A Review of Airborne Particulate Matter Effects on Young Children’s Respiratory Symptoms and Diseases
Abstract
1. Introduction
1.1. What Is Particulate Matter and Where Does It Come From?
1.2. Chemical Composition of Particulate Matter and Its Potential Health Effects
1.3. Objective of the Study
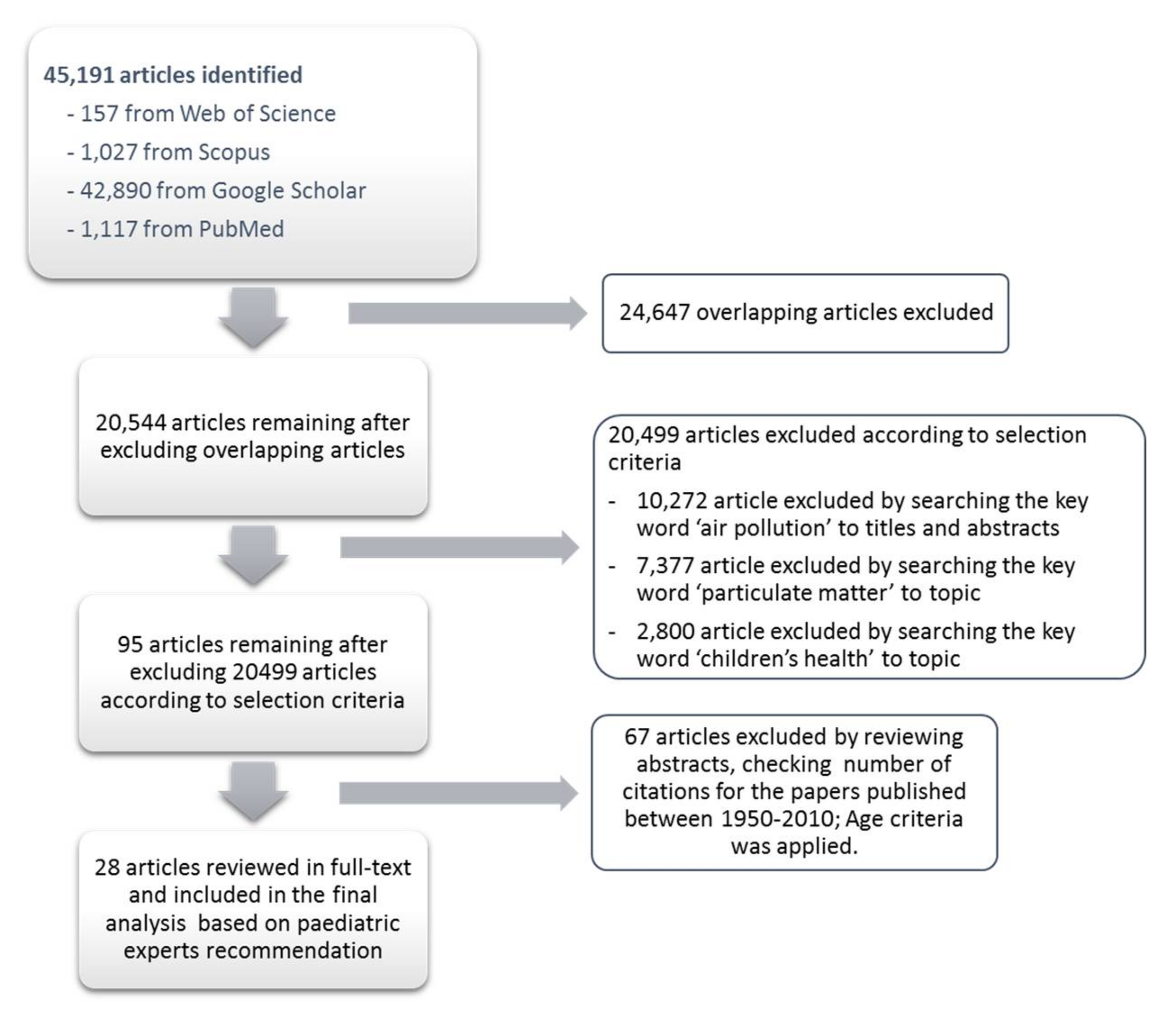
2. Methodology
3. Results
3.1. Databases’ Automatic Search Results
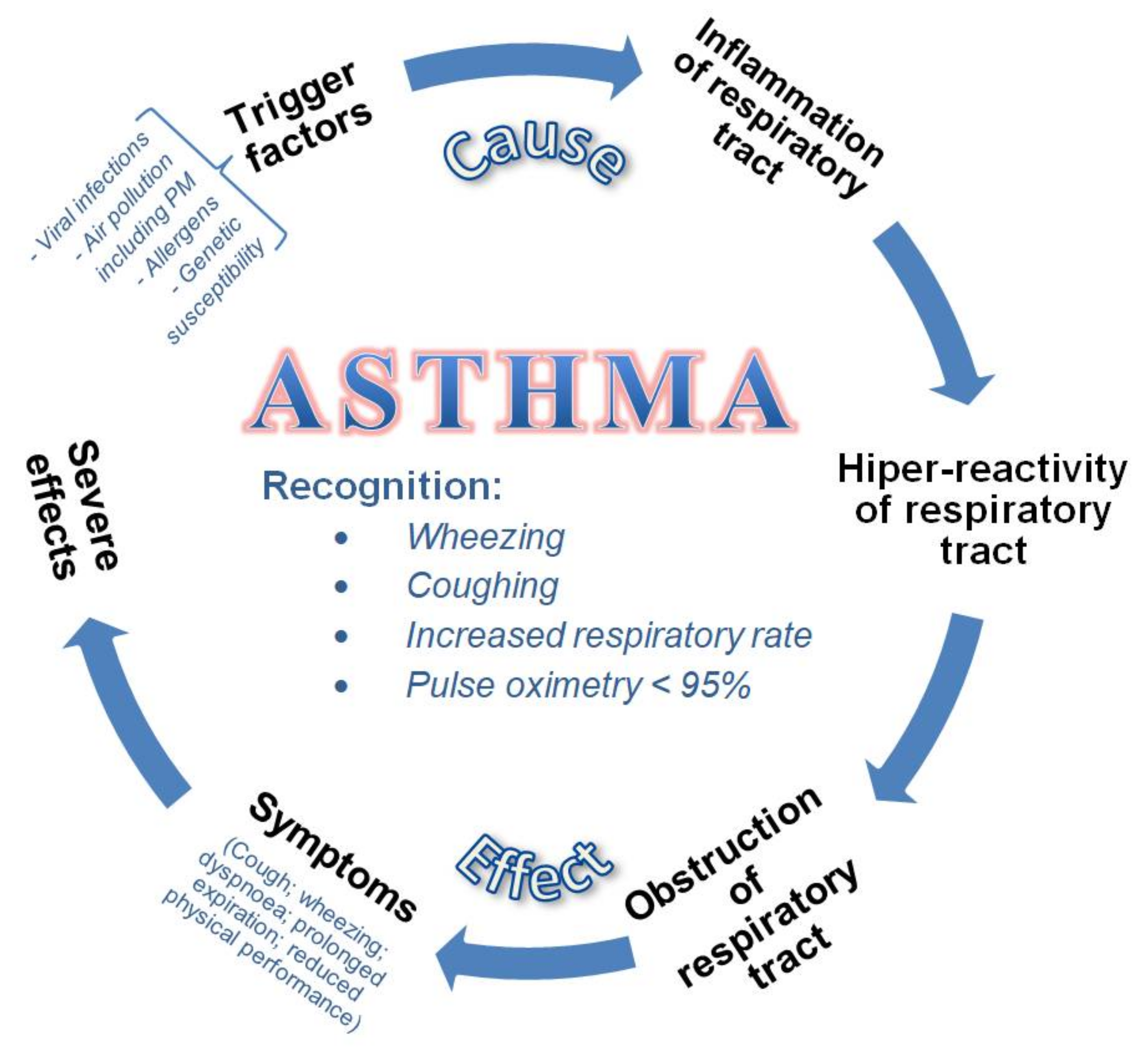
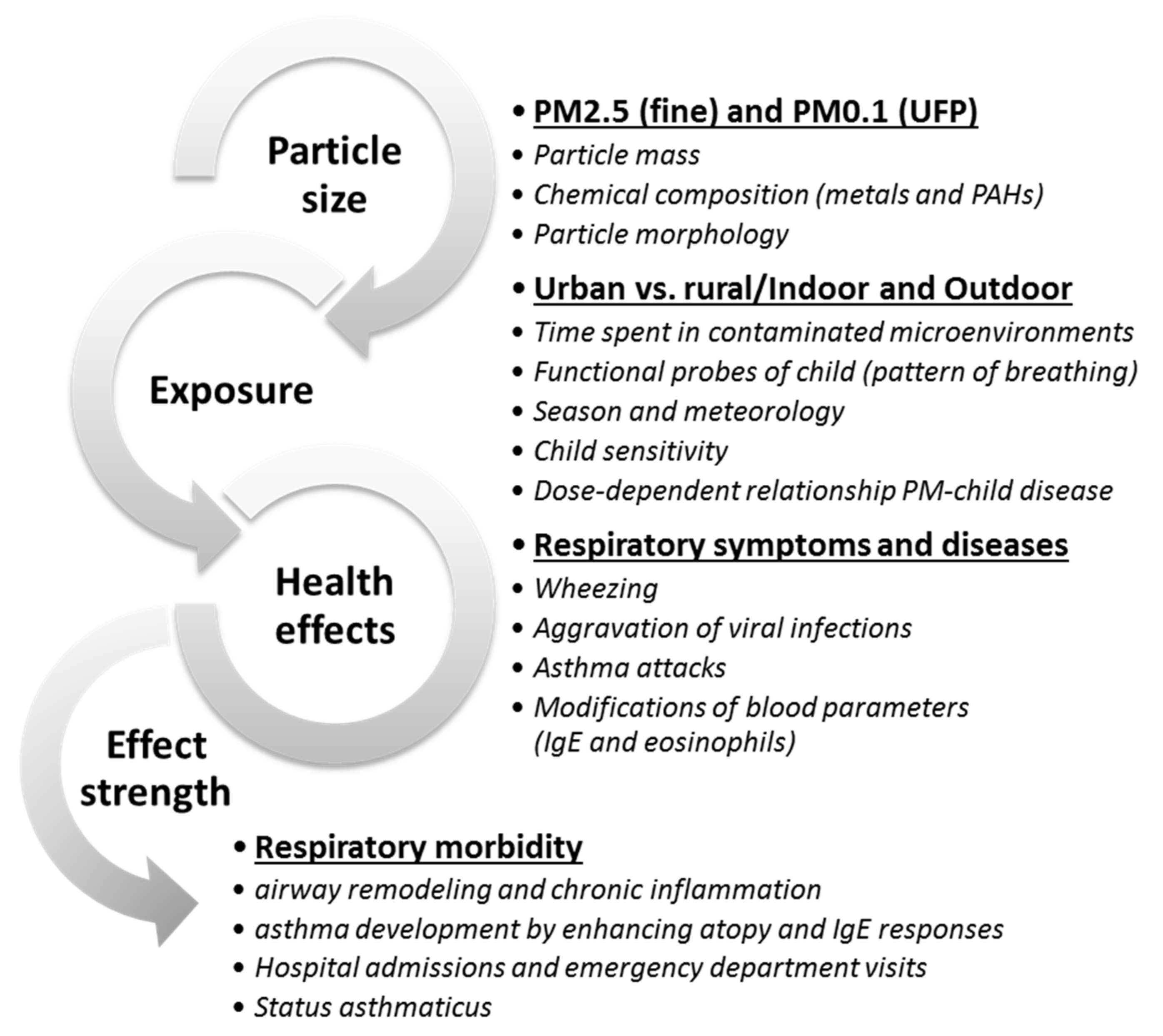
3.2. Current Knowledge on the Effects of Particulate Matter on Children’s Health
3.2.1. Growth and Development of Children in Relation to Exposure to Air Pollution
3.2.2. Effects of Exposure to Particulate Matter on the Pulmonary Function in Children
Pulmonary Function and PM Impairment Mechanism
Quantitative Effects of PM on Pulmonary Function
3.2.3. Effects of Exposure to Particulate Matter on Children’s Health: Short- vs. Long-Term and Outdoor vs. Indoor
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kampa, M.; Castanas, E. Human health effects of air pollution. Environ. Pollut. 2007, 151, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-Y.; Bartonova, A.; Schindler, M.; Sharma, M.; Behera, S.N.; Katiyar, K.; Dikshit, O. Respiratory disease in relation to outdoor air pollution in Kanpur, India. Arch. Environ. Occup. Health 2013, 68, 204–217. [Google Scholar] [CrossRef] [PubMed]
- Health Effects of Particulate Matter—Policy Implications for Countries in Eastern Europe, Caucasus and Central Asia. Available online: http://www.euro.who.int/__data/assets/pdf_file/0006/189051/Health-effects-of-particulate-matter-final-Eng.pdf (accessed on 24 April 2017).
- Moshammer, H.; Bartonova, A.; Hanke, W.; van den Hazel, P.; Koppe, J.G.; Krämer, U.; Ronchetti, R.; Sram, R.J.; Wallis, M.; Wallner, P.; et al. Air pollution: A threat to the health of our children. ACTA Paediatr. 2006, 95 (Suppl. S453), 93–105. [Google Scholar] [CrossRef] [PubMed]
- Henschel, S.; Atkinson, R.; Zeka, A.; Le Tertre, A.; Analitis, A.; Katsouyanni, K.; Chanel, O.; Pascal, M.; Forsberg, B.; Medina, S.; et al. Air pollution interventions and their impact on public health. Int. J. Public Health 2012, 57, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Beelen, R.; Hoek, G.; van den Brandt, P.A.; Goldbohm, R.A.; Fischer, P.; Schouten, L.J.; Jerrett, M.; Hughes, E.; Armstrong, B.; Brunekreef, B. Long-term effects of traffic-related air pollution on mortality in a Dutch cohort (NLCS-AIR Study). Environ. Health Perspect. 2008, 116, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Samoli, E.; Peng, R.; Ramsay, T.; Pipikou, M.; Touloumi, G.; Dominici, F.; Burnett, R.; Cohen, A.; Krewski, D.; Samet, J.; et al. Acute effects of ambient particulate matter on mortality in Europe and North America: Results from the APHENA Study. Environ. Health Perspect. 2008, 116, 1480–1486. [Google Scholar] [CrossRef] [PubMed]
- Extended Follow-Up and Spatial Analysis of the American Cancer Society Linking Particulate Air Pollution and Mortality Boston. Available online: http://www.healtheffects.org/Pubs/RR140-Krewski.pdf (accessed on 23 April 2017).
- Pope, C.A., III; Burnett, R.T.; Turner, M.C.; Cohen, A.; Krewski, D.; Jerrett, M.; Gapstur, S.M.; Thun, M.J. Lung Cancer and Cardiovascular Disease Mortality Associated with Ambient Air Pollution and Cigarette Smoke: Shape of the Exposure-Response Relationships. Environ. Health Perspect. 2011, 119, 1616–1621. [Google Scholar] [CrossRef] [PubMed]
- Ambient (Outdoor) Air Quality and Health—Fact Sheet. Available online: http://www.who.int/mediacentre/factsheets/fs313/en (accessed on 16 February 2017).
- Feng, S.; Gao, D.; Liao, F.; Zhou, F.; Wang, X. The health effects of ambient PM2.5 and potential mechanisms. Ecotox. Environ. Safe 2016, 128, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Children are Not Little Adults. Available online: http://www.who.int/ceh/capacity/Children_are_not_little_adults.pdf (accessed on 10 January 2017).
- Briggs, D. Environmental pollution and the global burden of disease. Br. Med. Bull. 2003, 68, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Stanek, L.W.; Sacks, J.D.; Dutton, S.J.; Dubois, J.J.B. Attributing health effects to apportioned components and sources of particulate: An evaluation of collective results. Atmos. Environ. 2011, 45, 5655–5663. [Google Scholar] [CrossRef]
- Brauer, M.; Amann, M.; Burnett, R.T.; Cohen, A.; Dentener, F.; Ezzati, M.; Henderson, S.B.; Krzyzanowski, M.; Martin, R.V.; Van Dingenen, R.; et al. Thurston GD. Exposure assessment for estimation of the global burden of disease attributable to outdoor air pollution. Environ. Sci. Technol. 2012, 46, 652–660. [Google Scholar] [CrossRef] [PubMed]
- Kliegman, R.; Stanton, B.; St. Geme, J.; Schor, N. Nelson textbook of Pediatrics, 20th ed.; Elsevier: Philadelphia, PA, USA, 2016; pp. 1–3888. ISBN 9781455775668. [Google Scholar]
- Gasana, J.; Dilikar, D.; Mendy, A.; Forno, E.; Ramos-Vieira, E. Motor vehicle air pollution and asthma in children: A meta-analysis. Environ. Res. 2012, 117, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Simpson, C.; Sheikh, A. Trends in the epidemiology of asthma in England: A national study of 333, 294 patients. J. R. Soc. Med. 2010, 103, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Dunea, D.; Iordache, S.; Pohoata, A. Fine particulate matter in urban environments: A trigger of respiratory symptoms in sensitive children. Int. J. Environ. Res. Public Health 2016, 13, 1246. [Google Scholar] [CrossRef] [PubMed]
- Dusseldorp, A.; Kruize, H.; Brunekreef, B.; Hofschreuder, P.; de Meer, G.; van Oudvorst, A.B. Associations of PM10 and airborne iron with respiratory health of adults near a steel factory. Am. J. Respir. Crit. Care 1995, 152, 1932–1939. [Google Scholar] [CrossRef] [PubMed]
- Quantitative Health Risk Assessment for Particulate Matter. Available online: https://www3.epa.gov/ttn/naaqs/standards/pm/data/PM_RA_FINAL_June_2010.pdf (accessed on 12 March 2018).
- Harrison, R.M.; Jones, A.M.; Lawrence, R.G. Major component composition of PM10 and PM2.5 from roadside and urban background sites. Atmos. Environ. 2004, 38, 4531–4538. [Google Scholar] [CrossRef]
- Iordache, S.; Dunea, D. Methods for the Assessment of Air Pollution with Particulate Matter to Children’s Health (in Romanian with English Abstracts), 1st ed.; Matrix Rom: Bucharest, Romania, 2014; ISBN 978-606-25-0121-1. [Google Scholar]
- Ostro, B.D.; Rothschild, S. Air pollution and acute respiratory morbidity: An observational study of multiple pollutants. Environ. Res. 1989, 50, 238–247. [Google Scholar] [CrossRef]
- Hiltermann, T.J.; Stolk, J.; van der Zee, S.C.; Brunekreef, B.; de Bruijne, C.R.; Fischer, P.H.; Ameling, C.B.; Sterk, P.J.; Hiemstra, P.S.; van Bree, L. Asthma severity and susceptibility to air pollution. Eur. Respir. J. 1998, 11, 686–693. [Google Scholar] [PubMed]
- Roemer, W.; Hoek, G.; Brunekreef, B. Pollution effects on asthmatic children in Europe: The PEACE study. Clin. Exp. Allergy 2000, 30, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Hajat, S.; Anderson, H.; Atkinson, R.; Haines, A.; Seaton, A. Effects of air pollution on general practitioner consultations for upper respiratory diseases in London. Occup. Environ. Med. 2002, 59, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Dhondt, S.; Beckx, C.; Degraeuwe, B.; Lefebvre, W.; Kochan, B.; Bellemans, T.; Panis, L.I.; Macharis, C.; Putman, K. Health impact assessment of air pollution using a dynamic exposure profile: Implications for exposure and health impact estimates. Environ. Impact Assess. 2012, 36, 42–51. [Google Scholar] [CrossRef]
- Patel, M.M.; Quinn, J.W.; Jung, K.H.; Hoepner, L.; Diaz, D.; Perzanowski, M.; Rundle, A.; Kinney, P.L.; Perera, F.P.; Miller, R.L. Traffic density and stationary sources of air pollution associated with wheeze, asthma, and immunoglobulin E from birth to age 5 years among New York City children. Environ. Res. 2011, 111, 1222–1229. [Google Scholar] [CrossRef] [PubMed]
- Pope, C.A., III; Dockery, D.W. Health effects of fine particulate air pollution: Lines that connect. J. Air Waste Manag. Assoc. 2006, 56, 709–742. [Google Scholar] [CrossRef] [PubMed]
- Fine Particulate Matter (PM2.5) in the United Kingdom. Available online: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/69635/pb13837-aqeg-fine-particle-matter-20121220.pdf (accessed on 12 March 2018).
- Seinfeld, J.; Pandis, S. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change, 2nd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1998; p. 97. ISBN 0-471-17816-0. [Google Scholar]
- Omidvarborna, H.; Kumar, A.; Kim, D.-S. Recent studies on soot modeling for diesel combustion. Renew. Sustain. Energy Rev. 2015, 48, 635–647. [Google Scholar] [CrossRef]
- Chereminisoff, N.P. Handbook of Air Pollution Prevention and Control, 1st ed.; Butterworth-Heinemann: Oxford, UK, 2002; pp. 1–477. [Google Scholar]
- Brown, J.S.; Gordon, T.; Price, O.; Asgharian, B. Thoracic and respirable particle definitions for human health risk assessment. Part Fibre Toxicol. 2013, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Quality Assurance Project Plan for Pittsburgh Air Quality Study (PAQS). Available online: https://www3.epa.gov/ttnamti1/files/ambient/super/pittqapp.pdf (accessed on 16 February 2017).
- Guideline on Speciated Particulate Monitoring. Available online: https://www3.epa.gov/ttnamti1/files/ambient/pm25/spec/drispec.pdf (accessed on 12 March 2018).
- Dai, Q.-L.; Bi, X.-H.; Wu, J.-H.; Zhang, Y.-F.; Wang, J.; Xu, H.; Yao, L.; Jiao, L.; Feng, Y.-C. Characterization and source identification of heavy metals on ambient PM10 and PM2.5 in an integrated iron and steel industry zone compared with a background site. Aerosol. Air Qual. Res. 2015, 15, 875–887. [Google Scholar] [CrossRef]
- Wiseman, C.L.S.; Zereini, F. Airborne particulate matter, platinum group elements and human health: A review of recent evidence. Sci. Total Environ. 2009, 407, 2493–2500. [Google Scholar] [CrossRef] [PubMed]
- Davidson, C.I.; Phalen, R.F.; Solomon, P.A. Airborne Particulate Matter and Human Health: A Review. Aerosol. Sci. Technol. 2005, 39, 737–749. [Google Scholar] [CrossRef]
- Kim, K.-H.; Kabir, E.; Kabir, S. A review on the human health impact of airborne particulate matter. Environ. Int. 2015, 74, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, M.; Vinzents, P.; Petersen, J.H.; Kleinjans, J.C.; Plas, G.; Kirsch-Volders, M.; Dostál, M.; Rössner, P.; Beskid, O.; Sram, R.J.; et al. Cytogenetic effects in children and mothers exposed to air pollution assessed by the frequency of micronuclei and fluorescence in situ hybridization (FISH): A family pilot study in the Czech Republic. Mutat. Res. 2006, 608, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscip. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Shafy, H.I.; Mansour, M.S.M. A review on polycyclic aromatic hydrocarbons: Source, environmental impact, effect on human health and remediation. Egypt. J. Pet. 2016, 25, 107–123. [Google Scholar] [CrossRef]
- Amodio, M.; Andriani, E.; de Gennaro, G.; Di Gilio, A.; Ielpo, P.; Placentino, C.M.; Tutino, M. How a steel plant affects air quality of a nearby urban area: A study on metals and PAH concentrations. Aerosol. Air Qual. Res. 2013, 13, 497–508. [Google Scholar] [CrossRef]
- Dunea, D.; Pohoata, A.; Iordache, S. Using wavelet-feedforward neural networks to improve air pollution forecasting in urban environments. Environ. Monit. Assess. 2015, 187, 477. [Google Scholar] [CrossRef] [PubMed]
- Development of Environment and Health Indicators for European Union countries: Results of a Pilot Study. Available online: http://apps.who.int/iris/bitstream/10665/107620/1/E85061.pdf (accessed on 12 March 2018).
- Liu, H.-Y.; Bartonova, A.; Pascal, M.; Smolders, R.; Skjetne, E.; Dusinska, M. Approaches to integrated monitoring for environment and health impact assessment. Environ. Health 2012, 11, 88. [Google Scholar] [CrossRef] [PubMed]
- Jedrychowski, W.A.; Perera, F.P.; Spengler, J.D.; Mroz, E.; Stigter, L.; Flak, E.; Majewska, R.; Klimaszewska-Rembiasz, M.; Jacek, R. Intrauterine exposure to fine particulate matter as a risk factor for increased susceptibility to acute broncho-pulmonary infections in early childhood. Int. J. Hyg. Environ. Health 2013, 216, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.H.; Chiu, Y.H.; Coull, B.A.; Kloog, I.; Schwartz, J.; Lee, A.; Wright, R.O.; Wright, R.J. Prenatal Particulate Air Pollution and Asthma Onset in Urban Children. Identifying Sensitive Windows and Sex Differences. Am. J. Respir. Crit. Care Med. 2015, 192, 1052–1059. [Google Scholar] [PubMed]
- Dunea, D.; Iordache, S.; Liu, H.-Y.; Bøhler, T.; Pohoata, A.; Radulescu, C. Quantifying the impact of PM2.5 and associated heavy metals on respiratory health of children near metallurgical facilities. Environ. Sci. Pollut. Res. 2016, 23, 15395–15406. [Google Scholar] [CrossRef] [PubMed]
- Athanazio, R. Airway disease: Similarities and differences between asthma, COPD and bronchiectasis. Clinics 2012, 67, 1335–1343. [Google Scholar] [CrossRef]
- Popescu, V. Pathology of Respiratory System in Children (In Romanian); Teora: Bucharest, Romania, 1999. [Google Scholar]
- Moshammer, H.; Neuberger, M. The active surface of suspended particles as a predictor of lung function and pulmonary symptoms in Austrian school children. Atmos. Environ. 2003, 37, 1737–1744. [Google Scholar] [CrossRef]
- Thamrin, C.; Taylor, D.R.; Jones, S.L.; Suki, B.; Frey, U. Variability of lung function predicts loss of asthma control following withdrawal of inhaled corticosteroid treatment Free. Asthma 2010, 65, 403–408. [Google Scholar]
- Noonan, C.W.; Ward, T.J. Asthma randomized trial of indoor wood smoke (ARTIS): Rationale and methods. Contemp. Clin. Trials 2012, 33, 1080–1087. [Google Scholar] [CrossRef] [PubMed]
- Pohoata, M.; Buzatoiu, C. Current knowledge regarding the problems generated by the particulate matters on children’s health. In Methods for the Assessment of Air Pollution with Particulate Matter to Children’s Health; Iordache, Ş., Dunea, D., Eds.; Matrix Rom: Bucharest, Romania, 2014; pp. 267–290. ISBN 978-606-25-0121-1. [Google Scholar]
- Greene, N.A.; Morris, V.R. Assessment of public health risks associated with atmospheric exposure to PM2.5 in Washington, DC, USA. Int. J. Environ. Res. Public Health 2006, 3, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Alves de Oliveira, B.F.; Ignotti, E.; Artaxo, P.; Hilário do Nascimento Saldiva, P.; Leite Junger, W.; Hacon, S. Risk assessment of PM2.5 to child residents in Brazilian Amazon region with biofuel production. Environ. Health 2012, 11, 64. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, A.; Henry, F.S.; Butler, J.P. Particle transport and deposition: Basic physics of particle kinetics. Compr. Physiol. 2013, 3, 1437–1471. [Google Scholar] [PubMed]
- Gauderman, W.J.; Avol, E.; Gilliland, F.; Vora, H.; Thomas, D.; Berhane, K.; McConnell, R.; Kuenzli, N.; Lurmann, F.; Rappaport, E.; et al. The Effect of Air Pollution on Lung Development from 10 to 18 Years of Age. N. Engl. J. Med. 2004, 351, 1057–1067. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.D.; Marty, M.A. Impact of Environmental Chemicals on Lung Development. Environ. Health Perspect. 2010, 118, 1155–1164. [Google Scholar] [CrossRef] [PubMed]
- Gauderman, W.J.; Urman, R.; Avol, E.; Berhane, K.; McConnell, R.; Rappaport, E.; Chang, R.; Lurmann, F.; Gilliland, F. Association of Improved Air Quality with Lung Development in Children. N. Engl. J. Med. 2015, 372, 905–913. [Google Scholar] [CrossRef] [PubMed]
- La Grutta, S.; Indinnimeo, L.; Di Coste, A.; Ferrante, G.; Landi, M.; Pelosi, U.; Rusconi, F. Environmental risk factors and lung diseases in children: From guidelines to health effects. Early Hum. Dev. 2013, 89 (Suppl. S3), S59–S62. [Google Scholar] [CrossRef] [PubMed]
- Battistini, A.; Marvasi, R. Inquinamento extradomiciliare e malattie respiratorie del bambino—Air pollution and lung diseases. Med. Bambino 2005, 24, 240–245. [Google Scholar]
- Tulic, M.K.; Christodoulopoulos, P.; Hamid, Q. Small airway inflammation in asthma. Respir. Res. 2001, 2, 333. [Google Scholar] [CrossRef] [PubMed]
- Brain, J.D.; Valberg, P.A. Deposition of aerosol in the respiratory tract. Am. Rev. Respir. Dis. 1979, 120, 1325–1373. [Google Scholar] [PubMed]
- Anderson, J.O.; Thundiyil, J.G.; Stolbach, A. Clearing the Air: A Review of the Effects of Particulate Matter Air Pollution on Human Health. J. Med. Toxicol. 2012, 8, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Risom, L.; Møller, P.; Loft, S. Oxidative stress-induced DNA damage by particulate air pollution. Mutat. Res. 2005, 592, 119–137. [Google Scholar] [CrossRef] [PubMed]
- Longhin, E.; Holme, J.A.; Gutzkow, K.B.; Arlt, V.M.; Kucab, J.E.; Camatini, M.; Gualtieri, M. Cell cycle alterations induced by urban PM2.5 in bronchial epithelial cells: Characterization of the process and possible mechanisms involved. Part Fibre Toxicol. 2013, 10, 63. [Google Scholar] [CrossRef] [PubMed]
- International Classification of Diseases (ICD) ICD-10 Version. 2016. Available online: http://apps.who.int/classifications/icd10/browse/2016/en#/J84.9 (accessed on 16 February 2017).
- Landrigan, P.J.; Etzel, R.A. Textbook of Children’s Environmental Health; Oxford University Press: Oxford, UK, 2013; pp. 1–608. ISBN 9780199929573. [Google Scholar]
- Scarlett, J.F.; Abbott, K.J.; Peacock, J.L.; Strachan, D.P.; Anderson, H.R. Acute effects of summer air pollution on respiratory function in primary school children in southern England. Thorax 1996, 51, 1109–1114. [Google Scholar] [CrossRef] [PubMed]
- Neuberger, M.; Moshammer, H. Schwebstaub und lungengesundheit—Suspended particulates and lung health (In German). Wien Klin Wochenschr 2004, 116 (Suppl. S1), 8–12. [Google Scholar] [PubMed]
- Watanabe, M.; Noma, H.; Kurai, J.; Sano, H.; Hantan, D.; Ueki, M.; Kitano, H.; Shimizu, E. Effects of Short-Term Exposure to Particulate Air Pollutants on the Inflammatory Response and Respiratory Symptoms: A Panel Study in Schoolchildren from Rural Areas of Japan. Int. J. Environ. Res. Public Health 2016, 13, 983. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Effects of Air Pollution on Children’s Health and Development—A Review of the Evidence; WHO Regional Office for Europe: Copenhagen, Demark, 2005; pp. 1–193. [Google Scholar]
- He, Q.Q.; Wong, T.W.; Du, L.; Jiang, Z.Q.; Gao, Y.; Qiu, H.; Liu, W.J.; Wu, J.G.; Wong, A.; Yu, T.S. Effects of ambient air pollution on lung function growth in Chinese schoolchildren. Respir. Med. 2010, 104, 1512–1520. [Google Scholar] [CrossRef] [PubMed]
- Exposure to Air Pollution (Particulate Matter) in Outdoor Air Copenhagen. Available online: http://www.who.int/ipcs/features/air_pollution.pdf (accessed on 24 April 2017).
- Oravisjärvi, K.; Rautio, A.; Ruuskanen, J.; Tiittanen, P.; Timonen, K.L. Air pollution and PEF measurements of children in the vicinity of a steel works. Boreal Environ. Res. 2008, 13, 93–102. [Google Scholar]
- Rice, M.B.; Rifas-Shiman, S.L.; Litonjua, A.A.; Oken, E.; Gillman, M.W.; Kloog, I.; Luttmann-Gibson, H.; Zanobetti, A.; Coull, B.A.; Schwartz, J.; et al. Lifetime Exposure to Ambient Pollution and Lung Function in Children. Am. J. Respir. Crit. Care Med. 2016, 193, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Klepeis, N.E.; Nelson, W.C.; Ott, W.R.; Robinson, J.P.; Tsang, A.M.; Switzer, P.; Behar, J.V.; Hern, S.C.; Engelmann, W.H. The National Human Activity Pattern Survey (NHAPS): A Resource for Assessing Exposure to Environmental Pollutants. J. Exp. Anal. Environ. Epidemol. 2001, 11, 231–252. [Google Scholar] [CrossRef] [PubMed]
- Paulin, L.; Hansel, N. Particulate air pollution and impaired lung function. F1000Res 2016, 5, 201. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Vitale, C.; De Martino, A.; Viegi, G.; Lanza, M.; Molino, A.; Sanduzzi, A.; Vatrella, A.; Annesi-Maesano, I.; D’Amato, M. Effects on asthma and respiratory allergy of Climate change and air pollution. Multidiscip. Respir. Med. 2015, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Nagar, J.K.; Kumar, H.; Kushwah, A.S.; Meena, M.; Kumar, P.; Raj, N.; Singhal, M.K.; Gaur, S.N. Association of indoor and outdoor air pollutant level with respiratory problems among children in an industrial area of Delhi, India. Arch. Environ. Occup. Health 2007, 62, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Siddique, S.; Banerjee, M.; Ray, M.R.; Lahiri, T. Effect of Air Pollution on Incidence of Asthma: A Case Study in Children. Environ. Int. J. Sci. Technol. 2010, 5, 163–175. [Google Scholar]
- Barakat-Haddad, C.; Elliott, S.J.; Pengelly, D. Does Chronic Exposure to Air Pollution in Childhood Impact Long-Term Respiratory Health? Prof. Geogr. 2012, 64, 446–463. [Google Scholar] [CrossRef]
- Oluwole, O.; Ana, G.R.; Arinola, G.O.; Wiskel, T.; Falusi, A.G.; Huo, D.; Olopade, O.I.; Olopade, C.O. Effect of stove intervention on household air pollution and the respiratory health of women and children in rural Nigeria. Air Qual. Atmos. Health 2013, 6, 553. [Google Scholar] [CrossRef]
- Nunes, R.A.; Branco, P.T.; Alvim-Ferraz, M.C.; Martins, F.G.; Sousa, S.I. Particulate matter in rural and urban nursery schools in Portugal. Environ. Pollut. 2015, 202, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, L.A.; Rey, J.J.; Herrera, A.B.; Castro, H.; Niederbacher, J.; Vera, L.M.; Cala, L.L.; Bolívar, F. Respiratory symptoms associated with asthma prevalence and air pollution in preschool children. Biomedica 2010, 30, 15–22. [Google Scholar] [PubMed]
- Nandasena, S.; Wickremasinghe, A.R.; Sathiakumar, N. Respiratory health status of children from two different air pollution exposure settings of Sri Lanka: A cross-sectional study. Am. J. Ind. Med. 2012, 55, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, M.; Giorgis-Allemand, L.; Bernard, C.; Aguilera, I.; Andersen, A.M.; Ballester, F.; Beelen, R.M.; Chatzi, L.; Cirach, M.; Danileviciute, A.; et al. Ambient air pollution and low birthweight: A European cohort study (ESCAPE). Lancet Respir. Med. 2013, 1, 695–704. [Google Scholar] [CrossRef]
- Gehring, U.; Wijga, A.H.; Brauer, M.; Fischer, P.; de Jongste, J.C.; Kerkhof, M.; Oldenwening, M.; Smit, H.A.; Brunekreef, B. Traffic-related air pollution and the development of asthma and allergies during the first 8 years of life. Am. J. Respir. Crit. Care Med. 2010, 181, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Butz, A.M.; Breysse, P.; Rand, C.; Curtin-Brosnan, J.; Eggleston, P.; Diette, G.B.; Williams, D.; Bernert, J.T.; Matsui, E.C. Household smoking behavior: Effects on indoor air quality and health of urban children with asthma. Matern. Child Health J. 2011, 15, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Butz, A.M.; Matsui, E.C.; Breysse, P.; Curtin-Brosnan, J.; Eggleston, P.; Diette, G.; Williams, D.; Yuan, J.; Bernert, J.T.; Rand, C. A randomized trial of air cleaners and a health coach to improve indoor air quality for inner-city children with asthma and second-hand smoke exposure. Arch. Pediatr. Adolesc. Med. 2011, 165, 741–748. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.L.; Baumann, L.M.; Romero, K.; Combe, J.M.; Gomez, A.; Gilman, R.H.; Cabrera, L.; Gonzalvez, G.; Hansel, N.N.; Wise, R.A.; et al. Effect of urbanisation on asthma, allergy and airways inflammation in a developing country setting. Thorax 2011, 66, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Lewin, A.; Buteau, S.; Brand, A.; Kosatsky, T.; Smargiassi, A. Short-term risk of hospitalization for asthma or bronchiolitis in children living near an aluminum smelter. J. Exp. Sci. Environ. Epidemiol. 2013, 23, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Madureira, J.; Paciencia, I.; Ramos, E.; Barros, H.; Pereira, C.; Teixeira, J.P.; Fernandes, E.O. Children’s Health and indoor air quality in Primary schools and homes in Portugal—Study design. J. Toxicol. Environ. Health A 2015, 78, 915–930. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Kwon, H.J.; Lim, J.A.; Choi, J.H.; Ha, M.; Hwang, S.S.; Choi, W.J. Short-term Effect of Fine Particulate Matter on Children’s Hospital Admissions and Emergency Department Visits for Asthma: A Systematic Review and Meta-analysis. J. Prev. Med. Public Health 2016, 49, 205–219. [Google Scholar] [CrossRef] [PubMed]
- Siddique, S.; Ray, M.R.; Lahiri, T. Effects of air pollution on the respiratory health of children: A study in the capital city of India. Air Qual. Atmos. Health 2011, 4, 95–102. [Google Scholar] [CrossRef]
- Zora, J.E.; Sarnat, S.E.; Raysoni, A.U.; Johnson, B.A.; Li, W.W.; Greenwald, R.; Holguin, F.; Stock, T.H.; Sarnat, J.A. Associations between urban air pollution and pediatric asthma control in El Paso, Texas. Sci. Total Environ. 2013, 448, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, G.; Marks, G.B.; Morawska, L. Health effects of daily airborne particle dose in children: Direct association between personal dose and respiratory health effects. Environ. Pollut. 2013, 180, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.; Cardoso, S.M. Effects of indoor air quality on respiratory function of children in the 1st cycle of basic education of Coimbra, Portugal. Occup. Saf. Hyg. II 2014, 2014, 347–350. [Google Scholar]
- Ramesh, B.Y.; Manjunath, N.; Sanjay, D.; Dhanya, Y. Association of indoor air pollution with acute lower respiratory tract infections in children under 5 years of age. Paediatr. Int. Child Health 2012, 32, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Sram, R.J.; Binkova, B.; Dostal, M.; Merkerova-Dostalova, M.; Libalova, H.; Milcova, A.; Rossner, P., Jr.; Rossnerova, A.; Schmuczerova, J.; Svecova, V.; et al. Health impact of air pollution to children. Int. J. Hyg. Environ. Health 2013, 216, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Marchettia, P.; Marcona, A.; Pesce, G.; Paolo, G.; Guarda, L.; Pironi, V.; Fracasso, M.E.; Ricci, P.; de Marco, R. Children living near chipboard and wood industries are at an increased risk of hospitalization for respiratory diseases: A prospective study. Int. J. Hyg. Environ. Health 2014, 217, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, F.; Zhang, L.; Zhao, Z.; Norback, D. A longitudinal study of sick building syndrome (SBS) among pupils in relation to SO2, NO2, O3 and PM10 in schools in China. PLoS ONE 2014, 9, e112933. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, M.; Bravo, M.A.; Jin, L.; Nori-Sarma, A.; Xu, Y.; Guan, D.; Wang, C.; Chen, M.; Wang, X.; et al. Air quality in Lanzhou, a major industrial city in China: Characteristics of air pollution and review of existing evidence from air pollution and health studies. Water Air Soil Pollut. 2014, 225, 2187. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Nagar, J.K.; Kumar, H.; Kushwah, A.S.; Meena, M.; Kumar, P.; Raj, N.; Singhal, M.K.; Gaur, S.N. Indoor air pollution and respiratory function of children in Ashok Vihar, Delhi: An exposure-response study. Asia Pac. J. Public Health 2008, 20, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Neuberger, M.; Moshammer, H.; Schietz, A. Exposure to ultrafine particles in hospitality venues with partial smoking bans. J. Exp. Sci. Environ. Epidemiol. 2013, 23, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Mahr, T.; Makka, J.; Spahn, J. Inflammometry in pediatric asthma: A review of fractional exhaled nitric oxide in clinical practice. Allergy Asthma Proc. 2013, 3, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Poulose, V. Usefulness of FeNO in the Diagnosis of Chronic Cough. Chest 2014, 145 (Suppl. S3), 567A. [Google Scholar] [CrossRef]
- Effects of Air Pollution on Children’s Health and Development—A Review of the Evidence. Available online: http://www.euro.who.int/__data/assets/pdf_file/0010/74728/E86575.pdf (accessed on 16 February 2017).
- Lakhani, N.; North, M.; Ellis, A.K. Clinical Manifestations of Allergic Rhinitis. J. Allergy Ther. 2012, S5, 7. [Google Scholar] [CrossRef]
- Spann, K.; Snape, N.; Baturcam, E.; Fantino, E. The Impact of Early-Life Exposure to Airborne Environmental Insults on the Function of the Airway Epithelium in Asthma. Ann. Glob. Health 2016, 82, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Baraldi, E.; Carraro, S.; Filippone, M. Bronchopulmonary dysplasia: Definitions and long-term respiratory outcome. Early Hum. Dev. 2009, 85 (Suppl. S10), S1–S3. [Google Scholar] [CrossRef] [PubMed]
- Harrison, R.M.; Yin, J. Particulate Matter in the Atmosphere: Which Particle Properties are Important for its Effects on Health? Sci. Total Environ. 2000, 249, 85–101. [Google Scholar] [CrossRef]
- Valet, R.S.; Gebretsadik, T.; Carroll, K.N.; Wu, P.; Dupont, W.D.; Mitchel, E.F.; Hartert, T.V. High asthma prevalence and increased morbidity among rural children in a Medicaid cohort. Ann. Allergy Asthma Immunol. 2011, 106, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Gehring, U.; Gruzieva, O.; Agius, R.M.; Beelen, R.; Custovic, A.; Cyrys, J.; Eeftens, M.; Flexeder, C.; Fuertes, E.; Heinrich, J.; et al. Air pollution exposure and lung function in children: The ESCAPE project. Environ. Health Perspect. 2013, 121, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Mölter, A.; Simpson, A.; Berdel, D.; Brunekreef, B.; Custovic, A.; Cyrys, J.; de Jongste, J.; de Vocht, F.; Fuertes, E.; Gehring, U.; et al. A multicentre study of air pollution exposure and childhood asthma prevalence: The ESCAPE project. Eur. Respir. J. 2015, 45, 610–624. [Google Scholar] [CrossRef] [PubMed]
| Guideline Values PM2.5 | Guideline Values PM10 |
|---|---|
| 10 µg/m3 (annual mean) | 20 µg/m3 (annual mean) |
| 25 µg/m3 (24-h mean) | 50 µg/m3 (24-h mean) |
| Health Effects | Author | Country | Location of Study | Number of Children |
|---|---|---|---|---|
| Respiratory symptoms and inflammatory response (shortness of breath, wet and dry cough, wheezing, chest tightness, throat congestion) | Kumar et al. (2007) [84] | India | Indoor & outdoor | 394 |
| Siddique et al. (2010) [85] | India | Outdoor | 16,164 | |
| Barakat-Haddad et al. (2012) [86] | Canada | Outdoor | 395 | |
| Oluwole et al. (2013) [87] | Nigeria | Indoor | 59 | |
| Nunes et al. (2015) [88] | Portugal | Indoor & outdoor | N/A | |
| Watanabe et al. (2016) [75] | Japan | Indoor & outdoor | 360 | |
| Wheezing and poor respiratory health status | Rodriguez et al. (2010) [89] | Colombia | Indoor & outdoor | 768 |
| Nandasena et al. (2012) [90] | Sri Lanka | Indoor &outdoor | N/A | |
| Pedersen et al. (2013) [91] | Europe | Outdoor | N/A | |
| Asthma, Asthma and bronchiolitis, atopy and airways inflammation, Hospital admissions and emergency department visits for asthma | Gehring et al. (2010) [92] | Germany | Outdoor | 3863 |
| Butz et al. (2011a) [93] | USA | Indoor | 126 | |
| Butz et al. (2011b) [94] | USA | Indoor | 41 | |
| Robinson et al. (2011) [95] | Peru | Indoor | 1441 | |
| Lewin et al. (2013) [96] | Canada | Outdoor | N/A | |
| Madureira et al. (2015) [97] | Portugal | Indoor | 1600 | |
| Lim et al. (2016) [98] | Global | Indoor & outdoor | N/A | |
| Deterioration and changes of lung function | He et al. (2010) [77] | China | Outdoor | 983 |
| Siddique et al. (2011) [99] | India | Indoor & outdoor | 5671 | |
| Zora et al. (2013) [100] | USA | Outdoor | N/A | |
| Buonanno et al. (2013) [101] | Italy | Indoor & outdoor | 103 | |
| Ferreira & Cardoso (2014) [102] | Portugal | Indoor & outdoor | 1019 | |
| Paulin & Hansel (2016) [82] | Global | Indoor & outdoor | N/A | |
| Rice et al. (2016) [80] | USA | Indoor & outdoor | 614 | |
| Acute lower respiratory tract infections | Ramesh et al. (2012) [103] | India | Indoor | 202 |
| Sram et al. (2013) [104] | Czech Republic | Outdoor | 1492 | |
| Increasing risk of hospitalization for all diagnoses | Marchettia et al. (2014) [105] | Italy | Outdoor | 3854 |
| Onset of skin, mucosal and general symptoms | Zhang et al. (2014a) [106] | China | Indoor | 2134 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, H.-Y.; Dunea, D.; Iordache, S.; Pohoata, A. A Review of Airborne Particulate Matter Effects on Young Children’s Respiratory Symptoms and Diseases. Atmosphere 2018, 9, 150. https://doi.org/10.3390/atmos9040150
Liu H-Y, Dunea D, Iordache S, Pohoata A. A Review of Airborne Particulate Matter Effects on Young Children’s Respiratory Symptoms and Diseases. Atmosphere. 2018; 9(4):150. https://doi.org/10.3390/atmos9040150
Chicago/Turabian StyleLiu, Hai-Ying, Daniel Dunea, Stefania Iordache, and Alin Pohoata. 2018. "A Review of Airborne Particulate Matter Effects on Young Children’s Respiratory Symptoms and Diseases" Atmosphere 9, no. 4: 150. https://doi.org/10.3390/atmos9040150
APA StyleLiu, H.-Y., Dunea, D., Iordache, S., & Pohoata, A. (2018). A Review of Airborne Particulate Matter Effects on Young Children’s Respiratory Symptoms and Diseases. Atmosphere, 9(4), 150. https://doi.org/10.3390/atmos9040150






