Riverbank Filtration for the Water Supply on the Nakdong River, South Korea
Abstract
:1. Introduction
2. Materials and Methods
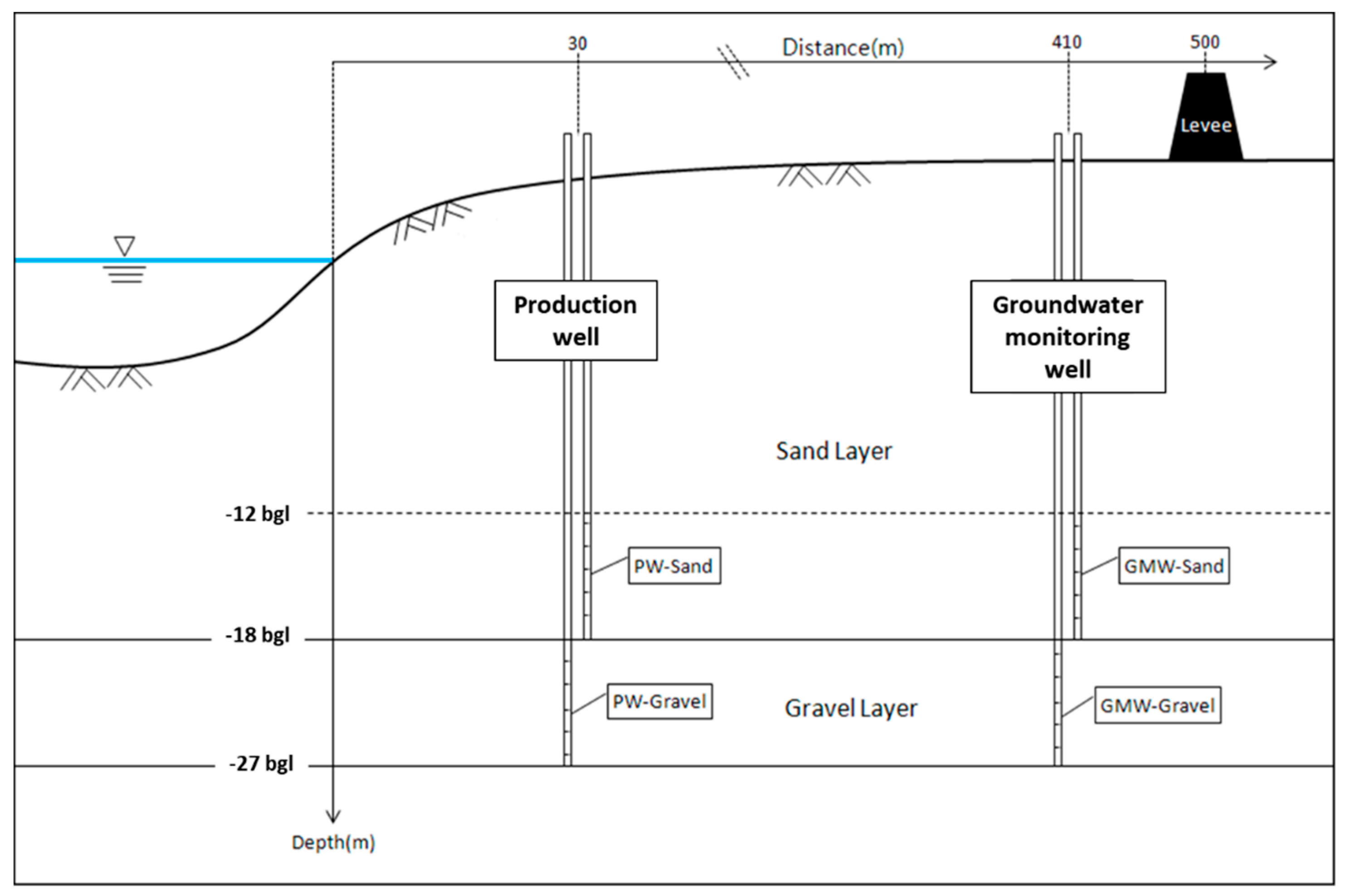
2.1. Field Site
2.2. Analytical Methods
3. Results and Discussion
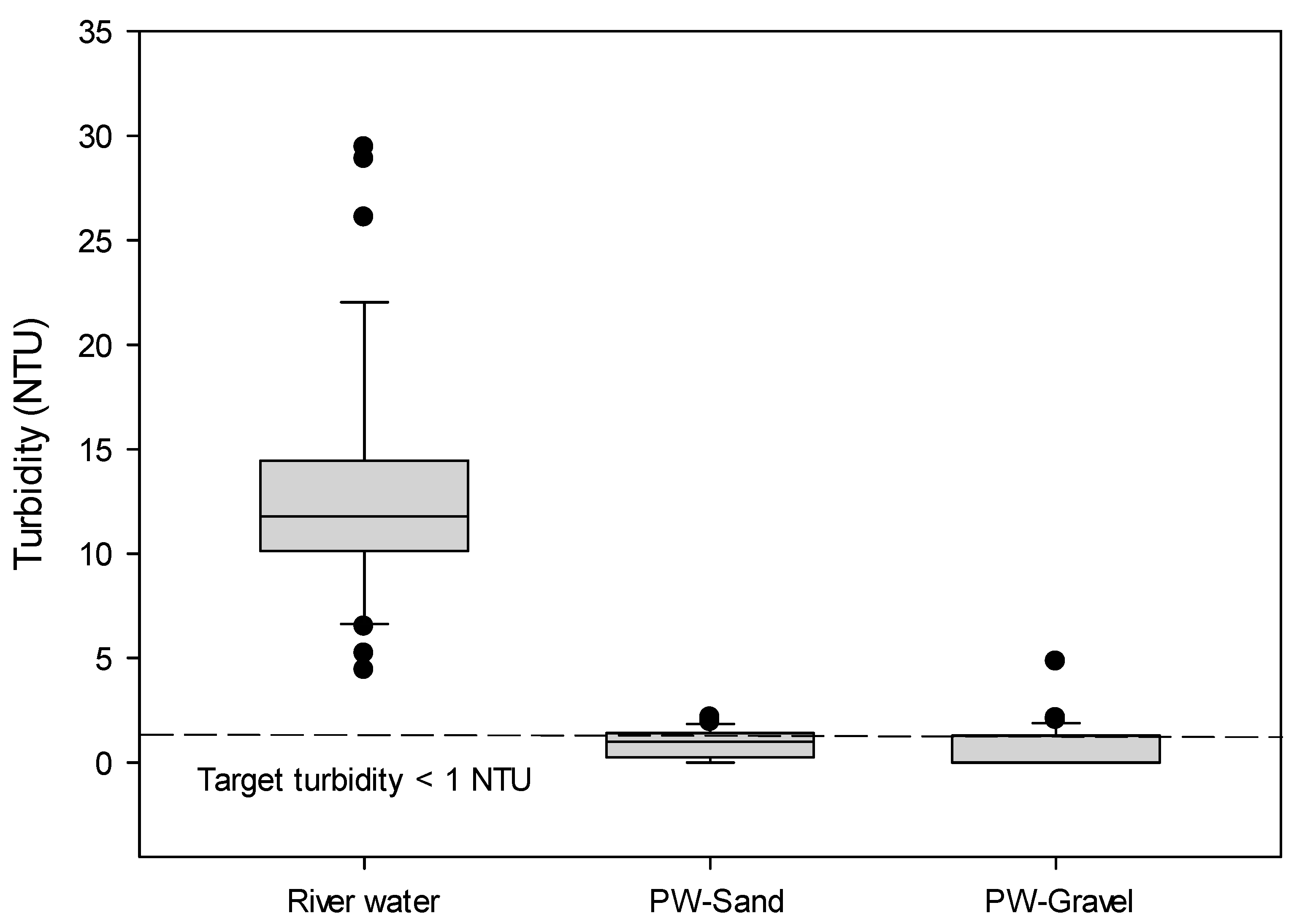
3.1. Turbidity
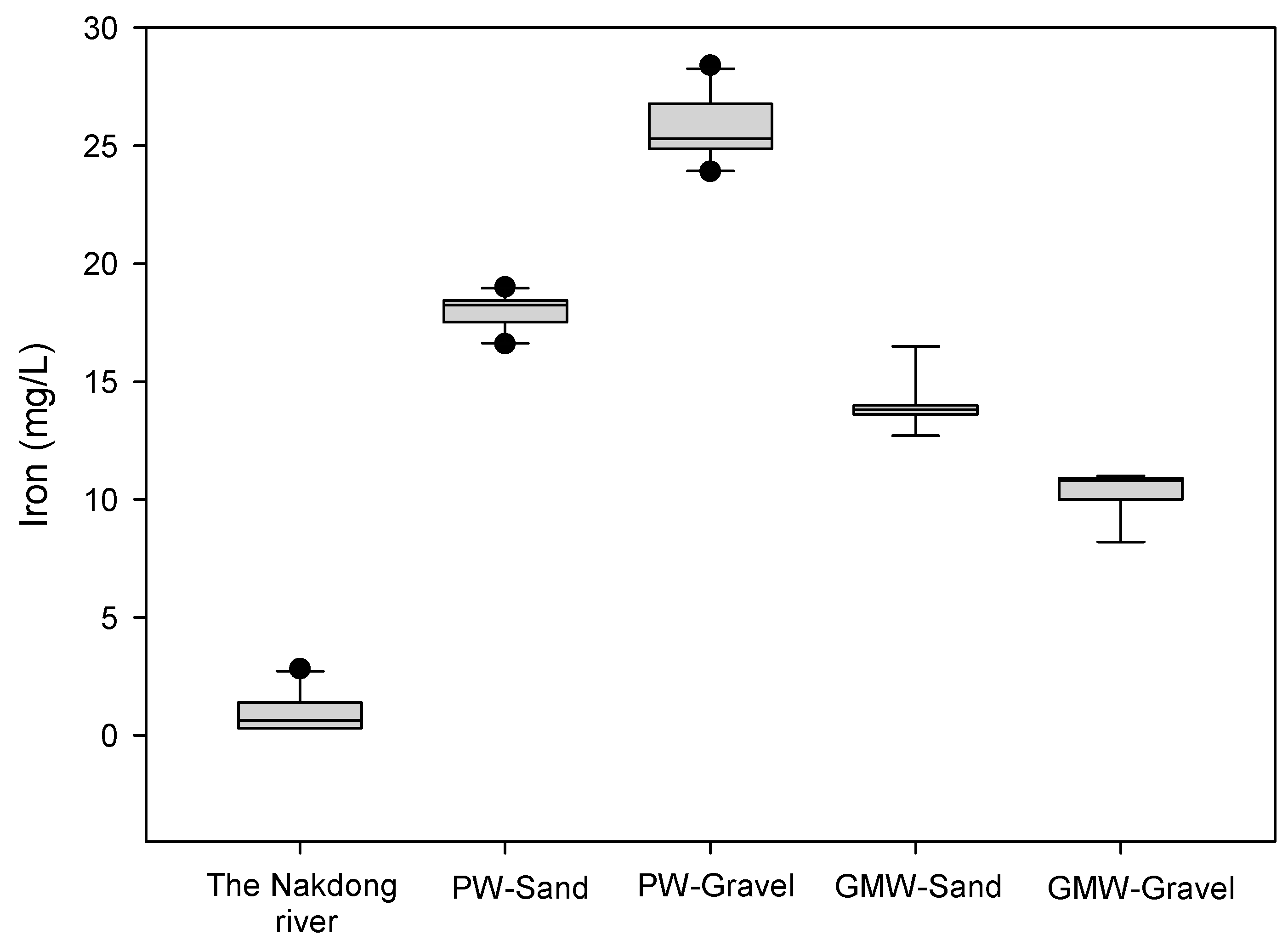
3.2. Iron and Manganese
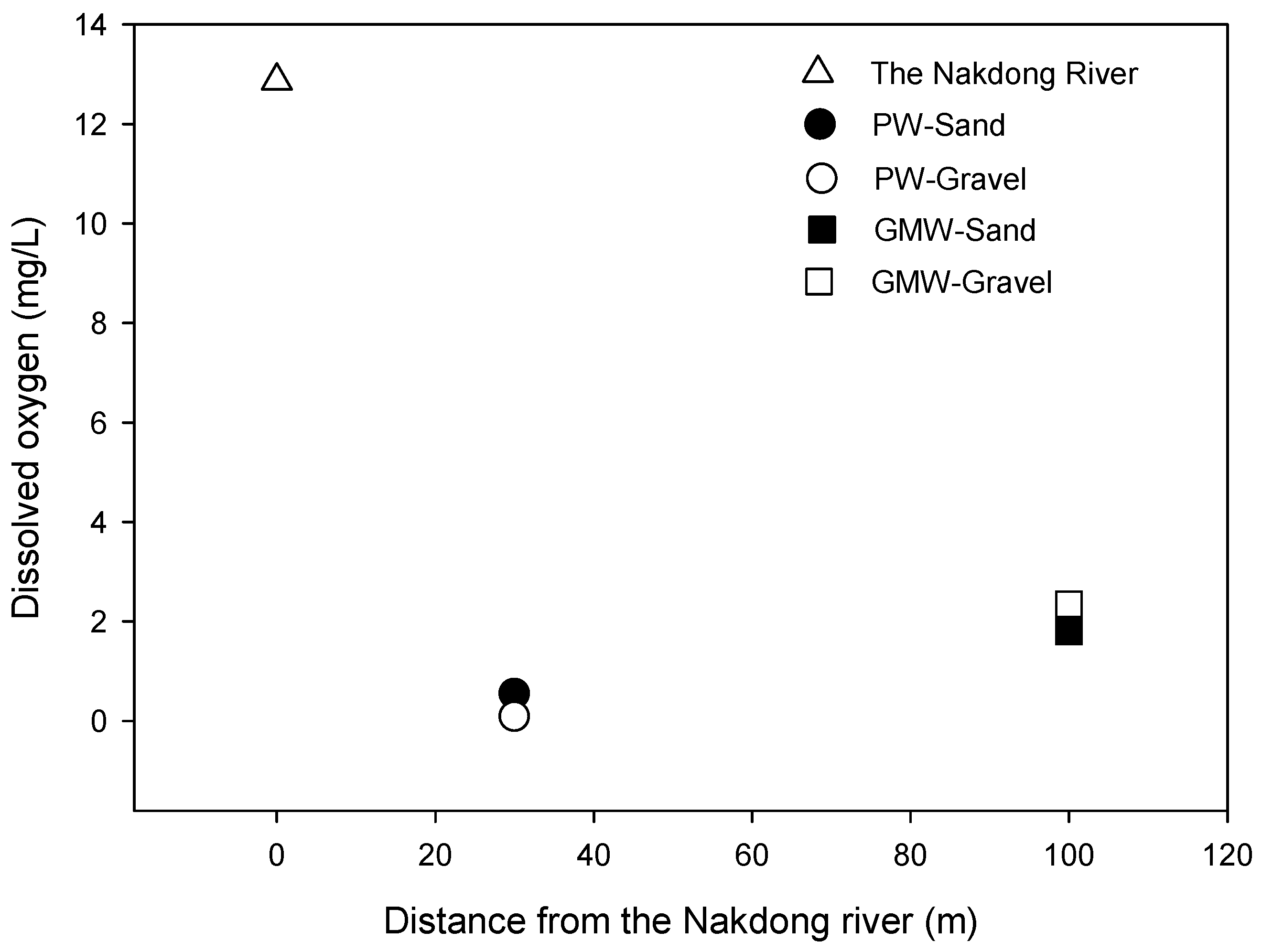
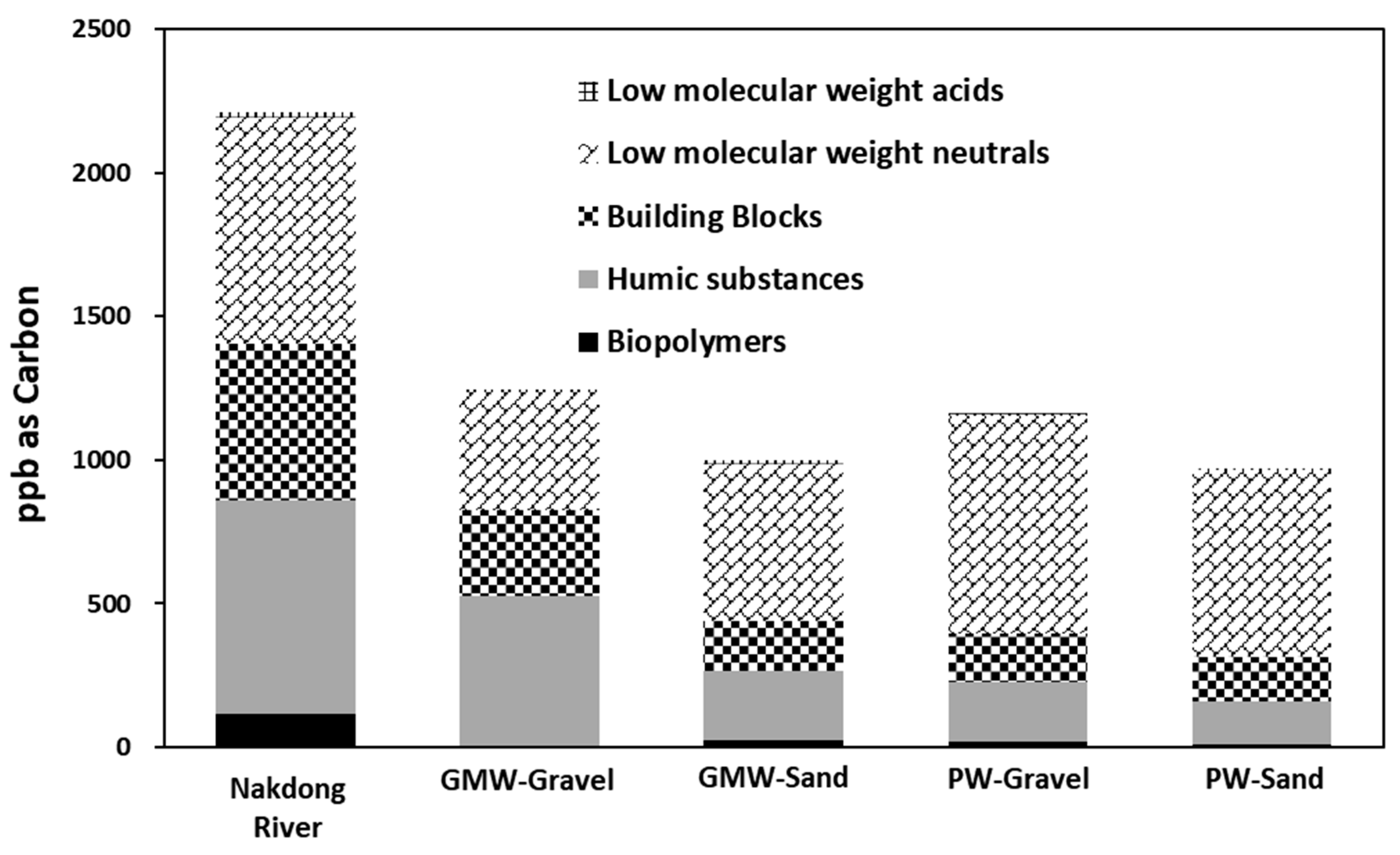
3.3. Bulk Organic Matter
3.3.1. Biochemical Oxygen Demand (BOD5)
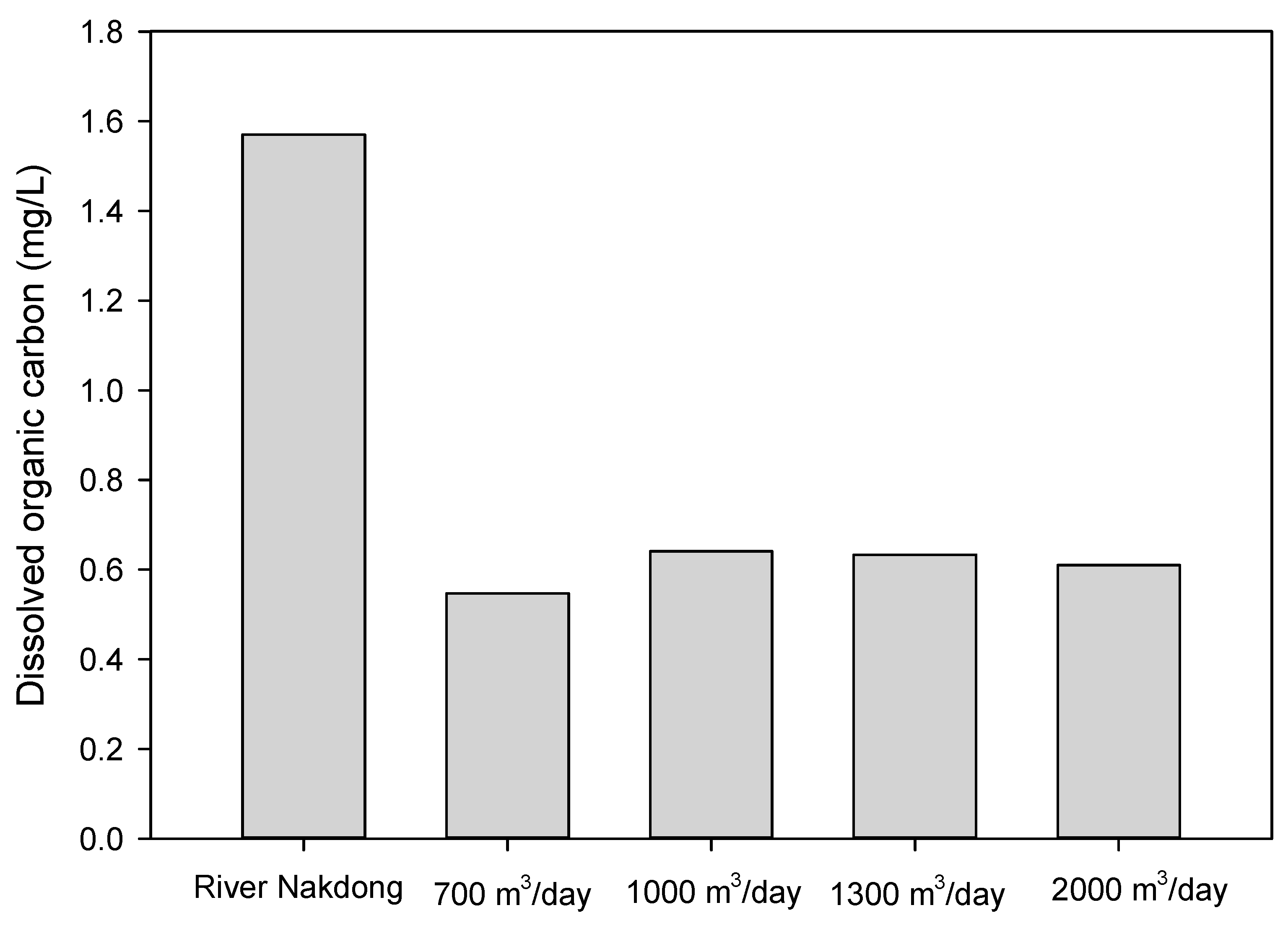
3.3.2. Dissolved Organic Carbon and UVA254
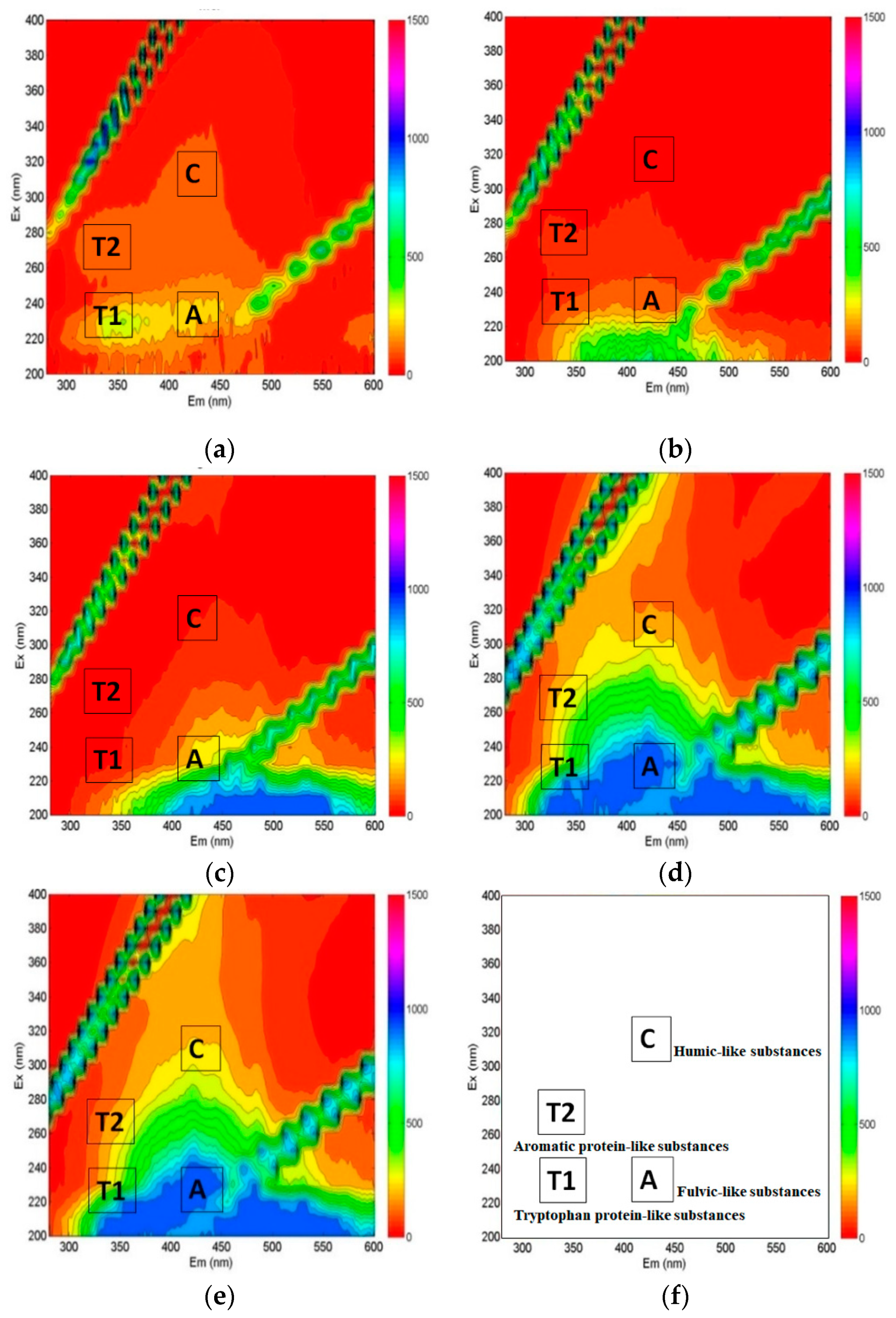
3.3.3. Dissolved Organic Matter’s Characteristics
3.4. Effect of Different Pumping Rates
4. Conclusions
- -
- The turbidity was effectively removed via RBF, although there were significant turbidity increases in the river water.
- -
- The iron concentrations from the RBF wells (i.e., PW-Sand and PW-Gravel) were higher than those of the groundwater monitoring wells (GMW-Sand and GMW-Gravel) and were 18.1 ± 0.8 and 25.9 ± 1.3 mg/L for the PW-Sand and PW-Gravel, respectively, during the study. The occurrence of a high iron concentration was due to the biodegradation of dissolved organic matter, which led to the reduced redox potential during soil passage. The reduced zone occurred between the river and RBF wells, which enhanced the mobilization of iron under more reducing conditions compared to that of land-side groundwater.
- -
- As a result of the dissolved organic matter characteristics via LC-OCD and fluorescence EEM, the biopolymers contained in the river were effectively removed while passing through the aquifer. It was also confirmed that most of the humic components, which are difficult to reduce biologically and were detected from the land-side groundwater, could influence the quality of the RBF filtrate.
- -
- The RBF wells (PW-Sand and PW-Gravel) in this study did not show any changes with respect to turbidity and DOC at different pumping rates (700, 1000, 1300 and 2000 m3/day).
- -
- Vertical wells at different layers (sand and gravel layers in the aquifer) were tested in this study in order to determine the depth of the laterals for the horizontal wells. This study used vertical wells in order to investigate the RBF site before the construction of the horizontal collectors, which usually cost much more than vertical wells.
Author Contributions
Funding
Conflicts of Interest
References
- Delpla, I.; Jung, A.-V.; Baures, E.; Clement, M.; Thomas, O. Impacts of climate change on surface water quality in relation to drinking water production. Environ. Int. 2009, 35, 1225–1233. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.-C.; Shin, H.-J.; Nguyen, T.T.; Tenhunen, J. Water policy reforms in South Korea: A historical review and ongoing challenges for sustainable water governance and management. Water 2017, 9, 717. [Google Scholar] [CrossRef]
- Pryor, F.L. Water stress and water wars. Econ. Peace Secur. J. 2007, 2, 7–18. [Google Scholar] [CrossRef]
- Kim, J.-S.; Jain, S. Precipitation trends over the Korean peninsula: Typhoon-induced changes and a typology for characterizing climate-related risk. Environ. Res. Lett. 2011, 6, 034033. [Google Scholar] [CrossRef]
- Lee, J.-H.; Hamm, S.-Y.; Cheong, J.-Y.; Kim, H.-S.; Ko, E.-J.; Lee, K.-S.; Lee, S.-I. Characterizing riverbank-filtered water and river water qualities at a site in the lower Nakdong River basin, Republic of Korea. J. Hydrol. 2009, 376, 209–220. [Google Scholar] [CrossRef]
- Greskowiak, J.; Prommer, H.; Massmann, G.; Johnston, C.D.; Nützmann, G.; Pekdeger, A. The impact of variably saturated conditions on hydrogeochemical changes during artificial recharge of groundwater. Appl. Geochem. 2005, 20, 1409–1426. [Google Scholar] [CrossRef]
- Henzler, A.F.; Greskowiak, J.; Massmann, G. Modeling the fate of organic micropollutants during river bank filtration (Berlin, Germany). J. Contam. Hydrol. 2014, 56, 78–92. [Google Scholar] [CrossRef]
- Kim, H.-C.; Lee, W.M.; Lee, S.; Choi, J.; Maeng, S.K. Characterization of organic precursors in DBP formation and AOC in urban surface water and their fate during managed aquifer recharge. Water Res. 2017, 123 (Suppl. C), 75–85. [Google Scholar] [CrossRef]
- Bertelkamp, C.; Reungoat, J.; Cornelissen, E.R.; Singhal, N.; Reynisson, J.; Cabo, A.J.; van der Hoek, J.P.; Verliefde, A.R.D. Sorption and biodegradation of organic micropollutants during river bank filtration: A laboratory column study. Water Res. 2014, 52, 231–241. [Google Scholar] [CrossRef]
- Maeng, S.K.; Sharma, S.K.; Abel, C.D.T.; Magic-Knezev, A.; Amy, G.L. Role of biodegradation in the removal of pharmaceutically active compounds with different bulk organic matter characteristics through managed aquifer recharge: Batch and column studies. Water Res. 2011, 45, 4722–4736. [Google Scholar] [CrossRef]
- Maeng, S.K.; Sharma, S.K.; Abel, C.D.T.; Magic-Knezev, A.; Song, K.-G.; Amy, G.L. Effects of effluent organic matter characteristics on the removal of bulk organic matter and selected pharmaceutically active compounds during managed aquifer recharge: Column study. J. Contam. Hydrol. 2012, 140–141, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Dillon, P. Future management of aquifer recharge. Hydrogeol. J. 2005, 13, 313–316. [Google Scholar] [CrossRef]
- Sharma, S.K.; Chaweza, D.; Bosuben, N.; Holzbecher, E.; Amy, G. Framework for feasibility assessment and performance analysis of riverbank filtration systems for water treatment. J. Water Supply Res. Technol. 2012, 61, 73–81. [Google Scholar] [CrossRef]
- Rustler, M.; Grützmacher, G. Bank Filtration Simulator-Manual, TECHNEAU Executive Summary. 2009. Available online: https://www.kompetenz-wasser.de/wp-content/uploads/2017/05/d5-2-5.pdf (accessed on 4 April 2018).
- Holzbecher, E.; Grützmacher, G.; Amy, G.; Wiese, B.; Sharma, S.K. The Bank Filtration Simulator—A MATLAB GUI. In Environmental Informatics and Industrial Ecology, Proceedings of the 22nd International Conference on Informatics for Environmental Protection (Enviroinfo 2008), Lüneburg, Germany, 10–12 September 2008; Möller, A., Page, B., Schreiber, M., Eds.; Shaker: Aachen, Germany, 2008. [Google Scholar]
- Chow, A.T.; Dahlgren, R.A.; Zhang, Q.; Wong, P.K. Relationships between specific ultraviolet absorbance and trihalomethane precursors of different carbon sources. J. Water Supply Res. Technol. 2008, 57, 471–480. [Google Scholar] [CrossRef]
- Ma, N.; Zhang, Y.; Quan, X.; Fan, X.; Zhao, H. Performing a microfiltration integrated with photocatalysis using an Ag-TiO2/HAP/Al2O3 composite membrane for water treatment: Evaluating effectiveness for humic acid removal and anti-fouling properties. Water Res. 2010, 44, 6104–6114. [Google Scholar] [CrossRef]
- Korean Ministry of Environment. Water Quality Standard and Test; Korean Ministry of Environment: Sejong City, Korea, 2011.
- Park, J.W.; Kim, H.C.; Meyer, A.S.; Kim, S.; Maeng, S.K. Influences of NOM composition and bacteriological characteristics on biological stability in a full-scale drinking water treatment plant. Chemosphere 2016, 160, 189–198. [Google Scholar] [CrossRef]
- Huber, S.A.; Balz, A.; Abert, M.; Pronk, W. Characterisation of aquatic humic and non-humic matter with size-exclusion chromatography–organic carbon detection–organic nitrogen detection (LC-OCD-OND). Water Res. 2011, 45, 879–885. [Google Scholar] [CrossRef]
- Kumar, P.; Mehrotra, I.; Gupta, A.; Kumar, S. Riverbank Filtration: A Sustainable Process to Attenuate Contaminants during Drinking Water Production. J. Sustain. Dev. Energy Water Environ. Syst. 2018, 6, 150–161. [Google Scholar] [CrossRef]
- Vanderzalm, J.L.; Page, D.W.; Barry, K.E.; Dillon, P.J. A comparison of the geochemical response to different managed aquifer recharge operations for injection of urban storm water in a carbonate aquifer. Appl. Geochem. 2010, 25, 1350–1360. [Google Scholar] [CrossRef]
- Vanderzalm, J.L.; Page, D.W.; Barry, K.E.; Scheiderich, K.; Gonzalez, D.; Dillon, P.J. Probabilistic approach to evaluation of metal(loid) fate during stormwater aquifer storage and recovery. Clean Soil Air Water 2016, 44, 1672–1684. [Google Scholar] [CrossRef]
- Maeng, S.K.; Sharma, S.K.; Amy, G.; Magic-Knezev, A. Fate of effluent organic matter (EfOM) and natural organic matter (NOM) through riverbank filtration. Water Sci. Technol. 2008, 57, 1999–2007. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.J.; Conrad, D.; Kothawala, D.N.; Baulch, H.M. Selective removal of dissolved organic matter affects the production and speciation of disinfection byproducts. Sci. Total Environ. 2019, 652, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Artinger, R.; Buckau, G.; Geyer, S.; Fritz, P.; Wolf, M.; Kim, J.I. Characterization of groundwater humic substances: Influence of sedimentary organic carbon. Appl. Geochem. 2000, 15, 97–116. [Google Scholar] [CrossRef]
- Schmidt, C.; Lange, F.; Brauch, H.; Kuhn, W. Experiences with riverbank filtration and infiltration in Germany. Proceeding of the International Symposium of Artificial Recharge of Groundwater, Daejon, Korea, 7–8 July 2003. [Google Scholar]
| City | Capacity (m3/day) | Since | Specification |
|---|---|---|---|
| Changwon | 70,000 | 2006 | 43 vertical wells |
| Changwon | 10,000 | 1998 | 7 vertical wells |
| Haman county | 20,000 | 2005 | 18 vertical wells |
| Gimhae | 180,000 | 2017 | 9 horizontal collector wells |
| Pumping Rate (m3/day) | Surface: Groundwater (%) | The Shortest Travel Time (days) |
|---|---|---|
| 2000 | 63:33 | 1 |
| 1300 | 60:40 | 2 |
| 1000 | 54:46 | 3 |
| 700 | 46:54 | 4 |
| UVA254 (cm−1) | DOC (mg/L) | |
|---|---|---|
| Nakdong River | 0.038 ± 0.005 | 1.9 ± 0.1 |
| PW-Sand | 0.010 ± 0.008 | 0.7 ± 0.1 |
| PW-Gravel | 0.012 ± 0.004 | 0.6 ± 0.1 |
| GMW-Sand | 0.36 ± 0.09 | 0.5 ± 0.1 |
| GMW-Gravel | 0.23 ± 0.03 | 0.7 ± 0.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maeng, S.K.; Lee, K.-H. Riverbank Filtration for the Water Supply on the Nakdong River, South Korea. Water 2019, 11, 129. https://doi.org/10.3390/w11010129
Maeng SK, Lee K-H. Riverbank Filtration for the Water Supply on the Nakdong River, South Korea. Water. 2019; 11(1):129. https://doi.org/10.3390/w11010129
Chicago/Turabian StyleMaeng, Sung Kyu, and Kyung-Hyuk Lee. 2019. "Riverbank Filtration for the Water Supply on the Nakdong River, South Korea" Water 11, no. 1: 129. https://doi.org/10.3390/w11010129
APA StyleMaeng, S. K., & Lee, K.-H. (2019). Riverbank Filtration for the Water Supply on the Nakdong River, South Korea. Water, 11(1), 129. https://doi.org/10.3390/w11010129




