Using Ionic Liquid Modified Zeolite as a Permeable Reactive Wall to Limit Arsenic Contamination of a Freshwater Lake—Pilot Tests
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Lab Zeolite Modification
2.3. Batch and Column Tests for As Removal
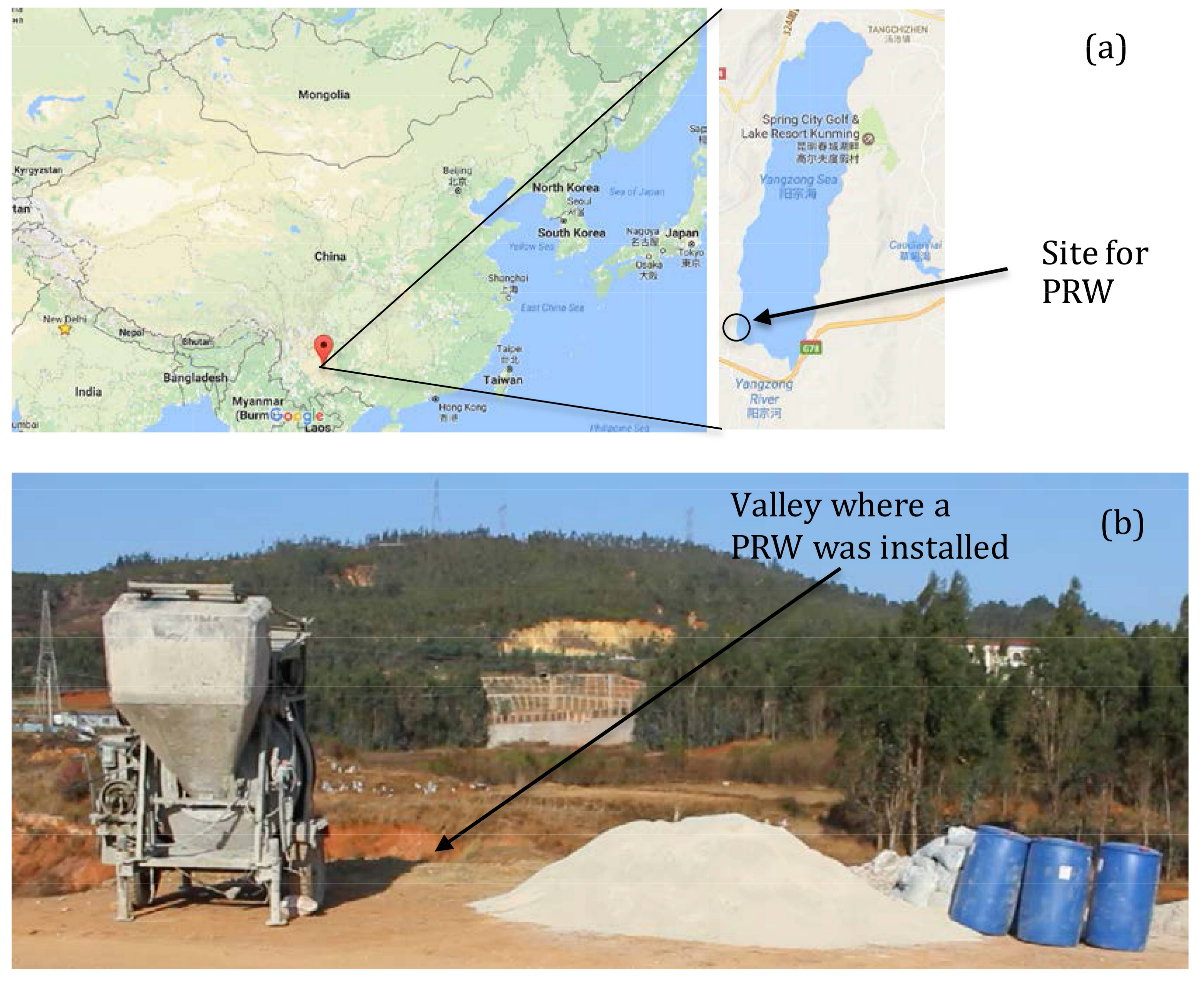
2.4. Pilot Scale PRW Design
2.5. Field Scale ILMZ Preparation
2.6. Pilot Scale PRW Installation
2.7. Sampling
2.8. Chemical Analyses
3. Results and Discussion
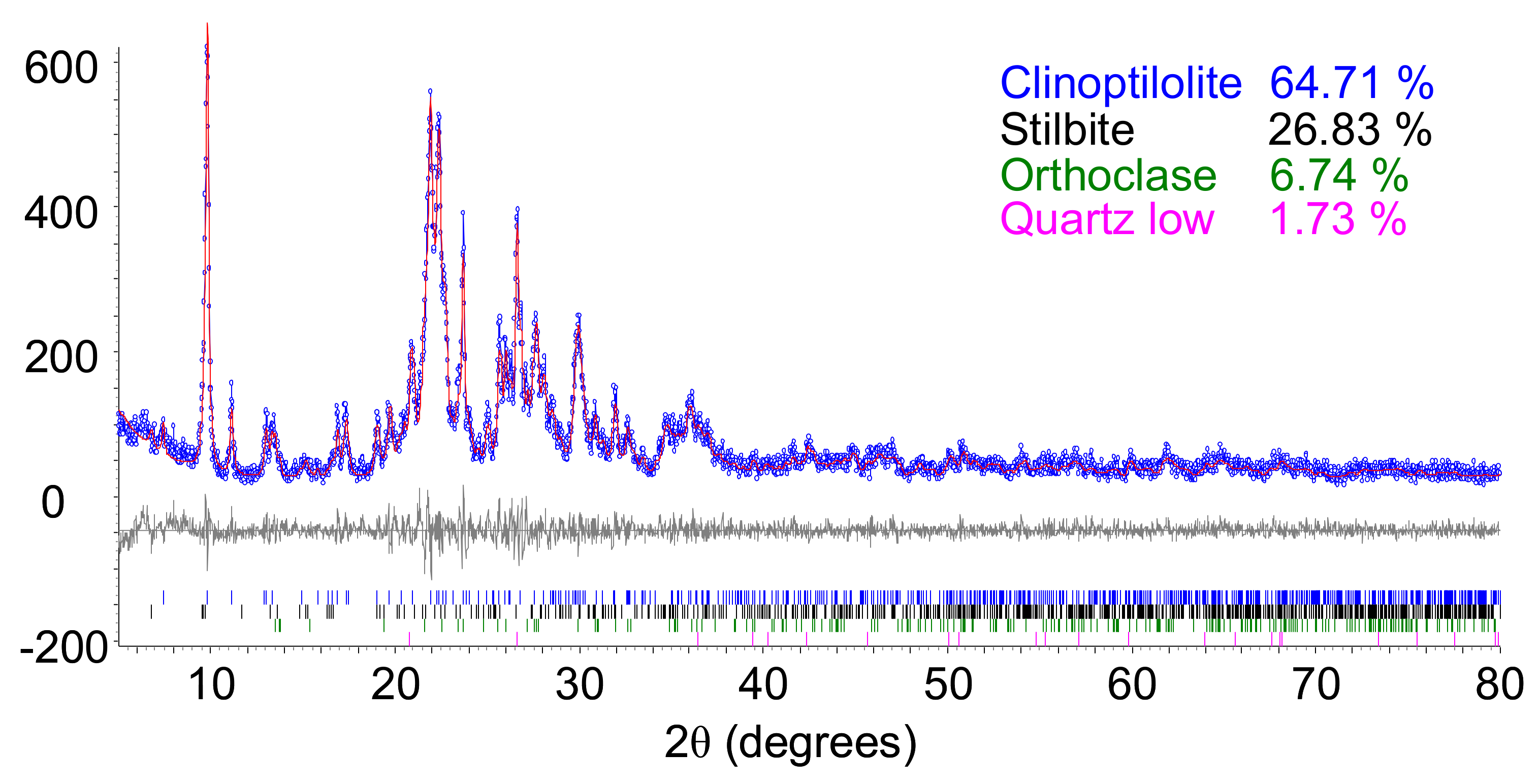
3.1. The Zeolite Characterization
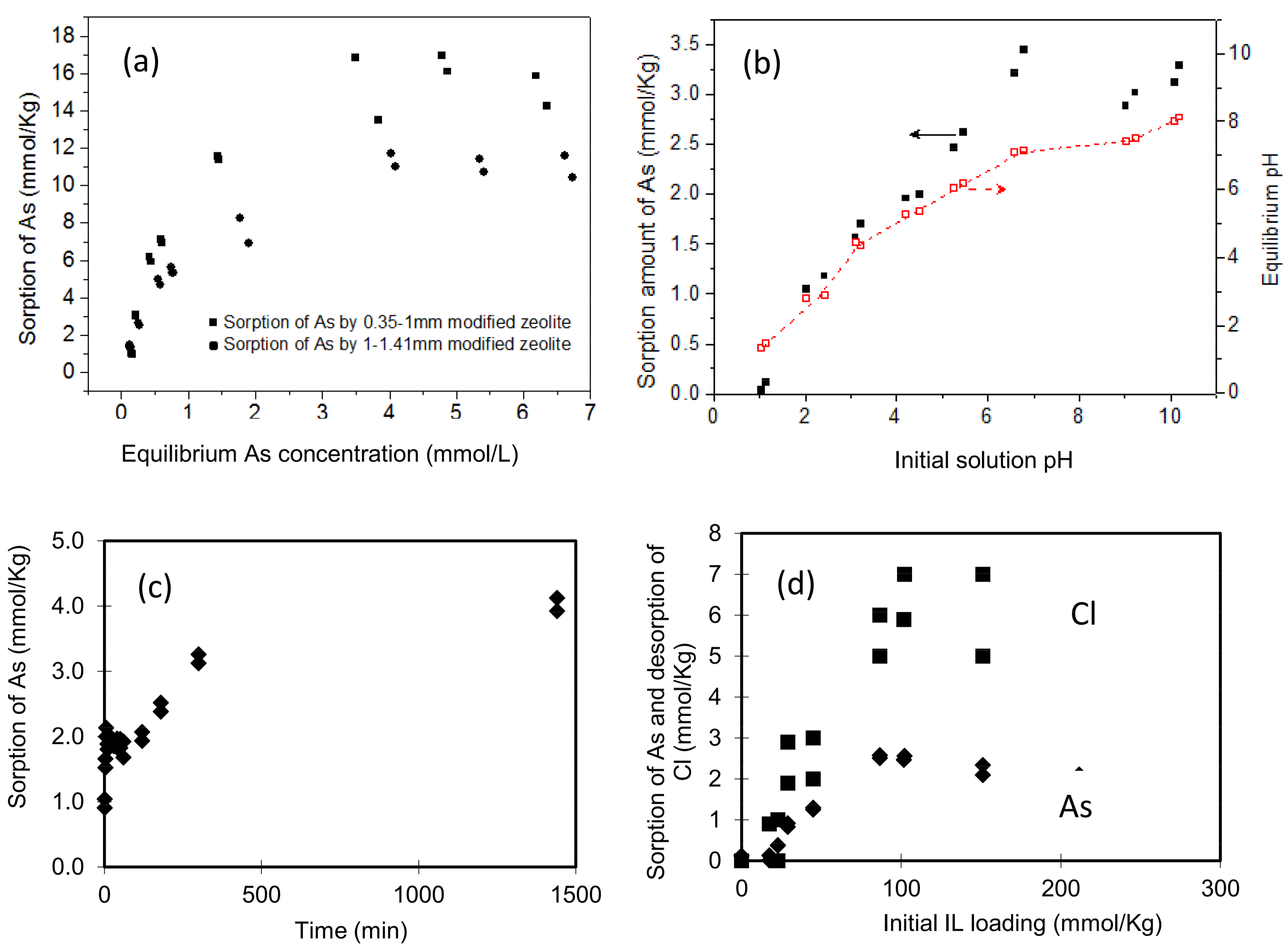
3.2. Batch Results on As Removal
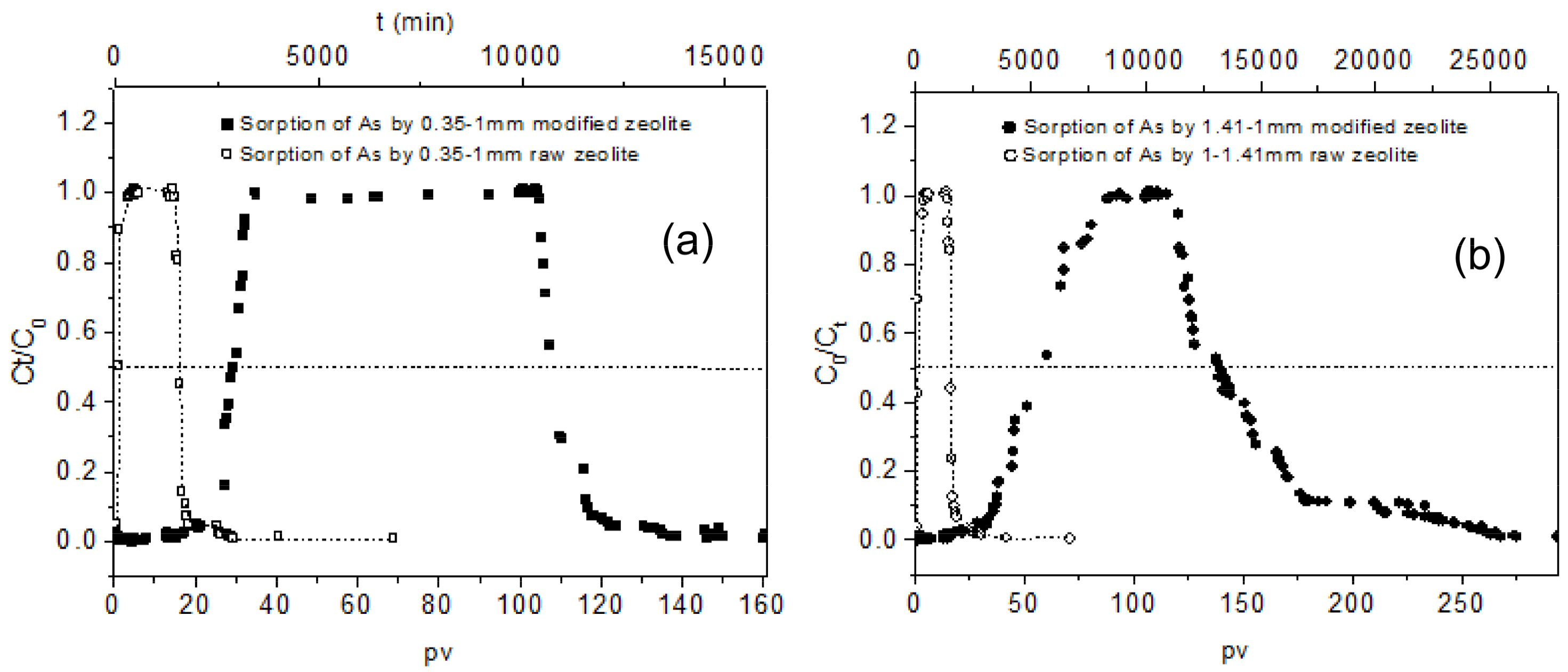
3.3. Column Results on As Removal
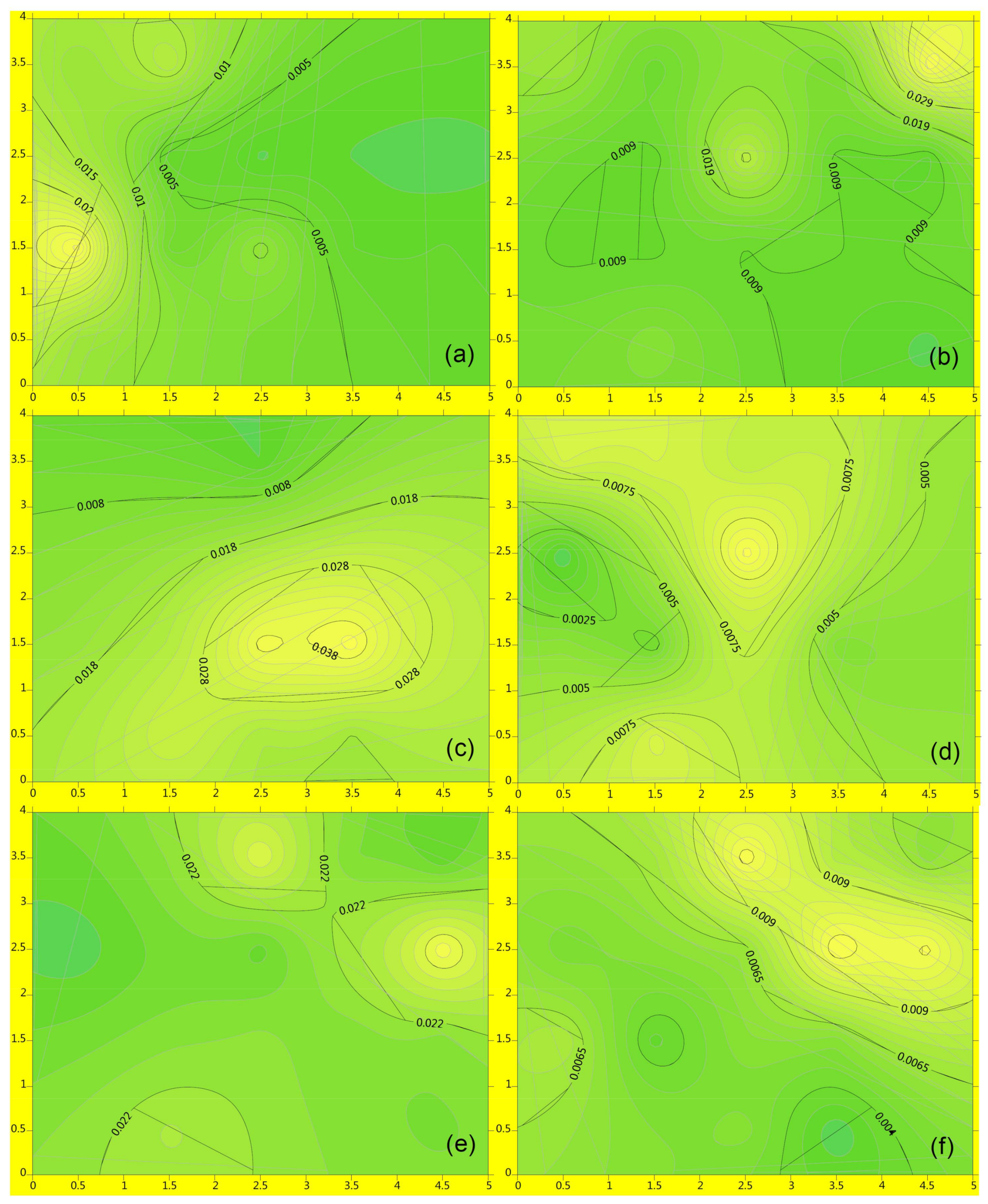
3.4. Pilot Tests on As Transport through PRW
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bowman, R.S.; Li, Z.; Roy, S.J.; Burt, T.; Johnson, T.L.; Johnson, R.L. Pilot Test of a Surfactant-Modified Zeolite Permeable Barrier for Groundwater Remediation. In Physicochemical Groundwater Remediation; Smith, J.A., Burns, S., Eds.; Kluwer Academic Publishers: Norwell, MA, USA, 2001; pp. 161–185. [Google Scholar]
- Wilkin, R.T.; Acree, S.D.; Ross, R.R.; Beak, D.G.; Lee, T.R. Performance of a zerovalent iron reactive barrier for the treatment of arsenic in groundwater: Part 1. Hydrogeochemical studies. J. Contam. Hydrol. 2009, 106, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Richards, P. Seven-year performance evaluation of a permeable reactive barrier. Remediat. J. 2008, 18, 63–78. [Google Scholar] [CrossRef]
- U.S. Department of Energy. Performance Assessment and Recommendations for Rejuvenation of a Permeable Reactive Barrier: Cotter Corporation’s Canon City, Colorado, Uranium Mill; U.S. Department of Energy: Grand Junction, CO, USA, 2005. Available online: https://www.energy.gov/sites/prod/files/canon_city_prb.pdf (accessed on 5 April 2018).
- Bi, J.; Liu, C.; Li, S. Variation of water quality of Yangzonghai Lake affected by arsenic pollution. Water Resour. Prot. 2014, 30, 84–89, (In Chinese with English Abstract). [Google Scholar]
- Wang, Z.; He, B.; Pan, X.; Zhang, K.; Wang, C.; Sun, J.; Yun, Z.; Jiang, G. The levels, trends and risk assessment of arsenic pollution in Yangzonghai Lake, Yunnan. Sci. Sin. Chim. 2011, 41, 556–564. [Google Scholar] [CrossRef]
- Zhang, Y.; Yun, X.; Zhang, Y.; Chen, X.; Liu, J.; Wang, J.; Zhang, Y.; Sun, C. Distribution and sources of arsenic in Yangzonghai Lake, China. Environ. Sci. 2012, 33, 3768–3777, (In Chinese with English Abstract). [Google Scholar]
- Chen, J.; Zhang, S.; Yang, X.; Huang, Z.; Wang, S.; Wang, C.; Wei, Y.; Zhang, G.; Xiao, J. Arsenic removal by coagulation process and the field expanding experiments for Yangzonghai Lake. Environ. Sci. 2015, 36, 202–208, (In Chinese with English Abstract). [Google Scholar]
- Chen, J.; Wang, S.; Zhang, S.; Yang, X.; Huang, Z.; Wang, C.; Wei, Q.; Zhang, G.; Xiao, J.; Jiang, F.; et al. Arsenic pollution and its treatment in Yangzonghai lake in China: In situ remediation. Ecotoxicol. Environ. Saf. 2015, 122, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Yang, C.; Li, S.; Sun, P.; Shen, S.; Li, Z. Arsenic mobility in the arsenic-contaminated Yangzonghai Lake in China. Ecotoxicol. Environ. Saf. 2014, 107, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Puls, R.W.; Paul, C.J.; Powell, R.M. The application of in situ permeable reactive (zero-valent iron) barrier technology for the remediation of chromate-contaminated groundwater: a field test. Appl. Geochem. 1999, 14, 989–1000. [Google Scholar] [CrossRef]
- Benner, S.G.; Blowes, D.W.; Ptacek, C.J. A full-scale porous reactive wall for prevention of acid mine drainage. Groundw. Monit. Remediat. 1997, 17, 99–107. [Google Scholar] [CrossRef]
- Weihermüller, L.; Siemens, J.; Deurer, M.; Knoblauch, S.; Rupp, H.; Göttlein, A.; Pütz, T. In situ soil water extraction: A review. J. Environ. Qual. 2007, 36, 1735–1748. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Jean, J.S.; Jiang, W.T.; Chang, P.H.; Chen, C.J.; Liao, L. Removal of arsenic from water using Fe-exchanged natural zeolite. J. Hazard. Mater. 2011, 187, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Bowman, R.S. Applications of surfactant-modified zeolites to environmental remediation. Microporous Mesoporous Mater. 2003, 61, 43–56. [Google Scholar] [CrossRef]
- Li, Z.; Roy, S.J.; Zou, Y.; Bowman, R.S. Long term chemical and biological stability of surfactant-modified zeolite. Environ. Sci. Technol. 1998, 32, 2628–2632. [Google Scholar] [CrossRef]
- Zhu, N.; Qiao, J.; Ye, Y.; Yan, T. Synthesis of mesoporous bismuth-impregnated aluminum oxide for arsenic removal: Adsorption mechanism study and application to a lab-scale column. J. Environ. Manag. 2018, 211, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Taleb, K.; Markovski, J.; Milosavljević, M.; Marinović-Cincović, M.; Rusmirović, J.; Ristić, M.; Marinković, A. Efficient arsenic removal by cross-linked macroporous polymer impregnated with hydrous iron oxide: Material performance. Chem. Eng. J. 2015, 279, 66–78. [Google Scholar] [CrossRef]
- Vogan, J.L.; Focht, R.M.; Clark, D.K.; Graham, S.L. Performance evaluation of a permeable reactive barrier for remediation of dissolved chlorinated solvents in groundwater. J. Hazard. Mater. 1999, 68, 97–108. [Google Scholar] [CrossRef]
- Ludwig, R.D.; McGregor, R.G.; Blowes, D.W.; Benner, S.G.; Mountjoy, K. A permeable reactive barrier for treatment of heavy metals. Groundwater 2002, 40, 59–66. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, L.; Li, Z.; Lv, G.; Mei, L.; Wang, H.; Shi, S.; Wei, Y.; Wang, X.; Ning, P.; Wei, Y. Using Ionic Liquid Modified Zeolite as a Permeable Reactive Wall to Limit Arsenic Contamination of a Freshwater Lake—Pilot Tests. Water 2018, 10, 448. https://doi.org/10.3390/w10040448
Liao L, Li Z, Lv G, Mei L, Wang H, Shi S, Wei Y, Wang X, Ning P, Wei Y. Using Ionic Liquid Modified Zeolite as a Permeable Reactive Wall to Limit Arsenic Contamination of a Freshwater Lake—Pilot Tests. Water. 2018; 10(4):448. https://doi.org/10.3390/w10040448
Chicago/Turabian StyleLiao, Libing, Zhaohui Li, Guocheng Lv, Lefu Mei, Haijuan Wang, Shuliu Shi, Yaozu Wei, Xiaoyu Wang, Ping Ning, and Yanke Wei. 2018. "Using Ionic Liquid Modified Zeolite as a Permeable Reactive Wall to Limit Arsenic Contamination of a Freshwater Lake—Pilot Tests" Water 10, no. 4: 448. https://doi.org/10.3390/w10040448
APA StyleLiao, L., Li, Z., Lv, G., Mei, L., Wang, H., Shi, S., Wei, Y., Wang, X., Ning, P., & Wei, Y. (2018). Using Ionic Liquid Modified Zeolite as a Permeable Reactive Wall to Limit Arsenic Contamination of a Freshwater Lake—Pilot Tests. Water, 10(4), 448. https://doi.org/10.3390/w10040448





