CD99: A Cell Surface Protein with an Oncojanus Role in Tumors
Abstract
1. Introduction
- The opposite, dual functions of CD99 isoforms.
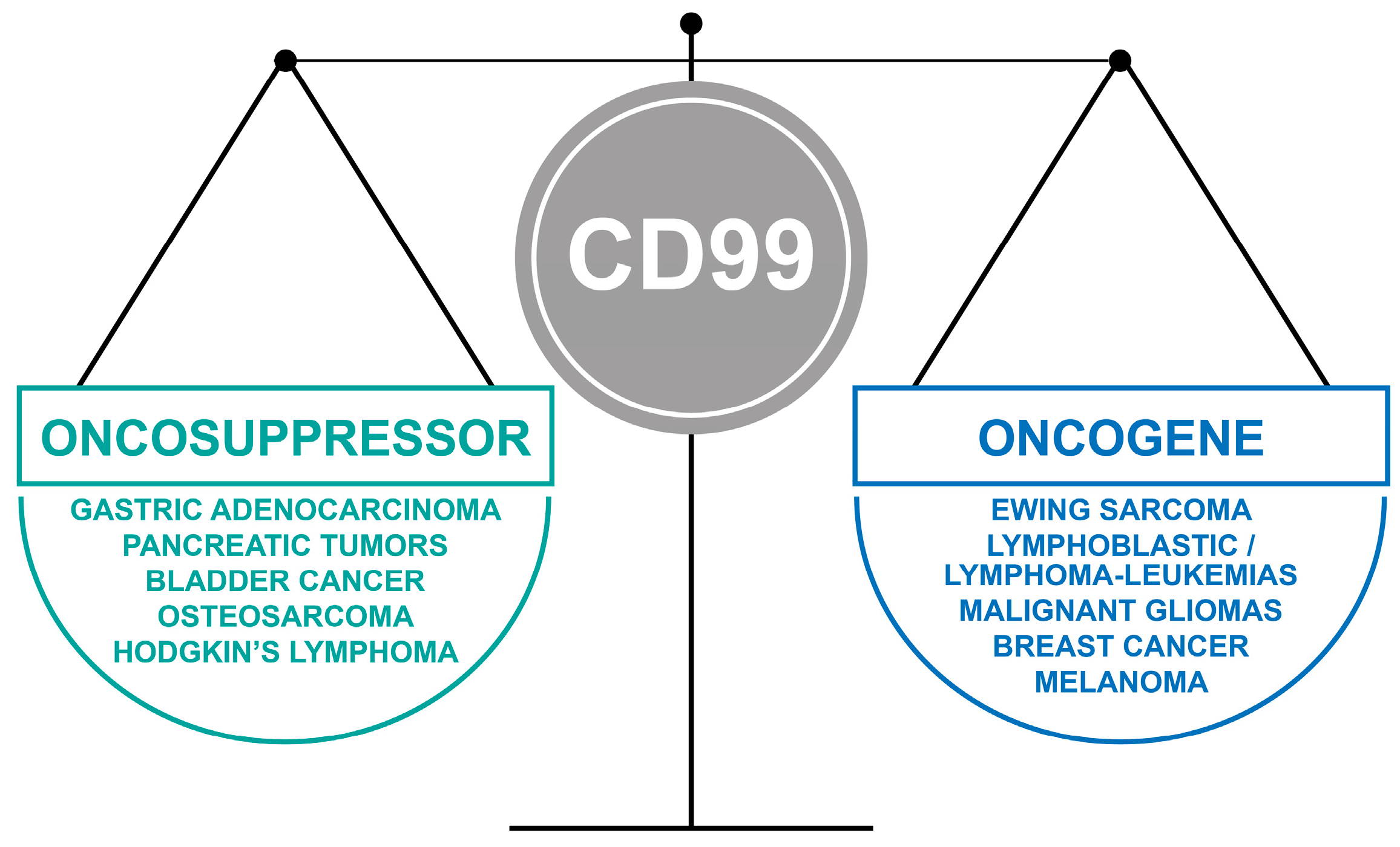
- The oncojanus function of CD99 in tumors (Figure 2).
- The therapeutic potential of strategies targeting CD99.
- Critical issues and novel perspectives.
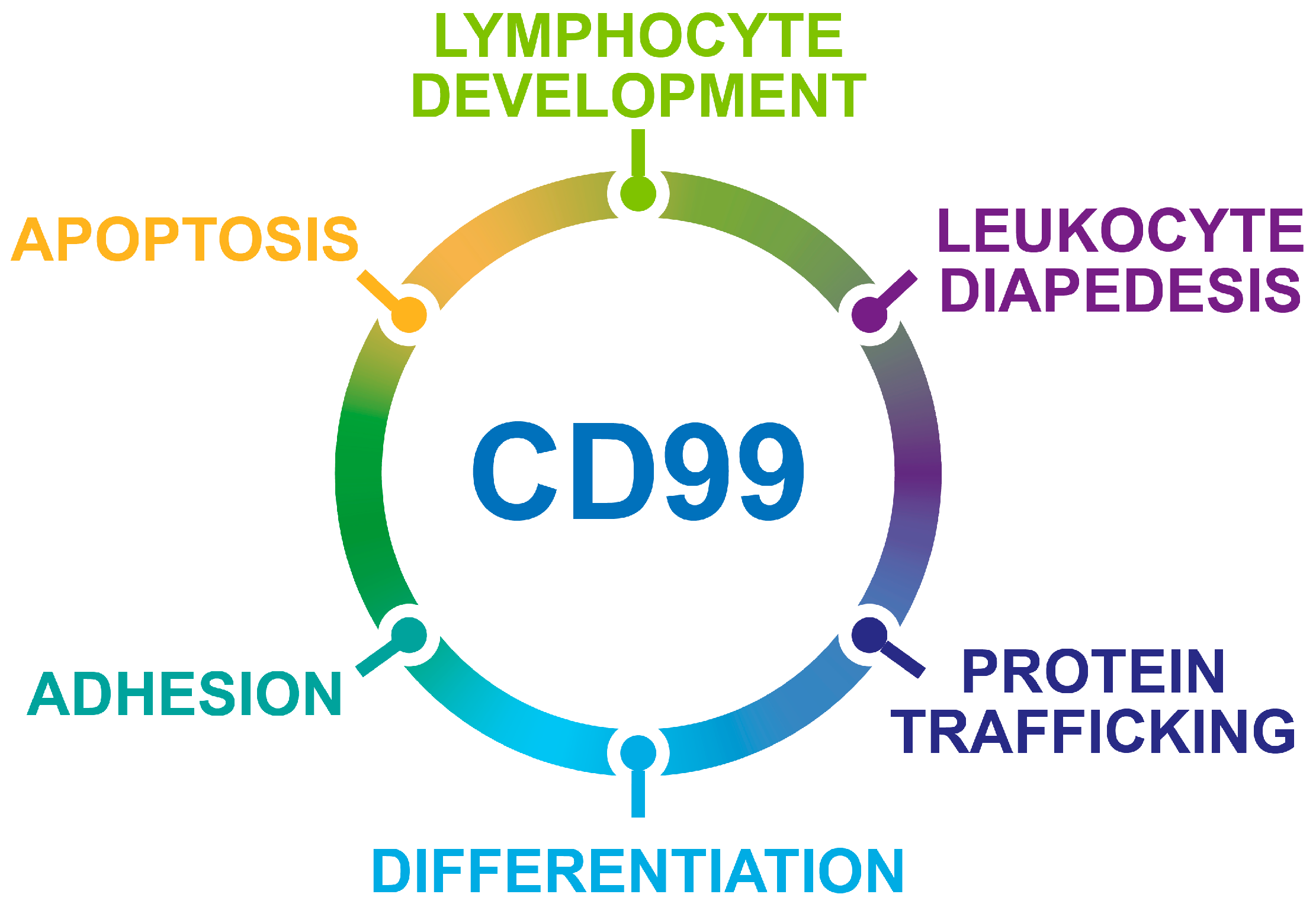
2. CD99 Structure and Expression in Normal Tissues
3. CD99 Isoforms Dictate Opposite Functions in Physiology and Malignancy
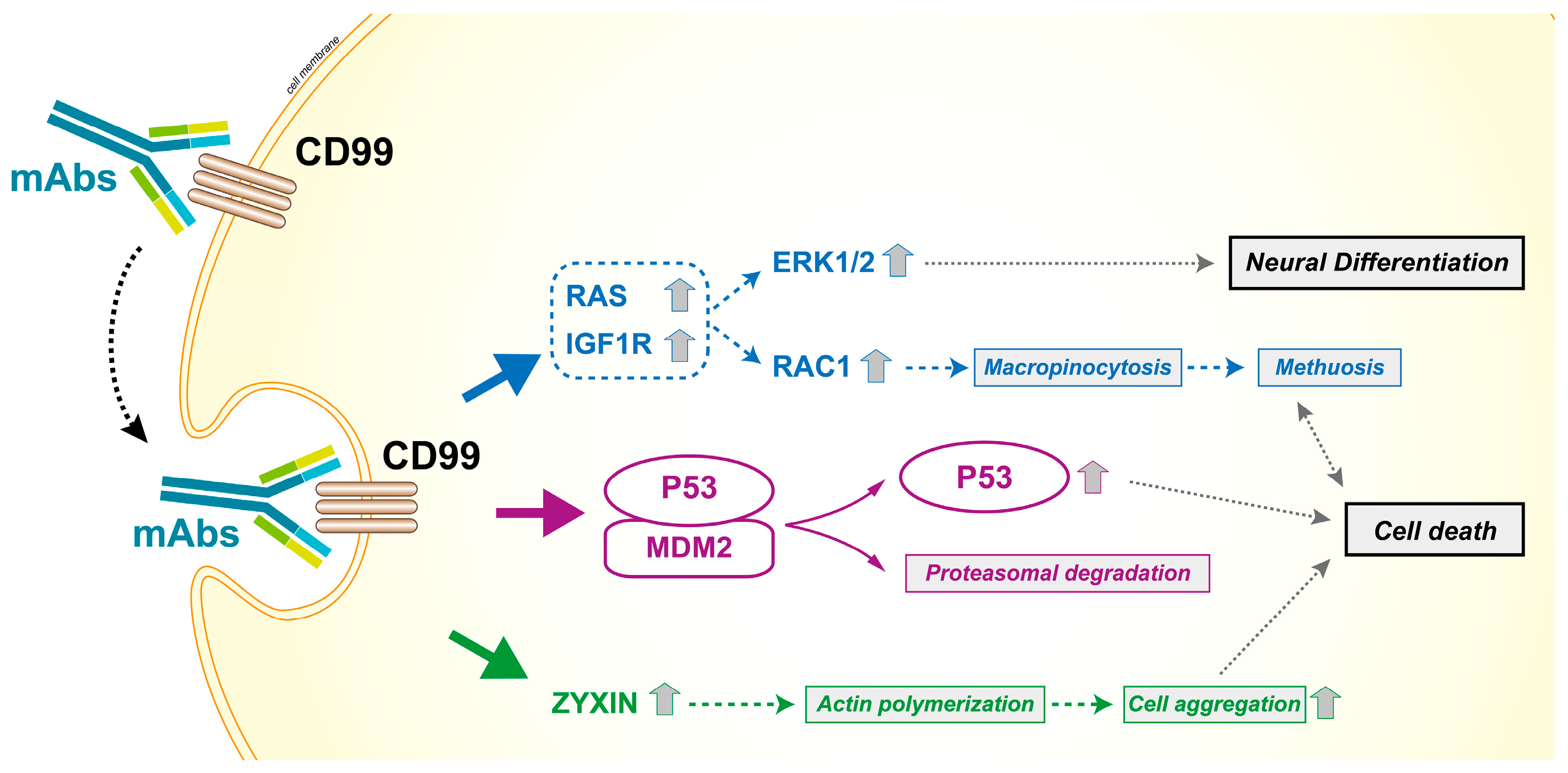
4. When CD99 Is Associated with Malignancy and Highly Expressed
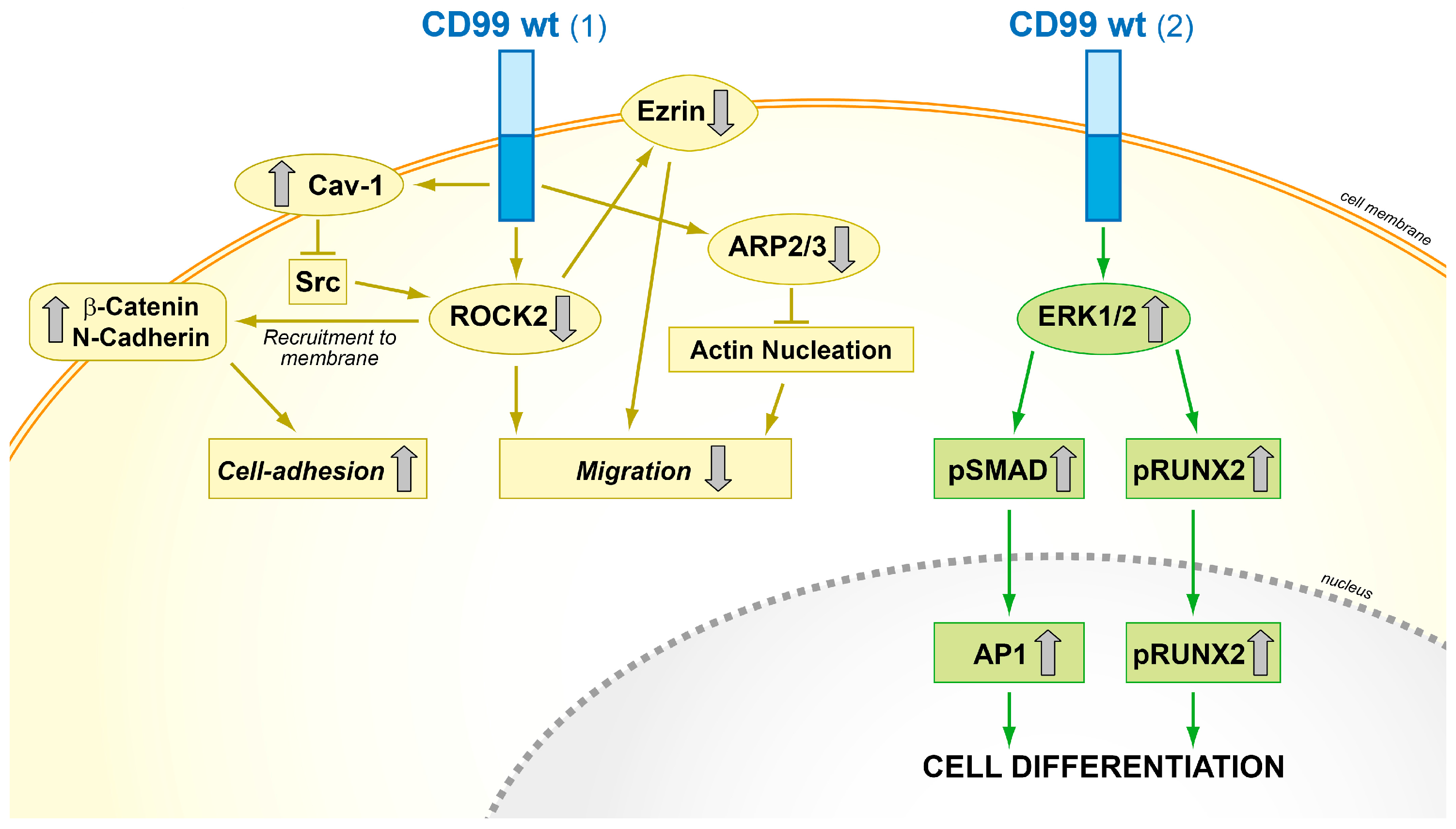
5. When CD99 Acts as an Oncosuppressor and is Lost in Malignancy
6. Therapeutic Perspectives of Strategies Targeting CD99
- In immune and tumor cells, CD99 is expressed in both short and long forms. Both isoforms play functional roles, and their differential expression can lead to distinct functional outcomes. Although the conjunction of the distinct functional outcomes via a single receptor might be coincidental, it is tempting to speculate that this finding represents a coordinated mechanism. The discrepant roles of CD99 in different tumors may be due not only to the difference in cancer types but also to the difference in the relative expression levels between the two CD99 isoforms. However, the currently available anti-CD99 antibodies cannot distinguish between the two CD99 isoforms, and other approaches must be considered in evaluating the expression levels of each CD99 isoform separately.
- The regulation of CD99 expression is still far from being understood. Considering the role that the loss of CD99 has in many common tumors, a true appreciation of the possible mechanisms involved (DNA methylation, LOH, transcriptional, and post-transcriptional mechanisms) is urgently required. In addition, more detailed studies are necessary to clarify how and when CD99sh is expressed with respect to the preeminent CD99wt isoform in different tumors.
- Considering the high dependence of CD99 functions on the cellular context, any therapeutic strategy targeting the molecule must be thoroughly studied in the appropriate context before any further clinical development.
- The application of chimeric antigen receptor (CAR) T cell therapy is emerging as a very promising, novel strategy, particularly after its success in the treatment of certain hematologic cancers. However, CAR T cell therapy for solid tumors faces some challenges, and its application potential is still far from being fully exploited because of the lack of specific tumor antigens, tumor heterogeneity, and the presence of immunosuppressive factors in the tumor microenvironment [109,110]. CD99 and the recently developed dAbd C7 [53,94,95] may represent an opportunity to address the problem of enhancing the specificity of CAR T cells for those tumors with a maintained and diffuse expression of CD99 and might be considered for generating CARs that are capable of recognizing multiple antigens.
- Recent advances in our understanding of cancer have revealed that tumors should be considered as complex tissues in which cancer cells communicate directly and indirectly with the surrounding cellular microenvironment to promote their own survival [111,112]. Because it is expressed on specific immune and stromal cells in addition to normal and cancer stem cells, CD99 appears to be at the crossroad between the regulation of tumor cell malignancy and tumor interconnections with its microenvironment. A full appreciation of how the presence or absence of CD99 may affect tumor infiltration and the cross-talk between tumor and normal host cells may open new therapeutic avenues.
- Interactions between tumor cells and their microenvironment involve extracellular vesicles, which are released by cells under physiological and pathological conditions and contain a bioactive cargo (miRNA, mRNA, proteins and lipids). The presence of CD99 in exosomes may provide another perspective on the role of this molecule in tumorigenesis.
7. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Ambros, I.M.; Ambros, P.F.; Strehl, S.; Kovar, H.; Gadner, H.; Salzer-Kuntschik, M. MIC2 is a specific marker for Ewing’s sarcoma and peripheral primitive neuroectodermal tumors. Evidence for a common histogenesis of Ewing’s sarcoma and peripheral primitive neuroectodermal tumors from MIC2 expression and specific chromosome aberration. Cancer 1991, 67, 1886–1893. [Google Scholar] [CrossRef]
- Llombart-Bosch, A.; Machado, I.; Navarro, S.; Bertoni, F.; Bacchini, P.; Alberghini, M.; Karzeladze, A.; Savelov, N.; Petrov, S.; Alvarado-Cabrero, I.; et al. Histological heterogeneity of Ewing’s sarcoma/pnet: An immunohistochemical analysis of 415 genetically confirmed cases with clinical support. Virchows Arch. Int. J. Pathol. 2009, 455, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Dworzak, M.N.; Froschl, G.; Printz, D.; Zen, L.D.; Gaipa, G.; Ratei, R.; Basso, G.; Biondi, A.; Ludwig, W.D.; Gadner, H. CD99 expression in T-lineage all: Implications for flow cytometric detection of minimal residual disease. Leukemia 2004, 18, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.S.; Eng, W.S.; Hu, W.; Khalaj, M.; Garrett-Bakelman, F.E.; Tavakkoli, M.; Levine, R.L.; Carroll, M.; Klimek, V.M.; Melnick, A.M.; et al. CD99 is a therapeutic target on disease stem cells in myeloid malignancies. Sci. Transl. Med. 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Seol, H.J.; Chang, J.H.; Yamamoto, J.; Romagnuolo, R.; Suh, Y.; Weeks, A.; Agnihotri, S.; Smith, C.A.; Rutka, J.T. Overexpression of CD99 increases the migration and invasiveness of human malignant glioma cells. Genes Cancer 2012, 3, 535–549. [Google Scholar] [CrossRef] [PubMed]
- Urias, U.; Marie, S.K.; Uno, M.; da Silva, R.; Evagelinellis, M.M.; Caballero, O.L.; Stevenson, B.J.; Silva, W.A., Jr.; Simpson, A.J.; Oba-Shinjo, S.M. CD99 is upregulated in placenta and astrocytomas with a differential subcellular distribution according to the malignancy stage. J. Neuro-Oncol. 2014, 119, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C. Synovial sarcoma. Ann. Diagn. Pathol. 1998, 2, 401–421. [Google Scholar] [CrossRef]
- Brown, R.E.; Boyle, J.L. Mesenchymal chondrosarcoma: Molecular characterization by a proteomic approach, with morphogenic and therapeutic implications. Ann. Clin. Lab. Sci. 2003, 33, 131–141. [Google Scholar] [PubMed]
- Ramani, P.; Rampling, D.; Link, M. Immunocytochemical study of 12E7 in small round-cell tumours of childhood: An assessment of its sensitivity and specificity. Histopathol. 1993, 23, 557–561. [Google Scholar] [CrossRef]
- Rajaram, V.; Brat, D.J.; Perry, A. Anaplastic meningioma versus meningeal hemangiopericytoma: Immunohistochemical and genetic markers. Hum. Pathol. 2004, 35, 1413–1418. [Google Scholar] [CrossRef] [PubMed]
- Goto, A.; Niki, T.; Terado, Y.; Fukushima, J.; Fukayama, M. Prevalence of CD99 protein expression in pancreatic endocrine tumours (PETs). Histopathology 2004, 45, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Baker, R.J.; Hildebrandt, R.H.; Rouse, R.V.; Hendrickson, M.R.; Longacre, T.A. Inhibin and CD99 (MIC2) expression in uterine stromal neoplasms with sex-cord-like elements. Hum. Pathol. 1999, 30, 671–679. [Google Scholar] [CrossRef]
- Wilkerson, A.E.; Glasgow, M.A.; Hiatt, K.M. Immunoreactivity of CD99 in invasive malignant melanoma. J. Cutan. Pathol. 2006, 33, 663–666. [Google Scholar] [CrossRef] [PubMed]
- Milanezi, F.; Pereira, E.M.; Ferreira, F.V.; Leitao, D.; Schmitt, F.C. CD99/MIC2 surface protein expression in breast carcinomas. Histopathology 2001, 39, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.C.; Park, W.S.; Bae, Y.M.; Hahn, J.H.; Hahn, K.; Lee, H.; Lee, H.W.; Koo, H.J.; Shin, H.J.; Shin, H.S.; et al. Immunoreactivity of CD99 in stomach cancer. J. Korean Med. Sci. 2002, 17, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Maitra, A.; Hansel, D.E.; Argani, P.; Ashfaq, R.; Rahman, A.; Naji, A.; Deng, S.; Geradts, J.; Hawthorne, L.; House, M.G.; et al. Global expression analysis of well-differentiated pancreatic endocrine neoplasms using oligonucleotide microarrays. Clin. Cancer Res. 2003, 9, 5988–5995. [Google Scholar] [PubMed]
- Choi, Y.L.; Xuan, Y.H.; Shin, Y.K.; Chae, S.W.; Kook, M.C.; Sung, R.H.; Youn, S.J.; Choi, J.W.; Kim, S.H. An immunohistochemical study of the expression of adhesion molecules in gallbladder lesions. J. Histochem. Cytochem. 2004, 52, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Manara, M.C.; Bernard, G.; Lollini, P.L.; Nanni, P.; Zuntini, M.; Landuzzi, L.; Benini, S.; Lattanzi, G.; Sciandra, M.; Serra, M.; et al. CD99 acts as an oncosuppressor in osteosarcoma. Mol. Biol. Cell 2006, 17, 1910–1921. [Google Scholar] [CrossRef] [PubMed]
- Hahn, J.H.; Kim, M.K.; Choi, E.Y.; Kim, S.H.; Sohn, H.W.; Ham, D.I.; Chung, D.H.; Kim, T.J.; Lee, W.J.; Park, C.K.; et al. CD99 (MIC2) regulates the LFA-1/ICAM-1-mediated adhesion of lymphocytes, and its gene encodes both positive and negative regulators of cellular adhesion. J. Immunol. 1997, 159, 2250–2258. [Google Scholar] [PubMed]
- Alberti, I.; Bernard, G.; Rouquette-Jazdanian, A.K.; Pelassy, C.; Pourtein, M.; Aussel, C.; Bernard, A. CD99 isoforms expression dictates t cell functional outcomes. FASEB J. 2002, 16, 1946–1948. [Google Scholar] [CrossRef] [PubMed]
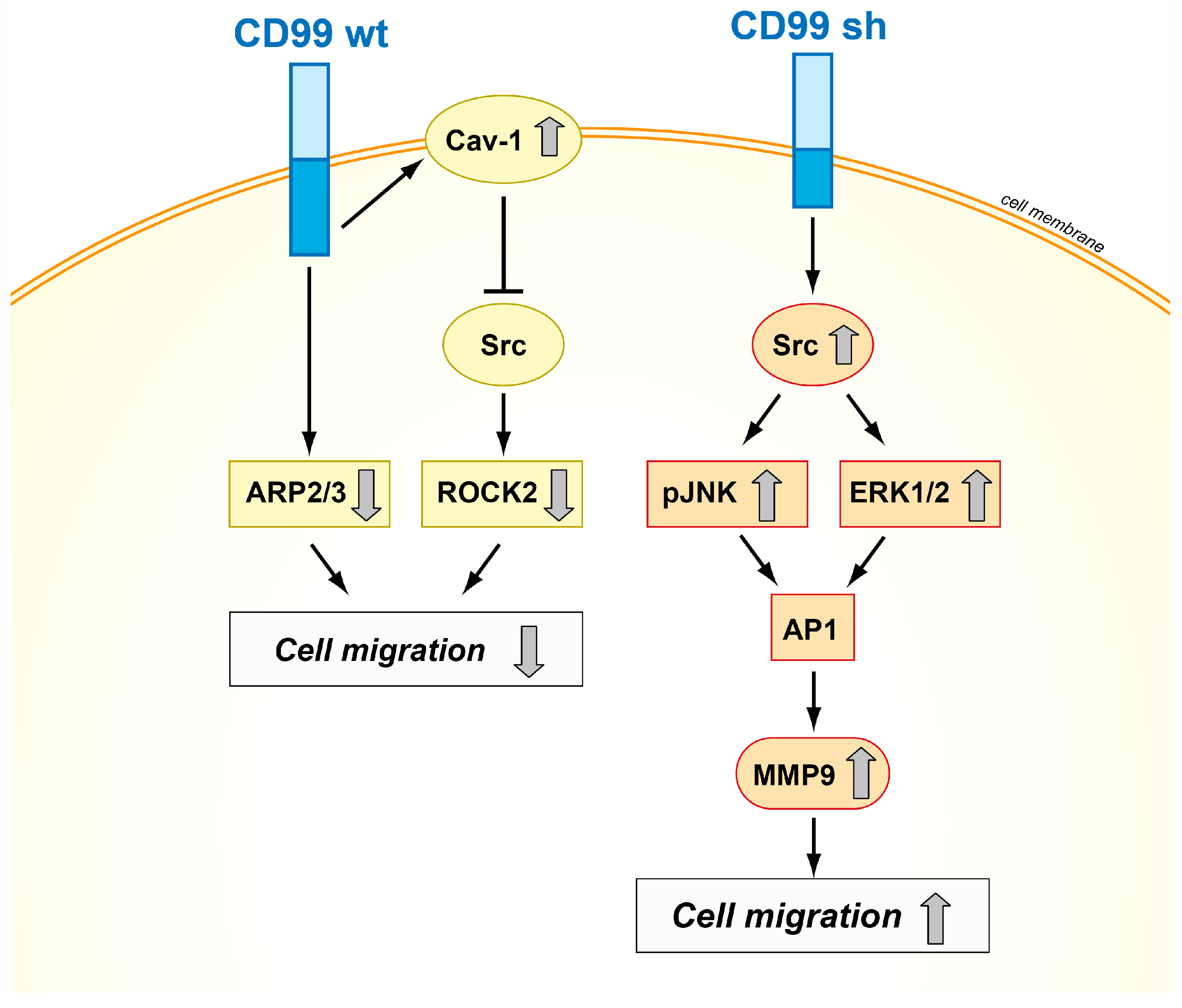
- Byun, H.J.; Hong, I.K.; Kim, E.; Jin, Y.J.; Jeoung, D.I.; Hahn, J.H.; Kim, Y.M.; Park, S.H.; Lee, H. A splice variant of CD99 increases motility and MMP-9 expression of human breast cancer cells through the AKT-, ERK-, and JNK-dependent AP-1 activation signaling pathways. J. Biol. Chem. 2006, 281, 34833–34847. [Google Scholar] [CrossRef] [PubMed]
- Scotlandi, K.; Zuntini, M.; Manara, M.C.; Sciandra, M.; Rocchi, A.; Benini, S.; Nicoletti, G.; Bernard, G.; Nanni, P.; Lollini, P.L.; et al. CD99 isoforms dictate opposite functions in tumour malignancy and metastases by activating or repressing c-Src kinase activity. Oncogene 2007, 26, 6604–6618. [Google Scholar] [CrossRef] [PubMed]
- Levy, R.; Dilley, J.; Fox, R.I.; Warnke, R. A human thymus-leukemia antigen defined by hybridoma monoclonal antibodies. Proc. Natl. Acad. Sci. USA 1979, 76, 6552–6556. [Google Scholar] [CrossRef] [PubMed]
- Petit, C.; Levilliers, J.; Weissenbach, J. Physical mapping of the human pseudo-autosomal region; comparison with genetic linkage map. EMBO J. 1988, 7, 2369–2376. [Google Scholar] [PubMed]
- Fouchet, C.; Gane, P.; Huet, M.; Fellous, M.; Rouger, P.; Banting, G.; Cartron, J.P.; Lopez, C. A study of the coregulation and tissue specificity of XG and MIC2 gene expression in eukaryotic cells. Blood 2000, 95, 1819–1826. [Google Scholar] [PubMed]
- Fouchet, C.; Gane, P.; Cartron, J.P.; Lopez, C. Quantitative analysis of XG blood group and CD99 antigens on human red cells. Immunogenetics 2000, 51, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Suh, Y.H.; Shin, Y.K.; Kook, M.C.; Oh, K.I.; Park, W.S.; Kim, S.H.; Lee, I.S.; Park, H.J.; Huh, T.L.; Park, S.H. Cloning, genomic organization, alternative transcripts and expression analysis of CD99L2, a novel paralog of human CD99, and identification of evolutionary conserved motifs. Gene 2003, 307, 63–76. [Google Scholar] [CrossRef]
- Meynet, O.; Scotlandi, K.; Pradelli, E.; Manara, M.C.; Colombo, M.P.; Schmid-Antomarchi, H.; Picci, P.; Bernard, A.; Bernard, G. XG expression in Ewing’s sarcoma is of prognostic value and contributes to tumor invasiveness. Cancer Res. 2010, 70, 3730–3738. [Google Scholar] [CrossRef] [PubMed]
- Banting, G.S.; Pym, B.; Darling, S.M.; Goodfellow, P.N. The MIC2 gene product: Epitope mapping and structural prediction analysis define an integral membrane protein. Mol. Immunol. 1989, 26, 181–188. [Google Scholar] [CrossRef]
- Bernard, G.; Breittmayer, J.P.; de Matteis, M.; Trampont, P.; Hofman, P.; Senik, A.; Bernard, A. Apoptosis of immature thymocytes mediated by E2/CD99. J. Immunol. 1997, 158, 2543–2550. [Google Scholar] [PubMed]
- Mahiddine, K.; Mallavialle, A.; Bziouech, H.; Larbret, F.; Bernard, A.; Bernard, G. CD99 isoforms regulate cd1a expression in human monocyte-derived dcs through ATF-2/CREB-1 phosphorylation. Eur. J. Immunol. 2016, 46, 1460–1471. [Google Scholar] [CrossRef] [PubMed]
- Husak, Z.; Printz, D.; Schumich, A.; Potschger, U.; Dworzak, M.N. Death induction by CD99 ligation in TEL/AML1-positive acute lymphoblastic leukemia and normal b cell precursors. J. Leukoc. Biol. 2010, 88, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Pasello, M.; Manara, M.C.; Scotlandi, K. CD99 at the crossroads of physiology and pathology. J. Cell Commun. Signal. 2018, 12, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Bernard, G.; Zoccola, D.; Deckert, M.; Breittmayer, J.P.; Aussel, C.; Bernard, A. The E2 molecule (CD99) specifically triggers homotypic aggregation of CD4+ CD8+ thymocytes. J. Immunol. 1995, 154, 26–32. [Google Scholar] [PubMed]
- Dworzak, M.N.; Fritsch, G.; Fleischer, C.; Printz, D.; Froschl, G.; Buchinger, P.; Mann, G.; Gadner, H. CD99 (MIC2) expression in paediatric B-lineage leukaemia/lymphoma reflects maturation-associated patterns of normal b-lymphopoiesis. Br. J. Haematol. 1999, 105, 690–695. [Google Scholar] [CrossRef] [PubMed]
- Waclavicek, M.; Majdic, O.; Stulnig, T.; Berger, M.; Sunder-Plassmann, R.; Zlabinger, G.J.; Baumruker, T.; Stockl, J.; Ebner, C.; Knapp, W.; et al. CD99 engagement on human peripheral blood T cells results in TCR/CD3-dependent cellular activation and allows for TH1-restricted cytokine production. J. Immunol. 1998, 161, 4671–4678. [Google Scholar] [PubMed]
- Wingett, D.; Forcier, K.; Nielson, C.P. A role for CD99 in T cell activation. Cell. Immunol. 1999, 193, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, R.D.; Bernard, G.; Olafsen, M.K.; Pourtein, M.; Lie, S.O. CD99 signals caspase-independent T cell death. J. Immunol. 2001, 166, 4931–4942. [Google Scholar] [CrossRef] [PubMed]
- Bernard, G.; Raimondi, V.; Alberti, I.; Pourtein, M.; Widjenes, J.; Ticchioni, M.; Bernard, A. CD99 (E2) up-regulates α4β1-dependent T cell adhesion to inflamed vascular endothelium under flow conditions. Eur. J. Immunol. 2000, 30, 3061–3065. [Google Scholar] [CrossRef]
- Kasinrerk, W.; Tokrasinwit, N.; Moonsom, S.; Stockinger, H. CD99 monoclonal antibody induce homotypic adhesion of jurkat cells through protein tyrosine kinase and protein kinase C-dependent pathway. Immunol. Lett. 2000, 71, 33–41. [Google Scholar] [CrossRef]
- Watson, R.L.; Buck, J.; Levin, L.R.; Winger, R.C.; Wang, J.; Arase, H.; Muller, W.A. Endothelial CD99 signals through soluble adenylyl cyclase and PKA to regulate leukocyte transendothelial migration. J. Exp. Med. 2015, 212, 1021–1041. [Google Scholar] [CrossRef] [PubMed]
- Dufour, E.M.; Deroche, A.; Bae, Y.; Muller, W.A. CD99 is essential for leukocyte diapedesis in vivo. Cell Commun. Adhes. 2008, 15, 351–363. [Google Scholar] [CrossRef] [PubMed]
- Sohn, H.W.; Shin, Y.K.; Lee, I.S.; Bae, Y.M.; Suh, Y.H.; Kim, M.K.; Kim, T.J.; Jung, K.C.; Park, W.S.; Park, C.S.; et al. CD99 regulates the transport of MHC class I molecules from the golgi complex to the cell surface. J. Immunol. 2001, 166, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Bremond, A.; Meynet, O.; Mahiddine, K.; Coito, S.; Tichet, M.; Scotlandi, K.; Breittmayer, J.P.; Gounon, P.; Gleeson, P.A.; Bernard, A.; et al. Regulation of HLA class I surface expression requires CD99 and p230/golgin-245 interaction. Blood 2009, 113, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.Y.; Park, W.S.; Jung, K.C.; Kim, S.H.; Kim, Y.Y.; Lee, W.J.; Park, S.H. Engagement of CD99 induces up-regulation of TCR and MHC class I and II molecules on the surface of human thymocytes. J. Immunol. 1998, 161, 749–754. [Google Scholar] [PubMed]
- Yoon, S.S.; Jung, K.I.; Choi, Y.L.; Choi, E.Y.; Lee, I.S.; Park, S.H.; Kim, T.J. Engagement of CD99 triggers the exocytic transport of ganglioside GM1 and the reorganization of actin cytoskeleton. FEBS Lett. 2003, 540, 217–222. [Google Scholar] [CrossRef]
- Choi, G.; Roh, J.; Park, C.S. CD99 is strongly expressed in basal cells of the normal adult epidermis and some subpopulations of appendages: Comparison with developing fetal skin. J. Pathol. Transl. Med. 2016, 50, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Sciandra, M.; Marino, M.T.; Manara, M.C.; Guerzoni, C.; Grano, M.; Oranger, A.; Lucarelli, E.; Lollini, P.L.; Dozza, B.; Pratelli, L.; et al. CD99 drives terminal differentiation of osteosarcoma cells by acting as a spatial regulator of ERK 1/2. J. Bone Min. Res. 2014, 29, 1295–1309. [Google Scholar] [CrossRef] [PubMed]
- Amaral, A.T.; Manara, M.C.; Berghuis, D.; Ordonez, J.L.; Biscuola, M.; Lopez-Garcia, M.A.; Osuna, D.; Lucarelli, E.; Alviano, F.; Lankester, A.; et al. Characterization of human mesenchymal stem cells from Ewing sarcoma patients. Pathogenetic implications. PLoS ONE 2014, 9, e85814. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Kim, Y.M.; Shin, Y.K.; Park, S.H.; Lee, W. Solution structure of the cytoplasmic domain of human CD99 type I. Mol. Cells 2004, 18, 24–29. [Google Scholar] [PubMed]
- Scotlandi, K.; Baldini, N.; Cerisano, V.; Manara, M.C.; Benini, S.; Serra, M.; Lollini, P.L.; Nanni, P.; Nicoletti, G.; Bernard, G.; et al. CD99 engagement: An effective therapeutic strategy for Ewing tumors. Cancer Res. 2000, 60, 5134–5142. [Google Scholar] [PubMed]
- Cerisano, V.; Aalto, Y.; Perdichizzi, S.; Bernard, G.; Manara, M.C.; Benini, S.; Cenacchi, G.; Preda, P.; Lattanzi, G.; Nagy, B.; et al. Molecular mechanisms of CD99-induced caspase-independent cell death and cell-cell adhesion in Ewing’s sarcoma cells: Actin and zyxin as key intracellular mediators. Oncogene 2004, 23, 5664–5674. [Google Scholar] [CrossRef] [PubMed]
- Guerzoni, C.; Fiori, V.; Terracciano, M.; Manara, M.C.; Moricoli, D.; Pasello, M.; Sciandra, M.; Nicoletti, G.; Gellini, M.; Dominici, S.; et al. CD99 triggering in Ewing sarcoma delivers a lethal signal through p53 pathway reactivation and cooperates with doxorubicin. Clin. Cancer Res. 2015, 21, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Pata, S.; Otahal, P.; Brdicka, T.; Laopajon, W.; Mahasongkram, K.; Kasinrerk, W. Association of CD99 short and long forms with MHC class I, MHC class II and tetraspanin CD81 and recruitment into immunological synapses. BMC Res. Notes 2011, 4, 293. [Google Scholar] [CrossRef] [PubMed]
- Zucchini, C.; Manara, M.C.; Pinca, R.S.; De Sanctis, P.; Guerzoni, C.; Sciandra, M.; Lollini, P.L.; Cenacchi, G.; Picci, P.; Valvassori, L.; et al. CD99 suppresses osteosarcoma cell migration through inhibition of ROCK2 activity. Oncogene 2014, 33, 1912–1921. [Google Scholar] [CrossRef] [PubMed]
- Hahn, M.J.; Yoon, S.S.; Sohn, H.W.; Song, H.G.; Park, S.H.; Kim, T.J. Differential activation of MAP kinase family members triggered by CD99 engagement. FEBS Lett. 2000, 470, 350–354. [Google Scholar] [CrossRef]
- Choe, Y.; Jung, H.; Khang, I.; Kim, K. Selective roles of protein kinase C isoforms on cell motility of GT1 immortalized hypothalamic neurones. J. Neuroendocrinol. 2003, 15, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Lee, H.G.; Park, S.H.; Choi, E.Y.; Park, S.H. CD99 type II is a determining factor for the differentiation of primitive neuroectodermal cells. Exp. Mol. Med. 2003, 35, 438–447. [Google Scholar] [CrossRef] [PubMed]
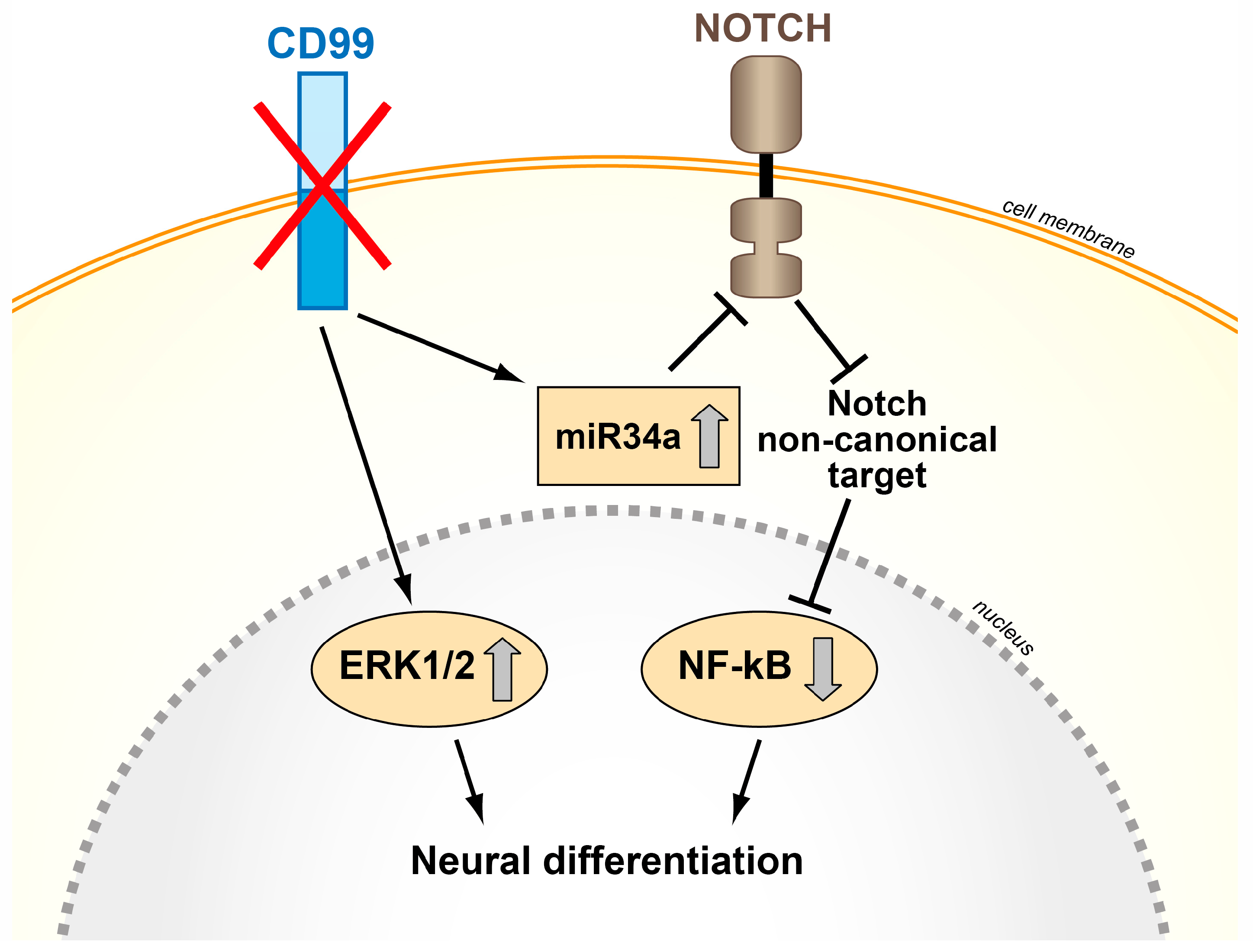
- Rocchi, A.; Manara, M.C.; Sciandra, M.; Zambelli, D.; Nardi, F.; Nicoletti, G.; Garofalo, C.; Meschini, S.; Astolfi, A.; Colombo, M.P.; et al. CD99 inhibits neural differentiation of human Ewing sarcoma cells and thereby contributes to oncogenesis. J. Clin. Investig. 2010, 120, 668–680. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Kim, E.; Jee, B.; Hahn, J.H.; Han, K.; Jung, K.C.; Park, S.H.; Lee, H. Functional involvement of src and focal adhesion kinase in a CD99 splice variant-induced motility of human breast cancer cells. Exp. Mol. Med. 2002, 34, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Fellinger, E.J.; Garin-Chesa, P.; Triche, T.J.; Huvos, A.G.; Rettig, W.J. Immunohistochemical analysis of Ewing’s sarcoma cell surface antigen p30/32MIC2. Am. J. Pathol. 1991, 139, 317–325. [Google Scholar] [PubMed]
- Antonescu, C.R.; Owosho, A.A.; Zhang, L.; Chen, S.; Deniz, K.; Huryn, J.M.; Kao, Y.C.; Huang, S.C.; Singer, S.; Tap, W.; et al. Sarcomas with cic-rearrangements are a distinct pathologic entity with aggressive outcome: A clinicopathologic and molecular study of 115 cases. Am. J. Surg. Pathol. 2017, 41, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Machado, I.; Navarro, L.; Pellin, A.; Navarro, S.; Agaimy, A.; Tardio, J.C.; Karseladze, A.; Petrov, S.; Scotlandi, K.; Picci, P.; et al. Defining Ewing and Ewing-like small round cell tumors (SRCT): The need for molecular techniques in their categorization and differential diagnosis. A study of 200 cases. Ann. Diagn. Pathol. 2016, 22, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Riggi, N.; Stamenkovic, I. The biology of Ewing sarcoma. Cancer Lett. 2007, 254, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sankar, S.; Bell, R.; Stephens, B.; Zhuo, R.; Sharma, S.; Bearss, D.J.; Lessnick, S.L. Mechanism and relevance of EWS/FLI-mediated transcriptional repression in Ewing sarcoma. Oncogene 2013, 32, 5089–5100. [Google Scholar] [CrossRef] [PubMed]
- Rorie, C.J.; Thomas, V.D.; Chen, P.; Pierce, H.H.; O’Bryan, J.P.; Weissman, B.E. The EWS/FLI-1 fusion gene switches the differentiation program of neuroblastomas to Ewing sarcoma/peripheral primitive neuroectodermal tumors. Cancer Res. 2004, 64, 1266–1277. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hu-Lieskovan, S.; Zhang, J.; Wu, L.; Shimada, H.; Schofield, D.E.; Triche, T.J. EWS-FLI1 fusion protein up-regulates critical genes in neural crest development and is responsible for the observed phenotype of Ewing’s family of tumors. Cancer Res. 2005, 65, 4633–4644. [Google Scholar] [CrossRef] [PubMed]
- Franzetti, G.A.; Laud-Duval, K.; Bellanger, D.; Stern, M.H.; Sastre-Garau, X.; Delattre, O. MiR-30a-5p connects EWS-FLI1 and CD99, two major therapeutic targets in Ewing tumor. Oncogene 2013, 32, 3915–3921. [Google Scholar] [CrossRef] [PubMed]
- Kreppel, M.; Aryee, D.N.; Schaefer, K.L.; Amann, G.; Kofler, R.; Poremba, C.; Kovar, H. Suppression of kcmf1 by constitutive high CD99 expression is involved in the migratory ability of Ewing’s sarcoma cells. Oncogene 2006, 25, 2795–2800. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Mi, L.; Yu, X.F.; Dong, J. Interaction of 14-3-σwith KCMF1 suppresses the proliferation and colony formation of human colon cancer stem cells. World J. Gastroenterol. 2013, 19, 3770–3780. [Google Scholar] [CrossRef] [PubMed]
- Ashton-Beaucage, D.; Lemieux, C.; Udell, C.M.; Sahmi, M.; Rochette, S.; Therrien, M. The deubiquitinase USP47 stabilizes MAPK by counteracting the function of the N-end rule ligase POE/UBR4 in drosophila. PLoS Biol. 2016, 14, e1002539. [Google Scholar] [CrossRef] [PubMed]
- Crespo, P.; Leon, J. Ras proteins in the control of the cell cycle and cell differentiation. Cell. Mol. Life Sci. CMLS 2000, 57, 1613–1636. [Google Scholar] [CrossRef] [PubMed]
- Traverse, S.; Gomez, N.; Paterson, H.; Marshall, C.; Cohen, P. Sustained activation of the mitogen-activated protein (MAP) kinase cascade may be required for differentiation of PC12 cells. Comparison of the effects of nerve growth factor and epidermal growth factor. Biochem. J. 1992, 288, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Ventura, S.; Aryee, D.N.; Felicetti, F.; De Feo, A.; Mancarella, C.; Manara, M.C.; Picci, P.; Colombo, M.P.; Kovar, H.; Care, A.; et al. CD99 regulates neural differentiation of Ewing sarcoma cells through miR-34a-Notch-mediated control of Nf-kappaB signaling. Oncogene 2016, 35, 3944–3954. [Google Scholar] [CrossRef] [PubMed]
- Angelini, D.F.; Ottone, T.; Guerrera, G.; Lavorgna, S.; Cittadini, M.; Buccisano, F.; De Bardi, M.; Gargano, F.; Maurillo, L.; Divona, M.; et al. A leukemia-associated CD34/CD123/CD25/CD99+ immunophenotype identifies FLT3-mutated clones in acute myeloid leukemia. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2015, 21, 3977–3985. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.J.; Barcos, M.; Stewart, C.C.; Block, A.W.; Sait, S.; Brooks, J.J. Immunoreactivity of MIC2 (CD99) in acute myelogenous leukemia and related diseases. Modern Pathol. 2000, 13, 452–458. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chan, J.K.; Tsang, W.Y.; Seneviratne, S.; Pau, M.Y. The MIC2 antibody 013. Practical application for the study of thymic epithelial tumors. Am. J. Surg. Pathol. 1995, 19, 1115–1123. [Google Scholar] [PubMed]
- Choi, Y.L.; Chi, J.G.; Suh, Y.L. CD99 immunoreactivity in ependymoma. Appl. Immunohistochem. Mol. Morphol. 2001, 9, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Mahfouz, S.; Aziz, A.A.; Gabal, S.M.; el-Sheikh, S. Immunohistochemical study of CD99 and EMA expression in ependymomas. Medscape J. Med. 2008, 10, 41. [Google Scholar] [PubMed]
- Kim, S.H.; Shin, Y.K.; Lee, I.S.; Bae, Y.M.; Sohn, H.W.; Suh, Y.H.; Ree, H.J.; Rowe, M.; Park, S.H. Viral latent membrane protein 1 (LMP-1)-induced CD99 down-regulation in B cells leads to the generation of cells with Hodgkin’s and reed-sternberg phenotype. Blood 2000, 95, 294–300. [Google Scholar] [PubMed]
- Pelosi, G.; Fraggetta, F.; Sonzogni, A.; Fazio, N.; Cavallon, A.; Viale, G. CD99 immunoreactivity in gastrointestinal and pulmonary neuroendocrine tumours. Virchows Arch. Int. J. Pathol. 2000, 437, 270–274. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, S.H.; Wang, L.H.; Choi, Y.L.; Kim, Y.C.; Kim, J.H.; Park, T.S.; Hong, Y.C.; Shin, Y.K. Clinical significance of CD99 down-regulation in gastric adenocarcinoma. Clin. Cancer Res. 2007, 13, 2584–2591. [Google Scholar] [CrossRef] [PubMed]
- Pelosi, G.; Leon, M.E.; Veronesi, G.; Spaggiari, L.; Pasini, F.; Viale, G. Decreased immunoreactivity of CD99 is an independent predictor of regional lymph node metastases in pulmonary carcinoid tumors. J. Thorac. Oncol. 2006, 1, 468–477. [Google Scholar] [CrossRef]
- Huang, X.; Zhou, X.; Wang, Z.; Li, F.; Liu, F.; Zhong, L.; Li, X.; Han, X.; Wu, Z.; Chen, S.; et al. CD99 triggers upregulation of miR-9-modulated PRDM1/BLIMP1 in Hodgkin/Reed-Sternberg cells and induces redifferentiation. Int. J. Cancer 2012, 131, E382–E394. [Google Scholar] [CrossRef] [PubMed]
- Xuan, Y.; Kim, S.; Lin, Z. Protein expression and gene promoter hypermethylation of CD99 in transitional cell carcinoma of urinary bladder. J. Cancer Res. Clin. Oncol. 2011, 137, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Cantiani, L.; Manara, M.C.; Zucchini, C.; De Sanctis, P.; Zuntini, M.; Valvassori, L.; Serra, M.; Olivero, M.; Di Renzo, M.F.; Colombo, M.P.; et al. Caveolin-1 reduces osteosarcoma metastases by inhibiting c-Src activity and met signaling. Cancer Res. 2007, 67, 7675–7685. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Zhang, Y.; Ye, L.; Jiang, W.G. The ferm family proteins in cancer invasion and metastasis. Front. Biosci. 2011, 16, 1536–1550. [Google Scholar] [CrossRef]
- Lee, K.J.; Yoo, Y.H.; Kim, M.S.; Yadav, B.K.; Kim, Y.; Lim, D.; Hwangbo, C.; Moon, K.W.; Kim, D.; Jeoung, D.; et al. CD99 inhibits CD98-mediated β1 integrin signaling through SHP2-mediated FAK dephosphorylation. Exp. Cell Res. 2015, 336, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Hall, P.A.; Russell, S.E. The pathobiology of the septin gene family. J. Pathol. 2004, 204, 489–505. [Google Scholar] [CrossRef] [PubMed]
- Doye, V.; Le Gouvello, S.; Dobransky, T.; Chneiweiss, H.; Beretta, L.; Sobel, A. Expression of transfected stathmin cdna reveals novel phosphorylated forms associated with developmental and functional cell regulation. Biochem. J. 1992, 287, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Jian, W.; Zhong, L.; Wen, J.; Tang, Y.; Qiu, B.; Wu, Z.; Yan, J.; Zhou, X.; Zhao, T. SEPTIN2 and stathmin regulate CD99-mediated cellular differentiation in Hodgkin’s lymphoma. PLoS ONE 2015, 10, e0127568. [Google Scholar] [CrossRef] [PubMed]
- Sohn, H.W.; Choi, E.Y.; Kim, S.H.; Lee, I.S.; Chung, D.H.; Sung, U.A.; Hwang, D.H.; Cho, S.S.; Jun, B.H.; Jang, J.J.; et al. Engagement of CD99 induces apoptosis through a calcineurin-independent pathway in Ewing’s sarcoma cells. Am. J. Pathol. 1998, 153, 1937–1945. [Google Scholar] [CrossRef]
- Husak, Z.; Dworzak, M.N. CD99 ligation upregulates HSP70 on acute lymphoblastic leukemia cells and concomitantly increases NK cytotoxicity. Cell Death Dis. 2012, 3, e425. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gellini, M.; Ascione, A.; Flego, M.; Mallano, A.; Dupuis, M.L.; Zamboni, S.; Terrinoni, M.; D’Alessio, V.; Manara, M.C.; Scotlandi, K.; et al. Generation of human single-chain antibody to the CD99 cell surface determinant specifically recognizing Ewing’s sarcoma tumor cells. Curr. Pharm. Biotechnol. 2013, 14, 449–463. [Google Scholar] [CrossRef] [PubMed]
- Moricoli, D.; Carbonella, D.C.; Dominici, S.; Fiori, V.; Balducci, M.C.; Guerzoni, C.; Manara, M.C.; Pasello, M.; Laguardia, M.E.; Cianfriglia, M.; et al. Process development of a human recombinant diabody expressed in E. coli: Engagement of CD99-induced apoptosis for target therapy in Ewing’s sarcoma. Appl. Microbiol. Biotechnol. 2016, 100, 3949–3963. [Google Scholar] [CrossRef] [PubMed]
- Manara, M.C.; Terracciano, M.; Mancarella, C.; Sciandra, M.; Guerzoni, C.; Pasello, M.; Grilli, A.; Zini, N.; Picci, P.; Colombo, M.P.; et al. CD99 triggering induces methuosis of Ewing sarcoma cells through IGF-1R/RAS/RAC1 signaling. Oncotarget 2016, 7, 79925–79942. [Google Scholar] [CrossRef] [PubMed]
- Maltese, W.A.; Overmeyer, J.H. Methuosis: Nonapoptotic cell death associated with vacuolization of macropinosome and endosome compartments. Am. J. Pathol. 2014, 184, 1630–1642. [Google Scholar] [CrossRef] [PubMed]
- Scotlandi, K.; Perdichizzi, S.; Bernard, G.; Nicoletti, G.; Nanni, P.; Lollini, P.L.; Curti, A.; Manara, M.C.; Benini, S.; Bernard, A.; et al. Targeting CD99 in association with doxorubicin: An effective combined treatment for Ewing’s sarcoma. Eur. J. Cancer 2006, 42, 91–96. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, A.F.; Dearling, J.L.; Wang, Y.; Tupper, T.; Sun, Y.; Aster, J.C.; Calicchio, M.L.; Perez-Atayde, A.R.; Packard, A.B.; Kung, A.L. Targeted imaging of Ewing sarcoma in preclinical models using a 64Cu-labeled anti-CD99 antibody. Clin. Cancer Res. 2014, 20, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Edlund, K.; Lindskog, C.; Saito, A.; Berglund, A.; Ponten, F.; Goransson-Kultima, H.; Isaksson, A.; Jirstrom, K.; Planck, M.; Johansson, L.; et al. CD99 is a novel prognostic stromal marker in non-small cell lung cancer. Int. J. Cancer 2012, 131, 2264–2273. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Tran, T.A.; Lee, H.J.; Jung, S.I.; Lee, J.J.; Jang, W.Y.; Moon, K.S.; Kim, I.Y.; Jung, S.; Jung, T.Y. Branched multipeptide immunotherapy for glioblastoma using human leukocyte antigen-A*0201-restricted cytotoxic T-lymphocyte epitopes from ERBB2, BIRC5 and CD99. Oncotarget 2016, 7, 50535–50547. [Google Scholar] [CrossRef] [PubMed]
- Robbins, P.F.; Morgan, R.A.; Feldman, S.A.; Yang, J.C.; Sherry, R.M.; Dudley, M.E.; Wunderlich, J.R.; Nahvi, A.V.; Helman, L.J.; Mackall, C.L.; et al. Tumor regression in patients with metastatic synovial cell sarcoma and melanoma using genetically engineered lymphocytes reactive with NY-ESO-1. J. Clin.Oncol. 2011, 29, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Thiel, U.; Schober, S.J.; Einspieler, I.; Kirschner, A.; Thiede, M.; Schirmer, D.; Gall, K.; Blaeschke, F.; Schmidt, O.; Jabar, S.; et al. Ewing sarcoma partial regression without GvHD by chondromodulin-I/HLA-A*02:01-specific allorestricted T cell receptor transgenic t cells. Oncoimmunology 2017, 6, e1312239. [Google Scholar] [CrossRef] [PubMed]
- Kirschner, A.; Thiede, M.; Grunewald, T.G.; Alba Rubio, R.; Richter, G.H.; Kirchner, T.; Busch, D.H.; Burdach, S.; Thiel, U. Pappalysin-1 T cell receptor transgenic allo-restricted T cells kill Ewing sarcoma in vitro and in vivo. Oncoimmunology 2017, 6, e1273301. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, D.; Grunewald, T.G.; Klar, R.; Schmidt, O.; Wohlleber, D.; Rubio, R.A.; Uckert, W.; Thiel, U.; Bohne, F.; Busch, D.H.; et al. Transgenic antigen-specific, HLA-A*02:01-allo-restricted cytotoxic T cells recognize tumor-associated target antigen steap1 with high specificity. Oncoimmunology 2016, 5, e1175795. [Google Scholar] [CrossRef] [PubMed]
- Celik, H.; Sciandra, M.; Flashner, B.; Gelmez, E.; Kayraklioglu, N.; Allegakoen, D.V.; Petro, J.R.; Conn, E.J.; Hour, S.; Han, J.; et al. Clofarabine inhibits Ewing sarcoma growth through a novel molecular mechanism involving direct binding to CD99. Oncogene 2018. [Google Scholar] [CrossRef]
- Braicu, C.; Tomuleasa, C.; Monroig, P.; Cucuianu, A.; Berindan-Neagoe, I.; Calin, G.A. Exosomes as divine messengers: Are they the hermes of modern molecular oncology? Cell Death Differ. 2015, 22, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, A.A.; Desai, N.N.; Qureshi, M.Z.; Librelotto, D.R.N.; Gasparri, M.L.; Bishayee, A.; Nabavi, S.M.; Curti, V.; Daglia, M. Exosome biogenesis, bioactivities and functions as new delivery systems of natural compounds. Biotechnol. Adv. 2018, 36, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Luo, F.; Yang, J.; Zhao, C.; Chu, Y. New chimeric antigen receptor design for solid tumors. Front. Immunol. 2017, 8, 1934. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, H.R.; Rodriguez, A.; Shepphird, J.; Brown, C.E.; Badie, B. Chimeric antigen receptors T cell therapy in solid tumor: Challenges and clinical applications. Front. Immunol. 2017, 8, 1850. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Achreja, A.; Iessi, E.; Logozzi, M.; Mizzoni, D.; Di Raimo, R.; Nagrath, D.; Fais, S. The key role of extracellular vesicles in the metastatic process. Biochim. Biophys. Acta 2017, 1869, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Cui, D.; Huang, Z.; Liu, Y.; Ouyang, G. The multifaceted role of periostin in priming the tumor microenvironments for tumor progression. Cell. Mol. Life Sci. 2017, 74, 4287–4291. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manara, M.C.; Pasello, M.; Scotlandi, K. CD99: A Cell Surface Protein with an Oncojanus Role in Tumors. Genes 2018, 9, 159. https://doi.org/10.3390/genes9030159
Manara MC, Pasello M, Scotlandi K. CD99: A Cell Surface Protein with an Oncojanus Role in Tumors. Genes. 2018; 9(3):159. https://doi.org/10.3390/genes9030159
Chicago/Turabian StyleManara, Maria Cristina, Michela Pasello, and Katia Scotlandi. 2018. "CD99: A Cell Surface Protein with an Oncojanus Role in Tumors" Genes 9, no. 3: 159. https://doi.org/10.3390/genes9030159
APA StyleManara, M. C., Pasello, M., & Scotlandi, K. (2018). CD99: A Cell Surface Protein with an Oncojanus Role in Tumors. Genes, 9(3), 159. https://doi.org/10.3390/genes9030159





