The Brine Shrimp Artemia Survives in Diluted Water of Lake Bunyampaka, an Inland Saline Lake in Uganda
Abstract
:1. Introduction
2. Materials and Methods
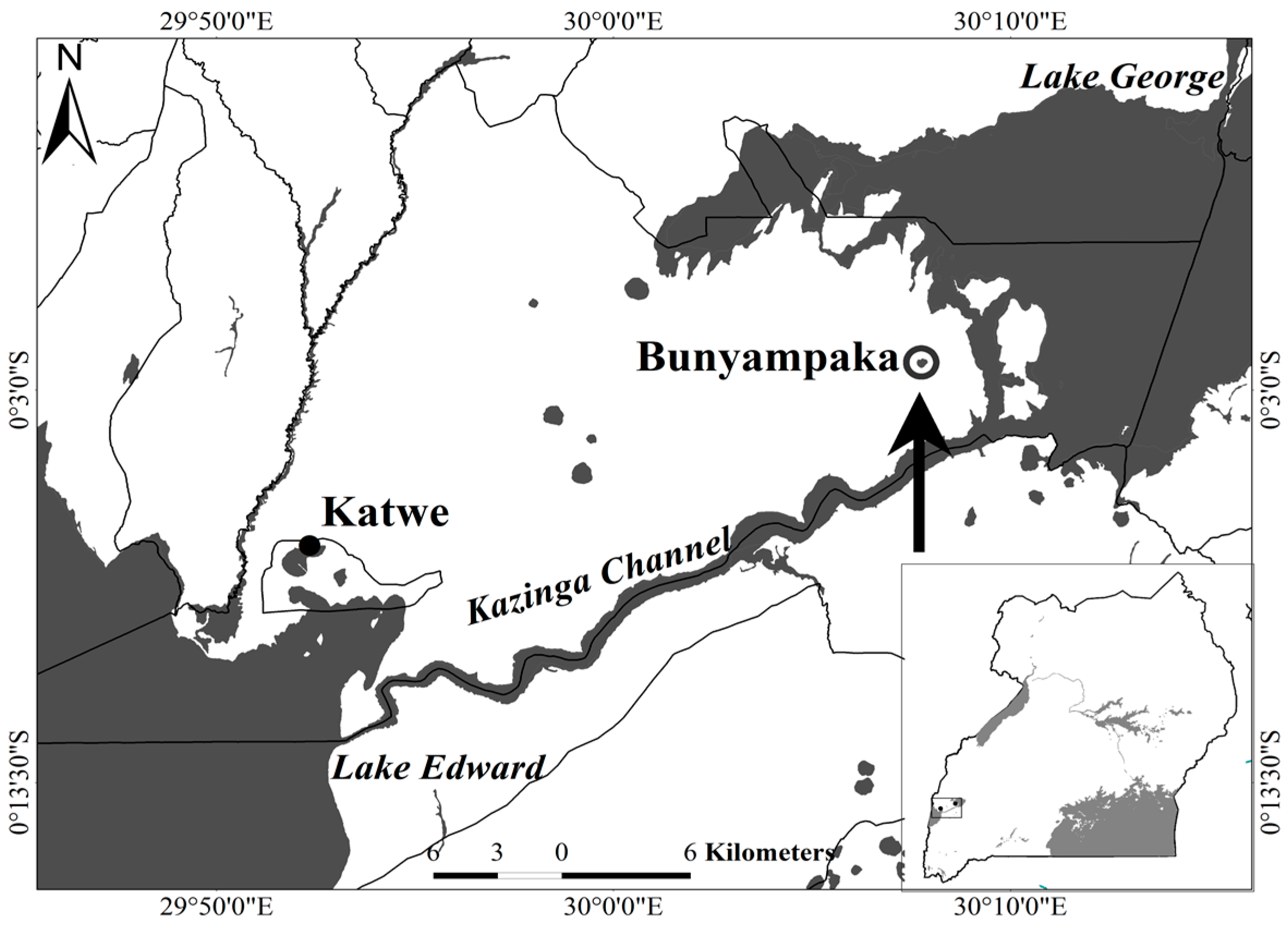
2.1. Saline Water Used
2.2. Experiment 1: Survival Experiment with Both GSL and VC Strains at 30 g L−1
2.3. Experiment 2: Survival and Growth Experiment with the VC Strain at 30 g L−1
2.4. Experiment 3: Reproduction Experiment with the VC Strain at 30 g L−1 and 50 g L−1
2.5. Statistical Analysis
3. Results
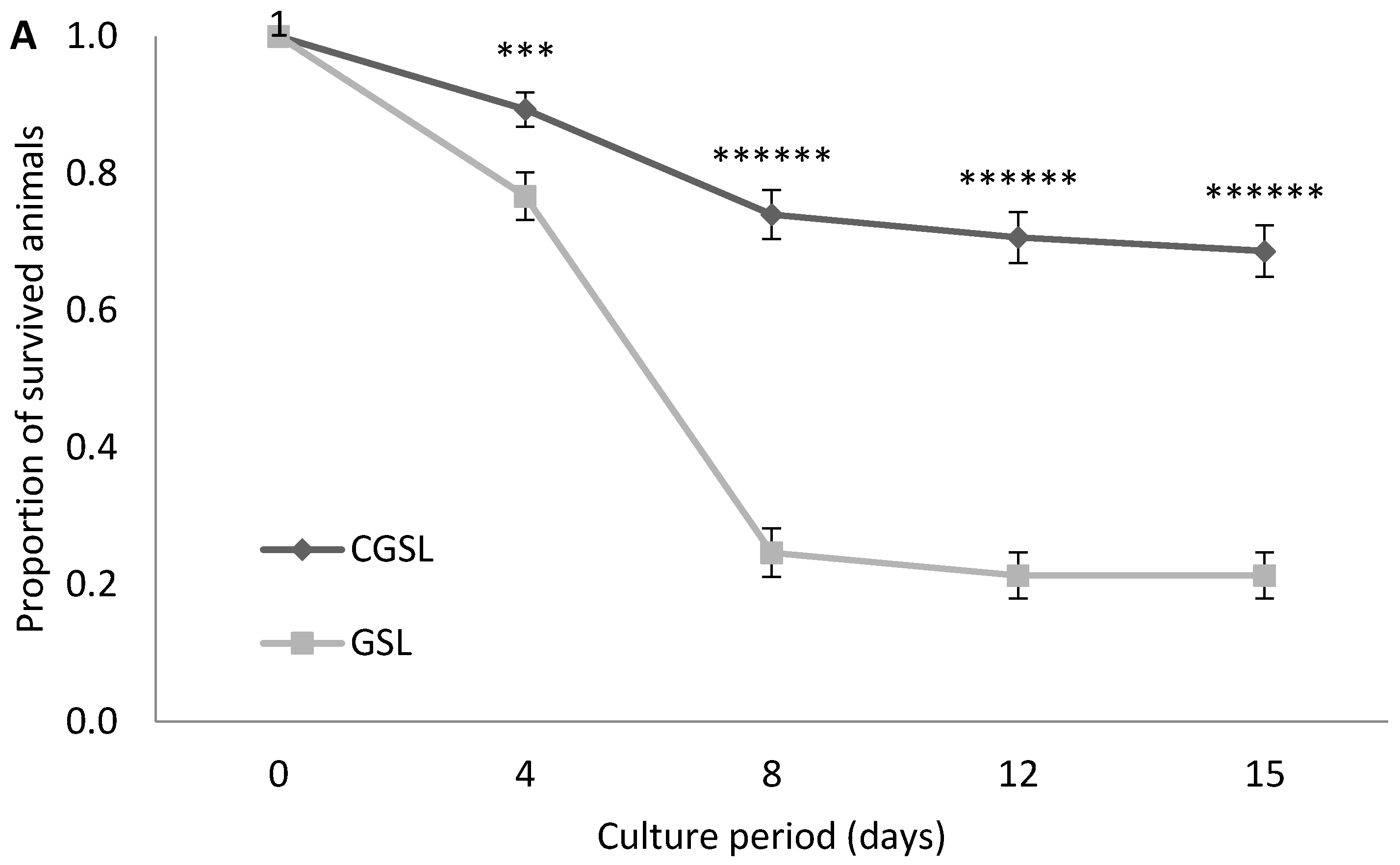
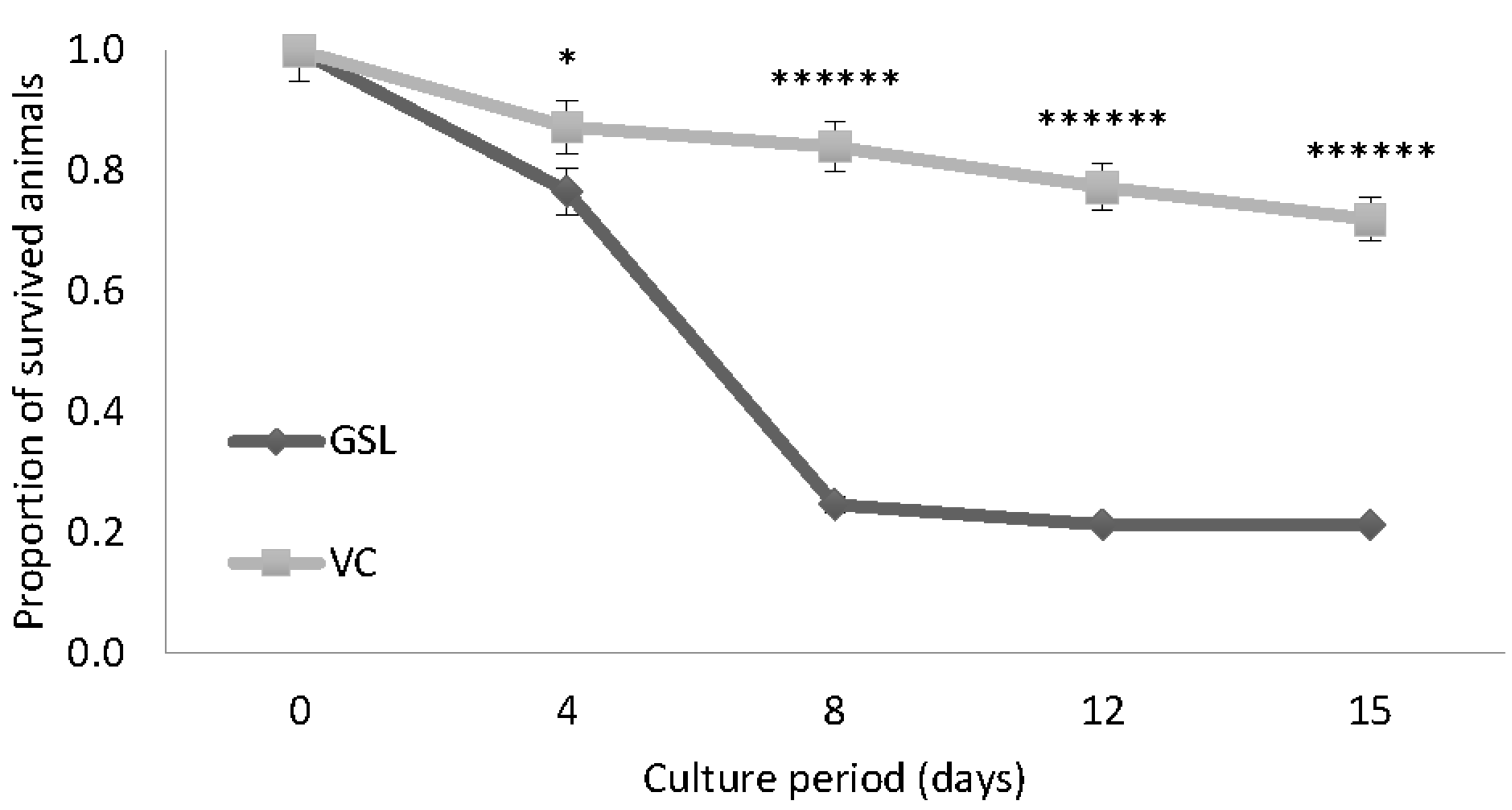
3.1. Experiment 1: Survival Experiment with Both GSL and VC Strains at 30 g L−1
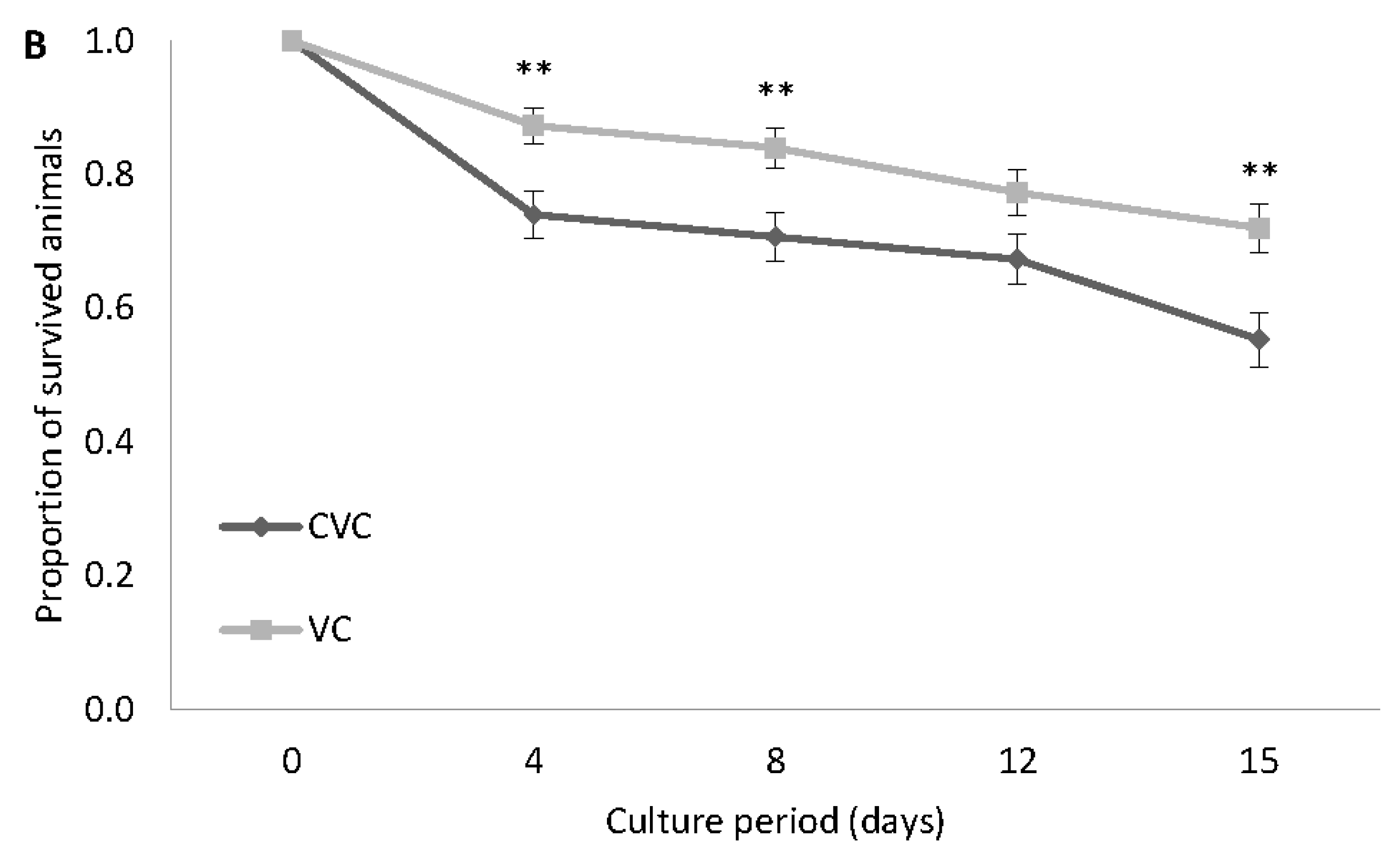
3.2. Experiment 2: Survival and Growth Experiment with the VC Strain at 30 g L−1
3.3. Experiment 3: Reproduction Experiment with the VC Strain at 30 g L−1 and 50 g L−1
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rutaisire, J.; Charo-Karisa, H.; Shoko, A.P.; Nyandat, B. Aquaculture for increased fish production in East Africa. Afr. J. Trop. Hydrobiol. Fish. 2009, 12, 74–79. [Google Scholar] [CrossRef]
- Ministry of Agriculture, Animal Industry and Fisheries (MAAIF). Agricultural Sector Development and Strategic Plan 2012/11–2014/15; MAAIF: Entebbe, Uganda, 2010.
- Walakira, J.; (National Fisheries Resources Research Institute, Jinja, Uganda). Personal communication, 2016.
- Sarker, S.D.; Hanaee, J.; Agh, N.; Hanaee, M.; Dalazar, A. Studies on the enrichment of Artemia urmiana cysts for improving fish food value. J. Dairy Sci. 2005, 120, 107–112. [Google Scholar]
- Van Stappen, G. Artemia. In Manual on the Production and Use of Live Food for Aquaculture; Lavens, P., Patrick Sorgeloos, P., Eds.; Food and Agriculture Organization (FAO): Rome, Italy, 1996; ISBN 9251039348. [Google Scholar]
- Hoa, N.V. Seasonal Farming of the Brine Shrimp Artemia Franciscana in Artisanal Salt Ponds in Vietnam: Effects of Temperature and Salinity. Ph.D. Thesis, Ghent University, Ghent, Belgium, 2002. [Google Scholar]
- Sorgeloos, P.; Hoa, D.N.; Wouters, R.; Wille, M.; Thanh, V.; Dong, T.K. A fresh-food maturation diet with an adequate HUFA composition for broodstock nutrition studies in black tiger shrimp Penaeus monodon (Fabricius, 1798). Aquaculture 2009, 297, 116–121. [Google Scholar]
- Toi, H.T.; Boeckx, P.; Sorgeloos, P.; Bossier, P.; Van Stappen, G. Bacteria contribute to Artemia nutrition in algae-limited conditions: A laboratory study. Aquaculture 2013, 388, 1–7. [Google Scholar] [CrossRef]
- Flüchter, J. Review of the present knowledge of rearing whitefish (Coregonidae) larvae. Aquaculture 1980, 19, 191–208. [Google Scholar] [CrossRef]
- Kräeuter, J.N.; Woods, C., III. Culture of striped bass and its hybrids: First feeding to six months. In Hybrid Striped Bass Culture: Status and Perspective; UNC Sea Grant Publication UNCSG-87-03; North Carolina State University: Raleigh, NC, USA, 1987; pp. 23–32. [Google Scholar]
- Bengtson, D.A.; Léger, P.; Sorgeloos, P. Use of Artemia as a food source for aquaculture. J. Artemia Biol. 1991, 11, 255–285. [Google Scholar]
- Guthrie, K.M.; Rust, M.B.; Langdon, C.J.; Barrows, F.T. Acceptability of various microparticulate diets to first-feeding walleye Stizostedion vitreum larvae. Aquac. Nutr. 2000, 6, 153–158. [Google Scholar] [CrossRef]
- Walakira, J.K.; Molnar, J.J.; Phelps, R.; Terhune, J. Culturing the African lungfish in Uganda: Effects of exogenous fish feed on growth performance in tanks. UJAS 2014, 15, 137–155. [Google Scholar]
- Soundarapanian, P.; Saravanakumar, G. Effect of different salinities on the survival and growth of Artemia Spp. Curr. Res. J. Biol. Sci. 2009, 1, 20–22. [Google Scholar]
- Lavens, P.; Sorgeloos, P. Manual on the Production and Use of Live Food for Aquaculture; Food and Agriculture Organization (FAO): Rome, Italy, 1996; ISBN 9251039348. [Google Scholar]
- Vanhaecke, P.; Tackaert, W.; Sorgeloos, P. The biogeography of Artemia: An updated review. J. Artemia Res. Appl. 1987, 1, 129–155. [Google Scholar]
- Xin, N.; Sun, J.; Zhang, B.; Triantaphyllidis, G.V.; Van Stappen, G.; Sorgeloos, P. International study on Artemia. LI. New survey of Artemia resources in the People’s Republic of China. Int. J. Salt Lake Res. 1994, 3, 105–112. [Google Scholar] [CrossRef]
- Triantaphyllidis, V.G.; Abatzopoulos, J.T.; Sorgeloos, P. Review of the biogeography of the genus Artemia (Crustacea, Anostraca). J. Biogeogr. 1998, 25, 213–226. [Google Scholar] [CrossRef]
- Browne, R.A.; Wanigasekera, G. Combined effects of salinity and temperature on survival and reproduction of five species of Artemia. J. Exp. Mar. Biol. Ecol. 2000, 244, 29–44. [Google Scholar] [CrossRef]
- Bowen, S.T.; Fogarino, E.A.; Hitchner, K.N.; Dana, G.L.; Chow, V.H.S.; Buoncristiani, M.R.; Carl, J.R. Ecological isolation in Artemia: Population differences in tolerance of anion concentrations. J. Crustac. Biol. 1985, 5, 106–129. [Google Scholar] [CrossRef]
- Bowen, S.T.; Buoncristiani, M.R.; Carl, J.R. Artemia habitats: Ion concentrations tolerated by one superspecies. Hydrobiologia 1988, 158, 201–214. [Google Scholar] [CrossRef]
- Asem, A.; Rastegar-Pouyani, N.; De Los Ríos-Escalante, P. The genus Artemia Leach, 1819 (Crustacea: Branchiopoda). I. True and false taxonomical descriptions. Submission article platform. Lat. Am. J. Aquat. Res. 2011, 38, 501–506. [Google Scholar] [CrossRef]
- Mwanja, M.; Rutaisire, J.; Ondhoro, C.; Ddungu, R.; Aruho, C. Current fish hatchery practices in Uganda: The potential for future investment. IJFAS 2015, 2, 224–232. [Google Scholar]
- Nkambo, M.; Bugenyi, F.W.; Naluwayilo, J.; Nayiga, S. Artemia occurrence, salinity and ionic rates in saline crater lakes of western Uganda. IJE 2015, 5, 59–65. [Google Scholar] [CrossRef]
- Nkambo, M.; (National Fisheries Resources Research Institute, Jinja, Uganda). Personal communication, 2016.
- Coutteau, P. Micro-algae. In Manual on the Production and Use of Live Food for Aquaculture; Lavens, P., Sorgeloos, P., Eds.; Food and Agriculture Organization (FAO): Rome, Italy, 1996; ISBN 9251039348. [Google Scholar]
- Dana, L.G.; Lenz, H.P. Effects of increasing salinity on an Artemia population from Mono Lake, California. Oecologia 1986, 68, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Van Stappen, G. Production, Harvest and Processing of Artemia from Natural Lakes. In Live Feeds in Marine Aquaculture; Støttrup, J.G., McEvoy, L.A., Eds.; Blackwell Publishing: Oxford, UK, 2003; pp. 122–144. ISBN 9780470995136. [Google Scholar]
- Van Stappen, G.; Sui, L.; Xin, N.; Sorgeloos, P. Characterisation of high-altitude Artemia populations from the Qinghai-Tibet Plateau, PR China. Hydrobiologia 2003, 500, 179–192. [Google Scholar] [CrossRef]
- Persoone, G.; Sorgeloos, P. General aspects of the ecology and biogeography of Artemia. In The Brine Shrimp Artemia Ecology, Culturing, Use in Aquaculture; Persoone, G., Sorgeloos, P., Roels, O., Jaspers, E., Eds.; Universal Press: Wetteren, Belgium, 1980; Volume 3, pp. 3–24. [Google Scholar]
- Cole, G.A.; Brown, R.J. The chemistry of Artemia habitats. Ecology 1967, 48, 858–861. [Google Scholar] [CrossRef]
- Bowen, S.T.; Durkin, J.P.; Sterling, G.; Clark, L.S. Artemia hemoglobins: Genetic variation in parthenogenetic and zygogenetic populations. Biol. Bull. 1978, 155, 273–287. [Google Scholar] [CrossRef]
- Kappas, I.; Abatzopoulos, T.J.; Van Hoa, N.; Sorgeloos, P.; Beardmore, J.A. Genetic and reproductive differentiation of Artemia franciscana in a new environment. Mar. Biol. 2004, 146, 103–117. [Google Scholar] [CrossRef]
- Clegg, J.S.; Jackson, S.A.; Hoa, N.V. Thermal resistance, developmental rate and heat shock proteins in Artemia franciscana, from San Francisco Bay and southern Vietnam. J. Exp. Mar. Biol. Ecol. 2000, 252, 85–90. [Google Scholar] [CrossRef]
- Clegg, J.S.; Hoa, N.V.; Sorgeloos, P. Thermal tolerance and heat shock proteins in encysted embryos of Artemia from widely different thermal habitats. Hydrobiologia 2001, 466, 221–229. [Google Scholar] [CrossRef]

| Parameter | Concentration | Method |
|---|---|---|
| Sodium (Na+) | 31,000 mg L−1 | ICP-OES |
| Potassium (K+) | 115 mg L−1 | ICP-OES |
| Calcium (Ca2+) | 1900 mg L−1 | ICP-OES |
| Magnesium (Mg2+) | 80 mg L−1 | ICP-OES |
| Ammonium (NH4+-N) | 12 mg L−1 N | Merck test kit: 114,752 |
| Nitrite (NO2−-N) | 0.13 mg L−1 N | Merck test kit: 114,776 |
| Nitrate (NO3−-N) | 1.2 mg L−1 N | Merck test kit: 114,773 |
| Phosphorus (PO43−-P) | 32 mg L−1 P | Merck test kit: 114,848 |
| Chloride (Cl−) | 35,700 mg L−1 Cl− | Merck test kit: 114,897 |
| Sulphate (SO42−) | 7500 mg L−1 SO42− | Merck-test kit: 114,548 |
| Characteristics | Lake Bunyampaka Water | Control | |
|---|---|---|---|
| Salinity | 30 g L−1 | 50 g L−1 | 35 g L−1 |
| Age at first offspring | 28 ± 0.4 ab | 35.5 ± 1.6 a | 23.7 ± 1.1 b |
| Brood size | 4.2 ± 0.6 a | 2.9 ± 0.9 a | 4.5 ± 0.5 a |
| Inter-brood interval (days) | 3 ± 0.4 a | 3.5 ± 0.3 a | 3.4 ± 0.3 a |
| Number of offspring/female/day | 18 ±1 ab | 10 ± 4 b | 28 ± 4 a |
| Total offspring production | 4574 ± 243 ab | 2650 ± 871 b | 6952 ± 555 a |
| Percentage offspring produced as cysts | 21 ± 6.8 a | 16 ± 5.1 a | 20.6 ± 10.3 a |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sserwadda, M.; Kagambe, E.; Van Stappen, G. The Brine Shrimp Artemia Survives in Diluted Water of Lake Bunyampaka, an Inland Saline Lake in Uganda. Water 2018, 10, 189. https://doi.org/10.3390/w10020189
Sserwadda M, Kagambe E, Van Stappen G. The Brine Shrimp Artemia Survives in Diluted Water of Lake Bunyampaka, an Inland Saline Lake in Uganda. Water. 2018; 10(2):189. https://doi.org/10.3390/w10020189
Chicago/Turabian StyleSserwadda, Martin, Edmond Kagambe, and Gilbert Van Stappen. 2018. "The Brine Shrimp Artemia Survives in Diluted Water of Lake Bunyampaka, an Inland Saline Lake in Uganda" Water 10, no. 2: 189. https://doi.org/10.3390/w10020189
APA StyleSserwadda, M., Kagambe, E., & Van Stappen, G. (2018). The Brine Shrimp Artemia Survives in Diluted Water of Lake Bunyampaka, an Inland Saline Lake in Uganda. Water, 10(2), 189. https://doi.org/10.3390/w10020189





