Water Resources Sustainability of Ulaanbaatar City, Mongolia
Abstract
1. Introduction
2. Methods
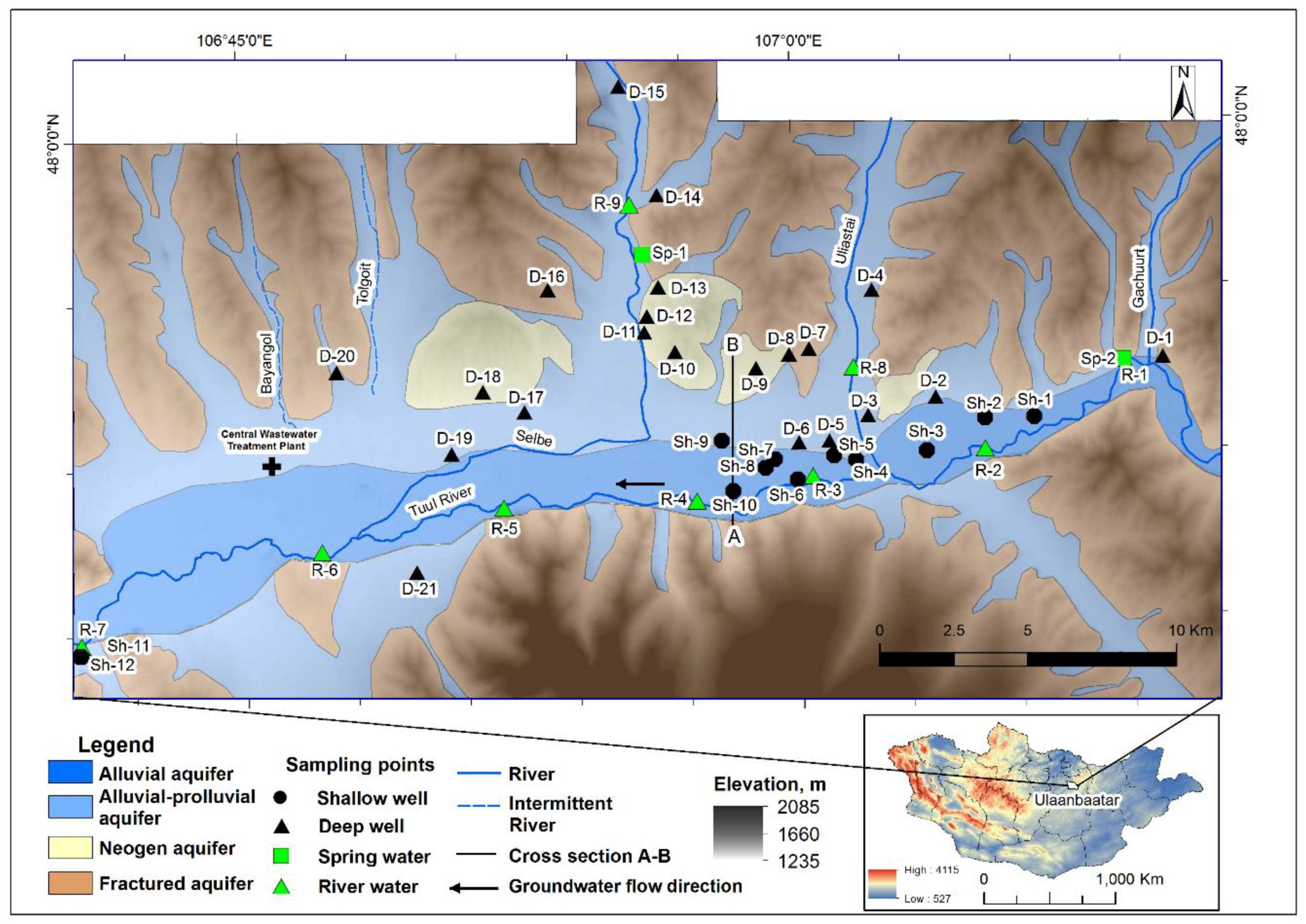
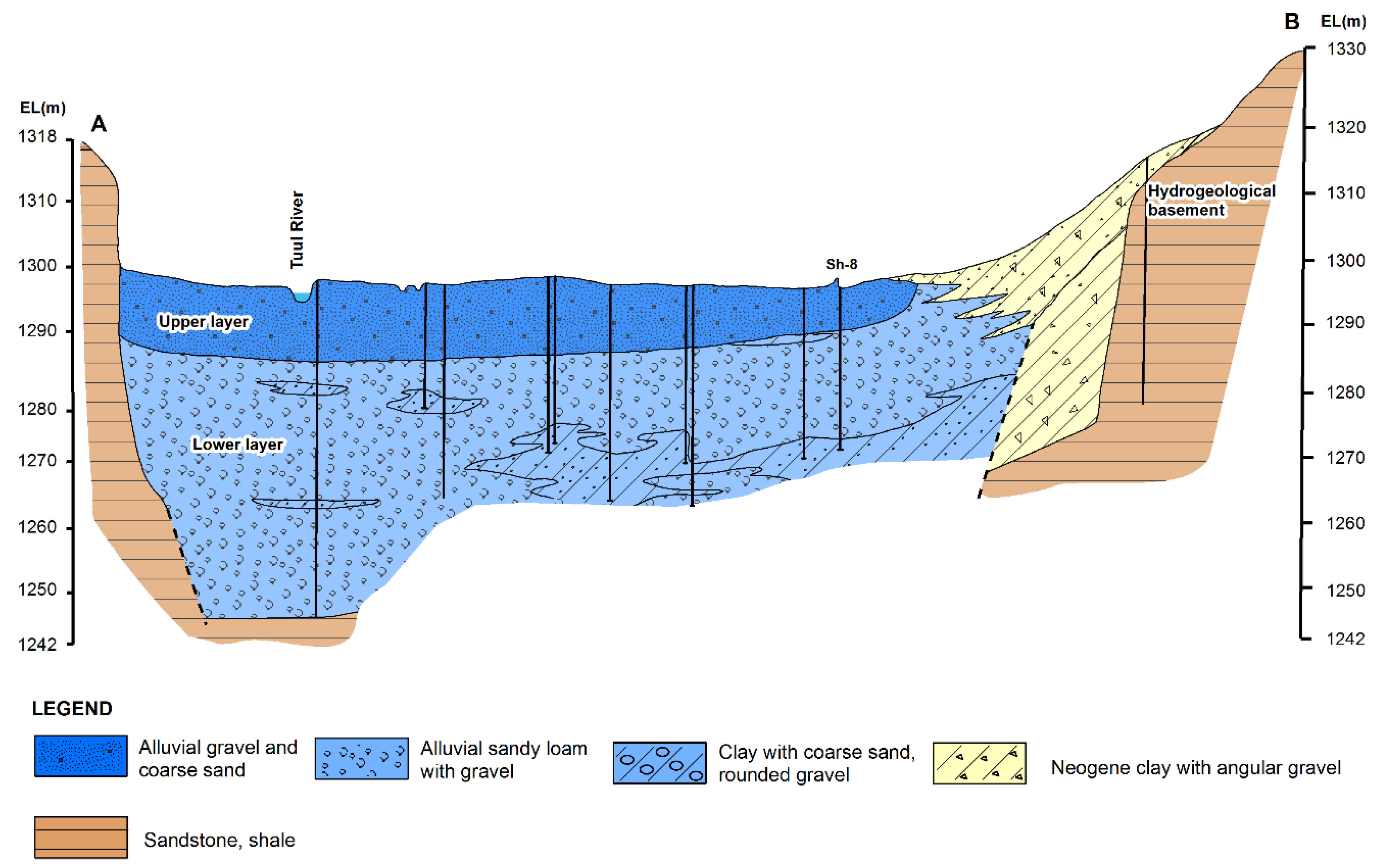
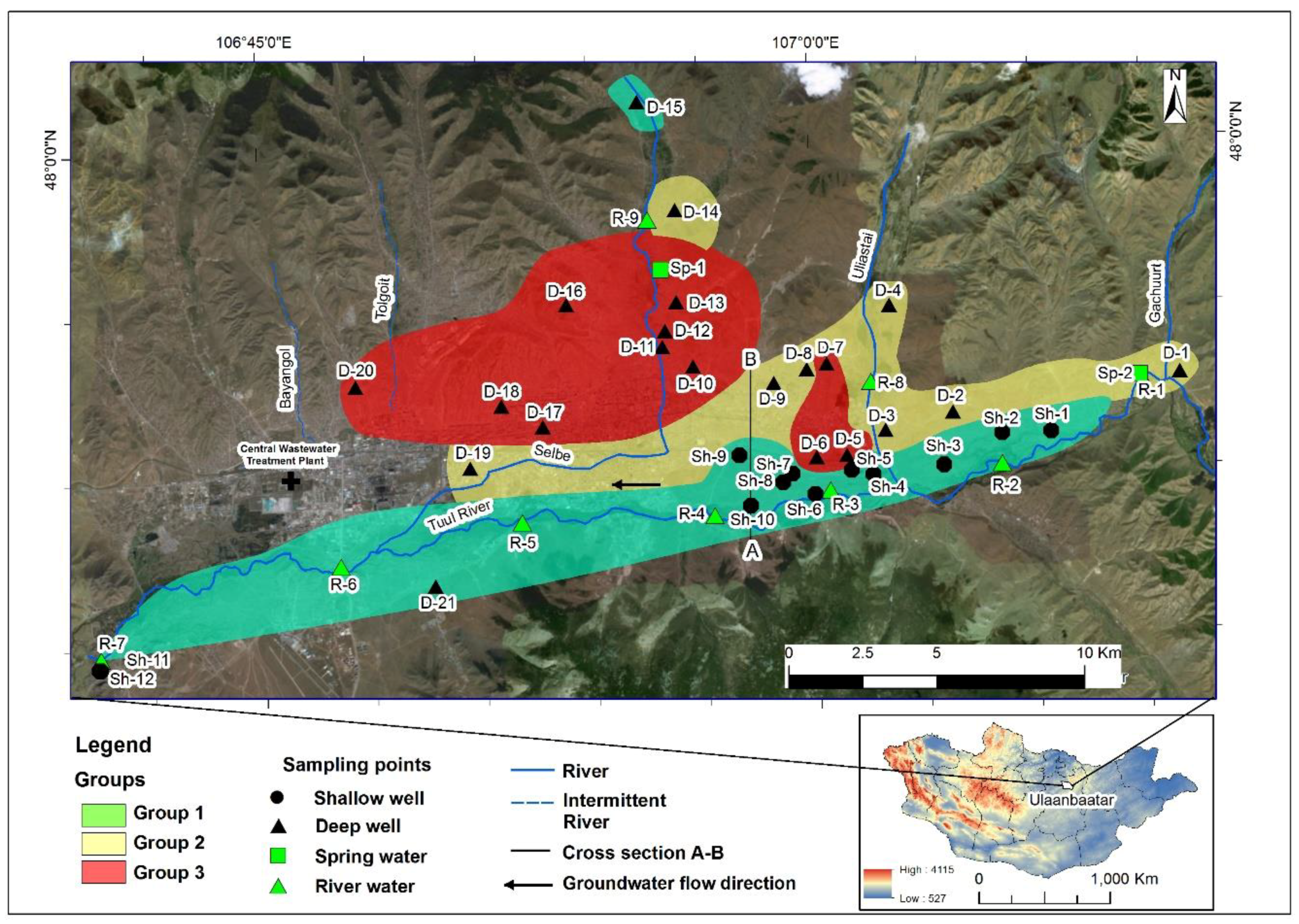
2.1. Study Area
2.2. Water Sample Collection and Analysis
3. Results and Discussion
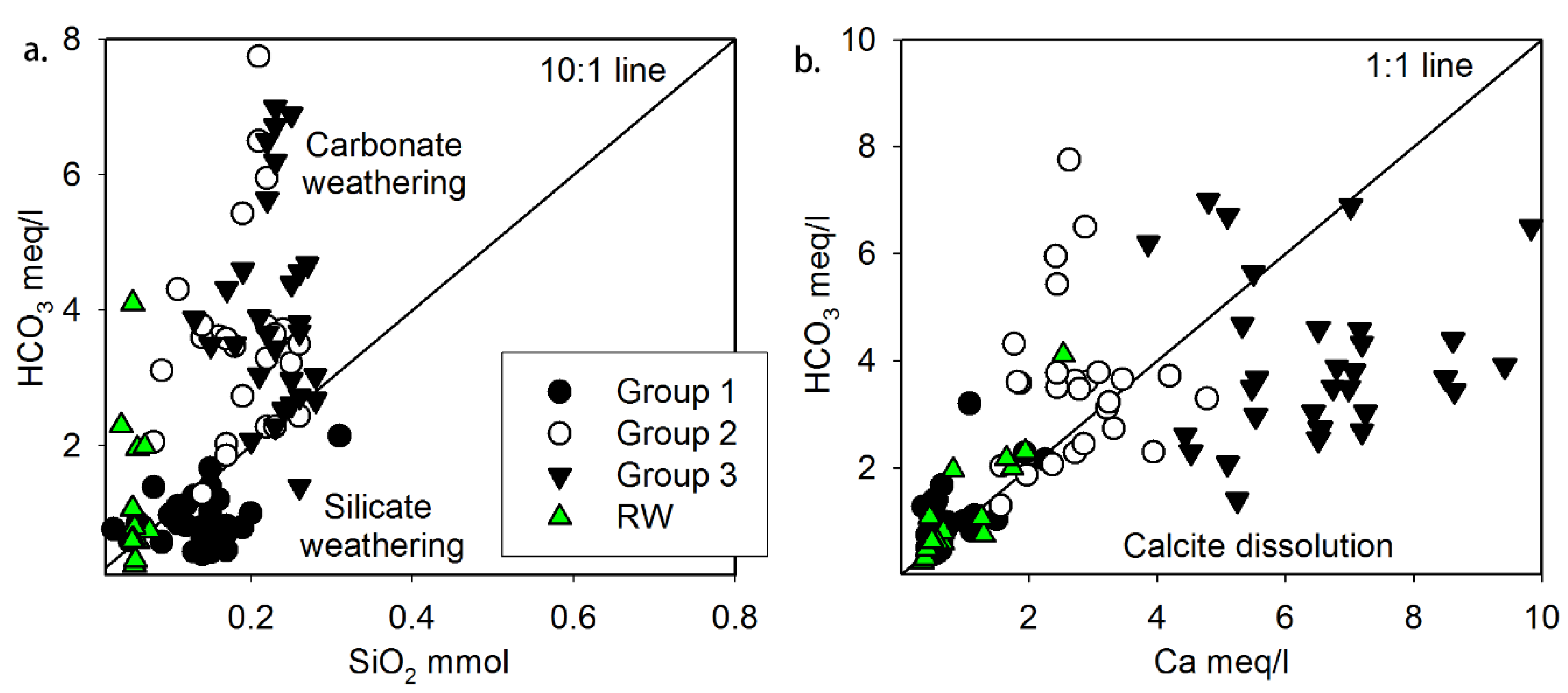
3.1. Hydrochemistry of Water Resources
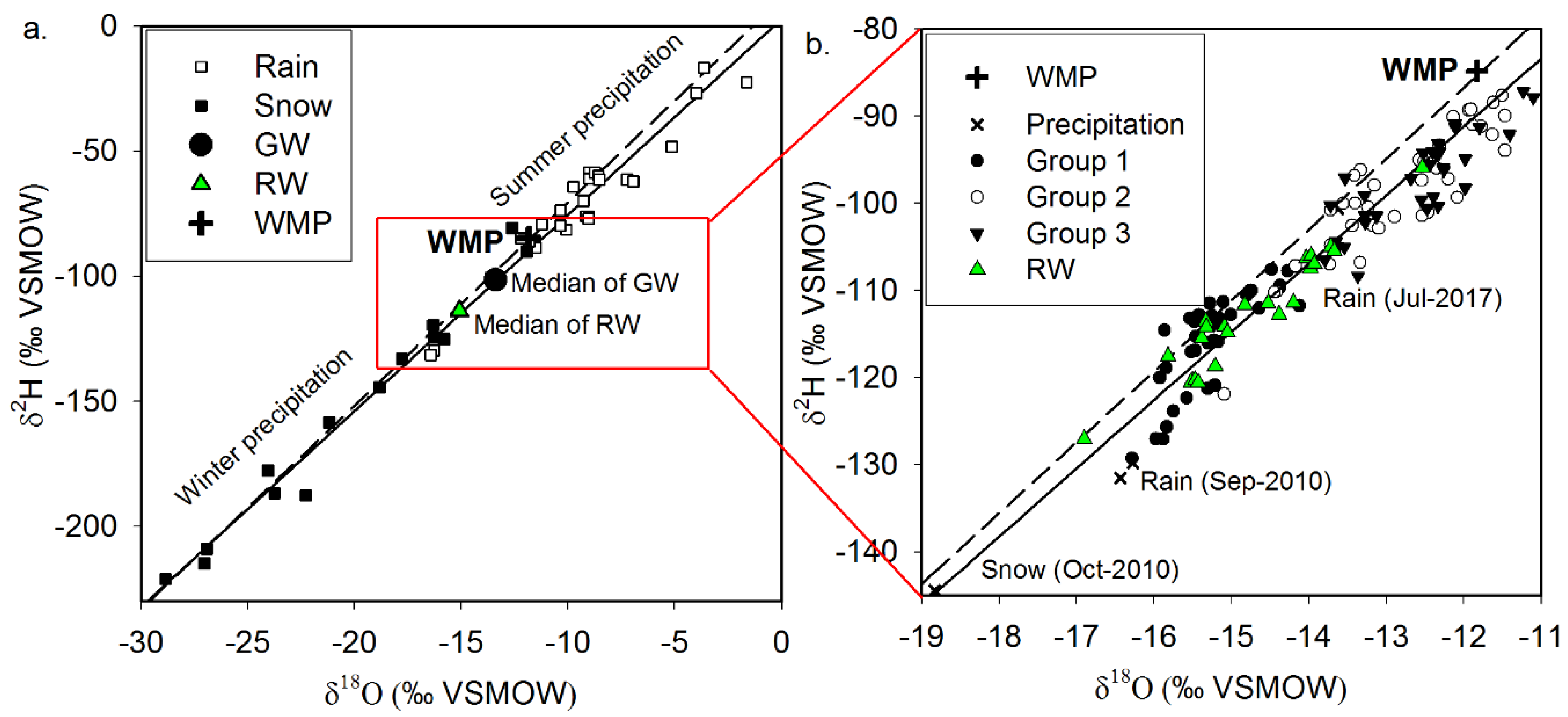
3.2. Isotopic Signatures of Water Resources
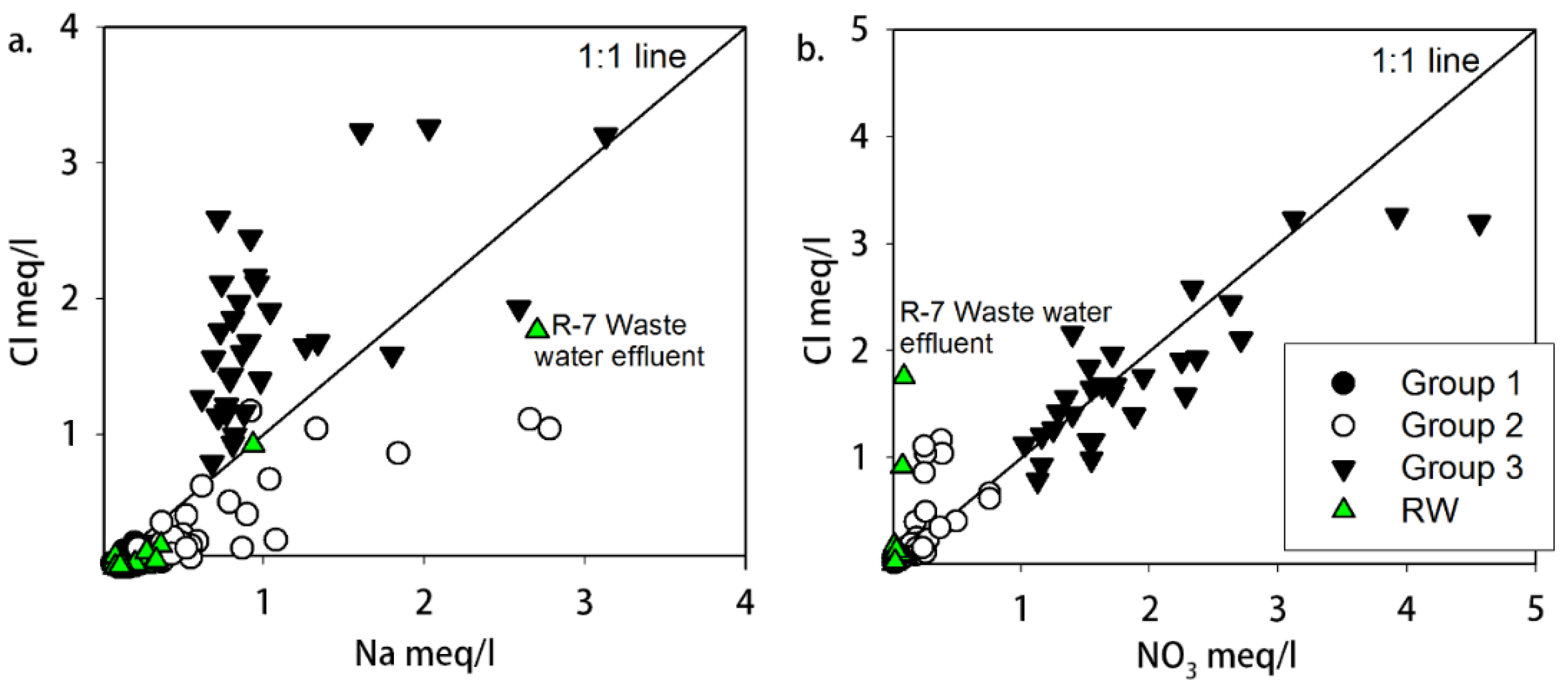
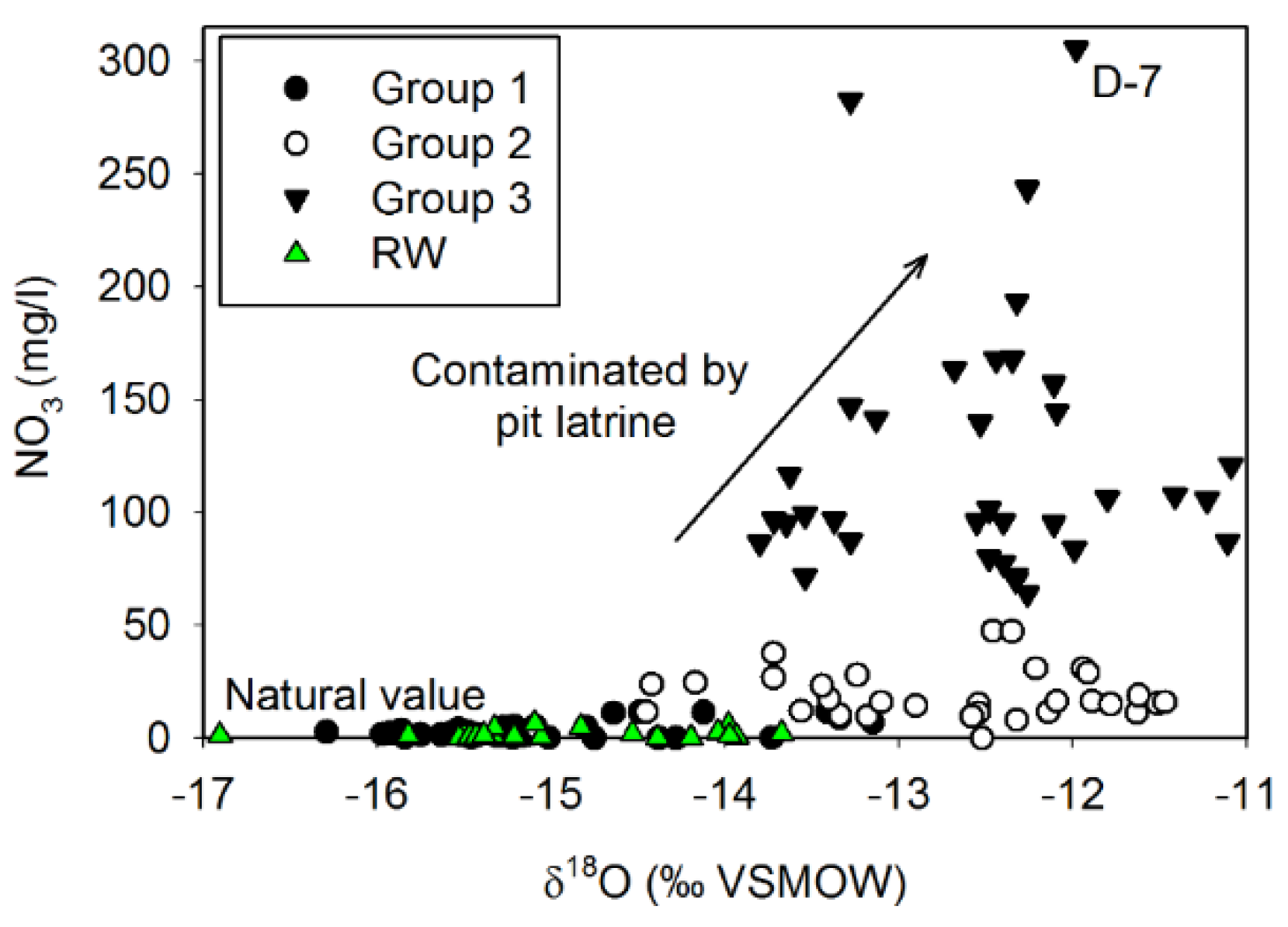
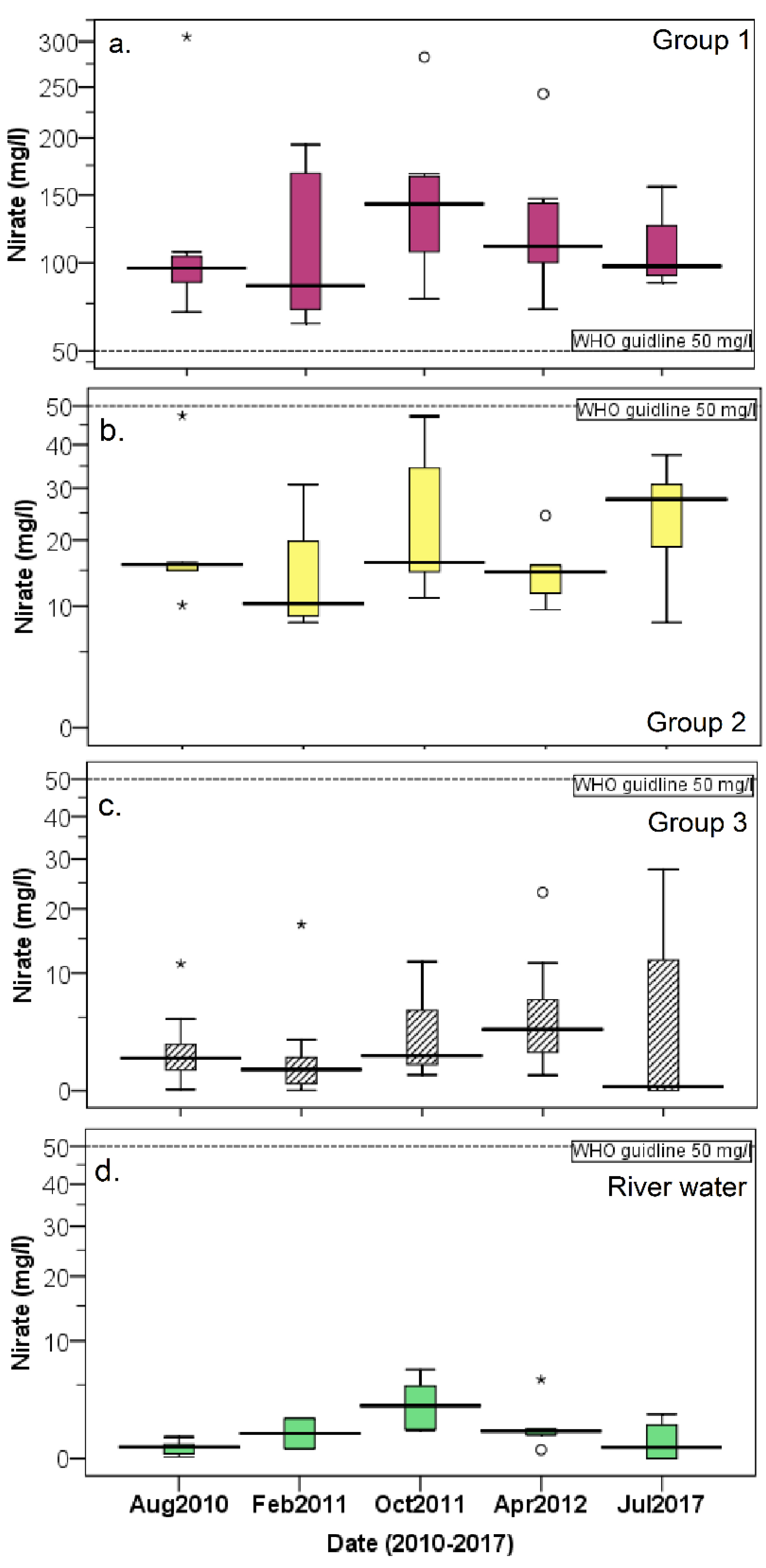
3.3. Nitrate Contamination of Water Resources
4. Conclusions
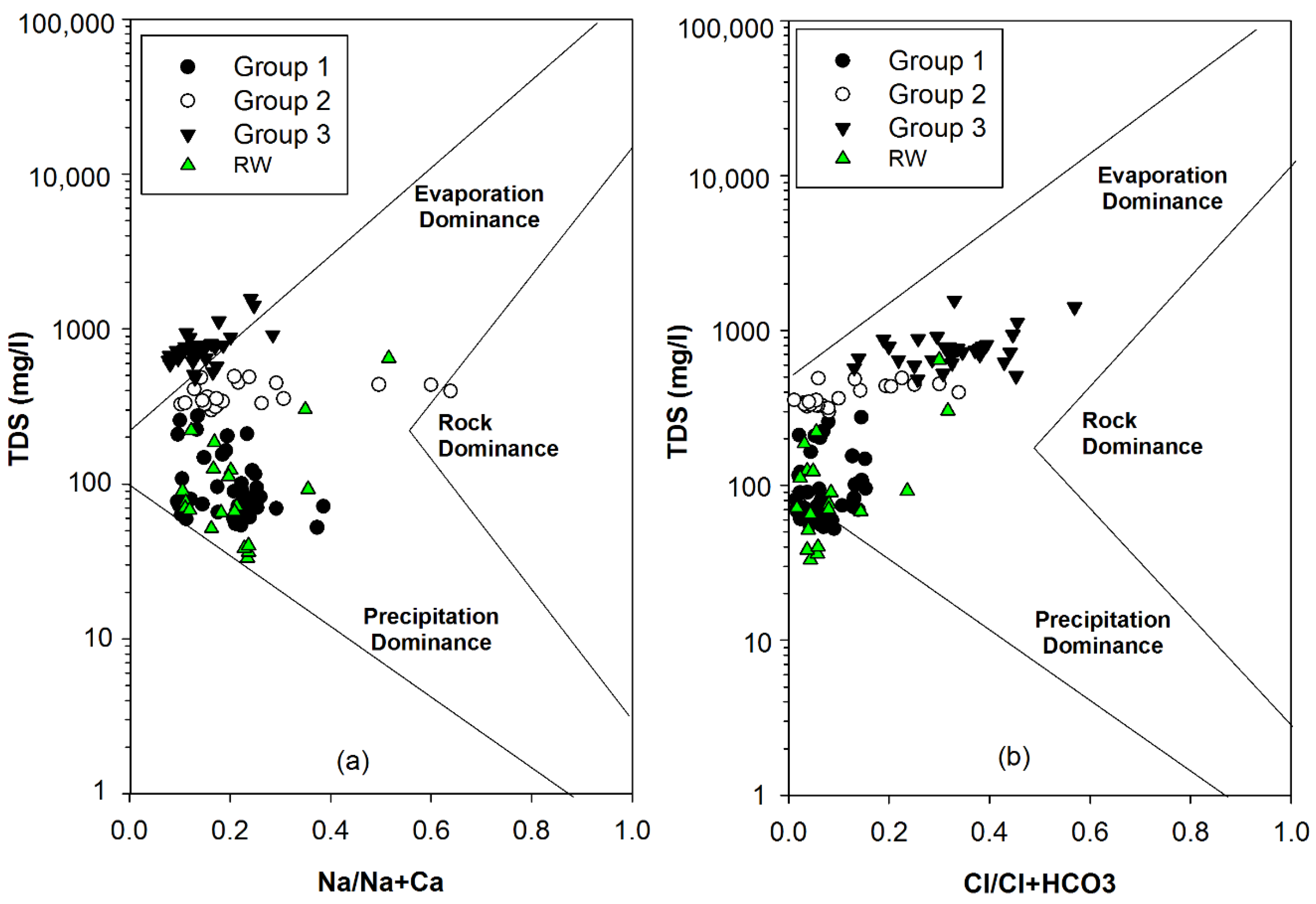
- The hydrochemistry of the water samples from various resources is typically dependent on two major factors: NO3 contamination processes and mineral weathering processes according to water–rock interactions of carbonate and silicate minerals in the geologic matrix.
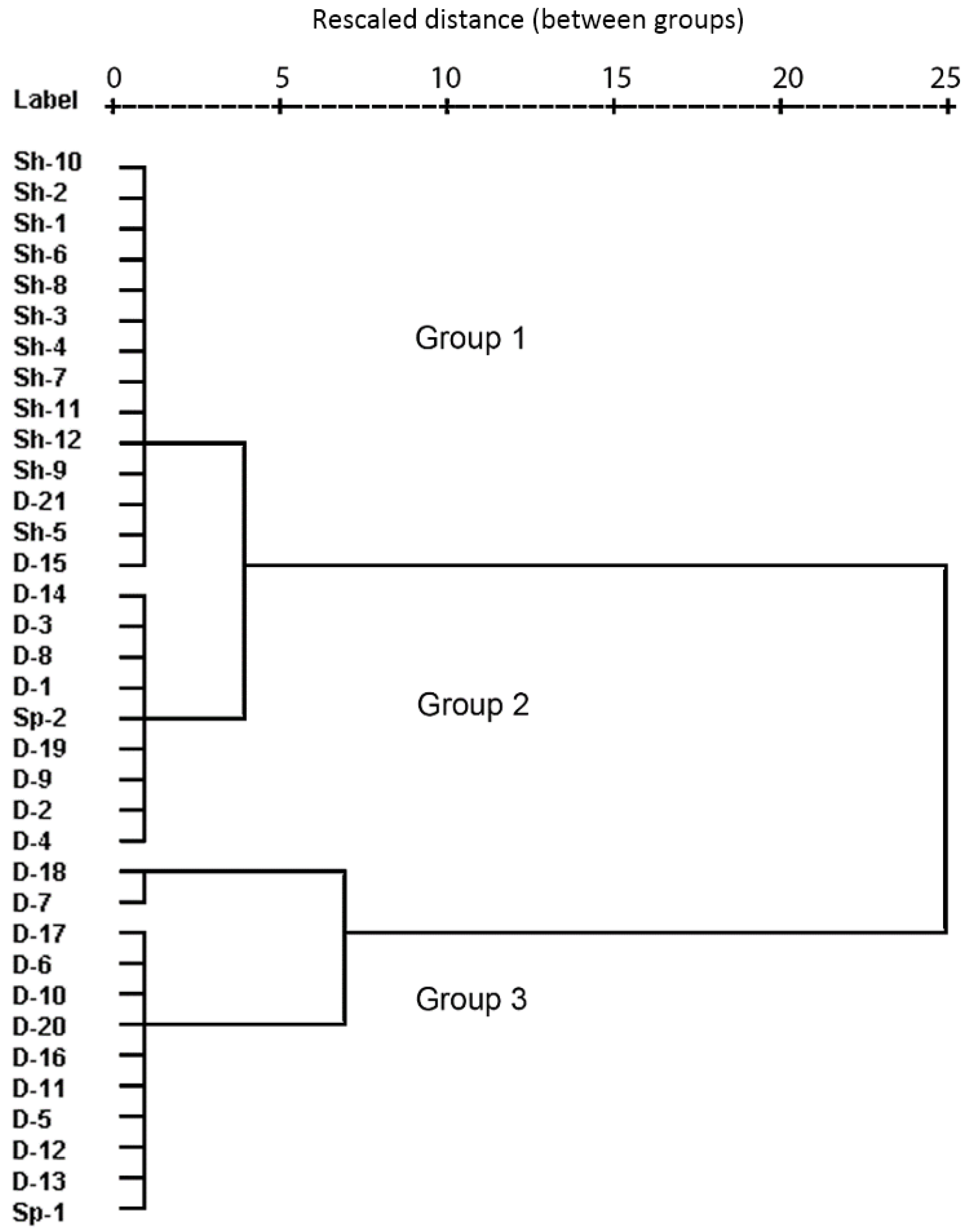
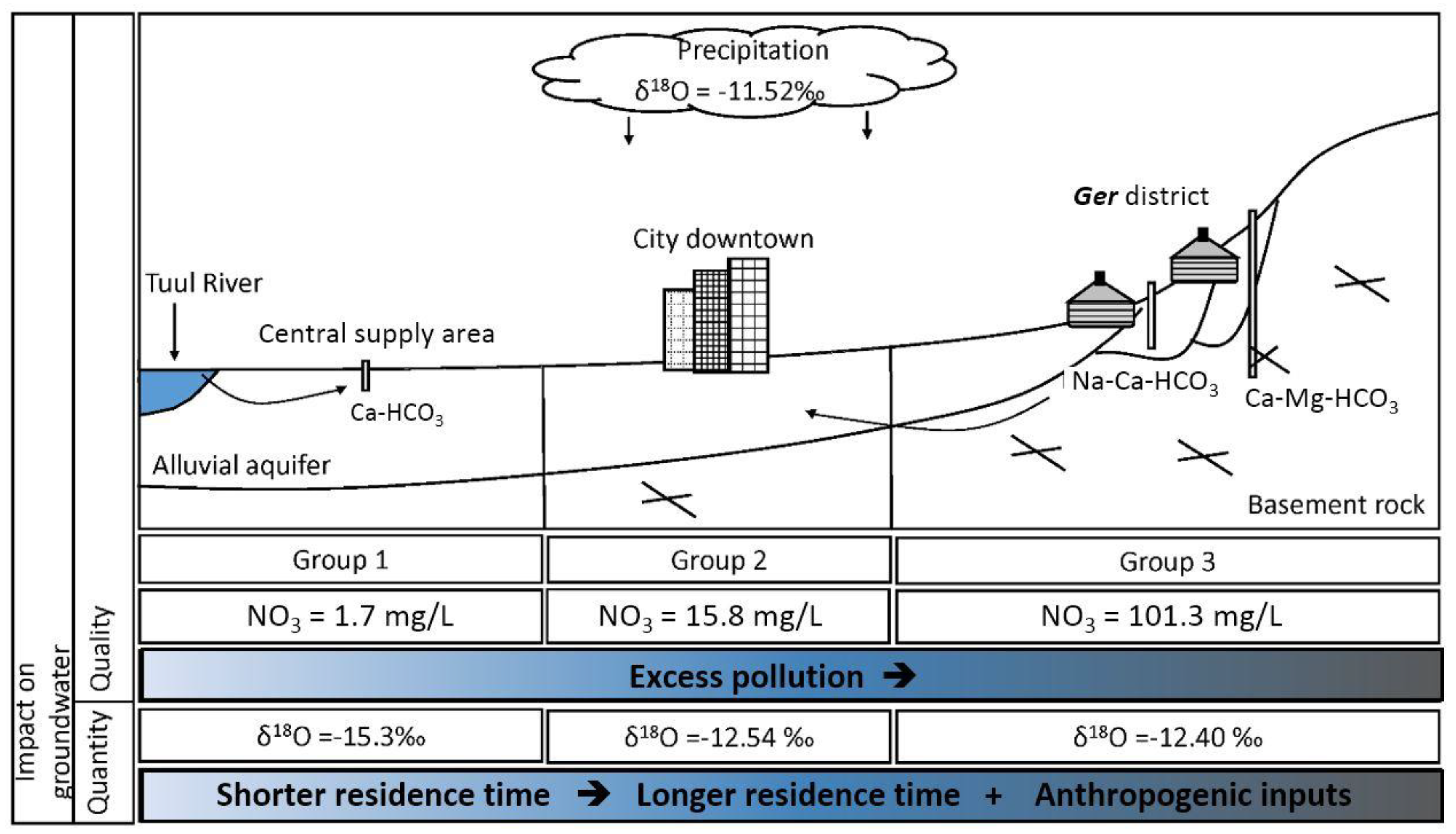
- Based on their chemical composition, the groundwater samples could be classified into three groups. Group 1 included shallow groundwater from the alluvial aquifer distributed along the Tuul River, exhibiting silicate weathering–dominant water–rock interactions with near-natural NO3 concentrations. Group 2 samples, distributed mostly on the north-eastern side of the study area, appeared to reflect carbonate hydrochemical reactions of carbonate weathering, as they had intermediate NO3 levels. Group 3 samples were dominated by CaCO3 dissolution, probably due to freshwater recharge, and had significantly elevated NO3 levels exceeding the World Health Organization drinking water guideline of 50 mg/L.
- The correlation of NO3 with Cl in the groundwater samples and the isotopic signatures implied that NO3 in groundwater resources originated from unsuitable sanitary and sewage disposal practices in the Ger districts, typically located in the upstream area surrounding UB with dense and poor living conditions.
- The stable isotope signatures delineated the typical groundwater flow in UB. Precipitation recharges into bedrock groundwater in the upslope area, flows downward, and eventually discharges to the local base level of the basin, the Tuul River, through the alluvial aquifer. In the alluvial aquifer along the river, active pumping from multiple wells using riverbank filtration drives dynamic mixing of the groundwater and river water.
- The Tuul River and shallow groundwater from the alluvial aquifer had natural background NO3 concentrations. Meanwhile, Group 3 groundwater, accounting for 32% of the total groundwater samples, showed NO3 contamination, with minimum and maximum concentrations of 64.0 and 305.6 mg/L, respectively. Finally, Group 2 groundwater samples had a maximum NO3 concentration of 47.4 mg/L and median of 15.8 mg/L, indicating that this groundwater was influenced by anthropogenic sources.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tsogtbaatar, J.; Janchivdorj, L.; Unurjargal, D.; Erdenechimeg, B. The Groundwater Problem in Mongolia. In UNESCO Chair Workshop on International Strategy for Sustainable Groundwater Management: Transboundary Aquifers and Integrated Watershed Management; UNESCO Office: Beijing, China, 2009; p. 25. [Google Scholar]
- Dagvadorj, D.; Natsagadorj, L.; Dorjpurev, J.; Namkhainyam, B. Mongolia Assessment Report on Climate Change; Ministry of Environment, Nature and Tourism of Mongolia: Ulaanbaatar, Mongolia, 2009. [Google Scholar]
- Enkhbaatar, D. Strategies for the Sustainable Redevelopment of Residential Buildings in Ulaanbaatar, Mongolia. Master’s Thesis, Rmit University, Melbourne, Australia, 2010. [Google Scholar]
- National Statistical Office of Mongolia. Bulletin of Statistics, Ulaanbaatar; National Statistical Office of Mongolia: Ulaanbaatar, Mongolia, 2016. [Google Scholar]
- Danzan, N. Migration into Ulaanbaatar City. In Workshop on Migration into Cities; Irmgard Coninx Foundation: Berlin, Germany, 2007. [Google Scholar]
- Buyankhishig, N.; Aley, M.; Enkhbayar, D. Abstraction Influence on Alluvial Aquifer of the Tuul River, Mongolia. In Trends and Sustainability of Groundwater in Highly Stressed Aquifers; IAHS-AISH Press: Hyderabad, India, 2009; pp. 80–88. [Google Scholar]
- Hiller, B.; Jadamba, N. Groundwater use in the Selenge river basin, Mongolia. J. Groundwater Sci. Eng. 2013, 1, 11–32. [Google Scholar]
- Jadambaa, N.; Tserenjav, G.; Unurjargal, D. Groundwater in Ulaanbaatar Area. In Ecology and Sustainable Development; The Institute of Geography and Geoecology: Ulaanbaatar, Mongolia, 2000. [Google Scholar]
- Battumur, R.; Chuluunbaatar, S. Present Situation and Future Prospective of Drinking and Industrial Water Supply of Ulaanbaatar; Urban Planning Designing Institute of Mongolia: Ulaanbaatar, Mongolia, 2000. [Google Scholar]
- Zandaryaa, S.; Borhculuun, U.; Munkhtuya, S. Reserves, consumption and contamination of groundwater in Ulaanbaatar, Mongolia. Atlas Urban Geol. 2003, 14, 445–488. [Google Scholar]
- Kelderman, P.; Batima, P. Water quality assessment of rivers in Mongolia. Water Sci. Technol. 2006, 53, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Itoh, M.; Takemon, Y.; Makabe, A.; Yoshimizu, C.; Kohzu, A.; Ohte, N.; Tumurskh, D.; Tayasu, I.; Yoshida, N.; Nagata, T. Evaluation of wastewater nitrogen transformation in a natural wetland (Ulaanbaatar, Mongolia) using dual-isotope analysis of nitrate. Sci. Total Environ. 2011, 409, 1530–1538. [Google Scholar] [CrossRef] [PubMed]
- Altansukh, O.; Davaa, G. Application of index analysis to evaluate the water quality of the Tuul River in Mongolia. J. Water Res. Prot. 2011, 3, 398–414. [Google Scholar] [CrossRef]
- Dalai, B.; Ishiga, H. Geochemical evaluation of present-day Tuul River sediments, Ulaanbaatar basin, Mongolia. Environ. Monit. Assess. 2013, 185, 2869–2881. [Google Scholar] [CrossRef] [PubMed]
- Senjim, B. Changes in River Bed Morphology and Ecology—Tuul River, Mongolia. Geological Issues in Mongolia. Proc. Mong. Acad. Sci. 2005, 5, 112–121. [Google Scholar]
- Batkhishig, O. Soil pollution of Ulaanbaatar. Proc. Mong. Acad. Sci. 2016, 53, 15–19. [Google Scholar]
- Graham, J.P.; Polizzotto, M.L. Pit latrines and their impacts on groundwater quality: A systematic review. Environ. Health Perspect. 2013, 121, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Batjargal, T.; Otgonjargal, E.; Baek, K.; Yang, J.S. Assessment of metals contamination of soils in Ulaanbaatar, Mongolia. J. Hazard. Mater. 2010, 184, 872–876. [Google Scholar] [CrossRef] [PubMed]
- Foster, S.S.D; Morris, B.L.; Chilton, P.J. Groundwater in urban development—A review of linkages and concerns. Impacts of Urban Growth on Surface Water and Groundwater Quality. In Proceedings of the IUGG 99, Symposium HS5, Birmingham, UK, 19–30 July 1999. [Google Scholar]
- Foster, S.S.D. The interdependence of groundwater and urbanisation in rapidly developing cities. Urban Water J. 2001, 3, 185–192. [Google Scholar] [CrossRef]
- Altai, Z.; Stoeckl, N.; King, D. Impacts of water demand side policies on Mongolian residential users. Water Pract. Technol. 2012, 7. [Google Scholar] [CrossRef]
- Karthe, D.; Chalov, S.; Borchardt, D. Water resources and their management in central Asia in the early twenty first century: Status, challenges and future prospects. Environ. Earth Sci. 2015, 73, 487–499. [Google Scholar] [CrossRef]
- Jadambaa, N. Groundwater Resource Evaluation and Flow System of Artesian Aquifers in Ulaanbaatar Area; Hydrogeology, Moscow University: Moscow, Russia, 1977. [Google Scholar]
- Engineering Geological Scientific-Research and Industrial Institute. Exploration for Evaluation Exploitable Groundwater Resource of an Alluvial Aquifer in the Tuul River Basin (Central Part); Engineering Geological Scientific-research and Industrial Institute: Ulaanbaatar, Mongolia, 1979; pp. 1–983. [Google Scholar]
- Japan International Cooperation Agency. The Study on Water Supply System in Ulaanbaatar and Surroundings; Japan International Cooperation Agency: Ulaanbaatar, Mongolia, 1995; pp. 1–1056. [Google Scholar]
- Buyankhishig, N. Hydrogeological Modeling for the Central Water Supply Source of the Ulaanbaatar City; Hydrogeology, Mongolian University of Science and Technology: Ulaanbaatar, Mongolia, 2008. [Google Scholar]
- Fenske, J.; Stephen, E. Development and Application of a Surface-Groundwater flow. Model, of the Tuul River Basin near Ulaanbaatar, Mongolia; US Army Corps of Engineers: Davis, CA, USA, 2016. [Google Scholar]
- Dandar, E. Water Resources Assessment in Cold Regions: The Upper Tuul River Basin, Mongolia; Technical University of Catalonia; UPC-Barcelona Tech.: Barcelona, Spain, 2017. [Google Scholar]
- Nriagu, J.; Nam, D.; Ayanwola, T.A.; Dinh, H.; Erdenechimeg, E.; Ochir, C.; Bolormaa, T. High levels of uranium in groundwater of Ulaanbaatar, Mongolia. Sci. Total Environ. 2012, 414, 722–726. [Google Scholar] [CrossRef] [PubMed]
- Bayartungalag, B. Evaluation of Urban Groundwater Quality at Ulaanbaatar, Mongolia Using Hydrochemical and Environmental Isotope Data; Korea University: Seoul, Korea, 2016. [Google Scholar]
- Basandorj, D.; Davaa, G. Tuul River Basin of Mongolia—Integrated Water Resource Management; Interpress: Ulaanbaatar, Mongolia, 2006. [Google Scholar]
- Jadambaa, N. Hydrogeology, 8th ed.; Geology and Mineralogy of Mongolia: Ulaanbaatar, Mongolia, 2010. [Google Scholar]
- Unudelger, G.; Banzragch, B. Hydrogeological Investigations in Ulaanbaatar Area; Geo-Information Center of Mongolia: Ulaanbaatar, Mongolia, 1993. [Google Scholar]
- Buyankhishig, N.; Altantuul, K. Groundwater condition in Ulaanbaatar city, Mongolia. In Proceedings of the 3rd International Forum on Strategic Technologies, Novosibirsk-Tomsk, Russia, 23–29 June 2008; pp. 644–647. [Google Scholar]
- IAEA/WISER. Global Networks of Isotopes in Precipitation; IAEA/WISER: Vienna, Austria, 2001. [Google Scholar]
- Mora, A.; Mahlknecht, J.; Rosales-Lagarde, L.; Hernández-Antonio, A. Assessment of major ions and trace elements in groundwater supplied to the Monterrey metropolitan area, Nuevo León, Mexico. Environ. Monit. Assess. 2017, 189, 394. [Google Scholar] [CrossRef] [PubMed]
- Nadiri, A.A.; Moghaddam, A.A.; Tsai, F.T.; Fijani, E. Hydrogeochemical analysis for Tasuj plain aquifer, Iran. J. Earth Syst. Sci. 2013, 122, 1091–1105. [Google Scholar] [CrossRef]
- Gu, H.; Chi, B.; Li, H.; Jiang, J.; Qin, W.; Wang, H. Assessment of groundwater quality and identification of contaminant sources of Liujiang basin in Qinhuangdao, North China. Environ. Earth Sci. 2015, 73, 6477–6493. [Google Scholar] [CrossRef]
- Chae, G.-T.; Yun, S.-T.; Kim, K.; Mayer, B. Hydrogeochemistry of sodium-bicarbonate type bedrock groundwater in the Pocheon spa area, South Korea: Water–rock interaction and hydrologic mixing. J. Hydrol. 2006, 321, 326–343. [Google Scholar] [CrossRef]
- Jiang, Y.; Wu, Y.; Groves, C.; Yuan, D.; Kambesis, P. Natural and anthropogenic factors affecting the groundwater quality in the Nandong karst underground river system in Yunan, China. J. Contam. Hydrol. 2009, 109, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, R.J. Mechanisms controlling world water chemistry. Science 1970, 170, 1088–1090. [Google Scholar] [CrossRef] [PubMed]
- Hounslow, W. Water Quality Data: Analysis and Interpretation; CRC Press: Boca Raton, FL, USA, 1995. [Google Scholar]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Craig, H. Isotopic variations in meteoric waters. Science 1961, 133, 1702–1703. [Google Scholar] [CrossRef] [PubMed]
- Tsujimura, M.; Abe, Y.; Tanaka, T.; Shimada, J.; Higuchi, S.; Yamanaka, T.; Davaa, G.; Oyunbaatar, D. Stable isotopic and geochemical characteristics of groundwater in Kherlen river basin, a semi-arid region in Eastern Mongolia. J. Hydrol. 2007, 333, 47–57. [Google Scholar] [CrossRef]
- Zhang, B.; Song, X.; Zhang, Y.; Han, D.; Tang, C.; Yu, Y.; Ma, Y. Hydrochemical characteristics and water quality assessment of surface water and groundwater in Songnen plain, Northeast China. Water Res. 2012, 46, 2737–2748. [Google Scholar] [CrossRef] [PubMed]
- Harvey, F.E.; Welker, J.M. Stable isotopic composition of precipitation in the semi-arid north-central portion of the US Great Plains. J. Hydrol. 2000, 238, 90–109. [Google Scholar] [CrossRef]
- Dogramaci, S.; Skrzypek, G.; Dodson, W.; Grierson, P.F. Stable isotope and hydrochemical evolution of groundwater in the semi-arid Hamersley basin of subtropical Northwest Australia. J. Hydrol. 2012, 475, 281–293. [Google Scholar] [CrossRef]
- Clark, I.D.; Fritz, P. Environmental Isotopes in Hydrogeology; CRC Press: Boca Raton, FL, USA, 1997. [Google Scholar]
- Jouzel, J.; Froehlich, K.; Schotterer, U. Deuterium and oxygen-18 in present-day precipitation: Data and modelling. Hydrol. Sci. J. 1997, 42, 747–763. [Google Scholar] [CrossRef]
- Kendall, C.; Doctor, D. Stable isotope applications in hydrologic studies. Treatise Geochem. 2003, 5, 319–364. [Google Scholar]
- Terwey, J.L. Isotopes in groundwater hydrology. In Challenges in African Hydrology and Water Resources (Proceedings of the Harare Symposium); International Association of Hydrological Sciences: Wallingford, UK, 1984. [Google Scholar]
- Gomo, M.; van Tonder, G.J.; Steyl, G. Investigation of the hydrogeochemical processes in an alluvial channel aquifer located in a typical Karoo basin of Southern Africa. Environ. Earth Sci. 2013, 70, 227–238. [Google Scholar] [CrossRef]
- Spaliding, R.; Exner, M. Occurrence of nitrate in groundwater. A review: J. Environ. Qual. 1993, 22, 392–402. [Google Scholar]
- World Health Organization. Guidelines for Drinking-Water Quality; First Addendum to the Fourth Edition; World Health Organization: Geneva, Switzerland, 2017; p. 541. [Google Scholar]
- Ward, M.H.; Mark, S.D.; Cantor, K.P.; Weisenburger, D.D.; Correa-Villasenor, A.; Zahm, S.H. Drinking water nitrate and the risk of Non-Hodgkin’s lymphoma. Epidemiology 1996, 7, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Weyer, P.J.; Cerhan, J.R.; Kross, B.C.; Hallberg, G.R.; Kantamneni, J.; Breuer, G.; Jones, M.P.; Zheng, W.; Lynch, C.F. Municipal drinking water nitrate level and cancer risk in older women: The Iowa women’s health study. Epidemiology 2001, 12, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Stallard, R.; Edmond, J. Geochemistry of the amazon: 2. The influence of geology and weathering environment on the dissolved load. J. Geophys. Res.-Oceans 1983, 88, 9671–9688. [Google Scholar] [CrossRef]

| Parameter | Ground Water (N = 95) | River Water (N = 20) | ||||||
|---|---|---|---|---|---|---|---|---|
| Min. | Max. | Median | Std. Dev | Min. | Max. | Median | Std. Dev | |
| EC (µS/cm) | 65.0 | 2035.0 | 426.0 | 424.1 | 43.0 | 841.0 | 96.0 | 182.6 |
| pH | 6.3 | 8.9 | 7.2 | 0.5 | 6.5 | 8.9 | 7.8 | 0.6 |
| Temp. (°C) | 1.2 | 14.9 | 5.9 | 2.5 | 1.9 | 21.4 | 8.0 | 6.0 |
| HCO3 (mg/L) | 24.4 | 473.5 | 154.7 | 111.2 | 14.6 | 250.5 | 38.6 | 59.1 |
| SO4 (mg/L) | BDL | 104.6 | 22.3 | 30.2 | 4.0 | 65.0 | 6.6 | 14.0 |
| Cl (mg/L) | 0.6 | 206.1 | 7.2 | 35.3 | 0.4 | 62.4 | 1.8 | 14.9 |
| NO3 (mg/L) | BDL | 305.4 | 12.0 | 65.7 | 0.1 | 6.5 | 1.3 | 1.9 |
| SiO2 (mg/L) | 0.2 | 18.6 | 10.4 | 4.4 | BDL | 9.6 | 7.0 | 3.1 |
| Ca (mg/L) | 7.0 | 197.1 | 48.9 | 53.2 | 6.8 | 50.9 | 11.5 | 12.8 |
| Mg (mg/L) | 1.4 | 73.6 | 9.9 | 11.8 | 1.0 | 7.9 | 1.6 | 2.0 |
| Na (mg/L) | 1.4 | 72.0 | 12.0 | 15.2 | 1.6 | 62.2 | 2.8 | 13.6 |
| K (mg/L) | BDL | 6.3 | 0.9 | 1.2 | BDL | 14.0 | 0.7 | 3.0 |
| Parameter | PC 1 | PC 2 |
|---|---|---|
| HCO3 | 0.30 | 0.70 |
| SO4 | 0.57 | 0.64 |
| Cl | 0.75 | 0.55 |
| NO3 | 0.76 | 0.56 |
| SiO2 | 0.04 | 0.87 |
| Ca | 0.62 | 0.72 |
| Mg | 0.68 | 0.63 |
| Na | 0.72 | 0.42 |
| K | 0.84 | −0.03 |
| % variance explained by eigenvalues | 67.8 | 10.0 |
| % variance explained by squared sum of loadings | 40.4 | 37.4 |
| Water Resource | δ18O | δ2H | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Min. | Max. | Median | Std. Dev. | Min. | Max. | Median | Std. Dev. | ||
| Precipitation | −30.5 | −1.6 | −11.5 | 7.5 | −236.5 | −16.6 | −81.4 | 59 | |
| River water | −16.9 | −12.5 | −15.1 | 0.9 | −127 | −96 | −113.9 | 6.8 | |
| Groundwater | Group 1 | −16.3 | −13.2 | −15.3 | 0.7 | −129.3 | −100 | −113.5 | 6.5 |
| Group 2 | −14.4 | −11.5 | −12.5 | 0.9 | −110.2 | −87.7 | −97.3 | 6.3 | |
| Group 3 | −13.7 | −11.1 | −12.4 | 0.7 | −108.4 | −87.2 | −96.7 | 5.3 | |
| Water Resource | Number of Samples | Min. | Max. | Median | Std. Dev. | |
|---|---|---|---|---|---|---|
| Groundwater | Group 1 | 48 | BDL | 27.8 | 1.7 | 6 |
| Group 2 | 28 | 8 | 47.4 | 15.8 | 11 | |
| Group 3 | 35 | 64 | 305.4 | 101.3 | 57.8 | |
| River water | 24 | BDL | 6.5 | 1.1 | 1.8 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batsaikhan, N.; Lee, J.M.; Nemer, B.; Woo, N.C. Water Resources Sustainability of Ulaanbaatar City, Mongolia. Water 2018, 10, 750. https://doi.org/10.3390/w10060750
Batsaikhan N, Lee JM, Nemer B, Woo NC. Water Resources Sustainability of Ulaanbaatar City, Mongolia. Water. 2018; 10(6):750. https://doi.org/10.3390/w10060750
Chicago/Turabian StyleBatsaikhan, Naranchimeg, Jae Min Lee, Buyankhishig Nemer, and Nam C. Woo. 2018. "Water Resources Sustainability of Ulaanbaatar City, Mongolia" Water 10, no. 6: 750. https://doi.org/10.3390/w10060750
APA StyleBatsaikhan, N., Lee, J. M., Nemer, B., & Woo, N. C. (2018). Water Resources Sustainability of Ulaanbaatar City, Mongolia. Water, 10(6), 750. https://doi.org/10.3390/w10060750






