Comparison of Element Concentrations (Ba, Mn, Pb, Sr, Zn) in the Bones and Teeth of Wild Ruminants from the West Carpathians and the Tian-Shan Mountains as Indicators of Air Pollution
Abstract
1. Introduction
2. Experiments
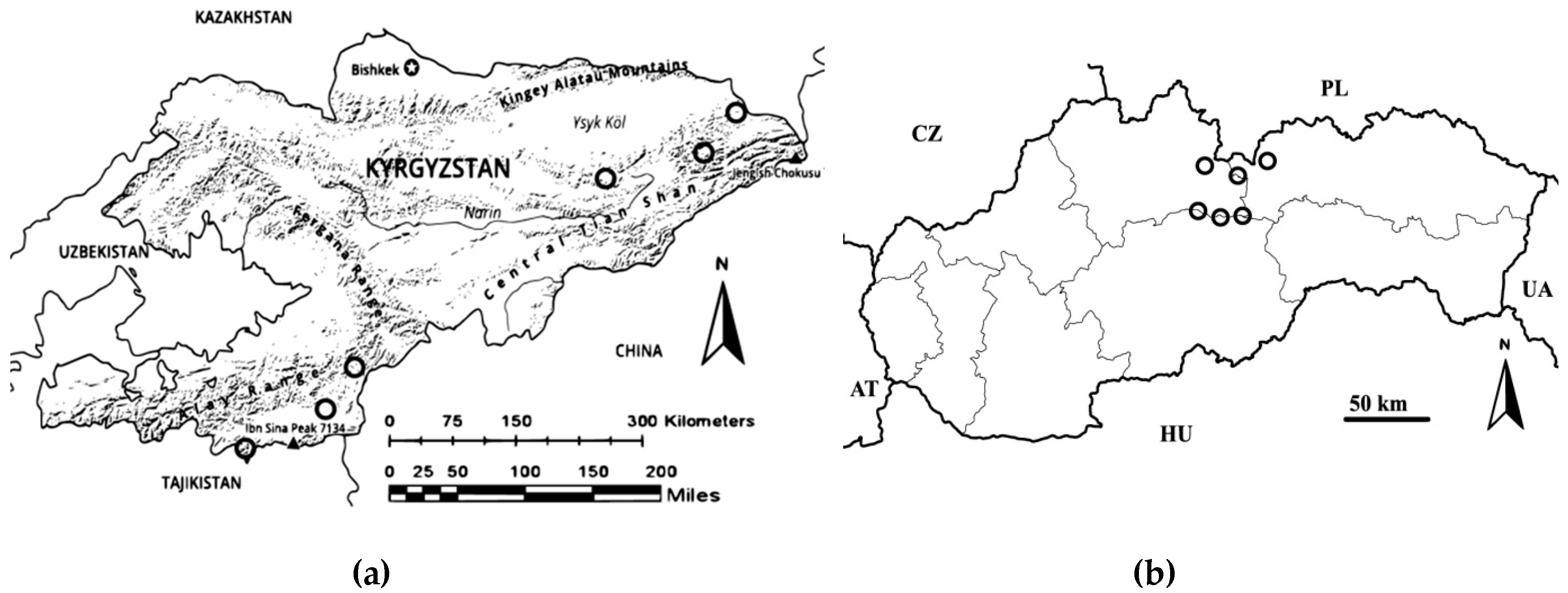
2.1. Sample Collection and Analyses
2.2. X-ray Fluorescence Spectrometry
2.3. Statistics
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Eichler, A.; Tobler, L.; Eyrikh, S.; Malygina, N.; Papina, T.; Schwikowski, M. Ice-core based assessment of historical anthropogenic heavy metal (Cd, Cu, Sb, Zn) emissions in the Soviet Union. Environ. Sci. Technol. 2014, 48, 2635–2642. [Google Scholar] [CrossRef] [PubMed]
- Grigholm, B.; Mayewski, P.A.; Aizen, V.; Kreutz, K.; Wake, C.P.; Aizen, E.; Kang, S.; Maasch, K.A.; Handley, M.J.; Sneed, S.B. Mid-twentieth century increases in anthropogenic Pb, Cd and Cu in central Asia set in hemispheric perspective using Tien Shan ice core. Atmos. Environ. 2016, 131, 17–28. [Google Scholar] [CrossRef]
- Zeng, H.; Wu, J.; Liu, W. Two-century sedimentary record of heavy metal pollution from lake Sayram: A deep mountain lake in central Tianshan, China. Quat. Int. 2014, 321, 125–131. [Google Scholar] [CrossRef]
- Wan, D.; Song, L.; Yang, J.; Jin, Z.; Zhan, C.; Mao, X.; Liu, D.; Shao, Y. Increasing heavy metals in the background atmosphere of central north China since the 1980s: Evidence from a 200-year lake sediment record. Atmos. Environ. 2016, 138, 183–190. [Google Scholar] [CrossRef]
- Steffen, W.; Persson, Å.; Deutsch, L.; Zalasiewicz, J.; Williams, M.; Richardson, K.; Crumley, C.; Crutzen, P.; Folke, C.; Gordon, L.; et al. The anthropocene: From global change to planetary stewardship. Ambio 2011, 40, 739–761. [Google Scholar] [CrossRef] [PubMed]
- Zhaoyong, Z.; Abuduwaili, J.; Fengqing, J. Heavy metal contamination, sources, and pollution assessment of surface water in the Tianshan mountains of China. Environ. Monit. Assess. 2015, 187, 33. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, Y.; Zhang, W.; Hong, S.; Hur, S.D.; Lee, K.; Pang, H.; Hou, S. High-resolution atmospheric cadmium record for ad 1776–2004 in a high-altitude ice core from the eastern Tien Shan, central Asia. Ann. Glaciol. 2016, 57, 265–272. [Google Scholar] [CrossRef]
- Ciriaková, A.; Mursaliev, N.; Šoltés, R.; Lukáň, M.; Janiga, M. Lead concentrations in soils and plants of two altitudinal transects in the eastern Kyrgyz Tian Shan mountains—A preliminary study. Oecol. Mont. 2011, 20, 19–26. [Google Scholar]
- Orozbaeva, K.D.; Kerimalieva, N.K.; Mombekov, S.T. Environment in the Kyrgyz Republic; National Statistical Committee of the Kyrgyz Republic: Bishkek, Kyrgyzstan, 2016; p. 118. [Google Scholar]
- Tataruch, F. Red deer antlers as biomonitors for lead contamination. Bull. Environ. Contam. Toxicol. 1995, 55, 332–337. [Google Scholar] [CrossRef]
- Khan, A.; Khan, S.; Khan, M.A.; Qamar, Z.; Waqas, M. The uptake and bioaccumulation of heavy metals by food plants, their effects on plants nutrients, and associated health risk: A review. Environ. Sci. Pollut. Res. 2015, 22, 13772–13799. [Google Scholar] [CrossRef]
- Li, R.; Bing, H.; Wu, Y.; Zhou, J.; Xiang, Z. Altitudinal patterns and controls of trace metal distribution in soils of a remote high mountain, southwest china. Environ. Geochem. Health 2018, 40, 505–519. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yu, Y.-Q.; Shi, L. Foraging and bedding site selection by Asiatic ibex (Capra sibirica) during summer in central Tianshan mountains. Pak. J. Zool. 2015, 47, 1–6. [Google Scholar]
- Reading, R.; Shank, C. Capra sibirica. The IUCN Red List of Threatened Species. 2008. e.T42398A10695735. Available online: https://www.iucnredlist.org/species/42398/10695735 (accessed on 11 April 2018).
- Yang, Y.; Wang, Y.; Zhao, Y.; Zhang, X.; Li, R.; Chen, L.; Zhang, G.; Jiang, Y.; Qiu, Q.; Wang, W. Draft genome of the Marco Polo sheep (Ovis ammon polii). GigaScience 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.B.; Reading, R. Ovis ammon. The IUCN Red List of Threatened Species. 2008. e.T15733A5074694. Available online: https://www.iucnredlist.org/species/15733/5074694 (accessed on 11 April 2018).
- Ballová, Z.; Janiga, M. Lead levels in the bones of small rodents from alpine and subalpine habitats in the Tian-shan mountains, Kyrgyzstan. Atmosphere 2018, 9, 35. [Google Scholar] [CrossRef]
- Janiga, M.; Hrehová, Z.; Dimitrov, K.; Gerasimova, C.; Lovari, S. Lead levels in the bones of snow voles Chionomys nivalis (Martins, 1842) (Rodentia) from European mountains: A comparative study of populations from the Tatra (Slovakia), Vitosha and Rila (Bulgaria). Acta Zool. Bulg. 2016, 68, 291–295. [Google Scholar]
- O’Neal, S.L.; Zheng, W. Manganese toxicity upon overexposure: A decade in review. Curr. Environ. Health Rep. 2015, 2, 315–328. [Google Scholar] [CrossRef] [PubMed]
- O’Neal, S.L.; Lee, J.W.; Zheng, W.; Cannon, J.R. Subacute manganese exposure in rats is a neurochemical model of early manganese toxicity. Neurotoxicology 2014, 44, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Roney, N.; Smith, V.C.; Willims, M.; Osier, M.; Paikoff, S.J. Toxicological Profile for Zinc; Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2005; p. 352. [Google Scholar]
- Plum, L.M.; Rink, L.; Haase, H. The essential toxin: Impact of zinc on human health. Int. J. Environ. Res. Public Health 2010, 7, 1342–1365. [Google Scholar] [CrossRef]
- Kravchenko, J.; Darrah, T.H.; Miller, R.K.; Lyerly, H.K.; Vengosh, A. A review of the health impacts of barium from natural and anthropogenic exposure. Environ. Geochem. Health 2014, 36, 797–814. [Google Scholar] [CrossRef]
- Demayo, A.; Taylor, M.C.; Taylor, K.W.; Hodson, P.V.; Hammond, P.B. Toxic effects of lead and lead compounds on human health, aquatic life, wildlife plants, and livestock. Crit. Rev. Environ. Control 1982, 12, 257–305. [Google Scholar] [CrossRef]
- Khandare, A.; Validandi, V.; Rao, S.; Dheeravath, S.; Nagalla, B. Synergistic effects of strontium and fluoride on nutritional status in guinea pigs (Cavia porcellus). Fluoride 2015, 48, 283. [Google Scholar]
- Mao, L.; Xia, L.; Chang, J.; Liu, J.; Jiang, L.; Wu, C.; Fang, B. The synergistic effects of Sr and Si bioactive ions on osteogenesis, osteoclastogenesis and angiogenesis for osteoporotic bone regeneration. Acta Biomater. 2017, 61, 217–232. [Google Scholar] [CrossRef] [PubMed]
- Priest, N.D.; Van de Vyver, F. Trace Metals and Fluoride in Bones and Teeth; CRC Press: Boca Raton, FL, USA; Ann Arbor, MI, USA; Boston, MA, USA, 1990. [Google Scholar]
- Martiniaková, M.; Omelka, R.; Jančová, A.; Stawarz, R.; Formicki, G. Concentrations of selected heavy metals in bones and femoral bone structure of bank (Myodes glareolus) and common (Microtus arvalis) voles from different polluted biotopes in Slovakia. Arch. Environ. Contam. Toxicol. 2011, 60, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Šoltés, R. Correlation between altitude and heavy metal deposition in the Tatra Mts (Slovakia). Biologia 1998, 53, 85–90. [Google Scholar]
- Parker, R.B.; Toots, H. Trace elements in bones as paleobiological indicators. In Fossils in the Making: Vertebrate Taphonomy and Paleoecology; Behrensmeyer, A.K., Hill, A.P., Eds.; The University of Chicago Press: Chicago, IL, USA; London, UK, 1988; p. 197. [Google Scholar]
- Shen, X.; Zhang, J.; Zhang, R. Phosphorus metabolic disorder of Guizhou semi-fine wool sheep. PLoS ONE 2014, 9, e89472. [Google Scholar] [CrossRef] [PubMed]
- Butler, O.T.; Cairns, W.R.L.; Cook, J.M.; Davidson, C.M. Atomic spectrometry update—A review of advances in environmental analysis. J. Anal. At. Spectrom. 2017, 32, 11–57. [Google Scholar] [CrossRef]
- Buddhachat, K.; Klinhom, S.; Siengdee, P.; Brown, J.L.; Nomsiri, R.; Kaewmong, P.; Thitaram, C.H.; Mahakkanukrauh, P.; Nganvongpanit, K. Elemental analysis of bone, teeth, horn and antler in different animal species using non-invasive handheld X-ray fluorescence. PLoS ONE 2016, 11, e0155458. [Google Scholar] [CrossRef] [PubMed]
- Conostan Calibration Standard. 316 Stainless Steel Alloy. Innov-X Systems: Waltham, MA, USA, 2011. [Google Scholar]
- Wise, S.A.; Watters, R.L., Jr. Certificate of Analysis Standard Reference Material 2710a. Montana I Soil, National Institute of Standards and Technology: Gaithersburg, MD, USA, 2009. Available online: https://www-s.nist.gov/srmors/view_detail.cfm?srm=2710A (accessed on 22 May 2009).
- Wise, S.A.; Watters, R.L., Jr. Certificate of Analysis Standard Reference Material 2711a. Montana II Soil, National Institute of Standards and Technology: Gaithersburg, MD, USA, 2009. Available online: https://www-s.nist.gov/srmors/view_detail.cfm?srm=2711A (accessed on 22 May 2009).
- Haizhou, W. Certificate of Certified Reference Material NCS ZC 71001. China National Analysis Center for Iron and Steel: Beijing, China, 2015. Available online: http://gsometal.ru/Catalogues%202011/NCS.pdf (accessed on 22 October 2018).
- Janiga, M.; Ballová, Z.; Angelovičová, M.; Korňan, J. The snow vole and Tatra marmot as different rodent bioindicators of lead pollution in an alpine environment: A hibernation effect. Pol. J. Environ. Stud. 2018, in press. [Google Scholar]
- Innov-X-Systems. Delta HHXRF Analyzers. Limits of Detection (LODs); Innov-X Systems, Inc.: Woburn, MA, USA; Available online: http://www.xrfrentals.com/images/documents/delta_detectable_elememts.pdf (accessed on 28 June 2018).
- Spears, J.W. Trace mineral bioavailability in ruminants. J. Nutr. 2003, 133, 1506S–1509S. [Google Scholar] [CrossRef]
- Aschner, J.L.; Aschner, M. Nutritional aspects of manganese homeostasis. Mol. Asp. Med. 2005, 26, 353–362. [Google Scholar] [CrossRef]
- Röllin, H.; Nogueira, C.M.C.A. Manganese: Environmental pollution and health effects. In Encyclopedia of Environmental Health; Elsevier: Burligton, NJ, USA, 2011; pp. 617–629. [Google Scholar]
- Szarlowicz, K.; Reczynski, W.; Misiak, R.; Kubica, B. Radionuclides and heavy metal concentrations as complementary tools for studying the impact of industrialization on the environment. J. Radioanal. Nucl. Chem. 2013, 298, 1323–1333. [Google Scholar] [CrossRef] [PubMed]
- Gusev, A.; Ilyin, I.; Travnikov, O.; Sokovych, V. Model Assessment of Transboundary Pollution by Lead and Pcb-153 of the Central Asian Countries: Kazakhstan, Kyrgyzstan, Tajikistan, Turkmenistan, Uzbekistan. EMEP/MSC-E Contribution to the UN ECE CAPACT Project; Meteorological Synthesizing Centre-East: Moscow, Russia, 2007; Available online: http://www.en.msceast.org/reports/6_2007.pdf (accessed on 7 August 2018).
- Monaci, F.; Bargagli, R. Barium and other trace metals as indicators of vehicle emissions. Water Air Soil Pollut. 1997, 100, 89–98. [Google Scholar] [CrossRef]
- Feng, X.; Li, G.; Qiu, G. A preliminary study on mercury contamination to the environment from artisanal zinc smelting using indigenous methods in Hezhang county, Guizhou, China—Part 1: Mercury emission from zinc smelting and its influences on the surface waters. Atmos. Environ. 2004, 38, 6223–6230. [Google Scholar] [CrossRef]
- Bi, X.; Feng, X.; Yang, Y.; Qiu, G.; Li, G.; Li, F.; Liu, T.; Fu, Z.; Jin, Z. Environmental contamination of heavy metals from zinc smelting areas in Hezhang county, western Guizhou, China. Environ. Int. 2006, 32, 883–890. [Google Scholar] [CrossRef] [PubMed]
- Janiga, M.; Hrehová, Z.; Kostková-Zelinová, V. Seasonal effects of lead uptake by snow vole Chionomys nivalis (Martins, 1842) in West Tatra Mts.: Bone metal concentrations and hematological indices. Pol. J. Ecol. 2012, 60, 611–619. [Google Scholar]
- Rybicka, E.H. Environmental impact of mining and smelting industries in Poland. Geol. Soc. Lond. Spec. Publ. 1996, 113, 183–193. [Google Scholar] [CrossRef]
- Stefanowicz, A.; Woch, M.; Kapusta, P. Soils from sites of historical metal mining in western Małopolska (S Poland) are strongly contaminated with Zn, Pb and Cd. E3S Web Conf. 2013. [Google Scholar] [CrossRef]
- Steblez, W.G. The Mineral industries of Central Europe. The Czech Republic, Hungary, Poland, and Slovakia. USGS Miner. Yearb. 2005, 3, 1–26. [Google Scholar]
- Angelovičová, M.; Janiga, M. Heavy metals and some other elements in the teeth of the Tatra marmot (Marmota marmota latirostris). Oecol. Mont. 2017, 26, 34–41. [Google Scholar]
- Ftáčniková, V.; Némethy, M. Bio-indication of environmental pollution in alpine environments using X-ray analysis in snow vole (Chionomys nivalis) population. Oecol. Mont. 2017, 26, 12–18. [Google Scholar]
- Stake, P.; Miller, W.; Gentry, R.; Neathery, M. Zinc metabolic adaptations in calves fed a high but nontoxic zinc level for varying time periods 1. J. Anim. Sci. 1975, 40, 132–137. [Google Scholar] [CrossRef]
- Suttle, N.F. Mineral Nutrition of Livestock, 4th ed.; MPG Books Group: London, UK, 2010. [Google Scholar]
- Martiniaková, M.; Omelka, R.; Jančová, A.; Formicki, G.; Stawarz, R.; Bauerová, M. Accumulation of risk elements in kidney, liver, testis, uterus and bone of free-living wild rodents from a polluted area in Slovakia. J. Environ. Sci. Health Part A 2012, 47, 1202–1206. [Google Scholar] [CrossRef] [PubMed]
- Jabłońska-Czapla, M.; Nocoń, K.; Szopa, S.; Łyko, A. Impact of the Pb and Zn ore mining industry on the pollution of the Biała Przemsza River, Poland. Environ. Monit. Assess. 2016, 188, 262. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, L.; Li, Y.; Li, H.; Wang, W.; Ye, B. Impacts of lead/zinc mining and smelting on the environment and human health in China. Environ. Monit. Assess. 2012, 184, 2261–2273. [Google Scholar] [CrossRef] [PubMed]
- Martiniaková, M.; Omelka, R.; Jančová, A.; Stawarz, R.; Formicki, G. Heavy metal content in the femora of yellow-necked mouse (Apodemus flavicollis) and wood mouse (Apodemus sylvaticus) from different types of polluted environment in Slovakia. Environ. Monit. Assess. 2010, 171, 651–660. [Google Scholar] [CrossRef]
- Monaci, F.; Moni, F.; Lanciotti, E.; Grechi, D.; Bargagli, R. Biomonitoring of airborne metals in urban environments: New tracers of vehicle emission, in place of lead. Environ. Pollut. 2000, 107, 321–327. [Google Scholar] [CrossRef]
- Bentley, R.A. Strontium isotopes from the earth to the archaeological skeleton: A review. J. Archaeol. Method Theory 2006, 13, 135–187. [Google Scholar] [CrossRef]
- Kostial, K.; Lutkić, A.; Gruden, N.; Vojvodić, S.; Harrison, G.E. The effect of dietary phosphorus on the metabolism of calcium and strontium in the rat. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1963, 6, 431–439. [Google Scholar] [CrossRef]
- Sivertsen, T.; Daae, H.L.; Godal, A.; Sand, G. Ruminant uptake of nickel and other elements from industrial air pollution in the norwegigan-russian border area. Environ. Pollut. 1995, 90, 75–81. [Google Scholar] [CrossRef]
- Klamárová, S.; Solár, J. Spatial distribution of elements in soils of experimental area, Ružomberok—X-ray analysis. Oecol. Mont. 2017, 26, 1–14. [Google Scholar]
- Sendecká, M.; Šoltés, R. Seasonal changes in the dust nuisance and contamination of mosses in the experimental study area Ružomberok. Oecol. Mont. 2017, 26, 24–34. [Google Scholar]
- Böhmová, P.; Šoltés, R. Accumulation of selected element deposition in the organs of Fallopia japonica during ontogeny. Oecol. Mont. 2017, 26, 35–46. [Google Scholar]
- Grešíková, S.; Janiga, M. Analysis of S, Cl, K, Ca, Cr, Mn, Fe, Zn, Rb, Sr, Mo, Ba and Pb concentrations in the needles of Abies alba and potential impact of paper mill industry. Oecol. Mont. 2017, 26, 47–55. [Google Scholar]
- Sobota, S.; Baranowska-Bosiacka, I.; Gutowska, I.; Kupiec, M.; Dusza, K.; Machoy, Z.; Chlubek, D. Biomonitoring of lead and fluoride contamination in forests using chemical analysis of hard tissues of roe deer (Capreolus capreolus L.). Pol. J. Environ. Stud. 2011, 20, 435–443. [Google Scholar]
- Turner-Walker, G. The chemical and microbial degradation of bones and teeth. Adv. Hum. Palaeopathol. 2008, 3–29. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1002/9780470724187.ch1 (accessed on 5 November 2018).
- Kasem, M.A.; Russo, R.E.; Harith, M.A. Influence of biological degradation and environmental effects on the interpretation of archeological bone samples with laser-induced breakdown spectroscopy. J. Anal. At. Spectrom. 2011, 26, 1733–1739. [Google Scholar] [CrossRef]
- Demesko, J.; Markowski, J.; Słaba, M.; Hejduk, J.; Minias, P. Age-related patterns in trace element content vary between bone and teeth of the European roe deer (Capreolus capreolus). Arch. Environ. Contam. Toxicol. 2018, 74, 330–338. [Google Scholar] [CrossRef] [PubMed]

| Element (ppm) | N | Mean | Median | Minimum | Maximum | SD |
|---|---|---|---|---|---|---|
| Ba | 13 | 101.08 | 96.00 | 54.00 | 211.00 | 41.22 |
| Pb | 1 | 8.00 | 8.00 | 8.00 | 8.00 | - |
| Sr | 38 | 200.61 | 187.50 | 129.00 | 593.00 | 77.75 |
| Zn | 32 | 57.53 | 37.50 | 14.00 | 253.00 | 54.62 |
| Mn | 20 | 82.45 | 62.50 | 17.00 | 377.00 | 83.12 |
| Element (ppm) | N | Mean | Median | Minimum | Maximum | SD |
|---|---|---|---|---|---|---|
| Ba | 19 | 99.79 | 85.00 | 52.00 | 199.00 | 40.27 |
| Pb | 17 | 15.12 | 9.00 | 8.00 | 40.00 | 10.97 |
| Sr | 30 | 156.47 | 140.00 | 55.00 | 711.00 | 112.99 |
| Zn | 30 | 175.73 | 143.00 | 53.00 | 470.00 | 93.95 |
| Mn | 29 | 167.34 | 125.00 | 20.00 | 764.00 | 163.20 |
| Sample Type | Mn (n = 68) | Ba (n = 68) | Pb (n = 68) | Sr (n = 68) | Zn (n = 68) |
|---|---|---|---|---|---|
| WC positive | (29) 42.6% | (19) 27.9% | (17) 25.0% | (30) 44.1% | (30) 44.1% |
| TS positive | (20) 29.4% | (13) 19.1% | (1) 1.5% | (38) 55.9% | (32) 47.1% |
| WC negative | (1) 1.5% | (11) 16.2% | (13) 19.1% | (0) 0.0% | (0) 0.000% |
| TS negative | (18) 26.5% | (25) 36.8% | (37) 54.4% | (0) 0.0% | (6) 8.8% |
| Chi-square WC/TS (df = 1) | 16.15, p = 0.0001 | 5.71, p = 0.0169 | 25.15, p = 0.000 | - | p = 0.0252 1 |
| Element | t-Value | df | p | N(TS) | N(WC) | F-Ratio | p |
|---|---|---|---|---|---|---|---|
| Ba | 0.09 | 30 | 0.9305 | 13 | 19 | 1.05 | 0.9019 |
| Pb | −0.63 | 16 | 0.5372 | 1 | 17 | 0.00 | 1.0000 |
| Sr | 1.91 | 66 | 0.0611 | 38 | 30 | 2.11 | 0.0326 * |
| Zn | −6.10 | 60 | 0.0000 *** | 32 | 30 | 2.96 | 0.0038 *** |
| Mn | −2.14 | 47 | 0.0377 ** | 20 | 29 | 3.86 | 0.0034 *** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ballová, Z.; Janiga, M.; Hančinský, R. Comparison of Element Concentrations (Ba, Mn, Pb, Sr, Zn) in the Bones and Teeth of Wild Ruminants from the West Carpathians and the Tian-Shan Mountains as Indicators of Air Pollution. Atmosphere 2019, 10, 64. https://doi.org/10.3390/atmos10020064
Ballová Z, Janiga M, Hančinský R. Comparison of Element Concentrations (Ba, Mn, Pb, Sr, Zn) in the Bones and Teeth of Wild Ruminants from the West Carpathians and the Tian-Shan Mountains as Indicators of Air Pollution. Atmosphere. 2019; 10(2):64. https://doi.org/10.3390/atmos10020064
Chicago/Turabian StyleBallová, Zuzana, Marián Janiga, and Richard Hančinský. 2019. "Comparison of Element Concentrations (Ba, Mn, Pb, Sr, Zn) in the Bones and Teeth of Wild Ruminants from the West Carpathians and the Tian-Shan Mountains as Indicators of Air Pollution" Atmosphere 10, no. 2: 64. https://doi.org/10.3390/atmos10020064
APA StyleBallová, Z., Janiga, M., & Hančinský, R. (2019). Comparison of Element Concentrations (Ba, Mn, Pb, Sr, Zn) in the Bones and Teeth of Wild Ruminants from the West Carpathians and the Tian-Shan Mountains as Indicators of Air Pollution. Atmosphere, 10(2), 64. https://doi.org/10.3390/atmos10020064





