Behavior of Organic Micropollutants During River Bank Filtration in Budapest, Hungary
Abstract
1. Introduction
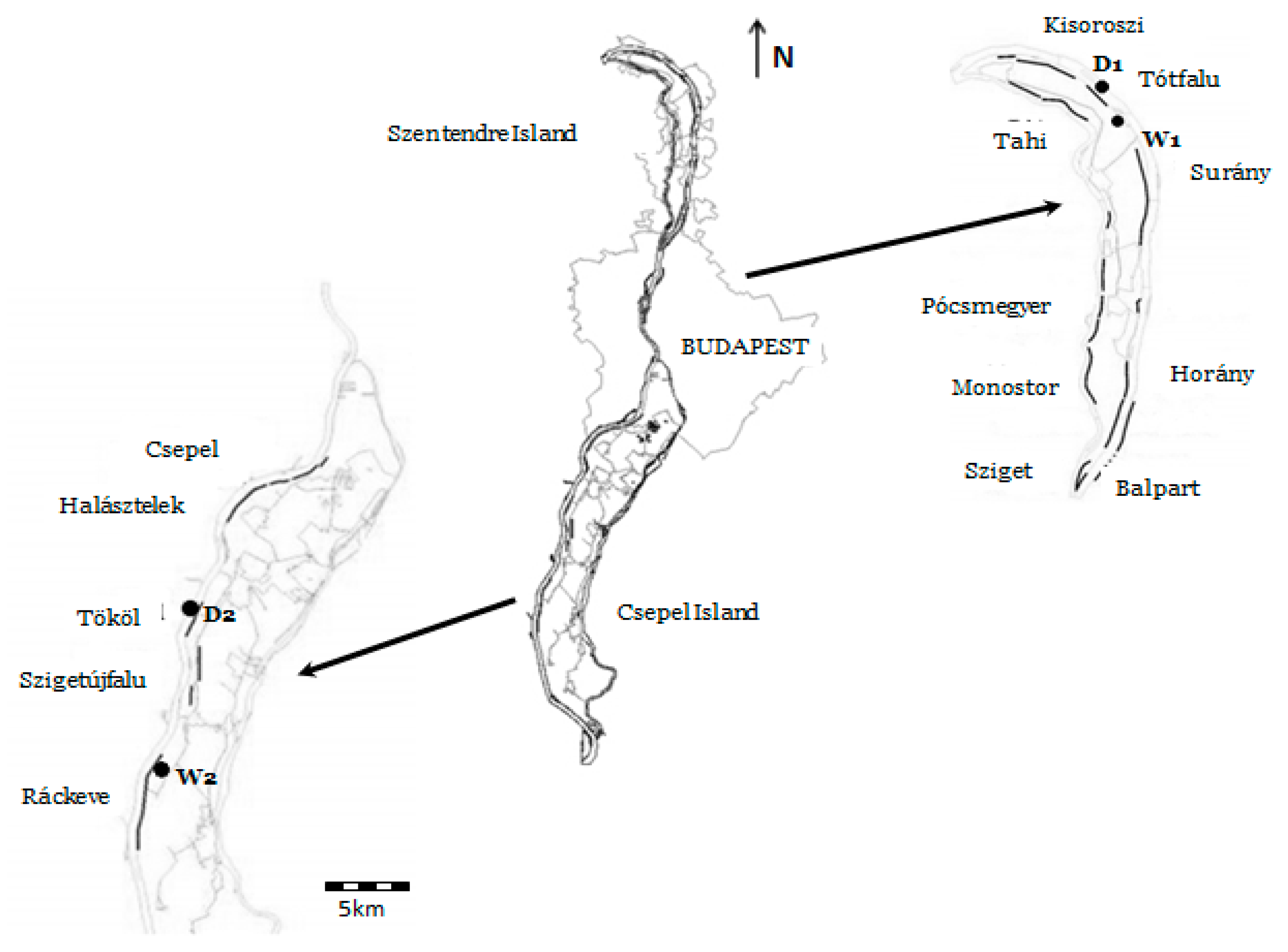
2. Materials and Methods
3. Results
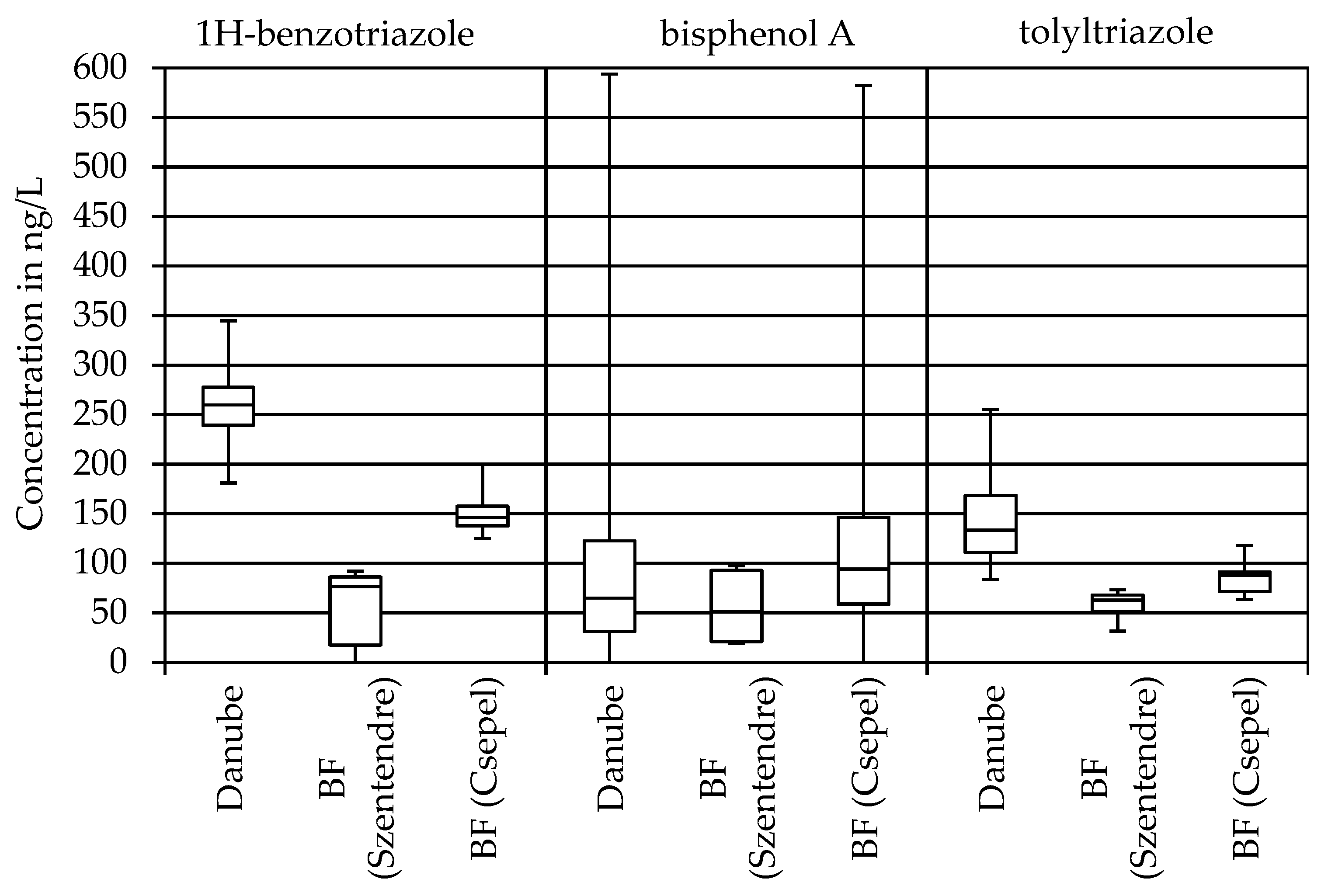
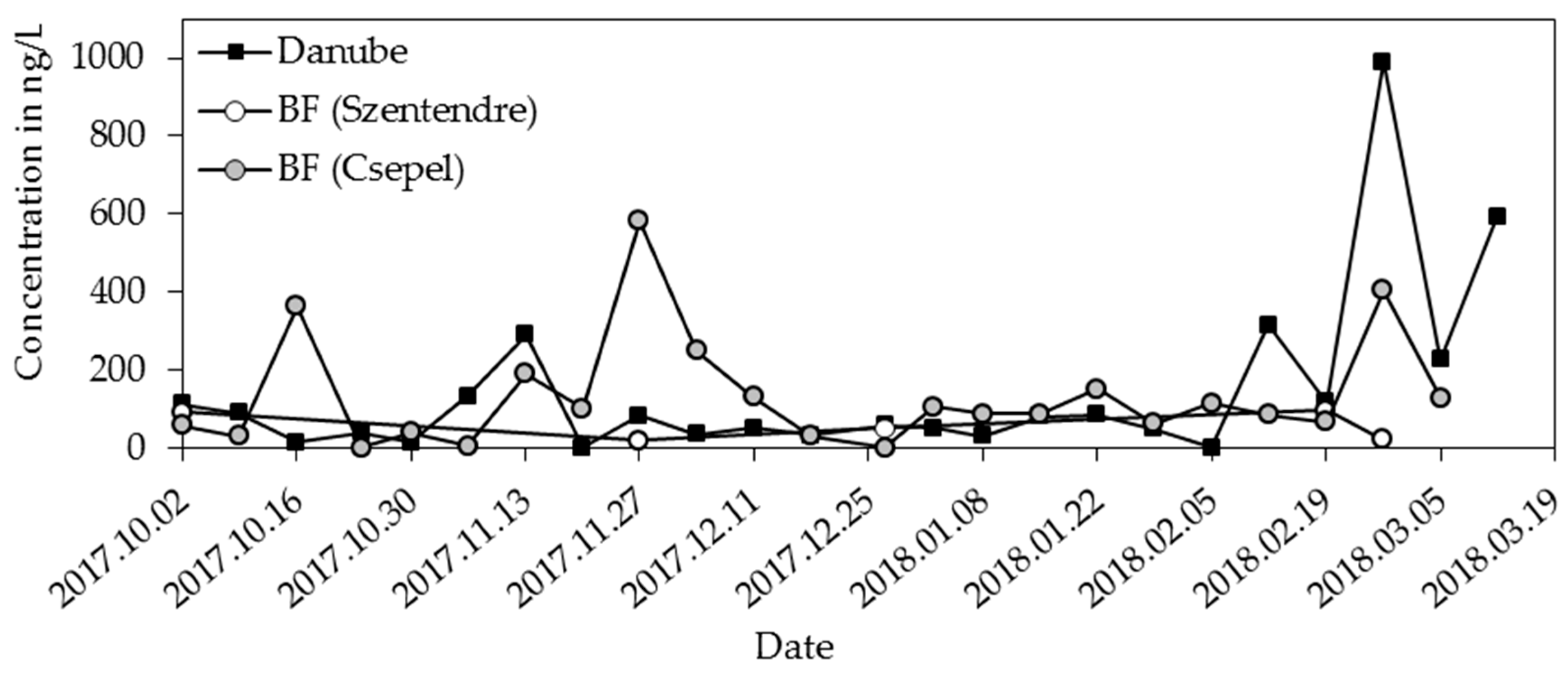
3.1. Industrial Products
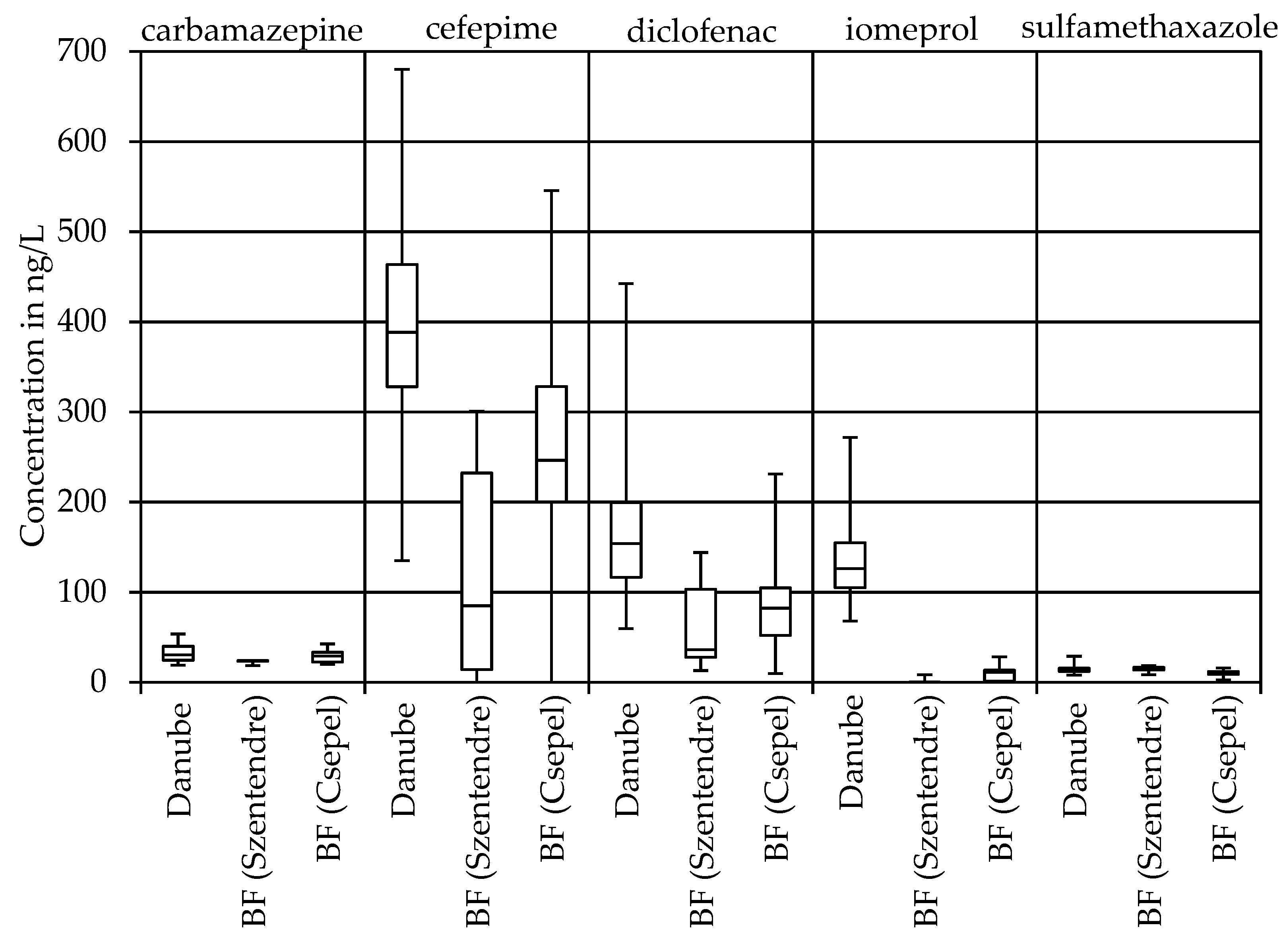
3.2. Pharmaceuticals and X-ray Contrast Agents
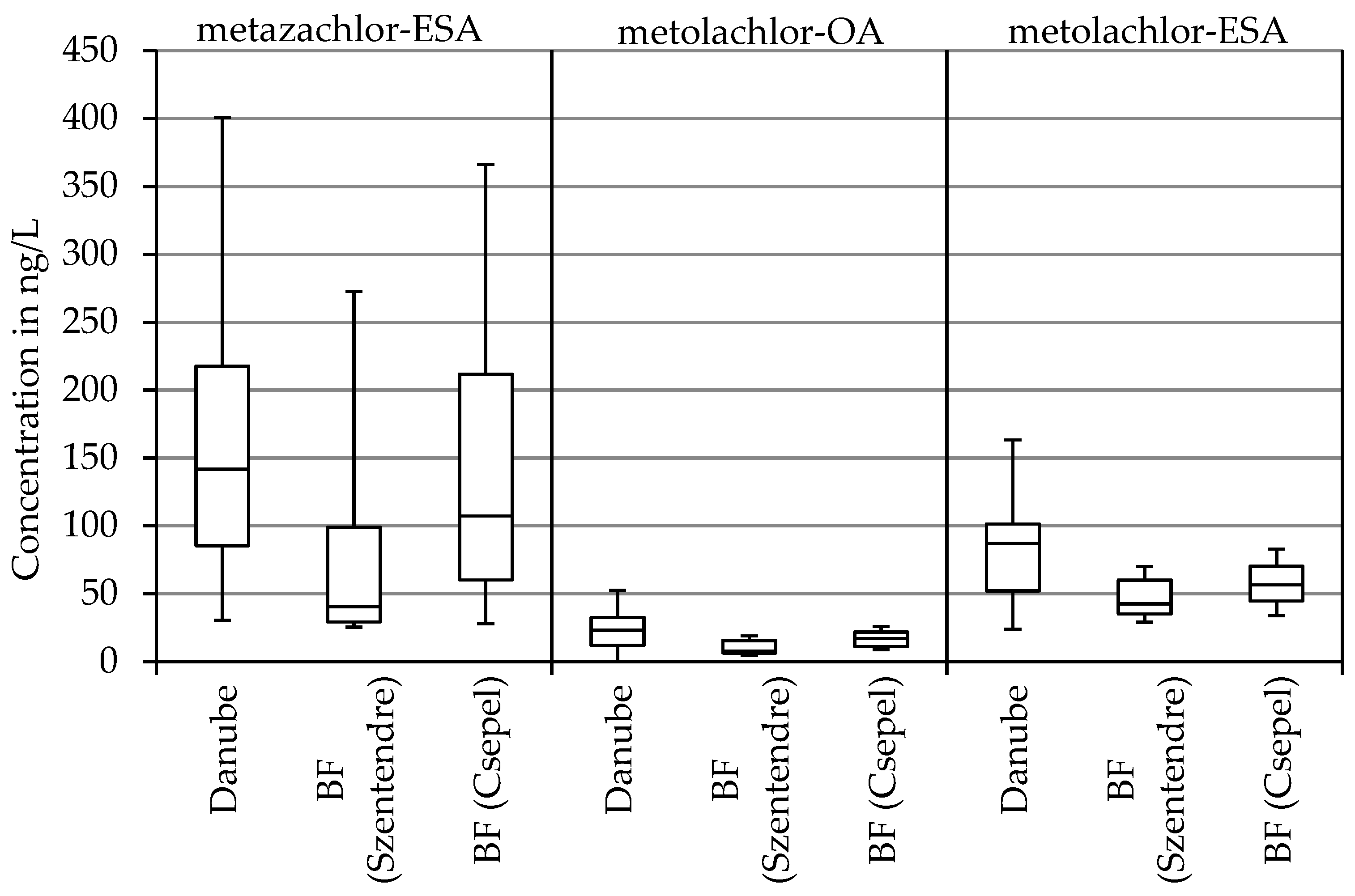
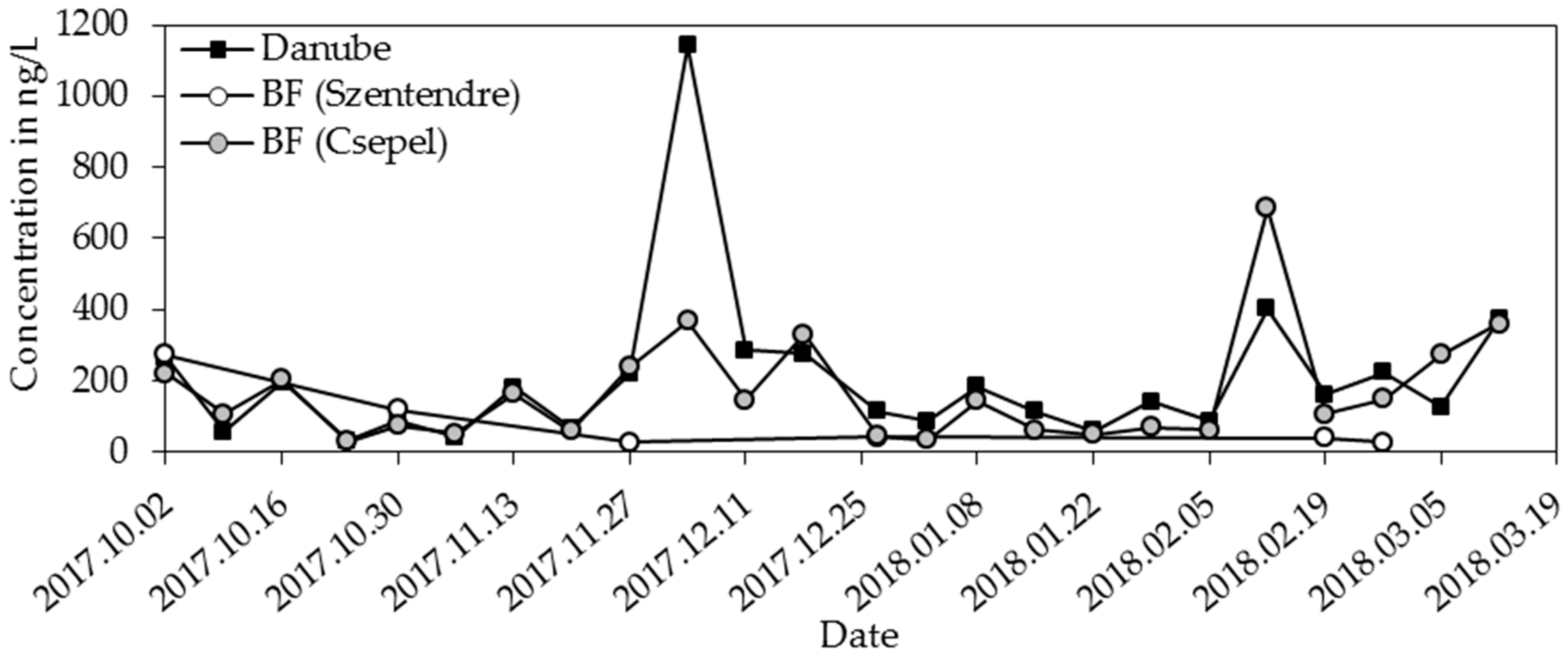
3.3. Herbicides, Pesticides, and Transformation Products
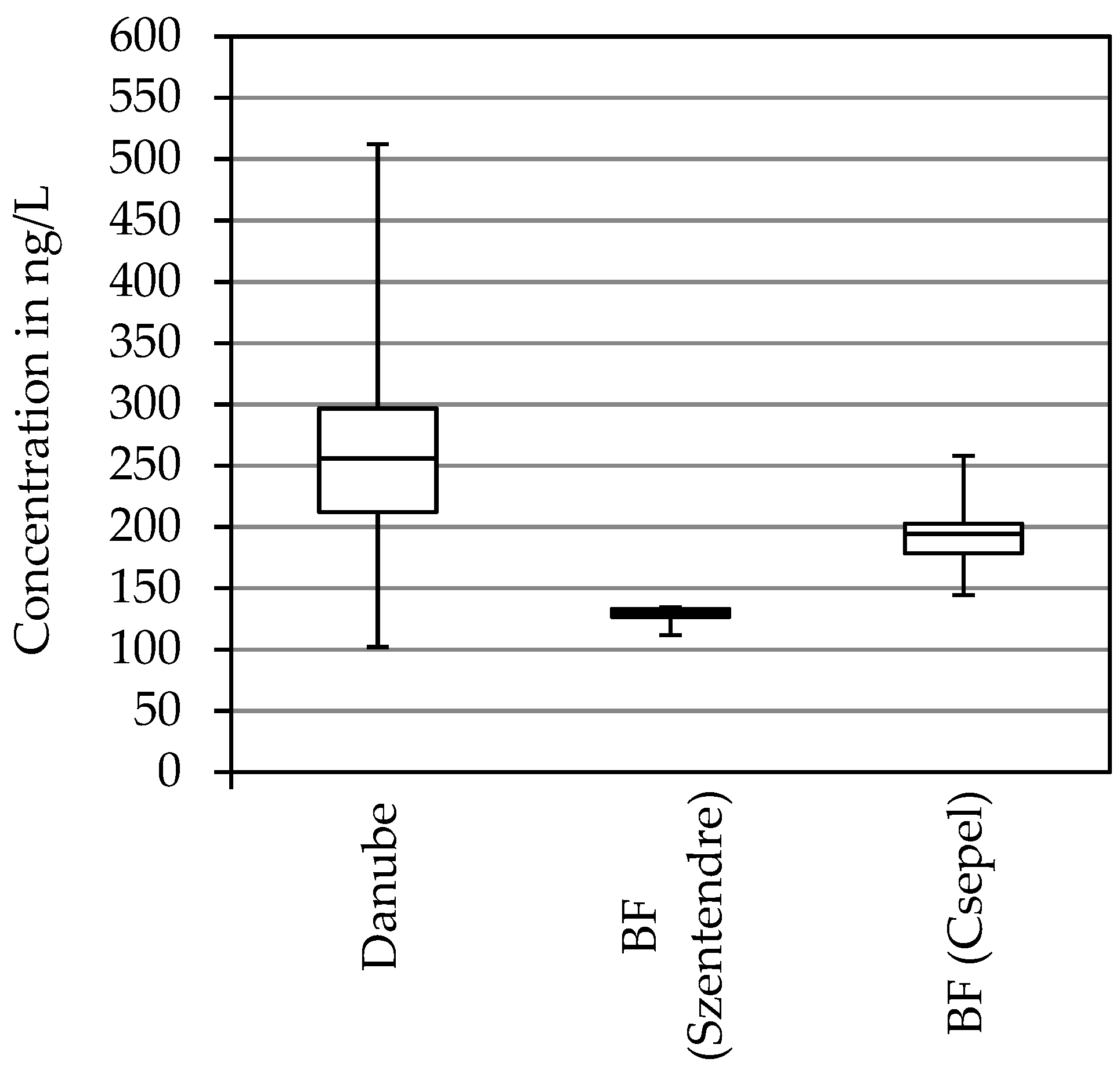
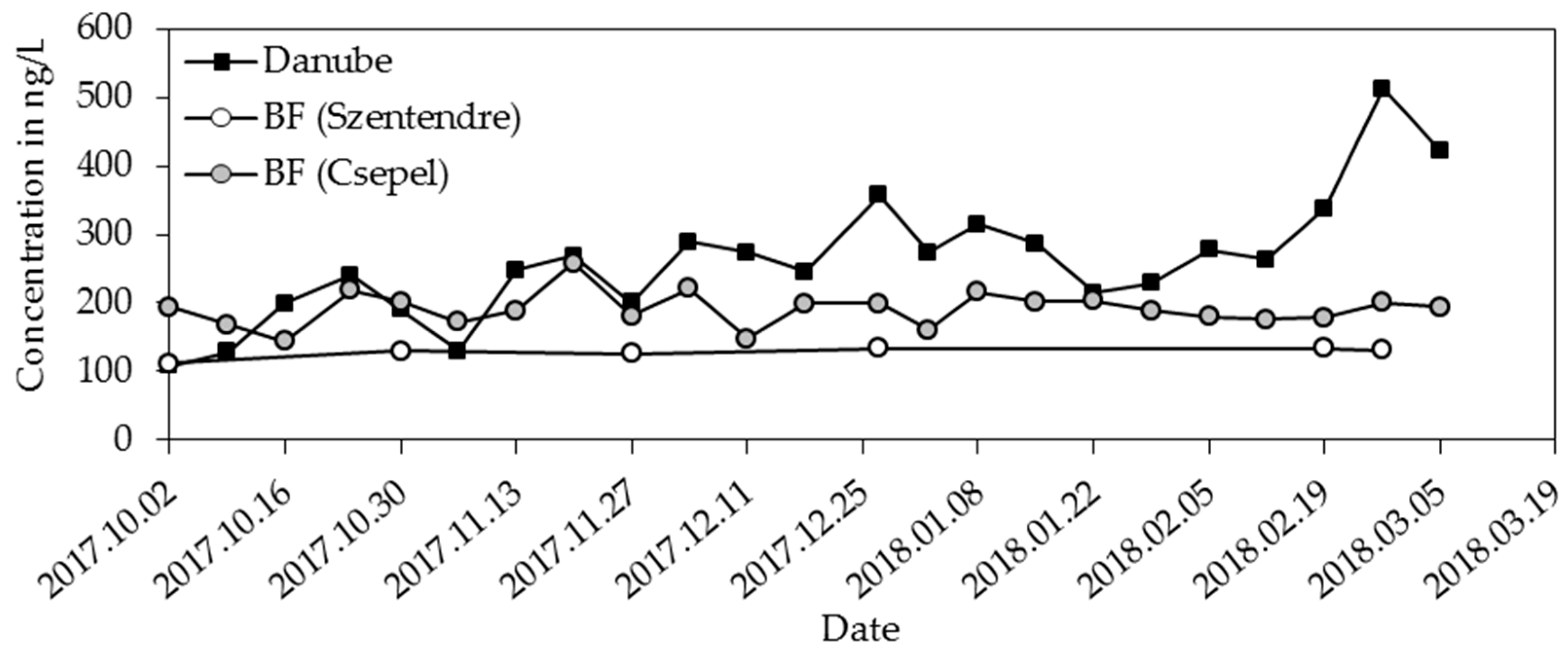
3.4. Food Additives
4. Discussion
5. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Loos, R.; Gawlik, B.M.; Locoro, G.; Rimaviciute, E.; Contini, S.; Bidoglio, G. EU-wide survey of polar organic persistent pollutants in European river waters. Environ. Pollut. 2009, 157, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Liška, I.; Wagner, F.; Sengl, M.; Deutsch, K.; Slobodník, J. Joint Danube Survey 3|ICPDR – International Commission for the Protection of the Danube River. Available online: https://www.icpdr.org/main/activities-projects/jds3 (accessed on 12 December 2018).
- Kim, M.-K.; Zoh, K.D. Occurrence and removals of micropollutants in water environment. Eng. Res. 2016, 21, 319–332. [Google Scholar] [CrossRef]
- Gutiérrez, J.P.; van Halem, D.; Rietveld, L. River bank filtration for the treatment of highly turbid Colombian rivers. Drinking Water Eng. Sci. 2017, 10, 13–26. [Google Scholar] [CrossRef]
- Hiscock, K.M.; Grischek, T. Attenuation of groundwater pollution by bank filtration. J. Hydrol. 2001, 266, 139–144. [Google Scholar] [CrossRef]
- Schmidt, C.K.; Lange, F.T.; Brauch, H.-J. Assessing the impact of different redox conditions and residence times on the fate of organic micropollutants during river bank filtration. In Proceedings of the 4th International Conference on Pharmaceuticals and Endocrine Disrupting Chemicals in Water; National Ground Water Association: Minneapolis, MN, USA, 2004; pp. 195–205. [Google Scholar]
- Regnery, J.; Wing, A.D.; Kautz, J.; Drewes, J.E. Introducing sequential managed aquifer recharge technology (SMART)-From laboratory to full-scale application. Chemosphere 2016, 154, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Hellauer, K.; Mergel, D.; Ruhl, A.S.; Filter, J.; Hübner, U.; Jekel, M.; Drewes, J.E. Advancing Sequential Managed Aquifer Recharge Technology (SMART) using different intermediate oxidation processes. Water 2017, 9, 221. [Google Scholar] [CrossRef]
- Storck, F.R.; Sacher, F.; Brauch, H.-J. Hazardous and emerging substances in drinking water resources in the Danube River Basin. In Danube River Basin; Liska, I., Ed.; Springer: Berlin, Germany, 2015; pp. 251–270. [Google Scholar]
- Greskowiak, J.; Hamann, E.; Burke, V.; Massmann, G. The uncertainty of biodegradation rate constants of emerging organic compounds in soil and groundwater-A compilation of literature values for 82 substances. Water Res. 2017, 126, 122–133. [Google Scholar] [CrossRef] [PubMed]
- AquaNES. Available online: http://www.aquanes-h2020.eu (accessed on 27 September 2018).
- Molnár, Z. Determination of the Production Well Capacities by Modelling (in Hungarian); Budapest Waterworks: Budapest, Hungary, 2013. (in Hungarian) [Google Scholar]
- AWE. AWE: Arbeitsgemeinschaft der Wasserversorger im Einzugsgebiet der Elbe, Gütebericht 2016–2017; Water Quality Report of Waterworks in the Elbe River Catchment: Torgau, Germany, 2018. [Google Scholar]
- Calvo-Flores, F.G.; Isac-Garcéa, J.; Dobado, J.A. Emerging Pollutants: Origin, Structure and Properties; Wiley-VCH: Weinheim, Germany, 2018. [Google Scholar]
- Milanović, M.; Sudji, J.; Grujić Letić, N.; Radonić, J.; Turk Sekulić, M.; Vojinović Miloradov, M.; Milić, N. Seasonal variations of bisphenol A in the Danube by the Novi Sad municipality. J. Serb. Chem. Soc. 2016, 80, 333–345. [Google Scholar] [CrossRef]
- Arnold, S.M.; Clark, K.E.; Staples, C.A.; Klecka, G.M.; Dimond, S.S.; Caspers, N.; Hentges, S.G. Relevance of drinking water as a source of human exposure to bisphenol A. J. Exposure Sci. Environ. Epidemiol. 2013, 23, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Technical University of Denmark. Benzotriazole and Tolyltriazole-Evaluation of Health Hazards and Proposal of Health Based Quality Criteria for Soil and Drinking Water, Toxicology and Risk Assessment; Technical University of Denmark: Lyngby, Denmark, 2013. [Google Scholar]
- Ray, C. Riverbank Filtration: Understanding Contaminant Biogeochemistry and Pathogen Removal; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002. [Google Scholar]
- Radović, T.; Grujić, S.; Dujaković, N.; Radišić, M.; Vasiljević, T.; Petković, A.; Boreli-Zdravkovic, Đ.; Dimkić, M.; Laušević, M. Pharmaceutical residues in the Danube River basin in Serbia–a two-year survey. Water Sci. Technol. 2012, 66, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Sacher, F.; Metziger, M.; Wenz, M.; Gabriel, S.; Brauch, H.-J. Arzneimittelrückstände in Grund- und Oberflächenwässern. In Spurenstoffe in Gewässern; Track, T., Kreysa, G., Eds.; Wiley-VCH: Weinheim, Germany, 2006; pp. 97–106. ISBN 3-527-31017-7. [Google Scholar]
- Hollender, J.; Huntscha, S. River Bank Filtration of Micropollutants, 2014. Available online: https://www.dora.lib4ri.ch/eawag/islandora/object/eawag%3A11880/datastream/PDF/view (accessed on 12 December 2018).
- Schittko, S.; Putschew, A.; Jekel, M. Bank filtration: A suitable process for the removal of iodinated X-ray contrast media? Water Sci. Technol. 2004, 50, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Soran, M.-L.; Lung, I.; Opriş, O.; Floare-Avram, V.; Coman, C. Determination of antibiotics in surface water by solid-phase extraction and high-performance liquid chromatography with diode array and mass spectrometry detection. Anal. Lett. 2017, 50, 1209–1218. [Google Scholar] [CrossRef]
- Hvězdová, M.; Kosubová, P.; Košíková, M.; Scherr, K.E.; Šimek, Z.; Brodský, L.; Šudoma, M.; Škulcová, L.; Sáňka, M.; Svobodová, M.; et al. Currently and recently used pesticides in Central European arable soils. Sci. Total Environ. 2018, 613, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K.A.; Tzilivakis, J.; Warner, D.J.; Green, A. An international database for pesticide risk assessments and management. Human Ecol. Risk Assess. Int. J. 2016, 22, 1050–1064. [Google Scholar] [CrossRef]
- Mai, C.; Theobald, N.; Lammel, G.; Hühnerfuss, H. Spatial, seasonal and vertical distributions of currently-used pesticides in the marine boundary layer of the North Sea. Atmos. Environ. 2013, 75, 92–102. [Google Scholar] [CrossRef]
- Lange, F.T.; Scheurer, M.; Brauch, H.-J. Artificial sweeteners-a recently recognized class of emerging environmental contaminants: A review. Anal. Bioanal.Chem. 2012, 403, 2503–2518. [Google Scholar] [CrossRef] [PubMed]
| Analyte | Range of Quantification in LOQ−10,000 ng/L | Quantifier MRM Transition Q1→Q3 (m/z) | Qualifier MRM Transition Q1→Q3 (m/z) |
|---|---|---|---|
| Industrial Chemicals | |||
| 1H-benzotriazole | 50−10000 | 120→65 | 120→92 |
| Bisphenol A | 5−10000 | 233→138 | 233→215 |
| Tolyltriazole | 10−10000 | 134→77 | 134→79 |
| Herbicides, Pesticides and Transformation Products | |||
| Dimethachlor-ESA | 1−10000 | 300→120 | 300→80 |
| Dimethachlor-OA | 10−10000 | 250→178 | 250→130 |
| Dimethoate | 10−10000 | 230→199 | 230→125 |
| Diuron | 10−10000 | 230→199 | 230→125 |
| Imidacloprid | 5−10000 | 256→209 | 256→175 |
| Irgarol | 1−10000 | 254→198 | 254→108 |
| Isoproturon | 1−10000 | 208→72 | 208→175 |
| Metazachlor-ESA | 5−10000 | 322→121 | 322→148 |
| Metazachlor-OA | 1−10000 | 271→67 | 271→65 |
| Metolachlor-ESA | 5−10000 | 328→120 | 328→80 |
| Metolachlor-OA | 5−10000 | 278→206 | 278→174 |
| Nicosulfuron | 5−10000 | 410→182 | 410→213 |
| Terbuthylazine-2-hydroxy | 1−10000 | 210→97 | 210→154 |
| Terbutryn | 5−10000 | 142→186 | 142→91 |
| Food Additives | |||
| Acesulfame | 1−10000 | 162→82 | 162→78 |
| Pharmaceuticals and X-ray Contrast Agents | |||
| Bezafibrate | 10−10000 | 362→316 | 362→139 |
| Carbamazepine | 1−10000 | 237→194 | 237→179 |
| Cefepime | 50−10000 | 481→396 | 481→324 |
| Cefotaxime | 50−10000 | 456→396 | 456→167 |
| Cefuroxime | 50−10000 | 447→386 | 447→342 |
| Clarithromycin | 10−10000 | 748→590 | 748→158 |
| Clindamycin | 5−10000 | 425→126 | 427→126 |
| Diclofenac | 50−10000 | 294→250 | 294→252 |
| Erythromycin | 5−10000 | 734→576 | 734→158 |
| Fluoxetin | 10−10000 | 310→148 | 310→44 |
| Gabapentin | 50−10000 | 172→154 | 172→137 |
| Ibuprofen | 5−10000 | 205→161 | 205→159 |
| Iomeprol | 50−10000 | 778→687 | 778→405 |
| Metoprolol | 5−10000 | 268→116 | 268→133 |
| Naproxen | 10−10000 | 229→185 | 229→169 |
| Paracetamol | 5−10000 | 152→110 | 152→93 |
| Roxithromycin | 50−10000 | 837→679 | 837→158 |
| Sulfamethoxazole | 1−10000 | 254→156 | 154→108 |
| Compound | Danube River Water (Szentendre) n = 6 | Danube River Water (Csepel) n = 24 | ||||
|---|---|---|---|---|---|---|
| Minimum | Median | Maximum | Minimum | Median | Maximum | |
| 1H-Benzotriazole | 181 | 272 | 345 | 183 | 256 | 338 |
| Bisphenol A | 15 | 33 | 124 | 14 | 86 | 990 |
| Tolyltriazole | 84 | 121 | 172 | 86 | 142 | 255 |
| Carbamazepine | 19 | 30 | 40 | 19 | 31 | 54 |
| Cefepime | 194 | 358 | 532 | 135 | 394 | 680 |
| Diclofenac | 70 | 153 | 442 | 59 | 154 | 418 |
| Iomeprol | 106 | 131 | 161 | 68 | 122 | 272 |
| Sulfamethoxazole | 6 | 14 | 17 | 7 | 13 | 45 |
| Metolachlor-ESA | 33 | 113 | 162 | 24 | 85 | 163 |
| Metolachlor-OA | 6 | 31 | 49 | 7 | 23 | 53 |
| Metazachlor-ESA | 52 | 180 | 359 | 31 | 152 | 1142 |
| Acesulfame | 102 | 219 | 343 | 115 | 266 | 512 |
| Compound | Bank Filtrate (Szentendre) n = 6 | Bank Filtrate (Csepel) n = 24 | ||||
|---|---|---|---|---|---|---|
| Minimum | Median | Maximum | Minimum | Median | Maximum | |
| 1H-Benzotriazole | 70 | 85 | 92 | 125 | 146 | 200 |
| Bisphenol A | 19 | 51 | 98 | 30 | 105 | 2381 |
| Tolyltriazole | 32 | 63 | 73 | 64 | 88 | 118 |
| Carbamazepine | 18 | 24 | 24 | 20 | 29 | 43 |
| Cefepime | 57 | 193 | 301 | 123 | 248 | 546 |
| Diclofenac | 36 | 103 | 144 | 13 | 87 | 231 |
| Iomeprol | <LOQ | <LOQ | <LOQ | <LOQ | <LOQ | <LOQ |
| Sulfamethoxazole | 9 | 13 | 18 | 6 | 9 | 16 |
| Metolachlor-ESA | 29 | 43 | 70 | 34 | 57 | 83 |
| Metolachlor-OA | 11 | 38 | 88 | 9 | 17 | 26 |
| Metazachlor-ESA | 25 | 40 | 273 | 28 | 125 | 686 |
| Acesulfame | 112 | 131 | 134 | 145 | 195 | 258 |
| Compound | Removal Rates in % (Szentendre) | Removal Rates in % (Csepel) |
|---|---|---|
| 1H-benzotriazole | 69 | 43 |
| bisphenol A | −54 | −22 |
| tolyltriazole | 48 | 38 |
| carbamazepine | 20 | 4 |
| cefepime | 46 | 37 |
| diclofenac | 32 | 44 |
| iomeprol | bank filtrate concentrations below LOQ | |
| sulfamethoxazole | 9 | 30 |
| metolachlor-ESA | 62 | 33 |
| metolachlor-OA | −20 | 25 |
| metazachlor-ESA | 78 | 18 |
| acesulfame | 40 | 27 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagy-Kovács, Z.; László, B.; Fleit, E.; Czichat-Mártonné, K.; Till, G.; Börnick, H.; Adomat, Y.; Grischek, T. Behavior of Organic Micropollutants During River Bank Filtration in Budapest, Hungary. Water 2018, 10, 1861. https://doi.org/10.3390/w10121861
Nagy-Kovács Z, László B, Fleit E, Czichat-Mártonné K, Till G, Börnick H, Adomat Y, Grischek T. Behavior of Organic Micropollutants During River Bank Filtration in Budapest, Hungary. Water. 2018; 10(12):1861. https://doi.org/10.3390/w10121861
Chicago/Turabian StyleNagy-Kovács, Zsuzsanna, Balázs László, Ernő Fleit, Katalin Czichat-Mártonné, Gábor Till, Hilmar Börnick, Yasmin Adomat, and Thomas Grischek. 2018. "Behavior of Organic Micropollutants During River Bank Filtration in Budapest, Hungary" Water 10, no. 12: 1861. https://doi.org/10.3390/w10121861
APA StyleNagy-Kovács, Z., László, B., Fleit, E., Czichat-Mártonné, K., Till, G., Börnick, H., Adomat, Y., & Grischek, T. (2018). Behavior of Organic Micropollutants During River Bank Filtration in Budapest, Hungary. Water, 10(12), 1861. https://doi.org/10.3390/w10121861






