Characterization of the Gaseous and Odour Emissions from the Composting of Conventional Sewage Sludge
Abstract
1. Introduction
2. Experiments
2.1. Characteristics of the Feedstocks
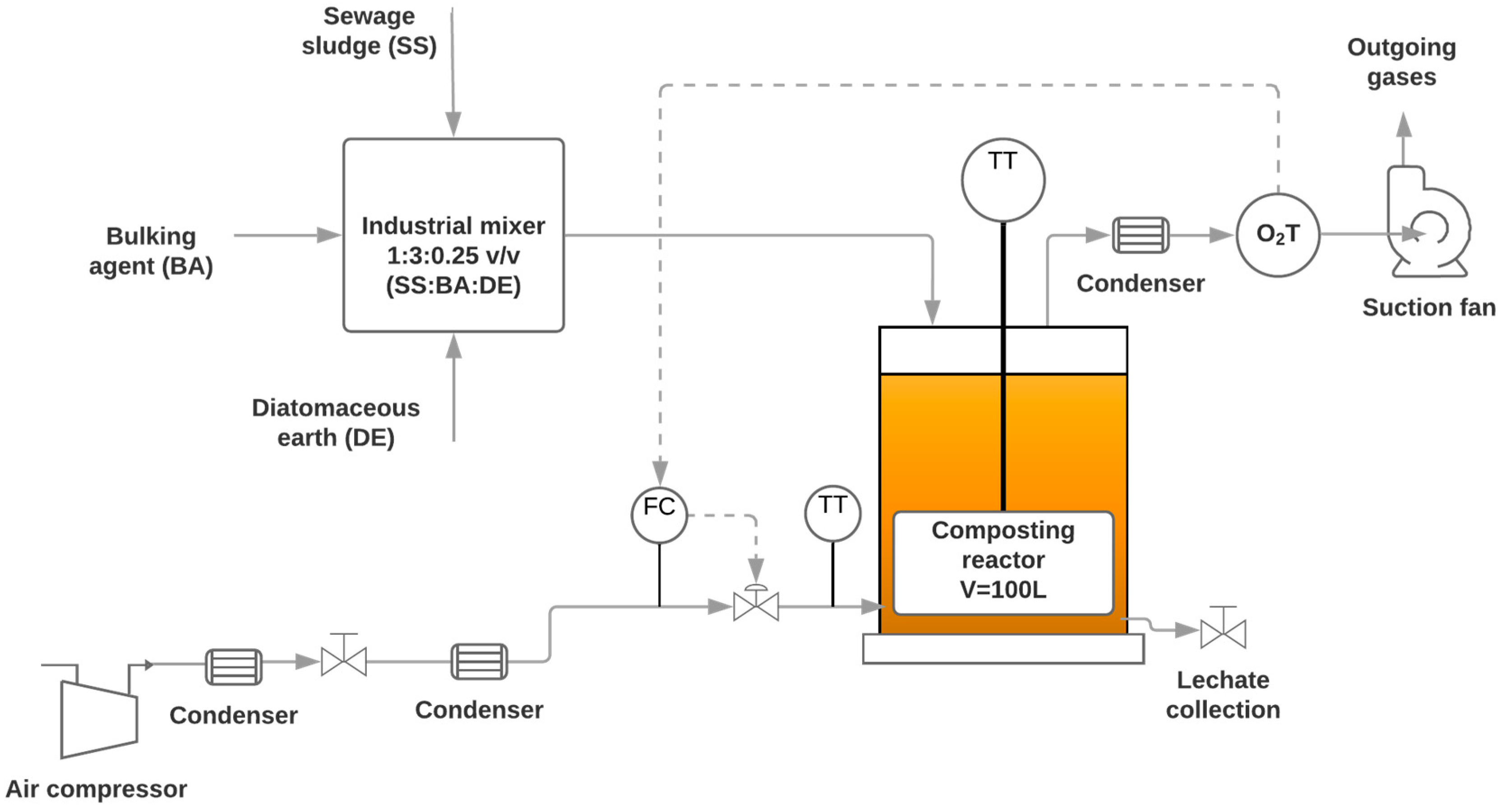
2.2. Sewage Sludge Composting Biorreactor Operation
2.3. Gas Sampling Methodology
2.4. Analytical Methodology
2.5. Estimation of the Emission Factors
2.6. Potential Odour Contributors
3. Results and Discussion
3.1. Sewage Sludge Composting Performance
3.2. Greenhouse Gases Emission
3.3. Odorant Compounds Emission
3.3.1. NH3, H2S, tVOCs and Odour Emission
3.3.2. VOCs’ Characterization
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- European Commission Council Directive 91/271/EEC concerning urban waste water treatment. Off. J. Eur. Union 1992, 28, 14–15.
- Eurostat Sewage Sludge Production and Disposal Database 2004–2015. Available online: http://ec.europa.eu/eurostat/tgm/table.do?tab=table&plugin=1&language=en&pcode=ten00030 (accessed on 4 July 2019).
- Colón, J.; Alarcón, M.; Healy, M.; Namli, A.; Ponsá, S.; Sanin, F.D.; Taya, C. Producing sludge for agricultural applications. In Innovative Wastewater Treatment and Resource Recovery Technologies; IWA: London, UK, 2014; pp. 292–314. [Google Scholar]
- Tchobanoglous, G. Gestión Integral de Residuos Sólidos; McGraw-Hill: Madrid, Spain, 1994. [Google Scholar]
- Lebrero, R.; Rangel, M.G.L.; Muñoz, R. Characterization and biofiltration of a real odorous emission from wastewater treatment plant sludge. J. Environ. Manag. 2013, 116, 50–57. [Google Scholar] [CrossRef]
- Maulini-Duran, C.; Artola, A.; Font, X.; Sánchez, A. A systematic study of the gaseous emissions from biosolids composting: Raw sludge versus anaerobically digested sludge. Bioresour. Technol. 2013, 147, 43–51. [Google Scholar] [CrossRef]
- Colón, J.; Cadena, E.; Pognani, M.; Barrena, R.; Sánchez, A.; Font, X.; Artola, A. Determination of the energy and environmental burdens associated with the biological treatment of source-separated Municipal Solid Wastes. Energy Environ. Sci. 2012, 5, 5731–5741. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifht Assessment Report of the Intergovernmental Panel on Climate Change; Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014; ISBN 9789291691432. [Google Scholar]
- Pagans, E.; Barrena, R.; Font, X.; Sánchez, A. Ammonia emissions from the composting of different organic wastes. Dependency on process temperature. Chemosphere 2006, 62, 1534–1542. [Google Scholar] [CrossRef] [PubMed]
- González, D.; Colón, J.; Gabriel, D.; Sánchez, A. The effect of the composting time on the gaseous emissions and the compost stability in a full-scale sewage sludge composting plant. Sci. Total Environ. 2019, 654, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Chen, T.B.; Gao, D.; Zheng, G.; Liu, H.; Yang, Q. Online monitoring of volatile organic compound production and emission during sewage sludge composting. Bioresour. Technol. 2012, 123, 463–470. [Google Scholar] [CrossRef]
- Sironi, S.; Capelli, L.; Céntola, P.; Del Rosso, R.; Il Grande, M. Odour emission factors for assessment and prediction of Italian MSW landfills odour impact. Atmos. Environ. 2005, 39, 5387–5394. [Google Scholar] [CrossRef]
- Capelli, L.; Sironi, S.; Del Rosso, R.; Céntola, P. Predicting odour emissions from wastewater treatment plants by means of odour emission factors. Water Res. 2009, 43, 1977–1985. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (USEPA). Compilation of Air Pollutant Emission Factors, AP-42, 5th ed.; Stationary Point and Area Sources; USEPA: Research Triangle Park, NC, USA, 1995; Volume 1.
- Sironi, S.; Capelli, L.; Céntola, P.; Del Rosso, R.; Il Grande, M. Odour emission factors for the prediction of odour emissions from plants for the mechanical and biological treatment of MSW. Atmos. Environ. 2006, 40, 7632–7643. [Google Scholar] [CrossRef]
- Capelli, L.; Sironi, S.; Rosso, R. Del Odour Emission Factors: Fundamental Tools for Air Quality Management. Chem. Eng. Trans. 2014, 40. [Google Scholar]
- Lucernoni, F.; Tapparo, F.; Capelli, L.; Sironi, S. Evaluation of an Odour Emission Factor (OEF) to estimate odour emissions from landfill surfaces. Atmos. Environ. 2016, 144, 87–99. [Google Scholar] [CrossRef]
- Capelli, L.; Sironi, S.; del Rosso, R. Odor sampling: Techniques and strategies for the estimation of odor emission rates from different source types. Sensors (Switzerland) 2013, 13, 938–955. [Google Scholar] [CrossRef] [PubMed]
- Puyuelo, B.; Gea, T.; Sánchez, A. A new control strategy for the composting process based on the oxygen uptake rate. Chem. Eng. J. 2010, 165, 161–169. [Google Scholar] [CrossRef]
- US Dept. of Agriculture; US Composting Council. Test methods for the examination of composting and compost purpose. In Edaphos International, Houston; US Composting Council: Raleigh, NC, USA, 2001. [Google Scholar]
- Water Environment Federation. Standard Methods for the Examination of Water and Wastewater; American Public Health Association, American Water Works Association and Water Environment Federation: Washington, DC, USA, 1995. [Google Scholar]
- Gea, T.; Barrena, R.; Artola, A.; Sánchez, A. Monitoring the biological activity of the composting process: Oxygen uptake rate (OUR), respirometric index (RI), and respiratory quotient (RQ). Biotechnol. Bioeng. 2004, 88, 520–527. [Google Scholar] [CrossRef]
- Ponsá, S.; Gea, T.; Sánchez, A. Different Indices to Express Biodegradability in Organic Solid Wastes. J. Environ. Qual. 2010, 39, 706–712. [Google Scholar] [CrossRef]
- González, D.; Colón, J.; Sánchez, A.; Gabriel, D. A systematic study on the VOCs characterization and odour emissions in a full-scale sewage sludge composting plant. J. Hazard. Mater. 2019, 373, 733–740. [Google Scholar] [CrossRef]
- Ribes, A.; Carrera, G.; Gallego, E.; Roca, X.; Berenguer, M.J.; Guardino, X. Development and validation of a method for air-quality and nuisance odors monitoring of volatile organic compounds using multi-sorbent adsorption and gas chromatography/mass spectrometry thermal desorption system. J. Chromatogr. A 2007, 1140, 44–55. [Google Scholar] [CrossRef]
- Blazy, V.; de Guardia, A.; Benoist, J.C.; Daumoin, M.; Guiziou, F.; Lemasle, M.; Wolbert, D.; Barrington, S. Correlation of chemical composition and odor concentration for emissions from pig slaughterhouse sludge composting and storage. Chem. Eng. J. 2015, 276, 398–409. [Google Scholar] [CrossRef]
- Nagata, Y. Odor measurement review, Measurement of odor threshold by triangle odor bag method. Minist. Enviornmental Gov. Japan 2003, 122–123. [Google Scholar]
- Blazy, V.; de Guardia, A.; Benoist, J.C.; Daumoin, M.; Lemasle, M.; Wolbert, D.; Barrington, S. Odorous gaseous emissions as influence by process condition for the forced aeration composting of pig slaughterhouse sludge. Waste Manag. 2014, 34, 1125–1138. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Wei, S.; Shao, L.; Lü, F. Emission potential of volatile sulfur compounds (VSCs) and ammonia from sludge compost with different bio-stability under various oxygen levels. Waste Manag. 2017, 73, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, A.; Gabarrell, X.; Barrena, R.; Colón, J.; Artola, A.; Font, X.; Komilis, D. Composting of Wastes. In Resource Recovery to Approach Zero Municipal Waste; Taherzadeh, M.J., Richards, T., Eds.; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Yuan, J.; Chadwick, D.; Zhang, D.; Li, G.; Chen, S.; Luo, W.; Du, L.; He, S.; Peng, S. Effects of aeration rate on maturity and gaseous emissions during sewage sludge composting. Waste Manag. 2016, 56, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Ruggieri, L.; Gea, T.; Artola, A.; Sánchez, A. Influence of different co-substrates biochemical composition on raw sludge co-composting. Biodegradation 2008, 19, 403–415. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Wu, Y.; Li, W.; Wu, C.; Chen, Z. Insight into effects of mature compost recycling on N2O emission and denitrification genes in sludge composting. Bioresour. Technol. 2018, 251, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Cai, H.; Zhang, Z.; Liu, H.; Li, R.; Mao, H.; Awasthi, M.K.; Wang, Q.; Zhai, L. Comparative evaluation of the use of acidic additives on sewage sludge composting quality improvement, nitrogen conservation, and greenhouse gas reduction. Bioresour. Technol. 2018, 270, 467–475. [Google Scholar] [CrossRef]
- He, Y.; Inamori, Y.; Mizuochi, M.; Kong, H.; Iwami, N.; Sun, T. Nitrous oxide emissions from aerated composting of organic waste. Environ. Sci. Technol. 2001, 35, 2347–2351. [Google Scholar] [CrossRef]
- Moënne-Loccoz, P.; Fee, J.A. Catalyzing NO to N2O in the nitrogen cycle. Science 2010, 330, 10–11. [Google Scholar] [CrossRef]
- Fukumoto, Y.; Osada, T.; Hanajima, D.; Haga, K. Patterns and quantities of NH3, N2O and CH4 emissions during swine manure composting without forced aeration - Effect of compost pile scale. Bioresour. Technol. 2003, 89, 109–114. [Google Scholar] [CrossRef]
- Ahn, H.K.; Mulbry, W.; White, J.W.; Kondrad, S.L. Pile mixing increases greenhouse gas emissions during composting of dairy manure. Bioresour. Technol. 2011, 102, 2904–2909. [Google Scholar] [CrossRef]
- Cai, L.; Chen, T.B.; Gao, D.; Yu, J. Bacterial communities and their association with the bio-drying of sewage sludge. Water Res. 2016, 90, 44–51. [Google Scholar] [CrossRef]
- He, Y.; Inamori, Y.; Mizuochi, M.; Kong, H.; Iwami, N.; Sun, T. Measurements of N2O and CH4 from the aerated composting of food waste. Sci. Total Environ. 2000, 254, 65–74. [Google Scholar] [CrossRef]
- Sánchez-Monedero, M.A.; Serramiá, N.; Civantos, C.G.O.; Fernández-Hernández, A.; Roig, A. Greenhouse gas emissions during composting of two-phase olive mill wastes with different agroindustrial by-products. Chemosphere 2010, 81, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Rincón, C.A.; De Guardia, A.; Couvert, A.; Le Roux, S.; Soutrel, I.; Daumoin, M.; Benoist, J.C. Chemical and odor characterization of gas emissions released during composting of solid wastes and digestates. J. Environ. Manag. 2019, 233, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Xin-Wu, W. Reduction of ammonia, hydrogen sulfide, and Short-Chain fatty acids emission during the sewage sludge composting. Clean Soil Air Water 2010, 38, 998–1005. [Google Scholar] [CrossRef]
- Zigmontiene, A.; Zuokaite, E. Investigation into emissions of gaseous pollutants during sewage sludge composting with wood waste. J. Environ. Eng. Landsc. Manag. 2010, 18, 128–136. [Google Scholar] [CrossRef]
- Hort, C.; Gracy, S.; Platel, V.; Moynault, L. Evaluation of sewage sludge and yard waste compost as a biofilter media for the removal of ammonia and volatile organic sulfur compounds (VOSCs). Chem. Eng. J. 2009, 152, 44–53. [Google Scholar] [CrossRef]
- Scaglia, B.; Orzi, V.; Artola, A.; Font, X.; Davoli, E.; Sanchez, A.; Adani, F. Odours and volatile organic compounds emitted from municipal solid waste at different stage of decomposition and relationship with biological stability. Bioresour. Technol. 2011, 102, 4638–4645. [Google Scholar] [CrossRef]
- Toledo, M.; Márquez, P.; Siles, J.A.; Chica, A.F.; Martín, M.A. Co-composting of sewage sludge and eggplant waste at full scale: Feasibility study to valorize eggplant waste and minimize the odoriferous impact of sewage sludge. J. Environ. Manage. 2019, 247, 205–213. [Google Scholar] [CrossRef]
- Moreno, A.I.; Arnáiz, N.; Font, R.; Carratalá, A. Chemical characterization of emissions from a municipal solid waste treatment plant. Waste Manag. 2014, 34, 2393–2399. [Google Scholar] [CrossRef] [PubMed]
- Schiavon, M.; Martini, L.M.; Corrà, C.; Scapinello, M.; Coller, G.; Tosi, P.; Ragazzi, M. Characterisation of volatile organic compounds (VOCs) released by the composting of different waste matrices. Environ. Pollut. 2017, 231, 845–853. [Google Scholar] [CrossRef] [PubMed]
- Cometto-Muñiz, J.E.; Cain, W.; Abraham, M.; Kumarsingh, R. Sensory properties of selected terpenes. Thresholds for odor, nasal pungency, nasal localization, and eye irritation. Ann. N. Y. Acad. Sci. 1998, 855, 648–651. [Google Scholar] [CrossRef] [PubMed]
- Leffingwell Odor & Flavor Detection Thresholds. Available online: http://www.leffingwell.com/odorthre.htm (accessed on 10 December 2018).
- Parker, D.B.; Koziel, J.A.; Cai, L.; Jacobson, L.D.; Akdeniz, N.; Bereznicki, S.; Lim, T.; Caraway, E.A.; Zhang, S.; Hoff, S.J.; et al. Odor and Odorous Chemical Emissions from Animal Buildings: Part 6. Odor Activity Value. Trans. ASABE 2012, 55, 2357–2368. [Google Scholar] [CrossRef]
- Wenjing, L.; Zhenhan, D.; Dong, L.; Jimenez, L.M.C.; Yanjun, L.; Hanwen, G.; Hongtao, W. Characterization of odor emission on the working face of landfill and establishing of odorous compounds index. Waste Manag. 2015, 42, 74–81. [Google Scholar] [CrossRef]
- Hanajima, D.; Kuroda, K.; Morishita, K.; Fujita, J.; Maeda, K.; Morioka, R. Key odor components responsible for the impact on olfactory sense during swine feces composting. Bioresour. Technol. 2010, 101, 2306–2310. [Google Scholar] [CrossRef]





| Physical–Chemical Properties | SS | PW | DE | Composting Mixture |
|---|---|---|---|---|
| Moisture content (%) | 83.5 ± 0.1 | 18.0 ± 0.0 | 3.9 ± 0.0 | 59.2 ± 0.0 |
| Organic matter content (%) | 68.9 ± 0.2 | 88.7 ± 0.0 | 35.2 ± 0.1 | 85.9 ± 0.0 |
| C/N ratio | 9.1 | 31.6 | 1321.6 | 13.6 |
| pH | 6.7 | 8.2 | 6.2 | – |
| Electrical conductivity (mS·cm−1) | 2.5 | 1.7 | 1.2 | – |
| DRI24 h (g O2·h−1·kg−1 VS) | 7.3 ± 0.9 | – | – | 6.3 ± 1.7 |
| CH4 emission factor (kg CH4·Mg−1 DM–SS) | 1.37 × 10−1 |
| N2O emission factor (kg N2O·Mg−1 DM–SS) | 8.53 × 10−1 |
| CH4 emission factor (kg CO2eq·Mg−1 DM–SS) a | 3.83 |
| N2O emission factor (kg CO2eq·Mg−1 DM–SS) a | 2.26 × 102 |
| GHG emission factor (kg CO2eq·Mg−1 DM–SS) a | 2.30 × 102 |
| NH3 emission factor (kg NH3·Mg−1 DM–SS) | 5.13 |
| H2S emission factor (kg H2S·Mg−1 DM–SS) | 1.66 × 10−1 |
| tVOCs emission factor (kg C-VOC·Mg−1 DM–SS) | 6.20 |
| Odour emission factor (ou·Mg−1 DM–SS) | 2.68 × 107 |
| VOC Concentration | OAVi | |||||
|---|---|---|---|---|---|---|
| Family | Compound | ODT | Day 2 | Day 11 | Day 2 | Day 11 |
| Alkanes | Heptane | 670 a | 58.2 | <DL | 8.7 × 10−2 | <DL |
| Aromatic HC | Benzene | 2700 a | 3.7 | <DL | 1.4 × 10−3 | <DL |
| Toluene | 330 a | 69.7 | <DL | 0.2 | <DL | |
| m-xylene | 0.041 a | 10.7 | <DL | 261.0 | <DL | |
| Styrene | 35 a | 254.9 | 7.3 | - | ||
| Ketones | 2-butanone | 440 a | 5793.9 | 13.2 | - | |
| 2-pentanone | 28 a | 2369.1 | 84.6 | - | ||
| N compounds | Pyridine | 63 a | 910.6 | 14.5 | - | |
| Indole | 0.3 a | 7.5 | 25.0 | - | ||
| S compounds | DMS | 3 a | - | 70.9 | - | 23.6 |
| DMDS | 2.2 a | 37,813.0 | 70.1 | 17,187.7 | 31.9 | |
| Terpenes | α-pinene | 18 a | 13,299.1 | 12.8 | 738.8 | 0.7 |
| β-pinene | 33 a | 6390.3 | - | 193.6 | - | |
| Limonene | 38 a | 5490.0 | 2.3 | 144.5 | 6.1 × 10−2 | |
| p-cymene | 1200 b | 1545.4 | 0.9 | 1.3 | 7.5 × 10−4 | |
| Eucalyptol | 12 c | 13,604.8 | - | 1133.7 | - | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González, D.; Guerra, N.; Colón, J.; Gabriel, D.; Ponsá, S.; Sánchez, A. Characterization of the Gaseous and Odour Emissions from the Composting of Conventional Sewage Sludge. Atmosphere 2020, 11, 211. https://doi.org/10.3390/atmos11020211
González D, Guerra N, Colón J, Gabriel D, Ponsá S, Sánchez A. Characterization of the Gaseous and Odour Emissions from the Composting of Conventional Sewage Sludge. Atmosphere. 2020; 11(2):211. https://doi.org/10.3390/atmos11020211
Chicago/Turabian StyleGonzález, Daniel, Nagore Guerra, Joan Colón, David Gabriel, Sergio Ponsá, and Antoni Sánchez. 2020. "Characterization of the Gaseous and Odour Emissions from the Composting of Conventional Sewage Sludge" Atmosphere 11, no. 2: 211. https://doi.org/10.3390/atmos11020211
APA StyleGonzález, D., Guerra, N., Colón, J., Gabriel, D., Ponsá, S., & Sánchez, A. (2020). Characterization of the Gaseous and Odour Emissions from the Composting of Conventional Sewage Sludge. Atmosphere, 11(2), 211. https://doi.org/10.3390/atmos11020211








