Diversity and Structure of the Tychoplankton Diatom Community in the Limnocrene Spring Zelenci (Slovenia) in Relation to Environmental Factors
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Sampling and Laboratory Analyses
2.3. Ecological Groups of Diatoms
2.4. Data Analyses
3. Results
3.1. Abiotic Factors
3.2. Diatom Species Richness
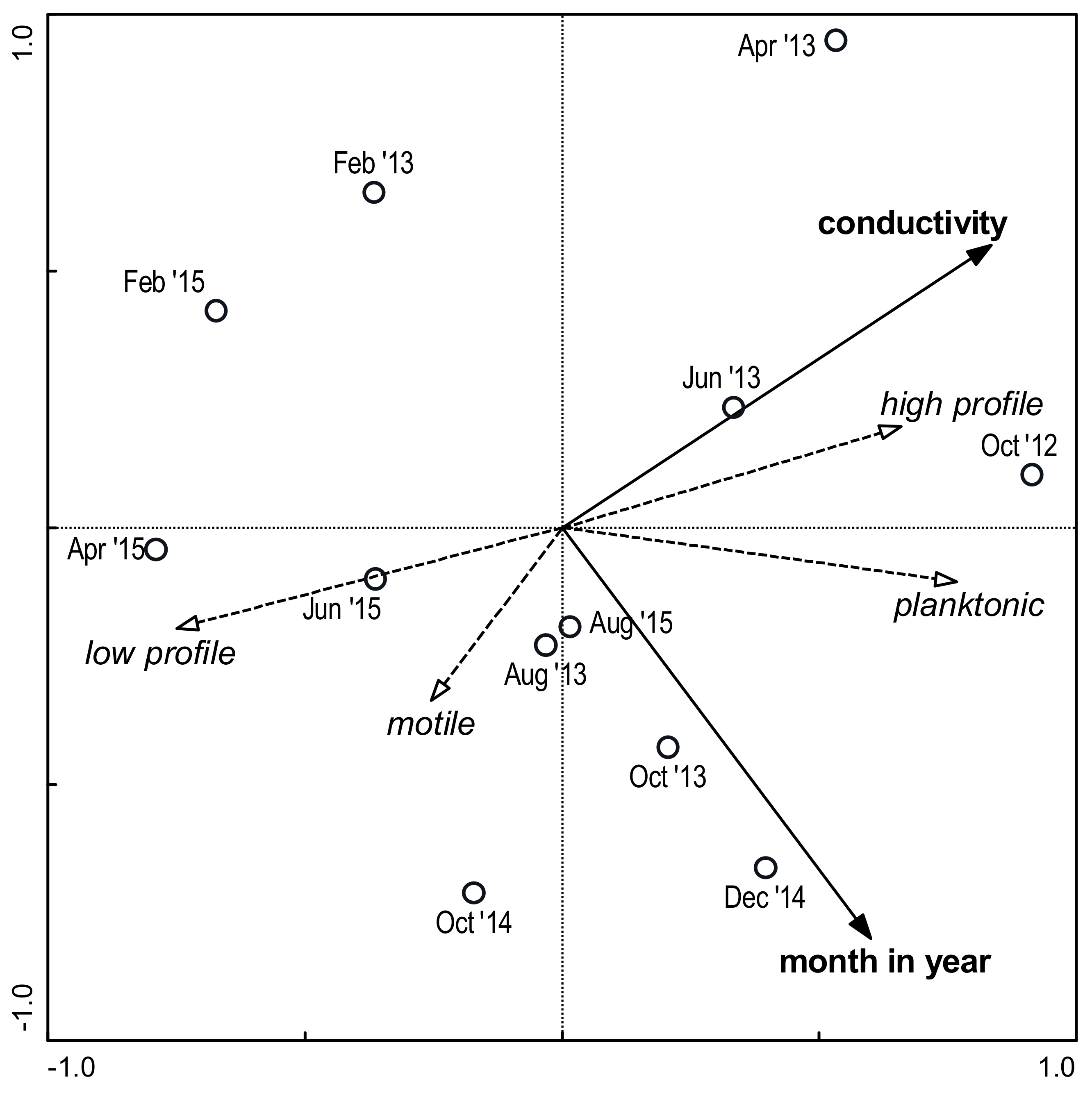
3.3. Environmental Factors and Species Composition
4. Discussion
4.1. Abiotic Factors
4.2. Diversity of the Tychoplanktonic Community
4.3. Environmental Factors and Species Composition
4.4. Correlations between Factors and Ecological Groups
4.5. Biodiversity Conservation and Water Resource Management
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stendera, S.; Adrian, R.; Bonada, N.; Cañedo-Argüells, M.; Hugueny, B.; Januschke, K.; Pletterbauer, F.; Hering, D. Drivers and stressors of freshwater biodiversity patterns across different ecosystems and scales: A review. Hydrobiologia 2012, 696, 1–28. [Google Scholar] [CrossRef]
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.I.; Knowler, D.J.; Leveque, C.; Naiman, R.J.; Prieur-Richard, A.H.; Soto, D.; Stiassny, M.L.J.; et al. Freshwater biodiversity: Importance, threats, status and conservation challenges. Biol. Rev. 2006, 81, 163–182. [Google Scholar] [CrossRef] [PubMed]
- Cantonati, M.; Gerecke, R.; Bertuzzi, E. Springs of the Alps—Sensitive ecosystems to environmental change: From biodiversity assessments to long-term studies. Hydrobiologia 2006, 562, 59–96. [Google Scholar] [CrossRef]
- Cantonati, M.; Angeli, N.; Bertuzzi, E.; Spitale, D.; Lange-Bertalot, H. Diatoms in springs of the Alps: Spring types, environmental determinants, and substratum. Freshw. Sci. 2012, 31, 499–524. [Google Scholar] [CrossRef]
- Mogna, M.; Cantonati, M.; Andreucci, F.; Angeli, N.; Berta, G.; Miserere, L. Diatom communities and vegetation of springs in the south-western Alps. Acta Bot. Croat. 2015, 74, 265–285. [Google Scholar] [CrossRef]
- Kløve, B.; Ala-Aho, P.; Bertrand, G.; Boukalova, Z.; Ertürk, A.; Goldscheider, N.; Ilmonen, J.; Karakaya, N.; Kupfersberger, H.; Kvoerner, J.; et al. Groundwater dependent ecosystems. Part I: Hydroecological status and trends. Environ. Sci. Policy 2011, 14, 770–781. [Google Scholar] [CrossRef]
- Cantonati, M.; Segadelli, S.; Ogata, K.; Tran, H.; Sanders, D.; Gerecke, R.; Rott, E.; Filippini, M.; Gargini, A.; Celico, F. A global review on ambient Limestone-Precipitating Springs (LPS): Hydrogeological setting, ecology, and conservation. Sci. Total Environ. 2016, 568, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Cantonati, M.; Lange-Bertalot, H.; Scalfi, A.; Angeli, N. Cymbella tridentina sp. nov. (Bacillariophyta), a crenophilous diatom from carbonate springs of the Alps. J. N. Am. Benthol. Soc. 2010, 29, 775–788. [Google Scholar] [CrossRef]
- Soininen, J. Environmental and spatial control of freshwater diatoms—A review. Diatom Res. 2007, 22, 473–490. [Google Scholar] [CrossRef]
- Cantonati, M.; Spitale, D. The role of environmental variables in structuring epiphytic and epilithic diatom assemblages in springs and streams of the Dolomiti Bellunesi national park (south-eastern Alps). Fundam. Appl. Limnol. 2009, 174, 117–133. [Google Scholar] [CrossRef]
- Cantonati, M. Diatom communities of springs in the Southern Alps. Diatom Res. 1998, 13, 201–220. [Google Scholar] [CrossRef]
- Menegalija, T.; Kosi, G. Distribution of diatoms in springs in the Julian Alps (NW Slovenia). Nat. Slov. 2008, 10, 21–37. [Google Scholar]
- Cantonati, M.; Lange-Bertalot, H. Diatom biodiversity of springs in the Berchtesgaden National Park (north-eastern Alps, Germany), with the ecological and morphological characterization of two species new to science. Diatom Res. 2010, 25, 251–280. [Google Scholar] [CrossRef]
- Gesierich, D.; Kofler, W. Epilithic diatoms from rheocrene springs in the eastern Alps (Vorarlberg, Austria). Diatom Res. 2010, 25, 43–66. [Google Scholar] [CrossRef]
- Taxböck, L.; Linder, H.P.; Cantonati, M. To What Extent Are Swiss Springs Refugial Habitats for Sensitive and Endangered Diatom Taxa? Water 2017, 9, 967. [Google Scholar] [CrossRef]
- Sabater, S.; Roca, J.R. Ecological and biogeographical aspects of diatom distribution in Pyrenean springs. Br. Phycol. J. 1992, 27, 203–213. [Google Scholar] [CrossRef]
- Aboal, M.; Puig, M.A.; Prefasi, M. Diatom assemblages in springs in Castellón province, Eastern Spain. Algological Stud. 1998, 90, 79–95. [Google Scholar]
- Werum, M.; Lange-Bertalot, H. Diatoms in Springs from Central Europe and Elsewhere under the Influence of Hydrogeology and Anthropogenic Impacts; A.R.G. Gantner Verlag K.G.: Ruggell, Liechtenstein, 2004; p. 480. [Google Scholar]
- Koh, C.; Khim, J.S.; Araki, H.; Yamanishi, H.; Mogi, H.; Koga, K. Tidal resuspension of microphytobenthic chlorophyll a in a Nanura mudflat, Saga, Arieke Sea, Japan: Flood-ebb and spring-neap variations. Mar. Ecol. Pror. Ser. 2006, 312, 85–100. [Google Scholar] [CrossRef]
- Gregori, J. Zelenci—Natural Reserve; Janez Gregori: Ljubljana, Slovenia, 1994; pp. 1–120. [Google Scholar]
- Quiroga, M.V.; Unrein, F.; Gonzalez Garraza, G.; Kuppers, G.; Lombardo, R.; Marinone, M.C.; Menu Marque, S.; Vinocur, A.; Mataloni, G. The plankton communities from peat bog pools: Structure, temporal variation and environmental factors. J. Plankton Res. 2013, 35, 1234–1253. [Google Scholar] [CrossRef]
- Hofmann, G.; Werum, M.; Lange-Bertalot, H. Diatomeen im Süsswasser-Benthos von Mitteleuropa. Bestimmungsflora Kieselalgen für die Ökologische Praxis. Über 700 der Häufigsten Arten und Ihre Ökologie; A.R.G. Gantner Verlag K.G.: Ruggell, Liechtenstein, 2011; p. 908. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae. Teil 1–4; Gustav Fischer Verlag: Stuttgart, Germany; New York, NY, USA, 1991. [Google Scholar]
- Passy, S.I. Diatom ecological guilds display distinct and predictable behavior along nutrient and disturbance gradients in running waters. Aquat. Bot. 2007, 86, 171–178. [Google Scholar] [CrossRef]
- Rimet, F.; Bouchez, A. Life-forms, cell-sizes and ecological guilds of diatoms in European rivers. Knowl. Manag. Aquat. Ecosyst. 2012, 406, 1–12. [Google Scholar] [CrossRef]
- Stenger-Kovács, C.; Lengyel, E.; Crossetti, L.O.; Üveges, V.; Padisák, J. Diatom ecological guilds as indicators of temporally changing stressors and disturbances in the small Torna-stream, Hungary. Ecol. Indic. 2013, 24, 138–147. [Google Scholar] [CrossRef]
- Körner, C. Scaling from species to vegetation: The usefulness of functional groups. In Biodiversity and Ecosystem Function; Schulze, E.D., Mooney, H.A., Eds.; Springer: Berlin, Germany, 1994; pp. 117–140. [Google Scholar]
- Grime, J.P. Plant Strategies, Vegetation Processes and Ecosystem Properties, 2nd ed.; Wiley: Chichester, UK, 2001. [Google Scholar]
- Tapolczai, K.; Bouchez, Á.; Stenger-Kovács, C.; Padisák, J.; Rimet, F. Trait-based ecological classifications for benthic algae: Review and perspectives. Hydrobiologia 2016, 776, 1–17. [Google Scholar] [CrossRef]
- Lange, K.; Liess, A.; Piggott, J.J.; Townsend, C.R.; Matthaei, C.D. Light, nutrients and grazing interact to determine stream diatom community composition and functional group structure: Diatom responses to light, nutrients and grazing. Freshw. Biol. 2011, 56, 264–278. [Google Scholar] [CrossRef]
- Lengyel, E.; Padisák, J.; Stenger-Kovács, C. Establishment of equilibrium states and effect of disturbances on benthic diatom assemblages of the Torna-stream, Hungary. Hydrobiologia 2015, 750, 43–56. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F.; Verdonschot, P.F.M. Canonical correspondence analysis and related multivariate methods in aquatic ecology. Aquat. Sci. 1995, 57, 255–289. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F.; Šmilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5); Microcomputer Power: Ithaca, NY, USA, 2002. [Google Scholar]
- Hudon, C.; Gagnon, P.; Amyot, J.-P.; Letourneau, G.; Jean, M.; Plant, C.; Rioux, D.; Deschenes, M. Historical changes in herbaceous wetland distribution induced by hydrological conditions in Lake Saint-Pierre (St. Lawrence River, Quebec, Canada). Hydrobiologia 2005, 539, 205–224. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Gerecke, R.; Cantonati, M.; Spitale, D.; Stur, E.; Wiedenbrug, S. The challenges of long-term ecological research in spring in the northern and southern Alps: Indicator groups, habitat diversity, and medium-term change. J. Limnol. 2011, 70, 168–187. [Google Scholar] [CrossRef]
- Van Dam, H.; Mertens, A.; Sinkeldam, J. A coded checklist and ecological indicator values of freshwater diatoms from the Netherlands. Neth. J. Aquat. Ecol. 1994, 28, 117–133. [Google Scholar]
- Giller, P.; Malmqvist, B. The Biology of Streams and Rivers; Oxford University Press: New York, NY, USA, 1998; pp. 1–296. [Google Scholar]
- Smith, H.; Wood, P.J. Flow permanence and macroinvertebrate community variability in limestone spring systems. Hydrobiologia 2002, 487, 45–58. [Google Scholar] [CrossRef]
- Falasco, E.; Bona, F. Diatom community biodiversity in an Alpine protected area: A study in the Maritime Alps Natural Park. J. Limnol. 2011, 70, 157–167. [Google Scholar] [CrossRef]
- Cantonati, M.; Ortler, K. Using spring biota of pristine mountain areas for long-term monitoring. Hydrology, Water Resources and Ecology in Headwaters. IAHS Publ. 1998, 248, 379–385. [Google Scholar]
- Maes, J.; Paracchini, M.L.; Zulian, G.; Dunbar, M.B.; Alkemade, R. Synergies and trade-offs between ecosystem service supply, biodiversity, and habitat conservation status in Europe. Biol. Conserv. 2012, 155, 1–12. [Google Scholar] [CrossRef]
- Grime, J.P. Competitive exclusion in herbaceous vegetation. Nature 1973, 242, 344–347. [Google Scholar] [CrossRef]
- Penning, W.; Mjelde, M.; Dudley, B.; Hellsten, S.; Hanganu, J.; Kolada, A.; Van Den Berg, M.; Poikane, S.; Phillips, G.; Willby, N.; et al. Classifying aquatic macrophytes as indicators of eutrophication in European lakes. Aquat. Ecol. 2008, 42, 237–251. [Google Scholar] [CrossRef]
- Toman, M.J.; Grošelj, A.M.; Zelnik, I. The influence of selected factors on the distribution of epilithic diatoms in a torrential river the Kamniška Bistrica (Slovenia). Acta Bot. Croat. 2014, 73, 447–463. [Google Scholar] [CrossRef]
- Cantonati, M.; Pipp, E. Longitudinal and seasonal differentiation of epilithic diatom communities in the uppermost sections of two mountain spring-fed streams. Verh. Int. Ver. Limnol. 2000, 27, 1591–1595. [Google Scholar] [CrossRef]
- B-Béres, V.; Török, P.; Kókai, Z.; Lukács, Á.; Krasznai, E.T.; Tóthmérész, B.; Bácsi, I. Ecological background of diatom functional groups: Comparability of classification systems. Ecol. Indic. 2017, 82, 183–188. [Google Scholar] [CrossRef]
- Porter, S.D.; Mueller, D.K.; Spahr, N.E.; Munn, M.D.; Dubrovsky, N.M. Efficacy of algal metrics for assessing nutrient and organic enrichment in flowing waters. Freshw. Biol. 2008, 53, 1036–1054. [Google Scholar] [CrossRef]
- Allan, J.D.; Castillo, M.M. Stream Ecology, 2nd ed.; Springer: Dordrecht, The Netherlands, 2007; p. 436. [Google Scholar]
- Smucker, N.J.; Vis, M.L. Using diatoms to assess human impacts on streams benefits from multiple-habitat sampling. Hydrobiologia 2010, 654, 93–109. [Google Scholar] [CrossRef]
- Jones, J.I.; Duerdoth, C.P.; Collins, A.L.; Sear, D.A. Interactions between diatoms and fine sediment. Hydrol. Process. 2014, 28, 1226–1237. [Google Scholar] [CrossRef]
- Balanč, T. Diversity of Diatom Communities in Different Habitats of the Limnocrene Spring Zelenci. Master’s Thesis, University of Ljubljana, Biotechnical Faculty, Ljubljana, Slovenia, December 2016. [Google Scholar]
- Grillas, P.; Gauthier, P.; Yaverkovski, N.; Perennou, C. Mediterranean Temporary Pools 1; Station Biologique de la Tour du Valat: Arles, France, 2004. [Google Scholar]
- URS Infrastructure & Environment UK Limited. South Stream Offshore Pipeline—Bulgarian Sector. Espoo Notification Document, URS-EIA-REP-201147, Published on 31 January 2013. Available online: http://www.moew.government.bg/static/media/ups/tiny/file/Industry/EIA/Transgranichni_EIA_procedures/notifkatsia_Morski_gazoprovod_en.pdf (accessed on 19 March 2018).

| Min | Max | Average | |
|---|---|---|---|
| pH | 7.2 | 7.8 | 7.6 |
| T [°C] | 5.3 | 9.6 | 7.4 |
| conductivity [µS/cm] | 234 | 290 | 257 |
| O2 saturation [%] | 80 | 103 | 88 |
| O2 [mg/L] | 9.1 | 11.7 | 10 |
| PO43− [mg/L] | * | 0.12 | 0.027 |
| NH4+ [mg/L] | 0.016 | 0.817 | 0.334 |
| NO3− [mg/L] | 1.5 | 2.8 | 2.0 |
| water level [cm] | 65 | 118 | 89 |
| LP % | 30 | 72 | 51 |
| HP % | 12 | 39 | 19 |
| M % | 15 | 33 | 23 |
| PL % | 0 | 20 | 7 |
| LP [%] | HP [%] | M [%] | PL [%] | Nr. spp. | SWI | |
|---|---|---|---|---|---|---|
| pH | 0.099 | −0.017 | −0.253 | 0.261 | 0.458 | 0.230 |
| T [°C] | 0.168 | −0.031 | 0.000 | 0.000 | −0.142 | −0.351 |
| conductivity [µS/cm] | −0.545 | 0.504 | −0.171 | 0.465 | 0.250 | 0.242 |
| O2 saturation [%] | 0.504 | −0.400 | 0.125 | −0.291 | −0.110 | −0.107 |
| O2 [mg/L] | 0.326 | −0.313 | 0.000 | 0.082 | 0.192 | 0.295 |
| PO43− [mg/L] | −0.031 | 0.016 | −0.016 | −0.281 | −0.645 | −0.500 |
| NH4+ [mg/L] | −0.064 | −0.016 | −0.146 | 0.504 | 0.377 | 0.191 |
| NO3− [mg/L] | 0.333 | −0.443 | 0.450 | −0.112 | −0.250 | −0.030 |
| potential irradiance | 0.080 | 0.065 | −0.197 | 0.153 | 0.017 | −0.208 |
| month in the year | −0.286 | 0.176 | 0.049 | 0.403 | 0.164 | 0.413 |
| month of sampling | 0.333 | −0.351 | 0.171 | −0.208 | −0.375 | −0.273 |
| water level | −0.394 | 0.321 | −0.326 | 0.497 | 0.406 | 0.515 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zelnik, I.; Balanč, T.; Toman, M.J. Diversity and Structure of the Tychoplankton Diatom Community in the Limnocrene Spring Zelenci (Slovenia) in Relation to Environmental Factors. Water 2018, 10, 361. https://doi.org/10.3390/w10040361
Zelnik I, Balanč T, Toman MJ. Diversity and Structure of the Tychoplankton Diatom Community in the Limnocrene Spring Zelenci (Slovenia) in Relation to Environmental Factors. Water. 2018; 10(4):361. https://doi.org/10.3390/w10040361
Chicago/Turabian StyleZelnik, Igor, Tadeja Balanč, and Mihael J. Toman. 2018. "Diversity and Structure of the Tychoplankton Diatom Community in the Limnocrene Spring Zelenci (Slovenia) in Relation to Environmental Factors" Water 10, no. 4: 361. https://doi.org/10.3390/w10040361
APA StyleZelnik, I., Balanč, T., & Toman, M. J. (2018). Diversity and Structure of the Tychoplankton Diatom Community in the Limnocrene Spring Zelenci (Slovenia) in Relation to Environmental Factors. Water, 10(4), 361. https://doi.org/10.3390/w10040361






