Groundwater Hydrochemical Zoning in Inland Plains and its Genetic Mechanisms
Abstract
1. Introduction
2. Research Area Overview
3. Materials and Methods
3.1. Collection and Testing of Groundwater Samples
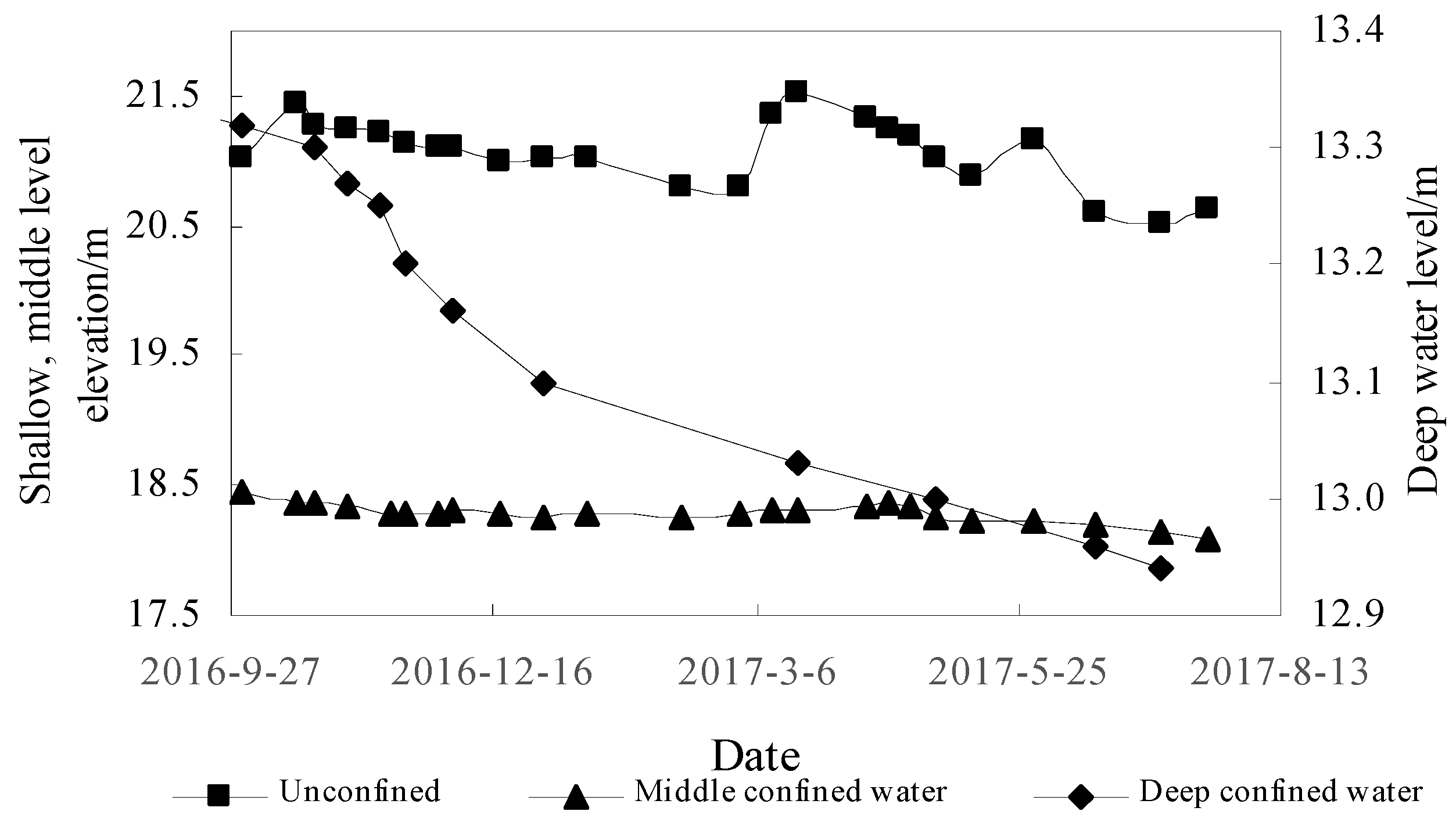
3.2. Dynamic Monitoring of Groundwater
- (1)
- Stratified monitoring of the groundwater level was undertaken at different depths in the observation holes on a weekly basis.
- (2)
- Stratified monitoring of groundwater quality characteristics was undertaken at different depths using an Aqualtroll 600-type multi-parameter water quality detector. Conductivity (EC) changes from the top to the bottom of the profiles were recorded using a test interval of 1 m. Test accuracy was ±0.5% + 1 μs/cm. Weekly monitoring commenced in November, 2016.
3.3. Adsorption Isothermal Experiment
3.3.1. Experimental Procedure
3.3.2. Calculation of the Retardation Coefficient
4. Results and Discussion
4.1. Groundwater Chemical Composition and Zoning Characteristics
4.1.1. Chemical Characteristics of Shallow Groundwater
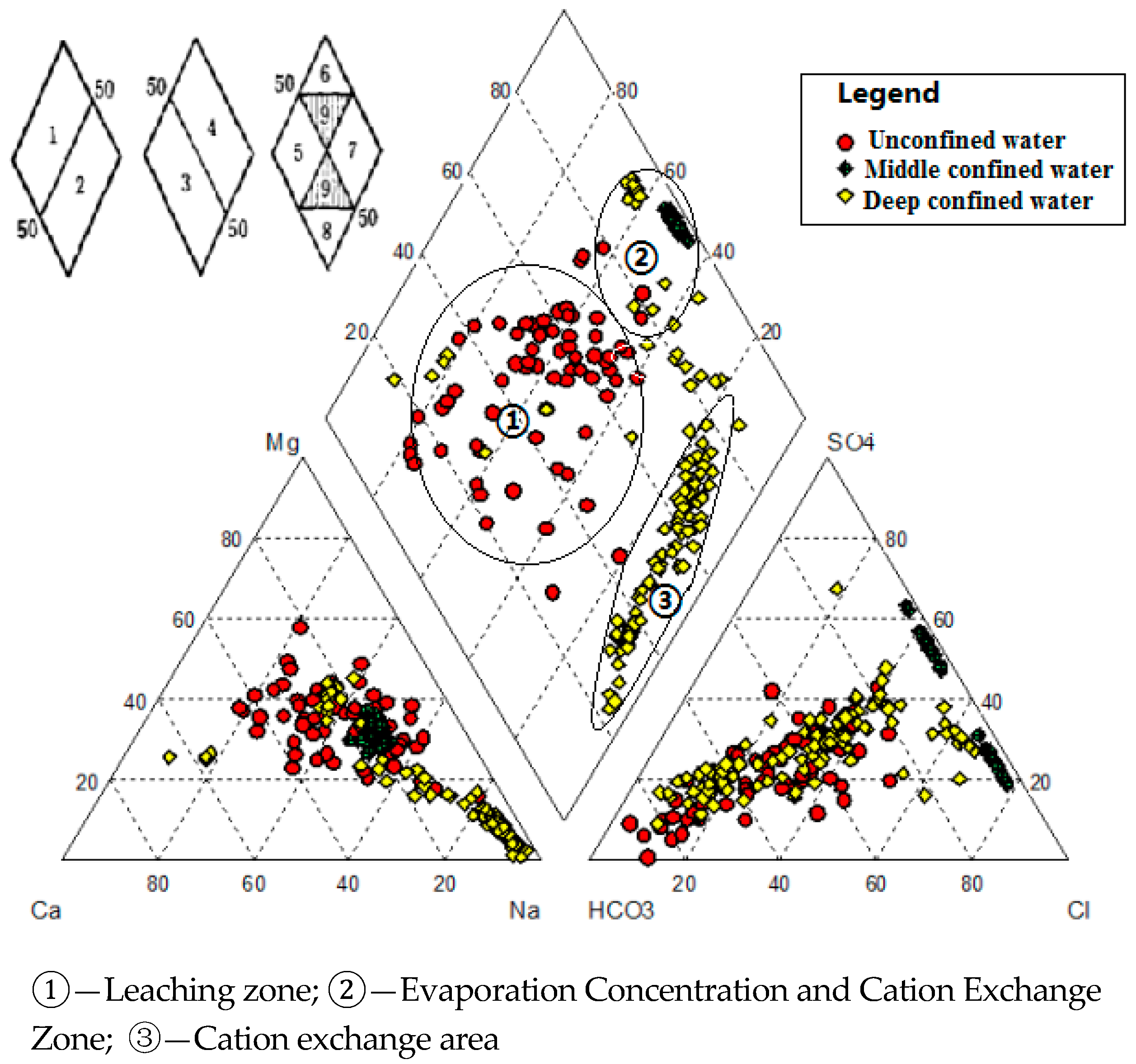
4.1.2. Hydrochemical Types and Horizontal Zoning Characteristics
- (1)
- Groundwater flow from the Southwest to the Northeast. The longer migration route, the greater the TDS of the groundwater. In Section I, Fengjiajing Village in Taiping Town (a located in the Southwest)—Hujiacun (b located in the Central)—Tanjia Village in Renfeng Town (c located in the Northeast) (Figure 1) of Taiping Town, groundwater was changed from freshwater with a TDS < 1 g·L−1 to brackish water with a TDS > 3 g·L−1. Na+, Ca2+, Mg2+, Cl−, HCO3−, SO42− and TDS all recorded a gradual increase. The hydrochemistry types were HCO3-Na·Mg ⟶ HCO3·Cl ·SO4-Na·Mg ⟶ Cl·HCO3-Na·Mg (Figure 3).
- (2)
- Brackish water and freshwater show an alternate distribution in local areas. Groundwater with a high salinity had a sporadic or irregular strip-shaped distributing interfluve between old channels (Figure 4). There are numerous seasonal streams and diversion channels in the Jiyang surface water system. Freshwater with a TDS < 2 g·L−1 is mainly distributed along the Yellow River, along the Tuhai River, near the Dasi River and around the Heishi River. The main hydrochemistry type is HCO3 type. The brackish water-saltwater area is mainly distributed between river, showing north-easterly. This area recorded a general TDS > 3 g·L−1, with results occasionally being >5 g·L−1. TDS samples in the Old Tang Temple were recorded to be >10 g·L−1. In the direction of Section II, the hydrochemistry types were HCO3·Cl-Mg·Na·Ca, Cl·SO4—Mg·Na·Ca, HCO3·Cl-Mg·Na and HCO3·Cl-Mg·Na, Cl·SO4—Na·Mg type, formed a freshwater-saltwater-freshwater-saltwater pattern.
4.2. Groundwater Chemical Composition of the Vertical Zone Characteristics
4.2.1. Changes in the Chemical Composition of Saltwater with Depth
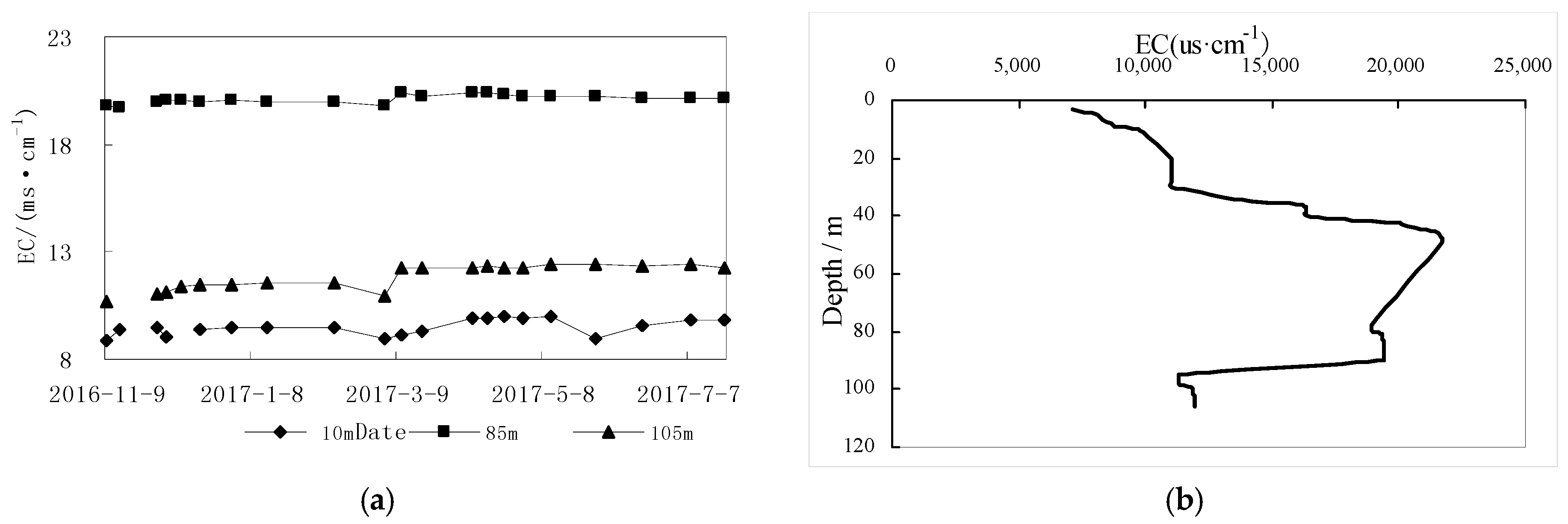
4.2.2. Groundwater Conductivity with Depth
4.2.3. Groundwater Hydrochemical Vertical Zoning Characteristics
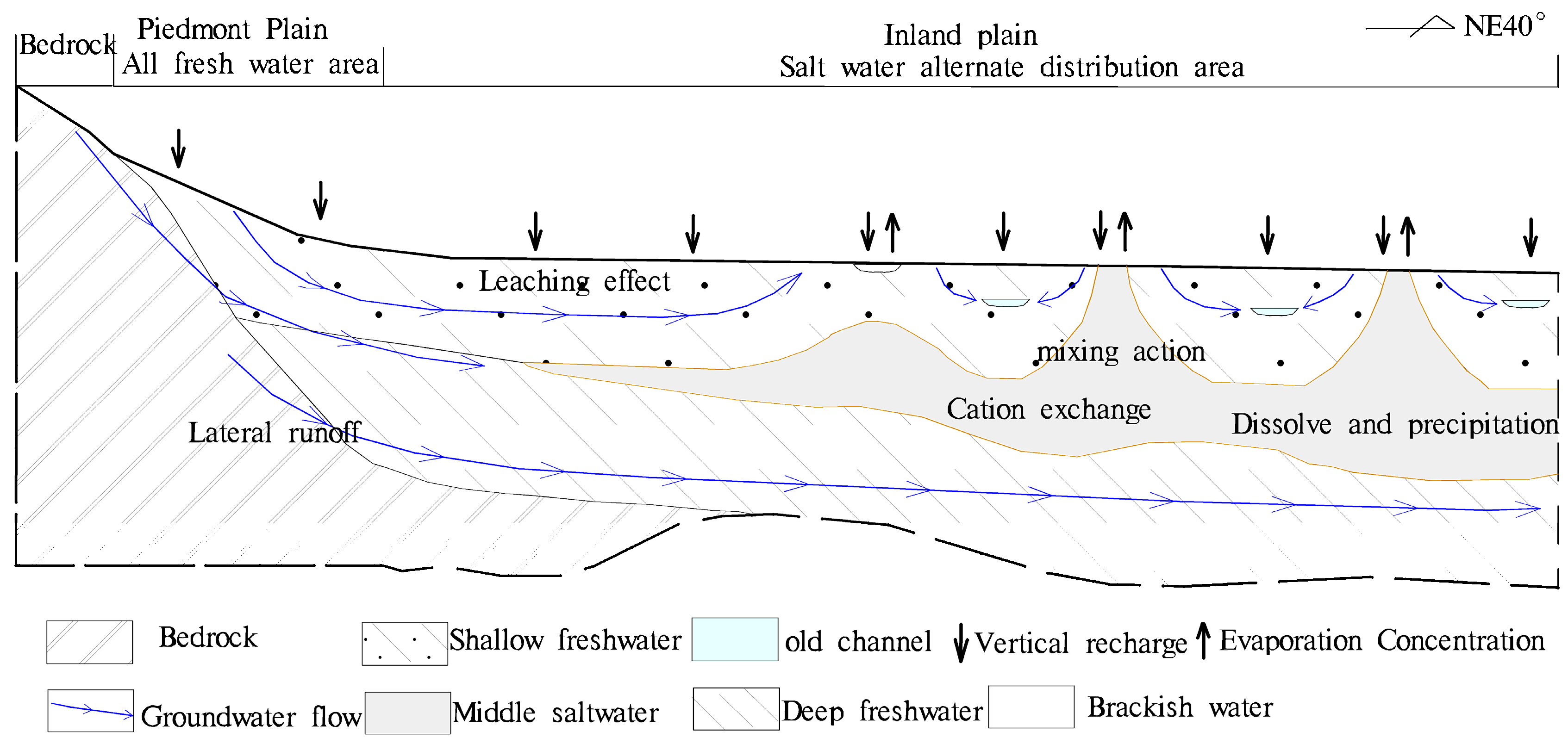
4.3. Inland Plain Groundwater Mechanisms
4.3.1. Groundwater Characteristics
4.3.2. Effect of Clay Retarding Action on Chemical Zoning in Water
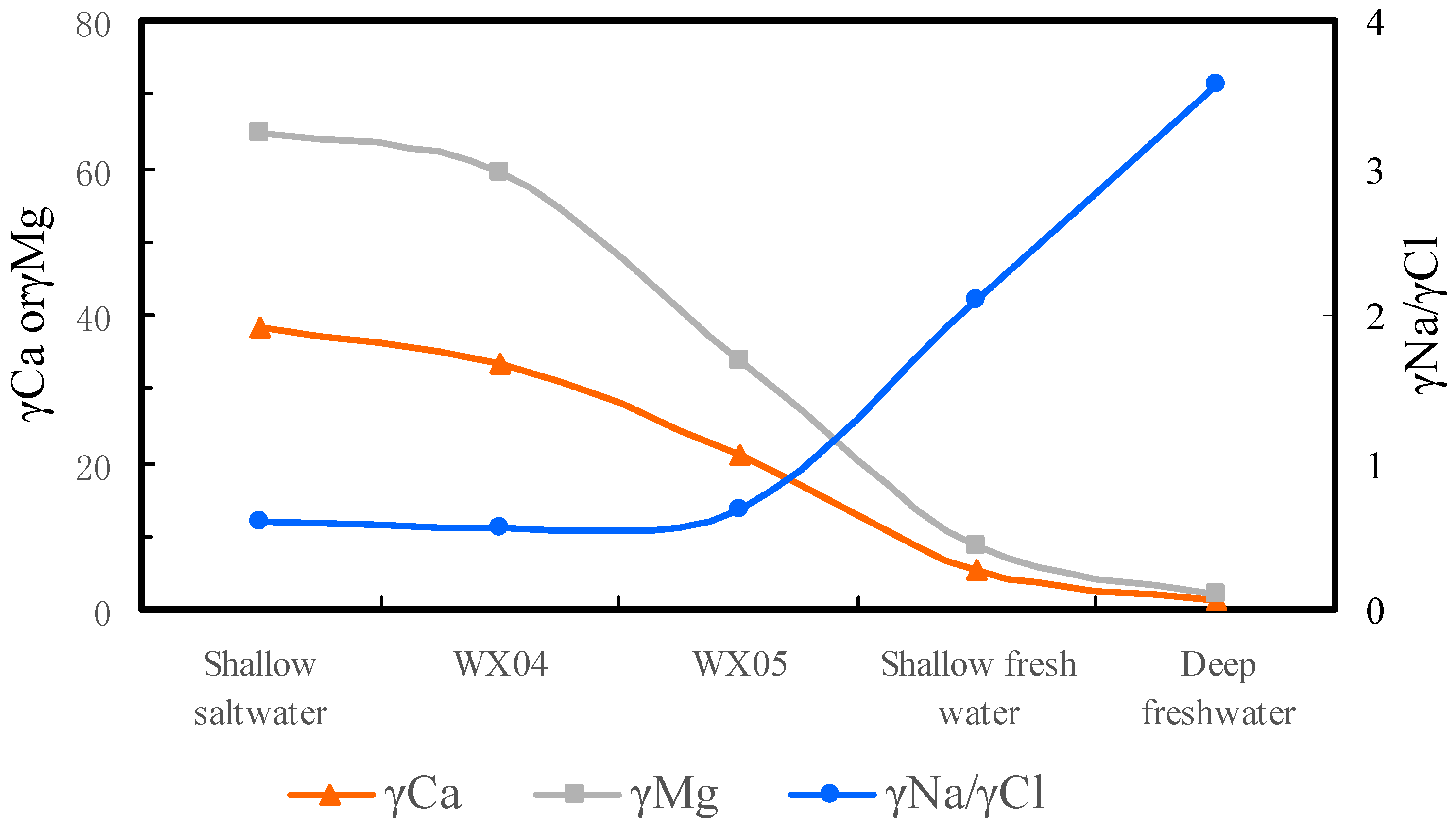
4.3.3. Groundwater Hydrogeochemistry Reflected in the Ion Ratio Coefficient
4.3.4. The Mechanism of Formation of Hydrochemistry
5. Conclusions
- (1)
- The influence of hydrodynamic conditions, sedimentary environment and topography has resulted in a complex groundwater state in the inland plain. TDS increases along the groundwater flow and with the horizontal distribution of freshwater and brackish water (saltwater), showing alternate distribution. Shallow saltwater is distributed in the interfluve between old channels as sporadic or irregular bands.
- (2)
- The optimal isothermal adsorption curves of Na+, Ca2+ and Mg2+ in groundwater over different depths accord with Henry’s equation and Langmuir equation, which is the monolayer chemical adsorption or linear adsorption process. The 6–112 m clay exhibits a large-small-large trend of Na+ and Ca2+ retardation in groundwater. The trend of Mg2+ was small-large-small, and the block strength of Na+ and Mg2+ in groundwater was higher than that of Ca2+, facilitating a Na-Mg or Mg-Na saltwater formation. The correlation between the block strength of clay for Na+, Ca2+ and Mg2+, and groundwater TDS was strong.
- (3)
- Water quality of middle saltwater was relatively stable. The main hydrochemical types were Cl·SO4-Na·Mg and SO4·Cl-Mg·Na, which belong to the intermediate circulation system. The dynamic stability of saltwater quality was affected by the stability of clay distribution, its weak permeability and blocking effect, and the sedimentary environment.
- (4)
- Groundwater in the study area is not only affected by complex hydrogeochemistry, such as the leaching effect, cation exchange and evaporation, it is also closely related to the retardation effect of clay. The inland plains area has horizontal and vertical hydrogeochemical zonation characteristics.
Author Contributions
Funding
Conflicts of Interest
References
- Mehta, S.; Fryar, A.E.; Brady, R.M.; Morin, R.H. Modeling regional salinization of the ogallala aquifer, Southern High Plains, TX, USA. J. Hydrol. 2000, 238, 44–64. [Google Scholar] [CrossRef]
- Weisbrod, N.; Dragila, M.I. Potential impact of convective fracture venting on salt-crust buildup and ground-water salinization in arid environments. J. Arid Environ. 2006, 65, 386–399. [Google Scholar] [CrossRef]
- Hunter, R.; Day, J.; Shaffer, G.; Lane, R.; Englande, A.; Reimers, R.; Kandalepas, D.; Wood, W.; Day, J.; Hillmann, E. Restoration and management of a degraded baldcypress swamp and freshwater marsh in Coastal Louisiana. Water 2016, 8, 71. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, M.; Li, X.; Qi, J.; Zhang, Q.; Guo, J.; Yu, L.; Zhao, R. Hydrochemical characteristics and multivariate statistical analysis of natural water system: A case study in Kangding County, Southwestern China. Water 2017, 10, 80. [Google Scholar] [CrossRef]
- Dedzo, M.G.; Tsozué, D.; Mimba, M.E.; Teddy, F.; Nembungwe, R.M.; Linida, S. Importance of rocks and their weathering products on groundwater quality in Central-East Cameroon. Hydrology 2017, 4, 23. [Google Scholar] [CrossRef]
- An, L.S.; Zhao, Q.S.; Ye, S.Y.; Liu, G.Q.; Ding, X.G. Hydrochemical characteristics and formation mechanism of shallow groundwater in the Yellow River Delta. Environ. Sci. 2012, 33, 370–378. [Google Scholar]
- Xing, L.; Zhang, F.; Changsuo, L.I.; Yang, L.; Wang, L. Hydro-chemical characteristics of shallow groundwater in the North Shandong Plain. J. Irrig. Drain. 2015, 17, 50–56. [Google Scholar]
- Wang, X.S.; Wan, L.; Jiang, X.W.; Li, H.; Zhou, Y.; Wang, J.; Ji, X. Identifying three-dimensional nested groundwater flow systems in a Tóthian Basin. Adv. Water Resour. 2017, 108, 139–156. [Google Scholar] [CrossRef]
- Li, Q.; Zhou, J.; Zhou, Y.; Bai, C.; Tao, H.; Jia, R.; Ji, Y.; Yang, G. Variation of groundwater hydrochemical characteristics in the plain area of the Tarim Basin, Xinjiang Region, China. Environ. Earth Sci. 2014, 72, 4249–4263. [Google Scholar] [CrossRef]
- Yuan, J.; Xu, F.; Deng, G.; Tang, Y.; Li, P. Hydrogeochemistry of shallow groundwater in a karst aquifer system of Bijie City, Guizhou Province. Water 2017, 9, 625. [Google Scholar] [CrossRef]
- Najib, S.; Fadili, A.; Mehdi, K.; Riss, J.; Makan, A. Contribution of hydrochemical and geoelectrical approaches to investigate salinization process and seawater intrusion in the coastal aquifers of Chaouia, Morocco. J. Contam. Hydrol. 2017, 198, 24–36. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.; Jean, J.S.; Chakraborty, S.; Lee, M.K.; Kar, S.; Yang, H.J.; Li, Z. Hydrogeochemistry of groundwater and arsenic adsorption characteristics of subsurface sediments in an Alluvial Plain, SW Taiwan. Sustainability 2016, 8, 1305. [Google Scholar] [CrossRef]
- Zhang, W.; Cao, Y.; Jiang, J.; Wang, W.; Duan, L. Numerical simulation of the formation of groundwater chemical components in the Mayukou area, Datong, China. In Proceedings of the International Symposium on Water-Rock Interaction, Kunming, China, 31 July–5 August 2007. [Google Scholar]
- Jilali, A.; Fagel, N.; Amar, M.; Abbas, M.; Zarhloule, Y. Hydrogeochemical processes constrained by multivariate statistical methods and isotopic evidence of groundwater recharge in the aquifer of Figuig, Eastern High Atlas of Morocco. Arab. J. Geosci. 2016, 9, 1–11. [Google Scholar] [CrossRef]
- Cao, W.; Yang, H.; Liu, C.; Li, Y.; Bai, H. Hydrogeochemical characteristics and evolution of the aquifer systems of Gonghe Basin, Northern China. Geosci. Front. 2018, 9, 907–916. [Google Scholar] [CrossRef]
- Awaleh, M.O.; Baudron, P.; Soubaneh, Y.D.; Boschetti, T.; Hoch, F.B.; Egueh, N.M.; Mohamed, J.; Dabar, O.A.; Masse-Dufresne, J.; Gassani, J. Recharge, groundwater flow pattern and contamination processes in an arid volcanic area: Insights from isotopic and geochemical tracers (bara aquifer system, Republic of Djibouti). J. Geochem. Explor. 2017, 175, 82–98. [Google Scholar] [CrossRef]
- Swaid, F.A.; Issa, A.A. Deep groundwater quality and its suitability for drinking and agricultural purposes, Wadibaye, Middle Region, Libya. In Sustainable Irrigation and Drainage V: Management, Technologies and Policies; WIT Press: Southampton, UK, 2014; pp. 183–196. [Google Scholar]
- Zhihong, L.I.; Fusheng, H.U.; Zhou, W.; Zhao, Z.; Wang, G.; Shi, Z. Hydrogeochemical characteristics and controlling factors of confined water in Yinchuan. Hydrogeol. Eng. Geol. 2017, 44, 31–39. [Google Scholar]
- Mustafa, O.; Merkel, B.; Weise, S. Assessment of hydrogeochemistry and environmental isotopes in karst springs of makook anticline, Kurdistan Region, Iraq. Hydrology 2015, 2, 48–68. [Google Scholar] [CrossRef]
- Sun, Y.Q.; Qian, H.; Wu, X.-H. Hydrogeochemical modeling of minerals in groundwater in Yinchuan Region. J. Saf. Environ. 2006, 6, 32. [Google Scholar]
- Banaś, K. The hydrochemistry of peatland lakes as a result of the morphological characteristics of their basins. Oceanol. Hydrobiol. Stud. 2013, 42, 28–39. [Google Scholar] [CrossRef]
- Kolpakova, M.N.; Naymushina, O.S. Zonal features of bog and lake water chemistry along a transect from boreal to arid landscapes in the south of Western Siberia, Russia. IOP Conf. Ser. Earth Environ. Sci. 2016, 44, 022025. [Google Scholar] [CrossRef]
- Chen, Q.; Mao, Y.; Cheng, L.I. Hydrochemistry characteristics of shallow groundwater in redbed of Luzhou, Sichuan. China Meas. Test 2013, 39, 42–45. [Google Scholar]
- Li, X.; Wu, H.; Qian, H.; Gao, Y. Groundwater chemistry regulated by hydrochemical processes and geological structures: A case study in Tongchuan, China. Water 2018, 10, 338. [Google Scholar] [CrossRef]
- Jiang, X.W.; Wan, L.; Wang, X.S.; Li, H.-L. Distribution of groundwater age in drainage basins. Hydrogeol. Eng. Geol. 2012, 4, 003. [Google Scholar]
- Feng, X.; Zhang, Y.Z. An analysis of the ions ratio coefficients of groundwater in Shenzhou area. China Rural Water Hydropower 2014, 4, 18–21. [Google Scholar]
- Zhang, G.X.; Wei, D.; Yan, H.E.; Salama, R. Hydrochemical characteristics and evolution laws of groundwater in Songnen Plain, Northeast China. Adv. Water Sci. 2006, 17, 20–28. [Google Scholar]
- Xing, W.; Ma, R.; Sun, Z.; Hu, Y.; Ge, M.; Chang, Q. Hydrochemical characteristics and water quality assessment of groundwater in the Dunhuang Basin, Northwestern China. Geol. Sci. Technol. Inf. 2016, 35, 196–202. [Google Scholar]
- Loni, O.A.; Zaidi, F.K.; Alhumimidi, M.S.; Alharbi, O.A.; Hussein, M.T.; Dafalla, M.; Alyousef, K.A.; Kassem, O.M.K. Evaluation of groundwater quality in an evaporation dominant arid environment; A case study from al asyah area in Saudi Arabia. Arab. J. Geosci. 2015, 8, 6237–6247. [Google Scholar] [CrossRef]
- Nakagawa, K.; Amano, H.; Kawamura, A.; Berndtsson, R. Classification of groundwater chemistry in Shimabara, using self-organizing maps. Hydrol. Res. 2017, 48, 840–850. [Google Scholar] [CrossRef]
- Sherwood, J.D.; Craster, B. Transport of water and ions through a clay membrane. J. Colloid Interface Sci. 2000, 230, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Yamei, L.I.; Cheng, J.; Minmin, L.I.; Cui, L. Experimental study on migration and transformation in cohesive soil of salt water. Water Resour. Prot. 2015, 31, 88–93. [Google Scholar]
- Ishiguro, M.; Matsuura, T.; Detellier, C. Reverse osmosis separation for a montmorillonite membrane. J. Membr. Sci. 1995, 107, 87–92. [Google Scholar] [CrossRef]
- Saber, M.; Abdelshafy, M.; Faragallah, E.A.A.; Abd-Alla, M.H. Hydrochemical and bacteriological analyses of groundwater and its suitability for drinking and agricultural uses at Manfalut District, Assuit, Egypt. Arab. J. Geosci. 2014, 7, 4593–4613. [Google Scholar] [CrossRef]
- Yin, J.; Deng, C.; Yu, Z.; Wang, X.; Xu, G. Effective removal of lead ions from aqueous solution using nano illite/smectite clay: Isotherm, kinetic, and thermodynamic modeling of adsorption. Water 2018, 10, 210. [Google Scholar] [CrossRef]
- Zhang, Y.; Fu, C.; Mao, L.; Gong, X.L.; Li, X.-Q. Hydrochemical characteristics and formation mechanism of the groundwater in Yancheng, Jiangsu Province. Resour. Environ. Yangtze Basin 2017, 26, 598–605. [Google Scholar]
- Xu, W.; Zhang, J.; Liu, Y. Degradation of organic matter and ammonia nitrogen in constructed rapid infiltration system. Fresenius Environ. Bull. 2011, 20, 1685–1690. [Google Scholar]
- Maleki, S.; Karimi-Jashni, A. Effect of ball milling process on the structure of local clay and its adsorption performance for Ni(II) removal. Appl. Clay Sci. 2017, 137, 213–224. [Google Scholar] [CrossRef]
- Yechieli, Y.; Wood, W.W. Hydrogeologic processes in saline systems: Playas, sabkhas, and saline lakes. Earth Sci. Rev. 2002, 58, 343–365. [Google Scholar] [CrossRef]
- Yang, Y.; Xing, L.T.; Zhang, W.J.; Xiang, H.; Song, G.Z.; Chi, G.Y.; Hou, X.Y. The retardation of clay in shallow groundwater system of inland plains. Sci. Technol. Eng. 2017, 17, 50–56. [Google Scholar]
- Huang, L.; Wang, L.; Zhang, Y.; Xing, L.; Hao, Q.; Xiao, Y.; Yang, L.; Zhu, H. Identification of groundwater pollution sources by a SCE-UA algorithm-based simulation/optimization model. Water 2018, 10, 193. [Google Scholar] [CrossRef]
- Hui, B.; Ping, H. Experimental study of water and salt redistribution of clay soil in an opening system with constant temperature. Environ. Geol. 2008, 55, 717–721. [Google Scholar]
- Zhang, Z.; Qin, Y.; Wang, G.X.; Fu, X.H. Numerical description of coalbed methane desorption stages based on isothermal adsorption experiment. Sci. China Earth Sci. 2013, 56, 1029–1036. [Google Scholar] [CrossRef]
- Vimonses, V.; Lei, S.; Bo, J.; Chow, C.W.K.; Saint, C. Kinetic study and equilibrium isotherm analysis of Congo red adsorption by clay materials. Chem. Eng. J. 2009, 148, 354–364. [Google Scholar] [CrossRef]
- Nelson, C.; Mildenhall, D.; Todd, A.; Pocknall, D. Subsurface stratigraphy, paleoenvironments, palynology, and depositional history of the late neogene tauranga group at ohinewai, lower waikato lowland, South Auckland, New Zealand. N. Z. J. Geol. Geophys. 1988, 31, 21–40. [Google Scholar] [CrossRef]
- Sugita, F.; Gillham, R.W. Pore scale variation in retardation factor as a cause of nonideal reactive breakthrough curves: 3. Column investigations. Water Resour. Res. 1995, 31, 121–128. [Google Scholar] [CrossRef]
- Zhang, Z.; Shi, D.; Ren, F.; Yin, Z.; Sun, J.; Zhang, C. Evolution of quaternary groundwater system in North China plain. Sci. China 1997, 40, 276–283. [Google Scholar] [CrossRef]
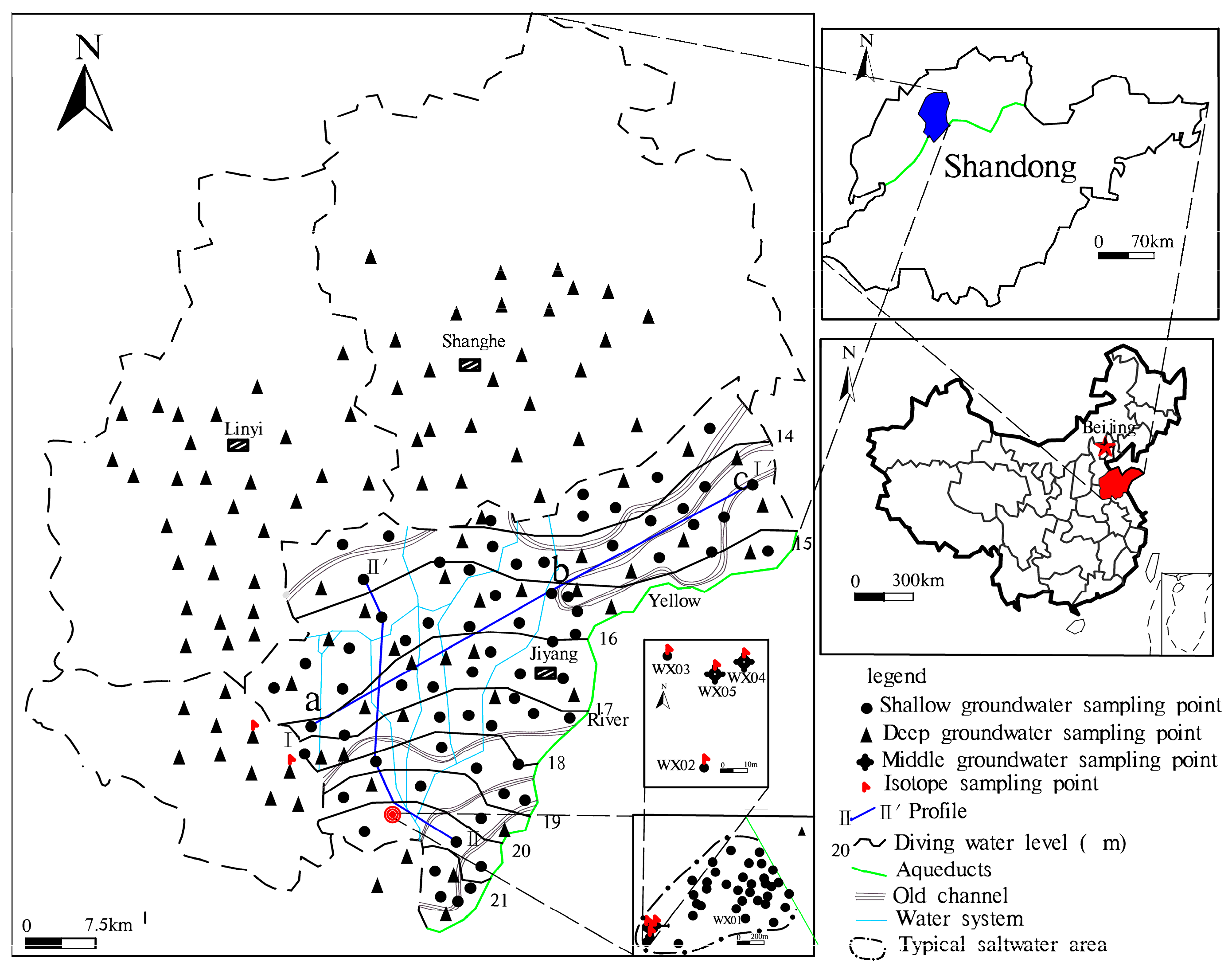
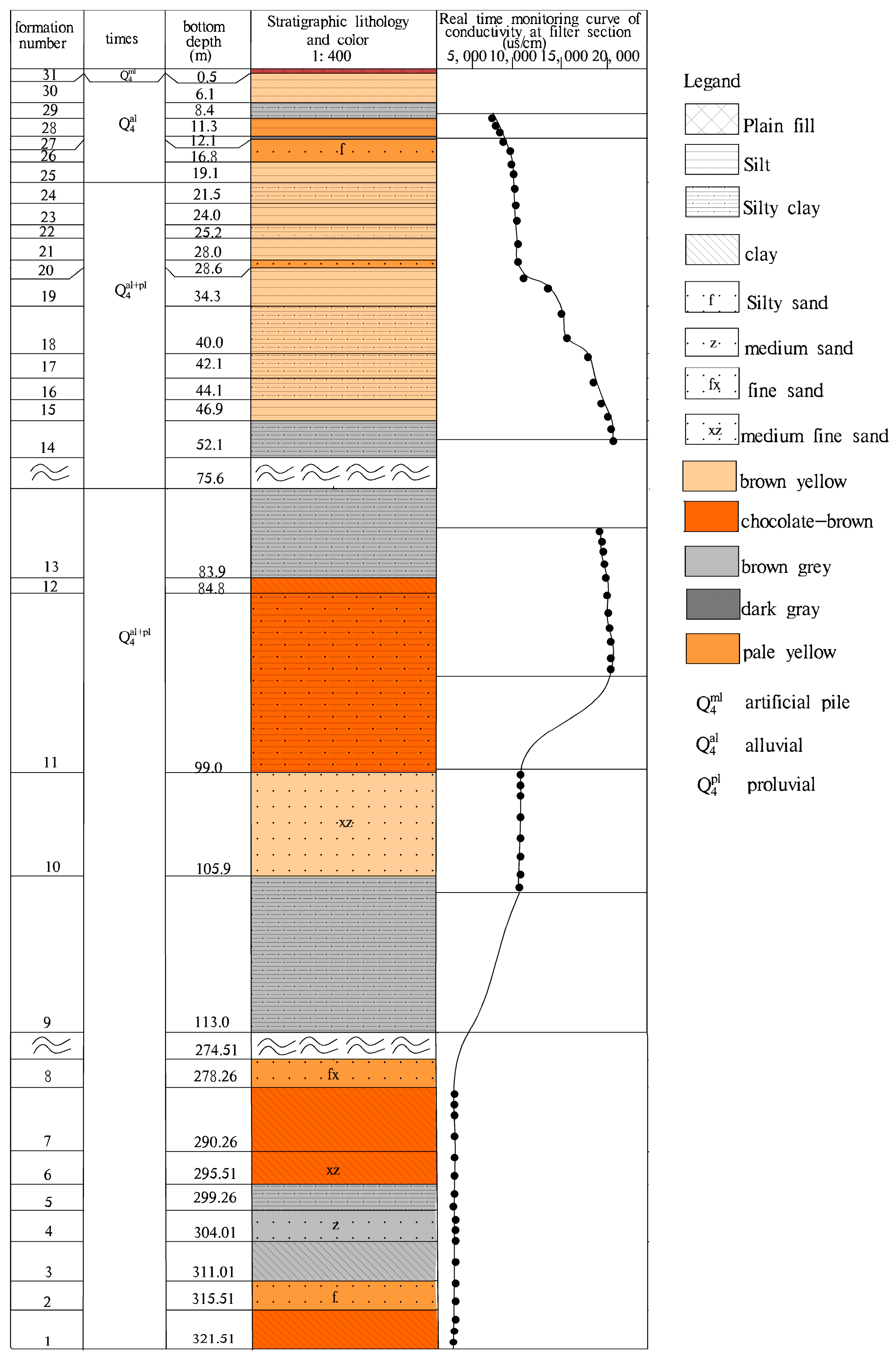
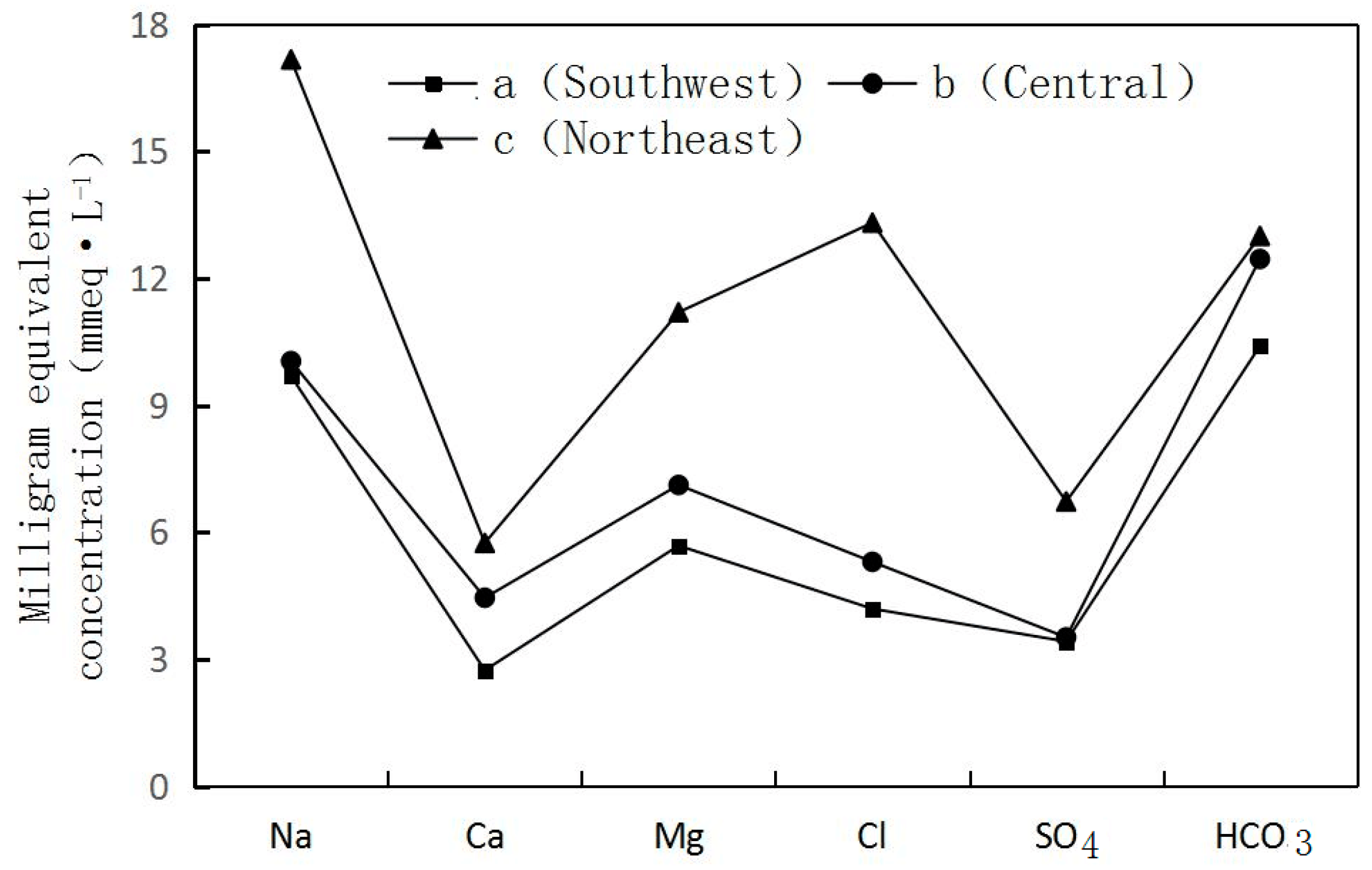
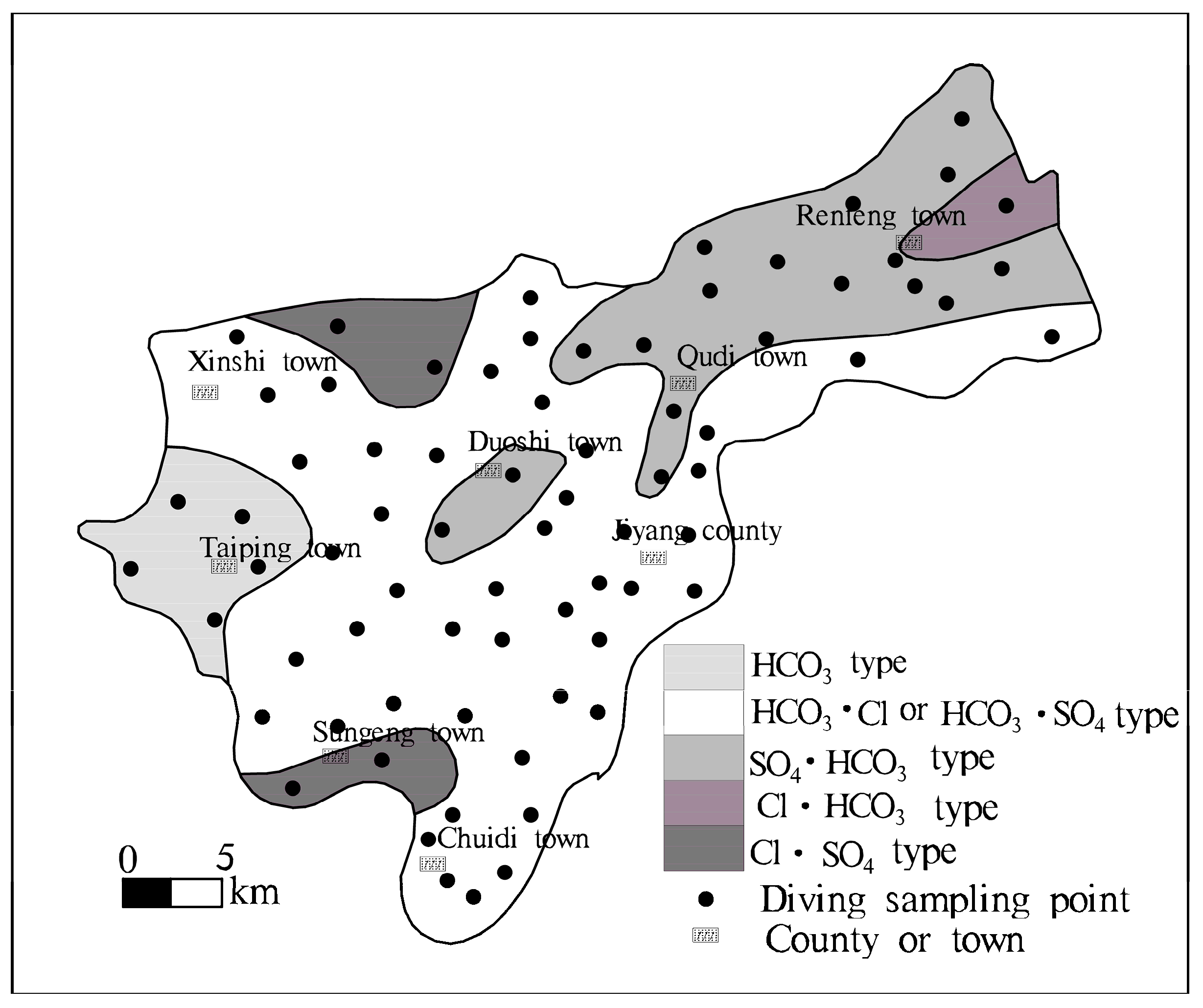

| Well Number | Well Diameter/mm | Well Depth/m | Filter Tube Depth/m | Groundwater Level/m | Aquifer Lithology |
|---|---|---|---|---|---|
| WX01 | 146 | 8 | 3–8 | 23.59 | Silty clay |
| WX02 | 146 | 12 | 8–12 | 21.03 | Silt |
| WX03 | 235 | 50 | 13–50 | 20.99 | Silty clay |
| WX04 | 140 | 91 | 78–90 | 19.16 | Silty clay |
| WX05 | 140 | 113 | 99–108 | 18.26 | silty sand |
| WX06 | 180 | 336 | 274–333 | 24.00 | Fine sand, fine sand, medium sand |
| Index | Na+ | Ca2+ | Mg2+ | HCO3− | SO42− | Cl− | TDS | |
|---|---|---|---|---|---|---|---|---|
| Freshwater (n = 50) | Maximum | 384.00 | 198.36 | 127.81 | 1009.28 | 460.45 | 358.99 | 1985.00 |
| Minimum | 61.40 | 50.74 | 46.48 | 58.56 | 4.00 | 23.98 | 549.89 | |
| Average | 167.95 | 95.39 | 76.90 | 580.06 | 174.85 | 159.07 | 1254.23 | |
| Coefficient of variation (%) | 46.16 | 29.89 | 33.07 | 33.35 | 57.37 | 55.15 | 46.16 | |
| Brackish water (n = 15) | Maximum | 688.45 | 342.62 | 226.69 | 1170.51 | 1165.65 | 907.13 | 3973.94 |
| Minimum | 223.50 | 70.14 | 101.1 | 637.65 | 178.04 | 167.92 | 2014.23 | |
| Average | 469.83 | 151.90 | 161.63 | 898.33 | 548.04 | 503.83 | 2733.57 | |
| Coefficient of variation (%) | 35.98 | 48.99 | 28.25 | 20.67 | 51.30 | 48.64 | 23.54 | |
| Saltwater (n = 5) | Maximum | 2223.69 | 646.68 | 310.74 | 540.08 | 6783.09 | 2738.77 | 13,482.32 |
| Minimum | 1244.00 | 155.32 | 470.96 | 1292.75 | 1470.89 | 1600.00 | 6255.17 | |
| Average | 1582.56 | 360.53 | 603.57 | 3191.94 | 3497.99 | 1177.33 | 9065.82 | |
| Coefficient of variation (%) | 20.40 | 32.24 | 16.64 | 37.46 | 19.04 | 37.80 | 23.45 | |
| Index | Na+ | Ca2+ | Mg2+ | HCO3− | SO42− | Cl− | PH | TDS | |
|---|---|---|---|---|---|---|---|---|---|
| Shallow salt water (n = 20) | Maximum | 4201.20 | 1062.51 | 1022.00 | 1183.8 | 6494.00 | 5321.24 | 7.11 | 16,984.20 |
| Minimum | 1094.24 | 264.47 | 283.21 | 645.54 | 1877.40 | 1217.00 | 6.99 | 5242.40 | |
| Average | 2029.10 | 798.65 | 791.81 | 886.96 | 3251.69 | 3791.59 | 7.04 | 10,077.96 | |
| Coefficient of variation (%) | 41.82 | 25.67 | 37.79 | 12.28 | 27.32 | 32.98 | 1.09 | 26.05 | |
| Middle confined water (n = 20) | Maximum | 2250.16 | 879.25 | 853.25 | 318.14 | 3266.29 | 3616.50 | 8.16 | 13,482.32 |
| Minimum | 1088.18 | 395.33 | 336.75 | 192.37 | 849.09 | 849.09 | 7.01 | 5572.77 | |
| Average | 2108.01 | 545.05 | 560.93 | 244.71 | 3266.29 | 2865.51 | 7.59 | 8991.46 | |
| Coefficient of variation (%) | 29.32 | 25.35 | 29.83 | 17.92 | 58.11 | 9.09 | 4.87 | 31.04 | |
| Deep freshwater (n = 101) | Maximum | 970.00 | 125.16 | 110.99 | 540.08 | 1274.01 | 1036.32 | \ | 3339.83 |
| Minimum | 11.00 | 5.18 | 1.36 | 186.20 | 16.97 | 21.52 | \ | 372.72 | |
| Average | 345.29 | 27.59 | 23.74 | 429.08 | 205.99 | 247.64 | \ | 1285.15 | |
| Coefficient of variation (%) | 34.59 | 91.56 | 76.67 | 26.15 | 74.87 | 59.03 | \ | 28.21 | |
| Clay Sampling Depth/m | Ion in Groundwater | Langmuir Equation Parameters | Henry Equation Parameters | |||
|---|---|---|---|---|---|---|
| Qm/(mg·kg−1) | KL/(L·mg−1) | R2 | Kd/(L·mg−1) | R2 | ||
| 6–6.2 | Na+ | 4.6 | 0.0011 | 0.9164 | 0.2396 | 0.6311 |
| Ca2+ | 55.87 | 0.0150 | 0.7438 | 0.102 | 0.4001 | |
| Mg2+ | 2.72 | 0.0269 | 0.0394 | 2.9292 | 0.3851 | |
| 48–48.2 | Na+ | 1009.09 | 0.0453 | 0.715 | 0.860 6 | 0.078 3 |
| Ca2+ | 466.67 | 0.0036 | 0.9408 | 0.8339 | 0.8511 | |
| Mg2+ | 600 | 0.0013 | 0.9853 | 0.8451 | 0.9637 | |
| 51–51.2 | Na+ | 23.92 | 0.0013 | 0.7574 | 1.0739 | 0.8907 |
| Ca2+ | 70.77 | 0.0016 | 0.8686 | 0.543 | 0.7074 | |
| Mg2+ | 549.45 | 0.0003 | 0.6605 | 0.8132 | 0.7468 | |
| 71–71.2 | Na+ | 130.21 | 0.0008 | 0.7464 | 0.4855 | 0.8182 |
| Ca2+ | 132.45 | 0.0220 | 0.9229 | 0.6738 | 0.9463 | |
| Mg2+ | 194.55 | 0.0085 | 0.9116 | 0.4338 | 0.7336 | |
| 84–84.2 | Na+ | 9.63 | 0.0025 | 0.2421 | 0.6365 | 0.1848 |
| Ca2+ | 3.57 | 0.0060 | 0.9976 | 0.0262 | 0.826 | |
| Mg2+ | 183.82 | 0.0004 | 0.5574 | 0.6570 | 0.9237 | |
| 97–97.2 | Na+ | 423.73 | 0.0006 | 0.9282 | 0.5368 | 0.9011 |
| Ca2+ | 40.68 | 0.0026 | 0.4554 | 0.1636 | 0.588 | |
| Mg2+ | 182.82 | 0.0008 | 0.8810 | 0.6876 | 0.8955 | |
| 102–102.2 | Na+ | 359.71 | 0.0022 | 0.8267 | 0.4518 | 0.7132 |
| Ca2+ | 22.52 | 0.0079 | 0.6343 | 0.207 | 0.8226 | |
| Mg2+ | 1373.33 | 0.0003 | 0.9836 | 0.4606 | 0.9069 | |
| 112–112.2 | Na+ | 4.16 | 0.0013 | 0.7698 | 1.5095 | 0.9251 |
| Ca2+ | 4.16 | 0.0014 | 0.9031 | 0.2111 | 0.5134 | |
| Mg2+ | 288.18 | 0.0178 | 0.9403 | 0.3774 | 0.8569 | |
| Clay Sampling Depth/m | Rd-Na | Rd-Ca | Rd-Mg | Rd-sum |
|---|---|---|---|---|
| 6 | 1.247 | 1.105 | 4.026 | 6.378 |
| 48 | 2.594 | 2.544 | 2.565 | 7.703 |
| 51 | 2.994 | 2.008 | 2.510 | 7.513 |
| 71 | 1.902 | 2.251 | 1.806 | 5.959 |
| 84 | 2.182 | 1.101 | 2.220 | 5.504 |
| 97 | 1.997 | 1.304 | 2.277 | 5.578 |
| 102 | 1.839 | 1.384 | 1.855 | 5.079 |
| 112 | 3.803 | 1.392 | 1.701 | 6.896 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xing, L.; Huang, L.; Hou, X.; Yang, L.; Chi, G.; Xu, J.; Zhu, H. Groundwater Hydrochemical Zoning in Inland Plains and its Genetic Mechanisms. Water 2018, 10, 752. https://doi.org/10.3390/w10060752
Xing L, Huang L, Hou X, Yang L, Chi G, Xu J, Zhu H. Groundwater Hydrochemical Zoning in Inland Plains and its Genetic Mechanisms. Water. 2018; 10(6):752. https://doi.org/10.3390/w10060752
Chicago/Turabian StyleXing, Liting, Linxian Huang, Xinyu Hou, Lizhi Yang, Guangyao Chi, Junxiang Xu, and Henghua Zhu. 2018. "Groundwater Hydrochemical Zoning in Inland Plains and its Genetic Mechanisms" Water 10, no. 6: 752. https://doi.org/10.3390/w10060752
APA StyleXing, L., Huang, L., Hou, X., Yang, L., Chi, G., Xu, J., & Zhu, H. (2018). Groundwater Hydrochemical Zoning in Inland Plains and its Genetic Mechanisms. Water, 10(6), 752. https://doi.org/10.3390/w10060752




