Groundwater Overexploitation and Seawater Intrusion in Coastal Areas of Arid and Semi-Arid Regions
Abstract
1. Overview Saltwater Intrusion into Coastal Aquifers
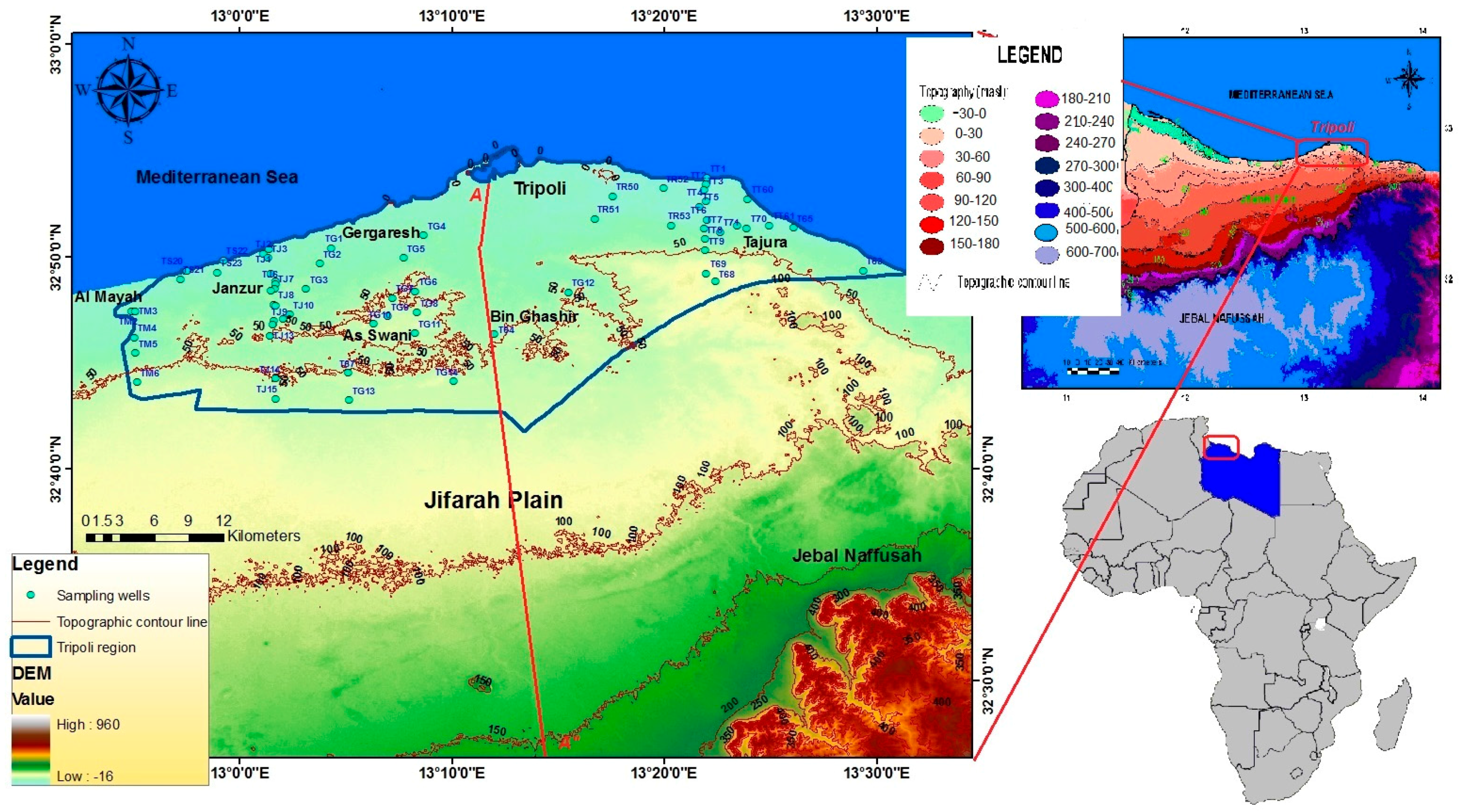
2. Introduction to the Study Area
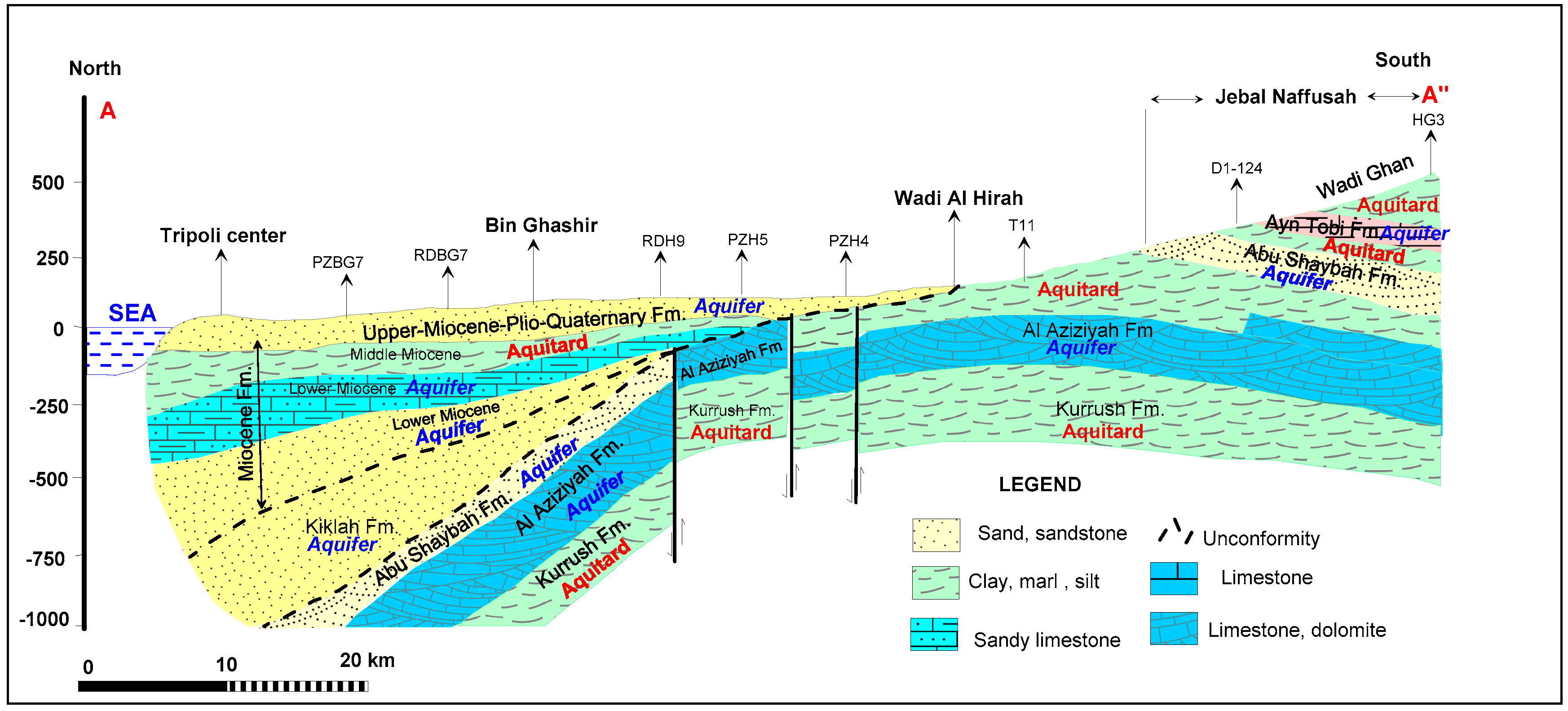
Geological and Hydrogeological Setting
3. Methodology
3.1. Sampling and Analytical Methods
3.2. Hydrochemical Evaluation Methods
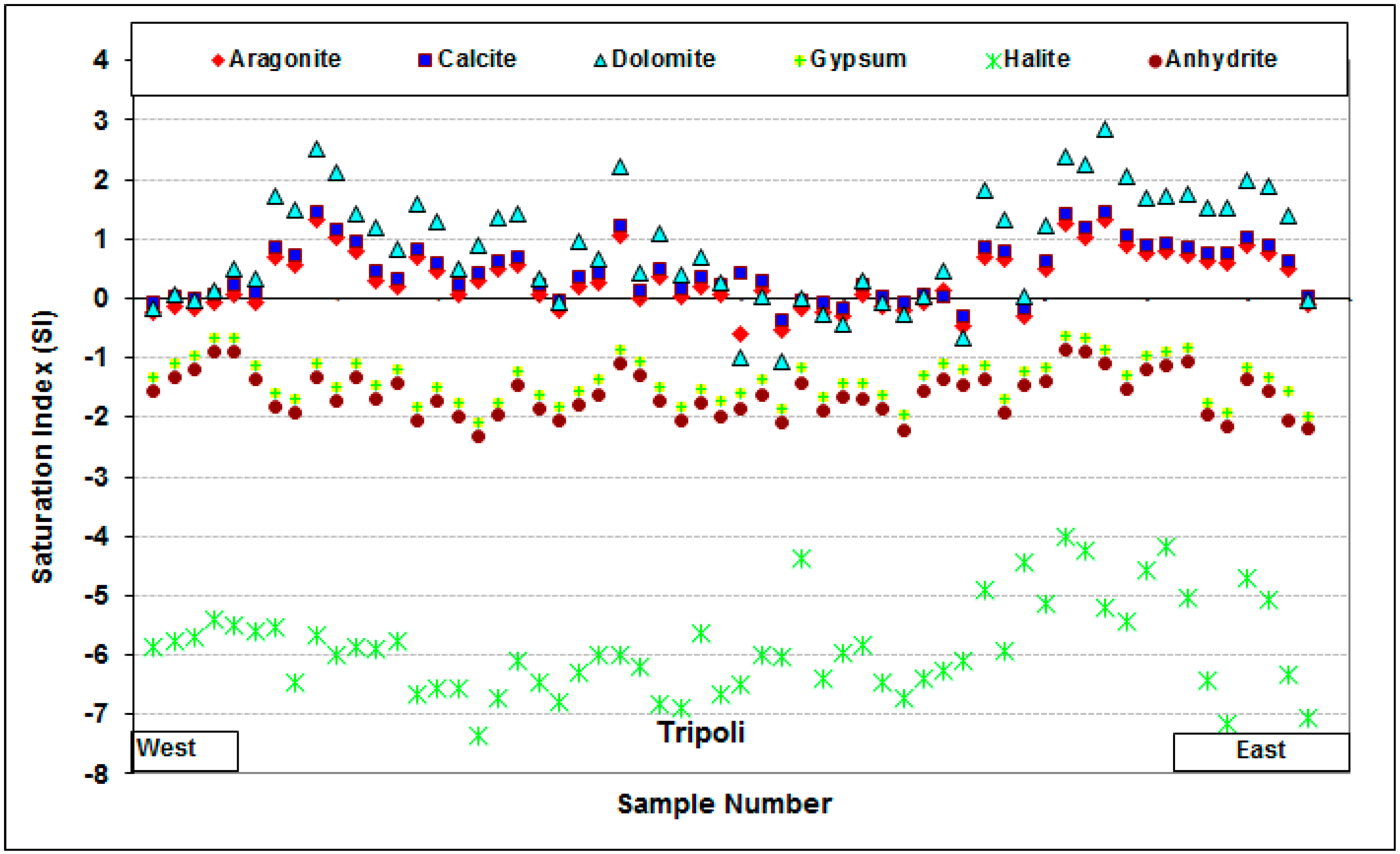
3.2.1. Saturation Indices
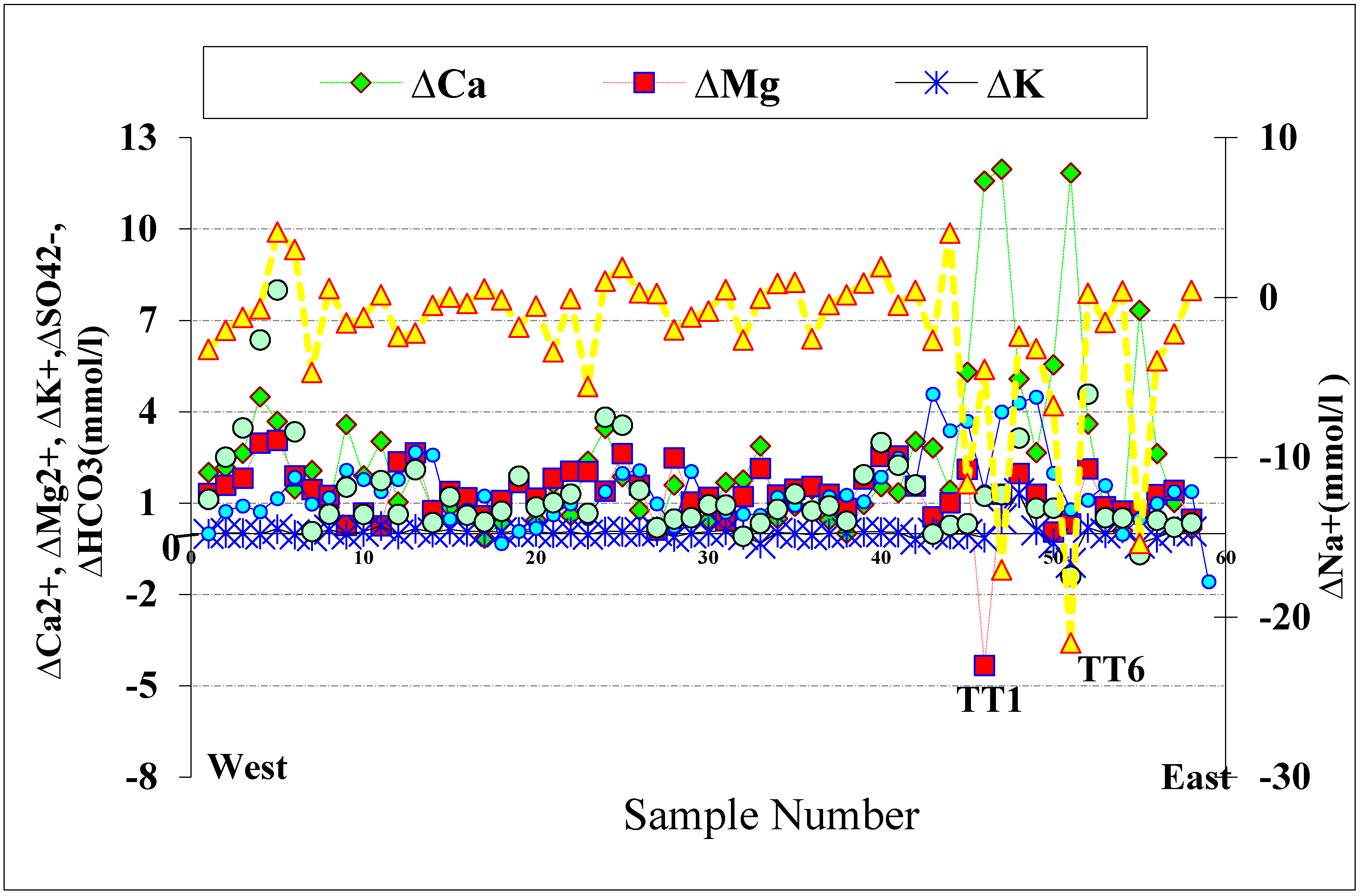
3.2.2. Ion Deviation from Conservative Freshwater/Seawater Mixing
- mCl−,sample = the concentration of Cl− in the sample expressed in mmol/L;
- mCl−,fresh = the concentration of Cl− in the freshwater expressed in mmol/L; and
- mCl−,sea = Cl− concentration in the seawater end member in mmol/L (for Mediterranean Seawater (possible end member), mCl−,sea = 645 mmol/L; Da’as and Walraevens, 2010).
3.2.3. Stuyfzand Classification
- – = often pointing at a saltwater intrusion;
- + = often pointing at a freshwater encroachment; and
- 0 = often pointing at an equilibrium.
4. Results and Discussion
4.1. Water Level and Hydrodynamics
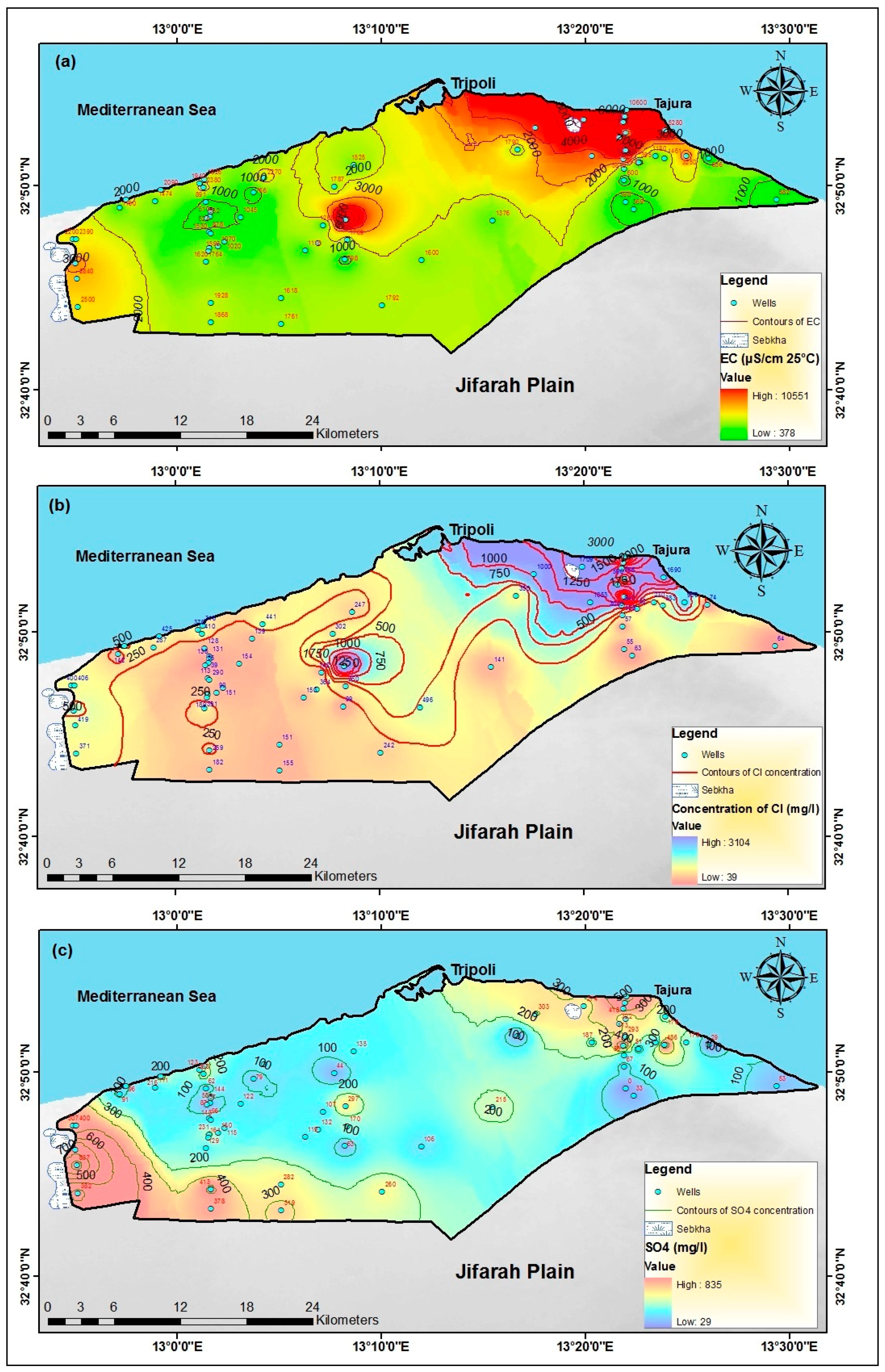
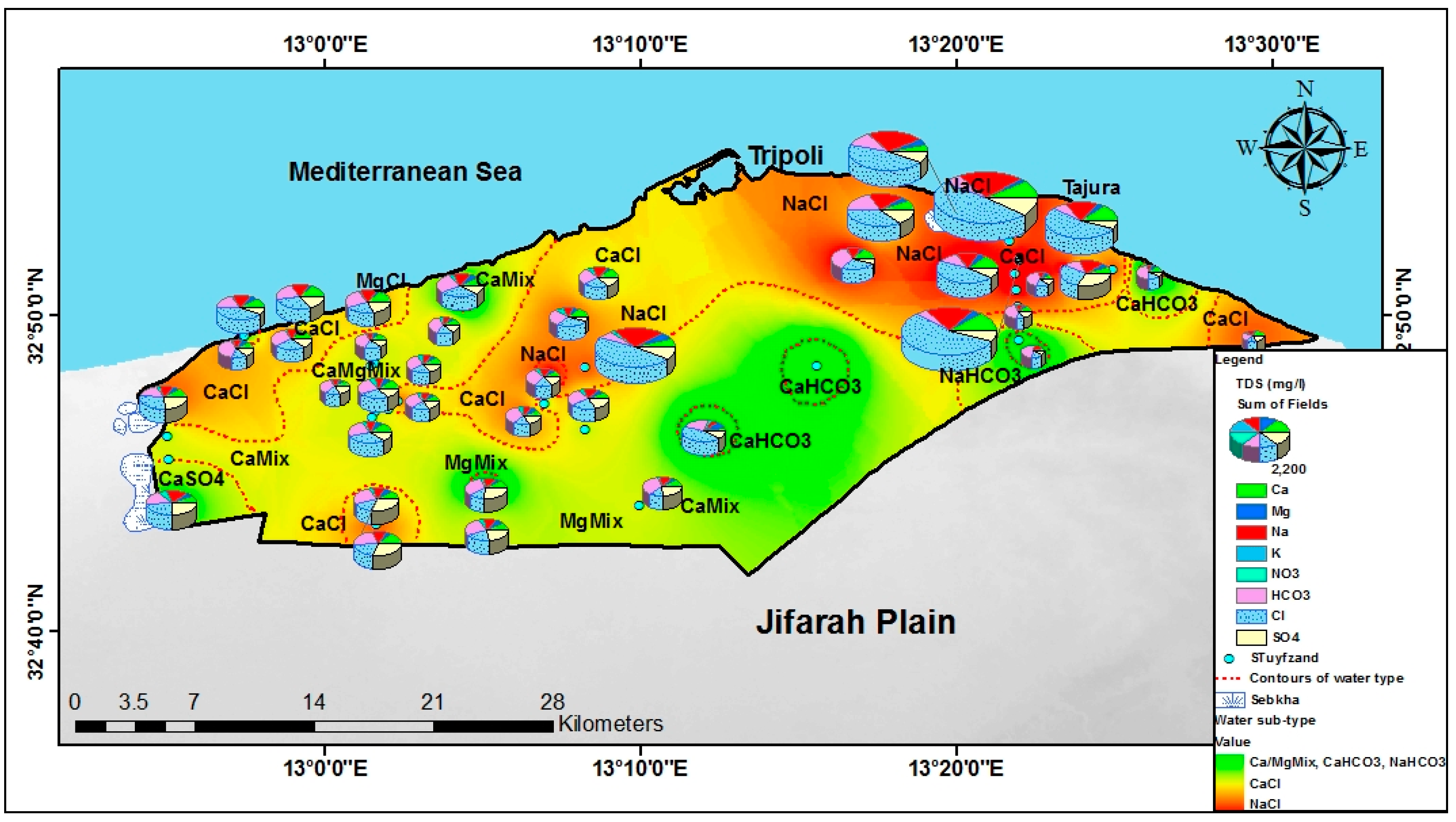
4.2. Major Hydrochemical Parameters
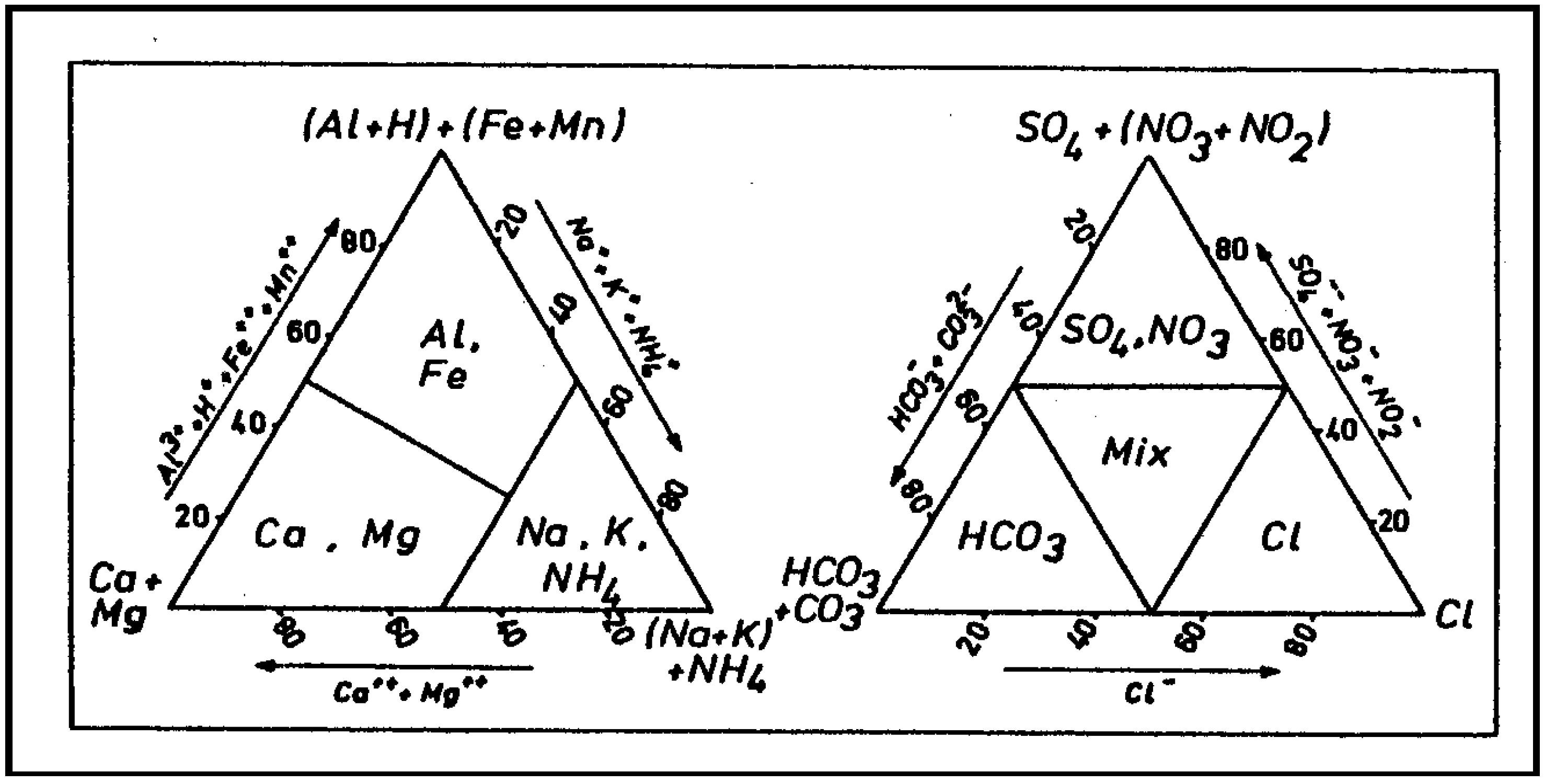
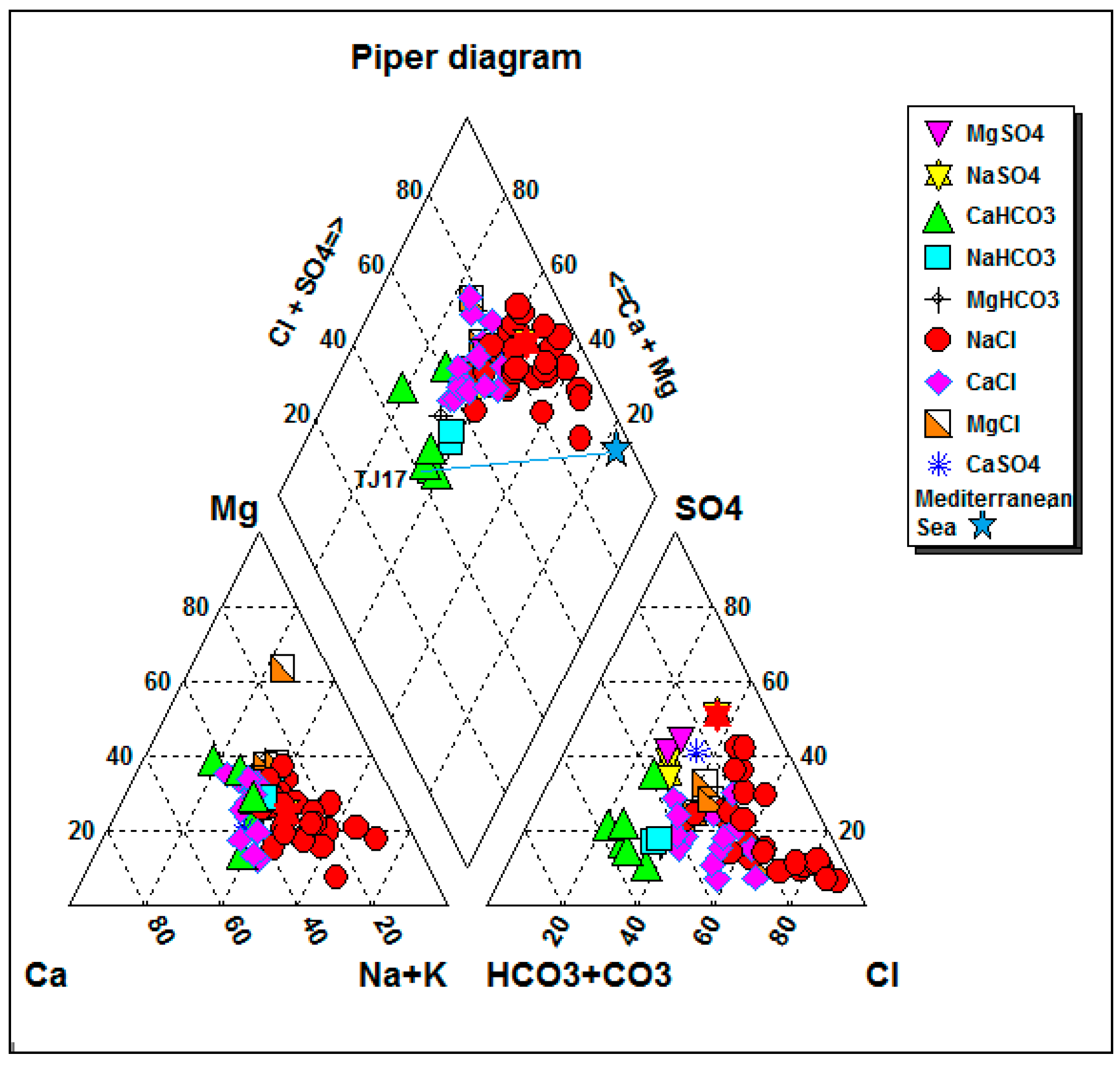
4.3. Water Types and Piper Diagram
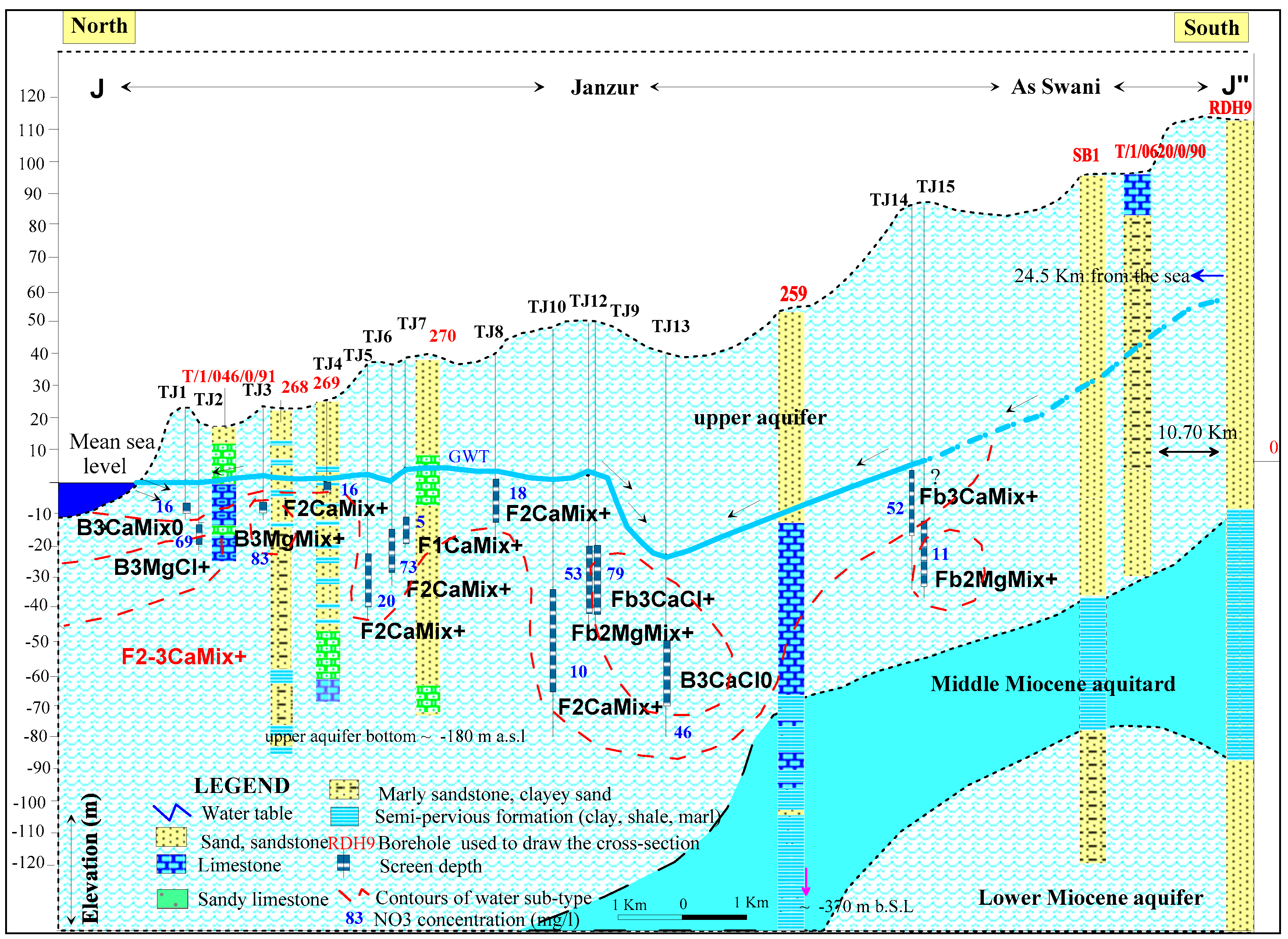
4.4. Hydrochemical Profile and Water Classification According to Stuyfzand
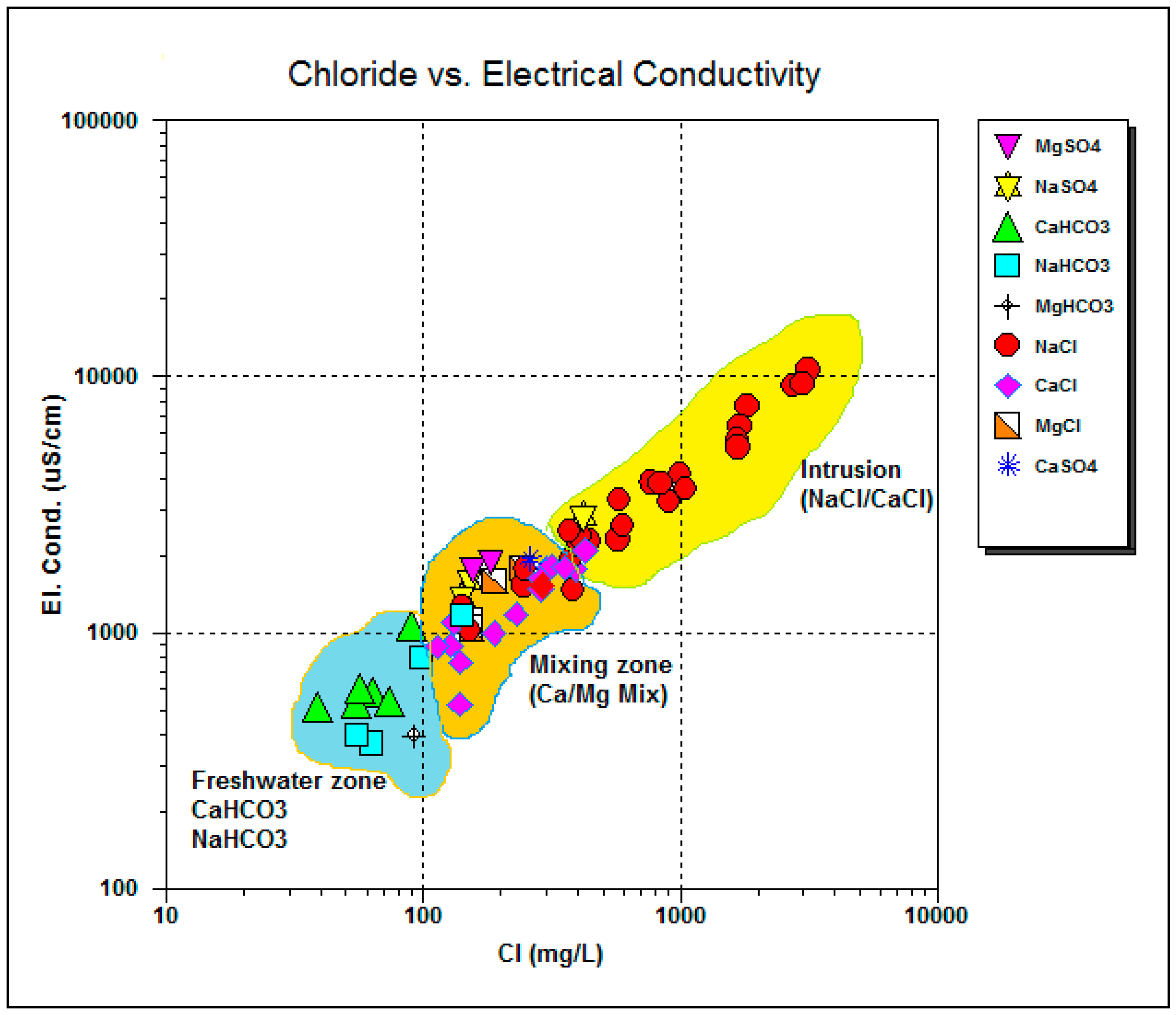
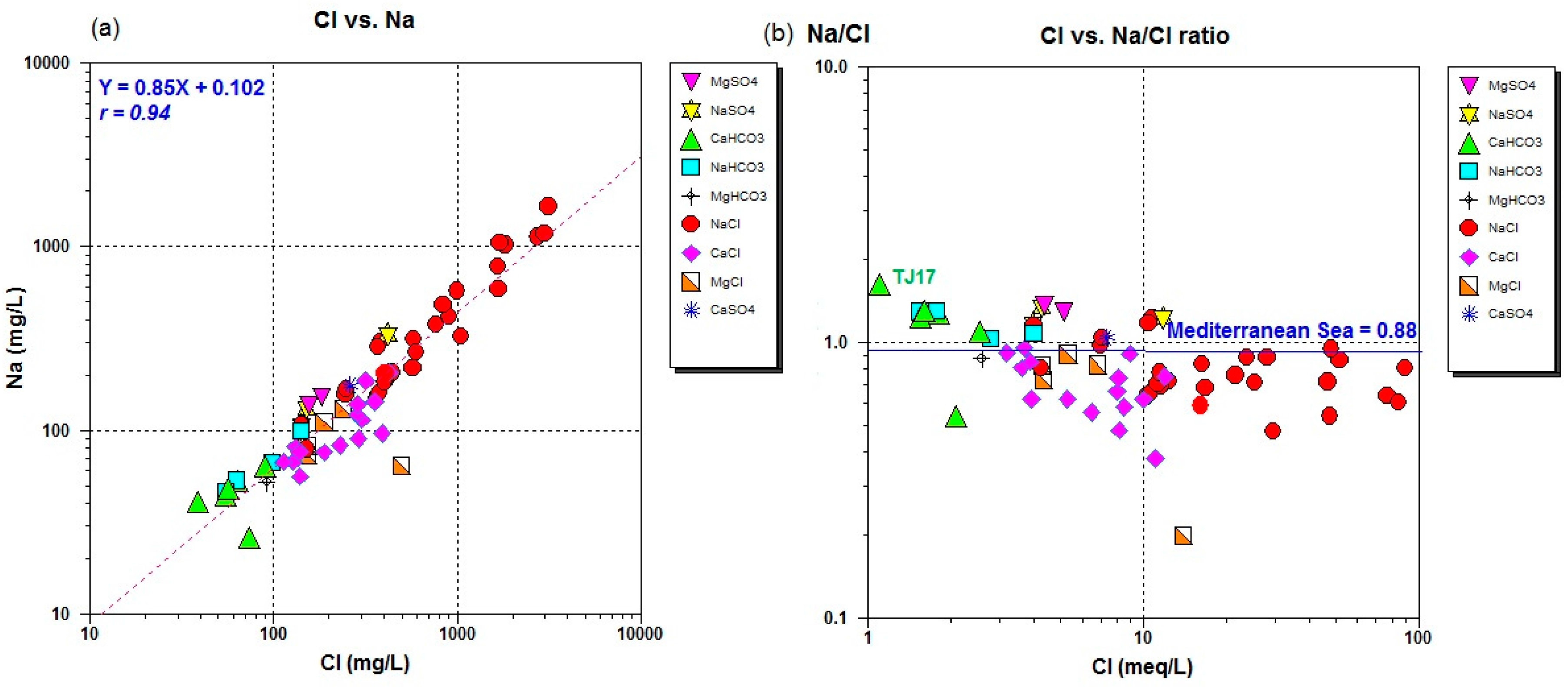
4.5. Ionic Ratio
4.6. Saturation Indices
4.7. Deviation from Conservative Mixture of End Member Fraction
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gaaloul, N.; Pliakas, F.; Kallioras, A.; Schuth, C.; Marinos, P. Simulation of Seawater Intrusion in Coastal Aquifers: Forty Five’Years exploitation in an Eastern Coast Aquifer in NE Tunisia. Open Hydrol. J. 2012, 6, 31–44. [Google Scholar] [CrossRef]
- Masciopinto, C. Simulation of coastal groundwater remediation: The case of Nardò fractured aquifer in Southern Italy. Environ. Model. Softw. 2006, 21, 85–97. [Google Scholar] [CrossRef]
- Mjemah, I.C.; Van Camp, M.; Walraevens, K. Groundwater exploitation and hydraulic parameter estimation for a Quaternary aquifer in Dar-es-Salaam, Tanzania. J. Afr. Earth Sci. 2009, 55, 134–146. [Google Scholar] [CrossRef]
- Van Camp, M.; Mtoni, Y.E.; Mjemah, I.C.; Bakundukize, C.; Walraevens, K. Investigating seawater intrusion due to groundwater pumping with schematic model simulations: The example of the Dar Es Salaam coastal aquifer in Tanzania. J. Afr. Earth Sci. 2014, 96, 71–78. [Google Scholar] [CrossRef]
- Jones, B.F.; Vengosh, A.; Rosenthal, E.; Yechieli, Y. Geochemical investigation of groundwater quality. In Seawater Intrusion in Coastal Aquifers-Concepts, Methods and Practices; Springer: Kluwer, The Netherlands, 1999; pp. 51–71. [Google Scholar]
- Meybeck, M.; Vorosmarty, C.; Schultze, R.; Becker, A. Conclusions: Scaling Relative Responses of Terrestrial Aquatic Systems to Global Changes. In Vegetation, Water, Humans and the Climate; Kabat, P., Claussen, M., Dirmeyer, P.A., Gash, J.H.C., Bravo de Guenni, L., Meybeck, M., Pielke, R.A., Vorosmarty, C.J., Hutjes, R.W.A., Lutkemeier, S., Eds.; Springer: Berlin/Heidelberg, Germany, 2003; pp. 455–464. [Google Scholar]
- Werner, A.D.; Bakker, M.; Post, V.E.A.; Vandenboede, A.; Lu, C.H.; Ataie-Ashtiani, B.; Simmons, C.T.; Barry, D.A. Seawater intrusion processes, investigation and management. Recent advances and futures challenges. Adv. Water Resour. 2013, 51, 3–26. [Google Scholar] [CrossRef]
- Andreasen, D.C.; Fleck, W.B. Use of bromide: Chloride ratios to differentiate potential sources of chloride in a shallow, unconfined aquifer affected by brackish-water intrusion. Hydrogeol. J. 1997, 5, 17–26. [Google Scholar] [CrossRef]
- Lin, J.; Snodsmith, J.B.; Zheng, C.; Wu, J. A modeling study of seawater intrusion in Alabama Gulf Coast, USA. Environ. Geol. 2009, 57, 119–130. [Google Scholar] [CrossRef]
- Meisler, H.; Leahy, P.P.; Knobel, LL. The Effect of Eustatic Sea-Level Changes on Saltwater-Freshwater Relations in the Northern Atlantic Coastal Plain; U.S. Geological Survey Water-Supply Paper; U.S. Geological Survey: Reston, VA, USA, 1985; Volume 2255, p. 28.
- Stringfield, V.T.; LeGrand, H.E. Relation of sea water to fresh water in carbonate rocks in coastal areas, with special reference to Florida, U.S.A. J. Hydrol. 1969, 9, 387–404. [Google Scholar] [CrossRef]
- Wicks, C.M.; Herman, J.S. Regional hydrogeochemistry of a modern coastal mixing zone. Water Resour. Res. 1996, 32, 401–407. [Google Scholar] [CrossRef]
- Wicks, C.M.; Herman, J.S.; Randazzo, A.F.; Jee, J.L. Water-rock interactions in a modern coastal mixing zone. Geol. Soc. Am. Bull. 1995, 107, 1023–1032. [Google Scholar] [CrossRef]
- Barlow, M.P. Groundwater in Freshwater-Saltwater Environments of the Atlantic Coast; U.S. Geological Survey Circular: Reston, GA, USA, 2003; p. 1262.
- Langman, J.B.; Ellis, A.S. A multi-isotope (δD, δ18O, 87Sr/86Sr, and δ11B) approach for identifying saltwater intrusion and resolving groundwater evolution along the Western Caprock Escarpment of the Southern High Plains, New Mexico. Appl. Geochem. 2010, 25, 159–174. [Google Scholar] [CrossRef]
- Izbicki, J.A. Chloride Sources in a California Coastal Aquifer, in Peters. In Ground Water in the Pacific Rim Countries; Peters, H.J., Ed.; American Society of Civil Engineers: Reston, GA, USA, 1991; pp. 71–77. [Google Scholar]
- Izbicki, J.A. Use of δ 18O and δD to define seawater intrusion. In North American Water and Environmental Congress; Bathala, C.T., Ed.; IR Div/ASCE; American Society of Civil Engineers: New York, NY, USA, 1996. [Google Scholar]
- Todd, D.K. Sources of Saline Intrusion in the 400-Foot Aquifer, Castroville Area, California; Monterey Country Flood Control and Water Conservation District: Salinas, CA, USA, 1989; p. 41.
- Vengosh, A.; Gill, J.; Davisson, M.L.; Hudson, G.B. A multi-isotope (B, Sr, O, H, and C) and age dating (3H-3He and 14C) study of groundwater from Salinas Valley, California: Hydrochemistry, dynamics, and contamination processes. Water Resour. Res. 2002, 38. [Google Scholar] [CrossRef]
- Cardoso, P. Raline water intrusion in Mexico. In Water Pollution 2018, 2nd ed.; Transactions on Ecology and the Environment; WIT Press: Southampton, UK, 1993. [Google Scholar] [CrossRef]
- Bocanegra, E.; Da Silva, G.C.; Custodio, E.; Manzano, M.; Montenegro, S. State of knowledge of coastal aquifer management in South America. Hydrogeol. J. 2010, 18, 261–267. [Google Scholar] [CrossRef]
- Werner, A.D. A review of seawater intrusion and its management in Australia. Hydrogeol. J. 2010, 18, 281–285. [Google Scholar] [CrossRef]
- Steyl, G.; Dennis, I. Review of coastal-area aquifers in Africa. Hydrogeol. J. 2010, 18, 217–225. [Google Scholar] [CrossRef]
- Chaouni Alia, A.; El Halimi, N.; Walraevens, K.; Beeuwsaert, E.; De Breuck, W. Investigation de la Salinisation de la Plaine de Bou-Areg (Maroc Nord-Oriental); Freshwater Contamination; International Association of Hydrological Sciences—IAHS Publication: London, UK, 1997; Volume 243, pp. 211–220. [Google Scholar]
- El Halimi, N.; Chaouni Alia, A.; Beeuwsaert, E.; Walraevens, K. Hydrogeological and Geophysical Investigation for Characterizing the Groundwater Reservoir in Saidia Plain (north-eastern Morocco). In Development of Water Resource Management Tools for Problems of Seawater Intrusion and Contamination of Fresh-Water Resources in Coastal Aquifers; Walraevens, K., Ed.; Ghent University: Ghent, Belgium, 2000; pp. 67–75. ISBN 90-76878-01-3. [Google Scholar]
- Lamrini, A.; Beeuwsaert, E.; Walraevens, K. Contribution to the characterization of the Martil Coastal Aquifer System. In Development of Water Resource Management Tools for Problems of Seawater Intrusion and Contamination of Fresh-Water Resources in Coastal Aquifers; Walraevens, K., Ed.; Ghent University: Ghent, Belgium, 2000; pp. 76–81. ISBN 90-76878-01-3. [Google Scholar]
- Tarhouni, J.; Jemai, S.; Walraevens, K.; Rekaya, M. Caractérisation de l’aquifère côtier de Korba au Cap Bon (Tunisie). In Development of Water Resource Management Tools for Problems of Seawater Intrusion and Contamination of Fresh-Water Resources in Coastal Aquifers; Walraevens, K., Ed.; Ghent University: Ghent, Belgium, 2000; pp. 11–27. ISBN 90-76878-01-3. [Google Scholar]
- Imerzoukene, S.; Walraevens, K.; Feyen, J. Salinization of the coastal and eastern zones of the alluvial and unconfined aquifer of the Mitidja Plain (Algeria). In Proceedings of the 13th Salt Water Intrusion Meeting, Cagliari, Italy, 5–4 June 1994; pp. 163–175. [Google Scholar]
- Van Camp, M.; Mjemah, I.C.; Alfarrah, N.; Walraevens, K. Modeling approaches and strategies for data-scarce aquifers: Example of the Dar es Salaam aquifer in Tanzania. Hydrogeol. J. 2013, 21, 341–356. [Google Scholar] [CrossRef]
- Walraevens, K.; Mjemah, I.C.; Mtoni, Y.; Van Camp, M. Sources of salinity and urban pollution in the Quaternary sand aquifers of Dar es Salaam, Tanzania. J. Afr. Earth Sci. 2015, 102, 149–165. [Google Scholar] [CrossRef]
- Oude Essink, G.H.P. Saltwater intrusion in 3D larg-scale aquifers a Duch case. Phys. Chem. Earth 2001, 26, 337–344. [Google Scholar] [CrossRef]
- Vandenbohede, A.; Walraevens, K.; De Breuck, W. What does the interface on the fresh-saltwater distribution map of the Belgian coastal plain represent? Geol. Belg. 2015, 18/1, 31–36. [Google Scholar]
- Alcala, F.J.; Custodio, E. Using the Cl/Br ratio as a tracer to identify the origin of salinity in aquifers in Spain and Portugal. J. Hydrol. 2008, 359, 189–207. [Google Scholar] [CrossRef]
- Custodio, E. Coastal aquifers of Europe: An overview. Hydrogeol. J. 2010, 18, 269–280. [Google Scholar] [CrossRef]
- De Montety, V.; Radakovitch, O.; Vallet-Coulomb, C.; Blavoux, B.; Hermitte, D.; Valles, V. Origin of groundwater salinity and hydrochemical processes in a confined coastal aquifer: Case of the Rhône delta (Southern France). Appl. Geochem. 2008, 23, 2337–2349. [Google Scholar] [CrossRef]
- UN—United Nations. Water for People, Water for Life; UN World Development Report (WWDR); UN: New York, NY, USA, 2003. [Google Scholar]
- WHO—World Health Organization. Guidelines for Drinking-Water Quality: First Addendum to Third Edition, Volume 1 Recommendations; WHO: Geneva, Switzerland, 2006. [Google Scholar]
- Ben-Asher, J.; Beltrao, J.; Costa, M. Modelling the effect of sea water intrusion on ground water salinity in agricultural areas in Israel, Portugal, Spain and Turkey. In Proceedings of the International Symposium on Techniques to Control Salination for Horticultural Productivity, Antalya, Turkey, 7–10 November 2000. [Google Scholar]
- Edmunds, W.M.; Milne, C.J. Palaeowaters in Coastal Europe: Evolution of Groundwater Since the Late Pleistocene; Geological Society of London: London, UK, 2001. [Google Scholar]
- Gordu, F.; Motz, L.H.; Yurtal, R. Simulation of Seawater Intrusion in the Goksu Delta at Silifke, Turkey. In Proceedings of the First International Conference on Saltwater Intrusion and Coastal Aquifers Monitoring, Modeling, and Management, Essaouira, Morocco, 23–25 April 2001. [Google Scholar]
- Yazicigil, H.; Ekmekci, M. Perspectives on Turkish ground water resources. Ground Water 2003, 41, 290–291. [Google Scholar] [CrossRef] [PubMed]
- Karahanoglu, N.; Doyuran, V. Finite element simulation of seawater intrusion into a quarry-site coastal aquifer, Kocaeli-Darica, Turkey. Environ. Geol. 2003, 44, 456–466. [Google Scholar] [CrossRef]
- Peters, H.J. Ground Water in the Pacific Rim Countries; IR Div/ASCE; American Society of Civil Engineers: New York, NY, USA, 1991; pp. 71–77. [Google Scholar]
- Demirel, Z. The history and evaluation of saltwater intrusion into a coastal aquifer in Mersin, Turkey. J. Environ. Manag. 2004, 70, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Camur, M.Z.; Yazicigil, H. Effects of the planned Ephesus recreational canal on freshwater-seawater interface in the Selcuk sub-basin, Izmir. Environ. Geol. 2005, 48, 229–237. [Google Scholar] [CrossRef]
- Alfarrah, N.; Hweesh, A.; Van Camp, M.; Walraevens, K. Groundwater flow and chemistry of the oases of Al Wahat, NE Libya. Environ. Earth Sci. 2016, 75, 1–24. [Google Scholar] [CrossRef]
- Gaaloul, N.; Pliakas, F.; Kallioras, A.; Marinos, P. Seawater intrusion in Mediterranean porous coastal aquifers: Cases from Tunisia and Greece. In Proceedings of the 8th International Hydrogeological Congress of Greece, Athens, Greece, 8–10 October 2008; pp. 281–290. [Google Scholar]
- World Bank. People’s Democratic Republic of Algeria—A Public Expenditure Review. Assuring High Quality Public Investment; Report No. 36270; World Bank: Washington, DC, USA, 2007. [Google Scholar]
- Djabri, L.; Laouar, R.; Hani, A.; Mania, J.; Mudry, J. The Origin of Water Salinity on the Annaba Coast (NE Algeria). In Proceedings of the Symposium HSO2a—IUGG2003, Sapporo, Japan, 30 June–11 July 2003; International Association of Hydrological Sciences—IAHS Publication: London, UK, 2003; Volume 280, pp. 229–235. [Google Scholar]
- Pulido-Bosch, A.; Tahiri, A.; Vallejos, A. Hydrogeochemical characteristics of processes in the Temara Aquifer in Northwestern Morocco. Water Air Soil Pollut. 1999, 114, 323–337. [Google Scholar] [CrossRef]
- Sherif, M.M. Nile Delta aquifer in Egypt. In Seawater Intrusion in Coastal Aquifers—Concepts, Methods and Practices; Bear, J., Cheng, A., Sorek, S., Ouazar, D., Herrera, A., Eds.; Theory and Application of Transport in Porous Media Book Series; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1999; pp. 559–590. [Google Scholar]
- ISARM. Internationally Shared (Transboundary) Aquifer Resources Management; IHP-VI, IHP Non Serial Publications in Hydrology November, Paris: UNESCO 2001; Internationally Shared (Transboundary) Aquifer Resources Management: Delft, The Netherlands, 2001. [Google Scholar]
- Khouri, J. Water resources of the Zarka River basin, Jordan. In Water Resources Management and Desertification Problems and Challenges; World Meteorological Organization (WMO): Geneva, Switzerland, 1996; pp. 84–96. [Google Scholar]
- Alfarrah, N. Hydrogeological and Hydrogeochemical Investigation of the Coastal Area of Jifarah Plain, NW Libya. Ph.D. Thesis, Laboratory of Applied Geology and Hydrogeology, Ghent University, Ghent, Belgium, 2011. [Google Scholar]
- Alfarrah, N.; Berhane, G.; Bakundukize, C.; Walraevens, K. Degradation of groundwater quality in coastal aquifer of Sabratah area, NW Libya. Environ. Earth Sci. 2017, 76, 664. [Google Scholar] [CrossRef]
- Alfarrah, N.; Van Camp, M.; Walraevens, K. Deducing transmissivity from specific capacity in the heterogeneous upper aquifer system of Jifarah Plain, NW-Libya. J. Afr. Earth Sci. 2013, 85, 12–21. [Google Scholar] [CrossRef]
- Gefli. Soil and Water Resources Survey for Hydro-Agricultural Development, Western Zone; Unpublished Report; Ground Water Authority: Tripoli, Libya, 1972.
- Libyan Industrial Research Centre (IRC). Geological Map of Jifarah Plain, 1st ed.; IRC: Tajura, Libya, 1975. [Google Scholar]
- Kruseman, G.P. Evaluation of Water Resources of the Gefara Plain; Unpublished Report; SDWR: Tripoli, Libya, 1977. [Google Scholar]
- Krummenacher, R. Gefara Plain Water Management Plan Project; Report on the Groundwater Resources of the Gefara Plain; Unpublished Report; 110p and 4 Annexes; GWA: Tripoli, Libya, 1982. [Google Scholar]
- APHA (American Public Health Association). Standard Methods for the Examination of Water and Wastewater; Greenberg, A.E., Trussell, R.R., Clesceri, L.S., Eds.; APHA: Washington, DC, USA, 1985. [Google Scholar]
- Parkhurst, D.L.; Appelo, C.A.J. User’s Guide to PHREEQC (Version 2)-A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations; U.S. Geological Survey Water-Resources Investigations Report 99-4259; U.S. Geological Survey: Reston, VA, USA, 1999; p. 312.
- Fidelibus, M.D.; Tulipano, L. Regional flow of intruding seawater in the carbonate aquifers of Apulia (Southern Italy). In Proceedings of the 14th Salt Water Intrusion Meeting, Malmö, Sweden, 17–21 June 1996; Geological Survey of Sweden: Uppsala, Sweden, 1996. [Google Scholar]
- Appelo, C.A.J. Cation and proton exchange, pH variations, and carbonate reactions in a freshening aquifer. Water Resour. Res. 1994, 30, 2793–2805. [Google Scholar] [CrossRef]
- Stuyfzand, P.J. A new hydrogeochemical classification of water types: Principles and application to the coastal dunes aquifer system of the Netherlands. In Proceedings of the 9th SWIM, Delft, The Netherlands, 12–16 May 1986; pp. 641–656. [Google Scholar]
- Stuyfzand, P.J. Hydrochemistry and Hydrology of the Coastal Dune Area of the Western Netherlands. Ph.D. Thesis, Free University (VU), Amsterdam, The Netherlands, 1993; p. 366. [Google Scholar]
- Mollema, P.N.; Antonellini, M.; Dinelli, E.; Gabbianelli, G.; Greggio, N.; Stuyfzand, P.J. Hydrochemical and physical processes influencing salinization and freshening in Mediterranean low-lying coastal environments. Appl. Geochem. 2013, 34, 207–221. [Google Scholar] [CrossRef]
- Mollema, P.N.; Antonellini, M.; Stuyfzand, P.J.; Juhasz-Holterman, M.H.A.; Van Diepenbeek, P.M.J.A. Metal accumulation in an artificially recharged gravel pit lake used for drinking water supply. J. Geochem. Explor. 2015, 150, 35–51. [Google Scholar] [CrossRef]
- Mollema, P.N. Water and Chemical Budgets of Gravel Pit Lakes: Case Studies of Fluvial Gravel Pit Lakes along the Meuse River (The Netherlands) and Coastal Gravel Pit Lakes along the Adriatric Sea (Ravenna, Italy). Ph.D. Thesis, Technische Universiteit Delft, Delft, The Netherlands, 2016. [Google Scholar]
- WHO—World Health Organization. Guidelines for Drinking-Water Quality (Electronic Resource): Incorporating 1st and 2nd Addenda, Volume 1, Recommendations, 3rd ed.; WHO Library Cataloguing-in Publication Data; WHO: Geneva, Switzerland, 2008; p. 668. [Google Scholar]
- Mercado, A. The use of hydrogeochemical patterns in carbonate sand and sandstone aquifers to identify intrusion and flushing of saline waters. Groundwater 1985, 23, 635–664. [Google Scholar] [CrossRef]
- El Moujabber, M.; Bou Samra, B.; Darwish, T.; Atallah, T. Comparison of different indicators for groundwater contamination by seawater intrusion on the Lebanese coast. Water Resour. Manag. 2006, 20, 161–180. [Google Scholar] [CrossRef]
- Walraevens, K.; Van Camp, M. Advances in understanding natural groundwater quality controls in coastal aquifers. In Proceedings of the 18th Salt Water Intrusion Meeting (SWIM), Cartagena, Spain, 31 May–3 June 2004; pp. 451–460. [Google Scholar]
- Vengosh, A.; Starinsky, A.; Melloul, A.; Fink, M.; Erlich, S. Salinization of the Coastal Aquifer Water by Ca-Chloride Solutions at the Interface Zone, Along the Coastal Plain of Israel; Hydrological Service: Jerusalem, Israel, 1991. [Google Scholar]
- Jeen, S.W.; Kim, J.M.; KO, K.S.; Yum, B.W.; Chang, H.W. Hydrogeochemical characteristics of groundwater in a Midwestern coastal aquifer system, Korea. Geosciences 2001, 5, 339–348. [Google Scholar] [CrossRef]
- Sanchez-Martos, F.; Pulido-Bosch, A.; Calaforra-Chordi, J.M. Hydrogeochemical processes in an arid region of Europe (Almeria, SE Spain). Appl. Geochem. 1999, 14, 735–745. [Google Scholar] [CrossRef]

| Period | Epoch | Deposits and Formations | Typical Lithology | Thickness (m) | |
|---|---|---|---|---|---|
| Quaternary | Holocene | Upper Miocene-Pliocene-Quaternary aquifer | Wadi deposits | Loose gravel, loam | 5–150 |
| Sand beach | Shell and silica sand | ||||
| Sand dunes and sand sheets | Shell fragments, silica sands and gypsum | ||||
| Sebkha deposits | Gypsum | ||||
| Fluvial-Eolian deposits | Silt, clay, marl and fine sand | ||||
| Gergaresh Formation | Conglomerate, sandstone, silt, sandy limestone | ||||
| Pleistocene | Jeffara Formation | Silt, sand and gravel caliche bands | |||
| Qsar al Haj Formation | Alluvial fans and cones | ||||
| Al Kums Formation | Limestone, dolomite | ||||
| Miocene | Volcanic rocks | Basalt and phonolite | 25–250 | ||
| Middle | Middle Miocene clay | ||||
| Parameter (Unit mg/L) | Analyzed Rainwater in Jifarah Plain | Analyzed Recharge Water in Tripoli (TJ17) | Mediterranean Seawater [54] |
|---|---|---|---|
| pH | 7.64 | 7.97 | - |
| Na+ | 31.50 | 43.75 | 12,700 |
| K+ | 3.00 | 4.50 | 470 |
| Ca2+ | 13.40 | 49.51 | 470 |
| Mg2+ | 3.15 | 8.70 | 1490 |
| Cl− | 26.10 | 55.15 | 22,900 |
| SO42− | 10.33 | 36.62 | 3190 |
| HCO3− | 102.48 | 174.46 | 173 |
| NO3− | 1.12 | 29.10 | 0 |
| NO2− | 0.16 | 0.001 | - |
| PO43− | 0.13 | 0.07 | - |
| Fe (Total) | 0.0 | 0.008 | - |
| Mn2+ | 0.01 | 0.01 | - |
| NH4+ | 0.22 | 0.001 | - |
| TDS | 199.24 | 401.88 | 41,393 |
| Main Type | Code | Cl (mg/L) |
|---|---|---|
| Fresh | F | ≤150 |
| Fresh-brackish | Fb | 150–300 |
| Brackish | B | 300–1000 |
| Brackish-salt | Bs | 1000–10,000 |
| Salt | S | 10,000–20,000 |
| Hyperhaline | H | >20,000 |
| Number | Name | Code | Total Hardness (mmol/L) | Natural Occurrence in Main Types |
|---|---|---|---|---|
| −1 | Very soft | * | 0–0.5 | F |
| 0 | Soft | 0 | 0.5–1 | F Fb B |
| 1 | Moderately hard | 1 | 1–2 | F Fb B Bs |
| 2 | Hard | 2 | 2–4 | F Fb B Bs |
| 3 | Very hard | 3 | 4–8 | F Fb B Bs |
| 4 | Extremely hard | 4 | 8–16 | Fb B Bs S |
| 5 | Extremely hard | 5 | 16–32 | Bs S H |
| 6 | Extremely hard | 6 | 32–64 | Bs S H |
| 7 | Extremely hard | 7 | 64–128 | S H |
| 8 | Extremely hard | 8 | 128–256 | H |
| 9 | Extremely hard | 9 | ≥256 | H |
| Class | Code | Condition (meq/L) |
|---|---|---|
| {Na + K + Mg} deficit | - | {Na + K + Mg} corrected < −√0.5 Cl |
| {Na + K + Mg} equilibrium | 0 | −√0.5 Cl ≤ {Na + K + Mg} corrected ≤ +√0.5 Cl |
| {Na + K + Mg} surplus | + | {Na + K + Mg} corrected > √0.5 Cl |
| ID | T (°C) | Water Level (m a.s.l) | pH | Eh (mV) | EC (µ/cm 25°) | Ca2+ mg/L | Mg2+ mg/L | Na+ mg/L | K+ mg/L | Mn2+ mg/L | Fe2+/3+ mg/L | NH4+ mg/L | NO3− mg/L | NO2− mg/L | CO32− mg/L | HCO3− mg/L | Cl− mg/L | SO42− mg/L |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TM5 | 22 | 19.11 | 7.30 | 234 | 2840 | 200 | 104 | 334 | 14 | 0.01 | 0.40 | 0.07 | 21 | 0.01 | 0 | 273 | 419 | 837 |
| TM6 | 22 | 45.20 | 7.45 | 234 | 2500 | 110 | 72 | 283 | 6 | 0.01 | 0.41 | 0.19 | 118 | 0.01 | 0 | 219 | 371 | 382 |
| TS21 | 21 | −8.01 | 7.97 | 133 | 1160 | 82 | 42 | 99 | 8 | 0 | 0 | 0 | 11 | 0 | 6 | 287 | 142 | 91 |
| TJ1 | 24 | 0 | 8.22 | 160 | 1826 | 172 | 29 | 186 | 16 | 0 | 0 | 0 | 16 | 0 | 24 | 269 | 316 | 221 |
| TJ2 | 24 | 0 | 7.82 | 142 | 1940 | 93 | 84 | 160 | 6 | 0.01 | 0.01 | 0.02 | 69 | 0.11 | 0 | 323 | 376 | 123 |
| TJ3 | 23 | 0 | 7.40 | 152 | 2380 | 140 | 93 | 184 | 14 | 0 | 0 | 0 | 83 | 0 | 0 | 317 | 410 | 268 |
| TJ4 | 24 | 1.50 | 8.17 | 188 | 891 | 78 | 29 | 67 | 4 | 0 | 0 | 0 | 16 | 0 | 1 | 189 | 128 | 62 |
| TJ5 | 24 | 2.25 | 8.16 | 143 | 1100 | 82 | 44 | 81 | 8 | 0 | 0 | 0 | 20 | 0 | 12 | 208 | 131 | 144 |
| TJ6 | 22 | 1.50 | 7.59 | 161 | 524 | 71 | 40 | 76 | 5 | 0 | 0 | 0.03 | 73 | 0.11 | 0 | 236 | 138 | 87 |
| TJ7 | 23 | 4.00 | 8.2 | 139 | 510 | 40 | 19 | 41 | 4 | 0 | 0 | 0 | 5 | 0 | 18 | 140 | 39 | 53 |
| TJ8 | 23 | 3.80 | 8.19 | 156 | 878 | 62 | 36 | 67 | 4 | 0 | 0 | 0 | 18 | 0 | 12 | 165 | 113 | 96 |
| TJ10 | 24 | 3.50 | 9.94 | 142 | 1020 | 66 | 41 | 79 | 5 | 0 | 0.08 | 0 | 53 | 0.14 | 0 | 198 | 151 | 115 |
| TJ11 | 22 | 0 | 7.54 | 169 | 1530 | 116 | 65 | 90 | 6 | 0 | 0.12 | 0 | 79 | 0.10 | 0 | 220 | 290 | 148 |
| TJ12 | 22 | 3.00 | 7.85 | 140 | 1592 | 72 | 64 | 111 | 6 | 0.01 | 0.04 | 0.10 | 46 | 0.11 | 0 | 190 | 189 | 161 |
| TJ13 | 24 | −23.90 | 7.77 | 150 | 1764 | 147 | 77 | 96 | 6 | 0.01 | 0.07 | 0.10 | 52 | 0.12 | 0 | 244 | 391 | 129 |
| TJ14 | 24 | 7.50 | 8.20 | 198 | 1928 | 188 | 53 | 175 | 8 | 0 | 0 | 0 | 11 | 0 | 12 | 281 | 259 | 413 |
| TJ15 | 24 | 9.00 | 7.30 | 175 | 1868 | 123 | 78 | 152 | 6 | 0 | 0.03 | 0.04 | 55 | 0.10 | 0 | 287 | 182 | 378 |
| TJ17 | 23 | 23.00 | 7.97 | 213 | 532 | 50 | 9 | 44 | 5 | 0.01 | 0 | 0 | 29 | 0 | 0 | 175 | 55 | 37 |
| TG1 | 19 | 26.35 | 7.75 | 149 | 2270 | 117 | 91 | 206 | 4 | 0.01 | 0.09 | 0.04 | 61 | 0.37 | 0 | 284 | 441 | 117 |
| TG6 | 22 | −24.50 | 7.40 | 151 | 7660 | 192 | 172 | 1023 | 22 | 0.01 | 0.16 | 0.04 | 77 | 0.12 | 0 | 232 | 1831 | 297 |
| TR52 | 22 | 0 | 7.95 | 166 | 6380 | 132 | 137 | 1049 | 31 | 0 | 0 | 0 | 38 | 0 | 39 | 384 | 1709 | 274 |
| TT2 | 22 | 0 | 7.73 | 148 | 9150 | 572 | 209 | 1129 | 70 | 0 | 0 | 0 | 0.80 | 0 | 6 | 421 | 2726 | 512 |
| TT3 | 22 | 0 | 8.22 | 142 | 3850 | 262 | 100 | 377 | 67 | 0 | 0 | 0 | 1 | 0 | 24 | 433 | 766 | 418 |
| TT6 | 21 | 0 | 7.73 | 162 | 9310 | 572 | 203 | 1178 | 20 | 0 | 0 | 0 | 23 | 0 | 3 | 226 | 3003 | 293 |
| TT7 | 21 | 1.00 | 7.97 | 168 | 3810 | 204 | 108 | 480 | 24 | 0 | 0 | 0 | 6 | 0 | 12 | 256 | 840 | 567 |
| TT9 | 24 | 5.80 | 8.30 | 183 | 600 | 50 | 24 | 48 | 8 | 0 | 0 | 0 | 7 | 0 | 9 | 189 | 57 | 67 |
| T63 | 23 | 9.00 | 8.03 | 191 | 582 | 52 | 18 | 53 | 4 | 0 | 0 | 0 | 6 | 0 | 77 | 64 | 64 | 53 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alfarrah, N.; Walraevens, K. Groundwater Overexploitation and Seawater Intrusion in Coastal Areas of Arid and Semi-Arid Regions. Water 2018, 10, 143. https://doi.org/10.3390/w10020143
Alfarrah N, Walraevens K. Groundwater Overexploitation and Seawater Intrusion in Coastal Areas of Arid and Semi-Arid Regions. Water. 2018; 10(2):143. https://doi.org/10.3390/w10020143
Chicago/Turabian StyleAlfarrah, Nawal, and Kristine Walraevens. 2018. "Groundwater Overexploitation and Seawater Intrusion in Coastal Areas of Arid and Semi-Arid Regions" Water 10, no. 2: 143. https://doi.org/10.3390/w10020143
APA StyleAlfarrah, N., & Walraevens, K. (2018). Groundwater Overexploitation and Seawater Intrusion in Coastal Areas of Arid and Semi-Arid Regions. Water, 10(2), 143. https://doi.org/10.3390/w10020143






