Abstract
In recent years, the need for a safe and modern composite barrier for the prevention of groundwater contamination and the provision of geo-environmental protection has been studied together with the need of designing a low-cost and effective liner for isolating landfill contents from the environment. In this study, various mix designs involving two natural adsorbents, Na-bentonite the pH-adjusted sawdust, were prepared for a series of geo-environmental experiments to be carried out to determine the adsorption capacity, buffering capacity, pH changes, and Chemical Oxygen Demand (COD) changes among others, in the presence of Pb(NO3)2 contaminant concentrations. Generally, the results showed an increase in adsorption capacity in the acidic segment of the treatment. An increase of 58% of the adsorption efficiency of the Na-bentonite in adsorbing the contaminant at the highest concentration was the most important achievement of the system while in the acidic segment.
1. Introduction
Soils, underground water, and surface water are continuously polluted by heavy metals issued by various resources such as industry and mining [1]. Soils contaminated with heavy metals have long been recognized as one of the most common outcomes of environmental pollution. The metal contaminants commonly found in soils are lead, copper, mercury, arsenic, chromium, and cadmium; but lead and copper are the ones that have received the most attention [2]. Only a small quantity of heavy metals is necessary for feeding into the biological cycles. The bulk that is expelled every day into the environment by human activities will add to the toxicity of these elements. Lead is among the few toxic heavy metals that can exist in the environment and be stable against micro-biological degradation [3]. However, once it gets into drinking water at more than 0.02 mg/L, the effects on human health include anemia, encephalopathy, diarrhea, hepatitis, and nephritic syndrome of the kidneys, among others [4]. Thus laws have been passed in many countries in order to ensure protection of the environment against lead contamination or to retain the contaminants rather than deal with the consequences.
In checking against infiltration of leachate that pollutes the soils as well as the groundwater, a clay-based obstruction is always necessary. These geo-chemical barriers, such as for nuclear waste reservoirs and hazardous waste landfills, involve linings made of bentonite, or to be more precise, montmorillonite. The low-cost, highly specific surface, and ubiquitous presence associated with bentonite have made it a precious material for geo-environmental projects [5]. Previous studies showed that bentonite is an appropriate adsorbent in solidifying and stabilizing heavy metal contaminants, even when these were in high concentrations. The clay soils, due to the specific capability at the molecular level, and also the specific characteristics which belong to the minerals would have a special interaction with the contaminants causing all or a portion of the hazardous substances in the solution to be assimilated [6,7,8]. This property has resulted in clay soils with bentonite in them becoming choice base liner materials for underlying industrial and municipal waste landfills when exposed to intensive physical–chemical attacks by the leachates of the wastes.
In many cases heavy metals are made to retain or entrapped in clay soils by three main particle properties of the clays, namely carbonation, cation exchange capacity (CEC), and specific surface area (SSA). Heavy metal retention capacity in each of the above cases also depends on the pH of the soil containing the heavy metal [9]. The environmental mobility and bioavailability of heavy metals in soils can be influenced by at least four possible factors such as: (1) the variation of acidity of the environment and following the system; (2) change in the system ionic strength; (3) changes in the oxidation-reduction potential of the system; and (4) formation of the complexes. Altogether, precipitation has a solid phase (oxide, hydroxides, carbonates), and complexation reactions. On the other hand, the ion exchange adsorption should be considered as ‘retardation’ because of the desorption of heavy metals which when adsorbed by the ion exchange mechanism can readily occur.
With increasing concentration of heavy metal pollutants, the pH of the system decreases. The presence of heavy metals decreases the pH of the system for two main reasons: first, due to the release of H+ caused by the retention of metal ions, and second, when the metal contaminant is dissolved in water, the hydrated ions are produced, and due to the hydrolysis process of metals, the solution will be acidified and the pH of the soil–metal system decreases [9]. Therefore, particularly for the latter, an acidification leads to an increase in mobility of the metals bound to the soil.
In an adsorption process, there are a lot of events, such as chemical sedimentation, ion exchange, and solvent extraction, which will affect the choice of an adsorbent to use. The choice in a landfill will also depend on cost, availability, ease of use, and adaptability with the surroundings. Recently researchers have nominated a number of inorganic adsorbent such as fly ash [10,11], zeolite [12], and agriculture adsorbents like onion skin [13], rice husks [14], clays, and activated carbon.
The new design approach has a preference for a low cost and biomass type of adsorbent. The sawdust proposed by this research is a lignocellulose material associated with wood. The cellulose, hemicellulose, and lignin, coming from the sawdust will provide for the adsorption capacity, strength, flexural, and impact properties of clay materials [15].
2. Materials and Methods
2.1. Physical and Chemical Properties of the Adsorbents
2.1.1. Na-Bentonite
Bentonite clay, provided by AMC Australia (216 Balcatta Road, Balcatta WA 6021 Australia), was used in this research. It was first washed with deionized water several times in order to remove dust, unwanted materials, and water-soluble impurities. The sample was then oven-dried at 150 to 200 °C for 48 h before being used as an adsorbent. At this point, the grey colored bentonite was in fine form passing mesh No. 30. Its physical properties such as specific gravity, particle size distribution, Atterberg limits, organic content, and moisture content were determined according to the ASTM D1237 [16]. Until the next stage, it was stored in cool, shaded, and dry environment. The sample was then air-dried and sieved through mesh No. 200 before undergoing chemical analyses which were in accordance with the laboratory manual of the Geotechnical Research Center, McGill University. The cation exchange capacity (CEC) was determined using the BaCl2 replacement method [17]. The X-ray powder diffraction (XRD) analysis was performed based on the method by Moore and Reynold [18]. The carbonate content was determined using the titration method [19]. The specific surface area (SSA) was determined according to the Brunauer–Emmett–Teller (BET) method [20]. The pH was measured on the bentonite-water solution of 1:10 ratio by mass—i.e., one portion of bentonite for every 10 portions of water. The resulting physical and chemical properties of the sample are given in Table 1, Table 2 and Table 3.

Table 1.
Physical properties of Na-bentonite.

Table 2.
Chemical properties of Na-bentonite.

Table 3.
Chemical composition of Na-bentonite.
2.1.2. Sawdust
The Red-Meranti sawdust was obtained from the workshop center of civil engineering faculty of University Sains Malaysia (5°08′49.6″ N 100°29′33.0″ E). The sawdust was washed with deionized water several times and it was oven-dried at 70 °C to evaporate the entire moisture content. The adsorbent was converted to a fine powder by the ball mill. The resulting material was sieved to the 100–150 (µm) particle size according to the ASTM D4779 method [21,22]. Then, 0.5 M HCL solution was used to remove the color and water soluble substances in the room temperature for 6 h. Eventually the sawdust was filtered and washed several times by the distilled water and after that oven dried at 70 °C. The adsorbent was kept in the airtight condition until the treatment process.
2.1.3. Alkali and Acidic Treatment of Sawdust
The pH of raw sawdust was measured in 1:10 adsorbent/water ratio by EUTECH measuremet700 pH meter. The sawdust placed into the five different groups of glass containers in order to adjusting the pH of the sawdust to 2, 4, 6, 8, and 10. To achieve this purpose, 0.1 M HCL for acidic treatment and 0.5 M NaOH for alkali treatment was used. The sawdust was soaked in deionized water at room temperature with continuous stirring, and the different amount of acidic and alkali reagents were added to the different containers in accordance with the pH targets, with the pH meter is located into the solutions. Until the stabilization of pH of different containers, the addition of HCL and NaOH drops continued. After this step, the solution was kept overnight and the pH was measured next day. This process was continued up to achieve the same amount of pH for two consecutive days. To eliminate the adsorbed alkali, the sawdust was then washed several times with distilled water and acetone. The alkali-treated and acidic-treated sawdust was dried in a hot-air oven at 60 °C and for subsequent analysis kept in a desiccator.
2.2. Sample Preparation and Experimental Methods
The equilibrium saturated mix test (EPA) was used in order to monitor the interaction of the bentonite clay soil and different mix designs of bentonite + % pH adjusted sawdust. Firstly, 20 g bentonite clay soil plus an addition of different percentage of the sawdust with various pH levels was added to the transparent bottles with a 1.5-L volume. Then, 1 L of each concentration of lead nitrate Pb(NO3)2 electrolyte was added to the bottles. The suspension of soil + sawdust + electrolyte was shacken for 3 h on the electric screed. To reach the equilibrium condition, all the shacken specimens were kept for 24 h. The suspensions were shacken again for another 3 h with an electric screed. In the following, the specimens were kept for 120 h to allow the system to become completely homogenous and for cation exchange to occur. After this step, the liquid phase of the specimens was separated from the solid phase by the means of centrifuge with 3000 rpm. Then, the concentration of lead ion was determined by the Atomic Adsorption Spectrometry (AAS) method.
To investigate the effect of the sawdust in different amounts and pH on the environmental behavior of the Bentonite clay soil in the presence of heavy metal (HM) Pb(NO3)2, a series of batch equilibrium tests was performed according to [23]. According to the different variable factors in this study, 80 numbers of the batches were prepared. The constant factor in all the specimens was 20 g Bentonite while the amount of sawdust was changing between 10%, 20%, and 30% weight of the Bentonite. The pH of each percent of sawdust was divided into five main groups (2, 4, raw sawdust (6), 8, and 10). The samples were prepared by adding solutions of lead nitrate Pb(NO3)2 in the range of 50 to 250 cmol/kg-soil to the bentonite + % pH adjusted samples. The suspensions of soil–electrolyte were prepared at a 1:10 soil/solution ratio.
3. Results and Discussion
3.1. Component Analysis of the Sawdust
An adjustment of the raw Meranti sawdust pH to the acidic and alkali was conducted to improve the environmental and mechanical properties. These properties are dependent to the lignin, cellulose, and hemicellulose content of the sawdust. So, to prove the effects of the chemical treatments, Fourier transform infrared spectroscopy (FTIR) was applied to the five groups of the sawdust after the adjusting process. Two samples belong to the acidic treatment (pH: 2 and pH: 4), one the raw sawdust without any treatment (pH: 6), and two others (pH: 8 and pH: 10) belong to the alkali treatment.
Figure 1 illustrates the wavenumbers of the different groups of sawdust versus the percentage of the transmittance. Cellulose, hemicellulose, and lignin are the main components of the sawdust. Each of them has a particular function and it was expected an improvement in the environmental and geotechnical properties of the clay by changes in the amount of components. Cellulose has prominent peaks at wavenumbers of 1606, 1461, 1251, 1213, and 1166 cm−1. Similarly hemicellulose spectrum has five distinct peaks at wavenumbers of 1431, 1373, 1338, 1319, and 1203 cm−1. The lignin spectrum illustrates the peaks at wavenumbers of 1599, 1511, 1467, 1429, 1157, and 1054 cm−1.
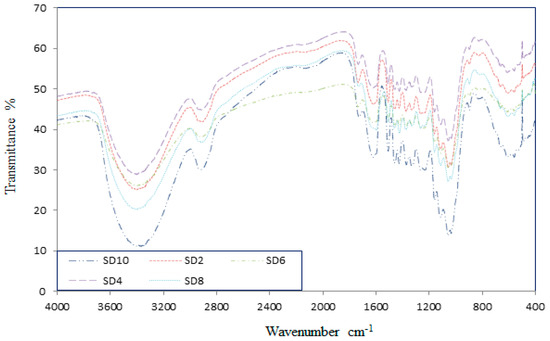
Figure 1.
Fourier-transform infrared spectroscopy (FTIR) of the different pH-adjusted sawdust.
As the previous study has demonstrated [24] the acidic treatment of the sawdust leading to an increase in the amount of lignin content while the alkali treatment has the same effect on the amount of the cellulose and hemicellulose. The acidic treatment increases the proportion of the active surfaces of the sawdust such as pores. The alkalization process not only directly influences the cellulosic fibril and degree of the polymerization, but also causes the extraction of the lignin [25]. From the geo-environmental point of view, the heavy metals are adsorbed by lignin rather than cellulose and hemicellulose [26]. From the mechanical properties of composite materials, the physical strength of clay soil system including sawdust was obtained using the cellulose and hemicellulose [27].
The adsorption bands at 2939 cm−1 and 2880 cm−1 are due to a contribution from C-H stretching that can prove the presence of the vinyl and methyl groups. Also, according to Figure 1 a strong band observed at 1046 cm−1 to 1053 cm−1 can be seen, indicating the presence of many C-OH and C-O-C bonds. The other strong band between 500 cm−1 and 600 cm−1 belongs to the C-X groups [28]. Thus, due to the different bands and wavenumbers an improvement in environmental properties by acidic treatment was expected.
3.2. Charge Characteristic
The zero point of charge (ZPC) is a fundamental description of a mineral surface, and is more or less the point where the total concentration of surface anionic sites is equal to the total concentration of surface cationic sites, and most of the sites are as the neutral hydroxide. At the pH above ZPC, the surface of the adsorbent is negatively charged and the surface will participate in the cation adsorption and cation exchange reactions. On the other hand, when the pH of the solution is below ZPC the surface has a net positive charge, and the surface will attract anions and participate in anion exchange reactions.
Each mineral and mineral crystal structures have zero point of charge. This value has been reported for montmorillonite as 2.5 [29]. The charge developed on the oxides surfaces are temporary charge and because it is dependent to the pH. This kind of charge is relatively weaker than the permanent charge of clay minerals. In the sawdust case (Figure 2), the pH over ZPC = 5.15 is favorable for chemical heavy metal cation adsorption. Thus, while the pH of the solution is over the ZPC, it can be expected that the sawdust participates in the cation adsorption process by the clay mineral. It should be considered that while the pH of the solution is below than ZPC the other adsorption mechanisms of the sawdust such as physically adsorption are still active.
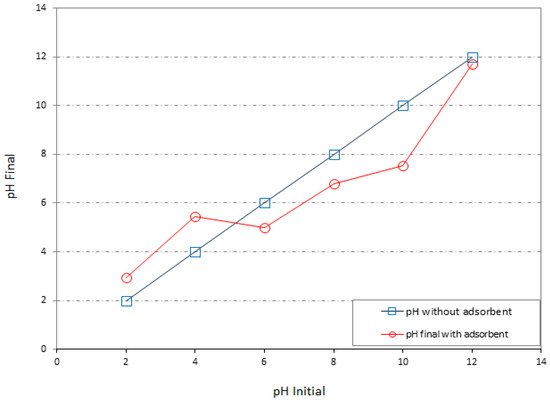
Figure 2.
Zero point of charge (ZPC) of the raw sawdust.
3.3. Brunauer-Emmett-Teller (BET) Surface Area and Particle Size
Specific surface area is one the important factors in the heavy metal adsorption case study. Figure 3 and Table 4 present the data of BET experiment on different samples of pH-adjusted sawdust. The surface area of raw sawdust (SD6) is 4.2 m2/g. This amount is relatively good compared with the other bio-adsorbents [30]. As was expressed before, the particle size of the sawdust was limited to 100–150 µm. The particle size directly effects on the surface area. This limitation was chosen based on the previous studies [27,31]. Intra-particle diffusion research shows that the adsorption rate is directly under the influence of particle size [32]. Reduction in the particle size can lead to an increase in the surface area and then, augmentation of the adsorption opportunity and capacity at the outer surface of the sawdust. On the other hand, the possibility of intra-particle diffusion from the outer surface into the inner parts and the pores of the material is a noteworthy fact. The diffusional resistance to mass transfer is less for fine particles. Diffusional path length, mass transfer resistance, contact time, and blockage of some diffusional path are a number of factors which can be effective on the adsorption capacity and are influenced from the particle size.
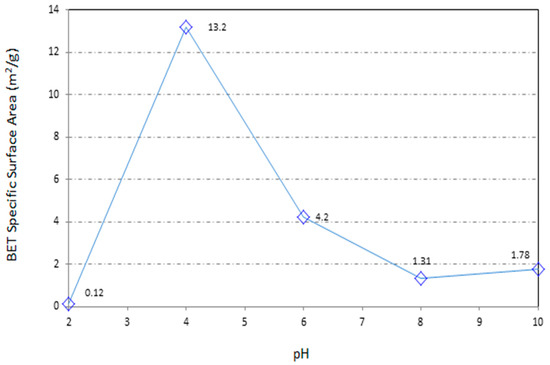
Figure 3.
Trend of the change in the specific surface area (SSA) of sawdust during the chemical treatments.

Table 4.
Pore Brunauer–Emmett–Teller (BET) data of different sawdust samples.
The acidic treatment of the sawdust as it was expected before, leads to activation of the surface and also generation of the pores. The optimum acidic treatment of the wood bio-waste in this research is pH = 4 to have a higher specific surface area.
3.4. Atomic Adsorption Spectrometry (AAS) “Adsorption Behavior” Results
Figure 4, Figure 5, Figure 6, Figure 7 and Figure 8 illustrate the comparison of Pb(NO3)2 adsorption between bentonite and different mix designs of the bentonite and the pH-adjusted sawdust. Due to the presented results, the sawdust at pH = 4 is more effective for improving the adsorption capacity of the bentonite than the others.
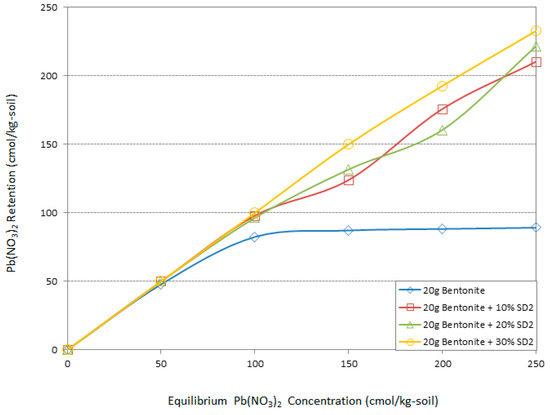
Figure 4.
The effect of different amounts of sawdust (pH = 2) on Pb adsorption by bentonite.
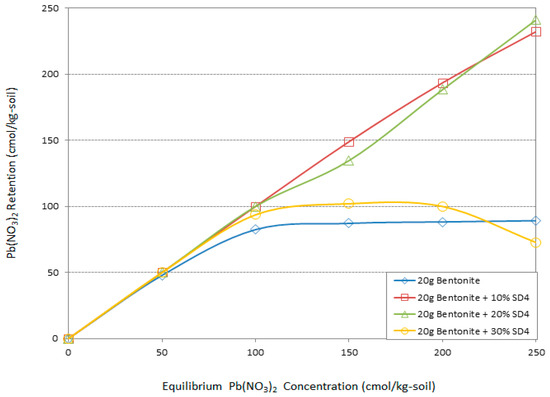
Figure 5.
The effect of different amounts of sawdust (pH = 4) on Pb adsorption by bentonite.
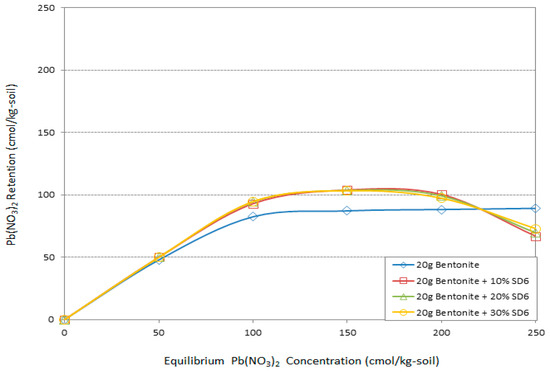
Figure 6.
The effect of different amounts of raw sawdust (pH = 6) on Pb adsorption by bentonite.
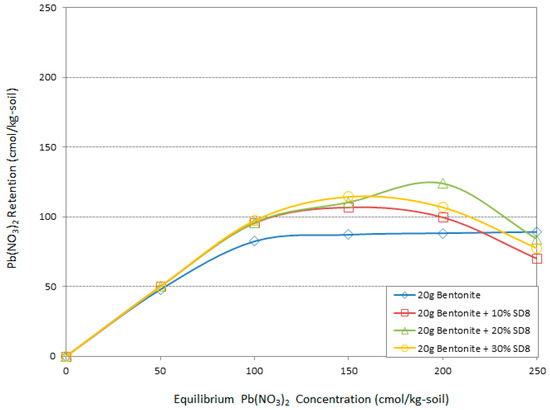
Figure 7.
The effect of different amount of sawdusts (pH = 8) on Pb adsorption by bentonite.
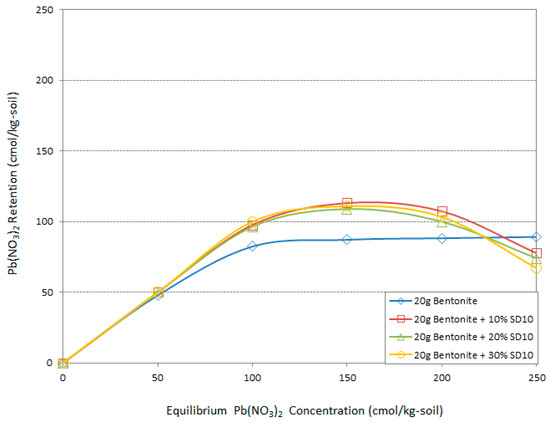
Figure 8.
The effect of different amounts of sawdust (pH = 10) on Pb adsorption by bentonite.
Figure 4 and Figure 5 demonstrate the effect of the acidic treatment of the sawdust on improving the environmental behavior of the bentonite. As it can be seen from the graphs, all the specimens have the same adsorption pattern in low Pb concentration (50 cmol/kg-soil). As the concentration increased to 100 cmol/kg-soil and more, the specimens with sawdust in pH = 2 behaved differently compared with the bentonite, while the specimens including sawdust in pH = 4 were divided into two trends of adsorption improvement. Overall, the acidic treatment had a significant effect on the Pb adsorption by bentonite. According to Figure 4 addition of 10%, 20%, and 30% sawdust in pH = 2 increased the adsorption capability of the bentonite in 100 cmol/kg-soil Pb by 15%, 15%, and 18%, respectively. As the heavy metal concentration increased to150 cmol/kg-soil, the adsorption behavior of the bentonite followed a constant line by sorption of 88 cmol/kg-soil of Pb contaminant. The involvement of 10%, 20%, and 30% SD2 ameliorated the adsorption capacity of the bentonite by 24%, 30%, and 42%, respectively, in contact with 150 cmol/kg-soil Pb.
In high Pb concentrations the same trend continued. According to the Figure 4 and Figure 5, while sawdust is treated in the most acidic environment (pH = 2), the maximum amount participation of sawdust had a more positive effect on the adsorption capacity of the bentonite. Conversely, this improvement occurred in the presence of the lowest percentage of the SD4. Increases of 52% and 58% in the adsorption efficiency of the bentonite were the consequences of the participation of 30% SD2 and 10% SD4 in 200 and 250 cmol/kg-soil concentration of Pb, respectively.
In the other part of the study, the sawdust was added in different amounts to the system without any chemical treatments. The main target was to monitor how much the chemical treatments improved the adsorption system. Thus, as with the other groups, three different amounts of raw sawdust (10%, 20%, 30%) were added to the bentonite and the system was put into contact with different Pb concentrations. There are two important points in Figure 6. The first is the same effect and behavior of the different amount of raw sawdust. The other point is the negative effect of raw sawdust on adsorption capacity of the system in the maximum Pb concentration.
It can be seen from the Figure 6 that 5%, 12%, 11%, and 6% are the maximum positive effect values of raw sawdust on the efficiency of the adsorption capacity of bentonite in contact with 50, 100, 150, and 200 cmol/kg-soil Pb concentrations, respectively. On the other hand, sawdust decreased the efficiency of the system up to 9% at a concentration of 250 cmol/kg-soil Pb.
The Pb(NO3)2 retention efficiency of bentonite including alkali sawdust is presented in Figure 7 and Figure 8. Generally, the addition of different amounts of alkali sawdust could be effective, except in 250 cmol/kg-soil Pb concentrations as in raw sawdust. The improvement was greater than that of raw sawdust but much less than that of acidic sawdust. According to Figure 7 from 50 to 150 cmol/kg-soil heavy metal concentration the presence of 30% SD8 was more effective than the others, while decreasing this amount to 20% SD8 helped the adsorption system better in high concentrations from 150 to 250 cmol/kg-soil Pb. An addition of 30% SD8 led to 5%, 15%, and 18% improvements in adsorption of Pb with the bentonite retention system. On the other hand, 20% SD8 increased the Pb retention by 18% at a 200 cmol/kg-soil concentration. Otherwise, loss adsorption rate was the lowest when 250 cmol/kg-soil Pb was presented in the system. Figure 8 demonstrates the maximum potential of SD10 to improving the Pb adsorption capability of bentonite. While the trends follow the same pattern as SD8, the effect of sawdust dosage is different. At almost all concentrations the minimum dosage 10% of the bentonite weight led to a better function than the others. The 5%, 15%, 17%, and 10% improvements in adsorption capacity were the result of the addition of 10% SD10. It is noticeable that increasing the alkaline degree did not meliorate the adsorption behavior of the bentonite in maximum Pb concentration in this study.
3.5. Adsorption Mechanisms
3.5.1. Na-Bentonite Adsorption Mechanism
The retention and attenuation of heavy metal characteristics in soils are particularly influenced by soil structure, composition, and the environmental condition of pore fluid. Na-bentonite is a montmorillonite-rich clay. Thus, it is expected that the geo-environmental behavior of the system follows montmorillonite characteristics. The high cation exchange capacity (CEC) of montmorillonite clay mineral is the most notable aspect, which is responsible for obtaining a favorable heavy metal retention capacity of the soil [33]. Otherwise, specific surface area (SSA) is the other part of bentonite to provide an appropriate condition to heavy metal adsorption. Montmorillonite has a very large specific surface area (800 m2/g) and a very small thickness (about 1 nm). Affected by these characteristics, the double -layer repulsive force can dominate over the van der Walls attractive force. Such conditions are likely to lead to more parallel layer arrangements. In sodium montmorillonite, unrestrained double layer swelling in pure water (due to water uptake) can result in the dispersion of the unit layers and the formation of a gel phase, where the plate-like unit layers form a “house-of-cards” structure [34]. Na-bentonite, through the montmorillonite mineral, can adsorb Pb via two different mechanisms [35,36]: (1) cation exchange in the interlayers resulting from the interactions between ions and negative permanent charge; and (2) formation of inner-sphere complexes through Si–O_ and Al–O_ groups at the clay particle edges.
Montmorillonite is a clay mineral with substantial isomorphic substitution. The negative charges which generated by isomorphic substitution balances by exchangeable cations in the 2:1 layers. In the different environmental conditions such as pressure, temperature, pH, and chemical and biological composition of the water, bentonite adsorbs the heavy metals in different types and concentrations. Cation exchange reactions are a response to changes in the environmental conditions of clay. This point is noteworthy in that the exchange reactions lead to the changes in physical and physicochemical properties while not affecting the structure of clay particles directly. In total, three sources of clay exchange capacity are known.
Isomorphous substitution: Mg2+ for Al3+ in the octahedral sheets and Al3+ for Si4+ in the silica sheet are the most common isomorphous substitution in the bentonite. This is the major source of bentonite exchange capacity.
Broken Bonds: The exchange sites along particle edges and on non-cleavage surfaces just contribute up to 20 percent of the total exchange capacity in the smectites. This source is the major source for kaolinite mineral exchange capacity. Otherwise, the importance of broken bond sites decrease with increasing the particle size.
Replacement: The hydrogen of an exposed hydroxyl is replaced by another type of cations.
3.5.2. Sawdust Adsorption Mechanism
According to the previous studies on the adsorption of heavy metals by sawdust, the ion exchange and hydrogen bounding were known as the principal sawdust heavy metal adsorption mechanism [31]. The components and complexing properties of the sawdust, the properties of heavy metals and the adsorption behavior, such as the effect of pH of the aqueous media are all the factors which support and effect on this concept. Pb(NO3)2 cations are readily form complexes with O-, N-, S-, or P-, containing functional groups in polymer materials [31]. Cellulose, lignin, and many hydroxyl groups, such as tannins or other phenolic compounds form the cell walls of sawdust while all of these components are active ion exchange compounds. Lignin, the third major component of the wood cell wall, is a polymer material. The lignin molecule is built up from the phenylpropane nucleus, i.e., an aromatic ring with a three-carbon side chain. Vanillin and syringaldehyde are the two of other basic structural units of lignin molecule. The lignin content of hardwoods is usually in the range of 18–25%, whereas that of softwoods varies between 25% and 35%. However, tropical hardwoods can exceed the lignin content of many types of softwood.
Due to the modification process of sawdust in many studies with some other materials, more extractive sites, such as N-, S-, and P-containing groups, are introduced to sawdust. The materials which were used for sawdust chemically treatments included dyestuff [37], polyacrylamide [32], carboxylated-polyethylenimine (CPEI) [38], ZrO2 [39], and other chemicals [40]. Consequently, the binding ability of modified sawdust will be enhanced. Based on the electron-donating nature of the O-, S-, N-, and P-containing groups in sawdust materials and the electron-accepting nature of heavy metal ions, the ion exchange mechanism could be preferentially considered. Pb2+ as a divalent heavy metal ion may attach itself to two adjacent hydroxyl groups and oxyl groups which can donate two pairs of electrons to the metal ion, following this reaction forming four coordination number compounds and releasing two hydrogen ions into solution.
These mechanisms are shown in the following equations [31]:
Ion exchange
2(−ROH) + M2+ = 2(RO) M + 2H+
−ROH + M(OH)+ = (−RO)M(OH) + H+
Hydrogen bonding
where M represents the metal ions and R represents the matrix of sawdust.
2(−ROH) + M(OH)2 = (−ROH)2 + M(OH)2
It can be difficult to distinguish between the two mechanisms. Adsorption of metal ions from solutions by solid phase can occur with formation of surface complex between the adsorbed ligand and the metal. However, the sites on modified sawdust can also contribute to the adsorption process.
The other part of the sawdust that functions in the retention of the contaminant process is the physical adsorption mechanism. The acidic modification led to the activation of the surface and creation of well-developed pores. Thus, beside the chemical adsorption mechanisms, due to the pore sizes the physical adsorption mechanism was the other factor which participated in the Pb retention process.
With respect to the optimum dosage of sawdust, it could be said that during a low adsorbent dose, all types of surface sites are entirely exposed for sorption, and the surface reaches saturation faster as a consequence, leading to a higher sorption capacity. However, at higher particle participation the availability of higher energy sites decreases with a larger fraction of lower energy sites becoming occupied, resulting in a lower sorption capacity.
3.6. pH Variation of the System
One of the other effective factors with respect to the interaction of the solid phase including bentonite + pH-adjusted sawdust and heavy metal electrolyte is the changes in pH of the system. The presence of heavy metal in the system leads to pH reduction [9] and the soils with high buffering capacity are more resistance to pH changes [41]. Figure 9 illustrates the changes of pH versus increasing Pb concentrations. As expected, with increasing Pb concentration the pH of the system was decreased. The pH reduction pattern follows the Pb retention pattern with different mix designs. As can be seen from Figure 9 from the optimum specimens and raw bentonite, the sample including 10% SD4 showed more resistance to the pH reduction. On increasing the Pb concentration to 200 cmol/kg-soil, more the curves coincide. In fact, at this concentration of contaminant and above, all the active phases in Pb retention are filled with the heavy metal ions and the system is not able to adsorb more contaminants.
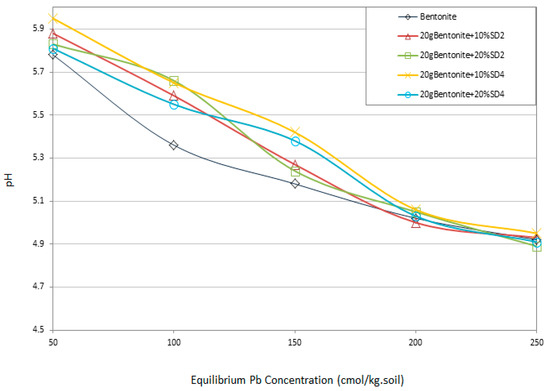
Figure 9.
pH variation of four optimum specimens and bentonite versus the Pb concentrations.
The equilibrium is quite dependent on pH of the aqueous solution. At lower pH, the H+ ions compete with Pb cations for the exchange sites on the sawdust, thereby partially releasing the latter. The Pb cations are completely released under circumstances of extreme acidic conditions. In most cases, the percentage of adsorption of Pb ions increased with an increase in pH up to a certain value and then decreased with further increase of pH. Due to different properties of various heavy metal ions and different sawdust materials, the maximum adsorption took place in a slightly different pH range for different metals.
In a certain pH range, for one specific heavy metal there may be a number of species present in solution, such as M, MOH+, M(OH)2, etc. At a lower pH, the positive charged Pb ion species may compete with H+ and be adsorbed at the surface of the sawdust by ion exchange mechanism. A decrease in adsorption at high pH is due to the formation of soluble hydroxyl complexes. With an increase in pH, metal ion species, mainly neutral, may be adsorbed by hydrogen bonding mechanism along with ion exchange.
As a result, the adsorption of Pb decreased with decreasing pH because the aluminol and silanol groups are more protonated and, hence, they are less available to retain the Pb contamination. This effect is strongly evident for Pb. The reason of this behavior is that the surface complexation reactions are influenced also by the electrostatic attraction between the surface charge and the dissolved ions. In fact, since lead has larger ionic radius (1.20), it has lower charge density and, therefore, is more affected by the protonation of the surface groups that determine a reduction of the adsorption sites on clay [42].
3.7. Buffering Capacity
In order to ensure the results presented in Figure 4, Figure 5, Figure 6, Figure 7 and Figure 8, the changes of pH in the optimum adsorption specimens versus changes of acid concentration were measured. The monitoring of this point could be the basic criteria in the evaluation of heavy metal adsorption and retention capacity in the geo-environmental projects [33]. According to Figure 10, the samples including 10% and 20% SD4 could improve the buffering capacity of the bentonite as well. The result of buffering capacity experiment complies with the results of adsorption behavior obtained from AAS.
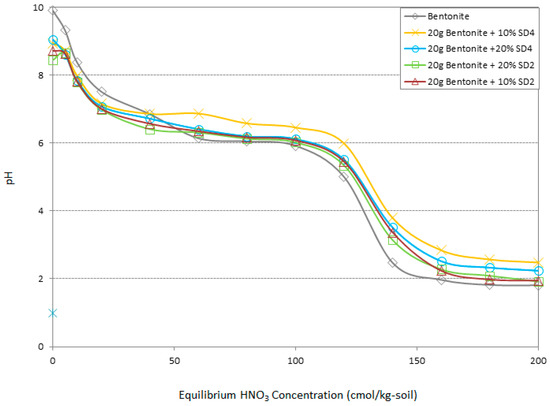
Figure 10.
Comparison of the buffering capacity in the optimum specimens and bentonite.
One of the major components of clayey soils in arid and semi-arid lands is carbonate. This component of clay soils contributes significantly to the contaminant attenuation properties of a soil [43]. The most common of the carbonate minerals found in soils is calcite (CaCO3), derived primarily from calcareous parent materials [44]. Generally, all carbonate groups, are known to be poorly soluble in water [45]. Heavy metals are, for the most part, retained in the clay soils by exchangeable, carbonate, hydroxide and organic phases and mechanisms. Among the entirely different components contributing to contaminant attenuation, carbonates play an important role [46,47]. Usually the carbonate phase contributes to the contaminant retention process when the pH is in excess of 5. It has been shown [41] that the buffer capacities of soils are strongly affected by the carbonate content and CEC of the soils. Calcite (CaCO3) mineral functions well as a buffer for pH changes in the soil–water system. This mineral is believed to be good adsorbent of heavy metals [48,49].
High contents of carbonates caused an increase of extract pH, changing the conditions for desorbing ions [50]. In the presence of carbonate, the CEC may or may not be changed, due to the fact that the CEC of carbonates is relatively small and its contribution to the overall CEC of the soil may not be effectively felt. Generally, carbonates contribute to the prevention of contaminant transport in soils in three different ways: firstly, by neutralizing the acidity of soil; secondly, by raising the pH level of the soil pore fluid; and finally by reacting with different components of contaminants to form insoluble constituents. Solid phase carbonates contribute to the attenuation characteristics of soil through an acid-consuming process.
This reaction process can be summarized as follows:
For the calcite reaction:
H+ + CaCO3 → Ca2+ + HCO3−
For the dolomite reaction:
2H+ + CaMg(CO3)2 → Ca2+ + Mg2+ + 2HCO3−
At low pH levels, the dissolution of carbonates originally present in the soil appears to be responsible for the decreased amounts of heavy metals retained by the carbonate phase [51,52]. So, according to the results from buffering capacity experiment sawdust could be nominated as the other phase of the system to improve the buffering capacity while the carbonate is not active due to the pH.
3.8. Scanning Electron Microscope (SEM)
The results of the morphological study are illustrated in Figure 11 and Figure 12. The raw sawdust is recognized by a structure in the form of filaments, which exists among various other materials. This type of arrangement causes the anisotropic character or nature of the sawdust. Thus, the main aim of the acidic and/or alkali treatment was to induce some changes in the structure of the sawdust in order to assign a special characteristic to the surfaces in each treatment. The acidic treatment was carried out in this research to make the surface active. The surface was activated with the well-developed pores. Our results show that the acidic treatment was effective and the operation was parallel to the intended aim of inducing or enhancing the environmental adsorption capacity of the raw sawdust. The empty channels and pores on the sawdust particles are the consequences the eliminating of filaments. The acidic treatment reacted with the sawdust components and created the honeycomb structure, which agreed with the findings by [53,54].

Figure 11.
Scanning Electron microscope (SEM) micrograph of acidic modified sawdust.

Figure 12.
Scanning Electron microscope (SEM) micrograph of Bentonite and sawdust Pb-loaded
A typical SEM micrograph of 150 cmol/kg-soil Pb loaded bentonite + 10% SD4 is given in Figure 12. The micrograph reveals the heterogeneous nature of surface morphology of the clay sample. The surface fabric changed to flocculated after Pb adsorption. The Pb-loaded sawdust fiber also appears beside the bentonite particle. Nearly all the macro-pores are filled by Pb through the sawdust functional surface adsorption mechanisms.
3.9. XRD Analysis
The interaction between clay and heavy metal contaminants changes the microstructure of the clayey soils by affecting the forces between the clay particles. Evaluation of the XRD spectrum is known as a criterion in determining the microstructure of the soils. The peak of the clay soils with the dispersive structure has a more intensity in comparison to the soils with the flocculated structure. In order to the investigate of the effect of Pb on the microstructure of the studied samples, the X-ray spectra of the optimum and the blank samples were prepared.
Figure 13 illustrates the result of the X-ray diffraction of the bentonite before and after the interaction with 200 (cmol/kg-soil) Pb, and bentonite with 10% SD4 in the presence of 200 (cmol/kg-soil). Based on the presented results, with the presence of the Pb, the main peak intensity decreased. This fits the fact that, the increasing of the salt concentration in the pore fluid leads to the formation of the flocculated structure and as a result the peak intensity of the major peaks decreased. Presence of the 10% SD4 improved the peak intensity of the sample in the presence of the 200 (cmol/kg-soil) Pb. So, the results of the atomic adsorption agree with the XRD diffraction results. It is noteworthy that, increasing the salt concentration of the pore fluid did not affect the position of the major peaks; it just affected the peak intensity.
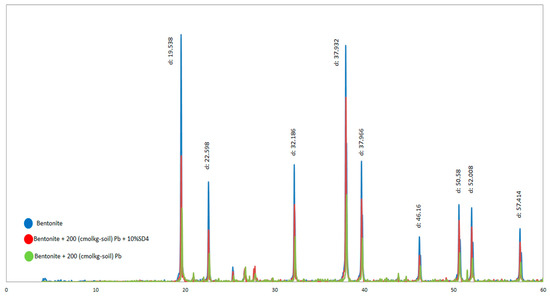
Figure 13.
Changes of the peak intensity of the optimum and the blank samples X-ray Diffraction (XRD).
3.10. Chemical Oxygen Demand (COD)
Decreasing in the non-biodegradable organic matters leads to decreasing the COD amount [55]. The interaction between clay minerals and organic compounds makes many complexes of different stabilities and properties. In this case there are many variables such as silicate layers, the inorganic cations, water, and the organic molecules. In the recent years, different studies have been conducted concerning the application of sawdust to heavy metal removal from aqueous solutions. Most of them have not monitored the amount of COD of the solution after the removal process. It is noticeable that the use of aquatic plants, microorganisms, and wood-based materials such as sawdust and bark increases the COD of water [24]. Thus, the main target of this part of experiment was to survey the effect of pH and dosage of sawdust, and Pb concentration on the presence of COD in the solution.
Figure 14 illustrates the trend of COD amounts of different specimens with changing Pb concentration. The maximum COD amount belongs to the specimens with the highest alkali degree (SD10) in this study and the minimum amount of COD appertains to the most acidic samples (SD2). It can be stated that in an acidic solution both surface functional groups of the bentonite and sawdust got the positive charge while in the alkali it converts to the negative. So, the adsorption efficiency of the organic matters with negative charge is high in the acidic situation. On the other hand, for the acidic treatment of sawdust HCl was used to increase the proportion of active surfaces and to prevent the elution of tannin compounds that would stain the treated water and that would greatly increase COD [24]. However, parallel with increasing the Pb concentration from 50 cmol/kg-soil to 150 cmol/kg-soil the COD amount decreased, but with increasing the cation concentration to 200 and 250 cmol/kg-soil the COD amount showed an incremental leap. Thus, the acidic treatment could be effective for controlling the COD amount in the solution.
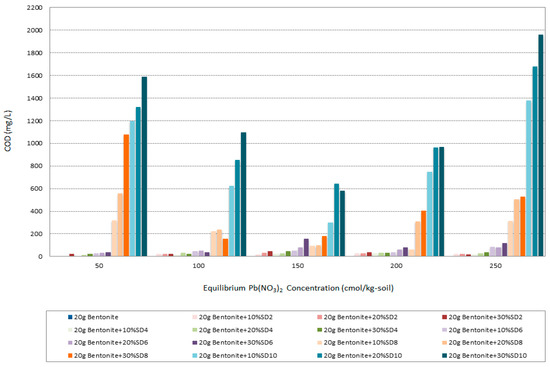
Figure 14.
Chemical Oxygen Demand (COD) variation of all the specimens in different Pb(NO3)2 concentrations.
4. Conclusions
The modification of raw sawdust into an acidic adsorbent has led to the improvement of its environmental behavior. The natural acidification processes are different for various sawdust types depending on the lignin, cellulose, and hemicellulose content and the structural characteristics of the sawdust. The results from this research have suggested that Na-bentonite plus 10% to 20% SD4 could eliminate a wide range of Pb contamination up to the 250 (cmol/kg-soil) and therefore can be used for the clay liner in a landfill. The given recommendation is based on the following findings from this current research.
- The acid treatment (also called acidification) of the sawdust has led to the improvement in its adsorption capacity through the activation of particle surfaces and the creation of the honeycomb structures.
- The acid treatment has caused an increase in the specific surface area of the sawdust particles. Thus, due to the improved pore sizes in the process, beside the chemically adsorption mechanisms the physical adsorption mechanisms have also participated in retaining the Pb contaminant.
- The reduction in the particle size can lead to increased specific surface area (SSA) and the subsequent augmentation in the adsorption opportunity and adsorption capacity of the outer sawdust surface.
- The high specific surface area (SSA) and high cation exchange capacity (CEC) of the Na-bentonite clay with high plasticity characteristics has increased the capability for high Pb retention through different adsorption mechanisms out of a solution with 100 (cmol/kg-soil) of Pb concentration, without using any sawdust additive.
- The 10% SD4 and the 30% SD2 designs had substantial effects on the Pb adsorption capacity of the Na-Bentonite and therefore were the optimum mix designs. These are the 10% sawdust additive of pH4 and the 30% sawdust additive of pH2.
- The presence of 10% SD4 or 30% SD2 could improve the adsorption capacity of the system as it was capable of adsorbing 100% of the Pb for concentrations up to 150 (cmol/kg-soil).
- For higher Pb concentrations, i.e., between 150 (cmol/kg-soil) and 250 (cmol/kg-soil), the given optimum mix designs of 10% SD4 and the 30% SD2 were capable of 58% adsorption of the Pb.
- The ion exchange and the hydrogen bonding were the main mechanisms introduced by the chemical adsorption capability of the sawdust. Also, the surface complexes and the functional groups have affected the cation adsorption capacity of the sawdust.
- The buffering capacity of the system has been improved by the addition of 10% SD4. Thus the change in pH and the resulting improved buffering capacity of a clay linear system in contact with heavy metals were the most significant geo-environmental behaviors that have aided the adsorption.
- The maximum COD was found with specimens of the highest alkali degree, i.e., involving the SD10 samples, while the minimum COD was found with specimens of the most acidic degree, i.e., involving the SD2 samples. Therefore, the acidification of the sawdust could solve the problem associated with the COD creation, which also corresponds to the maximum adsorption capacity.
Author Contributions
P.M. conducted Data curation, Formal analysis, Investigation, Software, Validation and Writing—original draft. Funding acquisition, Methodology, Project administration, Resources and Supervision were provided by M.R.S. and H.A.A. C.S. conducted Visualization and Writing—review & editing.
Funding
This research was partly funded by the iconic ‘Grant for Researches Associated with the Solid Waste Management Cluster’ (grant number 1001/CKT/870023), Universiti Sains Malaysia.
Acknowledgments
This research is funded by University Sains Malaysia (USM) under Ionic grant scheme (Grant no. 1001/CKT/870023) for research associated with the Solid Waste Management Cluster, Engineering campus, University Sains Malaysia. The authors would like to acknowledge the University Sains Malaysia (USM) for provision of research grant to conduct this work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bradbury, M.H.; Baeyens, B. Sorption modelling on illite. Part II: Actinide sorption and linear free energy relationships. Geochim. Cosmochim. Acta 2009, 73, 1004–1013. [Google Scholar] [CrossRef]
- Cruz-Guzmán, M.; Celis, R.; Hermosín, M.C.; Koskinen, W.C.; Nater, E.A.; Cornejo, J. Heavy Metal Adsorption by Montmorillonites Modified with Natural Organic Cations. Soil Sci. Soc. Am. J. 2006, 70, 215. [Google Scholar] [CrossRef]
- Yang, S.; Zhao, D.; Zhang, H.; Lu, S.; Chen, L.; Yu, X. Impact of environmental conditions on the sorption behavior of Pb(II) in Na-bentonite suspensions. J. Hazard. Mater. 2010, 183, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Ozdes, D.; Gundogdu, A.; Kemer, B.; Duran, C.; Senturk, H.B.; Soylak, M. Removal of Pb(II) ions from aqueous solution by a waste mud from copper mine industry: Equilibrium, kinetic and thermodynamic study. J. Hazard. Mater. 2009, 166, 1480–1487. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.E.; Olin, T.J.; Bricka, R.M.; Adrian, D.D. A review of potentially low-cost sorbents for heavy metals. Water Res. 1999, 33, 2469–2479. [Google Scholar] [CrossRef]
- Ho, S.H.; Wang, D.; Wei, Z.S.; Chang, J.S.; Ren, N.Q. Lead removal by a magnetic biochar derived from persulfate-ZVI treated sludge together with one-pot pyrolysis. Bioresour. Technol. 2018, 247, 463–470. [Google Scholar]
- Wei, Z.; Luo, S.; Xiao, R.; Khalfin, R.; Semiat, R. Characterization and quantification of chromate adsorption by layered porous iron oxyhydroxide: An experimental and theoretical study. J. Hazard. Mater. 2017, 338, 472–481. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Semiat, R. Applying a modified Donnan model to describe the surface complexation of chromate to iron oxyhydroxide agglomerates with heteromorphous pores. J. Colloid Interface Sci. 2017, 506, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Wei, Z.; Dionysiou, D.D.; Spinney, R.; Hu, W.P.; Chai, L.; Yang, Z.; Ye, T.; Xiao, R. Mechanistic insight into reactivity of sulfate radical with aromatic contaminants through single-electron transfer pathway. Chem. Eng. J. 2017, 327, 1056–1065. [Google Scholar] [CrossRef]
- Ferrero, F.; Gaglia Prati, M.P. Coal fly ash and alginate for the removal of heavy metals from aqueous solutions. Ann. Chim. 1996, 86, 125–132. [Google Scholar]
- Mathur, A.; Khare, S.K.; Rupainwar, D.C. Removal of heavy metals from main sewer-water of Varanasi city by adsorption on fly ash and blast furnace slag. J. Ind. Pollut. Control 1989, 5, 52–57. [Google Scholar]
- Ouki, S.; Kavannagh, M. Treatment of metals-contaminated wastewaters by use of natural zeolites. Water Sci. Technol. 1999, 39, 115–122. [Google Scholar] [CrossRef]
- Kumar, P.; Dara, S.S. Binding heavy metal ions with polymerized onion skin. J. Polym. Sci. Polym. Chem. Ed. 1981, 19, 397–402. [Google Scholar] [CrossRef]
- Khalid, N.; Ahmad, S.; Kiani, S.N.; Ahmed, J. Removal of Mercury from Aqueous Solutions by Adsorption to Rice Husks. Sep. Sci. Technol. 1999, 34, 3139–3153. [Google Scholar] [CrossRef]
- Wei, Z.; Seo, Y. Trichloroethylene (TCE) adsorption using sustainable organic mulch. J. Hazard. Mater. 2010, 181, 147–153. [Google Scholar] [CrossRef] [PubMed]
- American Society for Testing and Materials (ASTM). Annual Book of ASTM Standards; American Society for Testing and Materials: Philadelphia, PA, USA, 1992. [Google Scholar]
- Hendershot, W.; Duquette, M. A simple barium chloride method for determining cation exchange capacity and exchangeable cations. Soil Sci. Soc. Am. J. 1986, 50, 605–608. [Google Scholar] [CrossRef]
- Moore, D.; Reynolds, R. X-ray Diffraction and the Identification and Analysis of Clay Minerals; Oxford University Press: Oxford, UK, 1989. [Google Scholar]
- Hesse, P. A Textbook of Soil Chemical Analysis; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
- Fagerlund, G. Determination of specific surface by the BET method. Matériaux Constr. 1973, 6, 239–245. [Google Scholar] [CrossRef]
- ASTM D4749. StandardTest Method for Performing the Sieve Analysis of Coal and Designating Coal Size, USA; American Society for Testing and Materials: Philadelphia, PA, USA, 1994. [Google Scholar]
- Brown, P.; Jefcoat, I.; Parrish, D. Evaluation of the adsorptive capacity of peanut hull pellets for heavy metals in solution. Adv. Environ. Res. 2000, 4, 19–29. [Google Scholar] [CrossRef]
- EPA. Process Design Manual, Land Application of Municipal Sludge; EPA-625/1-83-016; Municipal Environmental Research Laboratory: Washington, DC, USA, 1983.
- Argun, M.; Dursun, S.; Ozdemir, C.; Karatas, M. Heavy metal adsorption by modified oak sawdust: Thermodynamics and kinetics. J. Hazard. Mater. 2007, 141, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Tabil, L.; Panigrahi, S. Chemical treatments of natural fiber for use in natural fiber-reinforced composites: A review. J. Polym. Environ. 2007, 15, 25–33. [Google Scholar] [CrossRef]
- Gaballah, I.; Kilbertus, G. Recovery of heavy metal ions through decontamination of synthetic solutions and industrial effluents using modified barks. J. Geochem. Explor. 1998, 62, 241–286. [Google Scholar] [CrossRef]
- Mohajeri, P.; Selamat, M.R.B.; Aziz, H.A.; Vakili, A.H.; Farraji, H. Geo-environmental Behaviour of Pb Contaminated Clay with Sawdust. Environ. Geotech. 2018, 1–40. [Google Scholar] [CrossRef]
- Singh, J.; Mishra, N.; Banerjee, S.; Sharma, Y. Comparative studies of physical characteristics of raw and modified sawdust for their use as adsorbents for removal of acid dye. BioResources 2011, 6, 2732–2743. [Google Scholar]
- Sakurai, K.; Teshima, A.; Kyuma, K. Changes in Zero Point of Charge (ZPC), Specific Surface Area (SSA), and Cation Exchange Capacity (CEC) of kaolinite and montmorillonite, and strongly weathered soils caused by Fe and Al coatings. Soil Sci. Plant Nutr. 1990, 36, 73–81. [Google Scholar] [CrossRef]
- Bulut, Y. Removal of heavy metals from aqueous solution by sawdust adsorption. J. Environ. Sci. 2007, 19, 160–166. [Google Scholar] [CrossRef]
- Ajmal, M.; Khan, A.; Ahmad, S.; Ahmad, A. Role of sawdust in the removal of copper (II) from industrial wastes. Water Res. 1998, 32, 3085–3091. [Google Scholar] [CrossRef]
- Raji, C.; Anirudhan, T. Kinetics of Pb (II) adsorption by polyacrylamide grafted sawdust. Indian J. Chem. Technol. 1997, 4, 157–162. [Google Scholar]
- Yong, R. Geoenvironmental Engineering: Contaminated Soils, Pollutant Fate, and Mitigation; CRC Press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Stewart, D.; Studds, P.; Cousens, T. The factors controlling the engineering properties of bentonite-enhanced sand. Appl. Clay Sci. 2003, 23, 97–110. [Google Scholar] [CrossRef]
- Kraepiel, A.; Keller, K.; Morel, F. A Model for Metal Adsorption on Montmorillonite. J. Colloid Interface Sci. 1999, 210, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Schindler, P.W.; Fürst, B.; Dick, R.; Wolf, P.U. Ligand properties of surface silanol groups. I. surface complex formation with Fe3+, Cu2+, Cd2+, and Pb2+. J. Colloid Interface Sci. 1976, 55, 469–475. [Google Scholar] [CrossRef]
- Suemitsu, R.; Uenishi, R.; Akashi, I.; Nakano, M. The use of dyestuff-treated rice hulls for removal of heavy metals from waste water. J. Appl. Polym. Sci. 1986, 31, 75–83. [Google Scholar] [CrossRef]
- Ibrahim, N. Animation of wood sawdust for removing anionic dyes from aqueous solutions. Polym. Plast. Technol. Eng. 1997, 36, 963–971. [Google Scholar] [CrossRef]
- Setyono, D.; Valiyaveettil, S. Chemically Modified Sawdust as Renewable Adsorbent for Arsenic Removal from Water. ACS Sustain. Chem. Eng. 2014, 2, 2722–2729. [Google Scholar] [CrossRef]
- Anirudhan, T.; Sreedhar, M. Adsorption thermodynamics of Co (II) on polysulphide treated sawdust. Indian J. Chem. Technol. 1998, 5, 41–47. [Google Scholar]
- Yong, R.; Warkentin, B. Buffer capacity and lead retention in some clay materials. Water Air Soil Pollut. 1990, 53, 53–67. [Google Scholar] [CrossRef]
- Abollino, O.; Aceto, M.; Malandrino, M.; Sarzanini, C.; Mentasti, E. Adsorption of heavy metals on Na-montmorillonite. Effect of pH and organic substances. Water Res. 2003, 37, 1619–1627. [Google Scholar] [CrossRef]
- Ouhadi, V.; Yong, R. Impact of carbonate on the efficiency of heavy metal removal from kaolinite soil by the electrokinetic soil remediation method. J. Hazard. Mater. 2010, 173, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, C.; Yong, R.; Gibbs, B. Remediation technologies for metal-contaminated soils and groundwater: An evaluation. Eng. Geol. 2001, 60, 193–207. [Google Scholar] [CrossRef]
- Mulligan, C.; Yong, R. Natural attenuation of contaminated soils. Environ. Int. 2004, 30, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Griffin, R.A.; Shimp, N.F.; Steele, J.D.; Ruch, R.R.; White, W.A.; Hughes, G.M. Attenuation of pollutants in municipal landfill leachate by passage through clay. Environ. Sci. Technol. 1976, 10, 1262–1268. [Google Scholar] [CrossRef] [PubMed]
- Plassard, F.; Winiarski, T.; Petit-Ramel, M. Retention and distribution of three heavy metals in a carbonated soil: Comparison between batch and unsaturated column studies. J. Contam. Hydrol. 2000, 42, 99–111. [Google Scholar] [CrossRef]
- Sipos, P.; Németh, T.; Mohai, I.; Dódony, I. Effect of soil composition on adsorption of lead as reflected by a study on a natural forest soil profile. Geoderma 2005, 124, 363–374. [Google Scholar] [CrossRef]
- Yong, R.; Ouhadi, V.; Goodarzi, A. Effect of Cu2+ ions and buffering capacity on smectite microstructure and performance. J. Geotech. Geoenviron. Eng. 2009, 135, 1981–1985. [Google Scholar] [CrossRef]
- Sulkowski, M.; Hirner, A. Element fractionation by sequential extraction in a soil with high carbonate content. Appl. Geochem. 2006, 21, 16–28. [Google Scholar] [CrossRef]
- Buckman, H.O.; Brady, N.C. The Nature and Properties of Soils; Macmillan: New York, NY, USA, 1968. [Google Scholar]
- Ouhadi, V.; Yong, R.; Sedighi, M. Desorption response and degradation of buffering capability of bentonite, subjected to heavy metal contaminants. Eng. Geol. 2006, 85, 102–110. [Google Scholar] [CrossRef]
- Alvarez, P.; Blanco, C.; Santamarıa, R.; Granda, M. Improvement of the thermal stability of lignocellulosic materials by treatment with sulphuric acid and potassium hydroxide. J. Anal. Appl. Pyrolysis 2004, 72, 131–139. [Google Scholar] [CrossRef]
- Camacho, F.; Gonzalez-Tello, P.; Jurado, E.; Robles, A. Microcrystalline-cellulose hydrolysis with concentrated sulphuric acid. J. Chem. Technol. Biotechnol. 1996, 67, 350–356. [Google Scholar] [CrossRef]
- Rodrıguez, J.; Castrillon, L.; Maranon, E. Removal of non-biodegradable organic matter from landfill leachates by adsorption. Water Res. 2004, 38, 3297–3303. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).