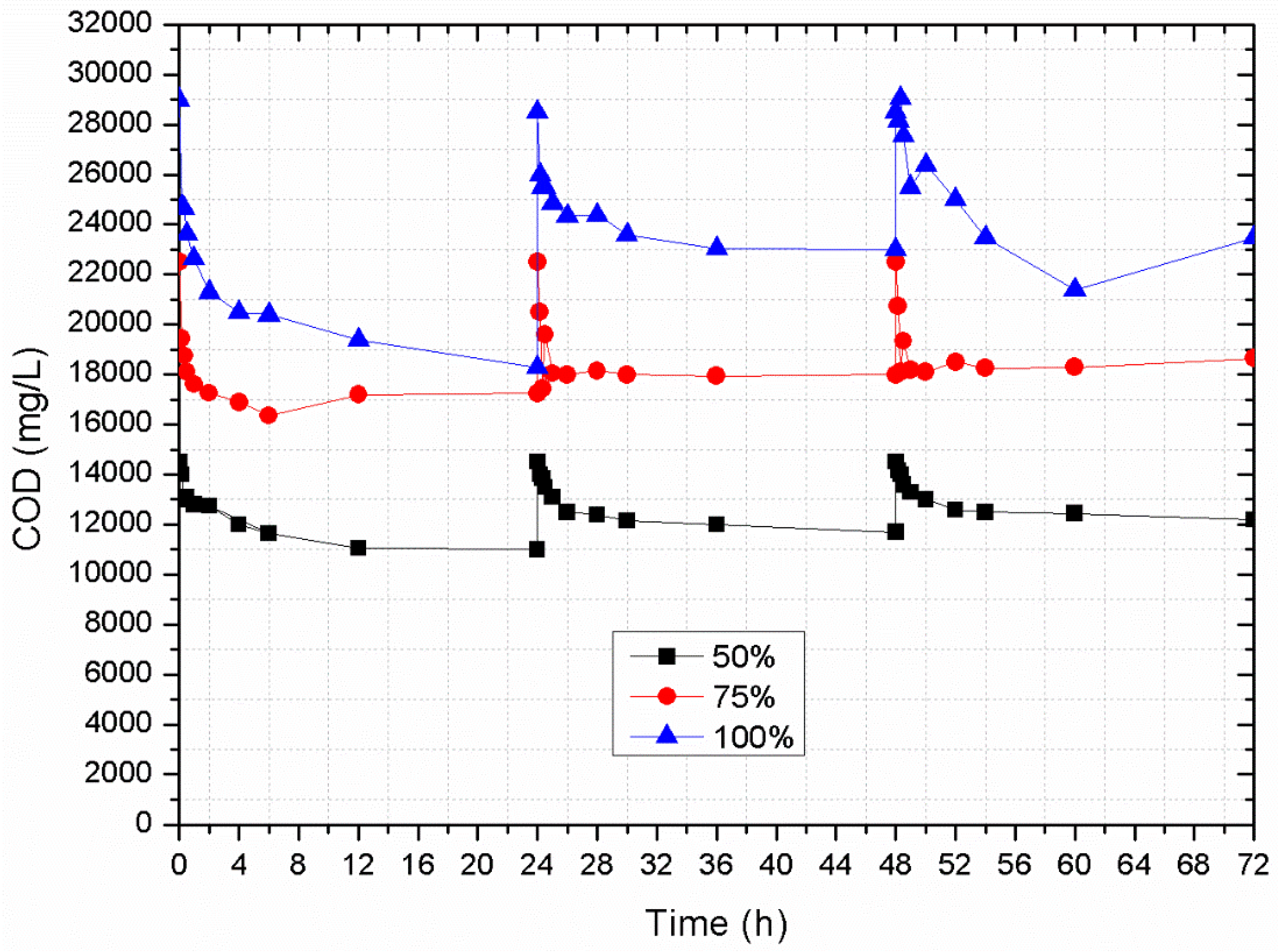
3.4. Sorption Column Studies Using SCW
3.5. Effect of Temperature and Biofilm Growth
4. Conclusions
- Batch experiments revealed that zeolite granulometry had no significant effect on its ability to absorb ammonium, while maximum removal efficiency was achieved at ammonium concentrations ranging from 10 to 80 mg/L.
- The pseudo-second order model fitted the experimental data thus revealing that chemisorption is the mechanism for the adsorption process of ammonium on zeolite. The Freundlich isotherm model best fit the experimental data.
- Experiments in columns using synthetic wastewater with an initial NH4+-N concentration of 200 mg/L revealed that for all granulometries tested, the zeolite was able to absorb almost all available NH4+-N (over 99%) in the first 120 min. The same was not observed for initial concentrations of 5000 mg/L where only zeolite with grain sizes of 0.71–1.0 mm managed to remove over 99% NH4+-N (99.9%) in 120 min. All other zeolite granulometries reached equilibrium much later (24 h, 96%–97% removal of NH4+-N).
- Natural zeolite may be used as an alternative substrate for second cheese whey treatment, as significant removal of organic load (up to 40%, 14.53 mg COD/g of zeolite) and NH4+-N (about 99%) can be achieved. Concerning PO43−-P, the zeolite appeared to saturate after day one of the experiments at a removal capacity of 0.15 mg P/g of zeolite. Desorption experiments with deionized water in batch and columns presented low desorption rates for NH4+-N and PO43−-P, thus indicating that this zeolite could be used as substrate for slow nitrogen release in soils.
- Prolonged use of zeolite in SCW treatment (after three days) led to the formation of biomass by increasing the percentage of organic load removal.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alayu, E.; Yirgu, Z. Advanced technologies for the treatment of wastewaters from agro-processing industries and cogeneration of by-products: A case of slaughterhouse, dairy and beverage industries. Int. J. Environ. Sci. Technol. 2018, 15, 1581–1596. [Google Scholar] [CrossRef]
- Abdallh, M.N.; Abdelhalim, W.S.; Abdelhalim, H.S. Industrial Wastewater Treatment of Food Industry Using Best Techniques. Int. J. Eng. Sci. Invent. 2016, 5, 15–28. [Google Scholar]
- Carvalho, F.; Prazeres, A.R.; Rivas, J. Cheese whey wastewater: Characterization and treatment. Sci. Total Environ. 2013, 445–446, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, A.A. Treatment of Cheese Processing Wastewater by Physicochemical and Biological Methods. Int. J. Microbiol. Res. 2013, 4, 321–332. [Google Scholar]
- Jasko, J.; Skripts, E.; Dubrovskis, V.; Zabarovskis, E.; Kotelenecs, V. Biogas production from cheese whey in two phase anaerobic digestion. Eng. Rural Dev. 2011, 373–376. [Google Scholar]
- Smithers, G.W. Whey and whey proteins—From ‘gutter-to-gold’. Int. Dairy J. 2008, 18, 695–704. [Google Scholar] [CrossRef]
- OECD-FAO, Agricultural Outlook 2014. Highlights. Paris. Available online: http://www.oecd-ilibrary.org/agriculture-and-food/oecdfao-agricultural-outlook_19991142 (accessed on 10 November 2014).
- Tatoulis, T.I.; Tekerlekopoulou, A.G.; Akratos, C.S.; Pavlou, S.; Vayenas, D.V. Aerobic biological treatment of second cheese whey in suspended and attached growth reactors. J. Chem. Technol. Biotechnol. 2015, 90, 2040–2049. [Google Scholar] [CrossRef]
- Chatzipaschali, A.A.; Stamatis, S.A. Biotechnological utilization with a focus on anaerobic treatment of cheese whey: Current status and prospects. Energies 2012, 5, 3492–3525. [Google Scholar] [CrossRef]
- Gazette of the Greek Government (GR) 2011/354B of 8 March 2011 on the “Establishment of Measures, Conditions and Procedures for the Reuse of Treated Wastewater and Other Provisions”. Available online: www.et.gr (accessed on 11 January 2019).
- Tatoulis, T.; Akratos, C.S.; Tekerlekopoulou, A.G.; Vayenas, D.V.; Stefanakis, A.I. A novel horizontal subsurface flow constructed wetland: Reducing area requirements and clogging risk. Chemosphere 2017, 186, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Sultana, M.-Y.; Mourti, C.; Tatoulis, T.; Akratos, C.S.; Tekerlekopoulou, A.G.; Vayenas, D.V. Effect of hydraulic retention time, temperature, and organic load on a horizontal subsurface flow constructed wetland treating cheese whey wastewater. J. Chem. Technol. Biotechnol. 2016, 91, 726–732. [Google Scholar] [CrossRef]
- Aly, A.A.; Hasan, N.Y.Y.; Al-Farraj, S.A. Olive mill wastewater treatment using a simple zeolite-based low cost method. J. Environ. Manag. 2014, 145, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Aly, A.A.; Alashgar, N.S.K.; Al-Farraj, S.A.; Ibrahim, M.H. Contaminants and salinity removal of olive mill wastewater using zeolite nanoparticles. Sep. Sci. Technol. 2018, 53, 1638–1653. [Google Scholar] [CrossRef]
- Huang, H.; Yang, L.; Xue, Q.; Liu, J.; Hou, L.; Ding, L. Removal of ammonium from swine wastewater by zeolite combined with chlorination for regeneration. J. Environ. Manag. 2015, 160, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Schierano, M.C.; Maine, M.A.; Panigatti, M.C. Dairy farm wastewater treatment using horizontal subsurface flow wetlands with Typhadomingensis and different substrates. Environ. Technol. 2017, 38, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Kolakovic, S.; Stefanovic, D.; Milicevic, D.; Trajkovic, S.; Milenkovic, S.; Kolakovic, S.; Andjelkovic, L. Effects of reactive filters based on modified zeolite in dairy industry wastewater treatment process. Chem. Ind. Chem. Eng. Q. 2013, 19, 583–592. [Google Scholar] [CrossRef]
- Samkutty, J.P.; Gough, H.R. Filtration treatment of dairy processing wastewater. J. Environ. Sci. Health Part A 2002, 37, 195–199. [Google Scholar] [CrossRef]
- An, S.-W.; Jeong, Y.-C.; Cho, H.-H.; Park, J.-W. Adsorption of NH4+-N and E. coli onto Mg2+-modified zeolites. Environ. Earth Sci. 2016, 75, 437. [Google Scholar] [CrossRef]
- Beebe, D.A.; Castle, J.W.; Rodgers, J.H., Jr. Treatment of ammonia in pilot-scale constructed wetland systems with clinoptilolite. J. Environ. Chem. Eng. 2013, 1, 1159–1165. [Google Scholar] [CrossRef]
- Das, P.; Prasad, B.; Singh, K.K.K. Applicability of zeolite based systems for ammonia removal and recovery from wastewater. Water Environ. Res. 2017, 89, 840. [Google Scholar] [CrossRef]
- Delkash, M.; Bakhshayesh, E.B.; Kazemian, H. Using zeolitic absorbents to clean up special wastewater streams: A review. Microporous Mesoporous Mater. 2015, 214, 224–241. [Google Scholar] [CrossRef]
- Ding, Y.; Sartaj, M. Statistical analysis and optimization of ammonia removal from aqueous solution by zeolite using factorial design and response surface methodology. J. Environ. Chem. Eng. 2015, 3, 807–814. [Google Scholar] [CrossRef]
- Dong, Y.-B.; Lin, H. Ammonia nitrogen removal from aqueous solution usingzeolite modified by microwave-sodium acetate. J. Cent. South Univ. 2016, 23, 1345–1352. [Google Scholar] [CrossRef]
- Guaya, D.; Valderrama, C.; Farran, A.; Armijos, C.; Cortina, J.L. Simultaneous phosphate and ammonium removal from aqueous solution by a hydrated aluminum oxide modified natural zeolite. Chem. Eng. J. 2015, 271, 204–213. [Google Scholar] [CrossRef]
- Guaya, D.; Valderrama, C.; Farran, A.; Cortina, J.L. Modification of a natural zeolite with Fe(III) for simultaneous phosphate and ammonium removal from aqueous solutions. J. Chem. Technol. Biotechnol. 2016, 91, 1737–1746. [Google Scholar] [CrossRef]
- Guo, H.; Zhang, X.; Liu, J. Ion-exchange capability for ammonium removal using zeolite modified by potassium permanganate. Chem. Eng. Trans. 2016, 55, 163–168. [Google Scholar] [CrossRef]
- He, Y.; Lin, H.; Dong, Y.; Wang, L. Preferable adsorption of phosphate using lanthanum-incorporated porous zeolite: Characteristics and mechanism. Appl. Surf. Sci. 2017, 426, 995–1004. [Google Scholar] [CrossRef]
- Huang, H.; Xiao, X.; Yan, B.; Yang, L. Ammonium removal from aqueous solutions by using natural Chinese (Chende) zeolite as adsorbent. J. Hazard. Mater. 2010, 175, 247–252. [Google Scholar] [CrossRef]
- Huo, H.; Lin, H.; Dong, Y.; Cheng, H.; Wang, H.; Cao, L. Ammonia-nitrogen and phosphates sorption from simulated reclaimed waters bymodified clinoptilolite. J. Hazard. Mater. 2012, 229–230, 292–297. [Google Scholar] [CrossRef]
- Khosravi, A.; Esmhosseini, M.; Khezri, S. Removal of ammonium ion from aqueous solutions using natural zeolite: Kinetic, equilibrium and thermodynamic studies. Res. Chem. Intermed. 2014, 40, 2905–2917. [Google Scholar] [CrossRef]
- Langwaldt, J. Ammonium removal from water by eight natural zeolites: A comparative study. Sep. Sci. Technol. 2008, 43, 2166–2182. [Google Scholar] [CrossRef]
- Lin, L.; Wan, C.; Lee, D.; Lei, Z.; Liu, X. Ammonium assists orthophosphate removal from high-strength wastewaters by natural zeolite. Sep. Purif. Technol. 2014, 133, 351–356. [Google Scholar] [CrossRef]
- Moharami, S.; Jalali, M. Use of modified clays for removal of phosphorus from aqueous solutions. Environ. Monit. Assess. 2015, 187, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.; Araújo, A.; Azevedo, G.; Pereira, M.F.R.; Neves, I.C.; Machado, A.V. Kinetic and equilibrium studies of phosphorous adsorption: Effect of physical and chemical properties of adsorption agent. Ecol. Eng. 2015, 82, 527–530. [Google Scholar] [CrossRef]
- Pizzaro, C.; Rubio, M.A.; Escudey, M.; Albornoz, M.F.; Muñoz, D.; Denardin, J.; Fabris, J.D. Nanomagnetite-zeolite composites in the removal of arsenate from aqueous systems. J. Braz. Chem. Soc. 2015, 26, 1887–1896. [Google Scholar] [CrossRef]
- Sancho, I.; Licon, E.; Valderrama, C.; de Arespacochaga, N.; López-Palau, S.; Cortina, J.L. Recovery of ammonia from domestic wastewater effluents as liquidfertilizers by integration of natural zeolites and hollow fibre membrane contactors. Sci. Total Environ. 2017, 584–585, 244–251. [Google Scholar] [CrossRef]
- Song, W.; Shi, T.; Yang, D.; Ye, J.; Zhou, Y.; Feng, Y. Pretreatment effects on the sorption of Cr(VI) onto surfactantmodified zeolite: Mechanism analysis. J. Environ. Manag. 2015, 162, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Stefanakis, A.I.; Tsihrintzis, V.A. Use of zeolite and bauxite as filter media treating the effluent of Vertical Flow Constructed Wetlands. Microporpous Mesoporous Mater. 2012, 155, 106–116. [Google Scholar] [CrossRef]
- Wan, C.; Ding, S.; Zhang, C.; Tan, X.; Zou, W.; Liu, X.; Yang, X. Simultaneous recovery of nitrogen and phosphorus from sludge fermentation liquid by zeolite adsorption: Mechanism and application. Sep. Purif. Technol. 2017, 180, 1–12. [Google Scholar] [CrossRef]
- Yapsakli, K.; Aktan, C.K.; Mertoglu, B. Anammox-zeolite system acting as buffer to achieves table effluent nitrogen values. Biodegradation 2017, 28, 69–79. [Google Scholar] [CrossRef] [PubMed]
- You, X.; Valderrama, C.; Queroland, J.L.C.X. Recovery of Ammonium by Powder Synthetic Zeolites from Wastewater Effluents: Optimization of the Regeneration Step. Water Air Soil Pollut. 2017, 228, 396. [Google Scholar] [CrossRef]
- Zendehdel, M.; Zendehnam, A.; Hoseini, F.; Azarkish, M. Investigation of removal of chemical oxygen demand (COD) wastewater and antibacterial activity of nanosilver incorporated in poly (acrylamide-coacrylic acid)/NaY zeolite nanocomposite. Polym. Bull. 2015, 72, 1281–1300. [Google Scholar] [CrossRef]
- Lakdawala, M.M.; Patel, Y.S. Studies on Adsorption Capacity of Zeolite for Removal of Chemical and Bio-Chemical Oxygen Demands. Chem. J. 2015, 1, 139–143. [Google Scholar]
- Zhou, L.; Boyd, C.E. Total ammonia nitrogen removal from aqueous solutions by the natural zeolite, mordenite: A laboratory test and experimental study. Aquaculture 2014, 432, 252–257. [Google Scholar] [CrossRef]
- Vocciante, M.; De Folly D’Auris, A.; Finocchi, A.; Tagliabue, M.; Bellettato, M.; Ferrucci, A.; Reverberi, A.P.; Ferro, S. Adsorption of ammonium on clinoptilolite in presence of competing cations: Investigation on groundwater remediation. J. Clean. Prod. 2018, 198, 480–487. [Google Scholar] [CrossRef]
- Yin, H.; Yang, C.; Jia, Y.; Chen, H.; Gu, X. Dual removal of phosphate and ammonium from high concentrations of aquaculture wastewaters using an efficient two-stage infiltration system. Sci. Total Environ. 2018, 635, 936–946. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Du, J.; Chen, P.; Huang, X.; Chen, O. Enhanced efficiency of swine wastewater treatment by the composite of modified zeolite and a bioflocculant enriched from biological sludge. Environ. Technol. 2018, 39, 3096–3103. [Google Scholar] [CrossRef] [PubMed]
- Cyrus, S.J.; Reddy, B.G. Sorption and Desorption of Ammonium by Zeolite: Batch and Column Studies. J. Environ. Sci. Health Part A 2011, 46, 408–414. [Google Scholar] [CrossRef]
- Lagergren, S.Y. Zur Theorie der sogenannten adsorption gelosterstoffe. Handlingar 1898, 24, 1–39. [Google Scholar]
- Blanchard, G.; Maunaye, M.; Martin, G. Removal of heavy metals from waters by means of natural zeolites. Water Res. 1984, 18, 1501–1507. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.-J.; Hosseini-Bandegharaei, A.; Chao, H.-P. Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: A critical review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef] [PubMed]
- Freundlich, H.M.F. Over the adsorption in solution. J. Phys. Chem. 1906, 57, 385–471. [Google Scholar]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Hall, K.R.; Eagleton, L.C.; Acrivos, A.; Vermeulen, T. Poreand solid-diffusion kinetics in fixed-bed adsorption under constant-pattern conditions. Ind. Eng. Chem. Fundam. 1966, 5, 212–223. [Google Scholar] [CrossRef]
- Worch, E. Adsorption Technology in Water Treatment: Fundamentals, Processes, and Modeling; De Gruyter: Berlin, Germany, 2012. [Google Scholar]
- Verdouw, H.; Van Echteld, C.J.A.; Dekkers, E.M.J. Ammonia determination based on indophenol formation with sodium salicylate. Water Res. 1978, 12, 399–402. [Google Scholar] [CrossRef]
- (American Public Health Association) APHA. Standard Methods for Examination of Water and Wastewater, 23rd ed.; American Public Health Association: New York, NY, USA, 2017. [Google Scholar]
- Wang, S.; Peng, Y. Natural zeolites as effective adsorbents in water and wastewater treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar] [CrossRef]
- Zhao, C.; Zheng, Z.; Zhang, J.; Wen, D.; Tang, X. Adsorption characteristics of ammonium exchange by zeolite and the optimal application in the tertiary treatment of coking wastewater using response surface methology. Water Sci. Technol. 2013, 67, 619–627. [Google Scholar] [CrossRef]
- Wang, W.; Zhao, J.; Zhang, X.; Cheng, Y.; Wang, J.; Zhang, Y.; Li, L. Adsorption properties of ammonium nitrogen in seawater by natural zeolite. Chin. J. Environ. Eng. 2015, 9, 4281–4286. [Google Scholar]
- Leyva-Ramos, R.; Aguilar-Armenta, G.; Gonzalez-Gutierrez, L.V.; Guerrero-Coronado, R.M.; Mendoza-Barron, J. Ammonia exchange on clinoptilolite from mineral deposits located in Mexico. J. Chem. Technol. Biotechnol. 2004, 79, 651–657. [Google Scholar] [CrossRef]
- Ören, A.H.; Kaya, A. Factors affecting adsorption characteristics of Zn2+ on two natural zeolites. J. Hazard. Mater. 2006, 131, 59–65. [Google Scholar] [CrossRef]
- Ziyath, A.M.; Mahbub, P.; Goonetilleke, A.; Adebajo, M.O.; Kokot, S.; Oloyede, A. Influence of physical and chemical parameters on the treatment of heavy metals in polluted stormwater using zeolite: A review. J. Water Resour. Prot. 2011, 3, 758–767. [Google Scholar] [CrossRef]
- Martins, T.H.; Souza, T.S.O.; Foresti, E. Ammonium removal from landfill leachate by Clinoptilolite adsorption followed by bioregeneration. J. Environ. Chem. Eng. 2017, 5, 63–68. [Google Scholar] [CrossRef]
- Alshameri, A.; Chunjie, Y.; Al-Ani, Y.; Dawood, A.S.; Ibrahim, A.; Zhou, C.; Wang, H. An investigation into the adsorption removal of ammonium by salt activated Chinese (Hulaodu) natural zeolite: Kinetics, isotherms, and thermodynamics. J. Taiwan Inst. Chem. Eng. 2014, 45, 554–564. [Google Scholar] [CrossRef]
- Karadag, D.; Koc, Y.; Turan, M.; Armagan, B. Removal of ammonium ion from aqueous solution using natural Turkish clinoptilolite. J. Hazard. Mater. 2006, 136, 604–609. [Google Scholar] [CrossRef]
- Sarioglu, M. Removal of ammonium from municipal wastewater using natural Turkish (Dogantepe) zeolite. Sep. Purif. Technol. 2005, 41, 1–11. [Google Scholar] [CrossRef]
- Townsend, R.P.; Loizidou, M. Ion exchange properties of natural clinoptilolite, ferrierite and mordenite: 1. Sodium—Ammonium equilibria. Zeolites 1984, 4, 191–195. [Google Scholar] [CrossRef]
- Thomas, J.M.; Thomas, W.J. Principles and Practice of Heterogeneous Catalysis; John Wiley &Sons: Hoboken, NJ, USA, 2014; p. 1560. [Google Scholar]
- Wen, D.; Ho, Y.S.; Tang, X. Comparative sorption kinetic studies of ammonium onto zeolite. J. Hazard. Mater. 2006, 133, 252–256. [Google Scholar] [CrossRef]
- Taddeo, R.; Prajapati, S.; Lepistö, R. Optimizing ammonium adsorption on natural zeolite for wastewaters with high loads of ammonium and solids. J. Porous Mater. 2017, 24, 1545–1554. [Google Scholar] [CrossRef]
- Shaban, M.; AbuKhadra, M.R.; Nasief, F.M.; El-Salam, H.A. Removal of ammonia from aqueous solutions, ground water, and wastewater using mechanically activated clinoptilolite and synthetic zeolite-a: Kinetic and equilibrium studies. Water Air Soil Pollut. 2017, 228, 450. [Google Scholar] [CrossRef]
- Cheng, Z.; Ding, W. Ammonium removal from water by naturaland modified zeolite: Kinetic, equilibrium, andthermodynamic studies. Desalin. Water Treat. 2015, 55, 978–985. [Google Scholar] [CrossRef]
- Sánchez-Hernández, R.; Padilla, I.; López-Andrés, S.; López-Delgado, A. Al-Waste-Based Zeolite Adsorbent Used for the Removal of Ammonium from Aqueous Solutions. Int. J. Chem. Eng. 2018, 2018, 1256197. [Google Scholar] [CrossRef]
- Celik, S.M.; Ozdemir, H.B.; Turan, E.Y.; Koyuncu, I.; Atesok, G.; Sarikaya, H.Z. Removal of ammonia by natural clay minerals using fixed and fluidised bed column reactors. Water Sci. Technol. 2001, 1, 81–88. [Google Scholar] [CrossRef]
- Hedström, A.; Amofah, L.R. Adsorption and desorption of ammonium by clinoptilolite adsorbent in municipal wastewater treatment systems. J. Environ. Eng. Sci. 2008, 7, 53–61. [Google Scholar] [CrossRef]
- Drizo, A.; Frost, C.A.; Grace, J.; Smith, K.A. Physico-chemical screening of phosphate-removing substrates for use in constructed wetland systems. Water Res. 1999, 33, 3595–3602. [Google Scholar] [CrossRef]
| Wastewater Type | Zeolite Type | Pollutant | Removal Rates | Reference |
|---|---|---|---|---|
| Olive mill wastewater | Natural (normal zeolite particles) | Nitrate Phosphorus Potassium | 78% 48.3% 66.6% | [13] |
| Olive mill wastewater | Natural (nanoparticles) | Nitrate Phosphorus Potassium | 92.79% 92.64% 99.96% | [14] |
| Simulated swine wastewater | Chemical modification with NaCl | Ammonium | 40%–95% | [15] |
| Dairy farm wastewater treated in constructed wetlands | Natural | Phosphorus Ammonium | 86%–99% 88%–99% | [16] |
| Dairy industry wastewater | Organo-zeolites | COD Nitrate nitrogen Phosphate | 50% 70% 20% | [17] |
| Dairy processing waters | Natural | COD | 76% | [18] |
| Aqueous solution/carcass leachates | Naturaland Mg2+ modified | Ammonium | 8.173 mg/g 7.759 mg/g | [19] |
| Municipal wastewater | Natural | Ammonium | 5.03 mg/g 75.6% | [20] |
| Aqueous solution | Modified with fly ash | Ammonium | 41.73%–45.25% | [21] |
| Review of acid mine drainage urban runofflandfill leachate | Natural/synthetic | Heavy metals | Zn (30%–95%)/Cu (33%–100%) Cd (54%–99%)/Cu (31%–90%)/Zn (81.892%)/Ammonium 78% Ammonium (over 50%)/Pb (71%)/Cd (74%) | [22] |
| Aqueous solution | Natural | Ammonium | 22.90 mg/g | [23] |
| Aqueous solution | Modified by microwave and sodium acetate | Ammonium | 92.90% | [24] |
| Secondary wastewater effluents | Natural Modified (Z-Al) | Phosphate (single) Ammonium(single) Phosphate (single) Ammonium (single) | 0.6 mg/g 33 mg/g 7.0 mg/g 30 mg/g | [25] |
| Municipal wastewater | Natural Modified (Z-Fe) | Phosphate (single) Ammonium (single) Phosphate (single) Ammonium (single) | 0.6 mg/g 33 mg/g 3.4 mg/g 27 mg/g | [26] |
| Aqueous solution | Natural Modified with potassium permanganate | Ammonium | 5.85 mg/g 3.68 mg/g | [27] |
| Aqueous solution | Modified with lanthanum oxide | Phosphorus Ammonium | 8.96 21.2 mg/g | [28] |
| Aqueous solution | Natural | Ammonium | 67.4%–81.1% | [29] |
| Simulated reclaimed wastewater | Modified with NaCl | Ammonia, nitrogen Phosphorus | 98.46% 99.8% | [30] |
| Aqueous solution | Natural | Ammonium | 75%–95.3% | [31] |
| Aqueous solution | Natural (eight different types) | Ammonium | 15.7%–32.4% | [32] |
| Municipal wastewater | Natural | Phosphorus Ammonium | 46%–100% 70% | [33] |
| Aqueous solution | Natural Modified | Phosphate | 0.28–1.82 mg/g 1.31–1.97 mg/g | [34] |
| Aqueous solution | Natural | Phosphate | Up to 26.48 mg/g | [35] |
| Aqueous solution | Modified with nanosized particles of magnetite | Arsenate | Up to 5.2 mg/g | [36] |
| Post-treated municipal wastewater | Natural | Ammonium | 23 ± 0.8 mg/g | [37] |
| Aqueous solution | Modified with cetylpyridinium chloride (CPC) | Hexavalent chromium | 5.8 mg/g | [38] |
| Simulated municipal wastewater (vertical flow constructed wetlands) | Natural | Ammonium | >90% | [39] |
| Fermentation liquids | Natural | Ammonium Phosphate | 94.06% 98.28% | [40] |
| Synthetic medium (Wastewater treatment in an ANAMMOX trickling filter) | Natural | Nitrogen | 95% | [41] |
| Aqueous solution | Modified with coal fly ash (Ze–Na) and potassium (Ze–K) | Ammonium | 109 ± 4 mg/g 33 ± 1 mg/g | [42] |
| Wastewater | Modified with silver | COD | 75% | [43] |
| Sugar industry wastewater | Natural | COD | 1%–26.6% | [44] |
| Aquaculture wastewater | Natural | Total ammonium nitrogen | 8.7 mg/g | [45] |
| Groundwater remediation | Natural | Ammonium | 14 mg/g | [46] |
| Aquaculture wastewater | Modified | Ammonium Phosphorus | 99.4% 99% | [47] |
| Swine wastewater | Modified by integrating calcinations with MgO at 400 °C (single) Bioflocculant and modified by integrating calcinations with MgO at 400 °C | COD Ammonia COD Ammonia Turbidity | almost 0% 73.4% 71.8% 54.5% 81.2% | [48] |
| Components | % |
|---|---|
| SiO2 | 69.62 |
| Al2O3 | 13.62 |
| TiO3 | 0.11 |
| Fe2O3 | 0.75 |
| CaO | 3.28 |
| MgO | 0.90 |
| Na2O | 0.55 |
| K2O | 2.94 |
| Loss of ignition (at 1050 °C) | 8.23 |
| Pseudo-First Order Model | Pseudo-Second Order Model | ||||||
|---|---|---|---|---|---|---|---|
| C0 (mg/L) | qe, exp (mg/g) | qe, cal (mg/g) | k1 (min−1) | R2 | qe, cal (mg/g) | k2 (g/mg min) | R2 |
| 500 | 7.36 | 5.16 | 0.002 | 0.932 | 7.52 | 0.001 | 0.997 |
| 1000 | 11.48 | 10.43 | 0.002 | 0.877 | 11.35 | 0.002 | 0.999 |
| 2000 | 15.79 | 14.59 | 0.004 | 0.967 | 15.86 | 0.001 | 0.998 |
| 3000 | 17.18 | 18.79 | 0.002 | 0.911 | 17.12 | 0.0008 | 0.998 |
| 4000 | 20.23 | 19.94 | 0.002 | 0.936 | 20.28 | 0.0003 | 0.997 |
| 5000 | 26.01 | 25.67 | 0.004 | 0.965 | 26.11 | 0.0006 | 0.995 |
| Freundlich Parameters | Langmuir Parameters | ||||
|---|---|---|---|---|---|
| 1/n | kf (mg/g)/(mg/L) | R2 | 1/qm (g/mg) | kL (L/mg) | R2 |
| 0.77 | 0.47 | 0.981 | 0.21 | 0.13 | 0.750 |
| Zeolite Granulometry (mm) | NH4+-N Absorbed Mass on Zeolite (mg/g) for Initial NH4+-N Concentration of 5000 mg/L | Time Required for the Final Desorption Concentration (min) | Final NH4+-N Concentration in the Aqueous Solution after Desorption (mg/L) | NH4+-N Recovery (%) |
|---|---|---|---|---|
| 4.0–5.0 | 3.638 | 11520 | 132.492 | 2.77 |
| 2.8–4.0 | 3.648 | 11520 | 160.402 | 3.35 |
| 2.0–2.8 | 3.671 | 11520 | 188.204 | 3.80 |
| 1.8–2.0 | 3.675 | 11520 | 196.330 | 4.06 |
| 0.71–1.0 | 3.798 | 11520 | 212.385 | 4.39 |
| Total Kjeldahl Nitrogen (TKN) (mg N/L) | N-NH4+ (mg/L) | N-NO2− (mg/L) | N-NO3− (mg/L) | P-PO43− (mg/L) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Secondary Cheese-Whey (SCW) Dilution Ration | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 | |
| 100% | Initial | 243 | 243 | 235.4 | 45.3 | 35.4 | 43.4 | 0 | 0 | 0 | 0.16 | 0.20 | 0.25 | 195.86 | 190.00 | 194.19 |
| Final | 73 | 73 | 0 | 0.41 | 0.9 | 0.9 | 0 | 0 | 0 | 2.50 | 0.60 | 0.36 | 45.92 | 141.03 | 180.62 | |
| 75% | Initial | 184 | 186 | 173 | 33.2 | 30.8 | 31.7 | 0 | 0 | 0 | 0.00 | 0.00 | 0.00 | 145.3 | 147.6 | 148.4 |
| Final | 50 | 48 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.18 | 0.22 | 0.19 | 33.2 | 99.8 | 137.6 | |
| 50% | Initial | 118.9 | 120.2 | 118.9 | 23.4 | 21.9 | 22.5 | 0 | 0 | 0 | 0.00 | 0.00 | 0.00 | 98.6 | 95.3 | 99.3 |
| Final | 32.3 | 30.9 | 0 | 0.07 | 0 | 0 | 0 | 0 | 0 | 0.09 | 0.10 | 0.12 | 20.3 | 65.8 | 80.9 | |
| COD (mg/L) | TKN (mg/L) | N-NO2− (mg/L) | N-NO3− (mg/L) | P-PO43− (mg/L) | |||
|---|---|---|---|---|---|---|---|
| Columns maintained at room temperature | Day 1 | Initial | 2350 ± 45 | 126.21 ± 2.1 | 0.13 ± 0.01 | 18.19 ± 0.28 | 25.02 ± 0.58 |
| Final | 1808 ± 252 | 7.36 ± 1.01 | 0.10 ± 0.01 | 10.52 ± 0.69 | 1.01 ± 0.02 | ||
| Day 2 | Initial | 2500 ± 32 | 110.47 ± 2.3 | 0.11 ± 0.01 | 14.02 ± 4.78 | 13.87 ± 9.39 | |
| Final | 2080 ± 20 | 8.78 ± 0.58 | 0.14 ± 0.001 | 9.67 ± 0.78 | 0.56 ± 0.01 | ||
| Day 3 | Initial | 2400 ± 15 | 115.26 ± 3.5 | 0.41 ± 0.36 | 17.19 ± 0.79 | 18.57 ± 2.53 | |
| Final | 2000 ± 204 | 10.58 ± 1.89 | 0.18 ± 0.03 | 8.56 ± 1.84 | 9.36 ± 1.53 | ||
| Day 4 | Initial | 2350 ± 14 | 128.56 ± 3.7 | 1.79 ± 0.20 | 7.23 ± 0.39 | 29.34 ± 4.69 | |
| Final | 1685 ± 490 | 9.78 ± 1.48 | 1.055 ± 0.51 | 15.47 ± 0.42 | 7.77 ± 1.29 | ||
| Day 5 | Initial | 2600 ± 18 | 117.45 ± 1.9 | 0.98 ± 0.60 | 7.41 ± 5.79 | 15.63 ± 0.86 | |
| Final | 1795 ± 434 | 11.01 ± 1.58 | 0.60 ± 0.02 | 10.44 ± 1.35 | 11.12 ± 1.68 | ||
| Day 6 | Initial | 2850 ± 32 | 123.98 ± 2.8 | 0.58 ± 0.03 | 9.14 ± 2.17 | 45.65 ± 2.11 | |
| Final | 1805 ± 100 | 15.89 ± 1.29 | 0.39 ± 0.02 | 0.4 ± 0.14 | 32.14 ± 2.94 | ||
| Refrigerated columns | Day 1 | Initial | 2350 ± 29 | 126.21 ± 4.2 | 0.13 ± 0.01 | 18.19 ± 0.89 | 25.02 ± 2.12 |
| Final | 2060 ± 149 | 8.01 ± 0.95 | 0.09 ± 0.02 | 11.69 ± 0.72 | 1.15 ± 0.02 | ||
| Day 2 | Initial | 2500 ± 56 | 110.47 ± 3.8 | 0.13 ± 0.05 | 17.55 ± 0.007 | 10.85 ± 3.94 | |
| Final | 1793 ± 585 | 8.52 ± 0.42 | 0.08 ± 0.02 | 10.51 ± 1.11 | 0.78 ± 0.05 | ||
| Day 3 | Initial | 2400 ± 87 | 115.26 ± 4.9 | 0.2 ± 0.05 | 15.18 ± 1.16 | 18.23 ± 0.94 | |
| Final | 2245 ± 84 | 12.96 ± 2.36 | 0.13 ± 0.02 | 0.54 ± 0.31 | 8.45 ± 0.66 | ||
| Day 4 | Initial | 2350 ± 54 | 128.56 ± 2.5 | 0.07 ± 0.007 | 11.24 ± 1.08 | 21.72 ± 2.1 | |
| Final | 1901 ± 331 | 11.22 ± 3.11 | 0.10 ± 0.06 | 14.62 ± 0.17 | 4.97 ± 4.68 | ||
| Day 5 | Initial | 2600 ± 68 | 117.45 ± 5.6 | 0.22 ± 0.16 | 8.31 ± 0.26 | 15.47 ± 0.50 | |
| Final | 2158 ± 34 | 14.63 ± 2.89 | 0.15 ± 0.02 | 9.75 ± 0.51 | 7.46 ± 7.32 | ||
| Day 6 | Initial | 2850 ± 44 | 123.98 ± 3.7 | 0.59 ± 0.02 | 12.29 ± 1.05 | 45.65 ± 2.6 | |
| Final | 2200 ± 60 | 17.89 ± 2.78 | 0.41 ± 0.02 | 9.59 ± 4.54 | 28.68 ± 2.84 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotoulas, A.; Agathou, D.; Triantaphyllidou, I.E.; Tatoulis, T.I.; Akratos, C.S.; Tekerlekopoulou, A.G.; Vayenas, D.V. Zeolite as a Potential Medium for Ammonium Recovery and Second Cheese Whey Treatment. Water 2019, 11, 136. https://doi.org/10.3390/w11010136
Kotoulas A, Agathou D, Triantaphyllidou IE, Tatoulis TI, Akratos CS, Tekerlekopoulou AG, Vayenas DV. Zeolite as a Potential Medium for Ammonium Recovery and Second Cheese Whey Treatment. Water. 2019; 11(1):136. https://doi.org/10.3390/w11010136
Chicago/Turabian StyleKotoulas, Aggelos, Dimitra Agathou, Irene E. Triantaphyllidou, Triantafyllos I. Tatoulis, Christos S. Akratos, Athanasia G. Tekerlekopoulou, and Dimitrios V. Vayenas. 2019. "Zeolite as a Potential Medium for Ammonium Recovery and Second Cheese Whey Treatment" Water 11, no. 1: 136. https://doi.org/10.3390/w11010136
APA StyleKotoulas, A., Agathou, D., Triantaphyllidou, I. E., Tatoulis, T. I., Akratos, C. S., Tekerlekopoulou, A. G., & Vayenas, D. V. (2019). Zeolite as a Potential Medium for Ammonium Recovery and Second Cheese Whey Treatment. Water, 11(1), 136. https://doi.org/10.3390/w11010136








