1. Introduction
The Millennium Development Goal to halve the number of people without access to improved water sources was achieved in 2015—five years ahead of schedule. By that, 2.6 billion people gained access to improved water sources. However, there is substantial evidence that improved sources of drinking water, including piped water, can contain fecal contamination and studies estimate that 1.8–2.0 billion people drink such water [
1,
2,
3,
4]. Every year 502,000 deaths are caused by diarrheal diseases that can be attributed to the consumption of unsafe water [
4]. Especially rural communities are prone to having no access to safe drinking water. Lack of infrastructure, technical expertise, user compliance, as well as the lack of supply of chemicals and electricity have been identified as reasons for the failure of rural water treatment and supply systems [
5]. Point-of-use (PoU) treatment approaches are often considered as alternatives and have shown to reduce the risk of diarrheal infections by 40% [
6]. However, the effectiveness of PoU disinfection (including chlorination) depends highly on the comprehension and willingness of the households to apply the treatment systems correctly, especially under varying source water conditions [
6,
7,
8]. Turbidity impedes the application of chlorine and other disinfection methods. In that case additional filtration is required, increasing the complexity and costs for PoU treatment. In the end, the responsibility for safe water supply is passed on to the end user and the educational and motivational efforts required for establishing a reliable application of PoU may not pay off.
The here presented combination of river bank filtration (RBF) and solar-driven electro-chlorination (ECl
2) could be a feasible option for the decentralized treatment of surface water in river bound communities. Reported data show that RBF can effectively remove many major water pollutants and micro-pollutants, including particulates, colloids, algae, pathogens, organic as well as inorganic compounds, microcystins, and heavy metals [
9,
10]. Log reductions for total coliforms of 5.5–6.1 and for bacteriophages of >4.4 were reported by [
11,
12]. Total organic carbon (TOC) removal rates of 60% are possible [
13]. Whereas conventional treatment methods, like coagulation-filtration, can reduce the disinfection by-product (DBP) formation potential by 25% [
13], the reduction can reach 50%–80% using RBF, without any waste sludge produced. Furthermore, RBF is able to attenuate temperature peaks and can provide protection against shock loads. Although inorganic contamination is less likely found in bank filtrate [
9], oxygen may be depleted during the passage of the water through the bank. Under anoxic conditions, iron, manganese, and even arsenic can re-mobilize and enter bank filtration wells [
14]. During the planning process of RBF abstraction sites such aspects have to be considered and recommendations for safe management of RBF sites in India were published [
15].
In the US, RBF has received log-credits for pathogen removal and is mainly used for the removal of suspended solids. The sites are often designed with shorter travel times compared to Europe, where RBF has been widely applied for more than 130 years to produce drinking water along the Rhine, Elbe, Danube, and Seine rivers. Furthermore, in developing regions, the interest in RBF is increasing and the feasibility of its application has been evaluated under different hydrological and hydrogeological conditions (e.g., in India [
16], Egypt [
17], and Thailand [
18]).
However, the application of RBF wells alone does not assure long-term microbial-safe water. Despite the cited removal rates, monitoring campaigns and risk assessment studies have repeatedly shown the presence of total coliforms and Escherichia coli (E. coli) in RBF wells, even at greater distances (48–190 m) to the river bank [
12,
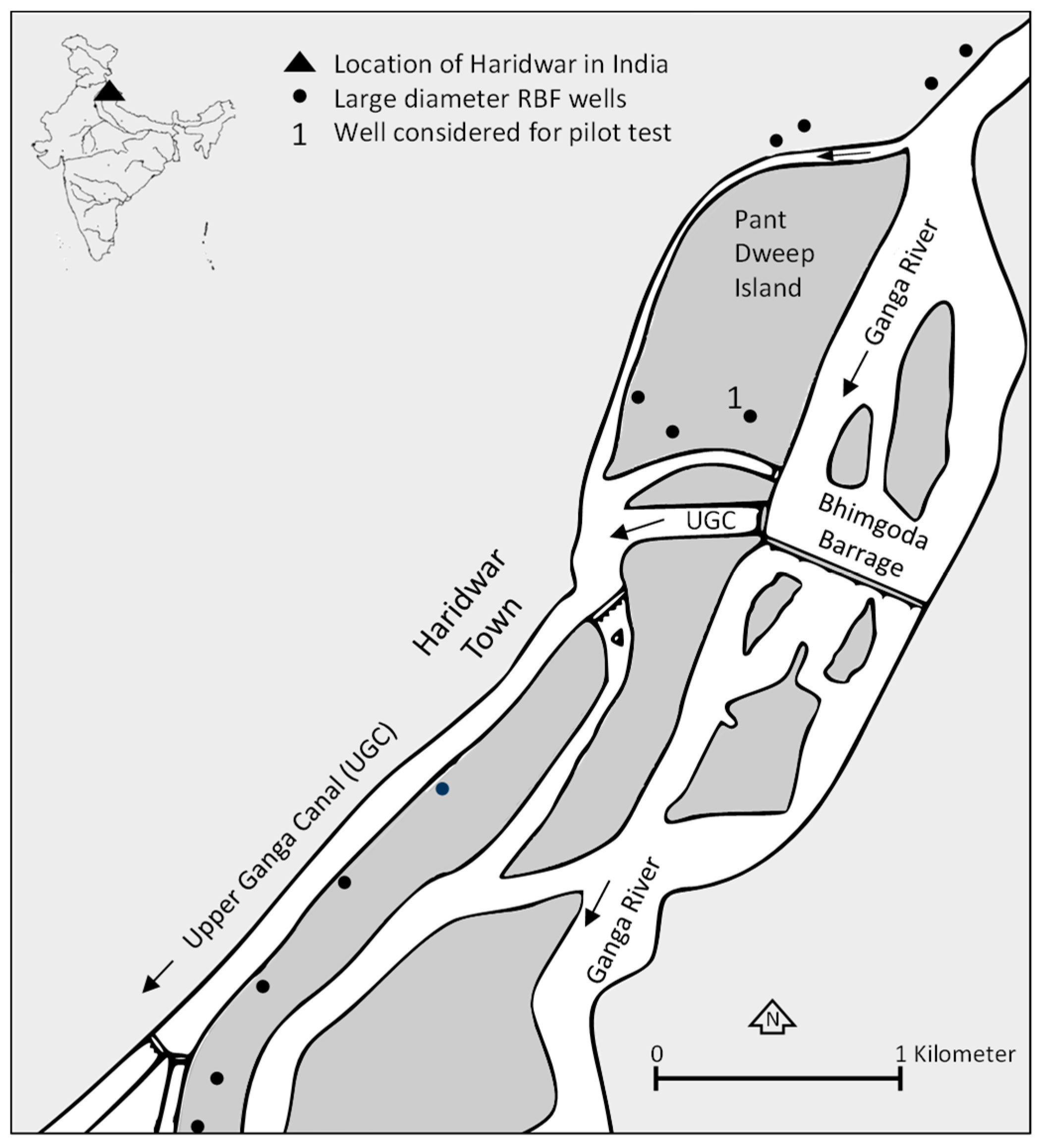
19]. In Haridwar (northern India), where the pilot site for this project is located, such incidents could be related to the seepage of fecal contamination in the direct vicinity of the wells [
19]. Further, recontamination may occur also during distribution and storage, justifying further disinfection. Here, chlorine, in contrast to, for example, UV-treatment or ultrafiltration (UF), has a long proven record of rendering water safe during storage and distribution—if handled correctly [
20,
21]. In rural communities; however, chlorination systems have failed for the same reasons as stated above, as they require constant availability of chemicals, skilled personnel capable in evaluating the chlorine demand of the water, and strict compliance with existing guidelines. Furthermore, the application of chlorine compounds is challenged by the formation of DBPs if applied in unfavorable source water conditions. Even though the risks for microbial contamination usually exceed the adverse side effects of chlorination [
22,
23], guideline values for chlorine dose and inorganic and organic DBP concentrations exist (
Table 1).
The inline-electrolytic production of chlorine (ECl
2) could pose a feasible alternative towards the dosing of chlorine. Here, gaseous chlorine is produced directly at the anode of an electrolytic cell from the chloride dissolved in the water that is to be treated (Equation (1)). The chlorine gas rapidly dissociates in water to hypochlorous acid, being chemically the same oxidizing agent as in conventional chlorine dosing systems (Equation (2)). The chlorine gas production is accompanied by a decrease of pH (Equation (3)) and the evolution of hydrogen gas at the cathode (Equation (4)) [
24].
| Anodic reaction chlorine: | | (1) |
| Dissociation of chlorine gas in water: | | (2) |
| Anodic reaction oxygen: | | (3) |
| Cathodic reaction: | | (4) |
To power this process a DC voltage is applied to dimension stable (DSA) titanium electrodes coated with platinum group metals. Studies have shown that coatings comprising iridium and or ruthenium oxides (MOX electrodes) produce consistently higher chlorine output compared to platinum coatings [
26,
27]. In comparison to the manifold in literature-described boron-doped diamond (BDD) electrodes, MOX electrodes are less prone to produce DBPs, especially considering chlorate and perchlorate [
28,
29].
To control the production of chlorine, fundamental knowledge about the functional interrelationship between chloride concentration, current, current density, electrode material, temperature, and source water quality is required and has become available only very recently [
30]. Systematical evaluation on the effectiveness of the produced disinfecting agents and the potential formation of disinfection by-products (DBP) has shown that the application of inline-electrolysis is comparable to the application of hypochlorous acid [
24,
27,
28,
30]. However, uncertainty towards the long-term operability, the effectiveness under very low chloride regimes, and elevated hardness levels persists [
27,
28].
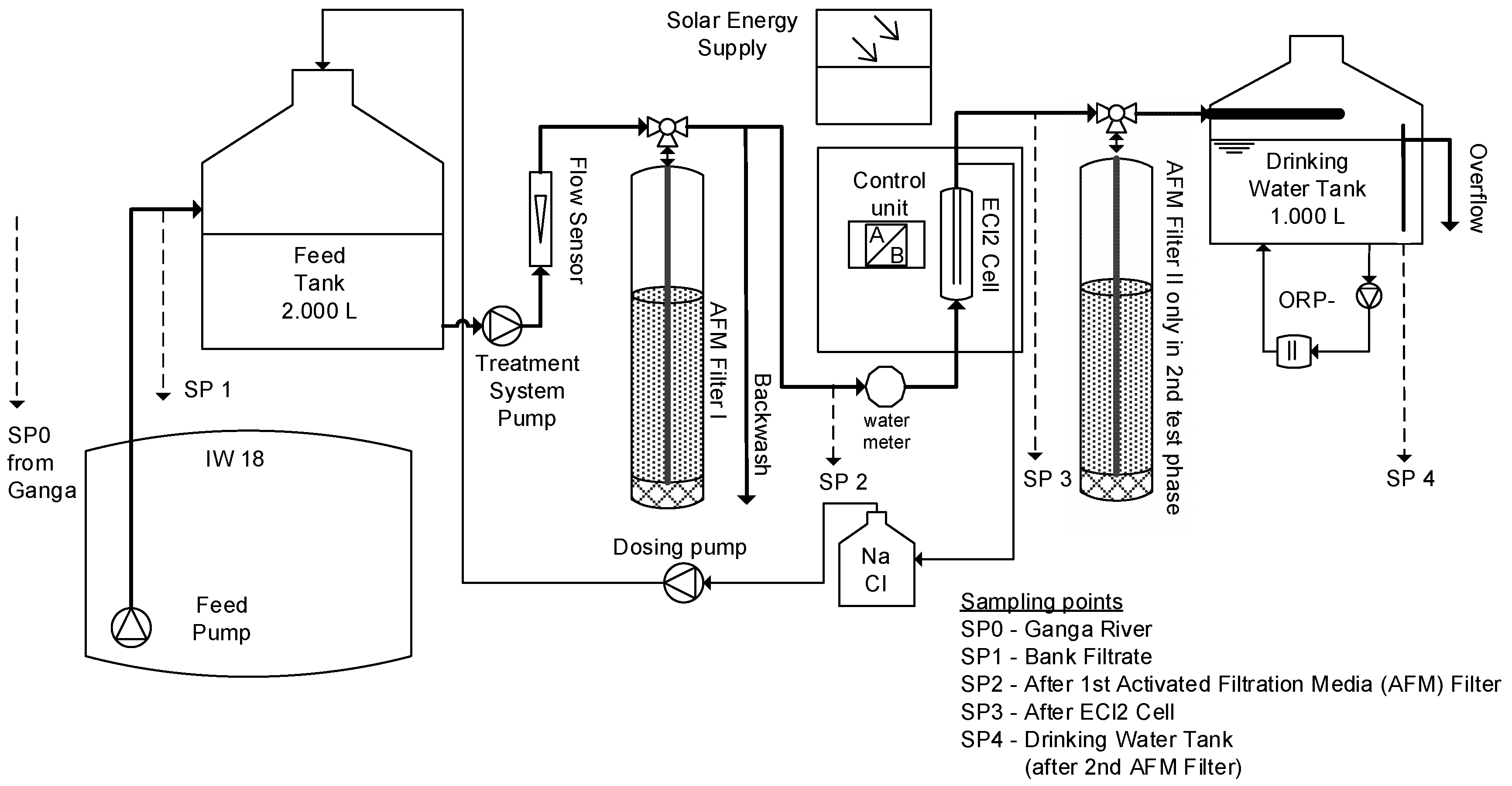
For the first time a combination of RBF and solar-driven inline-electrolysis was tested in a long term trial in northern India. The intention of this combination between natural and engineered solutions (cNES) was to merge the above-mentioned benefits of RBF for surface water treatment with the benefits of residual chlorination, eliminating the above-mentioned drawbacks of chemical dosing. The here presented data summarize the findings of two intensive sampling periods conducted within a two and a half year pilot trial. The main target was to evaluate the pathogen removal and residual disinfection capacity. The first eight-month sampling campaign lasted from March–November and included one monsoon season (July–September). The second sampling campaign lasted for two weeks and was conducted after system optimization. Further, the formation of DBPs and energy efficiency of this water treatment approach was evaluated and suggestions for long-term operation and maintenance requirements were derived.
3. Results and Discussion
3.1. System Operation Haridwar
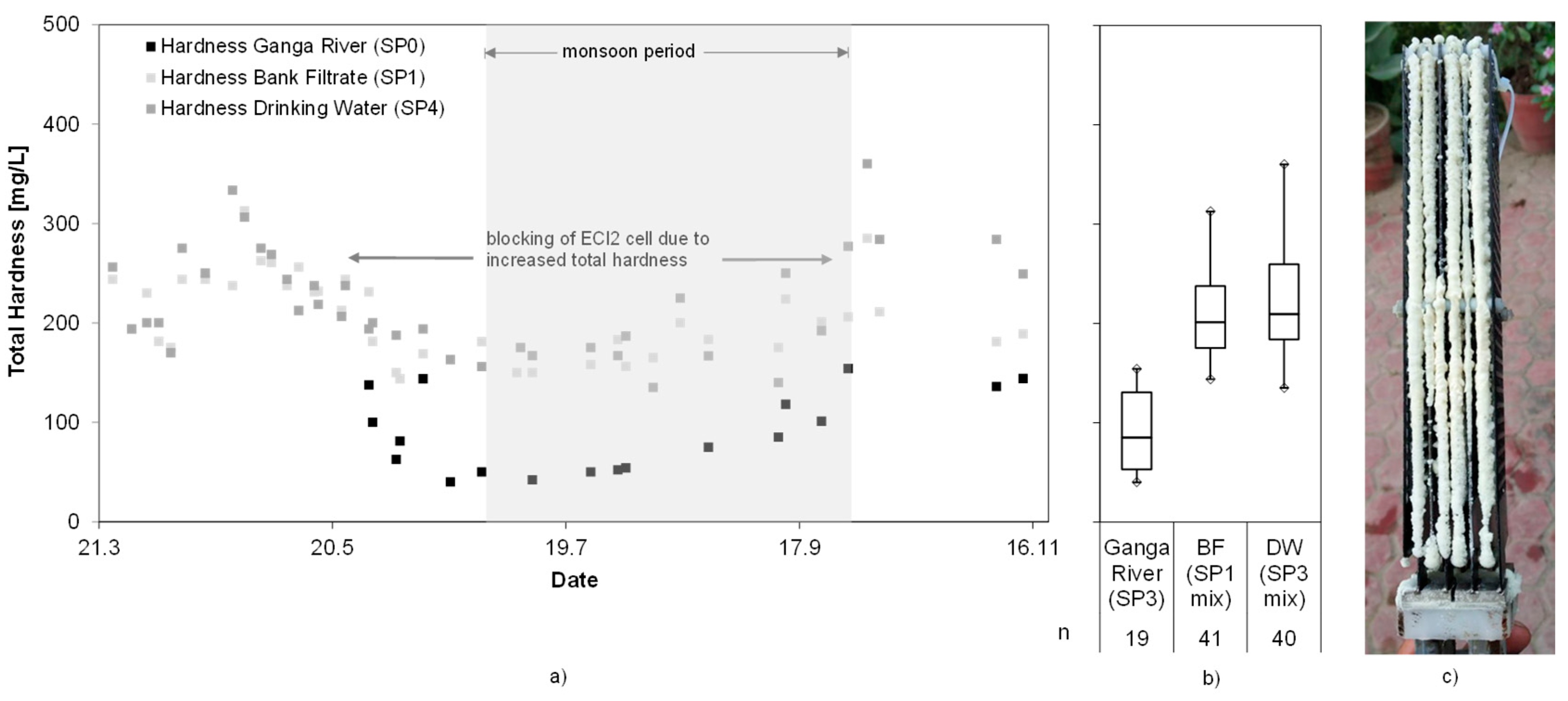
The evaluated trial phase lasted 244 days, including downtimes of in total 24 days. During most of the time the station operated without technical problems and allowed continuous sampling. Reasons for downtimes were, for example, low water levels in the well, infrequent cleaning of PV modules with subsequent power failure, pump failure. Despite the polarity inversion, the operation in Haridwar was challenged from time-to-time by sudden growth of calcareous deposits on the electrolytic cell following elevated levels of hardness in the source water. Those are believed to have initiated crystallization at the cell allowing fast build-up of deposits. Deposits needed to be manually removed from the cell using acid.
3.2. Water Quality Parameters Haridwar
Major water quality parameters of the Ganga River water and bank filtrate of the first sampling period are summarized in
Table 4.
3.3. Turbidity
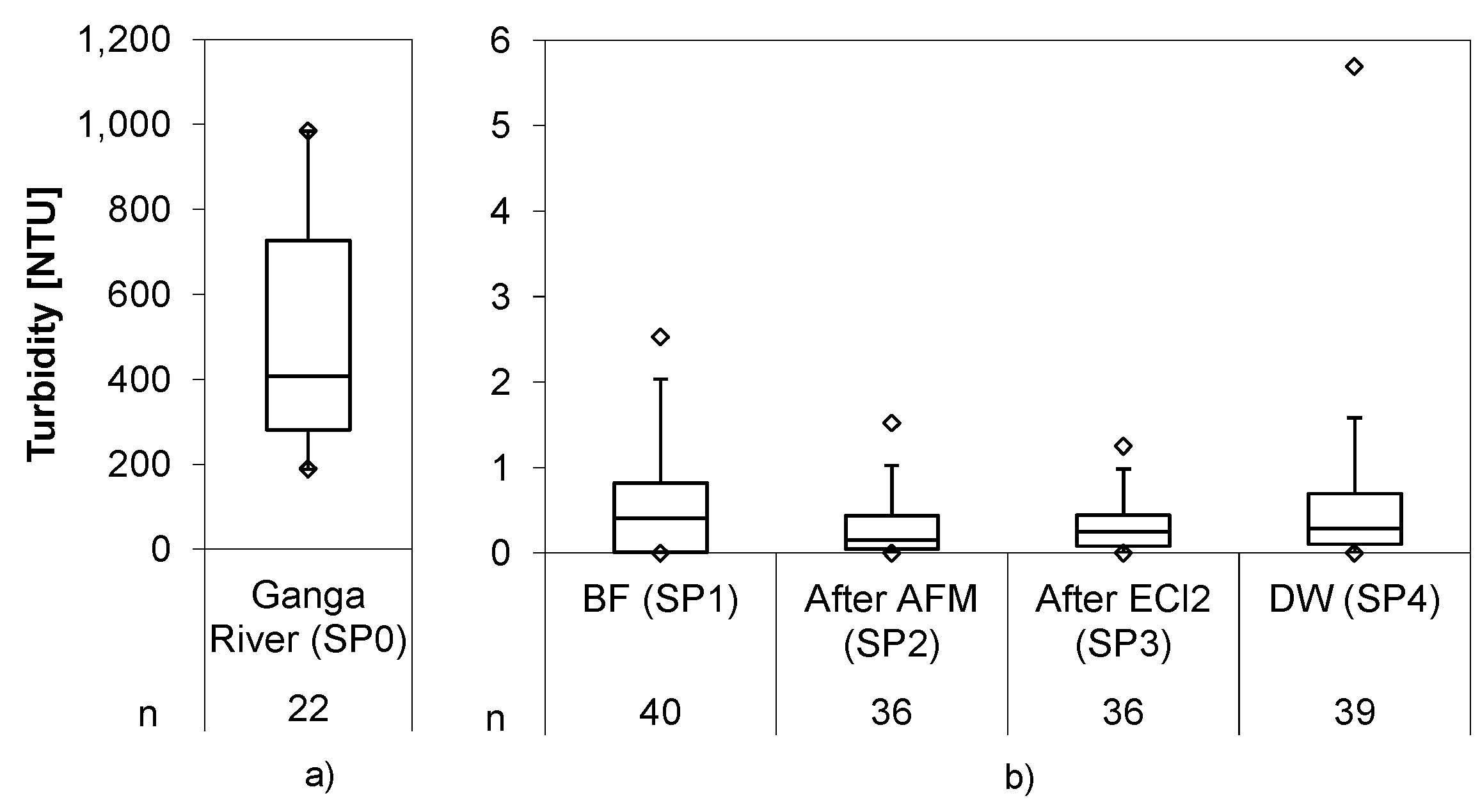
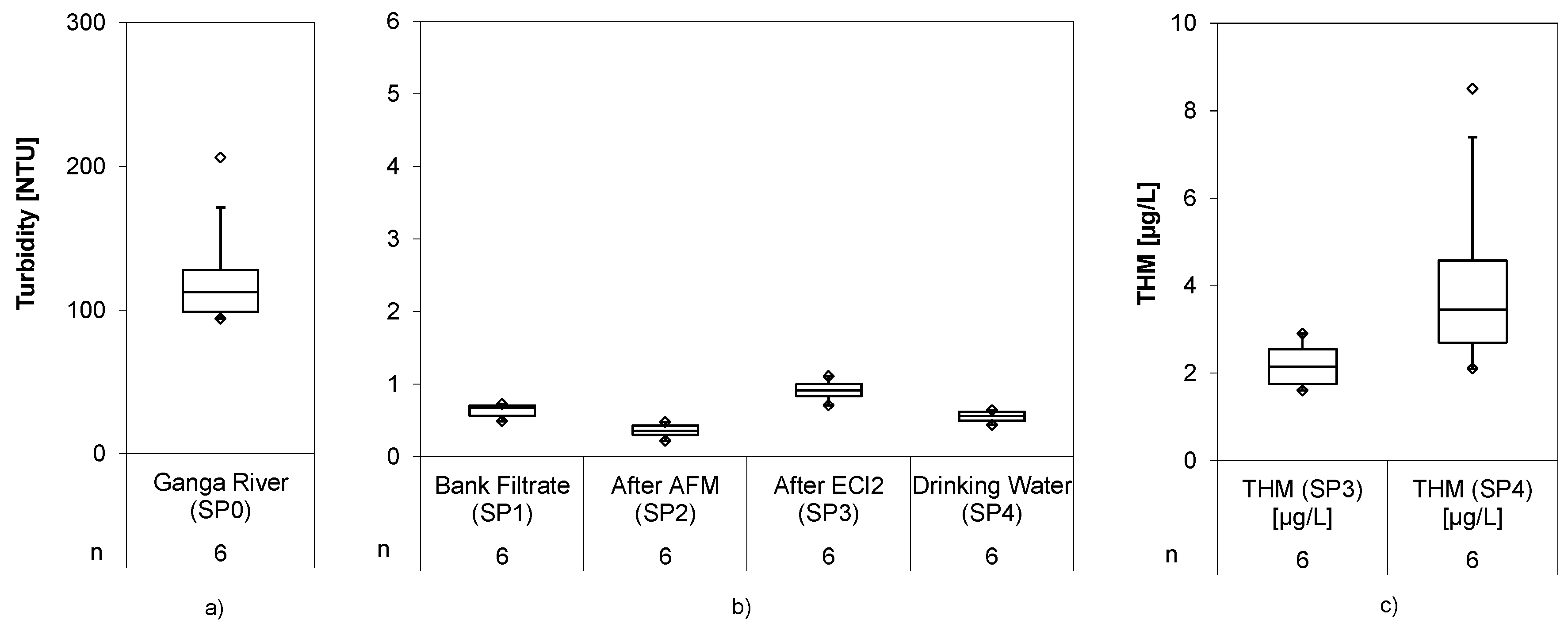
Turbidity in the Ganga River averaged to 501 ± 243 NTU and was reduced to 0.55 ± 0.63 NTU in the bank filtrate (
Figure 4), underlining the role of bank filtration as a barrier for particle ingress. The AFM filter further reduced the turbidity down to 0.30 ± 0.34 NTU, and by that substantially improved water quality prior to the chlorination. This value could be nearly maintained during the passage in the electrolysis cell but increased to 0.70 ± 1.16 NTU with outliers and 0.40 ± 0.38 NTU without outliers, which were caused by filter-breakthrough. The slight but constant increase of turbidity was traced back to calcareous deposits released from the electrolytic cell after polarity inversion. In the second short term trial, a second media filter was installed to remove those deposits.
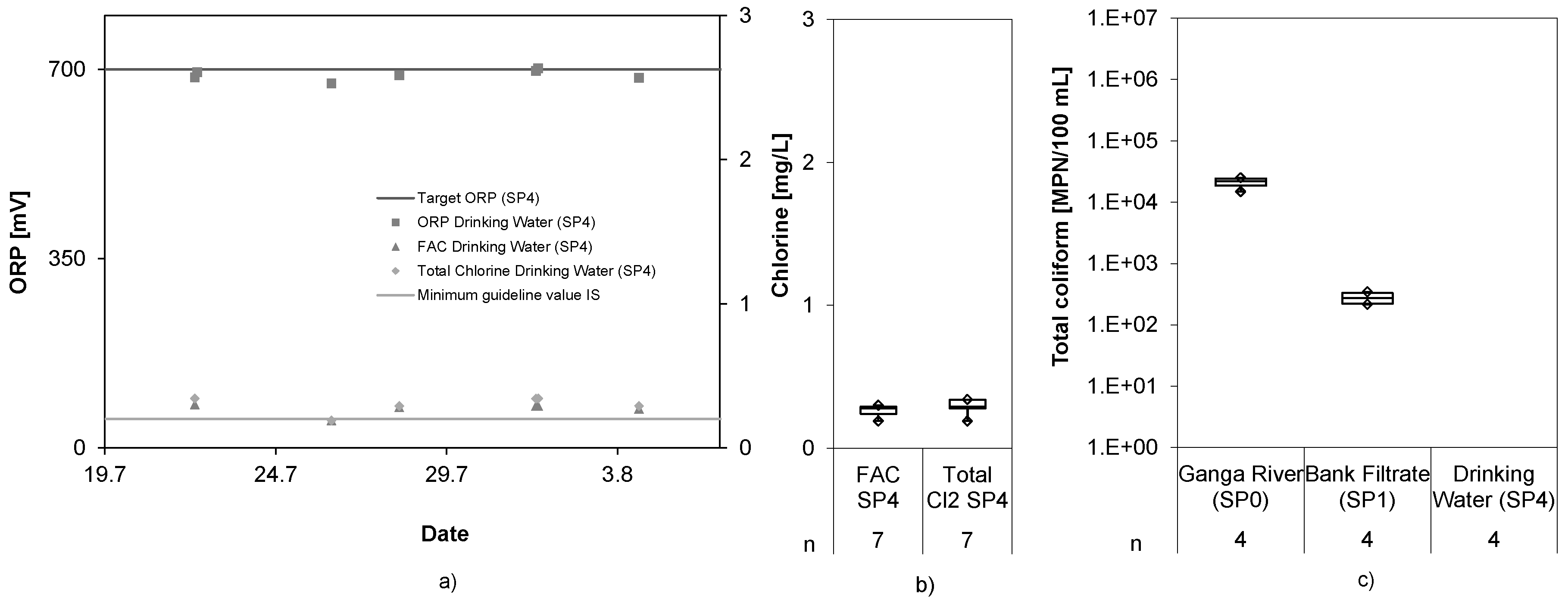
3.4. Disinfectant Production
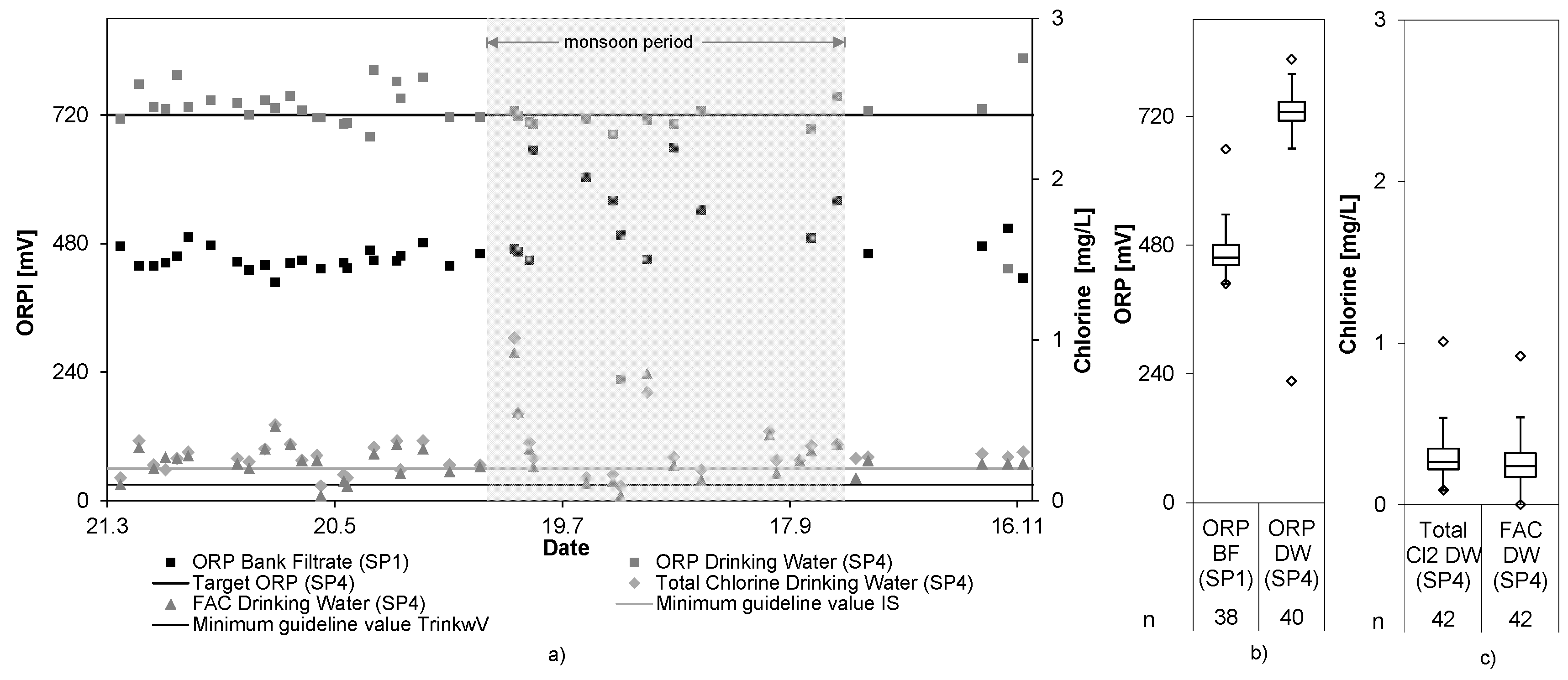
Figure 5 shows the ORP values of the bank filtrate and the final drinking water as well as the FAC and total chlorine in the drinking water storage tank.
The ORP increased from 476 ± 58 mV in the bank filtrate to 720 ± 85 mV in the drinking water tank. FAC and total chlorine values reached 0.27 ± 0.17 mg/L and 0.30 ± 0.16 mg/L, respectively. Despite the fact that increased ORP values indicate the presence of chlorine, no direct correlation between both values could be drawn. As reasons for that, the slow reaction time of the ORP sensors in combination with a relatively small storage volume for the tested flow rates were identified. Whenever the ORP sensor indicated a low reading, the system automatically increased the chlorine production and reduced the flow rate. On the other hand, whenever the ORP sensor signaled a high reading, the system automatically decreased the chlorine production and increased the flow rate. Due to the small volume in the drinking water tank, an increase or decrease of chlorine concentration was not detected quickly enough by the ORP sensor. As a consequence, the chlorine concentration oscillated while the system tried to maintain the target ORP value. Because of this oscillation, the chlorine value fell from time-to-time below the minimum target value of 0.2 mg/L and reached high chlorine values of around 1 mg/L. Disabling the automatic flow rate adaptation or using a larger drinking water storage tank would compensate for the delay of the ORP sensors in adjusting to changing chlorine levels, and thus could stabilize the chlorine concentration. The control mechanism was adapted in the second short term trial by removing the automatic adaptation of the flow rate, which proved to produce more constant results.
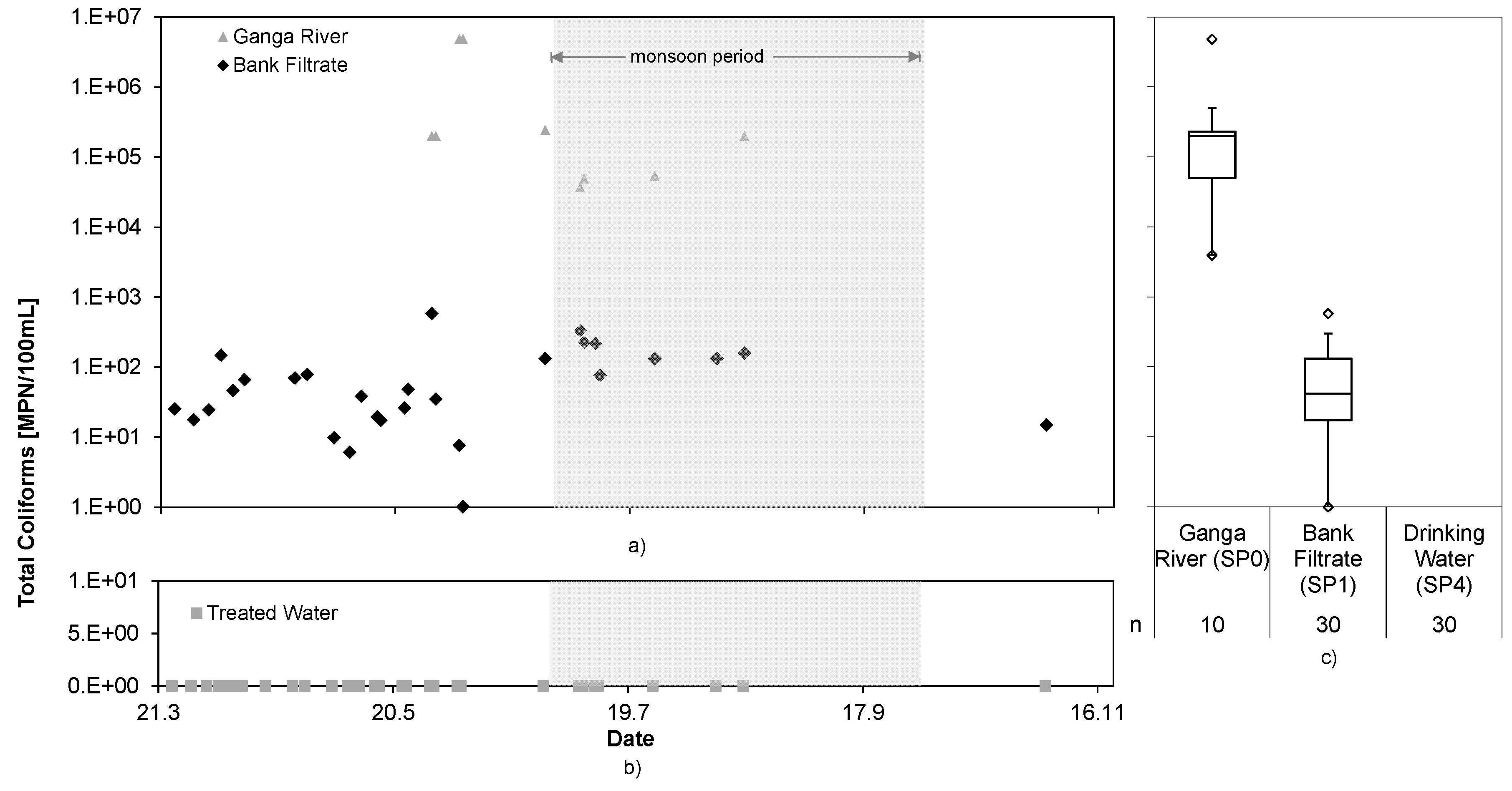
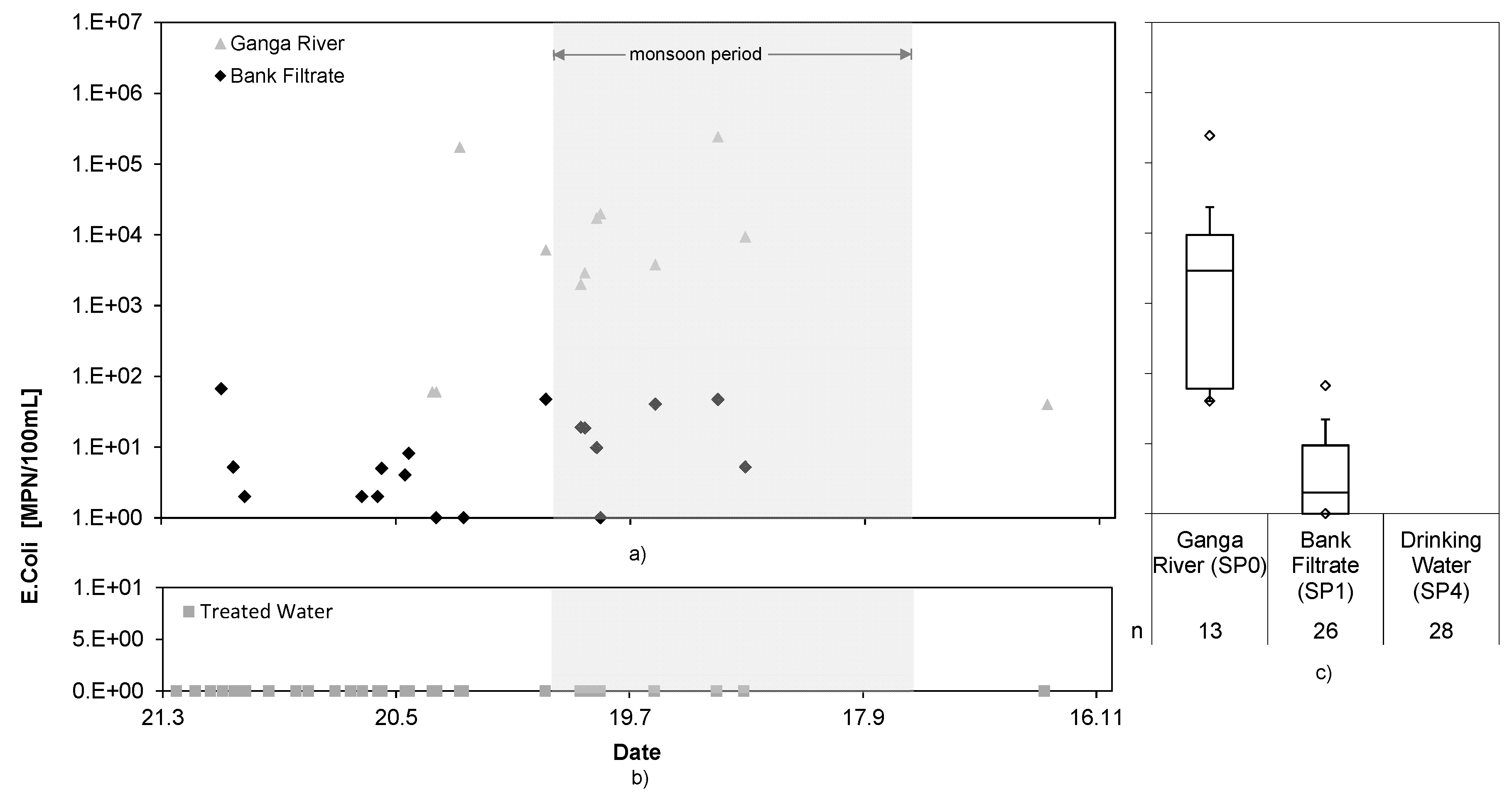
3.5. Pathogens
The analytical results of total coliforms and
E. coli, as indicator pathogens, for the first trial period are shown in
Figure 6 and
Figure 7.
The bank filtration achieved a log
10 reduction of 3.9 and 3.6 for total coliforms and
E. coli, respectively. It is assumed that the peak values in the bank filtrate did not originate from the Ganga River water, but rather came from seepage into the well from above, as described in [
19]. However, the ECl
2 system completely removed still present fecal indicators and water could be kept microbially safe at all times. The maximum log reduction of the RBF + ECl
2 cNES achieved was >6.7 for total coliforms and >5.4 for
E. coli. It can be assumed that even higher log reductions could be reached, considering presence of FAC in the treated water.
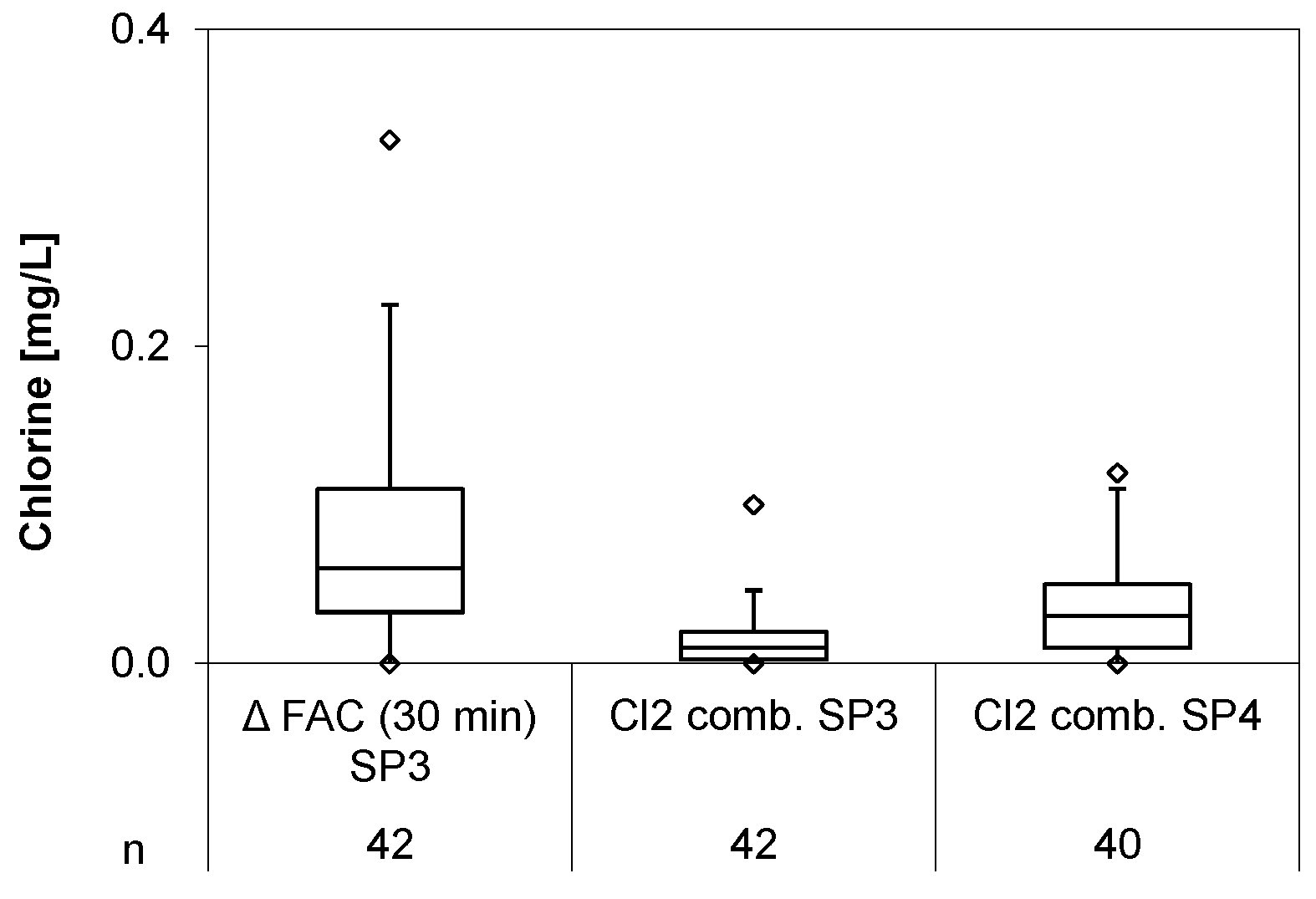
3.6. Chlorine Demand
During the trial period the chlorine demand (ΔFAC) was very low, with 0.03 ± 0.03 mg/L on average, but peaked to 0.33 mg/L. Combined chlorine, formed directly at SP3 and SP4, were 0.09 ± 0.08 mg/L and 0.02 ± 0.02 mg/L, respectively (
Figure 8).
The low average values resulted from the low concentrations of ammonium and organics in the bank filtrate (see
Table 4) and do not indicate any critical potential for organic disinfection by-product formation. However, as there are substantial fluctuations of organic and nitrogen compounds in the well water, an automated adaption of the chlorine production process is required to compensate for changing chlorine demand and combined chlorine formation. Even though there was no good correlation between ORP and chlorine concentration, the ORP can indicate insufficient supply of disinfectant and can; therefore, next to cell current and flow rate, be consideredas an additional parameter to control chlorine production.
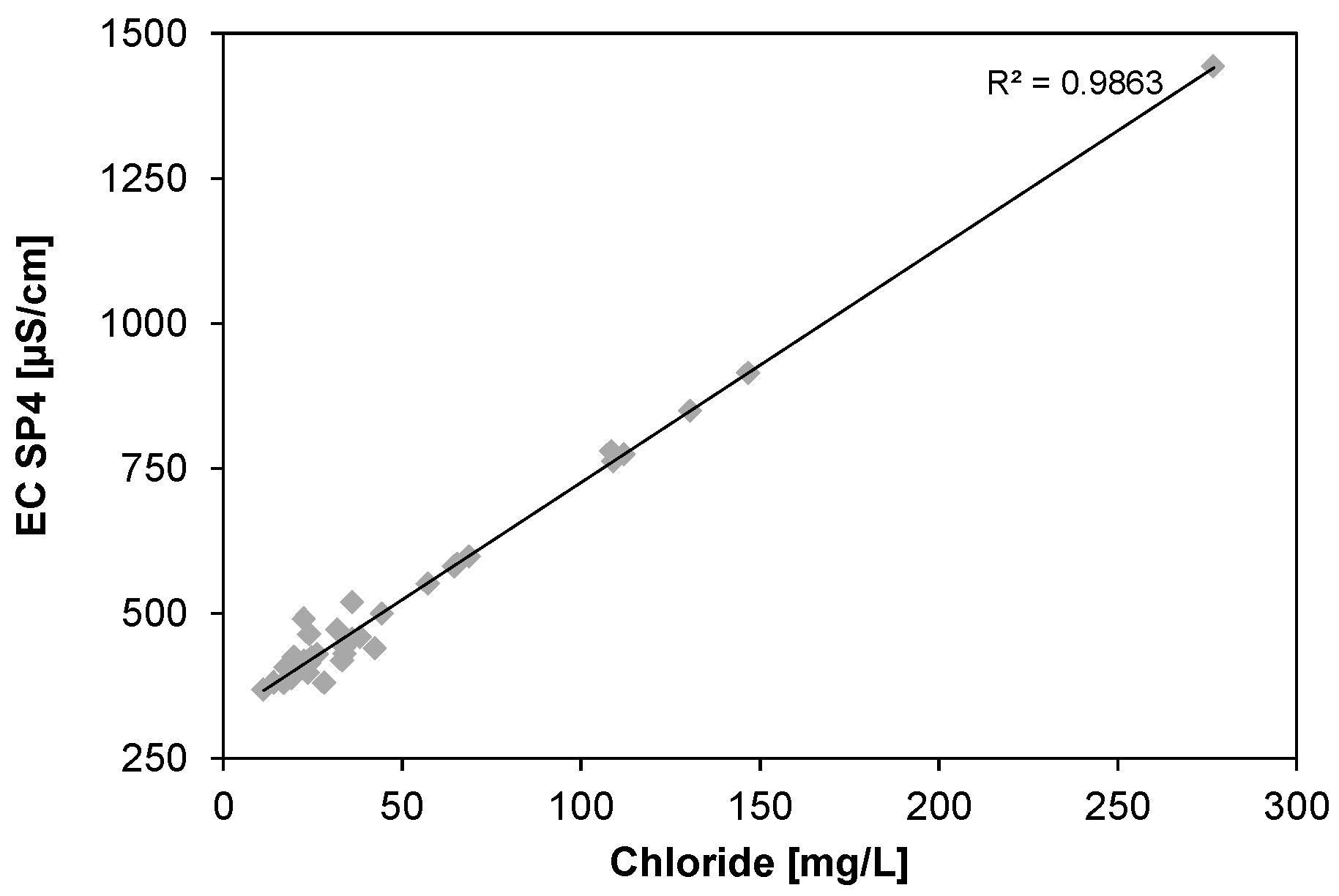
3.7. Electrical Conductivity and Chloride Concentration
The correlation between electrical conductivity and chloride concentration is shown in
Figure 9.
The scatterplot indicates that, especially at higher chloride concentrations, the effect of the chloride on the conductivity prevails towards other ions. In order to limit the NaCl consumption, the chloride concentration was supposed to be kept below 50 mg/L. The incidents when higher concentrations occurred could be tracked back to either a nearly empty feed tank, into which the dosing pump dosed too much chloride for the water available, or to calcearous deposits that were formed on the cell. Those deposits have hampered the ability to reach a voltage/current ratio of ≤3.
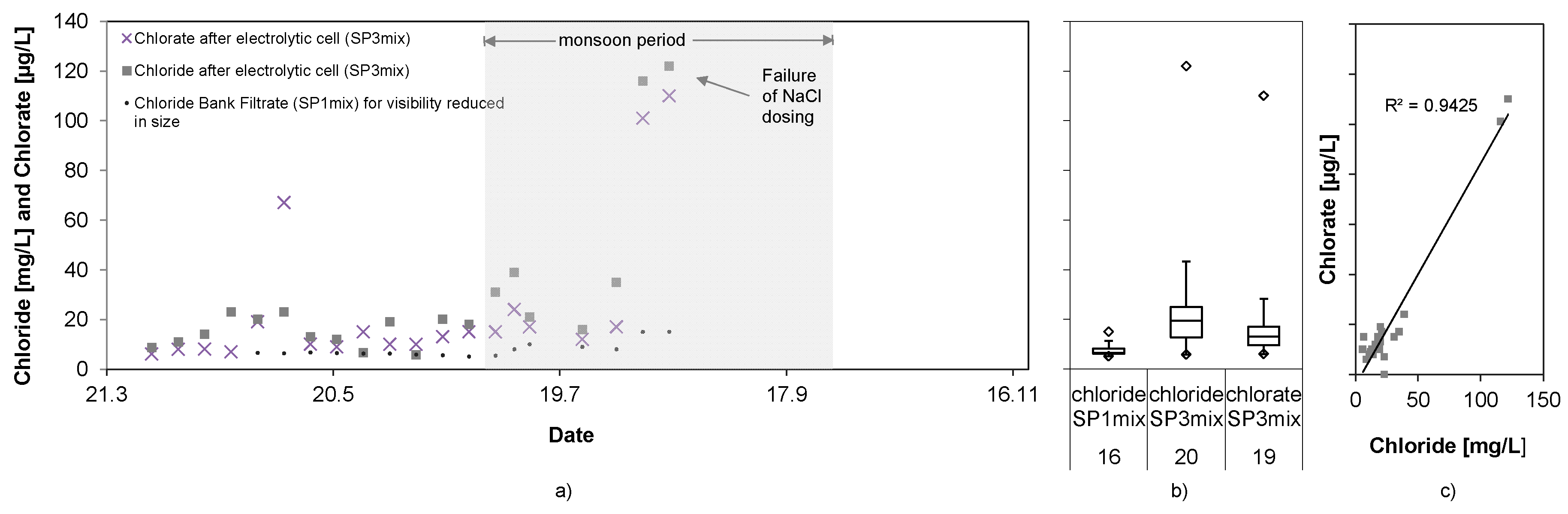
3.8. Inorganic Disinfection By-Products
The concentrations of the mixed samples for chloride and chlorate in SP3 (directly after the electrolysis cell) are shown in
Figure 10.
Even after the long storage period of several weeks until analysis in Germany, the chlorate concentrations reached only 22 ± 29 µg/L and is not of concern considering WHO and German guideline values (
Table 1). Uncertainty towards the maximum chlorate values exists due to the mixing of two to three random samples into one sample per week, as the concentration of samples with higher concentration might have been lowered with samples of lower concentration. However, the correlation between chloride concentration and chlorate production (
Figure 10c) show that higher chloride concentrations are required to reach elevated levels of chlorate. The two maximum chlorate concentrations above 100 µg/L went along with an excess of chloride added into the feed tank. Chlorite and perchlorate were always below the detection limit of 1 µg/L and are; therefore, not of concern when water is disinfected by means of inline-electrolysis with the here applied MOX-electrodes.
3.9. Hardness
Figure 11 shows the total hardness values measured during the first system trial.
The hardness values were fluctuating throughout the test period in the Ganga River (92 ± 39 mg/L) and the bank filtrate (207 ± 40 mg/L). Whereas the values in the bank filtrate ranged around an unproblematic 200 mg/L during monsoon, the levels reached ~300 mg/L before and after the monsoon season. Those values have shown to be problematic for system operation, as spontaneous growth of calcareous deposits on the ECl2 cell intermittently reduced chlorine production efficiency and required extra maintenance.
3.10. Second Optimized Test Phase
In the second short term test the automatic flow rate adaption was disabled and constant flow rates of 160, 220, and 280 L/h were established This was giving the ORP sensor sufficient time to detect changing chlorine concentrations and allowed the ability to test the system’s reaction on changing chlorine demand in SP4, adjusting the cell current only. The main results of the second short term test are presented in
Figure 12.
Despite the very short duration of this second pilot trial, it was sufficient to show that the fluctuation around the set target ORP of 700 mV could be reduced to an acceptable level. The constant flow rates permitted an adequate utilization of the ORP reading for controlling the chlorine production and keeping the concentration in the desired range of 0.26 ± 0.04 mg/L. The presence of FAC in this concentration range was represented by elevated ORP values of ~700 mV. It can be assumed that a larger drinking water storage tank (SP4) would have had a similar effect. Pathogens could still be completely removed through ECl2.
Further, the effect of the second AFM filter, placed behind the electrolytic cell to remove calcareous deposits, and the THM concentrations are shown in
Figure 13b.
The second filter reduced the turbidity down to 0.55 ± 0.08 NTU, after it had increased to 0.92 ± 0.13 NTU behind the electrolytic cell, improving overall water quality. The THM analysis showed concentrations of 2.2 ± 0.5 µg/L in SP3 after 30 min and 4.2 ± 2.1 µg/L in SP4. Those low concentrations were expected due to the low TOC and dissolved organic carbon DOC content of the water, allowing full compliance even with strict guideline values for DBPs (
Table 1).
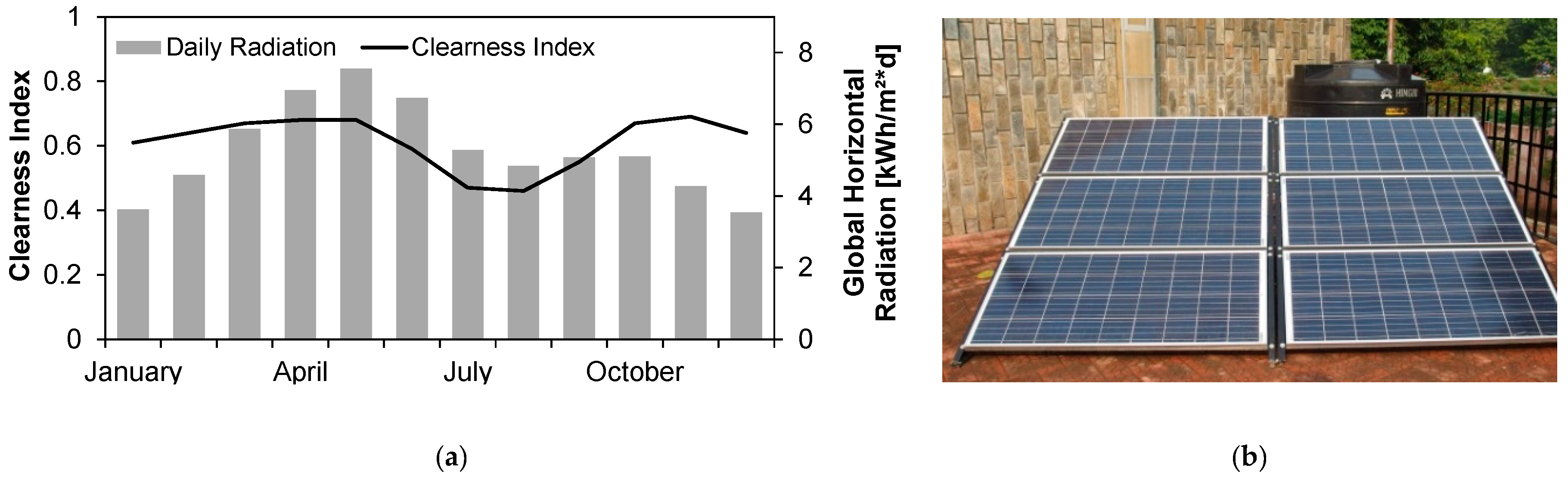
3.11. Energy Demand and Solar Energy Supply
During the first trial period a water volume of 1037 m
3 was treated and the total electricity demand was summed up to 271 kWh without and 412 kWh with bank filtrate pumping. The average flow rate through the system, including off times (e.g., at night, during maintenance or repair) in Haridwar, was 180 L/h. This resulted in an average power demand of 46 W without and 70 W with bank filtrate pumping, and a per m
3 energy consumption of 0.4 kWh (
Table 5).
The power requirement of 70 W was used for the sensitivity analysis using different sized PV generators and different battery capacities considering the parameters mentioned in
Table 3. The analysis shows that at least a 96 Ah battery system with minimum 800 Wp are required to power the water treatment system for 22 h per day and a permitted capacity shortage of 1%. The results are summarized in
Table 6.
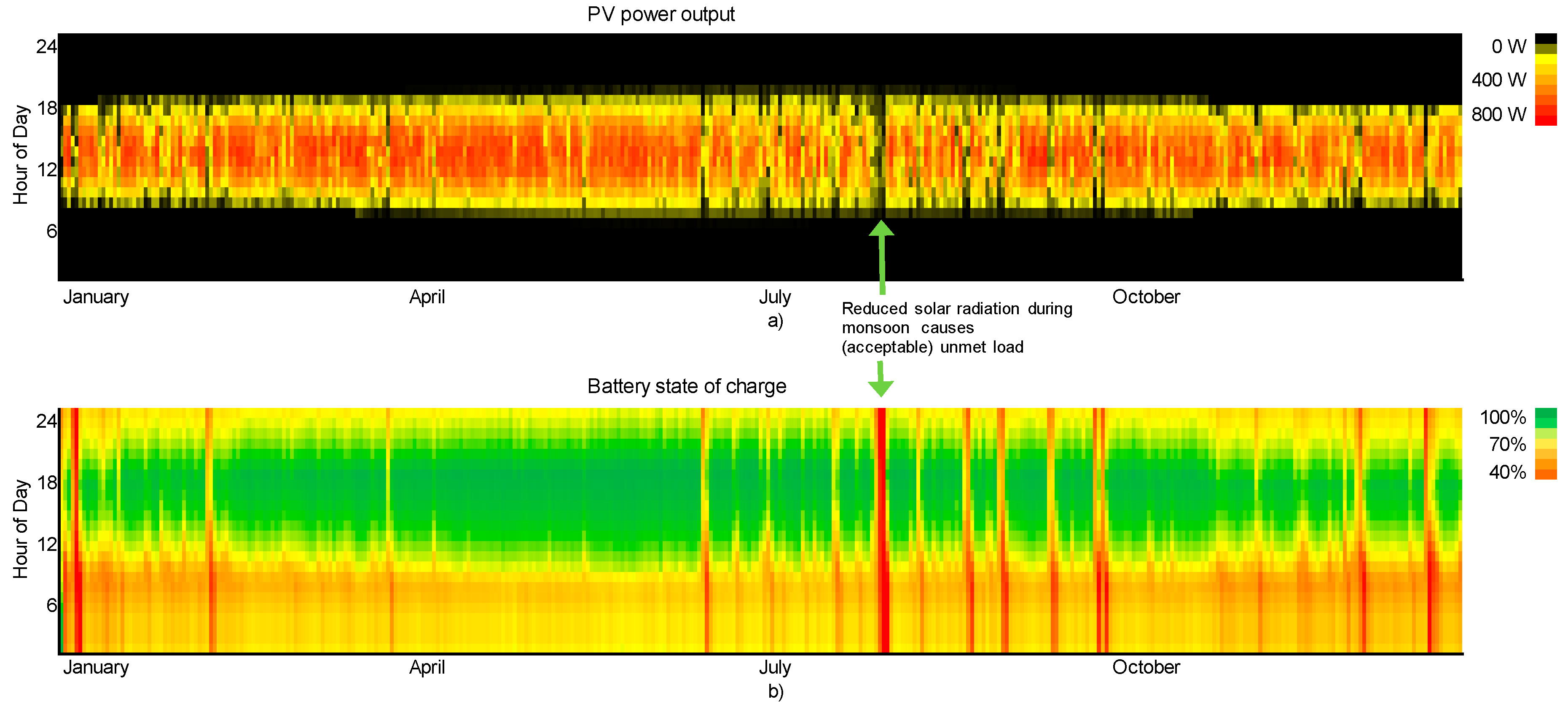
The simulation shows, that with the installed solar PV-battery combination of 900 Wp and 2 × 96 Ah, the required electricity can be supplied nearly throught the year. Only a few hours of electrictiy shortages including a 7-h long power cut in the middle of the monsoon in August occured. The total capacity shorage summed up to 0.4%. Whether this is permissible in a real case scenario and whether this could be compensated by, for example, an increase of the water storage capacity depends on local conditions. The simulated power production and SoC is shown in
Figure 14a,b, respectively.
During the trial the station was continuously running on the given solar PV system, as long as the modules were cleaned from dust frequently.
4. Conclusions
The presented results of a first long term and a second short term trial of a RBF ECl2 combination in India show that the tested system poses a feasible alternative for decentralized and safe drinking water supply for river bound communities in developing countries. RBF serves as a very efficient pre-treatment step, substantially reducing pathogens, turbidity, and DBP precursors. The installed AFM filter is capable of further reducing the already low turbidity values and the ECl2 system completely removes all still-present indicator pathogens, and supplies sufficient residual disinfectant for safe water distribution. The station complies with given water regulations concerning indicator pathogen and chlorine concentrations. Additionally, the production of DBPs is of no concern and stays well below the given guideline values. The first test period revealed some optimization potential of the control algorithm and the system setting, which was successfully implemented for the second trial. After that, the system reacted reliably to changing source water and operating conditions by keeping the residual disinfectant at a constant level. The used ORP sensor is able to indicate “sufficient” or “insufficient” disinfectant once it is given sufficient reaction time. For more accurate and faster readings, the application of chlorine probes may be an alternative to ORP probes.
Clogging of the electrolytic cell, due to increased levels of hardness, remains an operational challenge of the ECl2, which needs to be specifically addressed. With polarity inversion alone, and total hardness levels above 200 mg/L, the operation of an ECl2 system currently reduces the maintenance intervals to about once every two months. The operation of the ECl2 system at total hardness values above 300 mg/L is currently not advisable. With the implementation of an additional probe to measure electrical conductivity, cell overgrowth could be detected by monitoring current and voltage of the cell and comparing them with the actual conductivity of the water.
After this trial, the system is mature enough to be implemented in a real scenario and under favorable operational conditions and source water quality, and it should be possible to reduce the maintenance intervals of the station to six months. For this, the implemented SCADA system will play an important role. An increase of the treatment capacity is straight forward by increasing the ECl2-cell size and the solar energy supply accordingly.