Bromate Reduction by Iron(II) during Managed Aquifer Recharge: A Laboratory-Scale Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Anoxic Batch Reactors
2.3. Water and Chemicals
2.4. Analytical Methods
3. Results
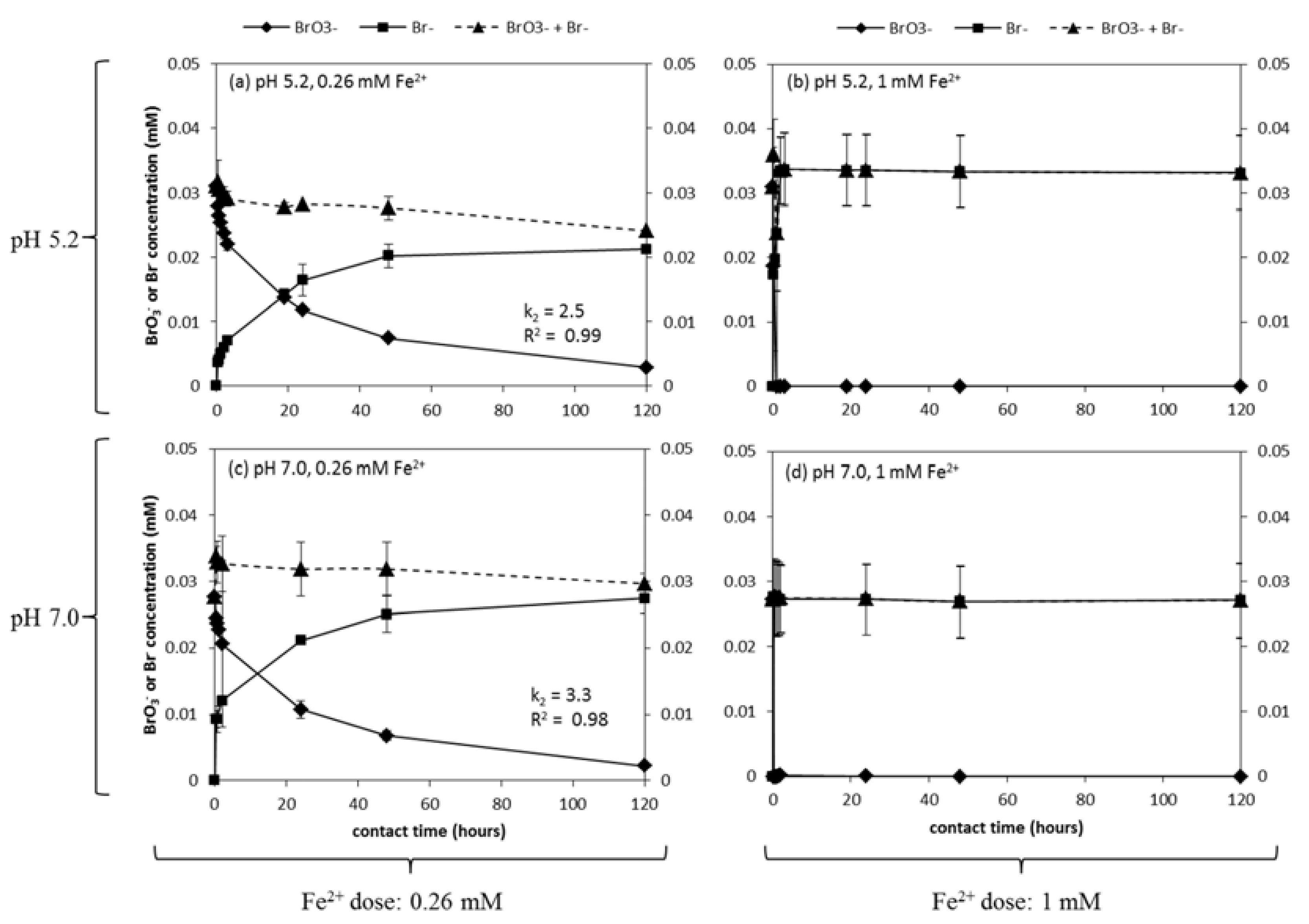
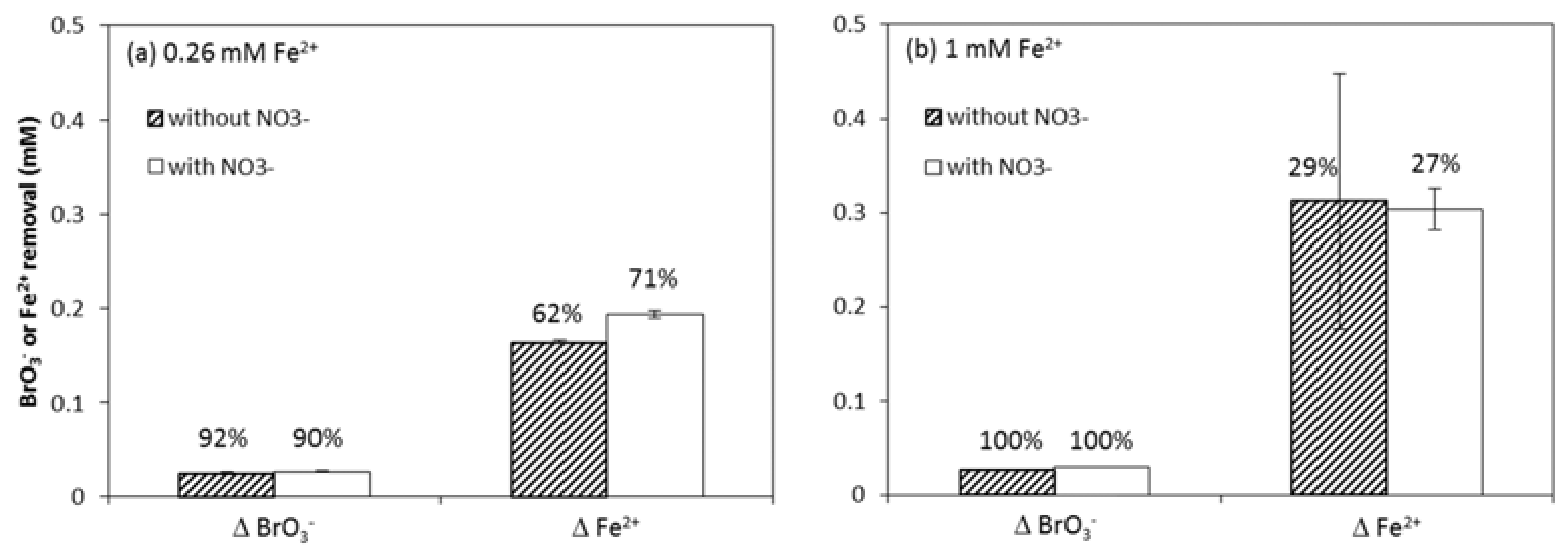
3.1. BrO3− Reduction Rate and Mass Balance
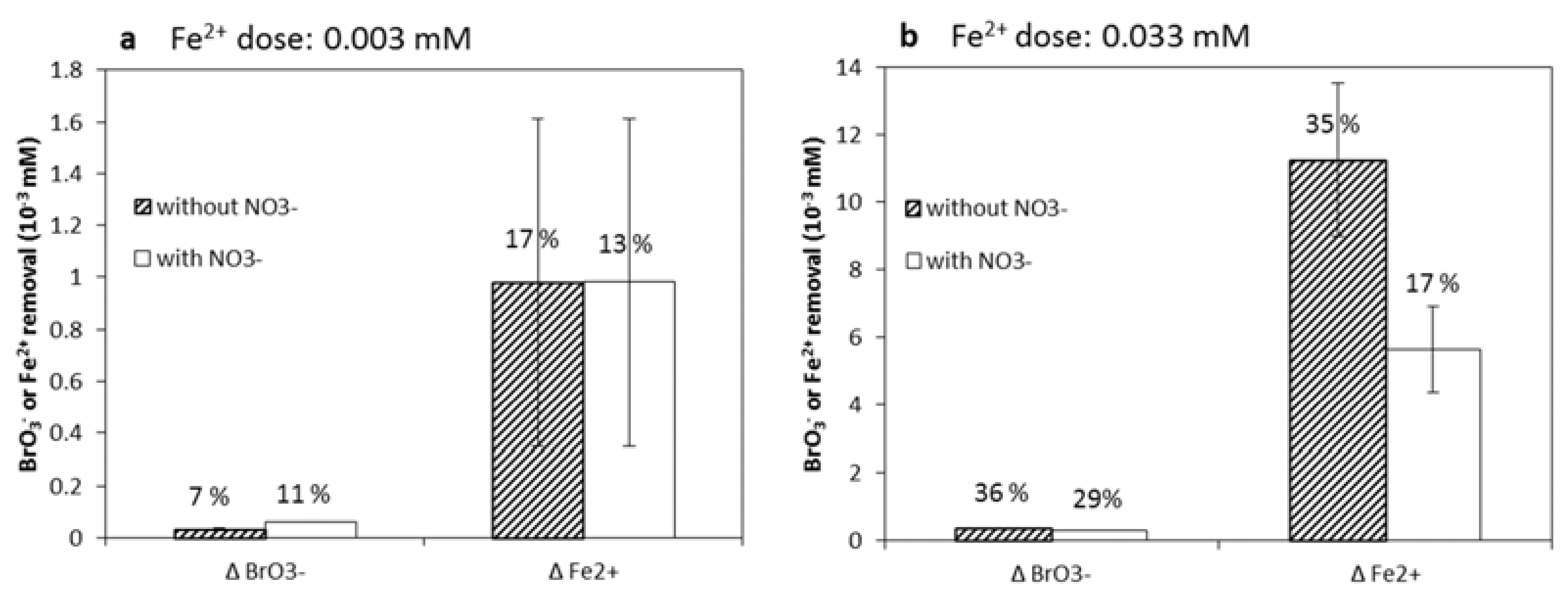
3.2. NO3−, A Competing Electron Acceptor?
3.3. pH Change and Fe3+ Hydrolysis
3.4. BrO3− Reduction under Concentrations Similar to MAR
4. Discussion
4.1. BrO3− Reduction Mechanism
4.2. Feasibility of BrO3− Reduction by Fe2+ during MAR
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Laws, B.V.; Dickenson, E.R.V.; Johnson, T.A.; Snyder, S.A.; Drewes, J.E. Attenuation of contaminants of emerging concern during surface-spreading aquifer recharge. Sci. Total Environ. 2011, 409, 1087–1094. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.C.; Noh, J.H.; Chae, S.R.; Choi, J.; Lee, Y.; Maeng, S.K. A multi-parametric approach assessing microbial viability and organic matter characteristics during managed aquifer recharge. Sci. Total Environ. 2015, 524–525, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Postigo, C.; Barceló, D. Synthetic organic compounds and their transformation products in groundwater: Occurrence, fate and mitigation. Sci. Total Environ. 2015, 503–504, 32–47. [Google Scholar] [CrossRef] [PubMed]
- Drewes, J.E.; Heberer, T.; Rauch, T.; Reddersen, K. Fate of pharmaceuticals during ground water recharge. Ground Water Monit. Remediat. 2003, 23, 64–72. [Google Scholar] [CrossRef]
- Ternes, T.A.; Meisenheimer, M.; McDowell, D.; Sacher, F.; Brauch, H.J.; Haist-Gulde, B.; Preuss, G.; Wilme, U.; Zulei-Seibert, N. Removal of pharmaceuticals during drinking water treatment. Environ. Sci. Technol. 2002, 36, 3855–3863. [Google Scholar] [CrossRef] [PubMed]
- Hübner, U.; Miehe, U.; Jekel, M. Optimized removal of dissolved organic carbon and trace organic contaminants during combined ozonation and artificial groundwater recharge. Water Res. 2012, 46, 6059–6068. [Google Scholar] [CrossRef] [PubMed]
- Hollender, J.; Zimmermann, S.G.; Koepke, S.; Krauss, M.; McArdell, C.S.; Ort, C.; Singer, H.; von Gunten, U.; Siegrist, H. Elimination of organic micropollutants in a municipal wastewater treatment plant upgraded with a full-scale post-ozonation followed by sand filtration. Environ. Sci. Technol. 2009, 43, 7862–7869. [Google Scholar] [CrossRef] [PubMed]
- Scheideler, J.; Lekkerkerker-Teunissen, K.; Knol, T.; Ried, A.; Verberk, J.; van Dijk, H. Combination of O3/H2O2 and uv for multiple barrier micropollutant treatment and bromate formation control—An economic attractive option. Water Pract. Technol. 2011, 6. [Google Scholar] [CrossRef]
- Lekkerkerker, K.; Scheideler, J.; Maeng, S.K.; Ried, A.; Verberk, J.Q.J.C.; Knol, A.H.; Amy, G.; van Dijk, J.C. Advanced oxidation and artificial recharge: A synergistic hybrid system for removal of organic micropollutants. Water Sci. Technol. Water Supply 2009, 9, 643–651. [Google Scholar] [CrossRef]
- Lekkerkerker-Teunissen, K.; Chekol, E.T.; Maeng, S.K.; Ghebremichael, K.; Houtman, C.J.; Verliefde, A.R.D.; Verberk, J.Q.J.C.; Amy, G.L.; van Dijk, J.C. Pharmaceutical removal during managed aquifer recharge with pretreatment by advanced oxidation. Water Sci. Technol. Water Supply 2012, 12, 755–767. [Google Scholar] [CrossRef]
- Oller, I.; Malato, S.; Sánchez-Pérez, J.A. Combination of Advanced Oxidation Processes and biological treatments for wastewater decontamination—A review. Sci. Total Environ. 2011, 409, 4141–4166. [Google Scholar] [CrossRef] [PubMed]
- Kurokawa, Y.; Maekawa, A.; Takahashi, M.; Hayashi, Y. Toxicity and carcinogenicity of potassium bromate—A new renal carcinogen. Environ. Health Perspect. 1990, 87, 309–335. [Google Scholar] [PubMed]
- Haag, W.R.; Holgne, J. Ozonation of bromide-containing waters: Kinetics of formation of hypobromous acid and bromate. Environ. Sci. Technol. 1983, 17, 261–267. [Google Scholar] [CrossRef]
- Assuncao, A.; Martins, M.; Silva, G.; Lucas, H.; Coelho, M.R.; Costa, M.C. Bromate removal by anaerobic bacterial community: Mechanism and phylogenetic characterization. J. Hazard. Mater. 2011, 197, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Shang, C. A review on bromate occurrence and removal strategies in water supply. Water Sci. Technol. Water Supply 2006, 6, 131–136. [Google Scholar] [CrossRef]
- Xiao, Q.; Yu, S.; Li, L.; Wang, T.; Liao, X.; Ye, Y. An overview of advanced reduction processes for bromate removal from drinking water: Reducing agents, activation methods, applications and mechanisms. J. Hazard. Mater. 2017, 324, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Kurokawa, Y.; Aoki, S.; Matsushima, Y.; Takamura, N.; Imazawa, T.; Hayashi, Y. Dose-response studies on the carcinogenicity of potassium bromate in F344 rats after long-term oral administration. J. Natl. Cancer Inst. 1986, 77, 977–982. [Google Scholar] [PubMed]
- Crofton, K.M. Bromate: Concern for developmental neurotoxicity? Toxicology 2006, 221, 212–216. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2011; Volume 216, pp. 303–304. [Google Scholar]
- U.S. EPA. Guidelines for Carcinogen Risk Assessment; Risk Assessment Forum: Washington, DC, USA, 2005.
- Carney, M. European drinking water standards. J. Am. Water Works Assoc. 1991, 83, 48–55. [Google Scholar] [CrossRef]
- Chen, R.; Yang, Q.; Zhong, Y.; Li, X.; Liu, Y.; Li, X.M.; Du, W.X.; Zeng, G.M. Sorption of trace levels of bromate by macroporous strong base anion exchange resin: Influencing factors, equilibrium isotherms and thermodynamic studies. Desalination 2014, 344, 306–312. [Google Scholar] [CrossRef]
- Xu, C.; Shi, J.; Zhou, W.; Gao, B.; Yue, Q.; Wang, X. Bromate removal from aqueous solutions by nano crystalline akaganeite (β-FeOOH)-coated quartz sand (CACQS). Chem. Eng. J. 2012, 187, 63–68. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, X. Preparation of Zn-Al CLDH to remove bromate from drinking water. J. Environ. Eng. (USA) 2014, 140. [Google Scholar] [CrossRef]
- Theiss, F.L.; Couperthwaite, S.J.; Ayoko, G.A.; Frost, R.L. A review of the removal of anions and oxyanions of the halogen elements from aqueous solution by layered double hydroxides. J. Coll. Interface Sci. 2014, 417, 356–368. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Yu, S.; Tang, Y. Adsorptive characteristics of bromate from aqueous solutions by modified granular activated carbon. Huanjing Kexue Xuebao/Acta Sci. Circumstantiae 2014, 34, 630–637. [Google Scholar]
- Gyparakis, S.; Diamadopoulos, E. Formation and reverse osmosis removal of bromate ions during ozonation of groundwater in coastal areas. Sep. Sci. Technol. 2007, 42, 1465–1476. [Google Scholar] [CrossRef]
- Van Der Hoek, J.P.; Rijnbende, D.O.; Lokin, C.J.A.; Bonné, P.A.C.; Loonen, M.T.; Hofman, J.A.M.H. Electrodialysis as an alternative for reverse osmosis in an integrated membrane system. Desalination 1998, 117, 159–172. [Google Scholar] [CrossRef]
- Wang, Q.; Snyder, S.; Kim, J.; Choi, H. Aqueous ethanol modified nanoscale zerovalent iron in Bromate reduction: Synthesis, characterization, and reactivity. Environ. Sci. Technol. 2009, 43, 3292–3299. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xu, Z.; Wan, H.; Zheng, J.; Yin, D.; Zheng, S. Aqueous bromate reduction by catalytic hydrogenation over Pd/Al2O3 catalysts. Appl. Catal. B Environ. 2010, 96, 307–313. [Google Scholar] [CrossRef]
- Siddiqui, M.; Amy, G.; Ozekin, K.; Zhai, W.; Westerhoff, P. Alternative strategies for removing bromate. J. Am. Water Works Assoc. (USA) 1994, 86, 81–96. [Google Scholar] [CrossRef]
- Kishimoto, N.; Matsuda, N. Bromate ion removal by electrochemical reduction using an activated carbon felt electrode. Environ. Sci. Technol. 2009, 43, 2054–2059. [Google Scholar] [CrossRef] [PubMed]
- Mao, R.; Zhao, X.; Qu, J. Electrochemical Reduction of Bromate by a Pd Modified Carbon Fiber Electrode: Kinetics and Mechanism. Electrochim. Acta 2014, 132, 151–157. [Google Scholar] [CrossRef]
- Kirisits, M.J.; Snoeyink, V.L.; Inan, H.; Chee-sanford, J.C.; Raskin, L.; Brown, J.C. Water quality factors affecting bromate reduction in biologically active carbon filters. Water Res. 2001, 35, 891–900. [Google Scholar] [CrossRef]
- Liu, J.; Yu, J.; Li, D.; Zhang, Y.; Yang, M. Reduction of bromate in a biological activated carbon filter under high bulk dissolved oxygen conditions and characterization of bromate-reducing isolates. Biochem. Eng. J. 2012, 65, 44–50. [Google Scholar] [CrossRef]
- Hijnen, W.A.M.; Jong, R.; van der Kooij, D. Bromate removal in a denitrifying bioreactor used in water treatment. Water Res. 1999, 33, 1049–1053. [Google Scholar] [CrossRef]
- Wang, F.; van Halem, D.; Ding, L.; Bai, Y.; Lekkerkerker-Teunissen, K.; van der Hoek, J.P. Effective removal of bromate in nitrate-reducing anoxic zones during managed aquifer recharge for drinking water treatment. Water Res. 2018, 130, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Hübner, U.; Kuhnt, S.; Jekel, M.; Drewes, J.E. Fate of bulk organic carbon and bromate during indirect water reuse involving ozone and subsequent aquifer recharge. J. Water Reuse Desal. 2016, 6, 413–420. [Google Scholar] [CrossRef]
- Xie, L.; Shang, C. The effects of operational parameters and common anions on the reactivity of zero-valent iron in bromate reduction. Chemosphere 2007, 66, 1652–1659. [Google Scholar] [CrossRef] [PubMed]
- Stefánsson, A. Iron(III) hydrolysis and solubility at 25 °C. Environ. Sci. Technol. 2007, 41, 6117–6123. [Google Scholar] [CrossRef] [PubMed]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Dong, Z.J.; Dong, W.Y.; Zhang, X.M.; Yu, X.H.; Ou, Y.F.; Du, H. Removal of bromate by ferrous sulfate reduction in drinking water. In Proceedings of the 2009 3rd International Conference on Bioinformatics and Biomedical Engineering (iCBBE), Beijing, China, 11–13 June 2009. [Google Scholar]
- Barbieri, M.; Carrera, J.; Sanchez-Vila, X.; Ayora, C.; Cama, J.; Köck-Schulmeyer, M.; de Alda, M.L.; Barceló, D.; Brunet, J.T.; García, M.H. Microcosm experiments to control anaerobic redox conditions when studying the fate of organic micropollutants in aquifer material. J. Contam. Hydrol. 2011, 126, 330–345. [Google Scholar] [CrossRef] [PubMed]
- Kedziorek, M.A.M.; Geoffriau, S.; Bourg, A.C.M. Organic matter and modeling redox reactions during river bank filtration in an alluvial aquifer of the Lot River, France. Environ. Sci. Technol. 2008, 42, 2793–2798. [Google Scholar] [CrossRef] [PubMed]
- Grischek, T.; Paufler, S. Prediction of iron release during riverbank filtration. Water 2017, 9, 317. [Google Scholar] [CrossRef]
- Song, X.; Wang, S.; Wang, Y.; Zhao, Z.; Yan, D. Addition of Fe2+ increase nitrate removal in vertical subsurface flow constructed wetlands. Ecol. Eng. 2016, 91, 487–494. [Google Scholar] [CrossRef]
- Huang, Y.H.; Zhang, T.C. Effects of low pH on nitrate reduction by iron powder. Water Res. 2004, 38, 2631–2642. [Google Scholar] [CrossRef] [PubMed]
- Buresh, R.J.; Moraghan, J. Chemical reduction of nitrate by ferrous iron. J. Environ. Qual. 1976, 5, 320–325. [Google Scholar] [CrossRef]
- Ottley, C.J.; Davison, W.; Edmunds, M.W. Chemical catalysis of nitrate reduction by iron (II). Geochim. Cosmochim. Acta 1997, 61, 1819–1828. [Google Scholar] [CrossRef]
- Krasner, S.W.; Glaze, W.H.; Weinberg, H.S.; Daniel, P.A.; Najm, I.N. Formation and control of bromate during ozonation of waters containing bromide. J. Am. Water Works Assoc. 1993, 85, 73–81. [Google Scholar] [CrossRef]
- Glaze, W.H.; Weinberg, H.S.; Cavanagh, J.E. Evaluating the formation of brominated DBPs during ozonation. J. Am. Water Works Assoc. 1993, 85, 96–103. [Google Scholar] [CrossRef]
- Amy, G.; Bull, R.; Craun, G.F.; Pegram, R.; Siddiqui, M. Disinfectants and Disinfectant by-Products; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Butler, R.; Godley, A.; Lytton, L.; Cartmell, E. Bromate environmental contamination: Review of impact and possible treatment. Crit. Rev. Environ. Sci. Technol. 2005, 35, 193–217. [Google Scholar] [CrossRef]
- Kirisits, M.J.; Snoeyink, V.L. Reduction of bromate in a BAC filter. J. Am. Water Works Assoc. 1999, 91, 74–84. [Google Scholar] [CrossRef]
- Thomas, D.; Rohrer, J. Determination of Chlorite, Bromate, Bromide, and Chlorate in Drinking Water by Ion Chromatography with an On-Line-Generated Postcolumn Reagent for Sub-µg/L Bromate Analysis; Thermo Fisher Scientific: Sunnyvale, CA, USA, 2017. [Google Scholar]
- Westerhoff, P. Reduction of nitrate, bromate, and chlorate by zero valent iron (Fe0). J. Environ. Eng. 2003, 129, 10–16. [Google Scholar] [CrossRef]
- Lengyel, I.; Nagy, I.; Bazsa, G. Kinetic study of the autocatalytic nitric acid-bromide reaction and its reverse, the nitrous acid-bromine reaction. J.Phys. Chem. 1989, 93, 2801–2807. [Google Scholar] [CrossRef]
- Britton, H.; Britton, H.G. 746. A potentiometric study of the reduction by potassium iodide of potassium bromate in sulphuric and hydrochloric acid solutions. J. Chem. Soc. (Resumed) 1952, 3887–3892. [Google Scholar] [CrossRef]
- Aksut, A. Investigation of Fe3+/Fe2+ redox reaction by electrochemical methods in aqueous solution. Commun. Fac. Sci. Univ. Ank. Ser. B 1994, 40, 83–93. [Google Scholar]
- Shen, W.; Lin, F.; Jiang, X.; Li, H.; Ai, Z.; Zhang, L. Efficient removal of bromate with core-shell Fe@Fe2O3 nanowires. Chem. Eng. J. 2017, 308, 880–888. [Google Scholar] [CrossRef]
- Hiemstra, T.; van Riemsdijk, W.H. Adsorption and surface oxidation of Fe(II) on metal (hydr)oxides. Geochim. Cosmochim. Acta 2007, 71, 5913–5933. [Google Scholar] [CrossRef]
- Williams, A.G.B.; Scherer, M.M. Spectroscopic evidence for Fe(II)-Fe(III) electron transfer at the iron oxide-water interface. Environ. Sci. Technol. 2004, 38, 4782–4790. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, S.A.; van Weert, G. On the catalysis of ferrous sulphate oxidation in autoclaves by nitrates and nitrites. Hydrometallurgy 1996, 42, 209–219. [Google Scholar] [CrossRef]
- Ohura, H.; Imato, T.; Kameda, K.; Yamasaki, S. Potentiometric determination of bromate using an Fe(III)-Fe(II) potential buffer by circulatory flow-injection analysis. Anal. Sci. 2004, 20, 513–518. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tresintsi, S.; Simeonidis, K.; Vourlias, G.; Stavropoulos, G.; Mitrakas, M. Kilogram-scale synthesis of iron oxy-hydroxides with improved arsenic removal capacity: Study of Fe (II) oxidation–precipitation parameters. Water Res. 2012, 46, 5255–5267. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; van Halem, D.; van der Hoek, J.P. The fate of H2O2 during managed aquifer recharge: A residual from advanced oxidation processes for drinking water production. Chemosphere 2016, 148, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; van Halem, D.; Liu, G.; Lekkerkerker-Teunissen, K.; van der Hoek, J.P. Effect of residual H2O2 from advanced oxidation processes on subsequent biological water treatment: A laboratory batch study. Chemosphere 2017, 185, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Maeng, S.K.; Sharma, S.K.; Lekkerkerker-Teunissen, K.; Amy, G.L. Occurrence and fate of bulk organic matter and pharmaceutically active compounds in managed aquifer recharge: A review. Water Res. 2011, 45, 3015–3033. [Google Scholar] [CrossRef] [PubMed]
- Stuyfzand, P.J. Hydrology and water quality aspects of Rhine bank groundwater in The Netherlands. J. Hydrol. 1989, 106, 341–363. [Google Scholar] [CrossRef]
- Eckert, P.; Appelo, C.A.J. Hydrogeochemical modeling of enhanced benzene, toluene, ethylbenzene, xylene (BTEX) remediation with nitrate. Water Resour. Res. 2002, 38. [Google Scholar] [CrossRef]
- Benz, M.; Brune, A.; Schink, B. Anaerobic and aerobic oxidation of ferrous iron at neutral pH by chemoheterotrophic nitrate-reducing bacteria. Arch. Microbiol. 1998, 169, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Oshiki, M.; Ishii, S.; Yoshida, K.; Fujii, N.; Ishiguro, M.; Satoh, H.; Okabe, S. Nitrate-dependent ferrous iron oxidation by anaerobic ammonium oxidation (anammox) bacteria. Appl. Environ. Microbiol. 2013, 79, 4087–4093. [Google Scholar] [CrossRef] [PubMed]
- Brons, H.J.; Hagen, W.R.; Zehnder, A.J.B. Ferrous iron dependent nitric oxide production in nitrate reducing cultures of Escherichia coli. Arch. Microbiol. 1991, 155, 341–347. [Google Scholar] [CrossRef] [PubMed]






© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, F.; Salgado, V.; Van der Hoek, J.P.; Van Halem, D. Bromate Reduction by Iron(II) during Managed Aquifer Recharge: A Laboratory-Scale Study. Water 2018, 10, 370. https://doi.org/10.3390/w10040370
Wang F, Salgado V, Van der Hoek JP, Van Halem D. Bromate Reduction by Iron(II) during Managed Aquifer Recharge: A Laboratory-Scale Study. Water. 2018; 10(4):370. https://doi.org/10.3390/w10040370
Chicago/Turabian StyleWang, Feifei, Vanida Salgado, Jan Peter Van der Hoek, and Doris Van Halem. 2018. "Bromate Reduction by Iron(II) during Managed Aquifer Recharge: A Laboratory-Scale Study" Water 10, no. 4: 370. https://doi.org/10.3390/w10040370
APA StyleWang, F., Salgado, V., Van der Hoek, J. P., & Van Halem, D. (2018). Bromate Reduction by Iron(II) during Managed Aquifer Recharge: A Laboratory-Scale Study. Water, 10(4), 370. https://doi.org/10.3390/w10040370






