Effect of Water Management Technology Used in Trout Culture on Water Quality in Fish Ponds
Abstract
:1. Introduction
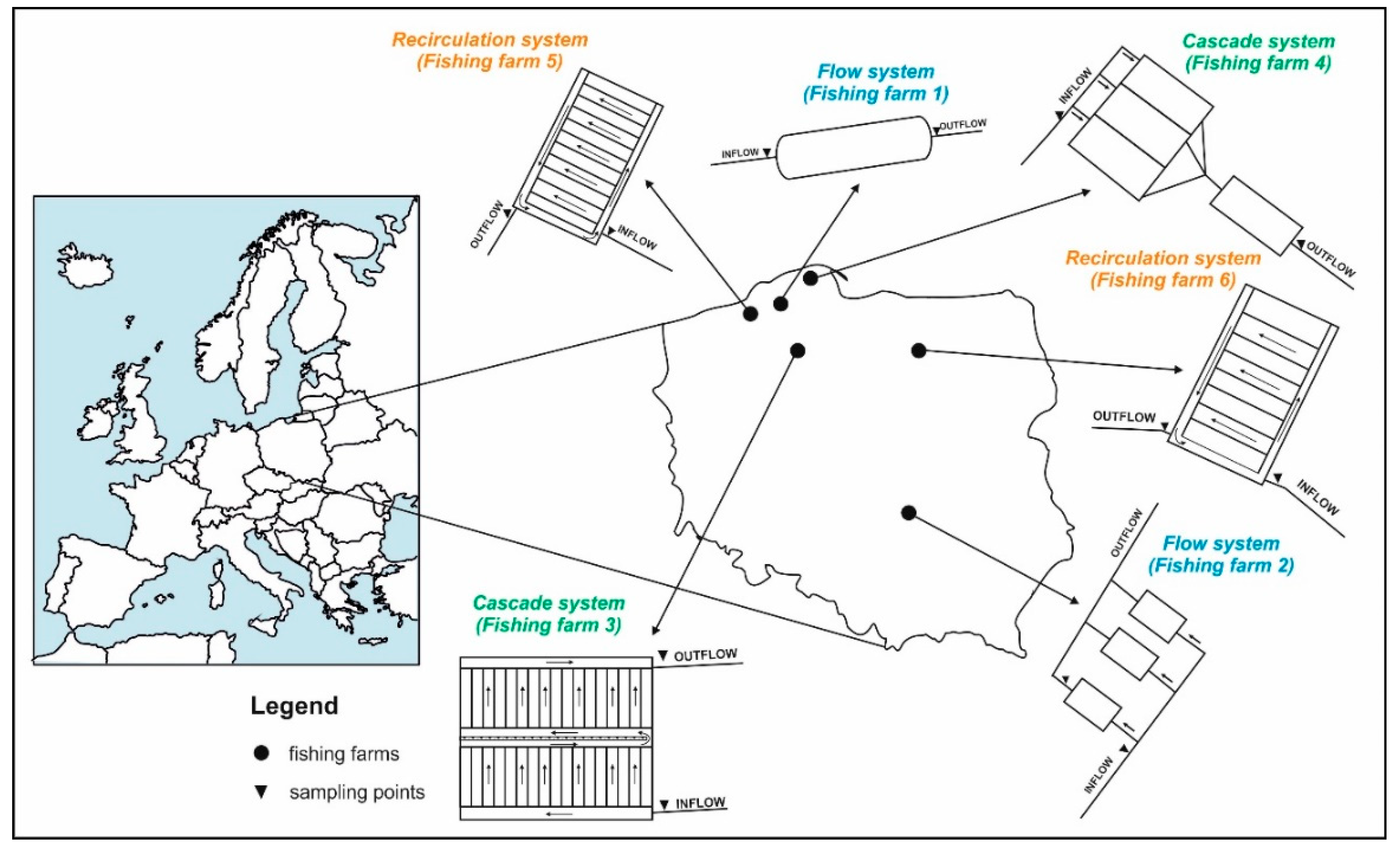
2. Materials and Methods
- (a)—mole reaction of un-ionized ammonia,
- TAN—total ammonia nitrogen (mg·dm−3),
- T—temperature of water in a fish pond,
- pH—pH of water in a fish pond.
Calculation of Water Quality Index
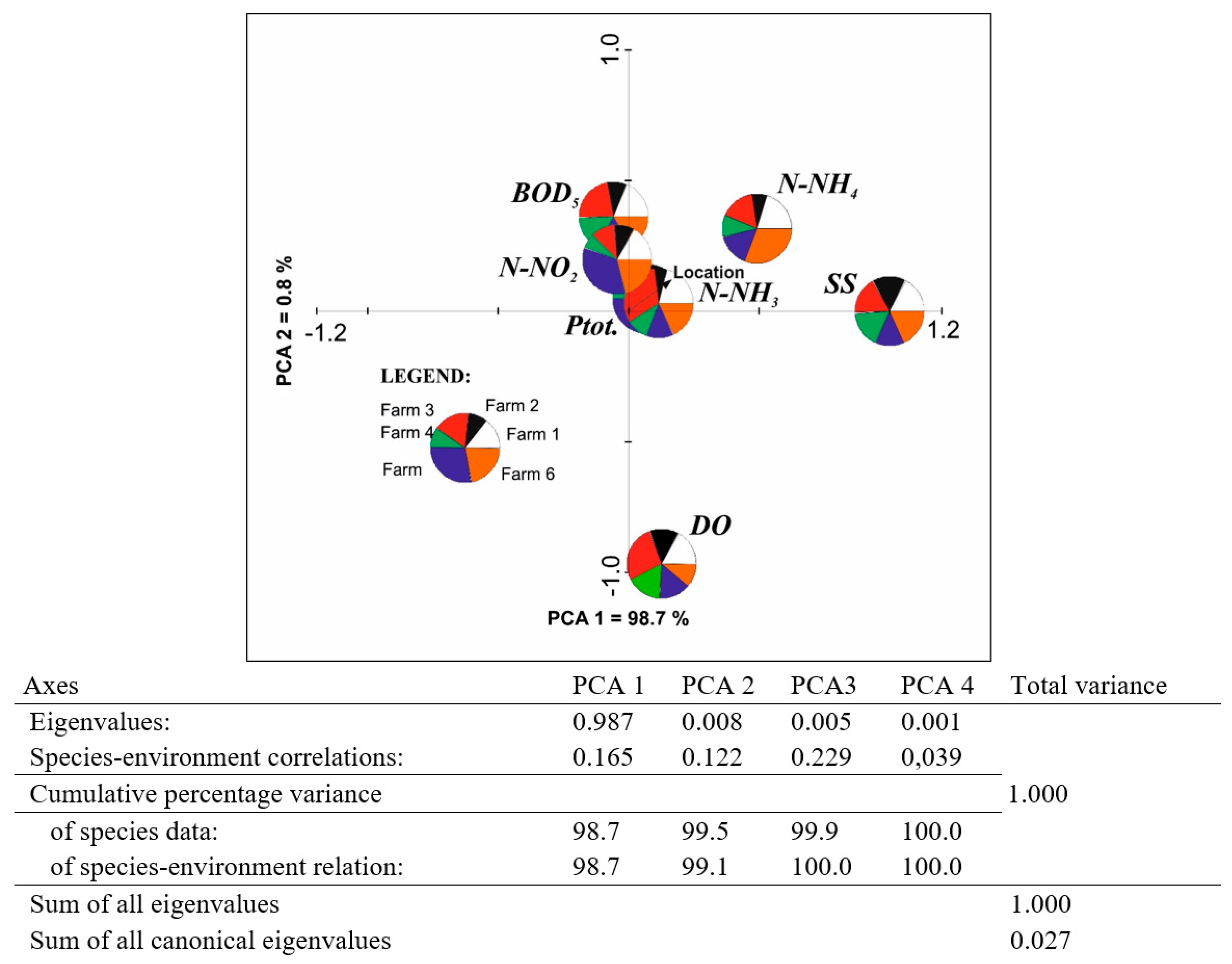
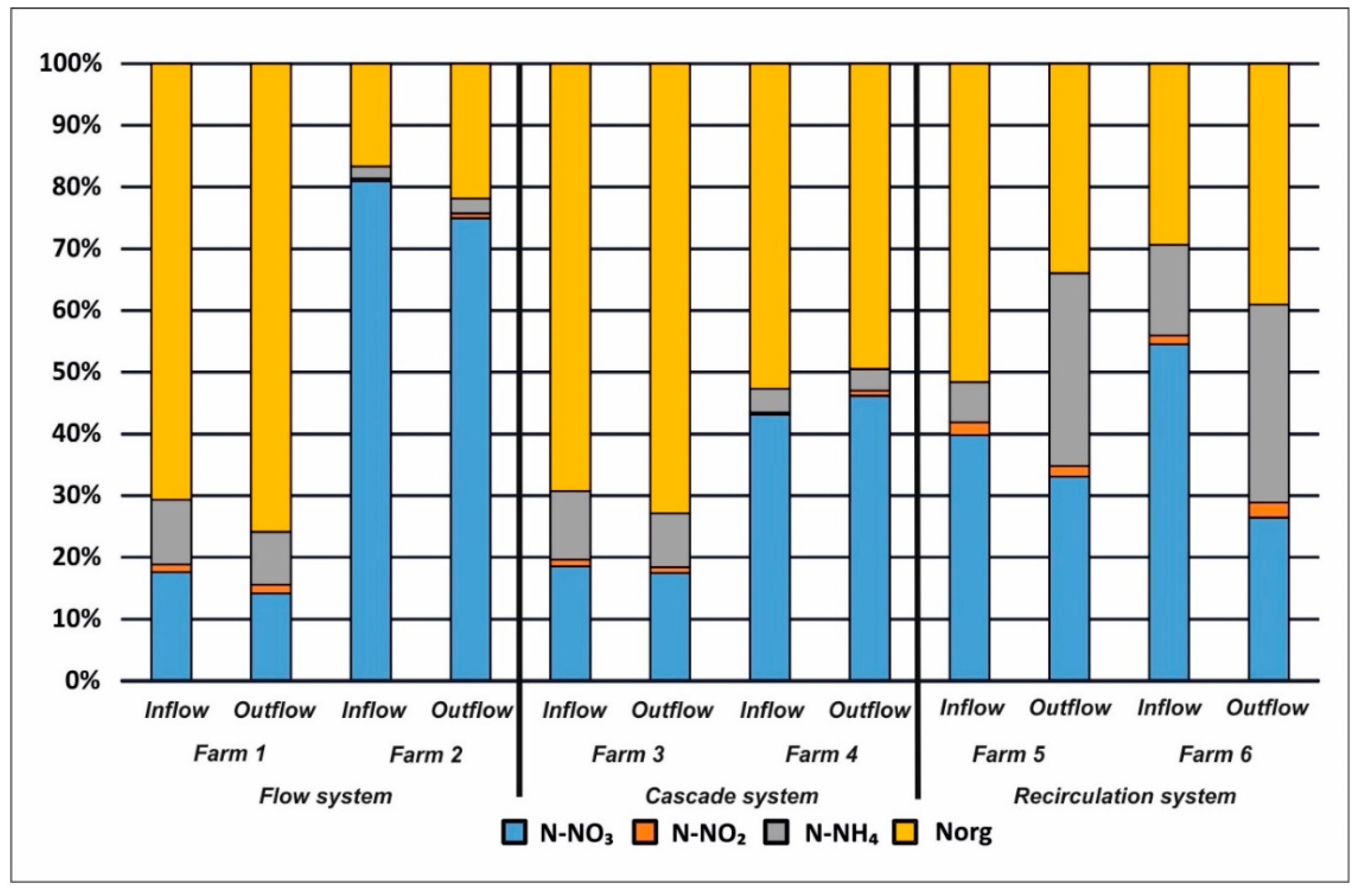
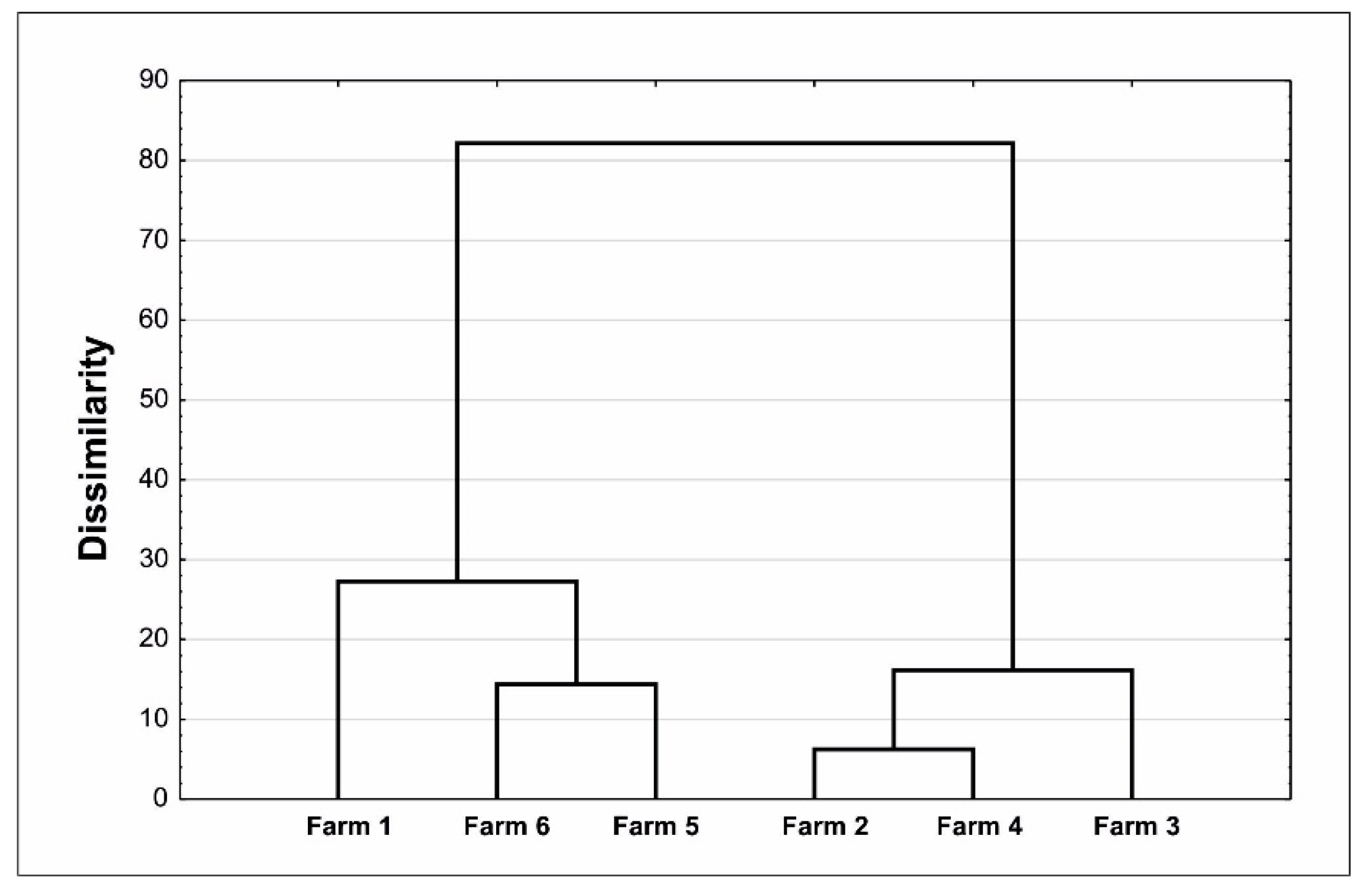
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations (FAO). The State of World Fisheries and Aquaculture; FAO: Rome, Italy, 2004. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO). The State of World Fisheries and Aquaculture; FAO: Rome, Italy, 2006. [Google Scholar]
- Amirkolaie, A.K. Reduction in the environmental impact of waste discharged by fish farms through feed and feeding. Rev. Aquac. 2011, 3, 19–26. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Garg, S.K. Causative factors of fish mortality in still water fish ponds under sub-tropical conditions. Aquaculture 2000, 1, 91–96. [Google Scholar]
- De Leo, F.; Miglietta, P.P.; Pavlinović, S. Marine ecological footprint of Italian Mediterranean fisheries. Sustainability 2014, 6, 7482–7495. [Google Scholar] [CrossRef]
- Brinker, A.; Koppe, W.; Rösch, R. Optimizing trout farm effluent treatment by stabilizing trout feces: A field trial. N. Am. J. Aquac. 2005, 67, 244–258. [Google Scholar] [CrossRef]
- Sindilariu, P.D.; Brinker, A.; Reiter, R. Waste and particle management in a commercial, partially recirculating trout farm. Aquac. Eng. 2009, 41, 127–135. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Devi, P. Water quality guidelines for the management of pond fish culture. Int. J. Environ. Sci. 2013, 3, 1980–2009. [Google Scholar]
- Yapo, M.L.; Yalamoussa, T.U.O.; Mouhamadou, K.O.N.E.; Atsé, B.C.; Kouassi, P. Can use the Biotic Index as an indication of fish farm pond water quality? J. Adv. Bot. Zool. 2017, 4, 419010529. [Google Scholar]
- Dhawan, A.; Kaur, S. Pig dung as pond manure: Effect on water quality, pond productivity and growth of carps in polyculture system. NAGA ICLARM Q. 2002, 25, 11–14. [Google Scholar]
- Kiran, B.R. Physico-chemical characteristics of fish ponds of Bhadra project at Karnataka. Rasayan J. Chem. 2010, 3, 671–676. [Google Scholar]
- Hlaváč, D.; Adámek, Z.; Hartman, P.; Másílko, J. Effects of supplementary feeding in carp ponds on discharge water quality: A review. Aquac. Int. 2014, 22, 299–320. [Google Scholar] [CrossRef]
- Mohammed, B.; Tewabe, D.; Zelalem, W.; Melaku, A. Physical, Chemical, Biological properties and fish species type of Geray reservoir,-W/Gojjam Zone, Ethiopia. Int. J. Aquac. Fish Sci. 2016, 2, 8–11. [Google Scholar]
- Beveridge, M.C.M.; Philips, M.J.; Macintosh, D.C. Aquaculture and environment: the supply and demand for environment goods and services by Asian aquaculture and the implications for sustainability. Aquac. Res. 1997, 28, 101–111. [Google Scholar] [CrossRef]
- Chen, S.; Coffin, D.E.; Malone, R.F. Sludge production and management for recirculating aquaculture system. J. World Aquac. Soc. 1997, 28, 303–315. [Google Scholar] [CrossRef]
- Blancheton, J.P. Developments in recirculation systems for Mediterranean fish species. Aquac. Eng. 2000, 22, 17–31. [Google Scholar] [CrossRef]
- Sikoki, F.D.; Veen, J.V. Aspects of water quality and the potential for fish production of Shinro Reservour, Nigeria. J. Fish. Aqua. Sci. 2012, 8, 186–204. [Google Scholar]
- Ray, L.I.P.; Panigrahi, P.K.; Mal, B.C. Temporal variation of water quality parameters in intensively IMC cultured lined pond. Univ. de Ştiinţe Agricole şi Medicină Veterinară Iaşi Lucrări Ştiinţifice, Seria Zootehnie. 2009, 52, 429–437. [Google Scholar]
- Bhatnagar, A.; Singh, G. Culture fisheries in village ponds: a multi-location study in Haryana, India. Agric. Biol. J. N. Am. 2010, 1, 961–968. [Google Scholar] [CrossRef]
- Dalsgaard, J.; Larsen, B.K.; Pedersen, P.B. Nitrogen waste from rainbow trout (Oncorhynchus mykiss) with particular focus on urea. Aquac. Eng. 2015, 65, 2–9. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 15th ed.; Water Pollution Control Federation: New York, NY, USA, 1992. [Google Scholar]
- Petit, J. Water supply, treatment and recycling in aquaculture. Aquaculture 1990, 1, 63–196. [Google Scholar]
- Breabăn, I.G.; Paiu, M. Application of DRASTIC Model and GIS for Evaluation of Aquifer Vulnerability: Study Case Barlad City Area. In Proceedings of the Water Resources and Wetlands, Tulcea, Romania, 14–16 September 2012; pp. 588–593. [Google Scholar]
- Mazaheri, K.Z.; Ghorbani, R.; Hajimoradloo, A.; Naeimi, A. The effect of trout farm effluents on the water quality parameters of Zaringol Stream (Golestan, Iran) based on NSFWQI and WQI indexes. Environ. Resour. Res. 2014, 1, 91–201. [Google Scholar]
- Mirsaeedghazi, H. Effect of trout farm on the water quality of river using Iran Water Quality Index (IRQWI): A case study on Deinachal River. J. Food Bioprocess Eng. 2015, 1, 17–26. [Google Scholar]
- Cymes, I. Use of water quality indices as a tool in water resources management. Fresenius Environ. Bull. 2018, 27, 2777–2784. [Google Scholar]
- Bharti, N.; Katyal, D. Water quality indices used for surface water vulnerability assessment. Int. J. Environ. Sci. 2011, 2, 154–173. [Google Scholar]
- Canadian Water Quality Guidelines for the Protection of Aquatic Life: CCME Water Quality Index 1.0, Technical Report. In Canadian Environmental Quality Guidelines; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 2001.
- Rozporządzenie, M.Z. Z dnia 16 października 2002 r. w sprawie wymagań, jakim powinna odpowiadać woda w kąpieliskach. Dz. U. 2002, 183, 1530. [Google Scholar]
- Directive Council 78/659/EEC of 18 July 1978 on the quality of fresh waters needing protection or improvement in order to support fish life. Eur. Commun. 1978, 222, 1–10.
- De Leo, F.; Miglietta, P.P.; Pavlinović, S. Marine fisheries and mariculture in Croatia: Economic and trade analysis. J. Econ. Financ. Stud. 2014, 2, 53–61. [Google Scholar] [CrossRef]
- Amirkolaie, A.K. Environmental impact of nutrient discharged by aquaculture waste water on Haraz the river. J. Fish. Aqua. Sci. 2008, 3, 275–279. [Google Scholar] [CrossRef]
- Farmaki, E.G.; Thomaidis, N.S.; Pasias, I.N.; Baulard, C.; Papaharisis, L.; Efstathiou, C.E. Environmental impact of intensive aquaculture: Investigation on the accumulation of metals and nutrients in marine sediments of Greece. Sci. Total Environ. 2014, 485, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Clark, M.; Tilman, D. Comparative analysis of environmental impacts of agricultural production systems, agricultural input efficiency, and food choice. Environ. Res. Lett. 2017, 12, 1–10. [Google Scholar] [CrossRef]
- Sidoruk, M.; Koc, J.; Cymes, I.; Rafałowska, M.; Rochwerger, A.; Sobczyńska-Wójcik, K.; Skibniewska, K.A.; Siemianowska, E.; Guziur, J.; Szarek, J. Risk assessment of surface waters associated with water circulation technologies on trout farms. J. Ecol. Eng. 2014, 15, 76–81. [Google Scholar]
- Fetherman, E.R.; Wardell, J.A.; Praamsma, Ch. J.; Hura, M.K. Critical Dissolved Oxygen Tolerances of Whirling Disease-Resistant Rainbow Trout. N. Am. J. Aquac. 2016, 78, 366–373. [Google Scholar] [CrossRef]
- Caldwell, C.A.; Hinshaw, J.M. Communications: Tolerance of Rainbow Trout to dissolved oxygen supplementation and a Yersinia ruckeri challenge. J. Aqua. Anim. Health 1995, 7, 168–171. [Google Scholar] [CrossRef]
- Sirakov, I.; Staykov, Y.; Djanovski, G. Consumption of dissolved oxygen in rainbow trout (Oncorhynchus mykiss) cultivated in raceway. Agric. Sci. Technol. 2011, 3, 220–223. [Google Scholar]
- Roze, T.; Christen, F.; Amerand, A.; Claireaux, G. Trade-off between thermal sensitivity, hypoxia tolerance and growth in fish. J. Therm. Biol. 2013, 38, 98–106. [Google Scholar] [CrossRef]
- Bonisławska, M.; Tański, A.; Mokrzycka, M.; Brysiewicz, A.; Nędzarek, A.; Tórz, A. The effect of effluents from rainbow trout ponds on water quality in the Gowienica River. J. Water Land Dev. 2013, 19, 23–30. [Google Scholar] [CrossRef]
- Sidoruk, M.; Koc, J.; Szarek, J.; Skibniewska, K.; Guziur, J.; Zakrzewski, J. Effect of trout production in concrete fish ponds with a cascade water flow on physical and chemical properties of surface waters. Inż. Ekolo. 2013, 34, 206–213. (In Polish) [Google Scholar] [CrossRef]
- Svobodová, Z.; Lloyd, R.; Máchová, J.; Vykusová, B. Water Quality and Fish Health; EIFAC Technical Paper; FAO: Rome, Italy, 1993; 54p. [Google Scholar]
- Kumar, D.; Karthik, M.; Rajakumar, R. Study of seasonal water quality assessment and fish pond conservation in Thanjavur, Tamil Nadu, India. J. Entomol. Zool. Stud. 2017, 5, 1232–1238. [Google Scholar]
- Hargreaves, J.A. Nitrogen biogeochemistry of aquaculture ponds. Aquaculture 2008, 166, 181–212. [Google Scholar] [CrossRef]
- Ahn, Y.H. Sustainable nitrogen elimination biotechnologies: A review. Process Biochem. 2006, 41, 1709–1721. [Google Scholar] [CrossRef]
- Robertson, G.P.; Vitousek, P.M. Nitrogen in agriculture: Balancing the cost of an essential resource. Annu. Rev. Environ. Resour. 2009, 34, 97–125. [Google Scholar] [CrossRef]
- Yossi, T.; Schreier, H.J.; Sowers, K.R.; Stubblefield, J.D.; Place, A.R.; Zohar, Y. Environmentally sustainable land-based marine aquaculture. Aquaculture 2009, 286, 28–35. [Google Scholar]
- Cymes, I.; Glinska-Lewczuk, K. The use of water quality indices (WQI and SAR) for multipurpose assessment of water in dam reservoirs. J. Elementol. 2016, 21, 1211–1224. [Google Scholar]
- de LG Solbé, J.F.; Shurben, D.G. Toxicity of ammonia to early life stages of rainbow trout (Salmo gairdneri). Wat. Res. 1989, 23, 127–129. [Google Scholar] [CrossRef]
- Randall, D.J.; Tsui, T.K.N. Ammonia toxicity in fish. Mar. Pollut. Bull. 2002, 45, 17–23. [Google Scholar] [CrossRef]
- Madeyski, M. Effect of fish ponds on selected components of the natural environment. Inż. Śr. 2001, 21, 139–144. (In Polish) [Google Scholar]
- Hernández, A.J.; Roman, D. Phosphorus and nitrogen utilization efficiency in rainbow trout (Oncorhynchus mykiss) fed diets with lupin (Lupinus albus) or soybean (Glycine max) meals as partial replacements to fish meal. Czech J. Anim. Sci. 2016, 61, 67–74. [Google Scholar] [CrossRef]
- Sugiura, S.H. Phosphorus, Aquaculture, and the Environment. Rev. Fish. Sci. Aquac. 2018, 26, 515–521. [Google Scholar] [CrossRef]
- Brown, C.; Laland, K. Social learning and life skills training for hatchery reared fish. J. Fish Biol. 2001, 59, 471–493. [Google Scholar] [CrossRef]
- Brown, C.; Day, R.L. The future of stock enhancements: lessons for hatchery practice from conservation biology. Fish Fish. 2002, 3, 79–94. [Google Scholar] [CrossRef]
- Fevolden, S.-E.; RØed, K.H.; Fjalestad, K. A combined salt and confinement stress enhances mortality in rainbow trout (Oncorhynchus mykiss) selected for high stress responsiveness. Aquaculture 2003, 216, 67–76. [Google Scholar] [CrossRef]
- Bartel, R. Present situation of the Vistula sea trout. Arch. Pol. Fish. 1993, 1, 101–111. [Google Scholar]
- Augustyn, L.; Bartel, R.; Epler, P. Effects of fish size on post-stocking mortality and growth rate of Brown Trout (Salmo trutta trutta m. Fario L.) fry. Acta Sci. Pol. 2006, 5, 17–28. [Google Scholar]
- Milner, N.J.; Elliott, J.M.; Armstrong, J.D.; Gardiner, R.; Welton, J.S.; Ladle, M. The natural control of salmon and trout populations in streams. Fish. Res. 2003, 62, 111–125. [Google Scholar] [CrossRef]

| Wqi Value | Water Quality | Description |
|---|---|---|
| 95–100 | Excellent | Water quality is protected with a virtual absence of threat or impairment, conditions very close to natural or pristine levels |
| 80–94 | Good | Water quality is protected with only a minor degree of threat or impairment, conditions rarely depart from natural or desirable levels |
| 65–79 | Fair | Water quality is usually protected but occasionally threatened or impaired, conditions sometimes depart from natural or desirable levels |
| 45–64 | Marginal | Water quality is frequently threatened or impaired, conditions often depart from natural or desirable levels |
| 0–44 | Poor | Water quality is almost always threatened or impaired, conditions usually depart from natural or desirable levels |
| Index | Objective Levels for Each Constituents as in EEC/78/669 | Flow System | Cascade System | Recirculation System | |||
|---|---|---|---|---|---|---|---|
| Farm 1 | Farm 2 | Farm 3 | Farm 4 | Farm 5 | Farm 6 | ||
| Temperature * | <21.5 | 12.9 ± 3.6 | 10.8 ± 2.0 | 11.2 ± 1.2 | 10.8 ± 6.6 | 12.5 ± 3.7 | 8.9 ± 0.6 |
| DO | >7.0 | 9.22 ± 1.95 | 8.61 ± 0.38 | 10.21 ± 0.43 | 10.31 ± 1.22 | 9.46 ± 1.80 | 8.27 ± 1.70 |
| pH ** | 6.0–9.0 | 7.78–8.48 | 6.73–7.87 | 8.00–8.66 | 7.35–8.27 | 6.39–8.25 | 7.46–8.56 |
| SS | <25 | 29 ± 29 | 30 ± 12 | 21 ± 7 | 33 ± 7 | 26 ± 11 | 42 ± 19 |
| BOD5 | <3.0 | 2.5 ± 1.2 | 1.5 ± 0.5 | 2.1 ± 1.4 | 2.5 ± 1.1 | 2.6 ± 1.1 | 3.1 ± 1.1 |
| TP | <0.2 | 0.118 ± 0.048 | 0.086 ± 0.095 | 0.096 ± 0.029 | 0.087 ± 0.026 | 0.270 ± 0.332 | 0.249 ± 0.095 |
| N-NH4 | <0.78 | 0.106 ± 0.064 | 0.046 ± 0.029 | 0.059 ± 0.025 | 0.064 ± 0.043 | 0.094 ± 0.042 | 0.22 ± 0.136 |
| N-NH3 | <0.020 | 0.004 ± 0.002 | 0.001 ± 0.001 | 0.004 ± 0.002 | 0.002 ± 0.002 | 0.003 ± 0.002 | 0.004 ±0.005 |
| N-NO2 | <0.003 | 0.012 ± 0.010 | 0.008 ± 0.008 | 0.006 ± 0.001 | 0.006 ± 0.004 | 0.029 ± 0.010 | 0.021 ± 0.021 |
| N-NO3 | – | 0.177 ± 0.061 | 1.837 ± 0.290 | 0.099 ± 0.012 | 0.713 ± 0.205 | 0.575 ± 0.311 | 0.836 ± 0.258 |
| Nmin | – | 0.289 ± 0.118 | 1.887 ± 0.294 | 0.164 ± 0.035 | 0.782 ± 0.235 | 0.698 ± 0.314 | 1.084 ± 0.285 |
| Norg | – | 0.713 ± 0.266 | 0.377 ± 0.219 | 0.369 ± 0.070 | 0.871 ± 0.629 | 0.745 ± 0.246 | 0.450 ± 0.121 |
| Ntot | – | 1.01 ± 0.22 | 2.27 ± 0.30 | 0.53 ± 0.06 | 1.65 ± 0.70 | 1.44 ± 0.32 | 1.53 ± 0.35 |
| Index | Objective Levels for Each Constituents as in EEC/78/669 | Flow System | Cascade System | Recirculation System | |||
|---|---|---|---|---|---|---|---|
| Farm 1 | Farm 2 | Farm 3 | Farm 4 | Farm 5 | Farm 6 | ||
| Temperature * | <21.5 | 12.4 ± 3.8 | 10.3 ± 2.0 | 11.2 ± 1.5 | 10.9 ± 6.6 | 14.0 ± 3.1 | 11.4 ± 2.9 |
| DO | >7.0 | 8.66 ± 2.67 | 8.96 ± 0.74 | 8.97 ± 0.88 | 10.21 ± 1.54 | 8.23 ± 2.03 | 7.97 ± 1.57 |
| pH ** | 6.0–9.0 | 7.85–8.32 | 7.05–8.35 | 6.94–8.25 | 7.41–8.12 | 7.64–7.97 | 7.32–8.52 |
| SS | <25 | 35 ± 29 | 37 ± 25 | 23 ± 8 | 61 ± 13 | 32 ± 13 | 42 ± 13 |
| BOD5 | <3.0 | 3.2 ± 1.1 | 1.9 ± 0.4 | 2.4 ± 1.4 | 1.8 ± 0.9 | 5.8 ± 0.4 | 4.2 ± 1.1 |
| TP | <0.2 | 0.127 ± 0.036 | 0.076 ± 0.017 | 0.130 ± 0.037 | 0.111 ± 0.064 | 0.151 ± 0.035 | 0.336 ± 0.113 |
| N-NH4 | <0.78 | 0.097 ± 0.064 | 0.064 ± 0.032 | 0.062 ± 0.020 | 0.053 ± 0.057 | 0.703 ± 0.267 | 0.581 ± 0.604 |
| N-NH3 | <0.020 | 0.004 ± 0.002 | 0.005 ± 0.004 | 0.002 ± 0.001 | 0.002 ± 0.003 | 0.011 ± 0.004 | 0.010 ± 0.012 |
| N-NO2 | <0.003 | 0.016 ± 0.010 | 0.020 ± 0.016 | 0.006 ± 0.001 | 0.013 ± 0.007 | 0.040 ± 0.024 | 0.044 ± 0.054 |
| N-NO3 | – | 0.161 ± 0.041 | 1.975 ± 0.356 | 0.124 ± 0.026 | 0.702 ± 0.322 | 0.747 ± 0.216 | 0.479 ± 0.191 |
| Nmin | – | 0.275 ± 0.099 | 2.057 ± 0.373 | 0.192 ± 0.035 | 0.766 ± 0.341 | 1.490 ± 0.286 | 1.104 ± 0.540 |
| Norg | – | 0.864 ± 0.200 | 0.576 ± 0.206 | 0.516 ± 0.267 | 0.752 ± 0.228 | 0.766 ± 0.592 | 0.706 ± 0.248 |
| Ntot | – | 1.14 ± 0.23 | 2.63 ± 0.47 | 0.71 ± 0.25 | 1.52 ± 0.39 | 2.26 ± 0.50 | 1.81 ± 0.78 |
| Water Management System | |||||||
|---|---|---|---|---|---|---|---|
| Flow System | Cascade System | Recirculation System | |||||
| Farm 1 | Farm 2 | Farm 3 | Farm 4 | Farm 5 | Farm 6 | ||
| Mortality rate (%) | 0.62 ± 0.13 | 0.64 ± 0.16 | 0.63 ± 0.17 | 0.64 ± 0.15 | 0.96 ± 0.16 | 0.98 ± 0.20 | |
| Stock density | (kg·m−3) | 0.4 | 3.5 | 7.0 | |||
| (pcs/pond) | 615 ± 266 | 607 ± 263 | 1005 ± 435 | 1020 ± 451 | 1754 ± 760 | 1656 ± 717 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sidoruk, M.; Cymes, I. Effect of Water Management Technology Used in Trout Culture on Water Quality in Fish Ponds. Water 2018, 10, 1264. https://doi.org/10.3390/w10091264
Sidoruk M, Cymes I. Effect of Water Management Technology Used in Trout Culture on Water Quality in Fish Ponds. Water. 2018; 10(9):1264. https://doi.org/10.3390/w10091264
Chicago/Turabian StyleSidoruk, Marcin, and Ireneusz Cymes. 2018. "Effect of Water Management Technology Used in Trout Culture on Water Quality in Fish Ponds" Water 10, no. 9: 1264. https://doi.org/10.3390/w10091264
APA StyleSidoruk, M., & Cymes, I. (2018). Effect of Water Management Technology Used in Trout Culture on Water Quality in Fish Ponds. Water, 10(9), 1264. https://doi.org/10.3390/w10091264





