Riverbank Filtration Impacts on Post Disinfection Water Quality in Small Systems—A Case Study from Auburn and Nebraska City, Nebraska
Abstract
1. Introduction
2. Materials and Methods
2.1. Description of the RBF Sites
2.2. Water Quality Analysis
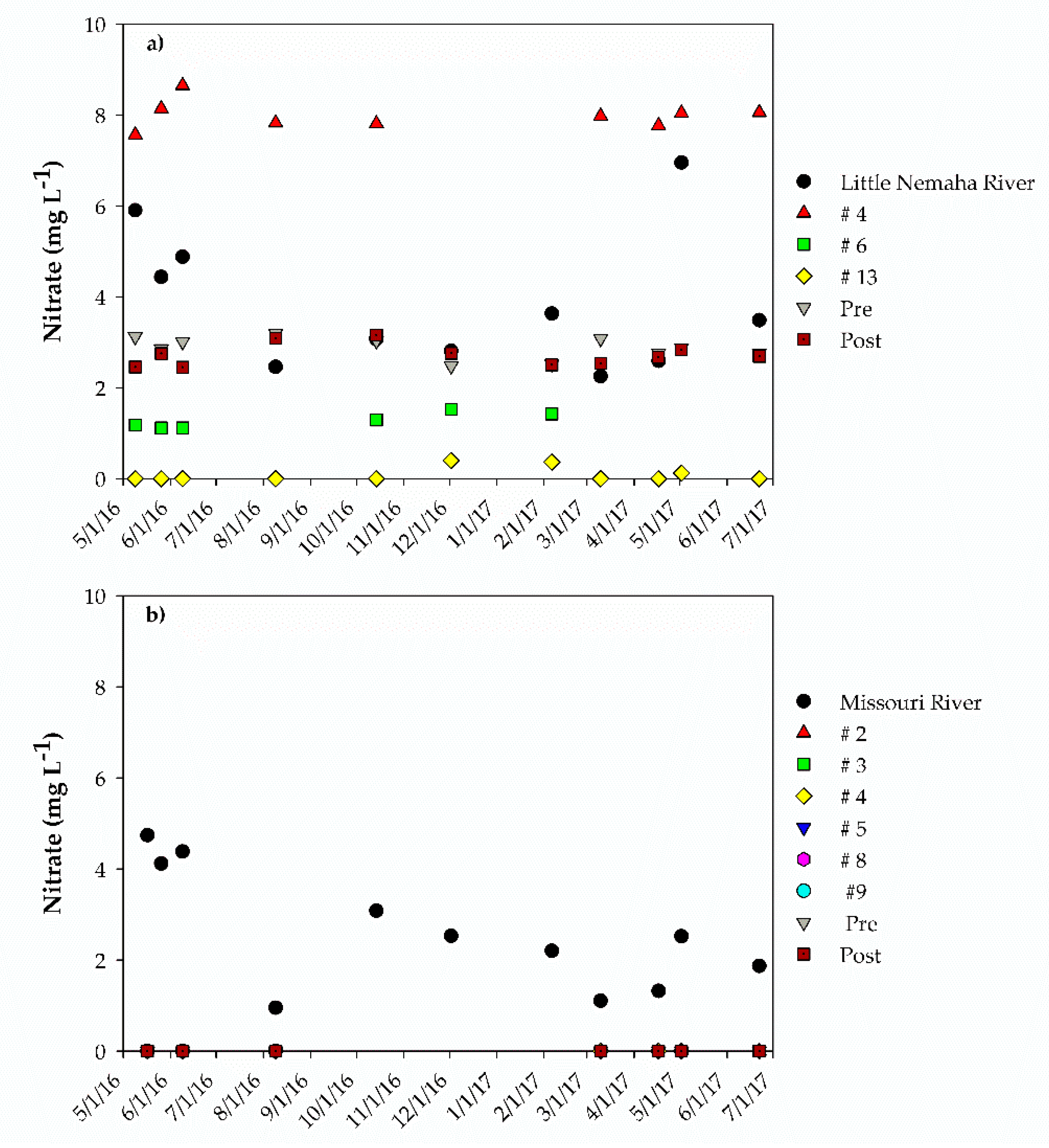
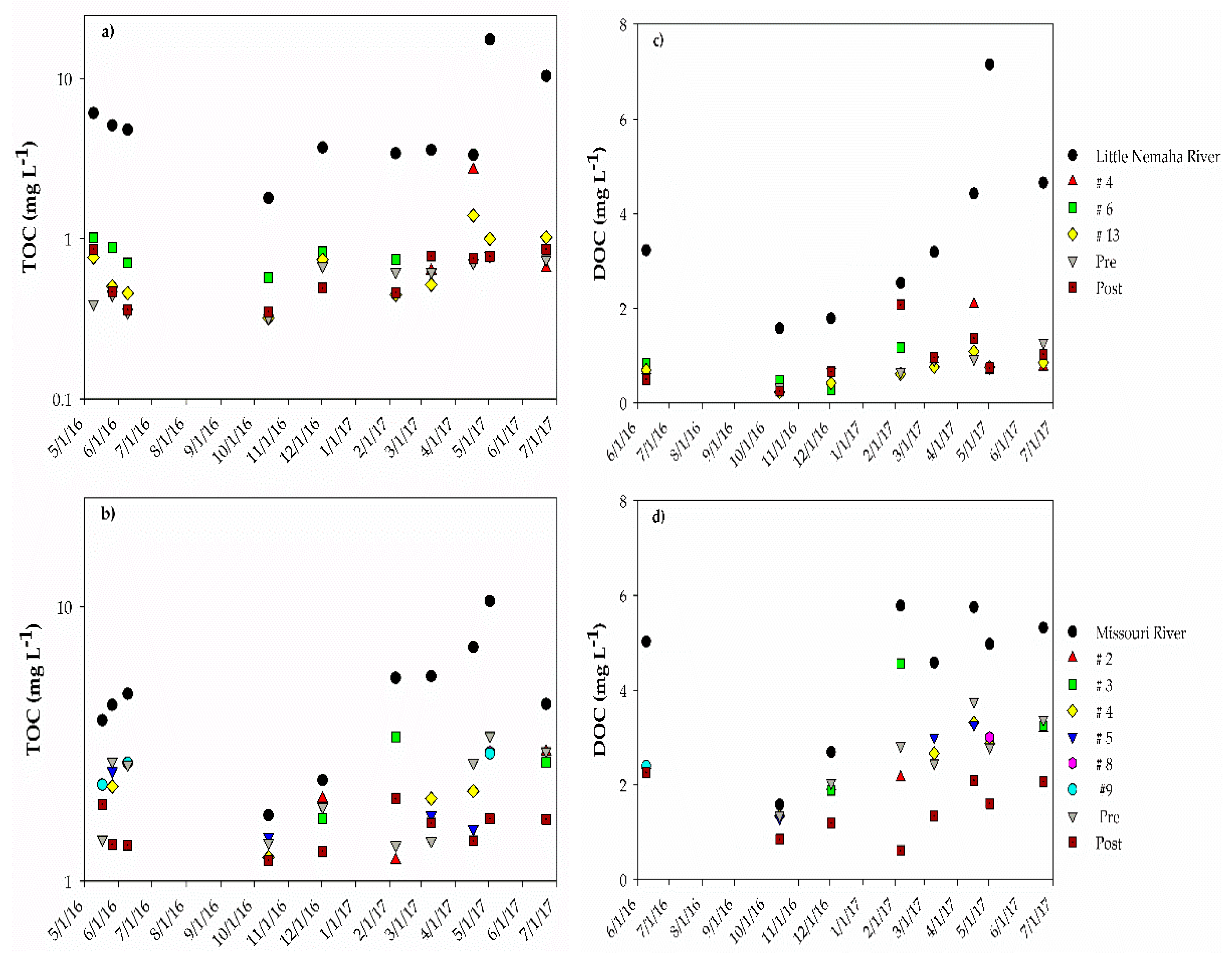
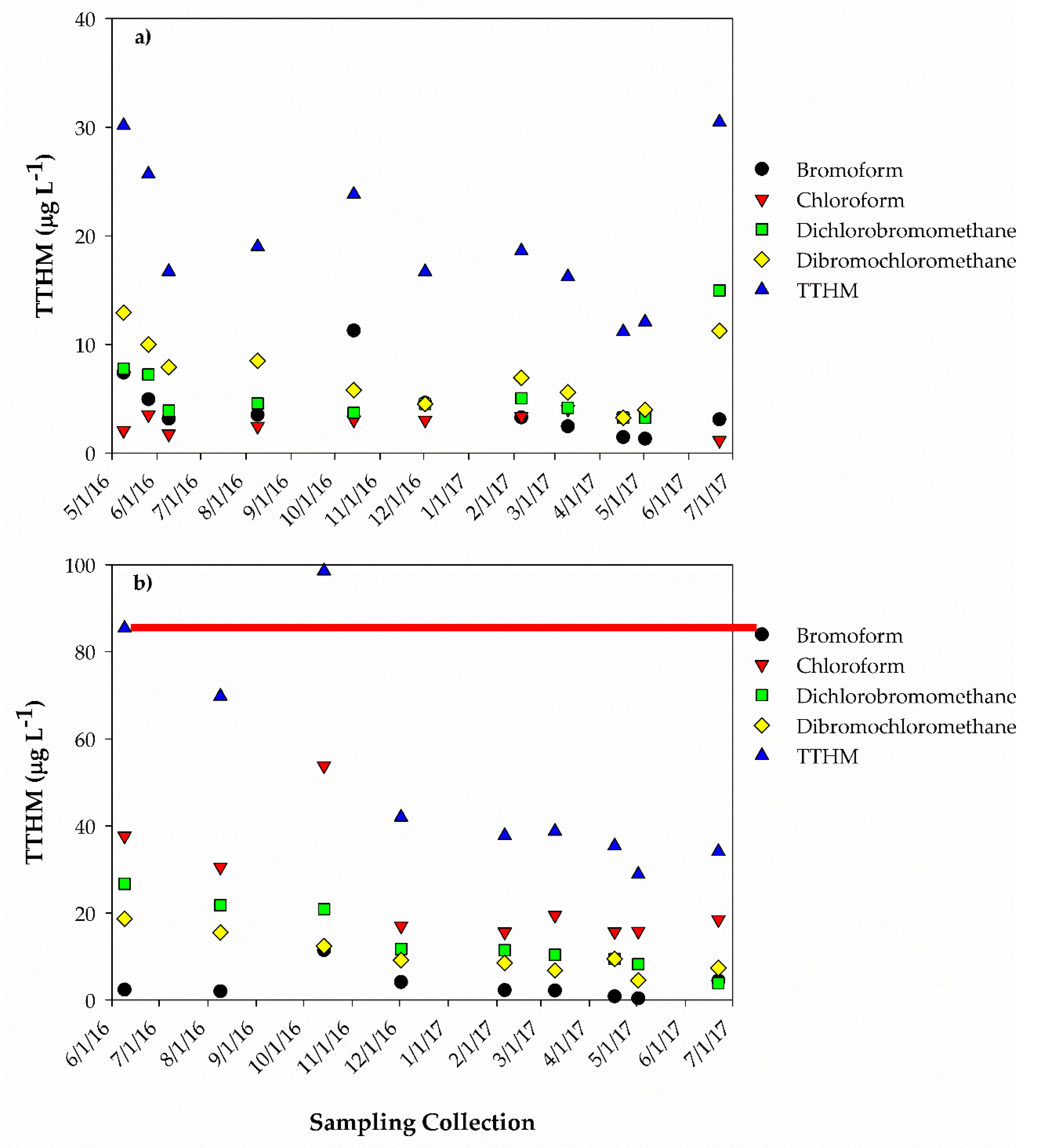
3. Results
3.1. Improvement in Water Quality: Auburn RBF Site
3.2. Improvement in Water Quality: Nebraska City RBF Site
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- U.S. Environmental Protection Agency. Building the Capacity of Drinking Water Systems. Available online: https://www.epa.gov/dwcapacity/learn-about-small-drinking-water-systems (accessed on 14 October 2018).
- Hua, B.; Mu, R.; Shi, H.; Inniss, E.; Yang, J. Water quality in selected small drinking water systems of Missouri rural communities. Beverages 2016, 2, 10. [Google Scholar] [CrossRef]
- Allard, S.; Tan, J.; Joll, C.A.; Von Gunten, U. Mechanistic study on the formation of Cl-/Br-/I-trihalomethanes during chlorination/chloramination combined with a theoretical cytotoxicity evaluation. Environ. Sci. Technol. 2015, 49, 11105–11114. [Google Scholar] [CrossRef] [PubMed]
- Roccaro, P.; Vagliasindi, F.; Korshin, G. Relationships between trihalomethanes, haloacetic acids, and haloacetonitriles formed by the chlorination of raw, treated, and fractionated surface waters. J. Water Supply Res. Tech. AQUA 2014, 63, 21–30. [Google Scholar] [CrossRef]
- Hua, G.; Reckhow, D.A. Characterization of disinfection byproduct precursors based on hydrophobicity and molecular size. Environ. Sci. Technol. 2007, 41, 3309–3315. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Love, N.; Edwards, M. Nitrification in drinking water systems. Crit. Rev. Environ. Control 2009, 39, 153–208. [Google Scholar] [CrossRef]
- Odell, L.H.; Kirmeyer, G.J.; Wilczak, A.; Jacangelo, J.G.; Marcinko, J.P.; Wolfe, R.L. Controlling nitrification in chloraminated systems. J. AWWA 1996, 88, 86–98. [Google Scholar] [CrossRef]
- Wilczak, A.; Jacangelo, J.G.; Marcinko, J.P.; Odell, L.H.; Kirmeyer, G.J. Occurrence of nitrification in chloraminated distribution systems. J. AWWA 1996, 88, 74–85. [Google Scholar] [CrossRef]
- Grellier, J.; Rushton, L.; Briggs, D.J.; Nieuwenhuijsen, M.J. Assessing the human health impacts of exposure to disinfection by-products—A critical review of concepts and methods. Environ. Int. 2015, 78, 61–81. [Google Scholar] [CrossRef]
- Richardson, S.D.; Plewa, M.J.; Wagner, E.D.; Schoeny, R.; DeMarini, D.M. Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: A review and roadmap for research. Mutat. Res. Rev. Mutat. Res. 2007, 636, 178–242. [Google Scholar] [CrossRef]
- Singer, R.C. Human substances as precursors for potentially harmful disinfection by-products. Water Sci. Technol. 1999, 40, 25–30. [Google Scholar] [CrossRef]
- Krasner, S.W.; Kostopoulou, M.; Toledano, M.B.; Wright, J.; Patelarou, E.; Kogevinas, M.; Villanueva, C.M.; Carrasco-Turigas, G.; Marina, L.S.; Fernández-Somoano, A.; et al. Occurrence of DBPs in drinking water of European regions for epidemiology studies. J. AWWA 2016, 108, E501–E512. [Google Scholar] [CrossRef]
- Guilherme, S.; Rodriguez, M.J. Occurrence of regulated and non-regulated disinfection by-products in small drinking water systems. Chemosphere 2014, 117, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Stage 1 and Stage 2 Disinfectants and Disinfection Byproducts Rules. Available online: http://water.epa.gov/lawsregs/rulesregs/sdwa/stage2/regs_factsheet.cfm (accessed on 3 September 2018).
- Ringenberg, D. Regulatory Barriers to Approval of New Technologies for Small Drinking Water Systems. Master’s Thesis, University of Nebraska, Lincoln, NE, USA, 2017. Available online: http://digitalcommons.unl.edu/envengdiss/13/ (accessed on 3 September 2018).
- Ray, C. Worldwide potential of riverbank filtration. Clean Technol. Environ. Policy 2008, 10, 223–225. [Google Scholar] [CrossRef]
- Ray, C.; Melin, G.; Linsky, R.B. River Bank Filtration—Improving Source-Water Quality; Kluwer: Dordrecht, The Netherlands, 2003; 364p. [Google Scholar]
- D’Alessio, M.; Yoneyama, B.; Ray, C. Fate of selected pharmaceutically active compounds during simulated riverbank filtration. Sci. Total Environ. 2015, 505, 615–622. [Google Scholar] [CrossRef] [PubMed]
- D’Alessio, M.; Yoneyama, B.; Kirs, M.; Kisand, V.; Ray, C. Pharmaceutically active compounds: Their removal during slow sand filtration and their impact on slow sand filtration bacterial removal. Sci. Total Environ. 2015, 524–525, 124–135. [Google Scholar] [CrossRef]
- Sudhakaran, S.; Lattemann, S.; Amy, G.L. Appropriate drinking water treatment processes for organic micropollutants removal based on experimental and model studies—A multi-criteria analysis study. Sci. Total Environ. 2013, 442, 478–488. [Google Scholar] [CrossRef] [PubMed]
- Storck, F.R.; Schmidt, C.K.; Lange, F.T.; Henson, J.W.; Hahn, K. Factors controlling micropollutant removal during riverbank filtration. J. AWWA 2012, 4, E643–E652. [Google Scholar] [CrossRef]
- Hoppe-Jones, C.; Oldham, G.; Drewes, J.E. Attenuation of total organic carbon and unregulated trace organic chemicals in U.S. riverbank filtration systems. Water Res. 2010, 44, 4643–4659. [Google Scholar] [CrossRef]
- Hiscock, K.M.; Grischeck, T. Attenuation of groundwater pollution by bank filtration. J. Hydrol. 2002, 266, 139–144. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. National Primary Drinking Water Regulations. Available online: https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations#one (accessed on 15 October 2018).
- Wells, M.J.; Gilmore, T.E.; Mittelstet, A.R.; Snow, D.D.; Sibray, S.S. Assessing decadal trends of a nitrate-contaminated shallow aquifer in Western Nebraska using groundwater isotopes, age-dating, and monitoring. Water 2018, 10, 1047. [Google Scholar] [CrossRef]
- Pennino, M.J.; Compton, J.E.; Leibowitz, S.G. Trends in drinking water nitrate violations across the United States. Environ. Sci. Technol. 2017, 51, 13450–13460. [Google Scholar] [CrossRef] [PubMed]
- Exner, M.E.; Hirsh, A.J.; Spalding, R.F. Nebraska’s groundwater legacy: Nitrate contamination beneath irrigated cropland. Water Resour. Res. 2014, 50, 4474–4489. [Google Scholar] [CrossRef] [PubMed]
- Spalding, R.F.; Exner, M.E. Occurrence of nitrate in groundwater—A review. J. Environ. Qual. 1993, 22, 392–402. [Google Scholar] [CrossRef]
- Ringenberg, D.; Wilson, S.; Dvorak, B. State barriers to approval of drinking water technologies for small systems. J. AWWA 2017, 109, E343–E352. [Google Scholar] [CrossRef]
- Kim, H.-C.; Lee, W.M.; Lee, S.; Choi, J.; Maeng, S.K. Characterization of organic precursors in DBP formation and AOC in urban surface water and their fate during managed aquifer recharge. Water Res. 2017, 123, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Farré, M.J.; Keller, J.; Gernjak, W. Reducing natural organic matter and disinfection by-product precursors by alternating oxic and anoxic conditions during engineered short residence time riverbank filtration: A laboratory-scale column study. Sci. Total Environ. 2016, 565, 616–625. [Google Scholar] [CrossRef] [PubMed]
- Partinoudi, V.; Collins, M.R. Assessing RBF reduction/removal mechanisms for microbial and organic DBP precursors. J. AWWA 2007, 99, 61–71. [Google Scholar] [CrossRef]
- Weiss, W.J.; Bouwer, E.J.; Ball, W.P.; O’Melia, C.R.; Lechevallier, M.W.; Arora, H.; Speth, T.F. Riverbank filtration—Fate of DBP precursors and selected microorganisms. J. AWWA 2003, 95, 68–81. [Google Scholar] [CrossRef]
- Weiss, W.J.; Bouwer, E.J.; Ball, W.P.; O’Melia, C.R.; LeChevallier, M.W.; Arora, H.; Aboytes, R.; Speth, T.F. Study of water quality improvements during riverbank filtration at three Midwestern United States drinking water utilities. Geophys. Res. Abstr. 2003, 5, 04297. [Google Scholar]
- United States Census Bureau, Census 2010. Available online: https://factfinder.census.gov/faces/tableservices/jsf/pages/productview.xhtml?src=bkmk (accessed on 9 September 2018).
- High Plains Regional Climate Center CLIMOD. Available online: http://climod.unl.edu/ (accessed on 18 September 2018).
- U.S. Environmental Protection Agency (U.S. EPA). Drinking water guidance on disinfection by-products. In Disinfection By-Products in Drinking Water; Advice Note. No. 4 Version 2; U.S. Environmental Protection Agency: Washington, DC, USA, 2012; p. 27. [Google Scholar]
- WHO. Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2011; p. 518. [Google Scholar]
- Baghoth, S.A.; Sharma, S.K.; Amy, G.L. Tracking natural organic matter (NOM) in a drinking water treatment plant using fluorescence excitation–emission matrices and PARAFAC. Water Res. 2011, 45, 797–809. [Google Scholar] [CrossRef]
- Chen, W.; Westerhoff, P.; Leenheer, J.A.; Booksh, K. Fluorescence excitation–emission matrix regional integration to quantify spectra for dissolved organic matter. Environ. Sci. Technol. 2003, 37, 5701–5710. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (U.S. EPA). Method 1623: Cryptosporidium and Giardia in Water by Filtration/IMS/FA; EPA 815-R-05-002; Office of Water: Washington, DC, USA, 2005; p. 68.
- ISO. Water Quality—Enumeration of Escherichia coli and Coliform Bacteria—Part 2: Most Probable Number Method; ISO 9308-2: 2012; International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- Cady, P.; Boving, T.B.; Choudri, B.S.; Cording, A.; Patil, K.; Reddy, V. Attenuation of bacteria at a riverank filtration site in rural India. Water Environ. Res. 2013, 85, 2164–2174. [Google Scholar] [CrossRef]
- Balzer, M.; Witt, N.; Flemming, H.C.; Wingender, J. Faecal indicator bacteria in river biofilms. Water Sci. Technol. 2010, 61, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- USGS Water Data for the Nation. Available online: https://waterdata.usgs.gov/nwis (accessed on 18 September 2018).
- Dash, R.R.; Prakash, E.B.; Kumar, P.; Mehrotra, I.; Sandhu, C.; Grischek, T. River bank filtration in Haridwar, India: Removal of turbidity, organics and bacteria. Hydrogeol. J. 2010, 18, 973–983. [Google Scholar] [CrossRef]
- Maeng, S.K.; Sharma, S.K.; Lekkerkerker-Teunissen, K.; Amy, G.L. Occurrence and fate of bulk organic matter and pharmaceutically active compounds in managed aquifer recharge: A review. Water Res. 2011, 45, 3015–3033. [Google Scholar] [CrossRef] [PubMed]
- Quanrud, D.M.; Hafer, J.; Karpiscak, M.M.; Zhang, J.; Lansey, K.E.; Arnold, R.G. Fate of organics during soil-aquifer treatment: Sustainability of removals in the field. Water Res. 2003, 37, 3401–3411. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency (U.S. EPA). Occurrence Assessment for the Final Stage 2 Disinfectants and Disinfection Byproducts Rule; EPA 815-R-05-011; U.S. EPA: Washington, DC, USA, 2005.
- Nebraska Drinking Water Watch. Available online: https://sdwis-dhhs.ne.gov:8443/DWW/ (accessed on 9 September 2018).
- Chen, W.J.; Weisel, C.P. Halogenated DBP concentrations in a distribution system. J. AWWA 1998, 90, 151–163. [Google Scholar] [CrossRef]
- Skark, C.; Remmler, F.; Zullei-Seibert, N. Classification of Riverbank Filtration Sites and Removal Capacity. In Recent Progress in Slow Sand and Alternative Biofiltration Processes; Gimbel, R., Graham, N.J.D., Collins, M.R., Eds.; IWA Publishing: London, UK, 2006. [Google Scholar]
- Wang, J. Riverbank Filtration Case Study at Louisville, Kentucky. In Riverbank Filtration; Ray, C., Melin, G., Linsky, R.B., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 117–145. [Google Scholar]
- Edzwald, J.K.; Tobiason, J.E. Enhanced coagulations: US requirements and a broader view. Water Sci. Technol. 1999, 40, 63–70. [Google Scholar] [CrossRef]
- Hua, G.; Reckhow, D.A.; Abusallout, I. Correlation between SUVA and DBP formation during chlorination and chloramination of NOM fractions from different sources. Chemosphere 2015, 130, 82–89. [Google Scholar] [CrossRef]
- Kitis, M.; Yigita, N.O.; Harmana, B.I.; Muhammetoglu, H.; Muhammetoglu, A.; Karadirek, I.E.; Demirel, I.; Ozdenc, T.; Palancic, I. Occurrence of trihalomethanes in chlorinated groundwaters with very low natural organic matter and bromide concentrations. Environ. Forensics 2010, 11, 264–274. [Google Scholar] [CrossRef]
- Hua, G.; Reckhow, D.A.; Kim, J. Effect of bromide and iodide ions on the formation and speciation of disinfection byproducts during chlorination. Environ. Sci. Technol. Lett. 2006, 40, 3050–3056. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
D'Alessio, M.; Dvorak, B.; Ray, C. Riverbank Filtration Impacts on Post Disinfection Water Quality in Small Systems—A Case Study from Auburn and Nebraska City, Nebraska. Water 2018, 10, 1865. https://doi.org/10.3390/w10121865
D'Alessio M, Dvorak B, Ray C. Riverbank Filtration Impacts on Post Disinfection Water Quality in Small Systems—A Case Study from Auburn and Nebraska City, Nebraska. Water. 2018; 10(12):1865. https://doi.org/10.3390/w10121865
Chicago/Turabian StyleD'Alessio, Matteo, Bruce Dvorak, and Chittaranjan Ray. 2018. "Riverbank Filtration Impacts on Post Disinfection Water Quality in Small Systems—A Case Study from Auburn and Nebraska City, Nebraska" Water 10, no. 12: 1865. https://doi.org/10.3390/w10121865
APA StyleD'Alessio, M., Dvorak, B., & Ray, C. (2018). Riverbank Filtration Impacts on Post Disinfection Water Quality in Small Systems—A Case Study from Auburn and Nebraska City, Nebraska. Water, 10(12), 1865. https://doi.org/10.3390/w10121865





