Chemical Composition and Source Apportionment of Total Suspended Particulate in the Central Himalayan Region
Abstract
:1. Introduction
2. Study Area and Measurements
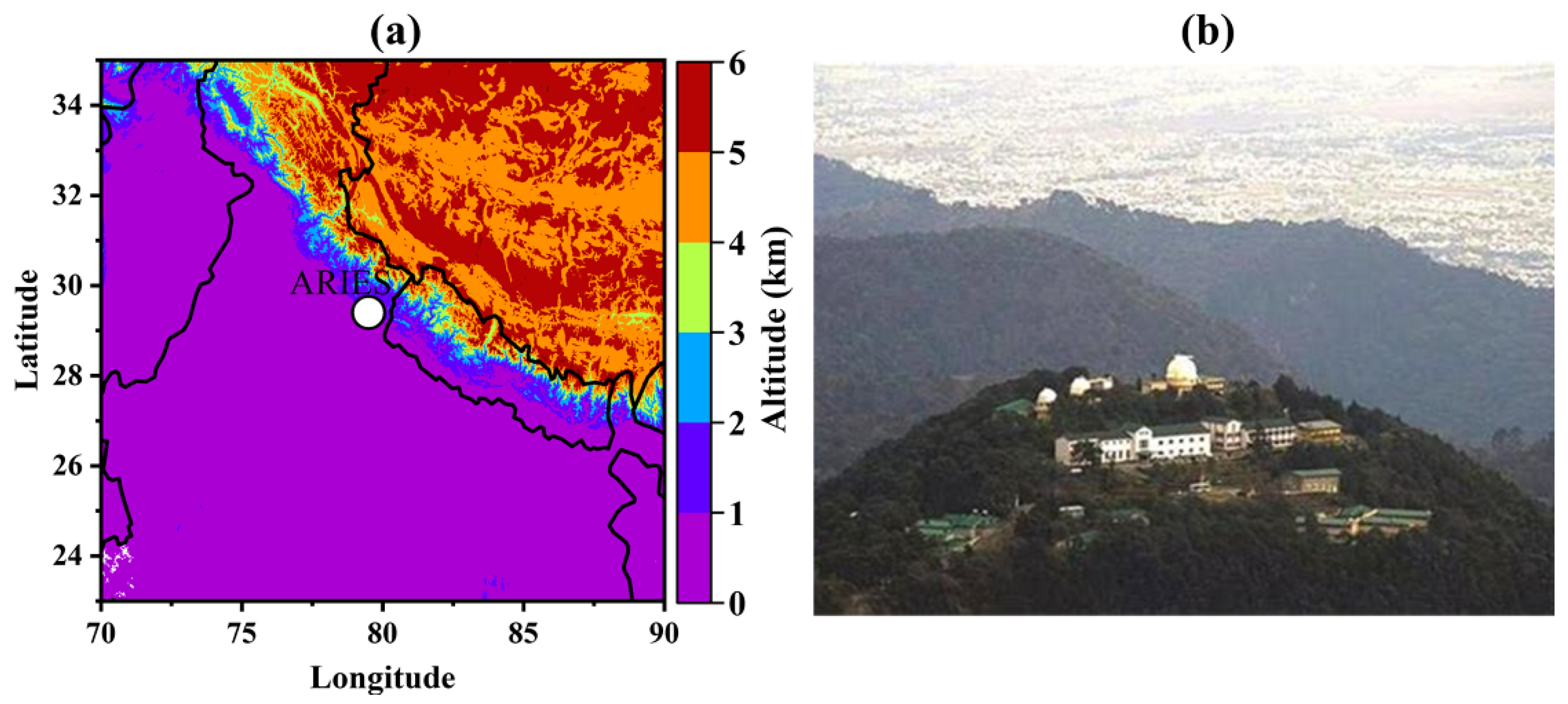
2.1. Site Description
2.2. Aerosol Sampling and Chemical Analysis
3. Methodology
3.1. Estimation of POC and SOC
3.2. Ion Balance and Neutralization Factors
3.3. Source Apportionment via PMF Receptor Model
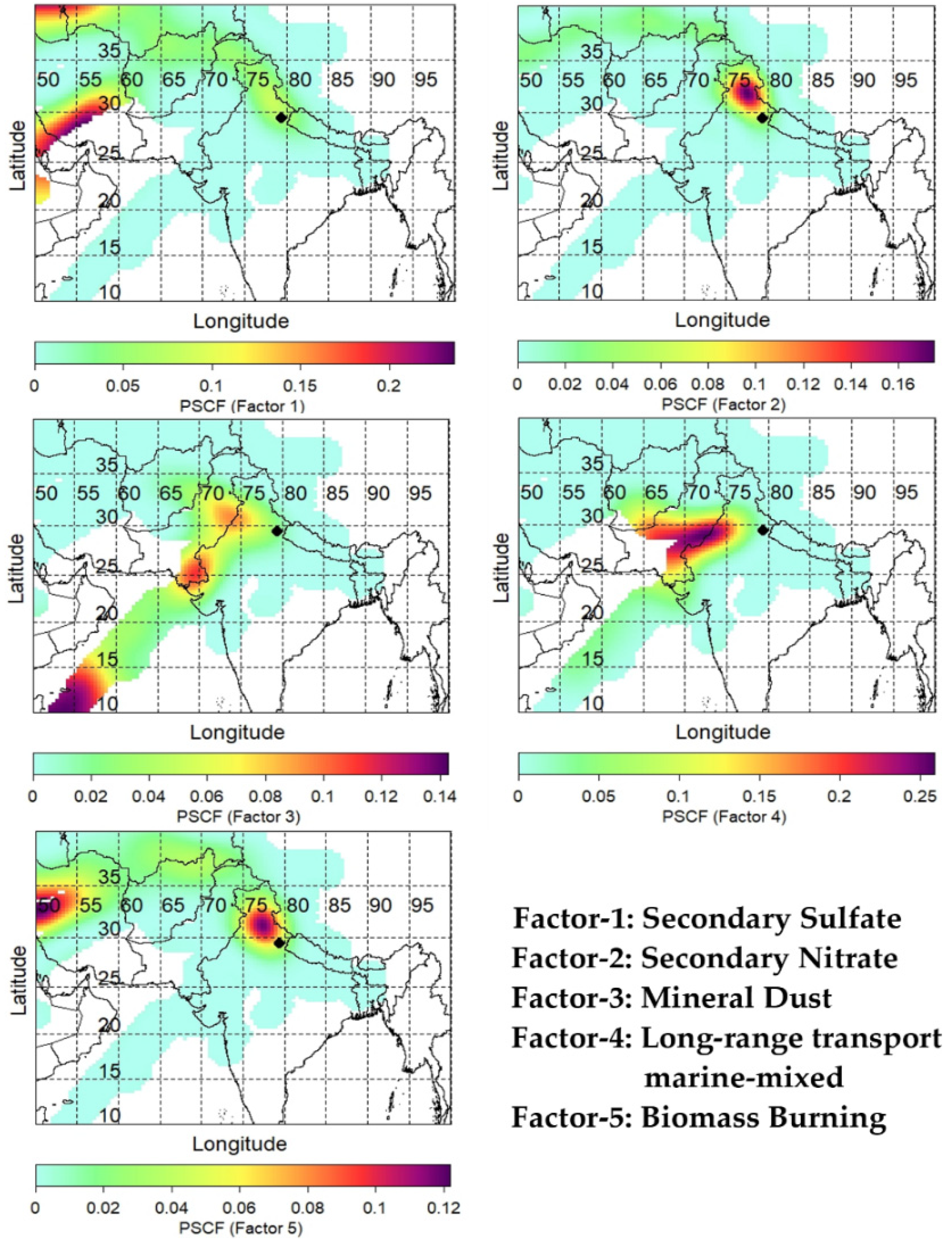
3.4. Air Mass Back-Trajectory and Probability Functions (PSCF and CWT) Analysis
4. Results and Discussion
4.1. TSP Mass and Chemical Composition
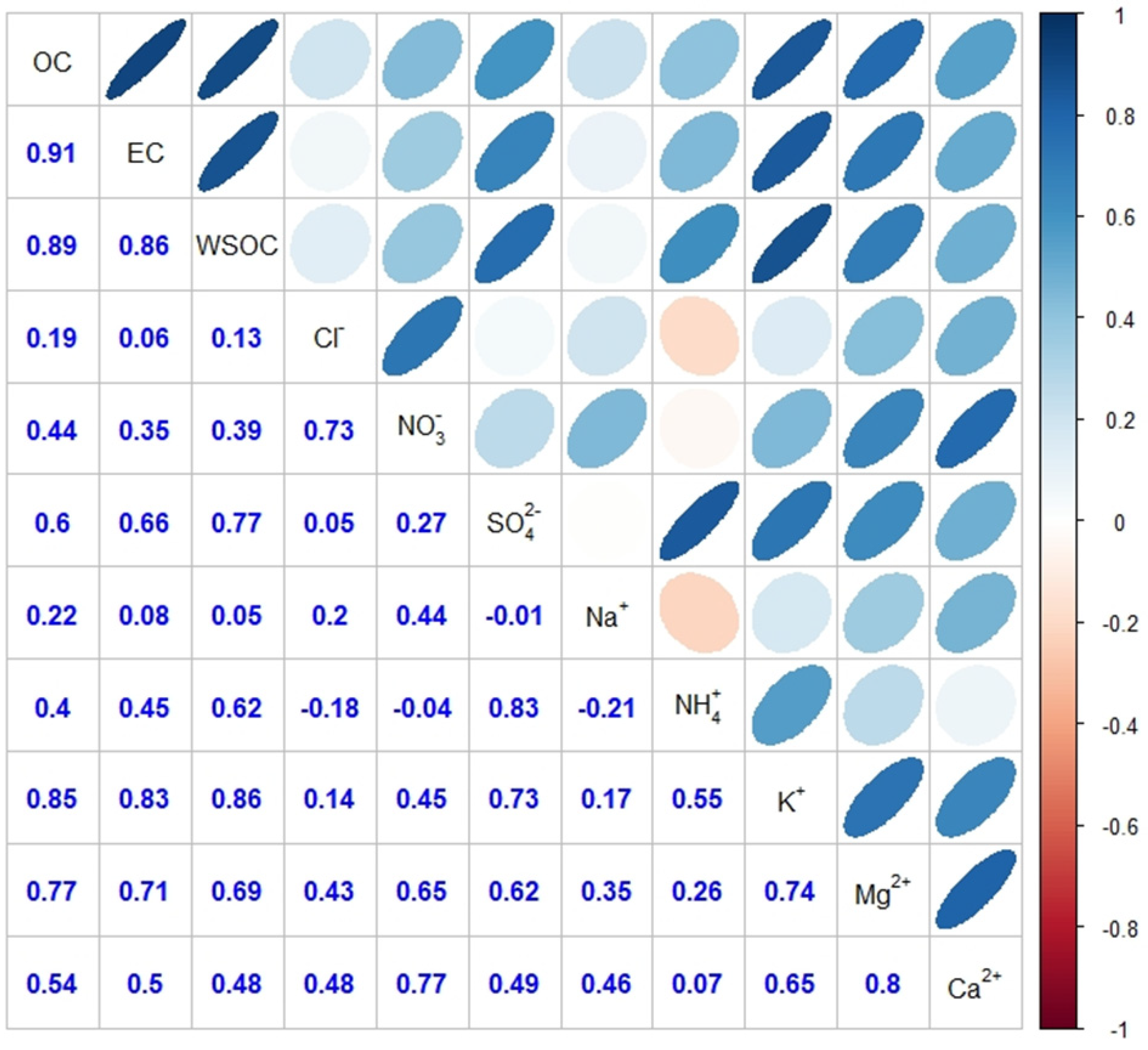
4.2. Ion Chemistry and Neutralization Factor
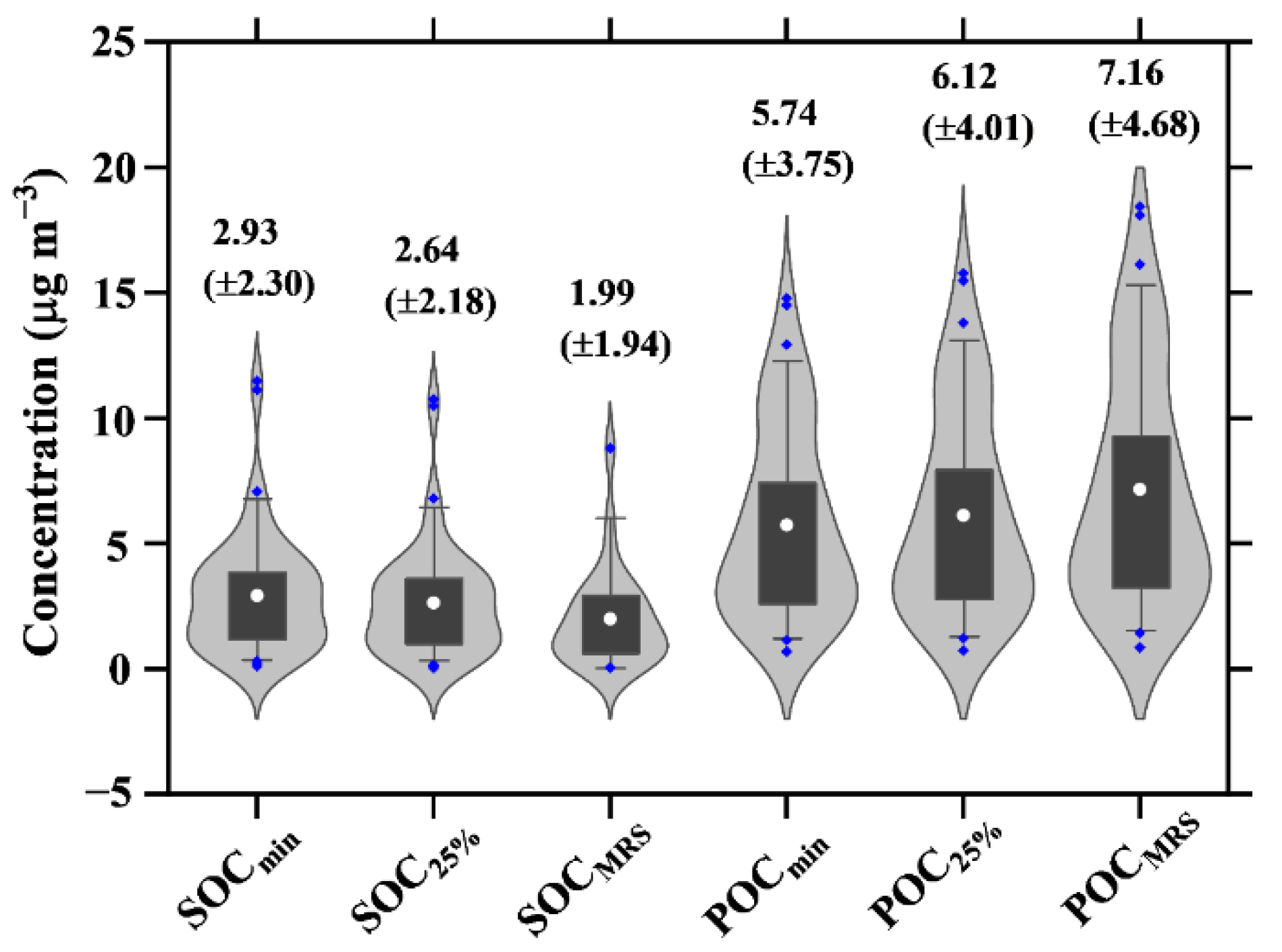
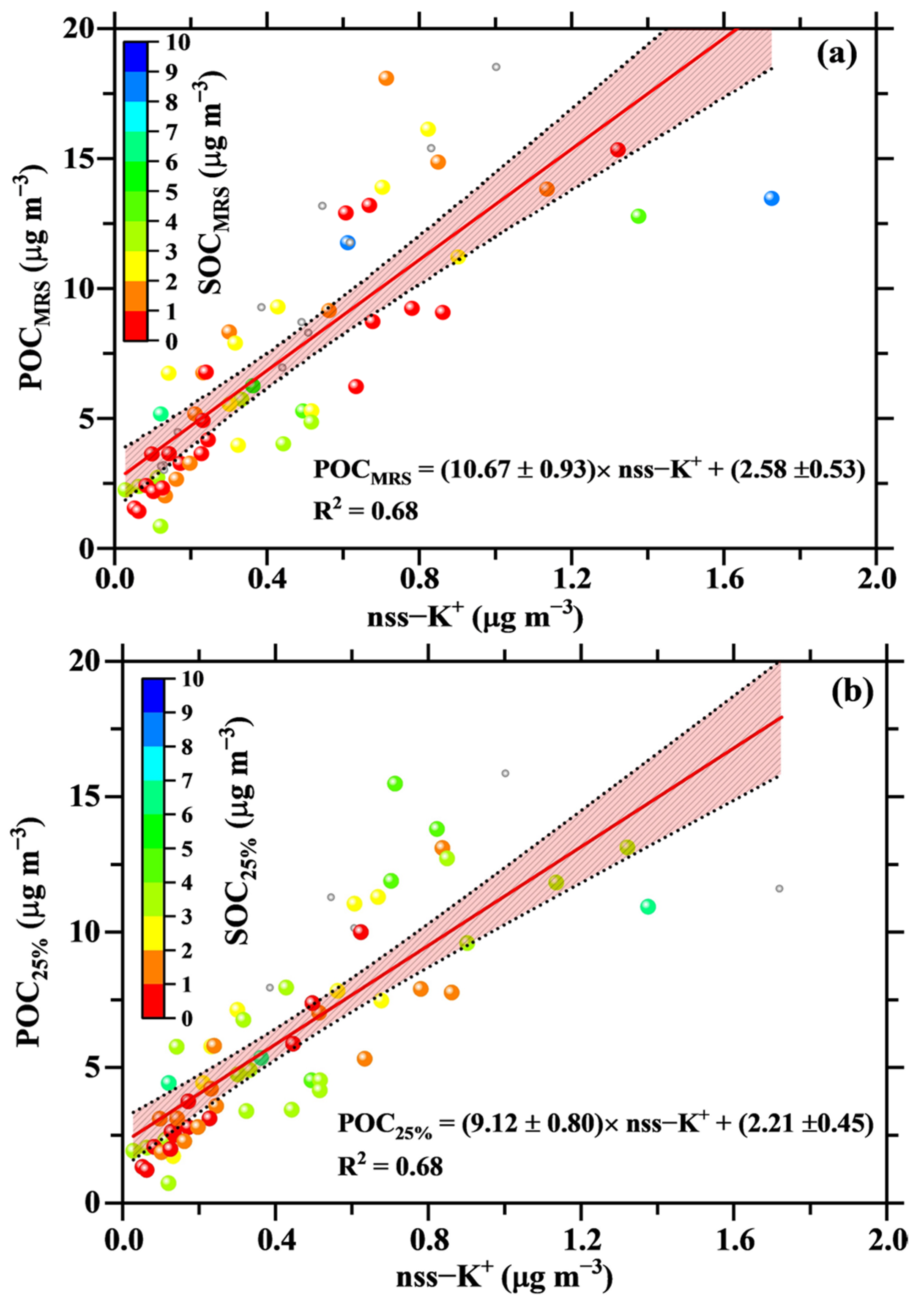
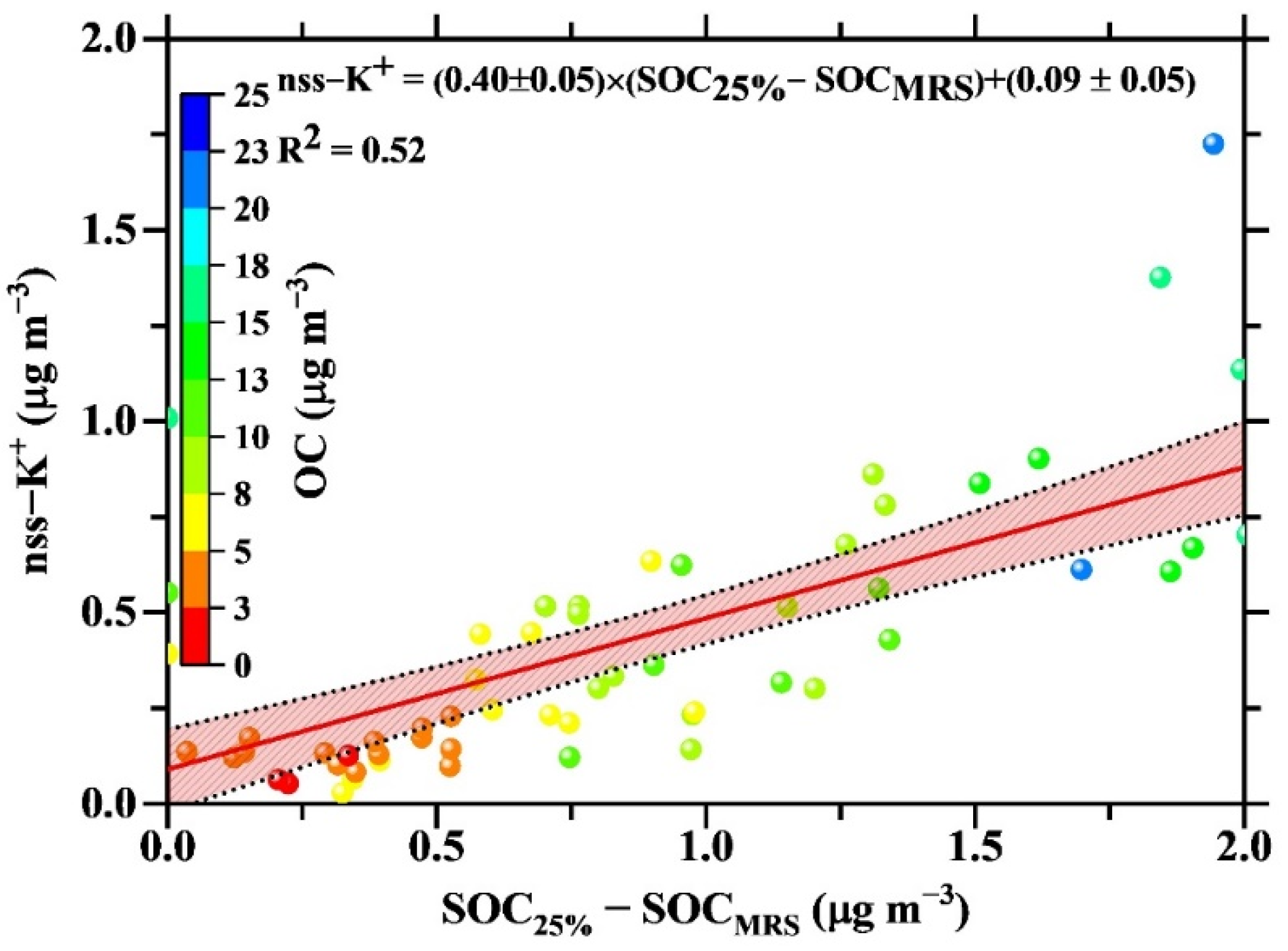
4.3. Estimates of POC and SOC
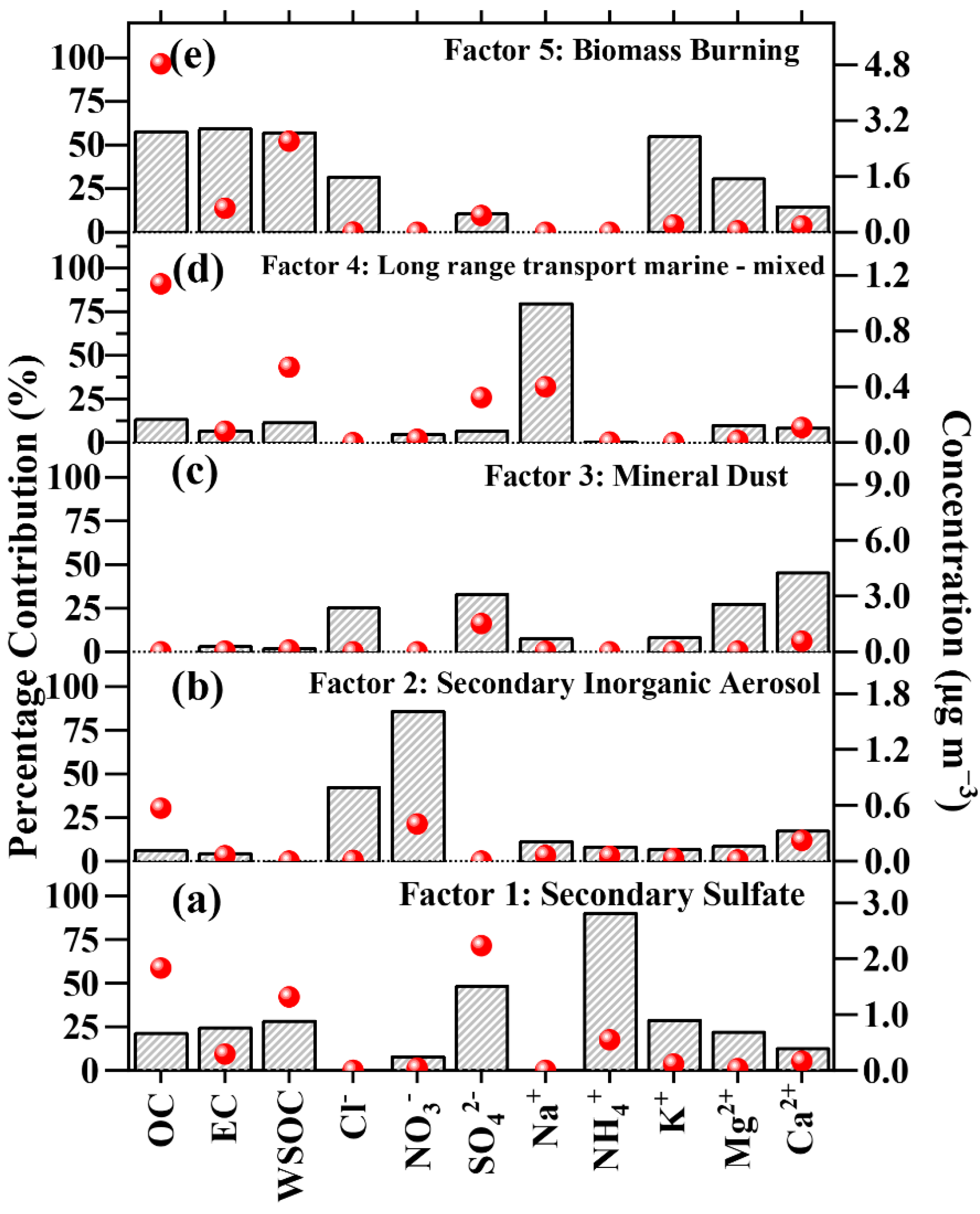
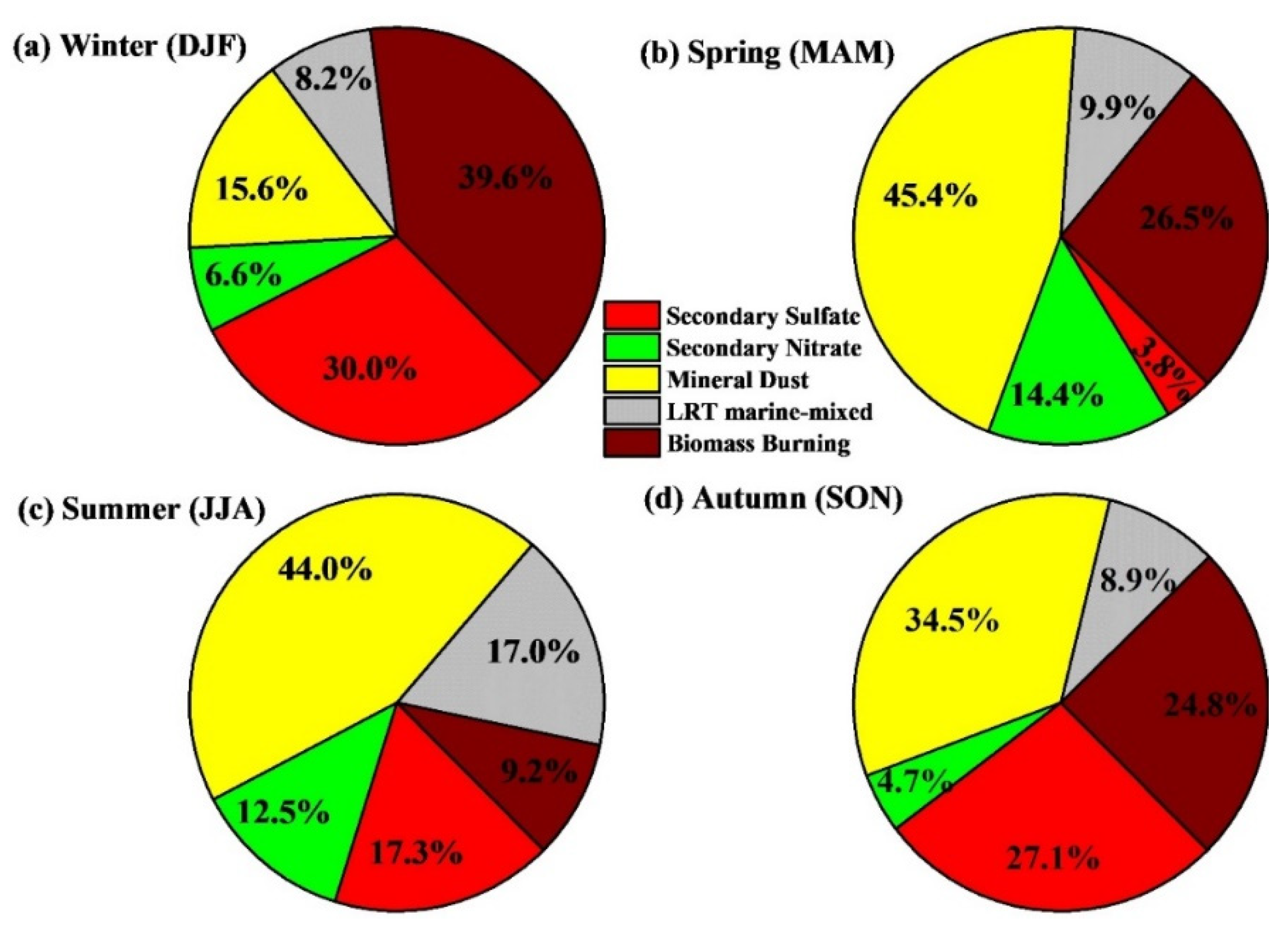
4.4. Source Identification via PMF Receptor Model
5. Summary and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tiwari, S.; Srivastava, A.K.; Chate, D.M.; Safai, P.D.; Bisht, D.S.; Srivastava, M.K.; Beig, G. Impacts of the High Loadings of Primary and Secondary Aerosols on Light Extinction at Delhi during Wintertime. Atmos. Environ. 2014, 92, 60–68. [Google Scholar] [CrossRef]
- Jain, S.; Sharma, S.K.; Choudhary, N.; Masiwal, R.; Saxena, M.; Sharma, A.; Mandal, T.K.; Gupta, A.; Gupta, N.C.; Sharma, C. Chemical Characteristics and Source Apportionment of PM2.5 Using PCA/APCS, UNMIX, and PMF at an Urban Site of Delhi, India. Environ. Sci. Pollut. Res. 2017, 24, 14637–14656. [Google Scholar] [CrossRef]
- Xu, Y.; Wu, X.; Kumar, R.; Barth, M.; Diao, C.; Gao, M.; Lin, L.; Jones, B.; Meehl, G.A. Substantial Increase in the Joint Occurrence and Human Exposure of Heatwave and High-PM Hazards Over South Asia in the Mid-21st Century. AGU Adv. 2020, 1, e2019AV000103. [Google Scholar] [CrossRef] [Green Version]
- Sharma, S.K.; Agarwal, P.; Mandal, T.K.; Karapurkar, S.G.; Shenoy, D.M.; Peshin, S.K.; Gupta, A.; Saxena, M.; Jain, S.; Sharma, A.; et al. Study on Ambient Air Quality of Megacity Delhi, India During Odd–Even Strategy. MAPAN 2017, 32, 155–165. [Google Scholar] [CrossRef]
- Bhowmik, H.S.; Naresh, S.; Bhattu, D.; Rastogi, N.; Prévôt, A.S.H.; Tripathi, S.N. Temporal and Spatial Variability of Carbonaceous Species (EC.; OC.; WSOC and SOA) in PM2.5 Aerosol over Five Sites of Indo-Gangetic Plain. Atmos. Pollut. Res. 2021, 12, 375–390. [Google Scholar] [CrossRef]
- Tiwari, S.; Srivastava, A.K.; Bisht, D.S.; Bano, T.; Singh, S.; Behura, S.; Srivastava, M.K.; Chate, D.M.; Padmanabhamurty, B. Black Carbon and Chemical Characteristics of PM10 and PM2.5 at an Urban Site of North India. J. Atmos. Chem. 2009, 62, 193–209. [Google Scholar] [CrossRef]
- Ram, K.; Sarin, M.M.; Hegde, P. Long-Term Record of Aerosol Optical Properties and Chemical Composition from a High-Altitude Site (Manora Peak) in Central Himalaya. Atmos. Chem. Phys. Discuss. 2010, 10, 7435–7467. [Google Scholar] [CrossRef]
- Ram, K.; Sarin, M.M. Day–Night Variability of EC, OC, WSOC and Inorganic Ions in Urban Environment of Indo-Gangetic Plain: Implications to Secondary Aerosol Formation. Atmos. Environ. 2011, 45, 460–468. [Google Scholar] [CrossRef]
- Hooda, R.K.; Hyvärinen, A.-P.; Vestenius, M.; Gilardoni, S.; Sharma, V.P.; Vignati, E.; Kulmala, M.; Lihavainen, H. Atmospheric aerosols local–regional discrimination for a semi-urban area in India. Atmos. Res. 2016, 168, 13–23. [Google Scholar] [CrossRef]
- Srinivas, B.; Rastogi, N.; Sarin, M.M.; Singh, A.; Singh, D. Mass Absorption Efficiency of Light Absorbing Organic Aerosols from Source Region of Paddy-Residue Burning Emissions in the Indo-Gangetic Plain. Atmos. Environ. 2016, 125, 360–370. [Google Scholar] [CrossRef]
- Rajput, P.; Singh, D.K.; Singh, A.K.; Gupta, T. Chemical Composition and Source-Apportionment of Sub-Micron Particles during Wintertime over Northern India: New Insights on Influence of Fog-Processing. Environ. Pollut. 2018, 233, 81–91. [Google Scholar] [CrossRef]
- Pant, P.; Harrison, R.M. Critical Review of Receptor Modelling for Particulate Matter: A Case Study of India. Atmos. Environ. 2012, 49, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Singh, N.; Murari, V.; Kumar, M.; Barman, S.C.; Banerjee, T. Fine Particulates over South Asia: Review and Meta-Analysis of PM2.5 Source Apportionment through Receptor Model. Environ. Pollut. 2017, 223, 121–136. [Google Scholar] [CrossRef]
- Chatterjee, A.; Mukherjee, S.; Dutta, M.; Ghosh, A.; Ghosh, S.K.; Roy, A. High Rise in Carbonaceous Aerosols under Very Low Anthropogenic Emissions over Eastern Himalaya, India: Impact of Lockdown for COVID-19 Outbreak. Atmos. Environ. 2021, 244, 117947. [Google Scholar] [CrossRef]
- Hopke, P.K.; Dai, Q.; Li, L.; Feng, Y. Global Review of Recent Source Apportionments for Airborne Particulate Matter. Sci. Total Environ. 2020, 740, 140091. [Google Scholar] [CrossRef]
- Bhanuprasad, S.; Venkataraman, C.; Bhushan, M. Positive matrix factorization and trajectory modelling for source identification: A new look at Indian Ocean experiment ship observations. Atmos. Environ. 2008, 42, 4836–4852. [Google Scholar] [CrossRef]
- Paraskevopoulou, D.; Liakakou, E.; Gerasopoulos, E.; Mihalopoulos, N. Sources of Atmospheric Aerosol from Long-Term Measurements (5 years) of Chemical Composition in Athens, Greece. Sci. Total Environ. 2015, 527–528, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Grivas, G.; Cheristanidis, S.; Chaloulakou, A.; Koutrakis, P.; Mihalopoulos, N. Elemental Composition and Source Apportionment of Fine and Coarse Particles at Traffic and Urban Background Locations in Athens, Greece. Aerosol Air Qual. Res. 2018, 18, 1642–1659. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Kim, T.-Y.; Yi, S.-M.; Heo, J. Source Apportionment of PM2.5 Using Positive Matrix Factorization (PMF) at a Rural Site in Korea. J. Environ. Manag. 2018, 214, 325–334. [Google Scholar] [CrossRef]
- Taghvaee, S.; Sowlat, M.H.; Mousavi, A.; Hassanvand, M.S.; Yunesian, M.; Naddafi, K.; Sioutas, C. Source Apportionment of Ambient PM2.5 in Two Locations in Central Tehran Using the Positive Matrix Factorization (PMF) Model. Sci. Total Environ. 2018, 628–629, 672–686. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Sharma, S.K.; Srivastava, M.K.; Chaterjee, A.; Singh, R.K.; Saxena, M.; Mandal, T.K. Source Apportionment of PM10 over Three Tropical Urban Atmospheres at Indo-Gangetic Plain of India: An Approach Using Different Receptor Models. Arch. Environ. Contam. Toxicol. 2019, 76, 114–128. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Sharma, S.K.; Vijayan, N.; Mandal, T.K. Seasonal Characteristics of Aerosols (PM2.5 and PM10) and Their Source Apportionment Using PMF: A four year study over Delhi, India. Environ. Pollut. 2020, 262, 114337. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.K.; Mehrotra, B.J.; Singh, A.; Singh, V.; Bisht, D.S.; Tiwari, S.; Srivastava, M.K. Implications of Different Aerosol Species to Direct Radiative Forcing and Atmospheric Heating Rate. Atmos. Environ. 2020, 241, 117820. [Google Scholar] [CrossRef]
- Singh, A.; Rastogi, N.; Kumar, V.; Slowik, J.G.; Satish, R.; Lalchandani, V.; Thamban, N.M.; Rai, P.; Bhattu, D.; Vats, P.; et al. Sources and Characteristics of Light-Absorbing Fine Particulates over Delhi through the Synergy of Real-Time Optical and Chemical Measurements. Atmos. Environ. 2021, 252, 118338. [Google Scholar] [CrossRef]
- Agarwal, A.; Satsangi, A.; Lakhani, A.; Kumari, K.M. Seasonal and Spatial Variability of Secondary Inorganic Aerosols in PM2.5 at Agra: Source Apportionment through Receptor Models. Chemosphere 2020, 242, 125132. [Google Scholar] [CrossRef]
- Rastogi, N.; Singh, A.; Singh, D.; Sarin, M.M. Chemical Characteristics of PM2.5 at a Source Region of Biomass Burning Emissions: Evidence for Secondary Aerosol Formation. Environ. Pollut. 2014, 184, 563–569. [Google Scholar] [CrossRef]
- Satish, R.; Shamjad, P.; Thamban, N.; Tripathi, S.; Rastogi, N. Temporal Characteristics of Brown Carbon over the Central Indo-Gangetic Plain. Environ. Sci. Technol. 2017, 51, 6765–6772. [Google Scholar] [CrossRef]
- Singh, N.; Banerjee, T.; Raju, M.P.; Deboudt, K.; Sorek-Hamer, M.; Singh, R.S.; Mall, R.K. Aerosol Chemistry, Transport, and Climatic Implications during Extreme Biomass Burning Emissions over the Indo-Gangetic Plain. Atmos. Chem. Phys. 2018, 18, 14197–14215. [Google Scholar] [CrossRef] [Green Version]
- Pratap, V.; Kumar, A.; Tiwari, S.; Kumar, P.; Tripathi, A.K.; Singh, A.K. Chemical Characteristics of Particulate Matters and Their Emission Sources over Varanasi during Winter Season. J. Atmos. Chem. 2020, 77, 83–99. [Google Scholar] [CrossRef]
- Pandey, C.P.; Singh, J.; Soni, V.K.; Singh, N. Yearlong First Measurements of Black Carbon in the Western Indian Himalaya: Influences of Meteorology and Fire Emissions. Atmos. Pollut. Res. 2020, 11, 1199–1210. [Google Scholar] [CrossRef]
- Tripathee, L.; Kang, S.; Chen, P.; Bhattarai, H.; Guo, J.; Shrestha, K.L.; Sharma, C.M.; Sharma Ghimire, P.; Huang, J. Water-Soluble Organic and Inorganic Nitrogen in Ambient Aerosols over the Himalayan Middle Hills: Seasonality, Sources, and Transport Pathways. Atmos. Res. 2021, 250, 105376. [Google Scholar] [CrossRef]
- Bisht, D.S.; Srivastava, A.K.; Joshi, H.; Ram, K.; Singh, N.; Naja, M.; Srivastava, M.K.; Tiwari, S. Chemical Characterization of Rainwater at a High-Altitude Site “Nainital” in the Central Himalayas, India. Environ. Sci. Pollut. Res. 2017, 24, 3959–3969. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Mukherjee, S.; Choudhary, N.; Rai, A.; Ghosh, A.; Chatterjee, A.; Vijayan, N.; Mandal, T.K. Seasonal Variation and Sources of Carbonaceous Species and Elements in PM2.5 and PM10 over the Eastern Himalaya. Environ. Sci. Pollut. Res. 2021. [Google Scholar] [CrossRef]
- Dumka, U.C.; Kaskaoutis, D.G.; Mihalopoulos, N.; Sheoran, R. Identification of Key Aerosol Types and Mixing States in the Central Indian Himalayas during the GVAX Campaign: The Role of Particle Size in Aerosol Classification. Sci. Total Environ. 2021, 761, 143188. [Google Scholar] [CrossRef]
- Srivastava, P.; Naja, M. Characteristics of Carbonaceous Aerosols Derived from Long-Term High-Resolution Measurements at a High-Altitude Site in the Central Himalayas: Radiative Forcing Estimates and Role of Meteorology and Biomass Burning. Environ. Sci. Pollut. Res. 2021, 28, 14654–14670. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, P.; Naja, M.; Seshadri, T.R.; Joshi, H.; Dumka, U.C.; Gogoi, M.M.; Babu, S.S. Implications of Site-specific Mass Absorption Cross-section (MAC) to Black Carbon Observations at a High-altitude Site in the Central Himalaya. Asia Pac. J. Atmos. Sci. 2021. [Google Scholar] [CrossRef]
- Dumka, U.C.; Kaskaoutis, D.G.; Srivastava, M.K.; Devara, P.C.S. Scattering and Absorption Properties of Near-Surface Aerosol over Gangetic–Himalayan Region: The Role of Boundary-Layer Dynamics and Long-Range Transport. Atmos. Chem. Phys. 2015, 15, 1555–1572. [Google Scholar] [CrossRef] [Green Version]
- Sagar, R.; Dumka, U.C.; Naja, M.; Singh, N.; Phanikumar, D.V. 2015 ARIES, Nainital: A strategically important location for climate change studies in the Central Gangetic Himalayan region. Curr. Sci. 2015, 109, 703–715. [Google Scholar]
- Bhardwaj, P.; Naja, M.; Kumar, R.; Chandola, H.C. Seasonal, Inter annual, and Long-Term Variabilities in Biomass Burning Activity over South Asia. Environ. Sci. Pollut. Res. 2016, 23, 4397–4410. [Google Scholar] [CrossRef]
- Choudhary, V.; Singh, G.K.; Gupta, T.; Paul, D. Absorption and Radiative Characteristics of Brown Carbon Aerosols during Crop Residue Burning in the Source Region of Indo-Gangetic Plain. Atmos. Res. 2021, 249, 105285. [Google Scholar] [CrossRef]
- Rana, A.; Jia, S.; Sarkar, S. Black Carbon Aerosol in India: A Comprehensive Review of Current Status and Future Prospects. Atmos. Res. 2019, 218, 207–230. [Google Scholar] [CrossRef]
- Dumka, U.C.; Kaskaoutis, D.G.; Francis, D.; Chaboureau, J.-P.; Rashki, A.; Tiwari, S.; Singh, S.; Liakakou, E.; Mihalopoulos, N. The Role of the Intertropical Discontinuity Region and the Heat Low in Dust Emission and Transport over the Thar Desert, India: A Premonsoon Case Study. J. Geophys. Res. Atmos. 2019, 124, 13197–13219. [Google Scholar] [CrossRef]
- Raatikainen, T.; Hyvärinen, A.-P.; Hatakka, J.; Panwar, T.S.; Hooda, R.K.; Sharma, V.P.; Lihavainen, H. The Effect of Boundary Layer Dynamics on Aerosol Properties at the Indo-Gangetic Plains and at the Foothills of the Himalayas. Atmos. Environ. 2014, 89, 548–555. [Google Scholar] [CrossRef]
- Hooda, R.K.; Kivekäs, N.; O’Connor, E.J.; Collaud Coen, M.; Pietikäinen, J.-P.; Vakkari, V.; Backman, J.; Henriksson, S.V.; Asmi, E.; Komppula, M.; et al. Driving Factors of Aerosol Properties over the Foothills of Central Himalayas Based on 8.5 Years Continuous Measurements. J. Geophys. Res. Atmos. 2018, 123, 13421–13442. [Google Scholar] [CrossRef]
- Dumka, U.C.; Ningombam, S.S.; Kaskaoutis, D.G.; Madhavan, B.L.; Song, H.-J.; Angchuk, D.; Jorphail, S. Long-Term (2008–2018) Aerosol Properties and Radiative Effect at High-Altitude Sites over Western Trans-Himalayas. Sci. Total Environ. 2020, 734, 139354. [Google Scholar] [CrossRef] [PubMed]
- Rengarajan, R.; Sarin, M.M.; Sudheer, A.K. Carbonaceous and Inorganic Species in Atmospheric Aerosols during wintertime over Urban and High-Altitude Sites in North India. J. Geophys. Res. 2007, 112, D21307. [Google Scholar] [CrossRef]
- Birch, M.E.; Cary, R.A. Elemental Carbon-Based Method for Monitoring Occupational Exposures to Particulate Diesel Exhaust. Aerosol Sci. Technol. 1996, 25, 221–241. [Google Scholar] [CrossRef]
- Ram, K.; Sarin, M.M.; Hegde, P. Atmospheric Abundances of Primary and Secondary Carbonaceous Species at Two High-Altitude Sites in India: Sources and Temporal Variability. Atmos. Environ. 2008, 42, 6785–6796. [Google Scholar] [CrossRef]
- Turpin, B.J.; Huntzicker, J.J. Identification of Secondary Organic Aerosol Episodes and Quantitation of Primary and Secondary Organic Aerosol Concentrations during SCAQS. Atmos. Environ. 1995, 29, 3527–3544. [Google Scholar] [CrossRef]
- Dumka, U.C.; Tiwari, S.; Kaskaoutis, D.G.; Hopke, P.K.; Singh, J.; Srivastava, A.K.; Bisht, D.S.; Attri, S.D.; Tyagi, S.; Misra, A.; et al. Assessment of PM2.5 Chemical Compositions in Delhi: Primary vs Secondary Emissions and Contribution to Light Extinction Coefficient and Visibility Degradation. J. Atmos. Chem. 2017, 74, 423–450. [Google Scholar] [CrossRef]
- Wu, C.; Wu, D.; Yu, J.Z. Estimation and Uncertainty Analysis of Secondary Organic Carbon Using 1 Year of Hourly Organic and Elemental Carbon Data. J. Geophys. Res. Atmos. 2019, 124, 2774–2795. [Google Scholar] [CrossRef]
- Grivas, G.; Cheristanidis, S.; Chaloulakou, A. Elemental and Organic Carbon in the Urban Environment of Athens. Seasonal and Diurnal Variations and Estimates of Secondary Organic Carbon. Sci. Total Environ. 2012, 414, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Kaskaoutis, D.G.; Grivas, G.; Theodosi, C.; Tsagkaraki, M.; Paraskevopoulou, D.; Stavroulas, I.; Liakakou, E.; Gkikas, A.; Hatzianastassiou, N.; Wu, C.; et al. Carbonaceous Aerosols in Contrasting Atmospheric Environments in Greek Cities: Evaluation of the EC-Tracer Methods for Secondary Organic Carbon Estimation. Atmosphere 2020, 11, 161. [Google Scholar] [CrossRef] [Green Version]
- Satsangi, A.; Pachauri, T.; Singla, V.; Lakhani, A.; Kumari, K.M. Water Soluble Ionic Species in Atmospheric Aerosols: Concentrations and Sources at Agra in the Indo-Gangetic Plain (IGP). Aerosol Air Qual. Res. 2013, 13, 1877–1889. [Google Scholar] [CrossRef]
- Kulshrestha, A.; Satsangi, P.G.; Masih, J.; Taneja, A. Metal Concentration of PM2.5 and PM10 Particles and Seasonal Variations in Urban and Rural Environment of Agra, India. Sci. Total Environ. 2009, 407, 6196–6204. [Google Scholar] [CrossRef]
- Tripathee, L.; Kang, S.; Huang, J.; Sharma, C.M.; Sillanpää, M.; Guo, J.; Paudyal, R. Concentrations of Trace Elements in Wet Deposition over the Central Himalayas, Nepal. Atmos. Environ. 2014, 95, 231–238. [Google Scholar] [CrossRef]
- Tripathee, L.; Kang, S.; Rupakheti, D.; Cong, Z.; Zhang, Q.; Huang, J. Chemical Characteristics of Soluble Aerosols over the Central Himalayas: Insights into Spatiotemporal Variations and Sources. Environ. Sci. Pollut. Res. 2017, 24, 24454–24472. [Google Scholar] [CrossRef]
- Wu, D.; Tie, X.; Deng, X. Chemical Characterizations of Soluble Aerosols in Southern China. Chemosphere 2006, 64, 749–757. [Google Scholar] [CrossRef]
- Jaiprakash; Singhai, A.; Habib, G.; Raman, R.S.; Gupta, T. Chemical Characterization of PM1.0 Aerosol in Delhi and Source Apportionment Using Positive Matrix Factorization. Environ. Sci. Pollut. Res. 2017, 24, 445–462. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. EPA Positive Matrix Factorization (PMF) 5.0 Fundamentals and User Guide. 2014. Available online: http://www.epa.gov/heasd/documents/EPA%20PMF%205.0%20User%20Guide.pd (accessed on 14 June 2021).
- Belis, C.A.; Larsen, B.R.; Amati, F.; Haddad, I.E.; Favez, O.; Harrison, R.M.; Hopke, P.K.; Nava, S.; Paatero, P.; Prévôt, A.; et al. European Guide on Air Pollution Source Apportionment with Receptor Models; Joint Research Centre Institute for Environment and Sustainability: Ispra, Italy, 2014; p. 27. [Google Scholar]
- Zabalza, J.; Ogulei, D.; Hopke, P.K.; Lee, J.H.; Hwang, I.; Querol, X.; Alastuey, A.; Santamaría, J.M. Concentration and Sources of PM10 and Its Constituents in Alsasua, Spain. Water Air Soil Pollut. 2006, 174, 385–404. [Google Scholar] [CrossRef]
- Kara, M.; Hopke, P.K.; Dumanoglu, Y.; Altiok, H.; Elbir, T.; Odabasi, M.; Bayram, A. Characterization of PM Using Multiple Site Data in a Heavily Industrialized Region of Turkey. Aerosol. Air Qual. Res. 2015, 15, 11–27. [Google Scholar] [CrossRef]
- Paatero, P.; Hopke, P.K. Discarding or Downweighting High-Noise Variables in Factor Analytic Models. Anal. Chim. Acta 2003, 490, 277–289. [Google Scholar] [CrossRef]
- Stein, A.F.; Draxler, R.R.; Rolph, G.D.; Stunder, B.J.B.; Cohen, M.D.; Ngan, F. NOAA’s HYSPLIT Atmospheric Transport and Dispersion Modeling System. Bull. Am. Meteorol. Soc. 2015, 96, 2059–2077. [Google Scholar] [CrossRef]
- Rolph, G.; Stein, A.; Stunder, B. Real-Time Environmental Applications and Display SYstem: READY. Environ. Model. Softw. 2017, 95, 210–228. [Google Scholar] [CrossRef]
- Dimitriou, K.; Kassomenos, P. A Meteorological Analysis of PM10 Episodes at a High Altitude City and a Low Altitude City in Central Greece–The Impact of Wood Burning Heating Devices. Atmos. Res. 2018, 214, 329–337. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Zhang, X.Y.; Draxler, R.R. TrajStat: GIS-Based Software That Uses Various Trajectory Statistical Analysis Methods to Identify Potential Sources from Long-Term Air Pollution Measurement Data. Environ. Model. Softw. 2009, 24, 938–939. [Google Scholar] [CrossRef]
- Li, D.; Liu, J.; Zhang, J.; Gui, H.; Du, P.; Yu, T.; Wang, J.; Lu, Y.; Liu, W.; Cheng, Y. Identification of Long-Range Transport Pathways and Potential Sources of PM2.5 and PM10 in Beijing from 2014 to 2015. J. Environ. Sci. 2017, 56, 214–229. [Google Scholar] [CrossRef]
- Rai, A.; Mukherjee, S.; Chatterjee, A.; Choudhary, N.; Kotnala, G.; Mandal, T.K.; Sharma, S.K. Seasonal Variation of OC, EC, and WSOC of PM10 and Their CWT Analysis Over the Eastern Himalaya. Aerosol. Sci. Eng. 2020, 4, 26–40. [Google Scholar] [CrossRef]
- Sandeep, K.; Negi, R.S.; Panicker, A.S.; Gautam, A.S.; Bhist, D.S.; Beig, G.; Murthy, B.S.; Latha, R.; Singh, S.; Das, S. Characteristics and Variability of Carbonaceous Aerosols over a Semi Urban Location in Garhwal Himalayas. Asia Pac. J. Atmos. Sci. 2020, 56, 455–465. [Google Scholar] [CrossRef]
- Rastogi, N.; Singh, A.; Sarin, M.M.; Singh, D. Temporal Variability of Primary and Secondary Aerosols over Northern India: Impact of Biomass Burning Emissions. Atmos. Environ. 2016, 125, 396–403. [Google Scholar] [CrossRef]
- Choudhary, V.; Rajput, P.; Gupta, T. Absorption Properties and Forcing Efficiency of Light-Absorbing Water-Soluble Organic Aerosols: Seasonal and Spatial Variability. Environ. Pollut. 2021, 272, 115932. [Google Scholar] [CrossRef] [PubMed]
- Pio, C.; Cerqueira, M.; Harrison, R.M.; Nunes, T.; Mirante, F.; Alves, C.; Oliveira, C.; Sanchez de la Campa, A.; Artíñano, B.; Matos, M. OC/EC Ratio Observations in Europe: Re-Thinking the Approach for Apportionment between Primary and Secondary Organic Carbon. Atmos. Environ. 2011, 45, 6121–6132. [Google Scholar] [CrossRef]
- Ram, K.; Sarin, M.M. Atmospheric carbonaceous aerosols from Indo-Gangetic Plain and Central Himalaya: Impact of anthropogenic sources. J. Environ. Manag. 2015, 148, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Andreae, M.O.; Merlet, P. Emission of Trace Gases and Aerosols from Biomass Burning. Glob. Biogeochem. Cycles 2001, 15, 955–966. [Google Scholar] [CrossRef] [Green Version]
- Favez, O.; Sciare, J.; Cachier, H.; Alfaro, S.C.; Abdelwahab, M.M. Significant Formation of Water-Insoluble Secondary Organic Aerosols in Semi-Arid Urban Environment. Geophys. Res. Lett. 2008, 35, L15801. [Google Scholar] [CrossRef]
- Hecobian, A.; Zhang, X.; Zheng, M.; Frank, N.; Edgerton, E.S.; Weber, R.J. Water-Soluble Organic Aerosol Material and the Light-Absorption Characteristics of Aqueous Extracts Measured over the Southeastern United States. Atmos. Chem. Phys. 2010, 10, 5965–5977. [Google Scholar] [CrossRef] [Green Version]
- Aswini, A.R.; Hegde, P.; Nair, P.R.; Aryasree, S. Seasonal Changes in Carbonaceous Aerosols over a Tropical Coastal Location in Response to Meteorological Processes. Sci. Total Environ. 2019, 656, 1261–1279. [Google Scholar] [CrossRef] [PubMed]
- Arun, B.S.; Aswini, A.R.; Gogoi, M.M.; Hegde, P.; Kumar Kompalli, S.; Sharma, P.; Suresh Babu, S. Physico-Chemical and Optical Properties of Aerosols at a Background Site (~4 Km a.s.l.) in the Western Himalayas. Atmos. Environ. 2019, 218, 117017. [Google Scholar] [CrossRef]
- Zhang, N.; Cao, J.; Ho, K.; He, Y. Chemical Characterization of Aerosol Collected at Mt. Yulong in Wintertime on the Southeastern Tibetan Plateau. Atmos. Res. 2012, 107, 76–85. [Google Scholar] [CrossRef]
- Sun, J.Y.; Wu, C.; Wu, D.; Cheng, C.; Li, M.; Li, L.; Deng, T.; Yu, J.Z.; Li, Y.J.; Zhou, Q.; et al. Amplification of Black Carbon Light Absorption Induced by Atmospheric Aging: Temporal Variation at Seasonal and Diel Scales in Urban Guangzhou. Atmos. Chem. Phys. 2020, 20, 2445–2470. [Google Scholar] [CrossRef] [Green Version]
- Pio, C.A.; Legrand, M.; Oliveira, T.; Afonso, J.; Santos, C.; Caseiro, A.; Fialho, P.; Barata, F.; Puxbaum, H.; Sanchez-Ochoa, A.; et al. Climatology of Aerosol Composition (Organic versus Inorganic) at Nonurban Sites on a West-East Transect across Europe. J. Geophys. Res. 2007, 112, D23S02. [Google Scholar] [CrossRef] [Green Version]
- Weber, R.J.; Sullivan, A.P.; Peltier, R.E.; Russell, A.; Yan, B.; Zheng, M.; de Gouw, J.; Warneke, C.; Brock, C.; Holloway, J.S.; et al. A Study of Secondary Organic Aerosol Formation in the Anthropogenic-influenced Southeastern United States. J. Geophys. Res. 2007, 112, 2007JD008408. [Google Scholar] [CrossRef]
- Shivani; Gadi, R.; Sharma, S.K.; Mandal, T.K. Seasonal Variation, Source Apportionment and Source Attributed Health Risk of Fine Carbonaceous Aerosols over National Capital Region, India. Chemosphere 2019, 237, 124500. [Google Scholar] [CrossRef]
- Pachauri, T.; Singla, V.; Satsangi, A.; Lakhani, A.; Kumari, K.M. Characterization of Carbonaceous Aerosols with Special Reference to Episodic Events at Agra, India. Atmos. Res. 2013, 128, 98–110. [Google Scholar] [CrossRef]
- Satsangi, A.; Pachauri, T.; Singla, V.; Lakhani, A.; Kumari, K.M. Organic and Elemental Carbon Aerosols at a Suburban Site. Atmos. Res. 2012, 113, 13–21. [Google Scholar] [CrossRef]
- Brown, K.W.; Bouhamra, W.; Lamoureux, D.P.; Evans, J.S.; Koutrakis, P. Characterization of Particulate Matter for Three Sites in Kuwait. J. Air Waste Manag. Assoc. 2008, 58, 994–1003. [Google Scholar] [CrossRef]
- Zhuang, H.; Chan, C.K.; Fang, M.; Wexler, A.S. Formation of Nitrate and Non-Sea-Salt Sulfate on Coarse Particles. Atmos. Environ. 1999, 33, 4223–4233. [Google Scholar] [CrossRef]
- Gupta, T.; Mandariya, A. Sources of Submicron Aerosol during Fog-Dominated Wintertime at Kanpur. Environ. Sci. Pollut. Res. 2013, 20, 5615–5629. [Google Scholar] [CrossRef] [PubMed]
- Sen, A.; Ahammed, Y.N.; Banerjee, T.; Chatterjee, A.; Choudhuri, A.K.; Das, T.; Chandara Deb, N.; Dhir, A.; Goel, S.; Khan, A.H.; et al. Spatial Variability in Ambient Atmospheric Fine and Coarse Mode Aerosols over Indo-Gangetic Plains, India and Adjoining Oceans during the Onset of Summer Monsoons, 2014. Atmos. Pollut. Res. 2016, 7, 521–532. [Google Scholar] [CrossRef]
- Cash, J.M.; Langford, B.; Di Marco, C.; Mullinger, N.J.; Allan, J.; Reyes-Villegas, E.; Joshi, R.; Heal, M.R.; Acton, W.J.F.; Hewitt, C.N.; et al. Seasonal analysis of submicron aerosol in Old Delhi using high-resolution aerosol mass spectrometry: Chemical characterisation, source apportionment and new marker identification. Atmos. Chem. Phys. 2021, 21, 10133–10158. [Google Scholar] [CrossRef]
- Ghosh, A.; Patel, A.; Rastogi, N.; Sharma, S.K.; Mandal, T.K.; Chatterjee, A. Size-segregated aerosols over a high altitude Himalayan and a tropical urban metropolis in Eastern India: Chemical characterization, light absorption, role of meteorology and long range transport. Atmos. Environ. 2021, 254, 118398. [Google Scholar] [CrossRef]
- Kumar, R.; Naja, M.; Satheesh, S.K.; Ojha, N.; Joshi, H.; Sarangi, T.; Pant, P.; Dumka, U.C.; Hegde, P.; Venkataramani, S. Influences of the Springtime Northern Indian Biomass Burning over the Central Himalayas. J. Geophys. Res. 2011, 116, D19302. [Google Scholar] [CrossRef]
- Vadrevu, K.P.; Lasko, K.; Giglio, L.; Justice, C. Vegetation Fires, Absorbing Aerosols and Smoke Plume Characteristics in Diverse Biomass Burning Regions of Asia. Environ. Res. Lett. 2015, 10, 105003. [Google Scholar] [CrossRef] [Green Version]
- Kaskaoutis, D.G.; Kumar, S.; Sharma, D.; Singh, R.P.; Kharol, S.K.; Sharma, M.; Singh, A.K.; Singh, S.; Singh, A.; Singh, D. Effects of Crop Residue Burning on Aerosol Properties, Plume Characteristics, and Long-Range Transport over Northern India: Effects of Crop Residue Burning. J. Geophys. Res. Atmos. 2014, 119, 5424–5444. [Google Scholar] [CrossRef]
- Sembhi, H.; Wooster, M.; Zhang, T.; Sharma, S.; Singh, N.; Agarwal, S.; Boesch, H.; Gupta, S.; Misra, A.; Tripathi, S.N.; et al. Post-Monsoon Air Quality Degradation across Northern India: Assessing the Impact of Policy-Related Shifts in Timing and Amount of Crop Residue Burnt. Environ. Res. Lett. 2020, 15, 104067. [Google Scholar] [CrossRef]
| Species | AM (µg m−3) | SD (µg m−3) | Min (µg m−3) | Max (µg m−3) | MDL (µg m−3) | No. of BDL Values | No. of Missing Values |
|---|---|---|---|---|---|---|---|
| TSP | 69.58 | 51.79 | 12.67 | 271.69 | -- | -- | -- |
| OC | 8.62 | 5.14 | 1.31 | 22.28 | 0.80 | 0 | 0 |
| EC | 1.19 | 0.78 | 0.14 | 3.07 | 0.15 | 1 | 0 |
| WSOC | 4.91 | 3.17 | 0.89 | 15.42 | 0.05 | 0 | 1 |
| TC (OC + EC) | 9.81 | 5.86 | 1.45 | 24.52 | -- | -- | -- |
| Cl− | 0.04 | 0.07 | 0.00 | 0.45 | 0.01 | 16 | 1 |
| NO3− | 0.57 | 0.73 | 0.00 | 3.18 | 0.03‘ | 5 | 4 |
| SO42− | 4.68 | 3.20 | 0.79 | 16.04 | 0.03 | 0 | 1 |
| Na+ | 0.52 | 0.45 | 0.04 | 1.41 | 0.02 | 0 | 1 |
| NH4+ | 0.61 | 0.73 | 0.01 | 3.65 | 0.02 | 5 | 3 |
| K+ | 0.46 | 0.37 | 0.02 | 1.77 | 0.02 | 1 | 1 |
| Mg2+ | 0.15 | 0.08 | 0.02 | 0.34 | 0.02 | 0 | 1 |
| Ca2+ | 1.40 | 0.99 | 0.14 | 4.65 | 0.03 | 0 | 1 |
| Total Ions | 8.24 | 5.14 | 0.93 | 26.98 | -- | -- | -- |
| Methods | Annual | Winter | Spring | Summer | Autumn | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EC (%) | POC (%) | SOC (%) | EC (%) | POC (%) | SOC (%) | EC (%) | POC (%) | SOC (%) | EC (%) | POC (%) | SOC (%) | EC (%) | POC (%) | SOC (%) | |
| EC tracer | 12.1 | 58.2 | 29.7 | 13.0 | 62.3 | 24.7 | 12.4 | 59.7 | 27.9 | 9.8 | 47.2 | 43.0 | 11.7 | 56.1 | 32.2 |
| 25% | 12.0 | 61.5 | 26.5 | 12.8 | 65.7 | 21.5 | 12.1 | 62.1 | 25.8 | 9.8 | 50.4 | 39.8 | 11.7 | 59.8 | 28.5 |
| MRS | 11.5 | 69.2 | 19.3 | 11.9 | 71.6 | 16.5 | 12.0 | 71.9 | 16.1 | 9.3 | 55.7 | 35.0 | 11.5 | 69.0 | 19.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sheoran, R.; Dumka, U.C.; Kaskaoutis, D.G.; Grivas, G.; Ram, K.; Prakash, J.; Hooda, R.K.; Tiwari, R.K.; Mihalopoulos, N. Chemical Composition and Source Apportionment of Total Suspended Particulate in the Central Himalayan Region. Atmosphere 2021, 12, 1228. https://doi.org/10.3390/atmos12091228
Sheoran R, Dumka UC, Kaskaoutis DG, Grivas G, Ram K, Prakash J, Hooda RK, Tiwari RK, Mihalopoulos N. Chemical Composition and Source Apportionment of Total Suspended Particulate in the Central Himalayan Region. Atmosphere. 2021; 12(9):1228. https://doi.org/10.3390/atmos12091228
Chicago/Turabian StyleSheoran, Rahul, Umesh Chandra Dumka, Dimitris G. Kaskaoutis, Georgios Grivas, Kirpa Ram, Jai Prakash, Rakesh K. Hooda, Rakesh K. Tiwari, and Nikos Mihalopoulos. 2021. "Chemical Composition and Source Apportionment of Total Suspended Particulate in the Central Himalayan Region" Atmosphere 12, no. 9: 1228. https://doi.org/10.3390/atmos12091228
APA StyleSheoran, R., Dumka, U. C., Kaskaoutis, D. G., Grivas, G., Ram, K., Prakash, J., Hooda, R. K., Tiwari, R. K., & Mihalopoulos, N. (2021). Chemical Composition and Source Apportionment of Total Suspended Particulate in the Central Himalayan Region. Atmosphere, 12(9), 1228. https://doi.org/10.3390/atmos12091228











