1. Introduction
Environmental remediation methods often employ the use of synthetic composites due to their enhanced material properties [
1,
2,
3,
4,
5]. Sulfate is one of commonly found anions in sources of water supplies, due the presence of atmospheric sulfur dioxide and many high water-soluble minerals containing sulfate, such as sodium sulfate, potassium sulfate, and magnesium sulfate in the environment [
6]. Recently, the removal of sulfate from drinking water has been targeted due to deleterious effects of sulfate on the environment [
7,
8]. Sulfates affect not only the environment but also human health. Abnormal changes resulting from sulfate consumption from drinking water may include diarrhea and dehydration. These ill effects are more prominent during infant years and decline with age [
6]. Previously, a novel composite made of polyvinyl chloride (PVC) loaded with barium carbonate (BaCO
3) was reported for sulfate removal [
9]. The implementation of such a material in a small-scale column test (SST) yielded promising results. However, reports indicate that potential contamination and leaching from conventional plastic like PVC can pose a severe threat to human health and the environment [
10,
11]. Monomethyl-, dimethyl-, monobutyl-, and dibutyl levels have been found at values up to 291 ng Tin (Sn)/L, 49.1 ng Sn/L, 28.5 ng Sn/L, and 52.3 ng Sn/L, respectively, in drinking water distributed through PVC pipes [
12,
13]. Successful remediation must involve the reduction of hazardous materials without introducing any further complications. Although the previously synthesized PVC/BaCO
3 composite [
10] showed promising results, the necessity for a benign and environmentally friendly remediation method still exists [
14].
The hydrolytic degradation of several common polyesters has provided an attractive new platform for composite synthesis. In particular, polycaprolactone (PCL) has become a focus of various medical device applications, including drug and bacterial encapsulation [
15,
16], antimicrobial film development [
17], and scaffolding in tissue engineering [
18]. PCL exhibits not only significant biocompatibility but also undergoes slow degradation to produce harmless byproducts. Additionally, the glass transition temperature of −60 °C and a low melting temperature near 60 °C result in PCL’s substantial amorphous thermoplastic properties. The simple ring-opening polymerization synthesis makes this polymer matrix attractive for nanocomposite formation. PCL shows significant solubility at room temperature in a gamut of solvents, including chloroform, dichloromethane, carbon tetrachloride, benzene, toluene, cyclohexanone, and 2-nitropropane. Such strong properties of PCL make it a suitable alternative to PVC in the previously reported facile synthesis of polymer/BaCO
3 composites [
7].
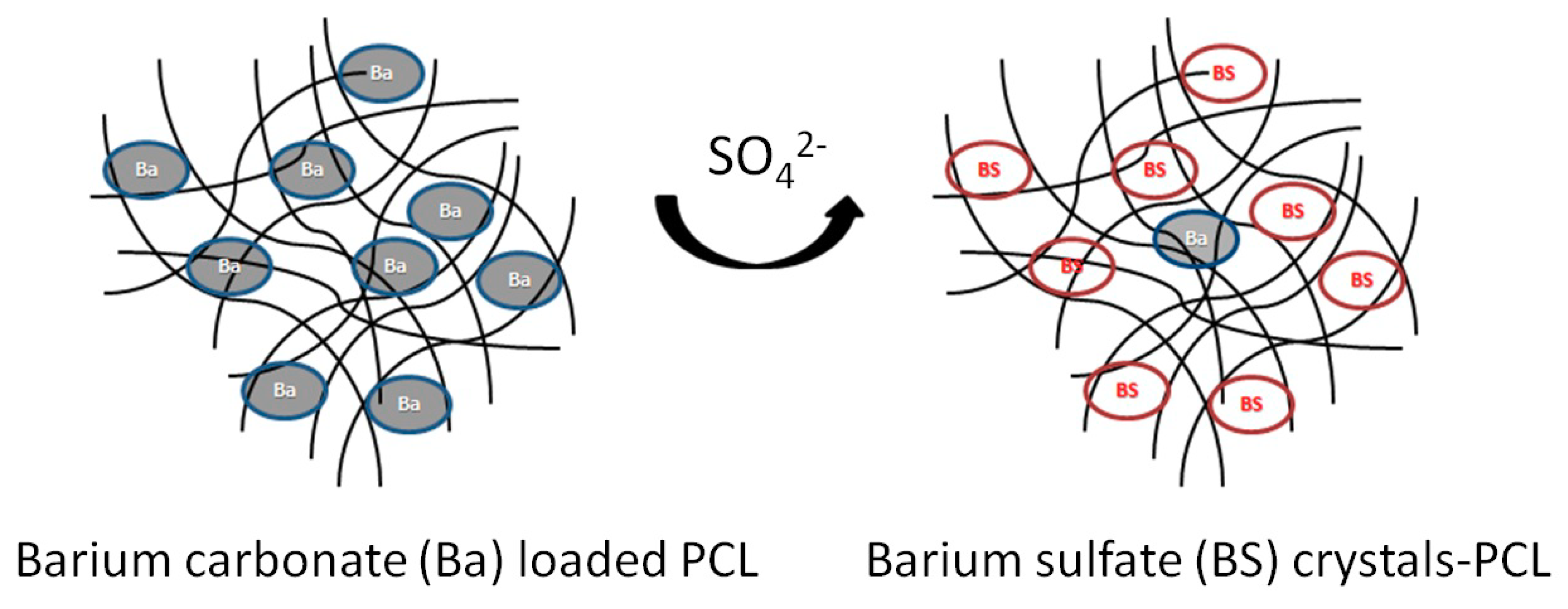
In the present work, incorporation of barium into a biodegradable and biocompatible polymer matrix for removal of sulfate from contaminated waters has been investigated. The porous nature of the PCL matrix promotes the use of such a material in column packing applications for environmental remediation. Several issues of previous schemes were addressed in the implementation of PCL. Primarily, hazardous and environmentally damaging PVC was replaced with PCL as a benign matrix. Furthermore, the easily accelerated hydrolytic degradation elucidates a new method for barium sulfate (BaSO
4) recovery following its use in the column studies. The resulting PCL and BaCO
3 composite (PCL/Ba) showed significant sulfate removal capacity similar to that observed in the previous studies [
7]. The process of precipitation is shown in
Scheme 1. Concerns of wall effects in column applications were addressed, and the results have been reported in this work.
3. Results and Discussion
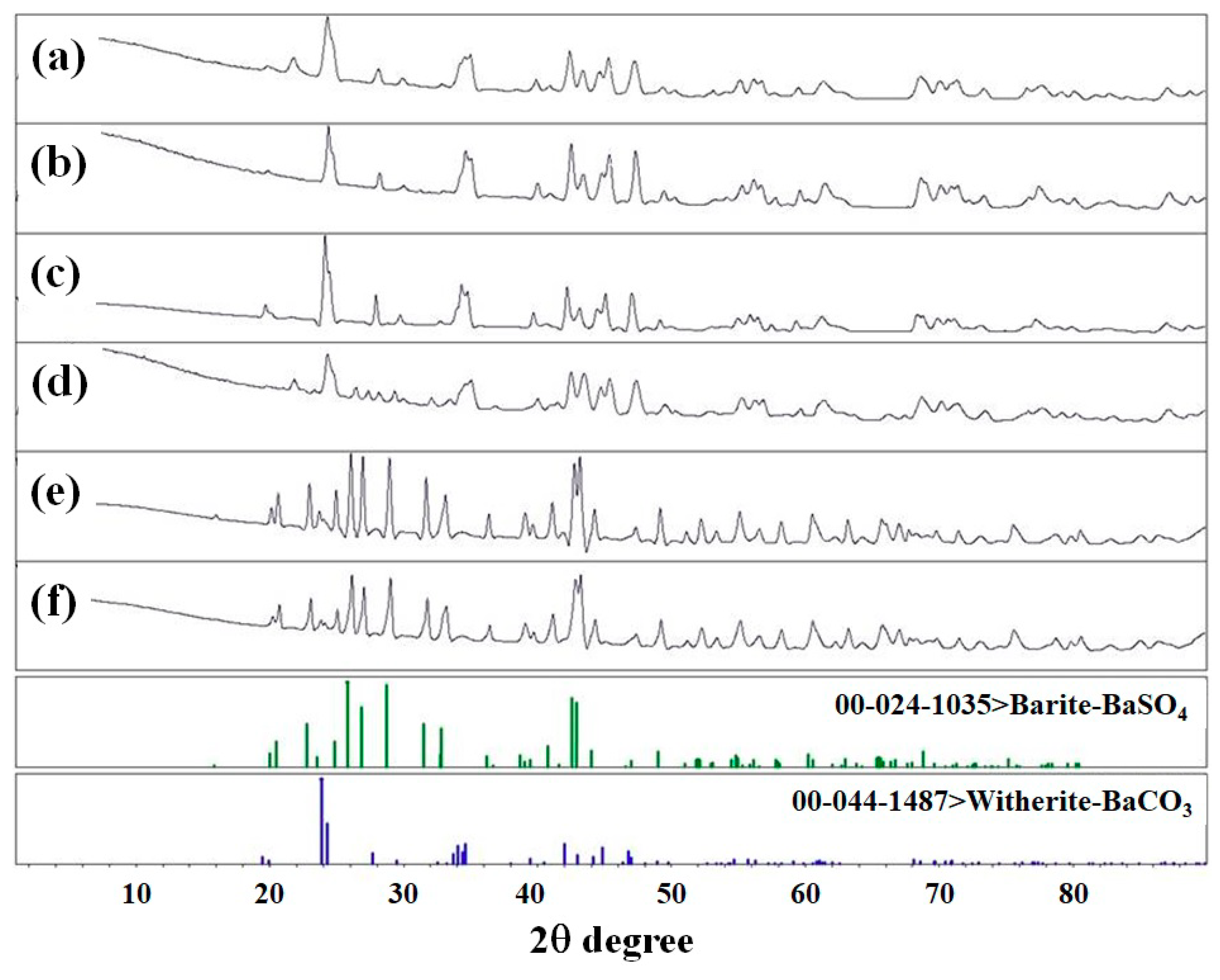
XRD analysis was performed to confirm the phase changes from BaCO
3 to BaSO
4 after sulfate removal. The obtained XRD patterns for PLC/Ba samples before and after sulfate adsorption are shown in
Figure 1. All the diffraction peaks were indexed with reference to the unit cell of the barite structure (JCPDS card: 24-1035) or witherite structure (JCPDS card: 00-044-1487). The diffraction peaks of (101), (111), (021), (121), (002), and (212) were characteristic peaks of orthorhombic BaSO
4 crystal, demonstrating that the product BaSO
4 formed. These results indicate that PCL/Ba samples effectively removed sulfate through the formation of BaSO
4. Additionally, these results are in good agreement with a previous report [
9], which showed the formation of BaSO
4 after sulfate adsorption using BaCO
3/PVC composites. However, in the case of the PCL/Ba (50/50) sample, the intensity of the peak corresponding to barium sulfate is weak due to the presence of increased polymer content that restricts the diffusion of sulfate molecules towards buried barium ions.
Figure 2a–h shows SEM images of control BaCO
3 and PCL/Ba samples with different magnifications.
Figure 2a,b represents control BaCO
3 with low and high magnification, respectively. The particle sizes ranged from a few hundred nanometers to microns in length. Elongated rod-shaped structures with hexagonal phases were observed.
Figure 2c–h show SEM images of PCL/Ba samples before barium sulfate precipitation. BaCO
3 particles were dispersed without surface morphology changes in the polymer matrix. BaCO
3 particles aggregated with an increase in the amount of polymer in the PCL/Ba composite (
Figure 2c), indicating less polymer content in the composites is better to effectively disperse BaCO
3 in the composites (
Figure 2e,g). The control BaCO
3 crystals dispersed in PCL had elongated structures with hexagonal faces as seen in
Figure 2d,f,h.
Figure 3a–f shows SEM images of PCL/Ba (50/50), (10/90), and (2.5/97.5) with lower and higher magnification after sulfate adsorption, respectively. Well-defined barium sulfate cubes were observed in all PCL/Ba composites. In a previous study [
9], the formation of BaSO
4 with a cubic structure was reported after sulfate adsorption using BaCO
3. In addition to the formation of BaSO
4 cubes, the bulk morphology of PCL in PCL/Ba (50/50) sample was observed due to the high content of PCL in the sample. Interestingly, random sized BaSO
4 cubes were observed in the samples containing less BaCO
3. Based on these results, it could be hypothesized that more controlled and well-defined BaSO
4 crystals can be precipitated when the barium content of the PCL/Ba composite is lower. The reasoning behind such a trend has been explained by Kucher et al., who studied the influence of supersaturation and free lattice ion ratio on barium sulfate particle formation. While heterogeneous nucleation dominates crystal formation at lower supersaturation levels, it is substituted by homogeneous nucleation at higher levels of supersaturation. Homogeneous nucleation promotes the formation of smaller crystals up to about 100 nm in size as opposed to larger well-defined crystals at lower levels. Similarly, in the present study, well-defined crystals were observed in composites with lower barium content as compared to random, smaller crystals formed with higher levels of barium-containing composites [
19].
Figure 4a–f shows TEM images of PCL/Ba composites before and after sulfate adsorption. Before sulfate adsorption, elongated BaCO
3 was observed in all samples, as seen in
Figure 2a–h. After sulfate removal, the transformation from a rod-shaped structure to the cubic structure was observed, which is in good agreement with the SEM images (
Figure 3b,d,f). These results indicate the formation of BaSO
4 cubes after sulfate adsorption. However, the formation of the BaSO
4 cube with a uniform size was not observed in the PCL/Ba (50/50) sample. Due to the high content of PCL in the sample, BaCO
3 particles were capped with PCL as seen in
Figure 4b. To confirm the formation of BaSO
4 after sulfate removal, HR-TEM analysis was performed in PCL/Ba (10/90) sample.
Figure 5a,b shows the HR-TEM images of the PCL/Ba (10/90) sample after sulfate removal.
As seen in
Figure 5a, the lattice spacing of 0.210 and 0.312 nm corresponding to (212) and (121) plane of Barite, respectively, was measured, which indicates the formation of BaSO
4 after sulfate adsorption. In addition, the measured lattice spacing in the sample after NOM removal as sulfate was 0.211 nm corresponding to (212) plane of Barite, indicating the PCL/Ba sample effectively removes sulfate in water. These results are in good agreement with the results of XRD analysis, confirming the formation of BaSO
4 after sulfate adsorption.
Table 2 summarizes theoretical and experimental sulfate adsorption capacity of PCL/Ba samples obtained from various SST, conducted using different size columns and various PCL/Ba composites. The theoretical capacity of the media was determined by taking into account that, according to the chemical reaction, 1 mole of barium reacts with 1 mole of sulfate to form barium sulfate. Therefore, the barium content in the media determined using EDS was used to calculate the theoretical sulfate removal capacity of the media in mg/g. The results shown in
Table 2 indicate that a lesser amount of polymer with a higher barium content in the composites helps improve the capacity of the media. The composite with lowest PCL (2.5%) and highest barium content (97.5%) was capable of achieving 72.4% of the theoretical capacity, as opposed to the PCL/Ba (50/50) which performed poorly.
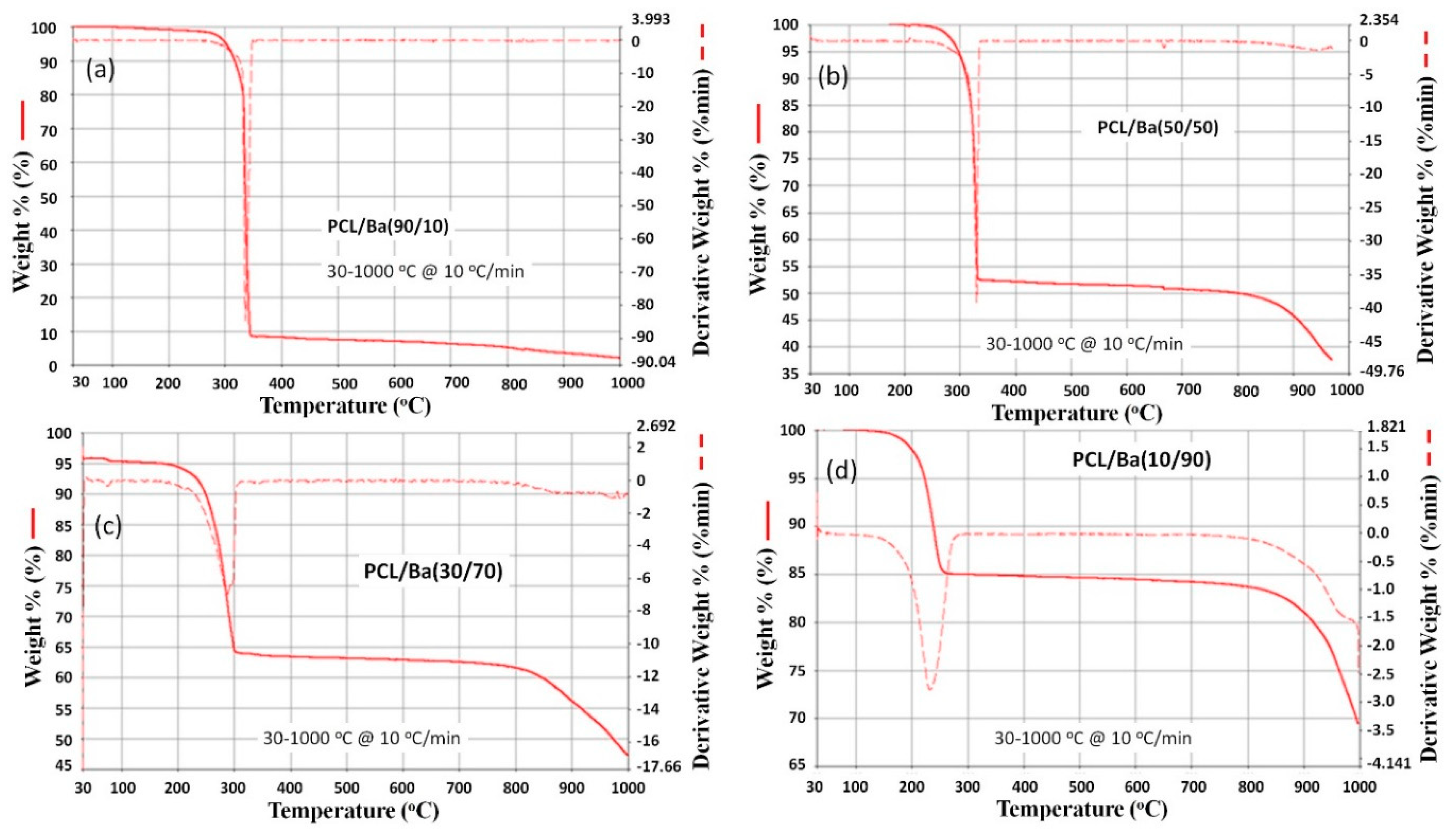
Thermogravimetric analysis was done on various BaCO
3 loaded polycaprolactone composites and these thermograms are shown in
Figure 6 and
Figure 7. The control polycaprolactone has a broad decomposition temperature around 380 to 415 °C. The addition of BaCO
3 decreased the decomposition temperatures (see
Figure 6). In addition, the decomposition temperatures are very sharp compared to pure polycaprolactone. The PCL/Ba (90/10) has a sharp decomposition temperature at around 335 °C, whereas PCL/Ba (50/50) has a decomposition temperature of 320 °C.
The increase in the quantity of BaCO3 loaded in the polymer composite in turn accelerated the early decomposition temperatures. For example, PCL/Ba (30/70) has a broad decomposition temperature from 250 to 300 °C and PCL/Ba (10/90) has a broad decomposition temperature of 200 to 250 °C, which is much lower compared to PCL/Ba (90/10).
The drastic decrease in decomposition temperatures resulting from increasing BaCO
3 content is unclear and requires detailed mechanistic studies. Similar observations were reported by Liu et al., wherein higher CaSO
4 loading in polycaprolactone composites decreased decomposition temperatures [
20]. Possible reasons for the abnormal decrease in temperature may have been due to poor recrystallization and weak bonding of polycaprolactone in the presence of BaCO
3. However, a different trend was observed by Lee et al. in the case of carbon nanotubes (CNT) dispersed polycaprolactone composites. Thermal stability increased even in the presence of a small amount of multi-walled CNT-Cl, which suggests the homogeneous dispersion in the composites CNT [
21]. Trapped solvent decomposition may have also played a role in causing the abnormal decrease in temperature.
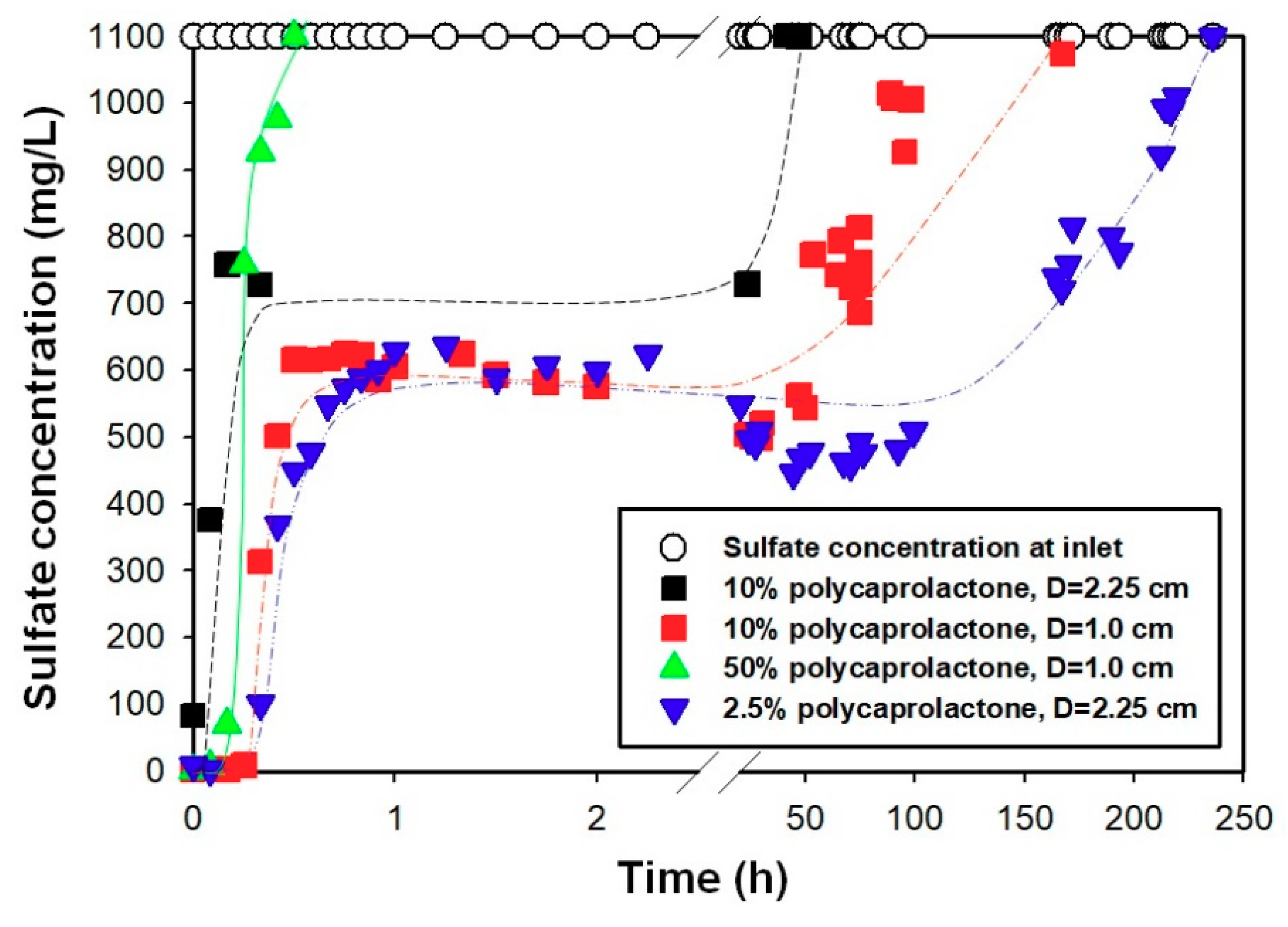
Figure 8 shows the sulfate removal of PCL/Ba composites with column tests. Symbols of white circles depict the influent sulfate concentration, which was kept constant at 1100 mg/L. There are two significant results shown in
Figure 8. Firstly, the sulfate removal depends on the amount of polymer and barium in the composites. The sample, containing a small amount of polymer (and consequently a large amount of barium), demonstrated higher sulfate removal compared to other samples. In the case of the PCL/Ba (50/50) sample, the column is saturated earlier compared to other samples due to a lower barium content, while a similar sulfate removal pattern is observed for PCL/Ba (2.5/97.5) and (10/90) composites. The existence of such a direct proportionality between the efficiency of sulfate removal and the cation content of the media has been studied by other researchers [
22]. Sulfate in the effluent was not detected for the first 10 minutes, while after this point it spiked to about 600 mg/L and maintained this level until about 4000 and 7000 min for PCL/Ba (2.5/97.5) and (10/90), respectively. Such a pattern in sulfate removal using PCL/Ba media can be explained by mass transfer principles. In the initial phase, barium available on the exterior of the packed column comes in contact with sulfate and quickly becomes saturated. After that, it takes a longer time for the sulfate molecules to access the barium molecules buried inside of the polymer matrix. Once all the barium molecules are saturated with adsorbed sulfate, the media reaches complete saturation and is not capable of any further sulfate removal.
The wall effect was studied in the process by conducting experiments in smaller and larger diameter columns loaded with the same amount of PCL/Ba media. As seen in
Figure 8, the friction was reduced significantly due to the wall surface when larger diameter columns were used. Although saturation in the larger diameter column (d = 2.25 cm) was achieved much quicker compared to the smaller diameter (d = 1.0 cm) column, the percentage of sulfate removal was higher in the smaller. Higher sulfate removal in the smaller column was the result of a longer retention time and hence better diffusion of sulfate molecules through the polymer matrix.
Figure 9 shows NOM removal as sulfate using PCL/Ba media. After passing sulfate containing NOM through the column for about 500 min, the PCL/Ba (10/90) media was able to remove more than 90% of the influent sulfate. A previous study on sulfate removal from wastes by precipitation by Beatti et al. [
23] reported that only 61.4% of the influent sulfate was removed using the barium precipitation technique under a sulfate concentration of 80 g/L in the influent. In the present study, the effluent sulfate concentrations remained well below 30–50 mg/L for the first two hours except for an outlier seen at 1 hour, removing more than 90% of influent sulfate because lower sulfate concentration (328 mg/L) was fed into the column. Such promising results show that the media used in this study has a very high capacity to remove low concentrations of sulfate, and further studies are required in pilot scale to evaluate a long-term activity of these media.
Table 3 shows the cumulative capacity of the PCL/Ba (10/90) composite used to remove sulfate from NOM containing Ohio River water. After passing sulfate containing water through the column for 450 min, the polymer composite was saturated, indicating that all the accessible barium molecules were bound with sulfate molecules to form barium sulfate. From
Table 3, it can be seen that PCL/Ba (10/90) exhibited a high capacity and removed more than 90% of the influent sulfate, and the cumulative capacity was calculated to be 175.8 mg/g.
Figure 10 shows an SEM image of the PCL/Ba (10/90) sample after sulfate removal in the Ohio River water containing NOM. The SEM image demonstrates irregular shaped barium sulfate crystals with different sizes ranging from 200 nm to 1 μm were formed after sulfate removal in the Ohio River water, which is in contrast to the well-defined cubes formed in the presence of synthetic sulfate containing solutions as shown in
Figure 3 and
Figure 4.