Correction of a Splicing Mutation Affecting an Unverricht-Lundborg Disease Patient by Antisense Therapy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Biological Material
2.2. Cycloheximide Treatment of Control and Patient Fibroblasts
2.3. Antisense Oligonucleotide Treatment and Analysis
2.4. CSTB Protein Immunodetection
3. Results
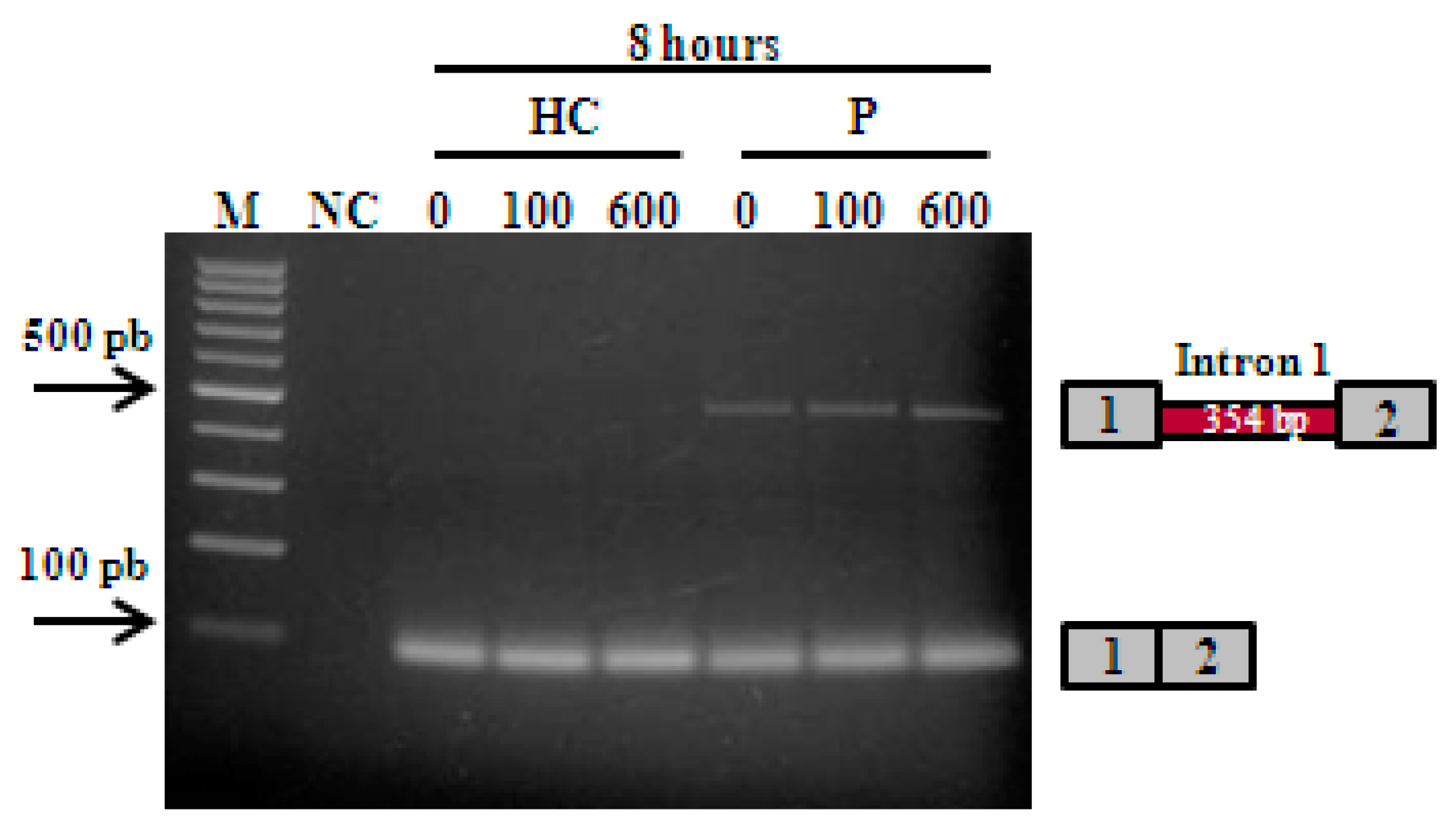
3.1. The CSTB c.66G>A Mutation Generates One Aberrant Transcript in Unverricht-Lundborg Disease Patient
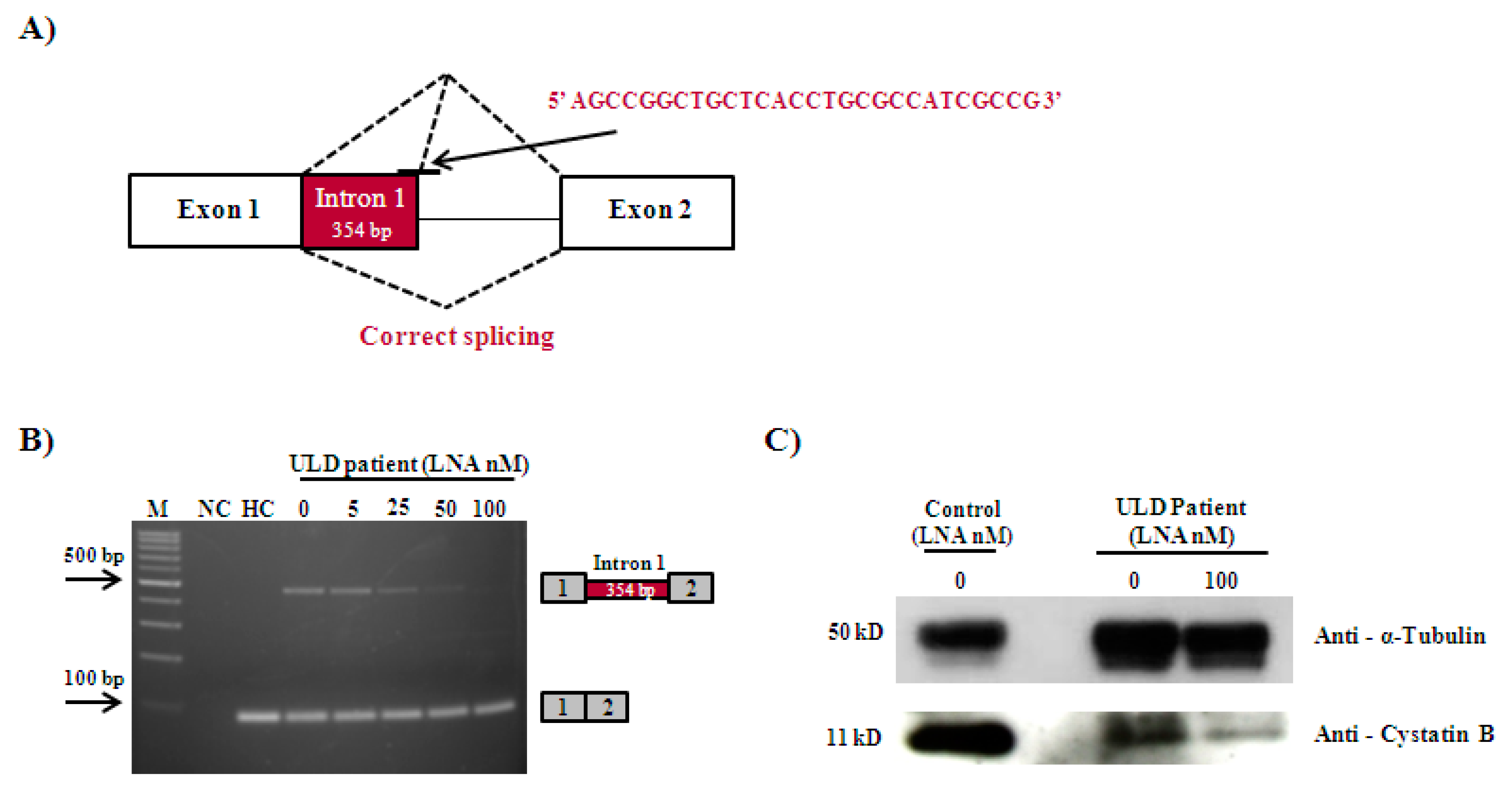
3.2. Correction of CSTB Mutation-Induced Missplicing Using an Locked Nucleic Acid Oligonucleotide in Patient-Derived Fibroblasts
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Merwick, A.; O’Brien, M.; Delanty, N. Complex single gene disorders and epilepsy. Epilepsia 2012, 53, 81–91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kälviäinen, R.; Khyuppenen, J.; Koskenkorva, P.; Eriksson, K.; Vanninen, R.; Mervaala, E. Clinical picture of EPM1-Unverricht-Lundborg disease. Epilepsia 2008, 49, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Genton, P. Unverricht-Lundborg disease (EPM1). Epilepsia 2010, 51, 37–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lehesjoki, A.E.; Koskiniemi, M.; Sistonen, P.; Miao, J.; Hästbacka, J.; Norio, R.; de la Chapelle, A. Localization of a gene for progressive myoclonus epilepsy to chromosome 21q22. Proc. Natl. Acad. Sci. USA 1991, 88, 3696–3699. [Google Scholar] [CrossRef] [PubMed]
- Turk, V.; Bode, W. The cystatins: Protein inhibitors of cysteine proteinases. FEBS Lett. 1991, 285, 213–219. [Google Scholar] [CrossRef] [Green Version]
- Crespel, A.; Ferlazzo, E.; Franceschetti, S.; Genton, P.; Gouider, R.; Kälviäinen, R.; Korja, M.; Lehtinen, M.K.; Mervaala, E.; Simonato, M.; et al. Unverricht-Lundborg disease. Epileptic Disord. 2016, 18, 28–37. [Google Scholar] [PubMed]
- Lalioti, M.D.; Scott, H.S.; Buresi, C.; Rossier, C.; Bottani, A.; Morris, M.A.; Malafosse, A.; Antonarakis, S.E. Dodecamer repeat expansion in cystatin B gene in progressive myoclonus epilepsy. Nature 1997, 386, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Joensuu, T.; Lehesjoki, A.E.; Kopra, O. Molecular background of EPM1-Unverricht-Lundborg disease. Epilepsia 2008, 49, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Koo, T.; Wood, M.J. Clinical trials using antisense oligonucleotides in Duchenne muscular dystrophy. Hum. Gene Ther. 2013, 24, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Mendell, J.R.; Rodino-Klapac, L.R.; Sahenk, Z.; Roush, K.; Bird, L.; Lowes, L.P.; Alfano, L.; Gomez, A.M.; Lewis, S.; Kota, J.; et al. Eteplirsen Study Group. Eteplirsen for the treatment of Duchenne muscular dystrophy. Ann. Neurol. 2013, 74, 637–647. [Google Scholar] [PubMed]
- Aartsma-Rus, A.; Krieg, A.M. FDA approves eteplirsen for Duchenne muscular dystrophy: The next chapter in the eteplirsen saga. Nucleic Acid Ther. 2017, 27, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Rigo, F.; Hua, Y.; Krainer, A.R.; Bennett, C.F. Antisense-based therapy for the treatment of spinal muscular atrophy. J. Cell Biol. 2012, 199, 21–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiriboga, C.A.; Swoboda, K.J.; Darras, B.T.; Iannaccone, S.T.; Montes, J.; De Vivo, D.C.; Norris, D.A.; Bennett, C.F.; Bishop, K.M. Results from a phase 1 study of nusinersen (ISIS-SMN(Rx)) in children with spinal muscular atrophy. Neurology 2016, 86, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Ottesen, E.W. ISS-N1 makes the first FDA-approved drug for spinal muscular atrophy. Transl. Neurosci. 2017, 8, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.M.; Nobumori, C.; Tu, Y.; Choi, C.; Yang, S.H.; Jung, H.J.; Vickers, T.A.; Rigo, F.; Bennett, C.F.; Young, S.G.; et al. Modulation of LMNA splicing as a strategy to treat prelamin A diseases. J. Clin. Investig. 2016, 126, 1592–1602. [Google Scholar] [CrossRef] [PubMed]
- Pérez, B.; Gutiérrez-Solana, L.G.; Verdú, A.; Merinero, B.; Yuste-Checa, P.; Ruiz-Sala, P.; Calvo, R.; Jalan, A.; Marín, L.L.; Campos, O.; et al. Clinical, biochemical, and molecular studies in pyridoxine-dependent epilepsy. Antisense therapy as possible new therapeutic option. Epilepsia 2013, 54, 239–248. [Google Scholar] [PubMed]
- Chauhan, N.B.; Siegel, G.J. Antisense inhibition at the beta-secretase-site of beta-amyloid precursor protein reduces cerebral amyloid and acetyl cholinesterase activity in Tg2576. Neuroscience 2007, 146, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Kalbfuss, B.; Mabon, S.A.; Misteli, T. Correction of alternative splicing of tau in frontotemporal dementia and parkinsonism linked to chromosome 17. J. Biol. Chem. 2001, 276, 42986–42993. [Google Scholar] [CrossRef] [PubMed]
- Kay, C.; Skotte, N.H.; Southwell, A.L.; Hayden, M.R. Personalized gene silencing therapeutics for Huntington disease. Clin. Genet. 2014, 86, 29–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magen, I.; Hornstein, E. Oligonucleotide-based therapy for neurodegenerative diseases. Brain Res. 2014, 1584, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Schoch, K.M.; Miller, T.M. Antisense oligonucleotides: translation from mouse models to human neurodegenerative diseases. Neuron 2017, 94, 1056–1070. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.; Freitas, J.; Duarte, A.J.; Ribeiro, I.; Ribeiro, D.; Lima, J.L.; Chaves, J.; Amaral, O. Unverricht-Lundborg disease: Homozygosity for a new splicing mutation in the cystatin B gene. Epilepsy Res. 2012, 99, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.J.; Ribeiro, D.; Chaves, J.; Amaral, O. Characterization of a rare Unverricht-Lundborg disease mutation. Mol. Genet. Metab. Rep. 2015, 4, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Hammond, S.M.; Wood, M.J. Genetic therapies for RNA mis-splicing diseases. Trends Genet. 2011, 27, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Douglas, A.G.; Wood, M.J. RNA splicing: Disease and therapy. Brief. Funct. Genom. 2011, 10, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Suñé-Pou, M.; Prieto-Sánchez, S.; Boyero-Corral, S.; Moreno-Castro, C.; El Yousfi, Y.; Suñé-Negre, J.M.; Hernández-Munain, C.; Suñé, C. Targeting splicing in the treatment of human disease. Genes 2017, 8, 87. [Google Scholar] [CrossRef] [PubMed]
- Arechavala-Gomeza, V.; Khoo, B.; Aartsma-Rus, A. Splicing modulation therapy in the treatment of genetic diseases. Appl. Clin. Genet. 2014, 7, 245–252. [Google Scholar] [PubMed]
- Rinaldi, C.; Wood, M.J.A. Antisense oligonucleotides: The next frontier for treatment of neurological disorders. Nat. Rev. Neurol. 2018, 14, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Havens, M.A.; Duelli, D.M.; Hastings, M.L. Targeting RNA splicing for disease therapy. Wiley Interdiscip. Rev. 2013, 4, 247–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeVos, S.L.; Miller, T.M. Antisense oligonucleotides: Treating neurodegeneration at the level of RNA. Neurotherapeutics 2013, 10, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Polajnar, M.; Ceru, S.; Kopitar-Jerala, N.; Zerovnik, E. Human stefin B normal and patho-physiological role: Molecular and cellular aspects of amyloid-type aggregation of certain EPM1 mutants. Front. Mol. Neurosci. 2012, 5, 88. [Google Scholar] [CrossRef] [PubMed]
- Polajnar, M.; Zavašnik-Bergant, T.; Kopitar-Jerala, N.; Tušek-Žnidarič, M.; Zerovnik, E. Gain in toxic function of stefin B EPM1 mutants aggregates: Correlation between cell death, aggregate number/size and oxidative stress. Biochim. Biophys. Acta 2014, 1843, 2089–2099. [Google Scholar] [CrossRef] [PubMed]
- Koskenkorva, P.; Hyppönen, J.; Aikiä, M.; Mervaala, E.; Kiviranta, T.; Eriksson, K.; Lehesjoki, A.E.; Vanninen, R.; Kälviäinen, R. Severer phenotype in Unverricht-Lundborg disease (EPM1) patients compound heterozygous for the dodecamer repeat expansion and the c.202C>T mutation in the CSTB gene. Neurodegener. Dis. 2011, 8, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Canafoglia, L.; Gennaro, E.; Capovilla, G.; Gobbi, G.; Boni, A.; Beccaria, F.; Viri, M.; Michelucci, R.; Agazzi, P.; Assereto, S.; et al. Electroclinical presentation and genotype-phenotype relationships in patients with Unverricht-Lundborg disease carrying compound heterozygous CSTB point and indel mutations. Epilepsia 2012, 53, 2120–2127. [Google Scholar] [CrossRef] [PubMed]
- Assenza, G.; Benvenga, A.; Gennaro, E.; Tombini, M.; Campana, C.; Assenza, F.; Di Pino, G.; Di Lazzaro, V. A novel c132-134del mutation in Unverricht-Lundborg disease and the review of literature of heterozygous compound patients. Epilepsia 2017, 58, e31–e35. [Google Scholar] [CrossRef] [PubMed]
- Brasil, S.; Viecelli, H.M.; Meili, D.; Rassi, A.; Desviat, L.R.; Pérez, B.; Ugarte, M.; Thöny, B. Pseudoexon exclusion by antisense therapy in 6-pyruvoyl-tetrahydropterin synthase deficiency. Hum. Mutat. 2011, 32, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Osorio, F.G.; Navarro, C.L.; Cadiñanos, J.; López-Mejía, I.C.; Quirós, P.M.; Bartoli, C.; Rivera, J.; Tazi, J.; Guzmán, G.; Varela, I.; et al. Splicing-directed therapy in a new mouse model of human accelerated aging. Sci. Transl. Med. 2011, 3, 106ra107. [Google Scholar] [CrossRef] [PubMed]
- Miller, T.M.; Pestronk, A.; David, W.; Rothstein, J.; Simpson, E.; Appel, S.H.; Andres, P.L.; Mahoney, K.; Allred, P.; Alexander, K.; et al. An antisense oligonucleotide against SOD1 delivered intrathecally for patients with SOD1 familial amyotrophic lateral sclerosis: A phase 1, randomised, first-in-man study. Lancet Neurol. 2013, 12, 435–442. [Google Scholar] [CrossRef]
- Regis, S.; Corsolini, F.; Grossi, S.; Tappino, B.; Cooper, D.N.; Filocamo, M. Restoration of the normal splicing pattern of the PLP1 Gene by means of an antisense oligonucleotide directed against an exonic mutation. PLoS ONE 2013, 8, e73633. [Google Scholar] [CrossRef] [PubMed]
- Bestas, B.; Moreno, P.M.; Blomberg, K.E.; Mohammad, D.K.; Saleh, A.F.; Sutlu, T.; Nordin, J.Z.; Guterstam, P.; Gustafsson, M.O.; Kharazi, S.; et al. Splice-correcting oligonucleotides restore BTK function in X-linked agammaglobulinemia model. J. Clin. Investig. 2014, 124, 4067–4081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Juliano, R.L. The delivery of therapeutic oligonucleotides. Nucleic Acids Res. 2016, 44, 6518–6548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.A.J.; Yokota, T. Antisense Therapy in Neurology. J. Pers. Med. 2013, 3, 144–176. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matos, L.; Duarte, A.J.; Ribeiro, D.; Chaves, J.; Amaral, O.; Alves, S. Correction of a Splicing Mutation Affecting an Unverricht-Lundborg Disease Patient by Antisense Therapy. Genes 2018, 9, 455. https://doi.org/10.3390/genes9090455
Matos L, Duarte AJ, Ribeiro D, Chaves J, Amaral O, Alves S. Correction of a Splicing Mutation Affecting an Unverricht-Lundborg Disease Patient by Antisense Therapy. Genes. 2018; 9(9):455. https://doi.org/10.3390/genes9090455
Chicago/Turabian StyleMatos, Liliana, Ana Joana Duarte, Diogo Ribeiro, João Chaves, Olga Amaral, and Sandra Alves. 2018. "Correction of a Splicing Mutation Affecting an Unverricht-Lundborg Disease Patient by Antisense Therapy" Genes 9, no. 9: 455. https://doi.org/10.3390/genes9090455







