1. Introduction
Due to the increasing strain on drinking water supplies and the energy demand for drinking water treatment, the combination of natural and engineered systems (CNES) gains more and more attention. While the currently reported annual volume of managed aquifer recharge (MAR) is only 1% of global groundwater use, in some countries it is considerably higher, especially where river bank filtration (RBF) is practiced, e.g., in Hungary, the Slovak Republic and Germany [
1]. Although the benefits of these processes are well-known and they have been studied extensively over the last century, some unresolved site-specific issues and ambiguous scientific terminology remain. In Hungary, with the exception of the RBF sites on islands in Budapest, it is typical for smaller rivers that had elevated concentrations of dissolved iron and manganese in the portion of pumped land-side groundwater to require further water treatment [
2].
The Fe and Mn issue is hydrogeology related, and it involves the distance between the surface water body (rivers in the case of RBF, infiltration trenches and basins in the case of MAR) and the abstraction point. If the abstraction point is far away (horizontally or vertically) from the surface water body, undesired flow from the background/land-side commonly increases, causing higher dissolved iron and manganese concentrations in the pumped water. On the other hand, if the flow path length is short, the withdrawal of surface water increases, but the short retention time reduces the efficiency of the porous media as a barrier and bioreactor.
A large number of publications are dealing with the question of how distance and travel time affect the removal capacity of MAR schemes [
1]. Hydraulic conditions and physical and chemical changes have been extensively studied. Due to limitations concerning the calibration of geohydrochemical models, there is a lack of data on the exact spatial extent of water quality changes on the subsurface. It is understood from the slow sand filtration process that the highest (micro)biological activity is found at the topmost few centimeters of the filter. The flow velocity in slow sand filters (1–50 cm/h) [
3,
4] are comparable with those documented for BF sites (0.1–50 cm/h) [
5,
6].
Both sand filtration and bank filtration were studied extensively in the past regarding the rejection of potential microbiological hazards. In slow sand filters, 1–3 log removal (90–99.9%) was observed by different authors, both by colony counting and other methods [
7,
8]. RBF was reported to be capable of 2–5 log reduction for pathogen indicator parameters, such as
E. coli and coliforms [
9,
10].
Consequently, with respect to the importance of the first few centimeters of the top layer, both RBF and MAR in general have some analogy with slow sand filtration and cake filtration. Since long residence times and flow path lengths can have an adverse effect on water quality, it raises the question about the extent that this thin boundary layer (or zone) can be utilized in order to avoid discharge from land-side or deeper groundwater.
Concerning RBF, a large number of scientific studies dealt with clogging and flow conditions in the riverbed, and various methods were applied and further developed to assess the permeability of the riverbed, i.e. the fragment approach and complex numerical flow simulation models [
11,
12]. The main difference between a pure slow sand filtration setup and RBF is the presence of the shear stress and sediment transport at the boundary coupled with an abstracted portion of land-side groundwater. With respect to MAR, the shear force at the boundary is usually negligible, depending on the specific layout and hydraulic conditions of the infiltration structures. Because of this, MAR is more closely related to slow sand filtration.
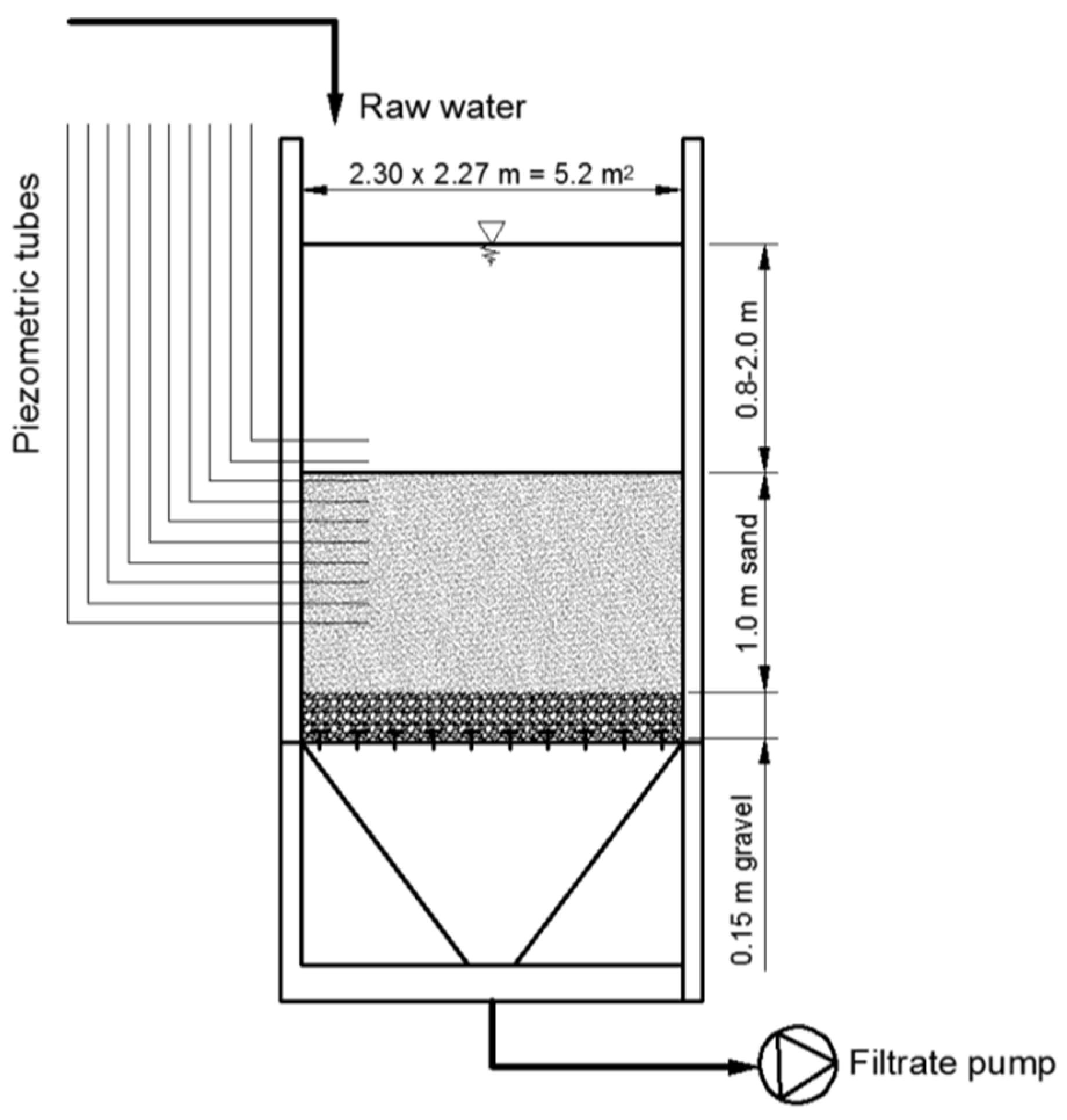
As part of the AquaNES “Demonstrating Synergies in Combined Natural and Engineered Processes for Water Treatment Systems” project, a slow sand filter was operated at the pilot water treatment plant (PWTP) of the National University of Public Service’s Faculty of Water Science in Baja, Hungary. The aim of the study described here was to assess the efficiency of slow sand filtration when a thin, clogged, biologically active top layer is present. This way an assessment can be made about the removal of pollutants and pathogen retention at the porous media–water body boundary. Results are compared with the efficiency of long retention time systems. The investigation is focused on determining the extent of reduction in microbiological indicator parameters.
The main application of reverse osmosis is seawater and brackish water desalination in the present day. Even though there is considerable drinking water production by reverse osmosis worldwide, most scientific research focuses on the process itself and extensive case studies do not exist for bank filtration coupled with reverse osmosis. The main area of the present research is the rejection of pollutants, which are known to be unaffected by bank filtration [
13]. Other research focuses on the energy efficiency [
14] as reverse osmosis (RO) equipment used for freshwater desalination operates on a considerably lower pressure and energy demand than those for seawater desalination. A similar pilot scale investigation with single element RO was carried out at three different sites in the United States [
15]. In developed countries, reverse osmosis of freshwater is typically used for wastewater reclamation and is usually located at the end of the process train either in wastewater treatment or drinking water treatment, for example in Orange County, California [
16].
Although large water companies and some authorities possess reverse osmosis equipment for emergency water treatment, the long-term treatment of bank filtered water on reverse osmosis has never been studied in Hungary. Bank filtration supplies almost 50% of the country’s drinking water demand and bank filtration sites are vulnerable to not just emerging micropollutants, but industrial and municipal wastewater discharge as well. Therefore, it is imperative to start investigations on the long-term sustainability of advanced treatment processes.
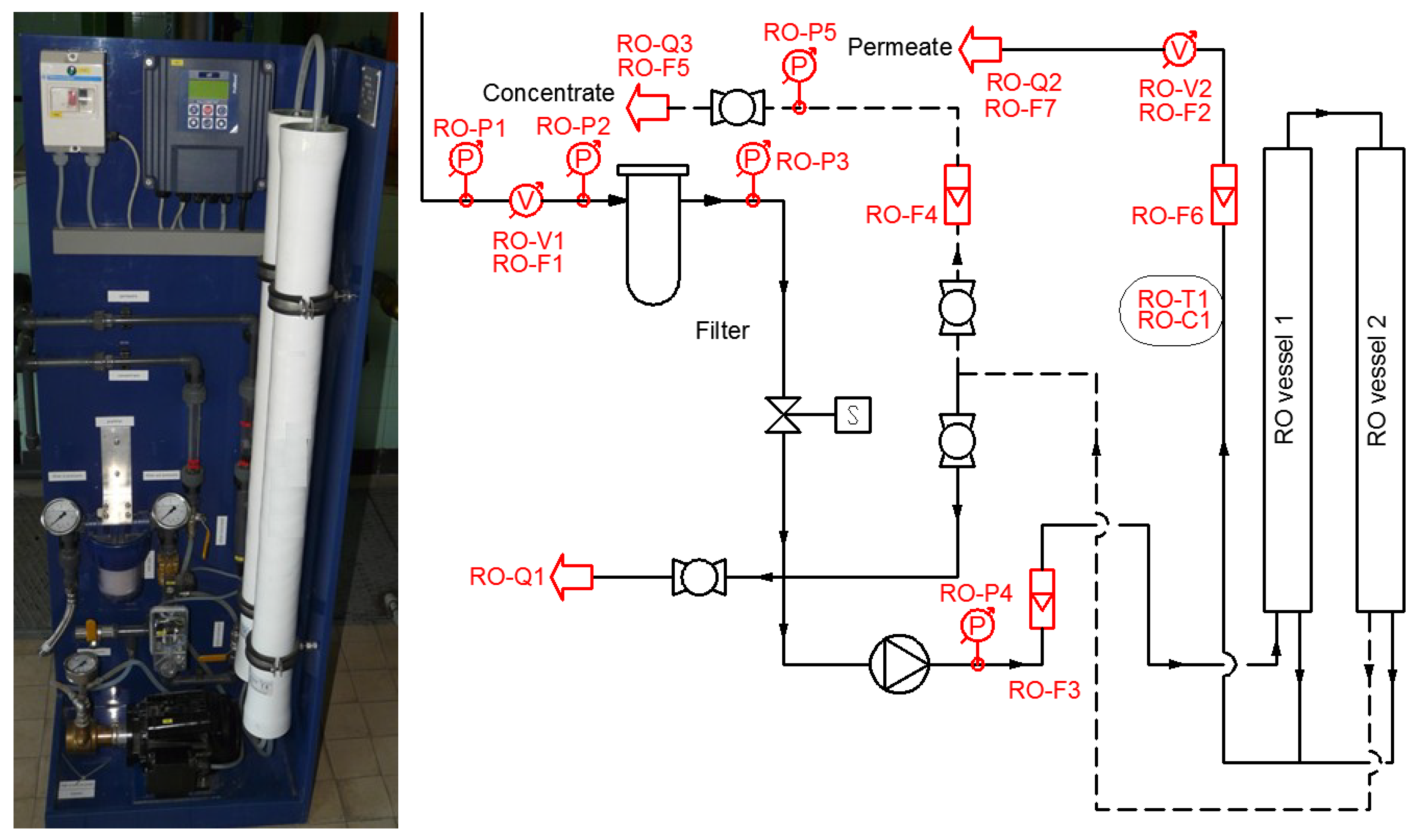
In order to investigate the impact of the bank filtration process on membrane filtration, a reverse osmosis (RO) filter was installed on the filtrate stream. This unique setup (despite RO never having been the first choice for treating fresh water in the past) was chosen because among all membranes, RO is the most sensitive to fouling. Because RO operating parameters respond very rapidly to fouling effects, it could be a good choice to investigate the impact of naturally treated water quality on engineered processes. If it can be proven that a thin boundary layer is adequate during MAR or even RBF to properly improve the water quality for membrane filtration, then more CNES systems with short retention times may be developed in the future.
3. Results
3.1. Clogging and Pressure Loss Development
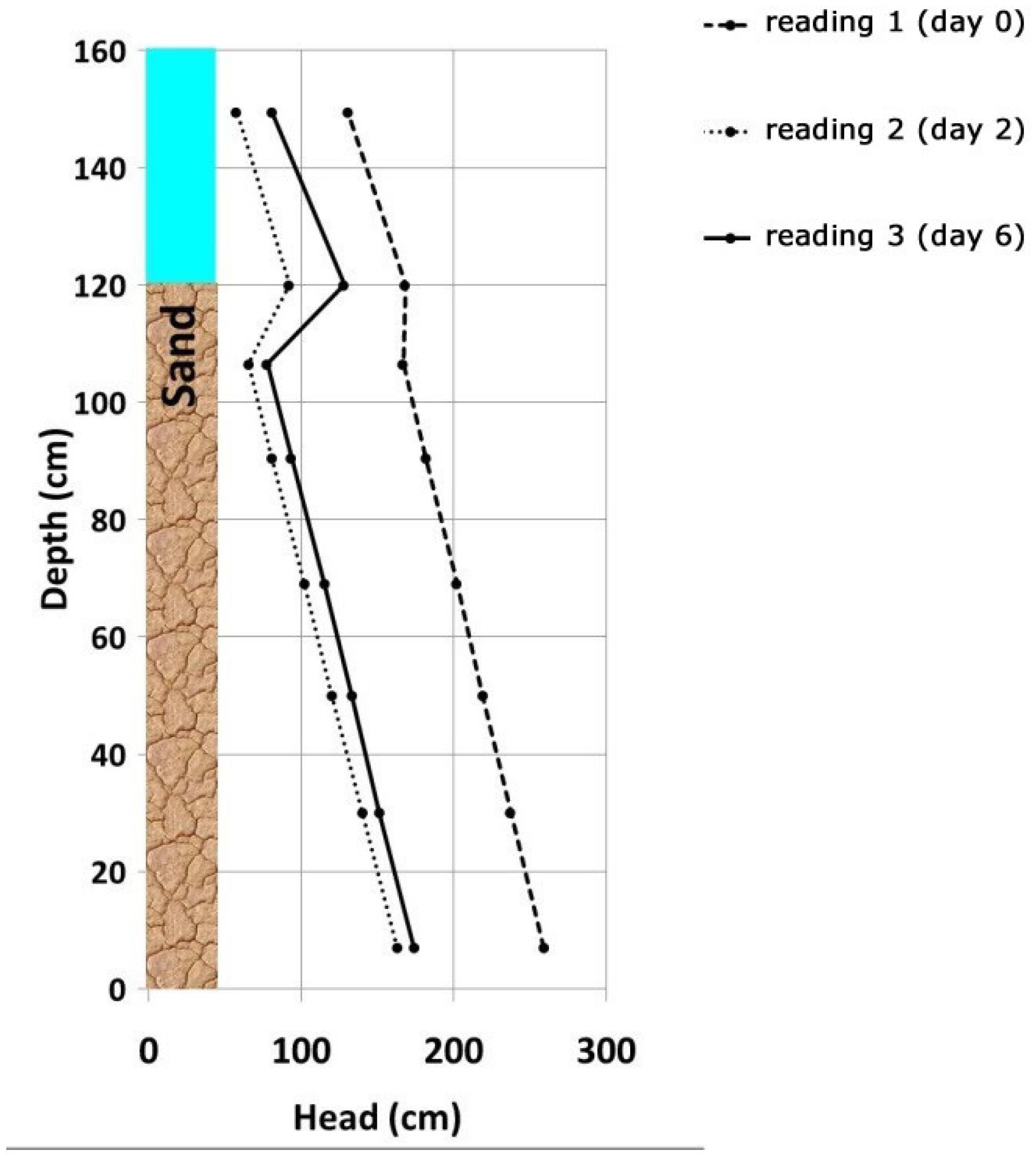
Based on the measured piezometric level differences at different depths in the sand layer of the filter, the pressure distribution (or hydraulic gradient) was calculated along the filter in the form of a Michau (or Lindquist) diagram (
Figure 3). The difference shown at a depth of 150 cm above the bottom of the filter indicates different water levels above the surface of the filter (water column of 1.0–1.5 m).
As expected, the pressure distribution shows the characteristics of a slow sand filter, with the steepest gradient present in the top 5 cm of the sand layer and no observable deviation from the linear gradient below. A deviance from the linear hydrostatic pressure can be attributed to the biofilm development on the geotextile covering the outlets of the piezometer tubes. This affected only the tubes near the top of the sand. Because of this, the equalization of the pressure differences in the piezometers takes a longer time.
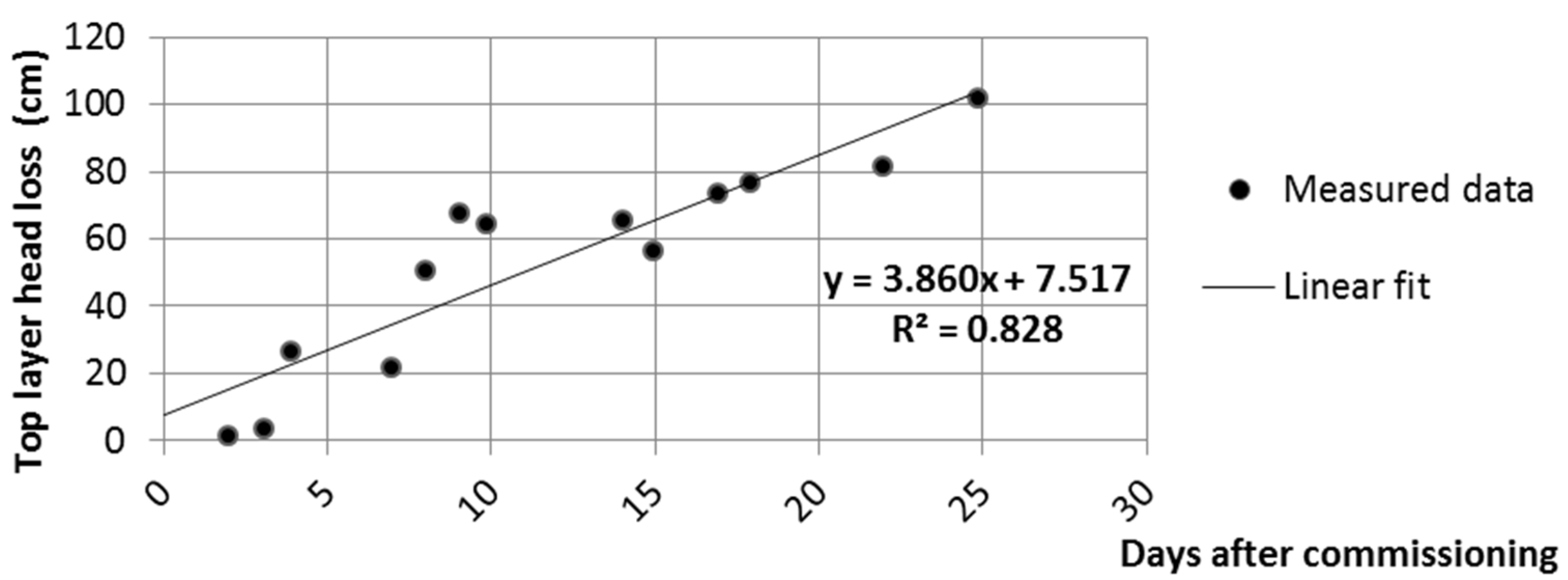
The gradient in the top layer plotted as a function of elapsed time shows an increasing linear tendency (
Figure 4). The calculated
p-value of the F-test performed to investigate the linear correlation is 1.6 × 10
−5. At a 0.05 confidence level, it can be rejected that there is no linear correlation. The error of the slope is 3.86 ± 0.53 cm/d. In spite of the large number of influencing factors, the increase of the gradient indicates an effective rejection of suspended solids and microbial growth.
The gradient and the resistance build-up are subject to many influencing factors apart from the microbiological growth and biofilm development. Combined, these factors can cause large random variations in the hydraulic gradient and the development of the resistance against filtration. Such factors include:
sedimentation of small particles (e.g., in stagnant river areas),
erosion of the top boundary layer (e.g., during floods or dredging),
three-phase flow (e.g., gas bubbles from biological activity),
non-zero order biomass growth (fluctuations in substrate concentration, temperature, pH, etc.),
changes in viscosity,
precipitation and dissolution of carbonates, iron-hydroxides and other compounds at the boundary.
Under the conditions of the experiment, the rate of the resistance build-up was 4 cm/d (cm stands for the measured difference in the piezometric levels). The developed biofilm and boundary layer could be observed with the naked eye after 1–2 weeks of operation (
Figure 5a–c). Its thickness was 2–3 mm.
3.2. Changes in Microbiological Indicator Parameters
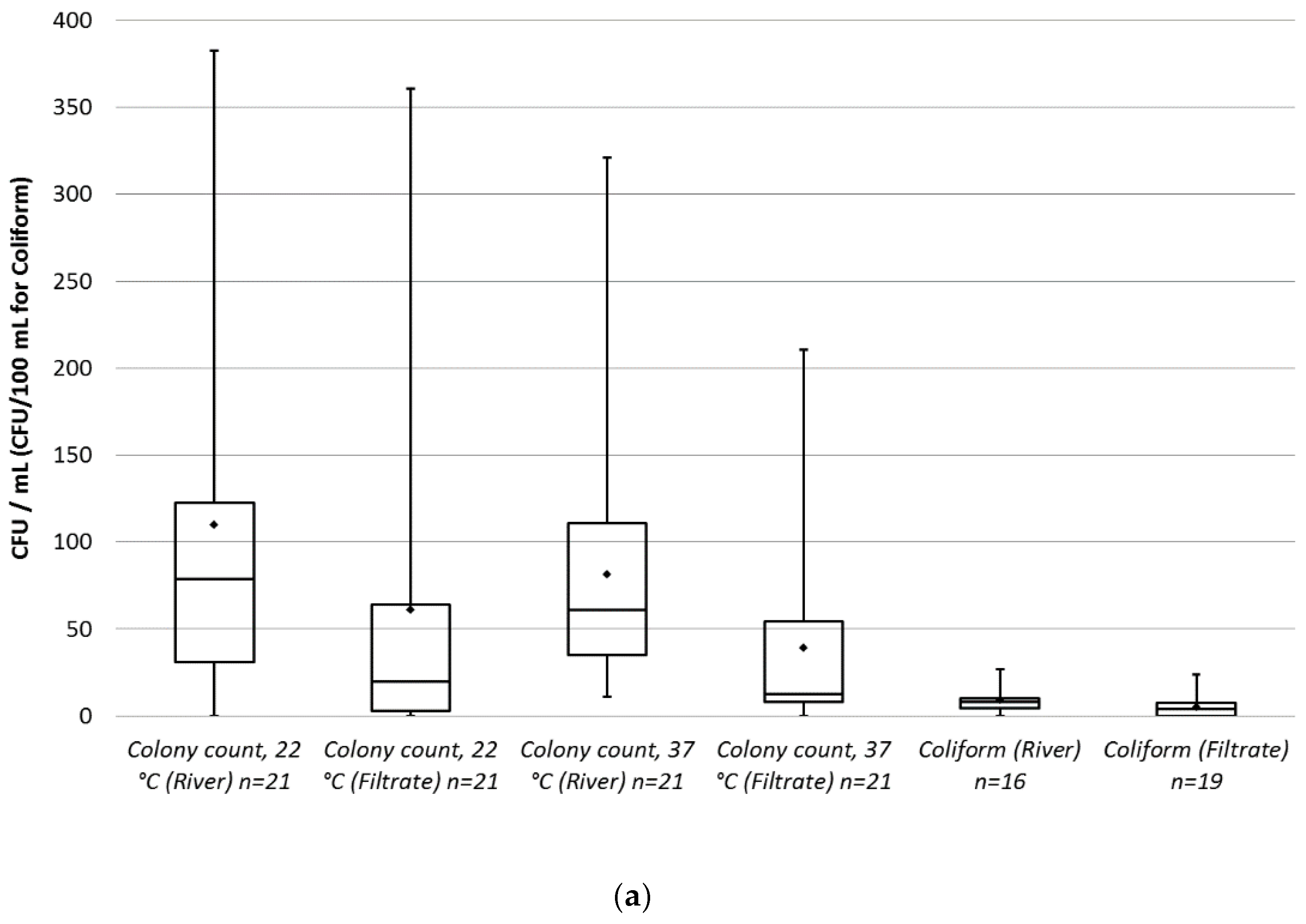
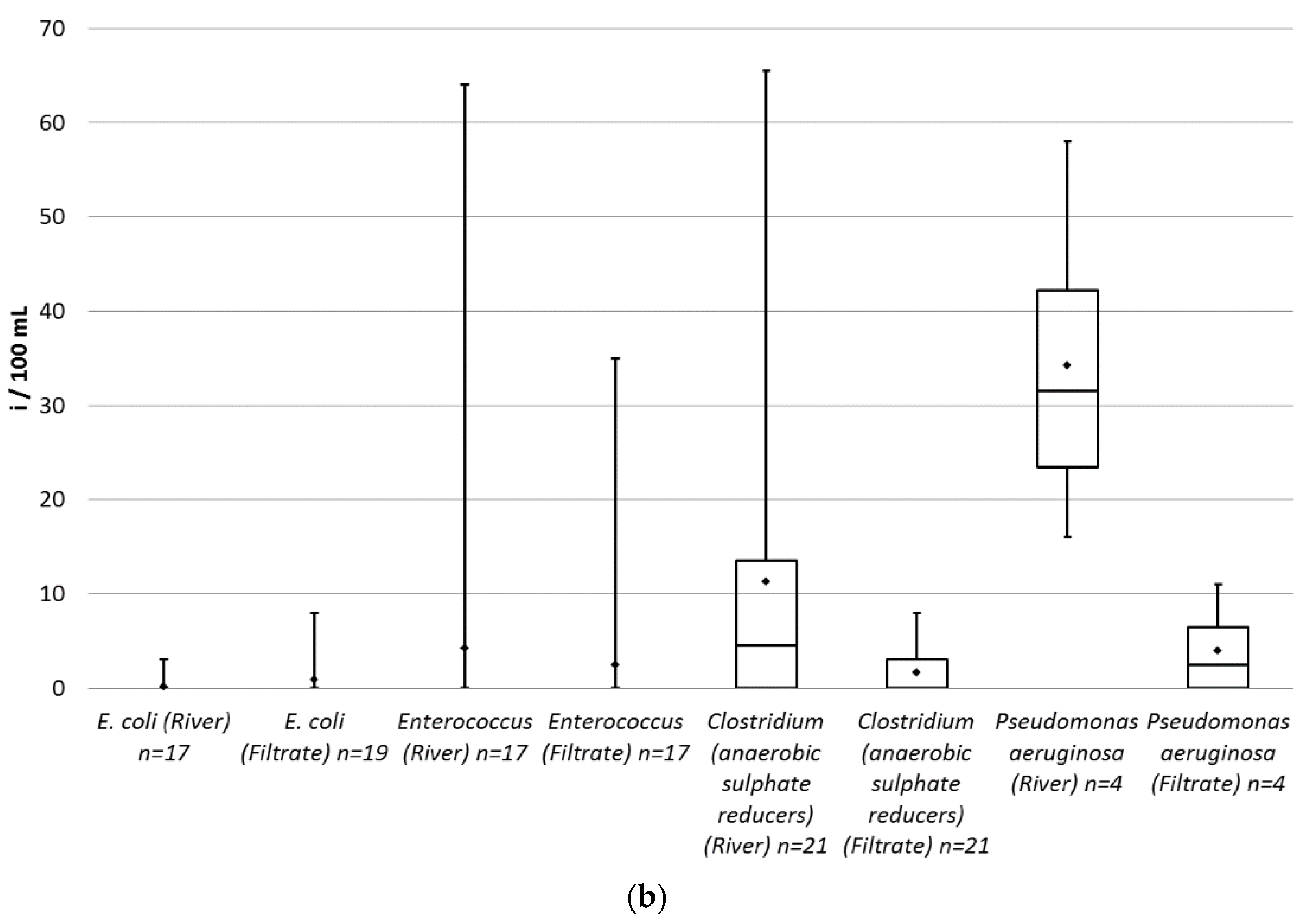
A box plot was created for the measured values of each parameter in the raw and in the filtered water (
Figure 6a,b). Some results for coliforms and
E. coli were omitted, because four samples could not be analyzed for
E. coli by the BUWW laboratory due to the unexpectedly high interference from all other microbiological growth. In case of
E. coli and
Enterococcus, only 1–3 non-zero results were obtained and only the minimum (0), the maximum and the average are shown on the graph. The number of samples is also given after the name of the parameters.
All parameters, except
E. coli, have a lower average and median in the filtrate compared to river water.
E. coli was present in only one river and two filtrate samples: 3 i/100 mL (individuum/100 mL), 6 i/100 mL and 3 i/100 mL respectively. Therefore, there is insufficient data to evaluate the change during the filtration for
E. coli. The reduction in percentage was calculated for every parameter based on the raw water samples with non-zero values (
Table 3).
Twenty samples were analyzed for each parameter, except for Enterococcus (17 samples) and Pseudomonas aeruginosa (four samples). Both the raw water and the filtered water showed high variation in the measured concentrations. Based on the average reductions, the box diagrams and the non-zero samples from river, the most effective reduction is achieved regarding Pseudomonas, Clostridium and colony count. Coliform and E. coli numbers were close to zero at all times. This is due to the clean state of the source water and to the lack of any significant fecal contamination and wastewater discharge during the sampling period.
A paired
t-test was carried out on all parameters except
E. coli, which has too much zero measured data (
Table 4). At a 0.05 significance level, it can be rejected that there is no difference between samples from the river and the filtered water for all parameters, except for coliforms and
Enterococcus.
3.3. Reverse Osmosis Results
The RO unit has been operated with slow filtered water produced purely from surface water from 1 February 2017 to 10 July 2017. The river water was spiked with groundwater extracted from a nearby well during a second test period from 12 July 2017 to 2 November 2017. The groundwater contained dissolved iron in concentrations of 5–10 mg/L and manganese in concentrations of 2–5 mg/L. Before spiking, dissolved iron concentrations were below 100 µg/L and manganese was below the detection limit (10 µg/L) above the filter. Dissolved iron concentration increased to 150–200 µg/L and manganese to 300 µg/L above the filter after spiking.
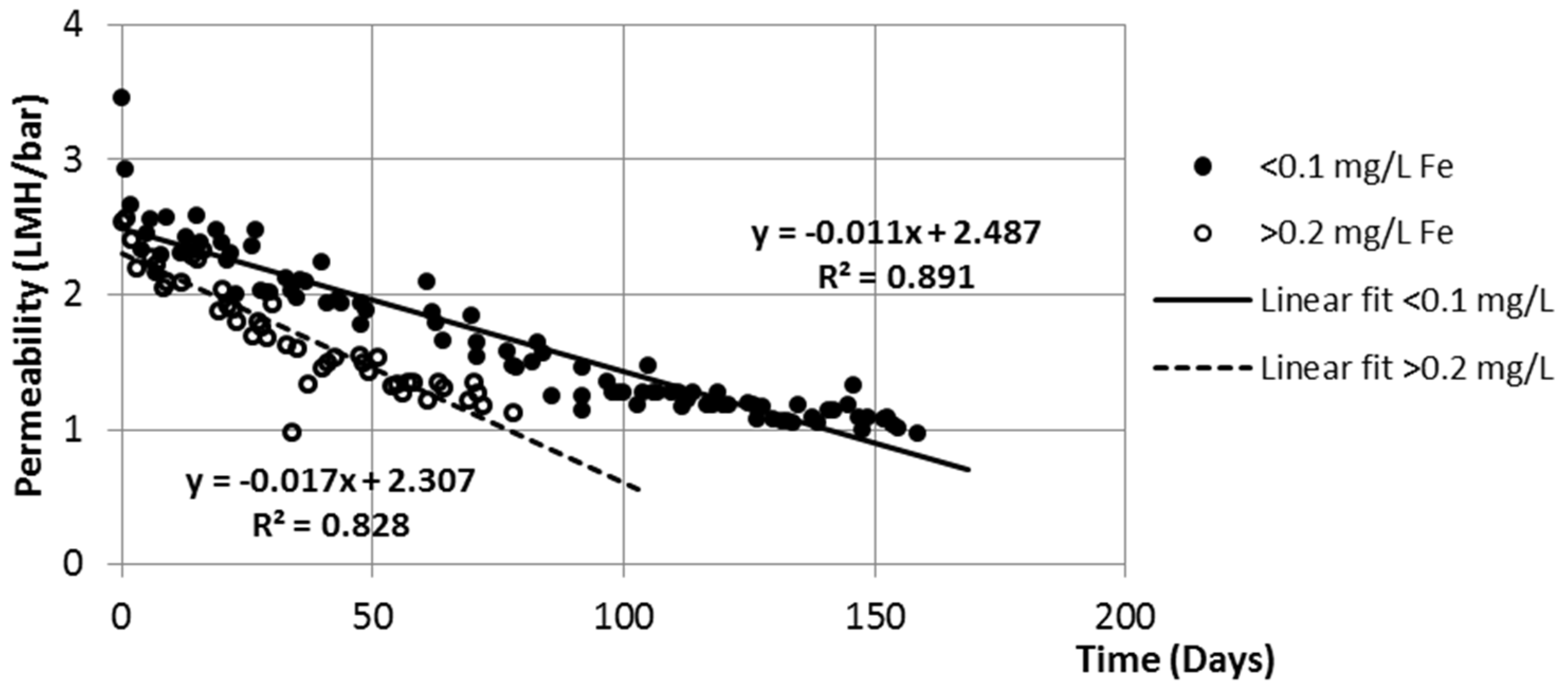
In order to push the slow filtration system more toward anoxic conditions, fresh riverbed material from the Danube was placed on top of the sand in a 10 cm thick layer. Even after this, 1–4 mg/L dissolved oxygen could be observed in the filtrate, but dissolved iron concentration in the filtrate increased to 200–250 µg/L. As shown in
Figure 7, the long-term permeability rate increased from 0.011 LMH/bar/day to 0.017 LMH/bar/day. LMH reprsenets the membrane flux and it stands for “liter per square meter per hour”, L·m
−2·h
−1. Divided by the transmembrane pressure and the elapsed time, it gives the average rate of permeability decrease.
Apart from the random errors due to the manual reading of pressure gauges and flow meters, the permeability shows a clear decreasing tendency, as expected. A sudden drop can be observed when the filtration is started after each chemical cleaning. After the first 1–2 days, the plotted linear tendency is present for a long duration (50–150 days). Even though the permeability decreased to one-third of its original value, it was possible to restore it.
4. Discussion
It has to be noted that indicator parameters are not well suited for the quantitative assessment of pathogens or microbial activity. These standard methods are primarily designed as safety indicators for distinguishing between the negative and positive (non-zero) samples. In order to assess microbiological activity and the biomass, other emerging methods are available such as ATP or RNA analysis, measurement of certain enzymes, etc. [
18].
Due to the low number of bacteria in the river water, only less than 1 log removal values could be observed during the time of the experiment. The good quality of the river water can be explained by no wastewater discharge nor any polluting activity in the vicinity and the location of the water intake structure at a stagnant zone in the river. Most suspended solids from the main stream settle before they reach the inlet of the water intake.
Because of these low initial values, a strong reduction could only be observed for three parameters: colony count at different temperatures and Pseudomonas. Although colony count has the largest range in reduction (10–100%), it gives the best proof of reduction in the number of microorganisms since it had the most positive samples in the river water. An even better reduction was achieved regarding Pseudomonas, however, two out of four samples from the river were found to be free of Pseudomonas.
The results obtained in this experiment for the reduction of microbiological indicator parameters are well within the range of previously reported values [
7]. For
Pseudomonas and total bacteria, 60% removal was obtained in small scale filter columns with 1 m thick sand layer and 0.15 m/h loading rate. It was also found that
Pseudomonas species dominate the bacterial biofilm [
19]. During the interpretation of the results, it has to be taken into account that even though there is an overall reduction in microbiological activity due to substrate consumption, the developed biofilm itself can detach from the solid surfaces of the filter and may cause higher results in the samples. Small slow sand filters with a thin 15 cm fine sand layer were reported to reject coliforms with 90–97% efficiency [
20].
Due to the very low pathogen content of surface water involved in this demonstration, it is problematic to compare results with similar studies. A similar investigation on the application of reverse osmosis directly after bank filtration described in “Desalination and Water Purification Research and Development Program Report No. 122” [
15], where coliforms ranged between 200–10,000 colony forming units (CFU)/100 mL in three investigated river sites. The report also mentions 0.052–1.62 mg/L of iron and 0.05–0.617 mg/L of manganese in the bank filtered water used for reverse osmosis membrane feed from the Ohio, Missouri and Raccoon rivers. Total rejection of coliforms on the reverse osmosis was also reported [
15].
The feed water quality requirement for the membranes involved in the current demonstration are less than 0.1 mg/L of total iron and manganese and less than 100 CFU/mL according to the manufacturer’s suggestions.
The transport of slowly biodegradable organic matter in the surface water should be taken into account when the short distance to the extraction point is considered. Such materials are humic acids, lignin and another plant residues, which can be found in surface waters used for bank filtration. The biologically active layer and the subsequent adsorption processes during slow filtration are capable of reducing the concentration of such components [
21]. If iron and manganese precipitates in the active layer due to the oxic conditions, it can be beneficial not only to the adsorption of the colloid material, but for the immobilization of microorganisms as well [
22], as long as severe clogging can be avoided.
Since the experiment was conducted under circumstances characteristic for pure slow sand filtration, its results support the hypothesis that the thin boundary layer and a short (1.2 m) flow through a porous media can be responsible for as much as 80% or more reduction in the common microbiological indicator parameters. Compared with common flow path lengths at Hungarian bank filtration sites of 10–50 m and the high iron and manganese concentration in land-side groundwater, it is important to define the ideal flow path length during the design. This requires a cost–benefit analysis, especially when iron and manganese sensitive membrane technologies are to be installed.
5. Conclusions
In general, it is customary to increase the distance of the extraction element from the surface water boundary for safety considerations. In case of combined systems, when new post-treatment technologies such as ultrafiltration, nanofiltration or reverse osmosis with high pathogen removal efficiency are applied, shorter flow path lengths could be an advantage to prevent increased concentration of dissolved iron and manganese. In the case of RBF, there are severe uncertainties and safety considerations for the distance of the extraction point from the river. The most important is riverbed erosion, which heavily puts the extent of the biologically active zone into question. On the other hand, artificial structures such as filter dams with geotextile support may be adequate to provide proper conditions for short travel distances.
It was found that as long as the bank filtered water fulfills the required quality standards set by the manufacturer, the permeability of the reverse osmosis membrane can be sustained for a long time, even for 50–100 days. The quality of the pumped water as a mixture of bank filtrate and land-side groundwater is site specific and in Hungary, especially at the lower Danube region, tends to contain more than 0.2 mg/L of dissolved iron due to the groundwater flow. In order to make the long-term operation of reverse osmosis possible, short distances between the river bank and the extraction point will help to maintain oxic conditions and minimize the iron input from groundwater flow. Reverse osmosis is known to be capable of rejecting pathogens almost completely. Even if a higher number of bacteria break through the bank filtration because of the shorter distances, the risks should not be higher than for classic water treatment processes.
In this way, the natural system (the biologically active zone) can be utilized not only to decrease turbidity and reject a significant number of pathogens, but to reduce the biofouling potential of the membrane by the removal of easily degradable substrates which otherwise contribute to biofilm growth. Membrane filtration techniques have a high efficiency in pathogen rejection, but are sensitive to fouling caused by iron or manganese precipitation. When membrane filtration is applied, the decrease in the portion of land-side groundwater is of first priority and the pathogen rejection can be safely carried out with the combination of short filtration by RBF or other MAR schemes and membrane treatment.