Microbial Environmental Pollution in ICUs: Results, Trends, and Suggestions from a Long-Lasting Surveillance
Abstract
1. Introduction
2. Materials and Methods
2.1. Setting
2.2. Air Sampling
2.3. Surface Sampling
2.4. Microclimatic Monitoring
2.5. Statistical Analysis
3. Results
3.1. Air Sampling Results
3.2. Surfaces Sampling Results
4. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- D’Alessandro, D.; Fara, G.M. Hospital environments and epidemiology of healthcare-associated infections. In Indoor Air Quality in Healthcare Facilities; SpringerBriefs in Public Health; Capolongo, S., Settimo, G., Gola, M., Eds.; Springer International Publishing: New York, NY, USA, 2017; pp. 41–52. [Google Scholar]
- Valentin, A.; Ferdinande, P.; ESICM Working Group on Quality Improvement. Recommendations on basic requirements for intensive care units: Structural and organizational aspects. Intensive Care Med. 2011, 37, 1575–1587. [Google Scholar] [CrossRef] [PubMed]
- Siegel, J.D.; Rhinehart, E.; Jackson, M.; Chiarello, L. Guideline for isolation precautions: Preventing transmission of infectious agents in health care settings. Am. J. Infect. Control 2007, 35, S65–S164. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.Y.; Tseng, L.; Yang, L.S. Microbial air contamination in an intensive care unit. Int. J. Public Health Sci. 2015, 4, 145–151. [Google Scholar] [CrossRef]
- Verderber, S.; Gray, S.; Suresh-Kumar, S.; Kercz, D.; Parshuram, C. Intensive Care Unit Built Environments: A Comprehensive Literature Review (2005–2020). Health Environ. Res. Des. J. 2021, 1–48. [Google Scholar] [CrossRef]
- Bracco, D.; Dubois, M.J.; Bouali, R.; Eggimann, P. Single rooms may help to prevent nosocomial bloodstream infection and cross-transmission of methicillin-resistant Staphylococcus aureus in intensive care units. Intensive Care Med. 2007, 33, 836–840. [Google Scholar] [CrossRef]
- Bloemendaal, A.L.; Fluit, A.C.; Jansen, W.M.; Vriens, M.R.; Ferry, T.; Argaud, L.; Amorim, J.M.; Resende, A.C.; Pascual, A.; Lopez-Cerero, L.; et al. Acquisition and cross-transmission of Staphylococcus aureus in European intensive care units. Infect. Control. Hosp. Epidemiol. 2009, 30, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Teltsch, D.Y.; Hanley, J.; Loo, V.; Goldberg, P.; Gursahaney, A.; Buckeridge, D.L. Infection acquisition following intensive care unit room privatization. Arch. Intern. Med. 2011, 171, 32–38. [Google Scholar] [CrossRef]
- Cepeda, J.A.; Whitehouse, T.; Cooper, B.; Hails, J.; Jones, K.; Kwaku, F.; Taylor, L.; Hayman, S.; Cookson, B.; Shaw, S.; et al. Isolation of patients in single rooms or cohorts to reduce the spread of MRSA in intensive care units: Prospective two-centre study. Lancet 2005, 364, 295–304. [Google Scholar] [CrossRef]
- Stiller, A.; Schröder, C.; Gropmann, A.; Schwab, F.; Behnke, M.; Geffers, C.; Sunder, W.; Holzhausen, J.; Gastmeier, P. ICU ward design and nosocomial infection rates: A cross-sectional study in Germany. J. Hosp. Infect. 2017, 95, 71–75. [Google Scholar] [CrossRef]
- Ministerio de Sanidad y Política Social. Unidades de Cuidados Intensivos. Estanderas y Recomendaciones; Ministerio de Sanidad y Política Social: Madrid, Spain, 2010; NIPO EN LINEA: 840-10-098-6. [Google Scholar]
- Russotto, V.; Cortegiani, A.; Raineri, S.M.; Giarratano, A. Bacterial contamination of inanimate surfaces and equipment in the intensive care unit. J. Intensive Care 2015, 3, 54. [Google Scholar] [CrossRef]
- Gastmeier, P.; Loui, A.; Stamm-Balderjahn, S.; Hansen, S.; Zuschneid, I.; Sohr, D.; Behnke, M.; Obladen, M.; Vonberg, R.P.; Rüden, H. Outbreaks in neonatal intensive care units—They are not like others. Am. J. Infect. Control 2007, 35, 172–176. [Google Scholar] [CrossRef]
- Mora, M.; Mahnert, A.; Koskinen, K.; Pausan, M.R.; Oberauner-Wappis, L.; Krause, R.; Perras, A.K.; Gorkiewicz, G.; Berg, G.; Moissl-Eichinger, C. Microorganisms in Confined Habitats: Microbial Monitoring and Control of Intensive Care Units, Operating Rooms, Cleanrooms and the International Space Station. Front. Microbiol. 2016, 7, 1573. [Google Scholar] [CrossRef] [PubMed]
- Tringe, S.G.; Hugenholtz, P. A renaissance for the pioneering 16S rRNA gene. Curr. Opin. Microbiol. 2008, 11, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Huntley, J.; Fierer, N.; Owens, S.M.; Betley, J.; Fraser, L.; Bauer, M.; et al. Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J. 2012, 6, 1621–1624. [Google Scholar] [CrossRef]
- Passaretti, C.L.; Otter, J.A.; Reich, N.G.; Myers, J.; Shepard, J.; Ross, T.; Carroll, K.C.; Lipsett, P.; Perl, T.M. An evaluation of environmental decontamination with hydrogen peroxide vapor for reducing the risk of patient acquisition of multidrug-resistant organisms. Clin. Infect. Dis. 2013, 56, 27–35. [Google Scholar] [CrossRef]
- Salgado, C.D.; Sepkowitz, K.A.; John, J.F.; Cantey, J.R.; Attaway, H.H.; Freeman, K.D.; Sharpe, P.A.; Michels, H.T.; Schmidt, M.G. Copper surfaces reduce the rate of healthcare-acquired infections in the intensive care unit. Infect. Control. Hosp. Epidemiol. 2013, 34, 479–486. [Google Scholar] [CrossRef]
- Handorean, A.; Robertson, C.E.; Harris, J.K.; Frank, D.; Hull, N.; Kotter, C.; Stevens, M.J.; Baumgardner, D.; Pace, N.R.; Hernandez, M. Microbial aerosol liberation from soiled textiles isolated during routine residuals handling in a modern health care setting. Microbiome 2015, 3, 72. [Google Scholar] [CrossRef]
- Wertheim, H.F.; Melles, D.C.; Vos, M.C.; van Leeuwen, W.; van Belkum, A.; Verbrugh, H.A.; Nouwen, J.L. The role of nasal carriage in Staphylococcus aureus infections. Lancet Infect. Dis. 2005, 5, 751–762. [Google Scholar] [CrossRef]
- Pittet, D.; Allegranzi, B.; Sax, H.; Dharan, S.; Pessoa-Silva, C.L.; Donaldson, L.; Boyce, J.M.; WHO Global Patient Safety Challenge; World Alliance for Patient Safety. Evidence-based model for hand transmission during patient care and the role of improved practices. Lancet Infect. Dis. 2006, 6, 641–652. [Google Scholar] [CrossRef]
- Finzi, G.; Mura, I.; Kob, K.; Sideli, C.; Lanzoni, L.; Mazzacane, S.; Odi, A.A.; Appicciafuoco, A.; Barchitta, M.; Bertinato, L.; et al. Linee Guida Sulla Valutazione del Processo di Sanificazione Ambientale Nelle Strutture Ospedaliere e Territoriali per Il Controllo Delle Infezioni Correlate All’assistenza (ICA); ANMDO: Bologna, Italy, 2018; Available online: https://www.anmdo.org/wp-content/uploads/2019/01/libro-uno-finzi-1.pdf (accessed on 8 July 2021).
- European Commission. EudraLex. The Rules Governing Medicinal Products in the European Union. Volume 4. EU Guidelines to Good Manifacturing Practice Medicinal Products for Human and Veterinary Use; European Commission: Brussels, Belgium, 2010. Available online: https://ec.europa.eu/health/sites/default/files/files/eudralex/vol-4/2011_intro_en.pdf (accessed on 12 July 2021).
- Saran, S.; Gurjar, M.; Baronia, A.; Sivapurapu, V.; Ghosh, P.S.; Raju, G.M.; Maurya, I. Heating, ventilation and air conditioning (HVAC) in intensive care unit. Crit Care. 2020, 24, 194. [Google Scholar] [CrossRef] [PubMed]
- Decree of the President of the Republic. Approvazione Dell’atto di Indirizzo e Coordinamento Alle Regioni e Alle Province Autonome di Trento e di Bolzano, in Materia di Requisiti Strutturali, Tecnologici ed Organizzativi Minimi per L’esercizio Delle Attivita’ Sanitarie da Parte Delle Strutture Pubbliche e Private. (GU Serie Generale n.42 del 20-02-1997—Suppl. Ordinario n. 37). 1997. Available online: https://www.gazzettaufficiale.it/eli/gu/1997/02/20/42/so/37/sg/pdf (accessed on 8 July 2021).
- Lazio region. Decree of the Commissioner ad Acta U0090 of 10 November 2010 (annex 1). Minimum Authorization Requirements for the Exercise of Health and Socio-Health Activities. Available online: http://www.regione.lazio.it/binary/rl_sanita/tbl_contenuti/Allegato_1_Decr_U0090_2010.pdf (accessed on 14 July 2021).
- College of Intensive Care Medicine of Australia and New Zealand. Minimum Standards for Intensive Care Units; CICM, 2011. Available online: https://www.cicm.org.au/CICM_Media/CICMSite/CICM-Website/Resources/Professional%20Documents/IC-1-Minimum-Standards-for-Intensive-Care-Units.pdf (accessed on 8 July 2021).
- Society of Critical Care Medicine. Guidelines for intensive care unit design. Guidelines/Practice Parameters Committee of the American College of Critical Care Medicine. Crit. Care Med. 1995, 23, 582–588. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services. Centers for Disease Control and Prevention (CDC). Guidelines for Environmental Infection Control in Health-Care Facilities. Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC); U.S. Department of Health and Human Services: Atlanta, GA, USA, 2003. Available online: https://www.cdc.gov/infectioncontrol/pdf/guidelines/environmental-guidelines-P.pdf (accessed on 8 July 2021).
- Pasquarella, C.; Vitali, P.; Saccani, E.; Manotti, P.; Boccuni, C.; Ugolotti, M.; Signorelli, C.; Mariotti, F.; Sansebastiano, G.E.; Albertini, R. Microbial air monitoring in operating theatres: Experience at the University Hospital of Parma. J. Hosp. Infect. 2012, 81, 50–57. [Google Scholar] [CrossRef]
- Vescia, N.; Brenier-Pinchart, M.P.; Osborn, J.F.; Cerquetani, F.; Cavarischia, R.; Grillot, R.; D’Alessandro, D. Field validation of a dusting cloth for mycological surveillance of surfaces. Am. J. Infect. Control 2011, 39, 156–158. [Google Scholar] [CrossRef]
- D’Alessandro, D.; Cerquetani, F.; Deriu, M.G.; Montagna, M.T.; Mura, I.; Napoli, C.; Vescia, N. Evaluation of fungal contamination in operating rooms using a dusting cloth pad: Comparison among different sampling methods. Am. J. Infect. Control 2013, 41, 658–660. [Google Scholar] [CrossRef]
- Rocha, A.C.; Baez, N.A.; Villaroel, E.V.; Quintero, G.M. Study of bioaerosols in surgical theatres and intensive care units from a public general hospital. JBM 2012, 2, 1–10. [Google Scholar] [CrossRef]
- EN (European Standard)—ISO (International Organization for Standardization). Clean Rooms and Associated Controlled Environments. Biocontamination Control. Part 1: General Principles and Methods; International Organization for Standardization: Geneva, Switzerland, 2004. [Google Scholar]
- Istituto Superiore per la Prevenzione e la Sicurezza del Lavoro (ISPESL). Linee Guida Sugli Standard di Sicurezza e di Igiene del Lavoro nel Reparto Operatorio; ISPESL: Roma, Italy, 2009. Available online: https://www.inail.it/cs/internet/docs/linee-guida-igiene-reparto-operatorio.pdf?section=attivita (accessed on 8 July 2021).
- CCLIn Sud-Ouest. Surveillance Microbiologique de L’environnement Dans les Établissements de Santé. Guide de Bonnes Pratiques. 2016. Available online: https://www.cpias-nouvelle-aquitaine.fr/wp-content/uploads/2015/08/Surv_microbio_environnement.pdf (accessed on 12 July 2021).
- Barca, S.; Caradonna, L.; Giaquinta, G.; Giovinazzo, R.; Guerrera, E.; Mameli, M.; Mansi, A.; Marena, G.; Mastromartino, T.; Sarto, D.; et al. Contaminazione Microbiologica Delle Superfici Negli Ambienti di Lavoro; INAIL, 2017. Available online: https://www.inail.it/cs/internet/docs/alg-pubbl-la-contaminazione-microbiologica-delle-superfici.pdf (accessed on 12 July 2021).
- European Commission. EudraLex. The Rules Governing Medicinal Products in the European Union. Volume 4. Goof Manifacturing Practice. Guidelines of Good Manifacturing Practice specific to Advanced Therapy Medicinal Products. 2017. Available online: https://ec.europa.eu/health/sites/default/files/files/eudralex/vol-4/2017_11_22_guidelines_gmp_for_atmps.pdf (accessed on 12 July 2021).
- Thompson, D.R.; Hamilton, D.K.; Cadenhead, C.D.; Swoboda, S.M.; Schwindel, S.M.; Anderson, D.C.; Schmitz, E.V.; St Andre, A.C.; Axon, D.C.; Harrell, J.W.; et al. Guidelines for intensive care unit design. Crit Care Med. 2012, 40, 1586–1600. [Google Scholar] [CrossRef] [PubMed]
- Chaudhury, H.; Mahmood, A.; Valente, M. Advantages and disadvantages of single versus multiple occupancy rooms in acute care environments: A review and analysis of the literature. Environ. Behav. 2005, 37, 760–786. [Google Scholar] [CrossRef]
- Harris, D.D.; Shepley, M.M.; White, R.D.; Kolberg, K.J.S.; Harrell, J.W. The impact of single family room design on patients and caregivers: Executive summary. J. Perinatol. 2006, 26, S38–S48. [Google Scholar] [CrossRef][Green Version]
- Chaudhury, H.; Mahmood, A.; Valente, M. Nurses’ perception of single-occupancy versus multioccupancy rooms in acute care environments: An exploratory comparative assessment. Appl. Nurs. Res. 2006, 19, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Gabor, J.Y.; Cooper, A.B.; Crombach, S.A.; Lee, B.; Kadikar, N.; Bettger, H.E.; Hanly, P.J. Contribution of the intensive care unit environment to sleep disruption in mechanically ventilated patients and healthy subjects. Am. J. Respir. Crit. Care Med. 2003, 167, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Appolloni, L.; D’Alessandro, D. Housing Spaces in Nine European Countries: A Comparison of Dimensional Requirements. Int. J. Environ. Res. Public Health 2021, 18, 4278. [Google Scholar] [CrossRef]
- Jones, N.R.; Qureshi, Z.U.; Temple, R.J.; Larwood, J.P.J.; Greenhalgh, T.; Bourouiba, L. Two metres or one: What is the evidence for physical distancing in covid-19. BMJ 2020, 370, m3223. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Otter, J.A.; Price, J.R.; Cimpeanu, C.; Garcia, D.M.; Kinross, J.; Boshier, P.R.; Mason, S.; Bolt, F.; Holmes, A.H.; et al. Investigating SARS-CoV-2 surface and air contamination in an acute healthcare setting during the peak of the COVID-19 pandemic in London. medRxiv 2020. [Google Scholar] [CrossRef] [PubMed]
- Phua, J.; Weng, L.; Ling, L.; Egi, M.; Lim, C.M.; Divatia, J.V.; Shrestha, B.R.; Arabi, Y.M.; Ng, J.; Gomersall, C.D.; et al. Asian Critical Care Clinical Trials Group. Intensive care management of coronavirus disease 2019 (COVID-19): Challenges and recommendations. Lancet Respir. Med. 2020, 8, 506–517. [Google Scholar] [CrossRef]
- Ichai, P.; Saliba, F.; Baune, P.; Daoud, A.; Coilly, A.; Samuel, D. Impact of negative air pressure in ICU rooms on the risk of polmunary aspergillosis in COVID-19 patients. Crit. Care 2020, 21, 538. [Google Scholar] [CrossRef]
- Agodi, A.; Auxilia, F.; Barchitta, M.; Cristina, M.L.; D’Alessandro, D.; Mura, I.; Nobile, M.; Pasquarella, C.; Italian Study Group of Hospital Hygiene. Operating theatre ventilation systems and microbial air contamination in total joint replacement surgery: Results of the GISIO-ISChIA study. J. Hosp. Infect. 2015, 90, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Balocco, C.; Petrone, G.; Cammarata, G. Assessing the effects of sliding doors on an operating theatre climate. Build. Simul. 2012, 5, 73–83. [Google Scholar] [CrossRef]
- Mears, S.C.; Blanding, R.; Belkoff, S.M. Door opening affects operating room pressure during joint arthroplasty. J. Orthoped. 2015, 38, 991–994. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, D.; Agodi, A.; Auxilia, F.; Brusaferro, S.; Calligaris, L.; Ferrante, M.; Montagna, M.T.; Mura, I.; Napoli, C.; Pasquarella, C.; et al. Prevention of healthcare associated infections: Medical and nursing students’ knowledge in Italy. Nurse Educ. Today 2014, 34, 191–195. [Google Scholar] [CrossRef]
- Masia, M.D.; Dettori, M.; Deriu, M.G.; Soddu, S.; Deriu, M.; Arghittu, A.; Azara, A.; Castiglia, P. Microbial monitoring as a tool for preventing infectious risk in the operating room: Results of 10 years activity. Atmosphere 2021, 12, 19. [Google Scholar] [CrossRef]
- Birgand, G.; Saliou, P.; Lucet, J.C. Influence of staff behavior on infectious risk in operating rooms: What is the evidence? Infect. Control. Hosp. Epidemiol. 2015, 36, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Pokrywka, M.; Byers, K. Traffic in the operating room: A review of factors influencing air flow and surgical wound contamination. Infect. Disord. Drug. Targets 2013, 13, 156–161. [Google Scholar] [CrossRef] [PubMed]
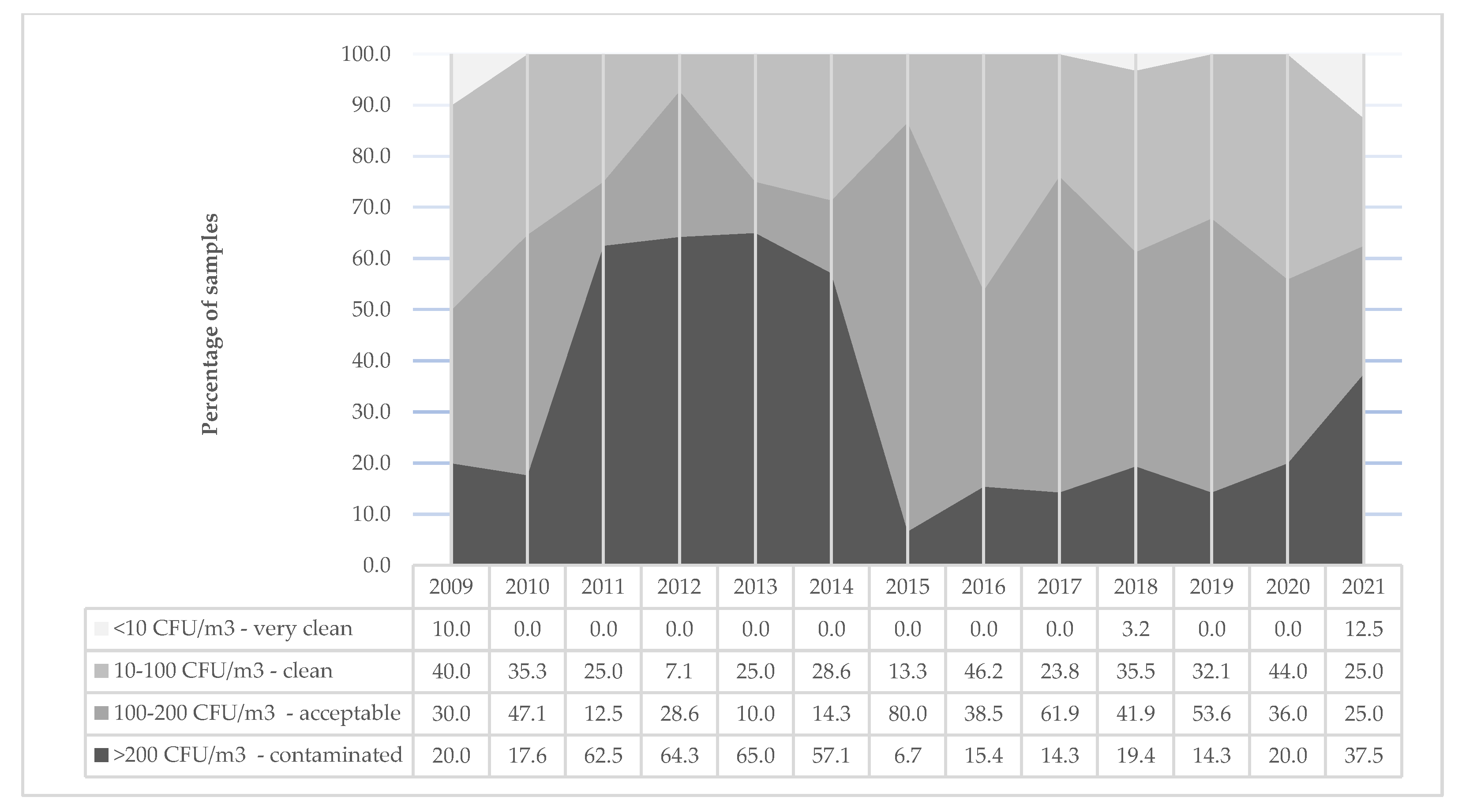
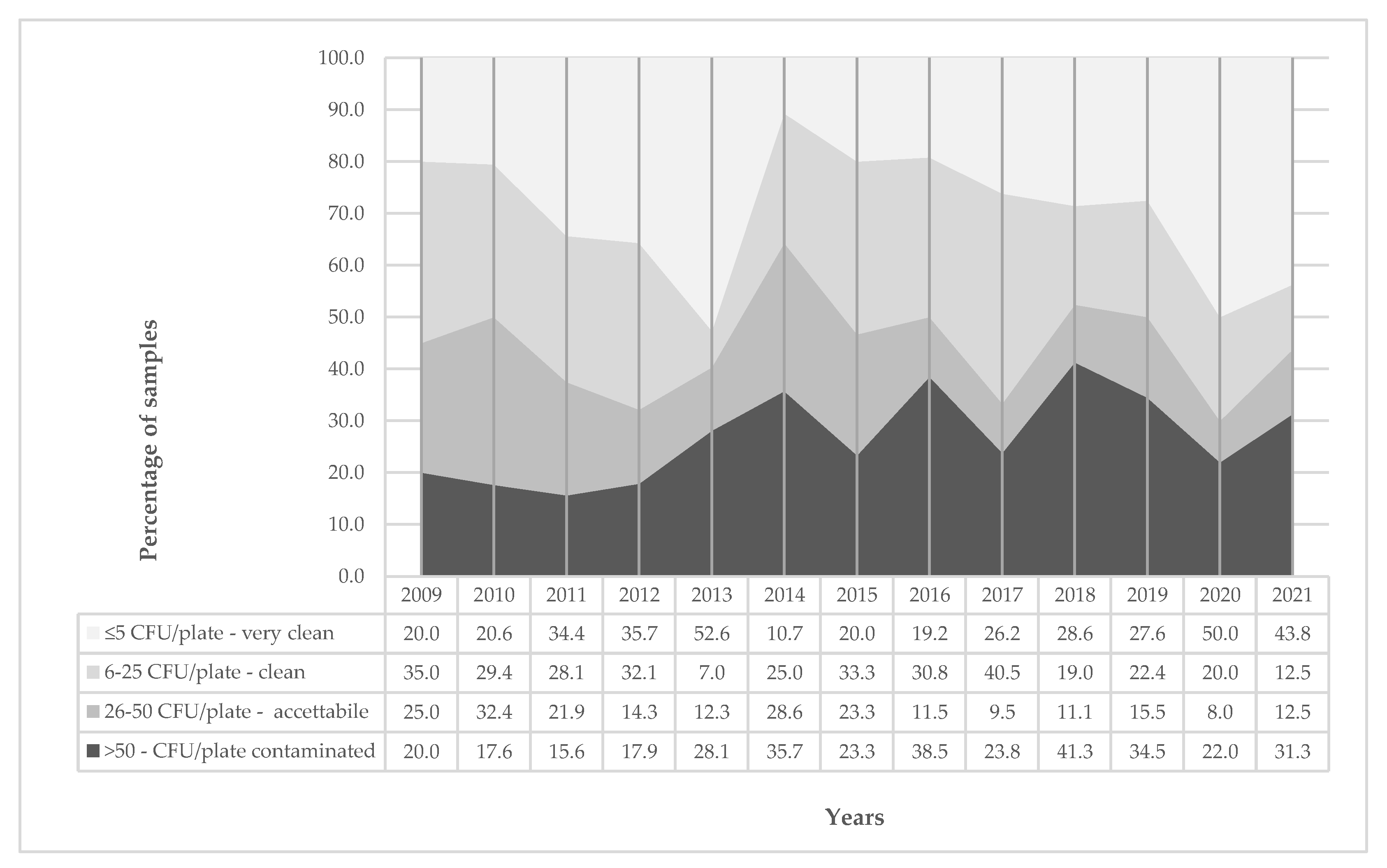

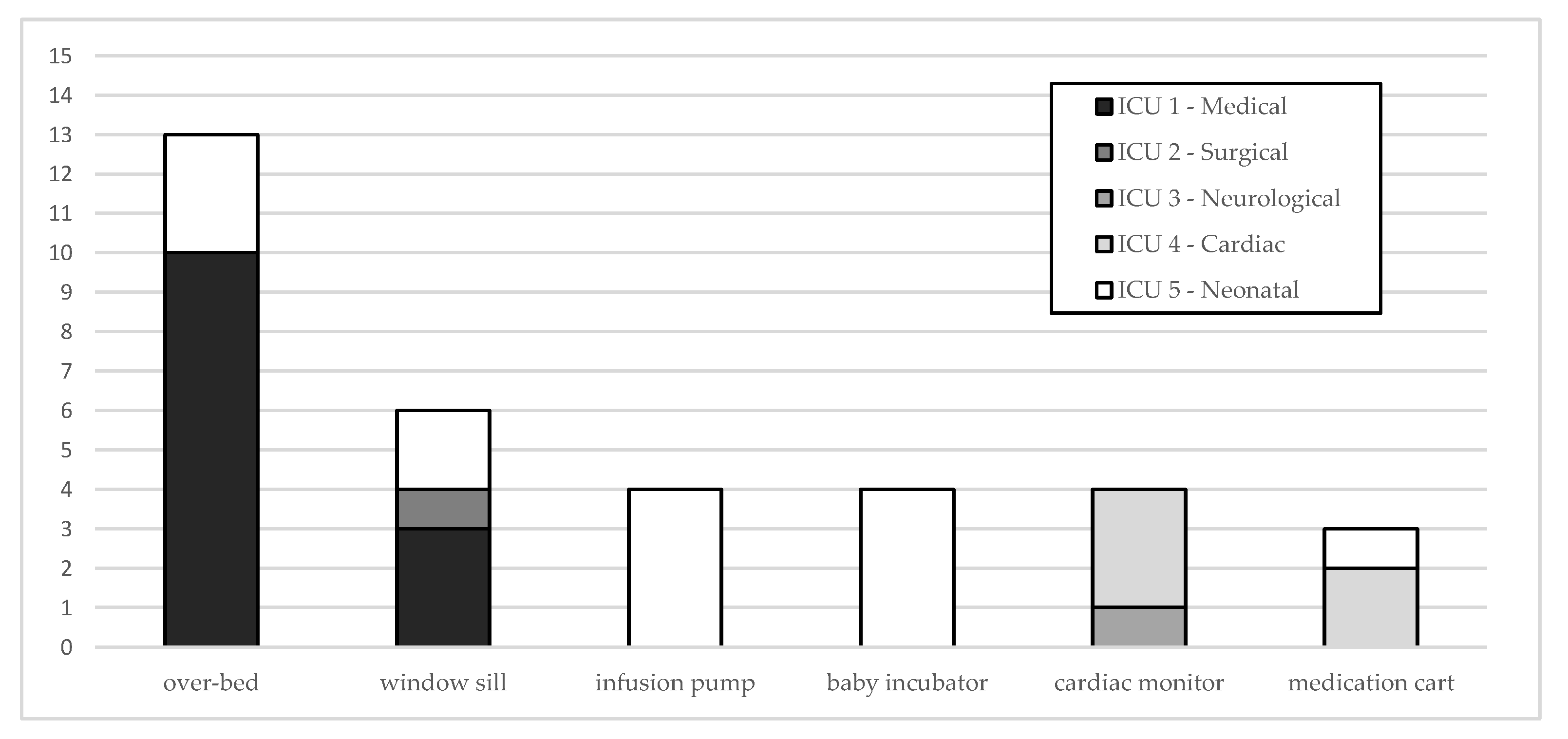
| ICUs | # Beds | # Rooms | HVAC System |
|---|---|---|---|
| ICU 1—Medical | 18 | 7 | centralized |
| ICU 2—Surgical | 6 | 1 | centralized |
| ICU 3—Neurologica | 9 | 1 | centralized |
| ICU 4—Cardiac | 9 | 1 | centralized |
| ICU 5—Neonatal | 15 | 4 | decentralized |
| ICUs | # Samples * | Mean | SD | Min–Max | 25th Percentile | Median | 75th Percentile |
|---|---|---|---|---|---|---|---|
| ICU 1—Medical | 64 | 146.1 | 125.2 | 10–610 | 78 | 105.5 | 168.7 |
| ICU 2—Surgical | 29 | 163.8 | 142.3 | 17–505 | 67 | 121.0 | 171.2 |
| ICU 3—Neurological | 30 | 158.6 | 144.5 | 1–594 | 58 | 127.5 | 186.0 |
| ICU 4—Cardiac | 24 | 216.5 | 211.8 | 11–972 | 98.5 | 166.5 | 229.7 |
| ICU 5—Neonatal | 84 | 201.8 | 163.7 | 6–972 | 118.7 | 167.5 | 249.2 |
| ICUs | 2009–2010 | 2011–2012 | 2013–2014 | 2015–2016 | 2017–2018 | 2019–2020 | X2 Per Trend | p-Value |
|---|---|---|---|---|---|---|---|---|
| ICU 1—Medical | 1/5 | 2/4 | 2/3 | 0/4 | 3/22 | 6/27 | 0.852 | n.s |
| ICU 2—Surgical | 0/6 | 3/6 | 1/4 | 1/3 | 1/4 | 1/5 | 0.052 | n.s |
| ICU 3—Neurological | 1/6 | 0/4 | 3/4 | 0/5 | 2/6 | 0/3 | 0.001 | n.s |
| ICU 4—Cardiac | 0/3 | 2/4 | 3/3 | 0/4 | 1/5 | 1/4 | 0.243 | n.s |
| ICU 5—Neonatal | 3/7 | 12/12 | 12/20 | 2/12 | 2/14 | 2/15 | 18.8 | <0.005 |
| Total | 5/27 | 19/30 | 21/34 | 3/28 | 9/51 | 10/54 | 11.6 | <0.05 |
| ICUs | # Samples * | Mean | SD | (Min–Max) | 25th Percentile | Median | 75th Percentile |
|---|---|---|---|---|---|---|---|
| ICU 1—Medical | 117 | 37.1 | 33.5 | 0–145 | 8 | 27 | 63 |
| ICU 2—Surgical | 57 | 22.2 | 22.7 | 0–104 | 3 | 16 | 35 |
| ICU 3—Neurological | 62 | 23.4 | 24.6 | 0–83 | 2 | 14 | 45 |
| ICU 4—Cardiac | 48 | 41.4 | 47.9 | 1–195 | 8.7 | 27 | 53.5 |
| ICU 5—Neonatal | 167 | 24.5 | 31.8 | 0–131 | 1 | 8 | 39.5 |
| ICUs | 2009–2010 | 2011–2012 | 2013–2014 | 2015–2016 | 2017–2018 | 2019–2020 | X2 Per Trend | p |
|---|---|---|---|---|---|---|---|---|
| ICU 1—Medical | 3/10 | 4/8 | 2/6 | 1/8 | 19/44 | 23/54 | 0.478 | n.s. |
| ICU 2—Surgical | 0/12 | 0/12 | 4/8 | 2/6 | 0/8 | 1/10 | 0.440 | n.s. |
| ICU 3—Neurological | 1/12 | 0/8 | 0/8 | 2/10 | 4/15 | 1/6 | 2.780 | n.s |
| ICU 4—Cardiac | 2/6 | 3/8 | 2/6 | 2/8 | 4/10 | 0/8 | 1.358 | n.s |
| ICU 5—Neonatal | 4/14 | 3/24 | 17/55 | 10/24 | 9/28 | 6/30 | 0.046 | n.s |
| Total | 10/54 | 10/60 | 25/83 | 17/56 | 36/105 | 31/108 | 4.597 | n.s |
| Temperature (°) | Relative Humidity (%) | Air Velocity (m/s) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| ICUs | Mean | Min–Max | % Compliance | Mean | Min–Max | % Compliance | Mean | Min–Max | % Compliance |
| ICU 1–Medical | 24.5 | 21.8–27.1 | 38.9 | 44.0 | 29.6–58.8 | 72.2 | 0.08 | 0.01–1.20 | 35.2 |
| ICU 2–Surgical | 24.2 | 20.0–27.1 | 40.0 | 48.9 | 32.2–80.5 | 60.0 | 0.05 | 0.00–0.15 | 60.0 |
| ICU 3–Neurological | 22.9 | 21.3–24.5 | 100.0 | 47.9 | 29.6–67.1 | 61.1 | 0.09 | 0.00–0.24 | 72.2 |
| ICU 4–Cardiac | 23.8 | 21.5–26.6 | 43.8 | 48.2 | 14.7–70.4 | 43.8 | 0.30 | 0.00–1.59 | 68.8 |
| ICU 5–Neonatal | 24.5 | 22.6–27.0 | 100.0 | 42.8 | 21.5–59.5 | 57.4 | 0.06 | 0.01–0.18 | 66.7 |
| Total | 24.2 | 20.7–27.1 | 67.5 | 44.9 | 14.7–80.5 | 61.8 | 0.10 | 0.00–1.60 | 56.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Alessandro, D.; Fabiani, M.; Appolloni, L. Microbial Environmental Pollution in ICUs: Results, Trends, and Suggestions from a Long-Lasting Surveillance. Atmosphere 2021, 12, 1174. https://doi.org/10.3390/atmos12091174
D’Alessandro D, Fabiani M, Appolloni L. Microbial Environmental Pollution in ICUs: Results, Trends, and Suggestions from a Long-Lasting Surveillance. Atmosphere. 2021; 12(9):1174. https://doi.org/10.3390/atmos12091174
Chicago/Turabian StyleD’Alessandro, Daniela, Massimo Fabiani, and Letizia Appolloni. 2021. "Microbial Environmental Pollution in ICUs: Results, Trends, and Suggestions from a Long-Lasting Surveillance" Atmosphere 12, no. 9: 1174. https://doi.org/10.3390/atmos12091174
APA StyleD’Alessandro, D., Fabiani, M., & Appolloni, L. (2021). Microbial Environmental Pollution in ICUs: Results, Trends, and Suggestions from a Long-Lasting Surveillance. Atmosphere, 12(9), 1174. https://doi.org/10.3390/atmos12091174







