Environmentally-Related Cherry Root Cambial Plasticity
Abstract
1. Introduction
2. Experiments
2.1. Research Aim Background
2.2. Study Site and Plant Material
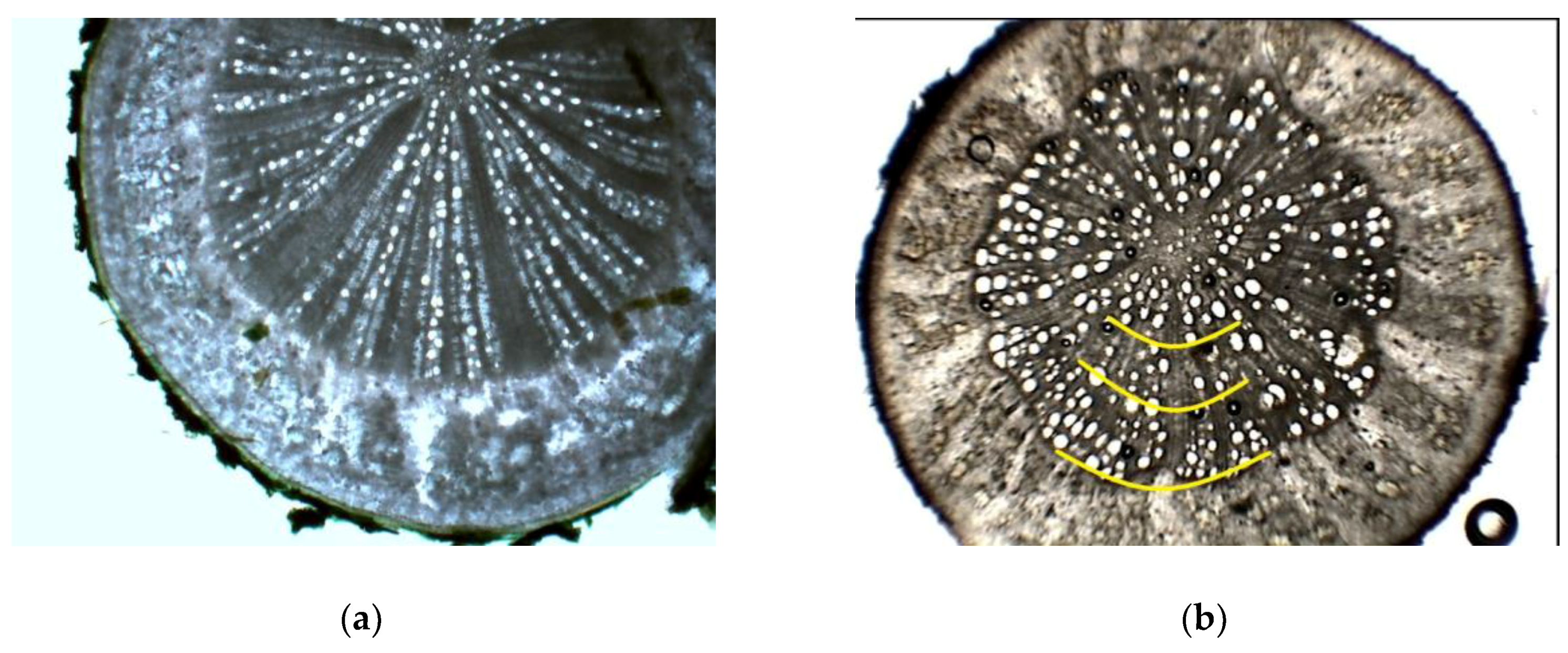
2.3. Anatomical Characteristics
3. Results
3.1. Root Anatomical Characteristics of Investigated Cherry Genotypes
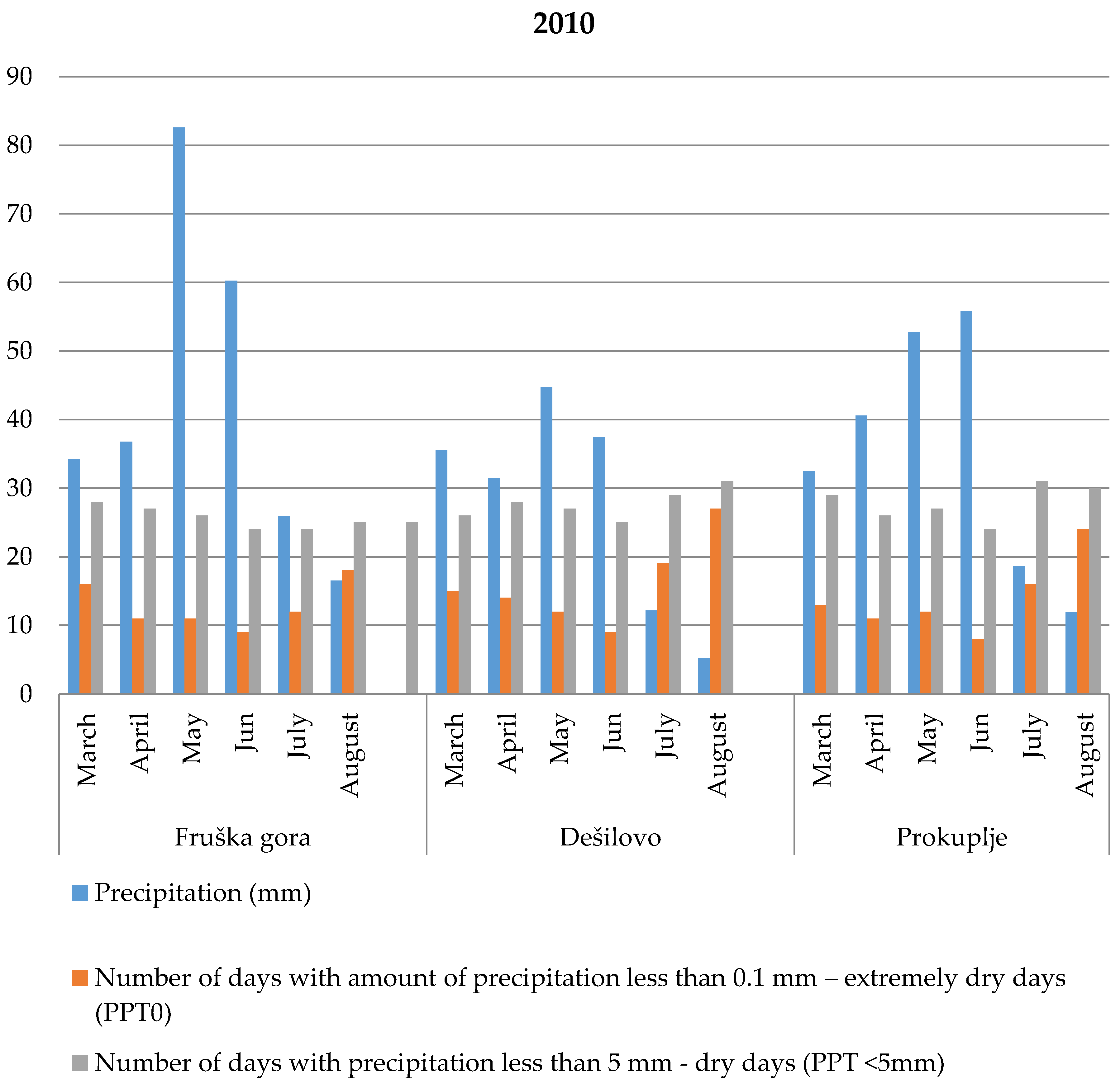
3.2. Meteorological Characteristics During Root Secondary Thickening
3.2.1. Fruška Gora
3.2.2. Dešilovo
3.2.3. Prokuplje
3.3. Correlation Analysis of the Investigated Anatomical Characteristics and Weather Conditions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Faostat. 2010. Available online: www.faostat.fao.org (accessed on 2 November 2010).
- Habib, M.; Bhat, M.; Dar, B.N.; Wani, A.A. Sweet cherries from farm to table: A review. Crit. Rev. Food Sci. 2017, 57, 1638–1649. [Google Scholar] [CrossRef] [PubMed]
- Ljubojević, M.; Zorić, L.; Maksimović, I.; Dulić, J.; Miodragović, M.; Barać, G.; Ognjanov, V. Anatomically assisted cherry rootstock selection. Sci. Hortic. 2017, 217, 197–208. [Google Scholar] [CrossRef]
- Baas, P.; Ewers, F.W.; Davis, S.D.; Wheeler, E.A. Evolution of xylem physiology. In The Evolution of Plant Physiology; Hemsley, A.R., Poole, I., Eds.; Elsevier Academic Press: London, UK; San Deigo, CA, USA, 2004; pp. 273–295. [Google Scholar]
- Ljubojević, M.; Ognjanov, V.; Zorić, L.; Maksimović, I.; Merkulov, L.; Bošnjaković, D.; Barać, G. Modeling of water movement trough cherry plant as preselecting tool for prediction of tree vigor. Sci. Hortic. 2013, 160, 189–197. [Google Scholar] [CrossRef]
- Hajagos, A.; Végvári, G. Investigation of tissue structure and xylem anatomy of eight rootstocks of sweet cherry (Prunus avium L.). Trees Struct. Funct. 2013, 27, 53–60. [Google Scholar] [CrossRef]
- Tyree, M.T.; Sperry, J.S. Do woody plants operate near the point of catastrophic xylem dysfunction caused by dynamic water stress? Answers from a model. Plant Physiol. 1988, 88, 574–580. [Google Scholar] [CrossRef] [PubMed]
- Bryukhanova, M.; Fonti, P. Xylem plasticity allows rapid hydraulic adjustment to annual climatic variability. Trees Struct. Funct. 2013, 27, 485–496. [Google Scholar] [CrossRef]
- Lovisolo, C.; Schubert, A. Effects of water stress on vessel size and xylem hydraulic conductivity in Vitis vinifera L. J. Exp. Bot. 1998, 49, 693–700. [Google Scholar] [CrossRef]
- Curtu, A.L.; Alizoti, P.; Ballian, D. Marginal/peripheral populations of forest tree species and their conservation status: Report for southeastern Europe. Ann. Silvic. Res. 2018, 41, 42–47. [Google Scholar] [CrossRef]
- Fonti, P.; von Arx, G.; García-González, I.; Eilmann, B.; Sass-Klaassen, U.; Gärtner, H.; Eckstein, D. Studying global change through investigation of the plastic responses of xylem anatomy in tree rings. New Phytol. 2010, 185, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Deslauriers, A.; Anfodillo, T. Assessment of cambial activity and xylogenesis by microsampling tree species: An example at the Alpine timberline. Iawa. J. 2006, 27, 383–394. [Google Scholar] [CrossRef]
- Von Arx, G.; Archer, S.R.; Hughes, M.K. Long-term functional plasticity in plant hydraulic architecture in response to supplemental moisture. Ann. Bot. 2012, 109, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- Dee, J.R.; Palmer, M.W. Application of herb chronology: annual fertilization and climate reveal annual ring signatures within the roots of US tallgrass prairie plants. Botany 2016, 94, 277–288. [Google Scholar] [CrossRef]
- Dee, J.R.; Palmer, M.W. Annual rings of perennial forbs and mature oaks show similar effects of climate but inconsistent responses to fire in the North American prairie–forest ecotone. Can. J. Forest Res. 2017, 47, 716–726. [Google Scholar] [CrossRef]
- Stojnic, S.; Sass-Klaassen, U.; Orlovic, S.; Matovic, B.; Eilmann, B. Plastic growth response of European beech provenances to dry site conditions. Iawa. J. 2013, 34, 475–484. [Google Scholar] [CrossRef]
- Brunner, I.; Herzog, C.; Dawes, M.A.; Arend, M.; Sperisen, C. How tree roots respond to drought. Front Plant Sci. 2015, 6, 547. [Google Scholar] [CrossRef] [PubMed]
- ERA 5. 2018. Available online: https://confluence.ecmwf.int//display/CKB/What+is+ERA5 (accessed on 16 September 2018).
- Opština Prokuplje. Available online: https://prokuplje.org.rs/planiranje-i-izgradnja/ (accessed on 30 April 2018).
- Opština Merošina. Available online: http://www.merosina.org.rs/cir/Planska%20dokumenta (accessed on 30 April 2018).
- Zorić, L.; Ljubojević, M.; Merkulov, L.; Luković, J.; Ognjanov, V. Anatomical characteristics of cherry rootstocks as possible preselecting tools for prediction of tree vigor. J. Plant Growth Regul. 2012, 31, 320–331. [Google Scholar] [CrossRef]
- Waisel, Y.; Eshel, A.; Kafkafi, U. Plant Roots: The Hidden Half; Marcel Dekker, Inc.: New York, NY, USA, 1996; p. 1002. [Google Scholar]
- Wasson, A.P.; Richards, R.A.; Chatrath, R.; Misra, S.C.; Prasad, S.V.; Rebetzke, G.J.; Kirkegaard, J.A.; Christopher, J.; Watt, M. Traits and selection strategies to improve root systems and water uptake in water-limited wheat crops. J. Exp. Bot. 2012, 63, 3485–3498. [Google Scholar] [CrossRef] [PubMed]
- Treder, W.; Konopacki, P. Impact of quantity and intensity of rainfall on soil water content in an orchard located in the central part of Poland. J. Water Land Dev. 1999, 3, 47–58. [Google Scholar]
- Worbes, M. How to measure growth dynamics in tropical trees a review. Iawa. J. 1995, 16, 337–351. [Google Scholar] [CrossRef]
- Worbes, M. Annual growth rings, rainfall-dependent growth and long-term growth patterns of tropical trees from the Caparo Forest Reserve in Venezuela. J. Ecol. 1999, 87, 391–403. [Google Scholar] [CrossRef]
- Menezes, M.; Berger, U.; Worbes, M. Annual growth rings and long-term growth patterns of mangrove trees from the Bragança peninsula, North Brazil. Wetl. Ecol. Manag. 2003, 11, 233–242. [Google Scholar] [CrossRef]
- Keserović, Z.; Magazin, N.; Milić, B.; Dorić, M. The ecology of fruit species. In Fruit Science and Viticulture (Part Fruit Science); Keserović, Z., Magazin, N., Milić, B., Dorić, M., Eds.; University of Novi Sad, Faculty of Agriculture, Feljton: Novi Sad, Serbia, 2016; p. 58. ISBN 978-86-7520-370-4. (In Serbian) [Google Scholar]
- Janjić, V.; Olar, O. Fertigation of stone trees. In Irrigation and Fertilization of Orchards, Vineyards and Gardens; Janjić, V., Olar, O., Eds.; Nolit: Belgrade, Serbia, 1992; p. 71. ISBN 86-19-01914-7. (In Serbian) [Google Scholar]
- Arend, M.; Fromm, J. Seasonal change in the drought response of wood cell development in poplar. Tree Physiol. 2007, 27, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Hrotkó, K.; Rozpara, E. Rootstocks and Improvement. In Cherries: Botany, Production and Uses; Quero-García, J., Iezzoni, A., Pulawska, J., Lang, G.A., Eds.; CABI: Boston, MA, USA, 2017; pp. 117–139. [Google Scholar]
- Singh, A.; Shamim, M.; Singh, K.N. Genotypic variation in root anatomy, starch accumulation, and protein induction in upland rice (Oryza sativa) varieties under water stress. Agr. Res. 2013, 2, 24–30. [Google Scholar] [CrossRef]
- Eilmann, B.; Zweifel, R.; Buchmann, N.; Fonti, P.; Rigling, A. Drought-induced adaptation of the xylem in Scots pine and pubescent oak. Tree Physiol. 2009, 29, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Tyree, M.T.; Ewers, F.W. The hydraulic architecture of trees and other woody plants. New Phytol. 1991, 119, 345–360. [Google Scholar] [CrossRef]
- Tombesi, S.; Johnson, R.S.; Day, K.R.; DeJong, T.M. Relationships between xylem vessel characteristics, calculated axial hydraulic conductance and size-controlling capacity of peach rootstocks. Ann. Bot. 2009, 105, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Sperry, J.S.; Saliendra, N.Z. Intra- and inter-plant variation in xylem cavitation in Betula occidentalis. Plant Cell Environ. 1994, 17, 1233–1241. [Google Scholar] [CrossRef]
- Maherali, H.; DeLucia, E.H. Xylem conductivity and vulnerability to cavitation of ponderosa pine growing in contrasting climates. Tree Physiol. 2000, 20, 859–867. [Google Scholar] [CrossRef] [PubMed]

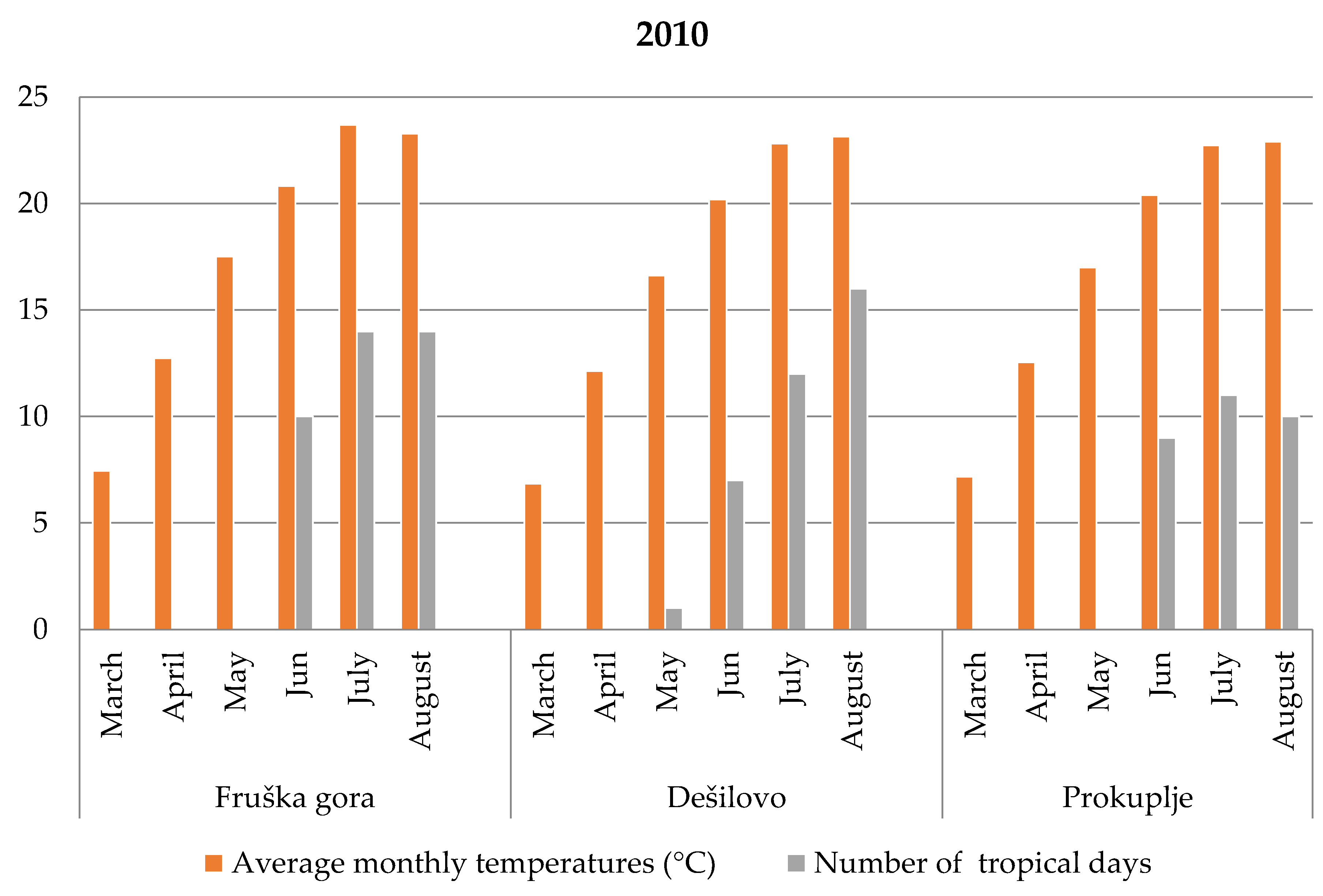
| Species | Locality | Longitude | Latitude | Altitude |
|---|---|---|---|---|
| European ground cherry | Fruška gora | 19°55′01′′ E | 45°09′39′′ N | 327 m |
| Oblačinska sour cherry | Dešilovo | 21°37′35′′ E | 43 17′34′′ N | 398 m |
| Oblačinska sour cherry | Prokuplje | 21°32′54′′ E | 43 13′46′′ N | 274 m |
| Genotype | Cross-Section Radius (mm) | Cross-Section Area (mm2) | Secondary Wood Area (%) | Secondary Cortex Area (%) | Wood/Cortex Ratio | Periderm Area (%) |
|---|---|---|---|---|---|---|
| European ground cherry—Locality Fruška gora | ||||||
| SV2 | 2.98 b | 27.9 ± 3.70 a,b | 52.5 ± 3.03 a | 43.8 ± 3.90 c | 1.21 ± 0.17 a | 3.70 ± 1.66 d |
| SV4 | 3.24 a | 33.4 ± 10.1 a | 39.5 ± 13.4 b,c,d | 47.8 ± 12.1 b,c | 0.89 ± 0.51 b | 12.7 ± 1.41 a |
| Oblačinska sour cherry—Locality Dešilovo | ||||||
| OV11 | 1.99 d | 12.7 ± 4.18 c–f | 35.4 ± 5.16 b-e | 60.1 ± 7.84 a | 0.61 ± 0.15 c,d,e | 4.32 ± 3.07 c,d |
| OV12 | 1.80 d | 10.3 ± 2.73 e,f | 39.6 ± 3.60 b,c,d | 52.8 ± 2.59 a,b | 0.76 ± 0.10 b-e | 7.64 ± 1.11 b,c,d |
| OV13 | 1.96 d | 12.3 ± 4.03 c–f | 37.3 ± 1.79 b,c,d | 54.0 ± 3.88 a,b | 0.70 ± 0.07 b-e | 8.70 ± 2.94 a,b,c |
| OV14 | 2.35 b,c | 17.5 ± 3.75 c,d,e | 34.1 ± 1.80 c,d,e | 58.1 ± 2.30 a | 0.59 ± 0.05 c,d,e | 7.73 ± 1.11 b,c,d |
| OV16 | 2.26 c,d | 16.5 ± 5.29 c,d,e | 33.3 ± 2.70 d,e | 59.3 ± 2.87 a | 0.56 ± 0.07 d,e | 7.64 ± 3.15 b,c,d |
| OV17 | 2.17 c,d | 14.9 ± 2.39 c–f | 38.3 ± 4.47 b,c,d | 52.8 ± 7.21 a,b | 0.75 ± 0.19 b-e | 8.85 ± 3.61 a,b,c |
| OV18 | 1.48 e | 7.24 ± 3.81 f | 30.3 ± 0.98 e | 59.1 ± 1.44 a | 0.51 ± 0.02 e | 10.7 ± 1.70 a,b |
| Oblačinska sour cherry—Locality Prokuplje | ||||||
| OV31 | 1.83 d | 10.9 ± 5.40 d,e,f | 35.2 ± 3.19 b–e | 58.4 ± 3.42 a | 0.61 ± 0.09 c,d,e | 6.42 ± 1.67 b,c,d |
| OV32 | 2.44 b,c | 19.0 ± 5.38 c,d | 41.4 ± 4.17 b | 51.5 ± 4.21 a,b,c | 0.81 ± 0.15 b,c | 7.03 ± 0.80 b,c,d |
| OV33 | 2.11 c,d | 14.1 ± 3.09 c–f | 40.6 ± 4.76 b,c | 51.8 ± 4.51 a,b,c | 0.80 ± 0.16 b,c,d | 7.55 ± 0.44 b,c,d |
| OV34 | 2.51 b | 20.8 ± 10.5 b,c | 35.2 ± 3.91 b-e | 54.9 ± 9.18 a,b | 0.66 ± 0.17 b–e | 9.85 ± 7.16 a,b |
| Genotype | Average Vessel Lumen Area in External Root Wood Zone (μm2) | Average Vessel Lumen Area in Middle root Wood Zone (μm2) | Average Vessel lumen Area in Inner Root Wood Zone (μm2) | Average Vessel Lumen Area on Complete Cross-Section (μm2) |
|---|---|---|---|---|
| European ground cherry—Locality Fruška gora | ||||
| SV2 | 916 ± 36.1 d | 917 ± 82.0 c | 944 ± 72.9 c,d | 926 ± 28.8 c,d |
| SV4 | 1399 ± 921 b,c,d | 1528 ± 841 a | 1530 ± 1307 a,b,c | 1486 ± 1023 b,c |
| Oblačinska sour cherry—Locality Dešilovo | ||||
| OV11 | 1511 ± 243 b,c,d | / d | 1468 ± 140 a,b,c | 1489 ± 154 b,c |
| OV12 | 1609 ± 350 b,c | / d | 1414 ± 431 b,c | 1512 ± 382 b |
| OV13 | 1402 ± 245 b,c,d | / d | 1264 ± 182 b,c,d | 1333 ± 204 b,c,d |
| OV14 | 1420 ± 212 b,c,d | / d | 1630 ± 235 a,b | 1525 ± 204 b |
| OV16 | 2353 ± 676 a | / d | 2099 ± 634 a | 2226 ± 622 a |
| OV17 | 2042 ± 225 a,b | / d | 1370 ± 228 b,c | 1706 ± 208 b |
| OV18 | 1276 ± 341 c,d | / d | 1524 ± 282 a,b,c | 1400 ± 309 b,c |
| Oblačinska sour cherry—Locality Prokuplje | ||||
| OV31 | 1719 ± 220 b,c | 1685 ± 102 a | 1651 ± 298 a,b | 1685 ± 220 b |
| OV32 | 1355 ± 284 b,c,d | 1220 ± 30.2 b | 1195 ± 290 b,c,d | 1257 ± 189 b,c,d |
| OV33 | 898 ± 31.7 d | 939 ± 271 c | 701 ± 92.9 d | 846 ± 102 d |
| OV34 | 1472 ± 864 b,c,d | 1329 ± 101 a,b | 1051 ± 468 b,c,d | 1284 ± 471 b,c,d |
| Genotype | Average Vessel Number in External Root Wood Zone | Average Vessel Number in Middle Root Wood Zone | Average Vessel Number in Inner Root Wood Zone | Average Vessel Number on the Complete Cross-Section |
|---|---|---|---|---|
| European ground cherry—Locality Fruška gora | ||||
| SV2 | 94.7 ± 9.98 a,b | 79.6 ± 8.52 c | 90.0 ± 21.3 c,d | 88.1 ± 13.3 c,d |
| SV4 | 82.4 ± 12.0 b | 78.1 ± 1.98 c | 88.0 ± 4.01 d | 82.8 ± 2.00 d |
| Oblačinska sour cherry—Locality Dešilovo | ||||
| OV11 | 85.8 ± 14.4 b | / d | 90.3 ± 10.5 b,c,d | 88.1 ± 10.1 c,d |
| OV12 | 92.0 ± 19.0 a,b | / d | 106 ± 3.10 a–d | 98.9 ± 9.03 a–d |
| OV13 | 94.5 ± 13.6 a,b | / d | 97.6 ± 14.0 b,c,d | 94.5 ± 13.6 c,d,e |
| OV14 | 127 ± 15.6 a | / d | 113 ± 31.3 a-d | 114 ± 17.9 a,b |
| OV16 | 105 ± 12.9 a,b | / d | 120 ± 5.82 a,b,c | 113 ± 8.31 a,b |
| OV17 | 87.4 ± 12.4 b | / d | 126 ± 21.9 a | 107 ± 11.0 a,b,c |
| OV18 | 95.9 ± 27.1 a,b | / d | 106 ± 15.8 a–d | 101 ± 20.5 a–s |
| Oblačinska sour cherry—Locality Prokuplje | ||||
| OV31 | 112 ± 20.4 a,b | 109 ± 6.75 a,b | 107 ± 8.86 a–d | 110 ± 8.71 a,b,c |
| OV32 | 109 ± 16.5 a,b | 111 ± 9.28 a,b | 109 ± 26.9 a–d | 110 ± 16.8 a,b,c |
| OV33 | 88.8 ± 15.5 b | 102 ± 26.6 b | 118 ± 27.4 a–d | 103 ± 11.8 a–d |
| OV34 | 125 ± 49.6 a | 116 ± 16.2 a | 121 ± 17.6 a,b | 121 ± 25.7 a |
| Anatomical Characteristic/Month | March | April | May | June | July | August | May–June | June–July | July–August |
|---|---|---|---|---|---|---|---|---|---|
| Cross-section radius (mm) | 0.40 | −0.30 | −0.80 | −0.56 | −0.65 | −0.83 | −0.75 | 0.29 | −0.57 |
| Cross-section area (mm2) | 0.40 | −0.29 | −0.85 | −0.57 | −0.70 | −0.87 | −0.77 | 0.33 | −0.58 |
| Secondary wood area (%) | 0.38 | −0.30 | −0.66 | −0.50 | −0.57 | −0.69 | −0.65 | 0.20 | −0.52 |
| Secondary cortex area (%) | −0.41 | 0.32 | 0.77 | 0.56 | 0.61 | 0.80 | 0.73 | −0.25 | 0.57 |
| Wood/cortex ratio | 0.38 | −0.29 | −0.78 | −0.54 | −0.64 | −0.81 | −0.73 | 0.29 | −0.56 |
| Periderm area (%) | −0.03 | 0.04 | −0.05 | 0.01 | −0.07 | −0.04 | −0.01 | 0.07 | 0.01 |
| AVL External root wood zone | −0.43 | 0.39 | 0.37 | 0.48 | 0.21 | 0.44 | 0.51 | 0.07 | 0.48 |
| AVL Middle root wood zone | 0.76 | −0.70 | −0.55 | −0.83 | −0.25 | −0.67 | −0.84 | −0.21 | −0.83 |
| AVL Inner root wood zone | −0.53 | 0.52 | 0.17 | 0.53 | −0.04 | 0.27 | 0.46 | 0.31 | 0.52 |
| AVL Complete cross-section | −0.50 | 0.47 | 0.29 | 0.52 | 0.10 | 0.38 | 0.51 | 0.19 | 0.53 |
| AVN External root wood zone | 0.24 | −0.29 | 0.35 | −0.13 | 0.45 | 0.27 | 0.05 | −0.47 | −0.12 |
| AVN Middle root wood zone | 0.64 | −0.60 | −0.48 | −0.71 | −0.23 | −0.59 | −0.72 | −0.17 | −0.71 |
| AVN Inner root wood zone | 0.06 | −0.14 | 0.61 | 0.10 | 0.65 | 0.56 | 0.33 | −0.53 | 0.11 |
| AVN Complete cross-section | 0.18 | −0.27 | 0.65 | 0.00 | 0.73 | 0.56 | 0.22 | −0.68 | 0.02 |
| Anatomical Characteristic/Month | May | June | July | August |
|---|---|---|---|---|
| Cross-section radius (mm) | −0.56 | −0.70 | 0.62 | −0.19 |
| Cross-section area (mm2) | −0.57 | −0.72 | 0.66 | −0.18 |
| Secondary wood area (%) | −0.50 | 0.61 | 0.48 | −0.21 |
| Secondary cortex area (%) | 0.56 | 0.69 | −0.57 | 0.21 |
| Wood/cortex ratio | −0.54 | −0.68 | 0.60 | −0.18 |
| Periderm area (%) | 0.01 | 0.00 | 0.07 | 0.04 |
| AVL External root wood zone | 0.48 | −0.51 | −0.17 | 0.34 |
| AVL Middle root wood zone | −0.83 | 0.85 | 0.19 | −0.33 |
| AVL Inner root wood zone | 0.53 | −0.48 | 0.08 | 0.50 |
| AVL Complete cross-section | 0.52 | −0.52 | −0.06 | 0.43 |
| AVN External root wood zone | −0.13 | 0.01 | −0.46 | −0.34 |
| AVN Middle root wood zone | −0.71 | −0.73 | 0.17 | −0.53 |
| AVN Inner root wood zone | 0.10 | −0.26 | −0.44 | −0.23 |
| AVN Complete cross-section | 0.00 | −0.19 | −0.73 | −0.36 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ljubojević, M.; Maksimović, I.; Lalić, B.; Dekić, L.; Narandžić, T.; Magazin, N.; Dulić, J.; Miodragović, M.; Barać, G.; Ognjanov, V. Environmentally-Related Cherry Root Cambial Plasticity. Atmosphere 2018, 9, 358. https://doi.org/10.3390/atmos9090358
Ljubojević M, Maksimović I, Lalić B, Dekić L, Narandžić T, Magazin N, Dulić J, Miodragović M, Barać G, Ognjanov V. Environmentally-Related Cherry Root Cambial Plasticity. Atmosphere. 2018; 9(9):358. https://doi.org/10.3390/atmos9090358
Chicago/Turabian StyleLjubojević, Mirjana, Ivana Maksimović, Branislava Lalić, Ljiljana Dekić, Tijana Narandžić, Nenad Magazin, Jovana Dulić, Maja Miodragović, Goran Barać, and Vladislav Ognjanov. 2018. "Environmentally-Related Cherry Root Cambial Plasticity" Atmosphere 9, no. 9: 358. https://doi.org/10.3390/atmos9090358
APA StyleLjubojević, M., Maksimović, I., Lalić, B., Dekić, L., Narandžić, T., Magazin, N., Dulić, J., Miodragović, M., Barać, G., & Ognjanov, V. (2018). Environmentally-Related Cherry Root Cambial Plasticity. Atmosphere, 9(9), 358. https://doi.org/10.3390/atmos9090358






