DNA Damage Tolerance Mechanisms Revealed from the Analysis of Immunoglobulin V Gene Diversification in Avian DT40 Cells
Abstract
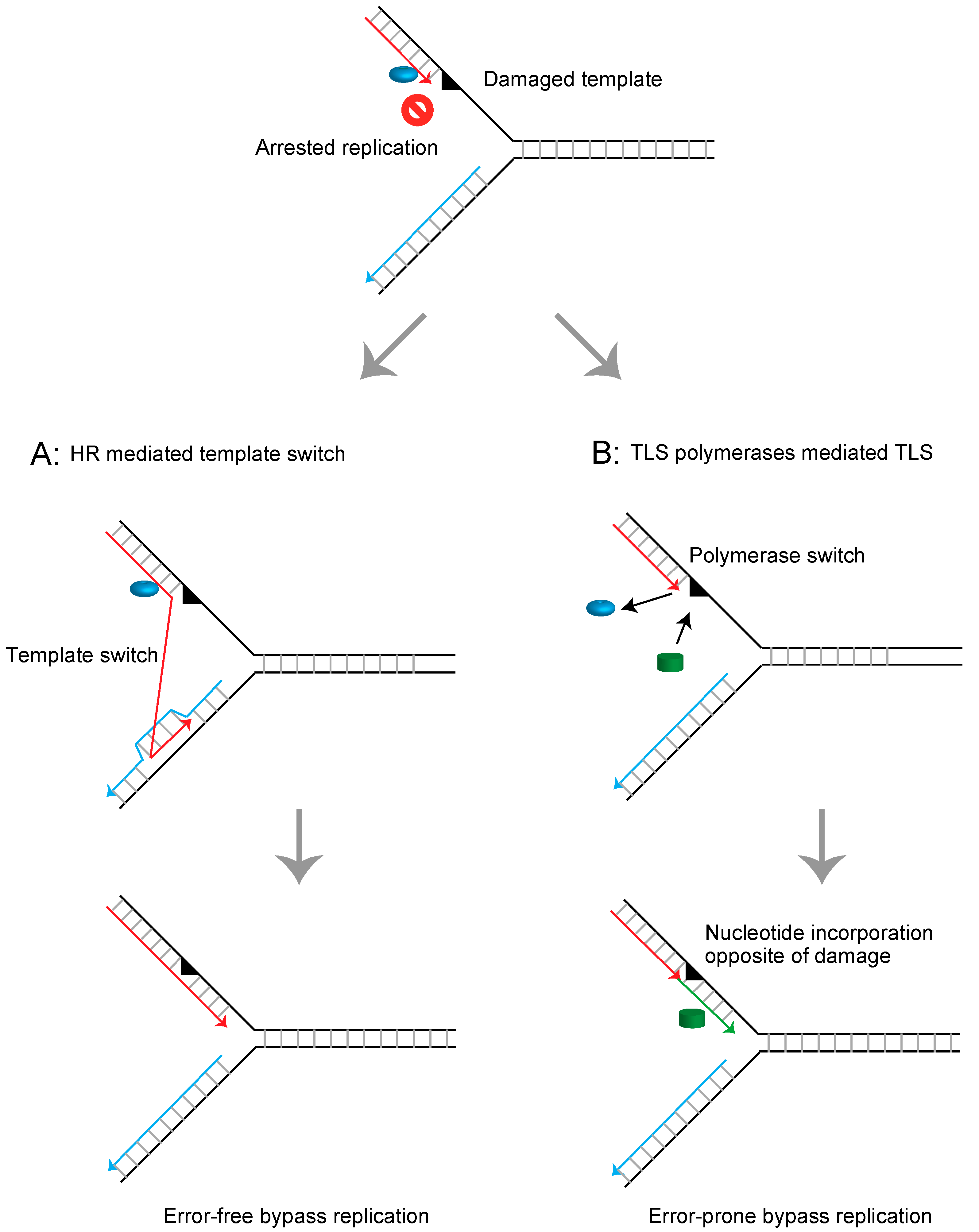
1. Introduction
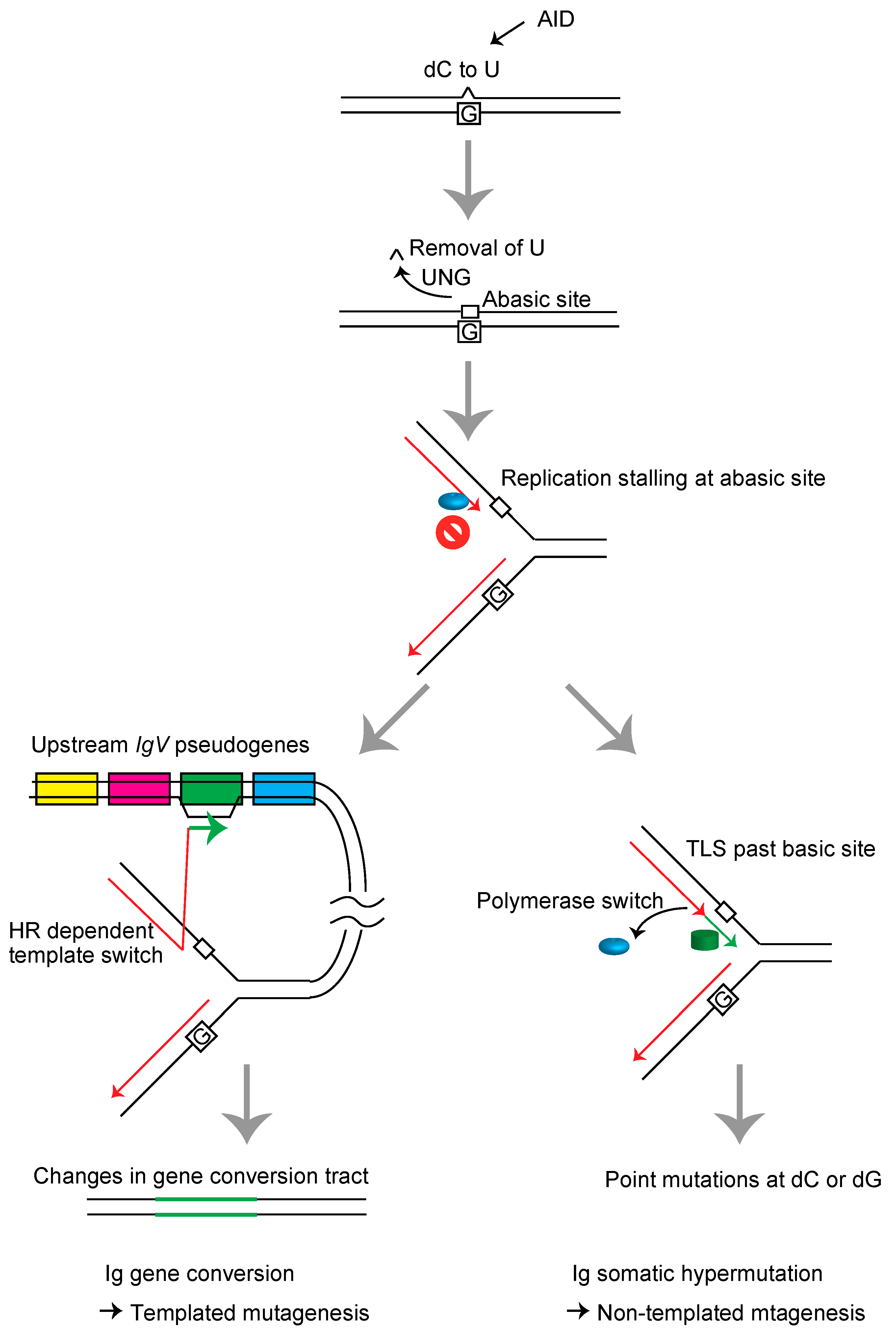
2. Immunoglobulin variable (IgV) Gene Diversification in DT40 Cells
3. Templated Mutagenesis by Gene Conversion
4. Nontemplated Somatic Hypermutation
5. Summary and Perspective
Funding
Conflicts of Interest
References
- McCulloch, S.D.; Kunkel, T.A. The fidelity of DNA synthesis by eukaryotic replicative and translesion synthesis polymerases. Cell Res. 2008, 18, 148–161. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, M.W.; Matsumoto, Y.; Loeb, L.A. High fidelity and lesion bypass capability of human DNA polymerase delta. Biochimie 2009, 91, 1163–1172. [Google Scholar] [CrossRef] [PubMed]
- Branzei, D.; Foiani, M. Regulation of DNA repair throughout the cell cycle. Nat. Rev. Mol. Cell Biol. 2008, 9, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, T. Instability and decay of the primary structure of DNA. Nature 1993, 362, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Friedberg, E.C.; Walker, G.C.; Siede, W.; Wood, R.D.; Schultz, R.A.; Ellenberger, T. DNA Repair and Mutagenesis; ASM Press: Boston, MA, USA, 2006. [Google Scholar]
- Barnes, D.E.; Lindahl, T.; Sedgwick, B. DNA repair. Curr. Opin. Cell Biol. 1993, 5, 424–433. [Google Scholar] [CrossRef]
- Klungland, A.; Rosewell, I.; Hollenbach, S.; Larsen, E.; Daly, G.; Epe, B.; Seeberg, E.; Lindahl, T.; Barnes, D.E. Accumulation of premutagenic DNA lesions in mice defective in removal of oxidative base damage. Proc. Natl. Acad. Sci. USA 1999, 96, 13300–13305. [Google Scholar] [CrossRef] [PubMed]
- Loeb, L.A. Apurinic sites as mutagenic intermediates. Cell 1985, 40, 483–484. [Google Scholar] [CrossRef]
- Prasad, R.; Horton, J.K.; Liu, Y.; Wilson, S.H. Central steps in mammalian BER and regulation by PARP1. Base Excision Repair Pathw. 2017, 253–280. [Google Scholar] [CrossRef]
- Branzei, D.; Psakhye, I. DNA damage tolerance. Curr. Opin. Cell Biol. 2016, 40, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Mutter-Rottmayer, E.; Zlatanou, A.; Vaziri, C.; Yang, Y. Mechanisms of post-replication DNA repair. Genes 2017, 8, 64. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Kasaishi, Y.; Nakada, S.; Takagi, T.; Era, S.; Motegi, A.; Chiu, R.K.; Takeda, S.; Hirota, K. Rad18 and Rnf8 facilitate homologous recombination by two distinct mechanisms, promoting Rad51 focus formation and suppressing the toxic effect of nonhomologous end joining. Oncogene 2015, 34, 4403–4411. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Heyer, W.D. Homologous recombination in DNA repair and DNA damage tolerance. Cell Res. 2008, 18, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Sonoda, E.; Kawamoto, T.; Motegi, A.; Masutani, C.; Hanaoka, F.; Szuts, D.; Iwai, S.; Sale, J.E.; Lehmann, A.; et al. Simultaneous disruption of two DNA polymerases, Poleta and Polzeta, in Avian DT40 cells unmasks the role of Poleta in cellular response to various DNA lesions. PLoS Genet. 2010, 6, e1001151. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Tsuda, M.; Tsurimoto, T.; Cohen, I.S.; Livneh, Z.; Kobayashi, K.; Narita, T.; Nishihara, K.; Murai, J.; Iwai, S.; et al. In vivo evidence for translesion synthesis by the replicative DNA polymerase delta. Nucleic Acids Res. 2016, 44, 7242–7250. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Yoshikiyo, K.; Guilbaud, G.; Tsurimoto, T.; Murai, J.; Tsuda, M.; Phillips, L.G.; Narita, T.; Nishihara, K.; Kobayashi, K.; et al. The POLD3 subunit of DNA polymerase δ can promote translesion synthesis independently of DNA polymerase ζ. Nucleic Acids Res. 2015, 43, 1671–1683. [Google Scholar] [CrossRef] [PubMed]
- Kohzaki, M.; Nishihara, K.; Hirota, K.; Sonoda, E.; Yoshimura, M.; Ekino, S.; Butler, J.E.; Watanabe, M.; Halazonetis, T.D.; Takeda, S. DNA polymerases ν and θ are required for efficient immunoglobulin V gene diversification in chicken. J. Cell Biol. 2010, 189, 1117–1127. [Google Scholar] [CrossRef]
- Sale, J.E. Competition, collaboration and coordination–determining how cells bypass DNA damage. J. Cell Sci. 2012, 125, 1633–1643. [Google Scholar] [CrossRef]
- Arakawa, H.; Buerstedde, J.M. Activation-induced cytidine deaminase-mediated hypermutation in the DT40 cell line. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 639–644. [Google Scholar] [CrossRef]
- Sale, J.E. Translesion DNA synthesis and mutagenesis in eukaryotes. Cold Spring Harb. Perspect. Biol. 2013, 5, a012708. [Google Scholar] [CrossRef]
- Sale, J.E.; Batters, C.; Edmunds, C.E.; Phillips, L.G.; Simpson, L.J.; Szuts, D. Timing matters: Error-prone gap filling and translesion synthesis in immunoglobulin gene hypermutation. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 595–603. [Google Scholar] [CrossRef]
- Romanello, M.; Schiavone, D.; Frey, A.; Sale, J.E. Histone H3.3 promotes IgV gene diversification by enhancing formation of AID-accessible single-stranded DNA. EMBO J. 2016, 35, 1452–1464. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Felsenfeld, G. Nucleosome stability mediated by histone variants H3.3 and H2A.Z. Genes Dev. 2007, 21, 1519–1529. [Google Scholar] [CrossRef] [PubMed]
- Di Noia, J.; Neuberger, M.S. Altering the pathway of immunoglobulin hypermutation by inhibiting uracil-DNA glycosylase. Nature 2002, 419, 43–48. [Google Scholar] [CrossRef]
- Saribasak, H.; Saribasak, N.N.; Ipek, F.M.; Ellwart, J.W.; Arakawa, H.; Buerstedde, J.M. Uracil DNA glycosylase disruption blocks Ig gene conversion and induces transition mutations. J. Immunol. 2006, 176, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Blagodatski, A.; Batrak, V.; Schmidl, S.; Schoetz, U.; Caldwell, R.B.; Arakawa, H.; Buerstedde, J.M. A cis-acting diversification activator both necessary and sufficient for AID-mediated hypermutation. PLoS Genet. 2009, 5, e1000332. [Google Scholar] [CrossRef]
- Arakawa, H.; Moldovan, G.L.; Saribasak, H.; Saribasak, N.N.; Jentsch, S.; Buerstedde, J.M. A role for PCNA ubiquitination in immunoglobulin hypermutation. PLoS Biol. 2006, 4, e366. [Google Scholar] [CrossRef]
- Arakawa, H.; Saribasak, H.; Buerstedde, J.M. Activation-induced cytidine deaminase initiates immunoglobulin gene conversion and hypermutation by a common intermediate. PLoS Biol. 2004, 2, e179. [Google Scholar] [CrossRef]
- Di Noia, J.M.; Neuberger, M.S. Molecular mechanisms of antibody somatic hypermutation. Annu. Rev. Biochem. 2007, 76, 1–22. [Google Scholar] [CrossRef]
- Sale, J.E. Immunoglobulin diversification in DT40: A model for vertebrate DNA damage tolerance. DNA Repair 2004, 3, 693–702. [Google Scholar] [CrossRef]
- Sale, J.E. Measurement of diversification in the immunoglobulin light chain gene of DT40 cells. Methods Mol. Biol. 2012, 920, 417–432. [Google Scholar] [CrossRef]
- Buerstedde, J.M.; Reynaud, C.A.; Humphries, E.H.; Olson, W.; Ewert, D.L.; Weill, J.C. Light chain gene conversion continues at high rate in an ALV-induced cell line. EMBO J. 1990, 9, 921–927. [Google Scholar] [CrossRef] [PubMed]
- Buerstedde, J.M.; Takeda, S. Increased ratio of targeted to random integration after transfection of chicken B cell lines. Cell 1991, 67, 179–188. [Google Scholar] [CrossRef]
- Seo, H.; Masuoka, M.; Murofushi, H.; Takeda, S.; Shibata, T.; Ohta, K. Rapid generation of specific antibodies by enhanced homologous recombination. Nat. Biotechnol. 2005, 23, 731–735. [Google Scholar] [CrossRef]
- Cummings, W.J.; Yabuki, M.; Ordinario, E.C.; Bednarski, D.W.; Quay, S.; Maizels, N. Chromatin structure regulates gene conversion. PLoS Biol. 2007, 5, e246. [Google Scholar] [CrossRef]
- Arakawa, H. Immunoglobulin gene conversion and hypermutation assay by FACs. Sub-Cell. Biochem. 2006, 40, 351–352. [Google Scholar]
- Shinkura, R.; Ito, S.; Begum, N.A.; Nagaoka, H.; Muramatsu, M.; Kinoshita, K.; Sakakibara, Y.; Hijikata, H.; Honjo, T. Separate domains of AID are required for somatic hypermutation and class-switch recombination. Nat. Immunol. 2004, 5, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Saberi, A.; Nakahara, M.; Sale, J.E.; Kikuchi, K.; Arakawa, H.; Buerstedde, J.M.; Yamamoto, K.; Takeda, S.; Sonoda, E. The 9-1-1 DNA clamp is required for immunoglobulin gene conversion. Mol. Cell. Biol. 2008, 28, 6113–6122. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yang, Z.; Rada, C.; Neuberger, M.S. AID upmutants isolated using a high-throughput screen highlight the immunity/cancer balance limiting DNA deaminase activity. Nat. Struct. Mol. Biol. 2009, 16, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Sale, J.E.; Calandrini, D.M.; Takata, M.; Takeda, S.; Neuberger, M.S. Ablation of XRCC2/3 transforms immunoglobulin V gene conversion into somatic hypermutation. Nature 2001, 412, 921–926. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.L.; Sale, J.E. The catalytic activity of REV1 is employed during immunoglobulin gene diversification in DT40. Mol. Immunol. 2006, 43, 1587–1594. [Google Scholar] [CrossRef] [PubMed]
- Longerich, S.; Orelli, B.J.; Martin, R.W.; Bishop, D.K.; Storb, U. Brca1 in immunoglobulin gene conversion and somatic hypermutation. DNA Repair 2008, 7, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, A.; Yamazoe, M.; Sale, J.E.; Takata, M.; Yamamoto, K.; Kitao, H.; Sonoda, E.; Kikuchi, K.; Yonetani, Y.; Takeda, S. Similar effects of Brca2 truncation and Rad51 paralog deficiency on immunoglobulin V gene diversification in DT40 cells support an early role for Rad51 paralogs in homologous recombination. Mol. Cell. Biol. 2005, 25, 1124–1134. [Google Scholar] [CrossRef] [PubMed]
- Bezzubova, O.; Silbergleit, A.; Yamaguchi-Iwai, Y.; Takeda, S.; Buerstedde, J.M. Reduced X-ray resistance and homologous recombination frequencies in a RAD54−/− mutant of the chicken DT40 cell line. Cell 1997, 89, 185–193. [Google Scholar] [CrossRef]
- Yamamoto, K.; Hirano, S.; Ishiai, M.; Morishima, K.; Kitao, H.; Namikoshi, K.; Kimura, M.; Matsushita, N.; Arakawa, H.; Buerstedde, J.M.; et al. Fanconi anemia protein FANCD2 promotes immunoglobulin gene conversion and DNA repair through a mechanism related to homologous recombination. Mol. Cell. Biol. 2005, 25, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, T.; Araki, K.; Sonoda, E.; Yamashita, Y.M.; Harada, K.; Kikuchi, K.; Masutani, C.; Hanaoka, F.; Nozaki, K.; Hashimoto, N.; et al. Dual roles for DNA polymerase eta in homologous DNA recombination and translesion DNA synthesis. Mol. Cell 2005, 20, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Holzschu, D.L.; Sugiyama, T. PCNA is efficiently loaded on the DNA recombination intermediate to modulate polymerase δ, η, and ζ activities. Proc. Natl. Acad. Sci. USA 2013, 110, 7672–7677. [Google Scholar] [CrossRef] [PubMed]
- McIlwraith, M.J.; Vaisman, A.; Liu, Y.; Fanning, E.; Woodgate, R.; West, S.C. Human DNA polymerase η promotes DNA synthesis from strand invasion intermediates of homologous recombination. Mol. Cell 2005, 20, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Sebesta, M.; Burkovics, P.; Haracska, L.; Krejci, L. Reconstitution of DNA repair synthesis in vitro and the role of polymerase and helicase activities. DNA Repair 2011, 10, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Sebesta, M.; Burkovics, P.; Juhasz, S.; Zhang, S.; Szabo, J.E.; Lee, M.Y.; Haracska, L.; Krejci, L. Role of PCNA and TLS polymerases in D-loop extension during homologous recombination in humans. DNA Repair 2013, 12, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Kannouche, P.L.; Wing, J.; Lehmann, A.R. Interaction of human DNA polymerase η with monoubiquitinated PCNA: A possible mechanism for the polymerase switch in response to DNA damage. Mol. Cell 2004, 14, 491–500. [Google Scholar] [CrossRef]
- Moldovan, G.L.; Pfander, B.; Jentsch, S. PCNA, the maestro of the replication fork. Cell 2007, 129, 665–679. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, H.D. Deubiquitinating PCNA: A downside to DNA damage tolerance. Nat. Cell Biol. 2006, 8, 303–305. [Google Scholar] [CrossRef] [PubMed]
- Navadgi-Patil, V.M.; Burgers, P.M. The unstructured C-Terminal tail of the 9-1-1 clamp subunit Ddc1 activates Mec1/ATR via two distinct mechanisms. Mol. Cell 2009, 36, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Navadgi-Patil, V.M.; Burgers, P.M. A tale of two tails: Activation of DNA damage checkpoint kinase Mec1/ATR by the 9-1-1 clamp and by Dpb11/TopBP1. DNA Repair 2009, 8, 996–1003. [Google Scholar] [CrossRef]
- Niida, H.; Nakanishi, M. DNA damage checkpoints in mammals. Mutagenesis 2006, 21, 3–9. [Google Scholar] [CrossRef]
- Bermudez, V.P.; Lindsey-Boltz, L.A.; Cesare, A.J.; Maniwa, Y.; Griffith, J.D.; Hurwitz, J.; Sancar, A. Loading of the human 9-1-1 checkpoint complex onto DNA by the checkpoint clamp loader hRad17-replication factor C complex in vitro. Proc. Natl. Acad. Sci. USA 2003, 100, 1633–1638. [Google Scholar] [CrossRef]
- Ellison, V.; Stillman, B. Biochemical characterization of DNA damage checkpoint complexes: Clamp loader and clamp complexes with specificity for 5’ recessed DNA. PLoS Biol. 2003, 1, E33. [Google Scholar] [CrossRef]
- Budzowska, M.; Jaspers, I.; Essers, J.; de Waard, H.; van Drunen, E.; Hanada, K.; Beverloo, B.; Hendriks, R.W.; de Klein, A.; Kanaar, R.; et al. Mutation of the mouse Rad17 gene leads to embryonic lethality and reveals a role in DNA damage-dependent recombination. EMBO J. 2004, 23, 3548–3558. [Google Scholar] [CrossRef]
- Pandita, R.K.; Sharma, G.G.; Laszlo, A.; Hopkins, K.M.; Davey, S.; Chakhparonian, M.; Gupta, A.; Wellinger, R.J.; Zhang, J.; Powell, S.N.; et al. Mammalian Rad9 plays a role in telomere stability, S- and G2-phase-specific cell survival, and homologous recombinational repair. Mol. Cell. Biol. 2006, 26, 1850–1864. [Google Scholar] [CrossRef]
- Bailey, C.; Fryer, A.E.; Greenslade, M. Warsaw Breakage Syndrome—A further report, emphasising cutaneous findings. Eur. J. Med. Genet. 2015, 58, 235–237. [Google Scholar] [CrossRef]
- Capo-Chichi, J.M.; Bharti, S.K.; Sommers, J.A.; Yammine, T.; Chouery, E.; Patry, L.; Rouleau, G.A.; Samuels, M.E.; Hamdan, F.F.; Michaud, J.L.; et al. Identification and biochemical characterization of a novel mutation in DDX11 causing Warsaw breakage syndrome. Hum. Mutat. 2013, 34, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Eppley, S.; Hopkin, R.J.; Mendelsohn, B.; Slavotinek, A.M. Clinical Report: Warsaw Breakage Syndrome with small radii and fibulae. Am. J. Med. Genet. A 2017, 173, 3075–3081. [Google Scholar] [CrossRef] [PubMed]
- Van der Lelij, P.; Chrzanowska, K.H.; Godthelp, B.C.; Rooimans, M.A.; Oostra, A.B.; Stumm, M.; Zdzienicka, M.Z.; Joenje, H.; de Winter, J.P. Warsaw breakage syndrome, a cohesinopathy associated with mutations in the XPD helicase family member DDX11/ChlR1. Am. J. Hum. Genet. 2010, 86, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Abe, T.; Ooka, M.; Kawasumi, R.; Miyata, K.; Takata, M.; Hirota, K.; Branzei, D. Warsaw breakage syndrome DDX11 helicase acts jointly with RAD17 in the repair of bulky lesions and replication through abasic sites. Proc. Natl. Acad. Sci. USA 2018, 115, 8412–8417. [Google Scholar] [CrossRef] [PubMed]
- Niedzwiedz, W.; Mosedale, G.; Johnson, M.; Ong, C.Y.; Pace, P.; Patel, K.J. The Fanconi anaemia gene FANCC promotes homologous recombination and error-prone DNA repair. Mol. Cell 2004, 15, 607–620. [Google Scholar] [CrossRef] [PubMed]
- Centore, R.C.; Yazinski, S.A.; Tse, A.; Zou, L. Spartan/C1orf124, a reader of PCNA ubiquitylation and a regulator of UV-induced DNA damage response. Mol. Cell 2012, 46, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Ghosal, G.; Leung, J.W.; Nair, B.C.; Fong, K.W.; Chen, J. Proliferating cell nuclear antigen (PCNA)-binding protein C1orf124 is a regulator of translesion synthesis. J. Biol. Chem. 2012, 287, 34225–34233. [Google Scholar] [CrossRef]
- Juhasz, S.; Balogh, D.; Hajdu, I.; Burkovics, P.; Villamil, M.A.; Zhuang, Z.; Haracska, L. Characterization of human Spartan/C1orf124, an ubiquitin-PCNA interacting regulator of DNA damage tolerance. Nucleic Acids Res. 2012, 40, 10795–10808. [Google Scholar] [CrossRef]
- Machida, Y.; Kim, M.S.; Machida, Y.J. Spartan/C1orf124 is important to prevent UV-induced mutagenesis. Cell Cycle 2012, 11, 3395–3402. [Google Scholar] [CrossRef]
- Toth, A.; Hegedus, L.; Juhasz, S.; Haracska, L.; Burkovics, P. The DNA-binding box of human SPARTAN contributes to the targeting of Poleta to DNA damage sites. DNA Repair 2017, 49, 33–42. [Google Scholar] [CrossRef]
- Davis, E.J.; Lachaud, C.; Appleton, P.; Macartney, T.J.; Nathke, I.; Rouse, J. DVC1 (C1orf124) recruits the p97 protein segregase to sites of DNA damage. Nat. Struct. Mol. Biol. 2012, 19, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Mosbech, A.; Gibbs-Seymour, I.; Kagias, K.; Thorslund, T.; Beli, P.; Povlsen, L.; Nielsen, S.V.; Smedegaard, S.; Sedgwick, G.; Lukas, C.; et al. DVC1 (C1orf124) is a DNA damage-targeting p97 adaptor that promotes ubiquitin-dependent responses to replication blocks. Nat. Struct. Mol. Biol. 2012, 19, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- Maskey, R.S.; Flatten, K.S.; Sieben, C.J.; Peterson, K.L.; Baker, D.J.; Nam, H.J.; Kim, M.S.; Smyrk, T.C.; Kojima, Y.; Machida, Y.; et al. Spartan deficiency causes accumulation of Topoisomerase 1 cleavage complexes and tumorigenesis. Nucleic Acids Res. 2017, 45, 4564–4576. [Google Scholar] [CrossRef] [PubMed]
- Morocz, M.; Zsigmond, E.; Toth, R.; Enyedi, M.Z.; Pinter, L.; Haracska, L. DNA-dependent protease activity of human Spartan facilitates replication of DNA-protein crosslink-containing DNA. Nucleic Acids Res. 2017, 45, 3172–3188. [Google Scholar] [CrossRef] [PubMed]
- Stingele, J.; Bellelli, R.; Alte, F.; Hewitt, G.; Sarek, G.; Maslen, S.L.; Tsutakawa, S.E.; Borg, A.; Kjaer, S.; Tainer, J.A.; et al. Mechanism and regulation of DNA-protein crosslink repair by the DNA-dependent metalloprotease SPRTN. Mol. Cell 2016, 64, 688–703. [Google Scholar] [CrossRef] [PubMed]
- Vaz, B.; Popovic, M.; Newman, J.A.; Fielden, J.; Aitkenhead, H.; Halder, S.; Singh, A.N.; Vendrell, I.; Fischer, R.; Torrecilla, I.; et al. Metalloprotease SPRTN/DVC1 orchestrates replication-coupled DNA-protein crosslink repair. Mol. Cell 2016, 64, 704–719. [Google Scholar] [CrossRef] [PubMed]
- Nakazato, A.; Kajita, K.; Ooka, M.; Akagawa, R.; Abe, T.; Takeda, S.; Branzei, D.; Hirota, K. SPARTAN promotes genetic diversification of the immunoglobulin-variable gene locus in avian DT40 cells. DNA Repair 2018, 68, 50–57. [Google Scholar] [CrossRef]
- Lee, K.Y.; Myung, K. PCNA modifications for regulation of post-replication repair pathways. Mol. Cells 2008, 26, 5–11. [Google Scholar]
- McIntyre, J.; Woodgate, R. Regulation of translesion DNA synthesis: Posttranslational modification of lysine residues in key proteins. DNA Repair 2015, 29, 166–179. [Google Scholar] [CrossRef]
- Kanao, R.; Masutani, C. Regulation of DNA damage tolerance in mammalian cells by post-translational modifications of PCNA. Mutat. Res. 2017, 803–805, 82–88. [Google Scholar] [CrossRef]
- Simpson, L.J.; Ross, A.L.; Szuts, D.; Alviani, C.A.; Oestergaard, V.H.; Patel, K.J.; Sale, J.E. RAD18-independent ubiquitination of proliferating-cell nuclear antigen in the avian cell line DT40. EMBO Rep. 2006, 7, 927–932. [Google Scholar] [CrossRef] [PubMed]
- Okada, T.; Sonoda, E.; Yoshimura, M.; Kawano, Y.; Saya, H.; Kohzaki, M.; Takeda, S. Multiple roles of vertebrate REV genes in DNA repair and recombination. Mol. Cell. Biol. 2005, 25, 6103–6111. [Google Scholar] [CrossRef] [PubMed]
- Edmunds, C.E.; Simpson, L.J.; Sale, J.E. PCNA ubiquitination and REV1 define temporally distinct mechanisms for controlling translesion synthesis in the avian cell line DT40. Mol. Cell 2008, 30, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Branzei, D.; Seki, M.; Enomoto, T. Rad18/Rad5/Mms2-mediated polyubiquitination of PCNA is implicated in replication completion during replication stress. Genes Cells 2004, 9, 1031–1042. [Google Scholar] [CrossRef] [PubMed]
- Hishida, T.; Kubota, Y.; Carr, A.M.; Iwasaki, H. RAD6-RAD18-RAD5-pathway-dependent tolerance to chronic low-dose ultraviolet light. Nature 2009, 457, 612–615. [Google Scholar] [CrossRef]
- Hoege, C.; Pfander, B.; Moldovan, G.L.; Pyrowolakis, G.; Jentsch, S. RAD6-dependent DNA repair is linked to modification of PCNA by ubiquitin and SUMO. Nature 2002, 419, 135–141. [Google Scholar] [CrossRef]
- Torres-Ramos, C.A.; Prakash, S.; Prakash, L. Requirement of RAD5 and MMS2 for postreplication repair of UV-damaged DNA in Saccharomyces cerevisiae. Mol. Cell. Biol. 2002, 22, 2419–2426. [Google Scholar] [CrossRef]
- Branzei, D.; Vanoli, F.; Foiani, M. SUMOylation regulates Rad18-mediated template switch. Nature 2008, 456, 915–920. [Google Scholar] [CrossRef]
- Pilzecker, B.; Buoninfante, O.A.; van den Berk, P.; Lancini, C.; Song, J.Y.; Citterio, E.; Jacobs, H. DNA damage tolerance in hematopoietic stem and progenitor cells in mice. Proc. Natl. Acad. Sci. USA 2017, 114, E6875–E6883. [Google Scholar] [CrossRef]
- Yang, Y.; Poe, J.C.; Yang, L.; Fedoriw, A.; Desai, S.; Magnuson, T.; Li, Z.; Fedoriw, Y.; Araki, K.; Gao, Y.; et al. Rad18 confers hematopoietic progenitor cell DNA damage tolerance independently of the Fanconi Anemia pathway in vivo. Nucleic Acids Res. 2016, 44, 4174–4188. [Google Scholar] [CrossRef]
- Huang, J.; Huen, M.S.; Kim, H.; Leung, C.C.; Glover, J.N.; Yu, X.; Chen, J. RAD18 transmits DNA damage signalling to elicit homologous recombination repair. Nat. Cell Biol. 2009, 11, 592–603. [Google Scholar] [CrossRef] [PubMed]
- Motegi, A.; Liaw, H.J.; Lee, K.Y.; Roest, H.P.; Maas, A.; Wu, X.; Moinova, H.; Markowitz, S.D.; Ding, H.; Hoeijmakers, J.H.; et al. Polyubiquitination of proliferating cell nuclear antigen by HLTF and SHPRH prevents genomic instability from stalled replication forks. Proc. Natl. Acad. Sci. USA 2008, 105, 12411–12416. [Google Scholar] [CrossRef] [PubMed]
- Motegi, A.; Sood, R.; Moinova, H.; Markowitz, S.D.; Liu, P.P.; Myung, K. Human SHPRH suppresses genomic instability through proliferating cell nuclear antigen polyubiquitination. J. Cell Biol. 2006, 175, 703–708. [Google Scholar] [CrossRef]
- Unk, I.; Hajdu, I.; Blastyak, A.; Haracska, L. Role of yeast Rad5 and its human orthologs, HLTF and SHPRH in DNA damage tolerance. DNA Repair 2010, 9, 257–267. [Google Scholar] [CrossRef]
- Unk, I.; Hajdu, I.; Fatyol, K.; Szakal, B.; Blastyak, A.; Bermudez, V.; Hurwitz, J.; Prakash, L.; Prakash, S.; Haracska, L. Human SHPRH is a ubiquitin ligase for Mms2-Ubc13-dependent polyubiquitylation of proliferating cell nuclear antigen. Proc. Natl. Acad. Sci. USA 2006, 103, 18107–18112. [Google Scholar] [CrossRef] [PubMed]
- Tomi, N.S.; Davari, K.; Grotzky, D.; Loos, F.; Bottcher, K.; Frankenberger, S.; Jungnickel, B. Analysis of SHPRH functions in DNA repair and immunoglobulin diversification. DNA Repair 2014, 24, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Kolas, N.K.; Chapman, J.R.; Nakada, S.; Ylanko, J.; Chahwan, R.; Sweeney, F.D.; Panier, S.; Mendez, M.; Wildenhain, J.; Thomson, T.M.; et al. Orchestration of the DNA-damage response by the RNF8 ubiquitin ligase. Science 2007, 318, 1637–1640. [Google Scholar] [CrossRef] [PubMed]
- Panier, S.; Durocher, D. Regulatory ubiquitylation in response to DNA double-strand breaks. DNA Repair 2009, 8, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Stewart, G.S.; Panier, S.; Townsend, K.; Al-Hakim, A.K.; Kolas, N.K.; Miller, E.S.; Nakada, S.; Ylanko, J.; Olivarius, S.; Mendez, M.; et al. The RIDDLE syndrome protein mediates a ubiquitin-dependent signaling cascade at sites of DNA damage. Cell 2009, 136, 420–434. [Google Scholar] [CrossRef] [PubMed]
- Moynahan, M.E.; Chiu, J.W.; Koller, B.H.; Jasin, M. Brca1 controls homology-directed DNA repair. Mol. Cell 1999, 4, 511–518. [Google Scholar] [CrossRef]
- Scully, R.; Ganesan, S.; Vlasakova, K.; Chen, J.; Socolovsky, M.; Livingston, D.M. Genetic analysis of BRCA1 function in a defined tumor cell line. Mol. Cell 1999, 4, 1093–1099. [Google Scholar] [CrossRef]
- Zhao, G.Y.; Sonoda, E.; Barber, L.J.; Oka, H.; Murakawa, Y.; Yamada, K.; Ikura, T.; Wang, X.; Kobayashi, M.; Yamamoto, K.; et al. A critical role for the ubiquitin-conjugating enzyme Ubc13 in initiating homologous recombination. Mol. Cell 2007, 25, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Keka, I.S.; Guilbaud, G.; Sale, J.; Narita, T.; Abdel-Aziz, H.I.; Wang, X.; Ogawa, S.; Sasanuma, H.; Chiu, R.; et al. The role of HERC2 and RNF8 ubiquitin E3 ligases in the promotion of translesion DNA synthesis in the chicken DT40 cell line. DNA Repair 2016, 40, 67–76. [Google Scholar] [CrossRef]
- Arakawa, H.; Hauschild, J.; Buerstedde, J.M. Requirement of the activation-induced deaminase (AID) gene for immunoglobulin gene conversion. Science 2002, 295, 1301–1306. [Google Scholar] [CrossRef]
- Oka, H.; Sakai, W.; Sonoda, E.; Nakamura, J.; Asagoshi, K.; Wilson, S.H.; Kobayashi, M.; Yamamoto, K.; Heierhorst, J.; Takeda, S.; et al. DNA damage response protein ASCIZ links base excision repair with immunoglobulin gene conversion. Biochem. Biophys. Res. Commun. 2008, 371, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Budzynska, P.M.; Kylaniemi, M.K.; Kallonen, T.; Soikkeli, A.I.; Nera, K.P.; Lassila, O.; Alinikula, J. Bach2 regulates AID-mediated immunoglobulin gene conversion and somatic hypermutation in DT40 B cells. Eur. J. Immunol. 2017, 47, 993–1001. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, K.; Abdel-Aziz, H.I.; Taniguchi, Y.; Yamazoe, M.; Takeda, S.; Hirota, K. Bloom DNA helicase facilitates homologous recombination between diverged homologous sequences. J. Biol. Chem. 2009, 284, 26360–26367. [Google Scholar] [CrossRef] [PubMed]
- Conticello, S.G.; Ganesh, K.; Xue, K.; Lu, M.; Rada, C.; Neuberger, M.S. Interaction between antibody-diversification enzyme AID and spliceosome-associated factor CTNNBL1. Mol. Cell 2008, 31, 474–484. [Google Scholar] [CrossRef]
- Kitao, H.; Kimura, M.; Yamamoto, K.; Seo, H.; Namikoshi, K.; Agata, Y.; Ohta, K.; Takata, M. Regulation of histone H4 acetylation by transcription factor E2A in Ig gene conversion. Int. Immunol. 2008, 20, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Pace, P.; Mosedale, G.; Hodskinson, M.R.; Rosado, I.V.; Sivasubramaniam, M.; Patel, K.J. Ku70 corrupts DNA repair in the absence of the Fanconi anemia pathway. Science 2010, 329, 219–223. [Google Scholar] [CrossRef]
- Kitao, H.; Nanda, I.; Sugino, R.P.; Kinomura, A.; Yamazoe, M.; Arakawa, H.; Schmid, M.; Innan, H.; Hiom, K.; Takata, M. FancJ/Brip1 helicase protects against genomic losses and gains in vertebrate cells. Genes Cells 2011, 16, 714–727. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, K.; Taniguchi, Y.; Hatanaka, A.; Sonoda, E.; Hochegger, H.; Adachi, N.; Matsuzaki, Y.; Koyama, H.; van Gent, D.C.; Jasin, M.; et al. Fen-1 facilitates homologous recombination by removing divergent sequences at DNA break ends. Mol. Cell. Biol. 2005, 25, 6948–6955. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Hashimoto, S.; Seo, H.; Shibata, T.; Ohta, K. Modulation of immunoglobulin gene conversion frequency and distribution by the histone deacetylase HDAC2 in chicken DT40. Genes Cells 2008, 13, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Simpson, L.J.; Sale, J.E. UBE2V2 (MMS2) is not required for effective immunoglobulin gene conversion or DNA damage tolerance in DT40. DNA Repair 2005, 4, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Campo, V.A.; Patenaude, A.M.; Kaden, S.; Horb, L.; Firka, D.; Jiricny, J.; Di Noia, J.M. MSH6- or PMS2-deficiency causes re-replication in DT40 B cells, but it has little effect on immunoglobulin gene conversion or on repair of AID-generated uracils. Nucleic Acids Res. 2013, 41, 3032–3046. [Google Scholar] [CrossRef]
- Nakahara, M.; Sonoda, E.; Nojima, K.; Sale, J.E.; Takenaka, K.; Kikuchi, K.; Taniguchi, Y.; Nakamura, K.; Sumitomo, Y.; Bree, R.T.; et al. Genetic evidence for single-strand lesions initiating Nbs1-dependent homologous recombination in diversification of Ig V in chicken B lymphocytes. PLoS Genet. 2009, 5, e1000356. [Google Scholar] [CrossRef] [PubMed]
- Paddock, M.N.; Buelow, B.D.; Takeda, S.; Scharenberg, A.M. The BRCT domain of PARP-1 is required for immunoglobulin gene conversion. PLoS Biol. 2010, 8, e1000428. [Google Scholar] [CrossRef] [PubMed]
- Hosono, Y.; Abe, T.; Ishiai, M.; Islam, M.N.; Arakawa, H.; Wang, W.; Takeda, S.; Ishii, Y.; Takata, M.; Seki, M.; et al. Tumor suppressor RecQL5 controls recombination induced by DNA crosslinking agents. Biochim. Biophys. Acta 2014, 1843, 1002–1012. [Google Scholar] [CrossRef] [PubMed]
- Simpson, L.J.; Sale, J.E. Rev1 is essential for DNA damage tolerance and non-templated immunoglobulin gene mutation in a vertebrate cell line. EMBO J. 2003, 22, 1654–1664. [Google Scholar] [CrossRef] [PubMed]

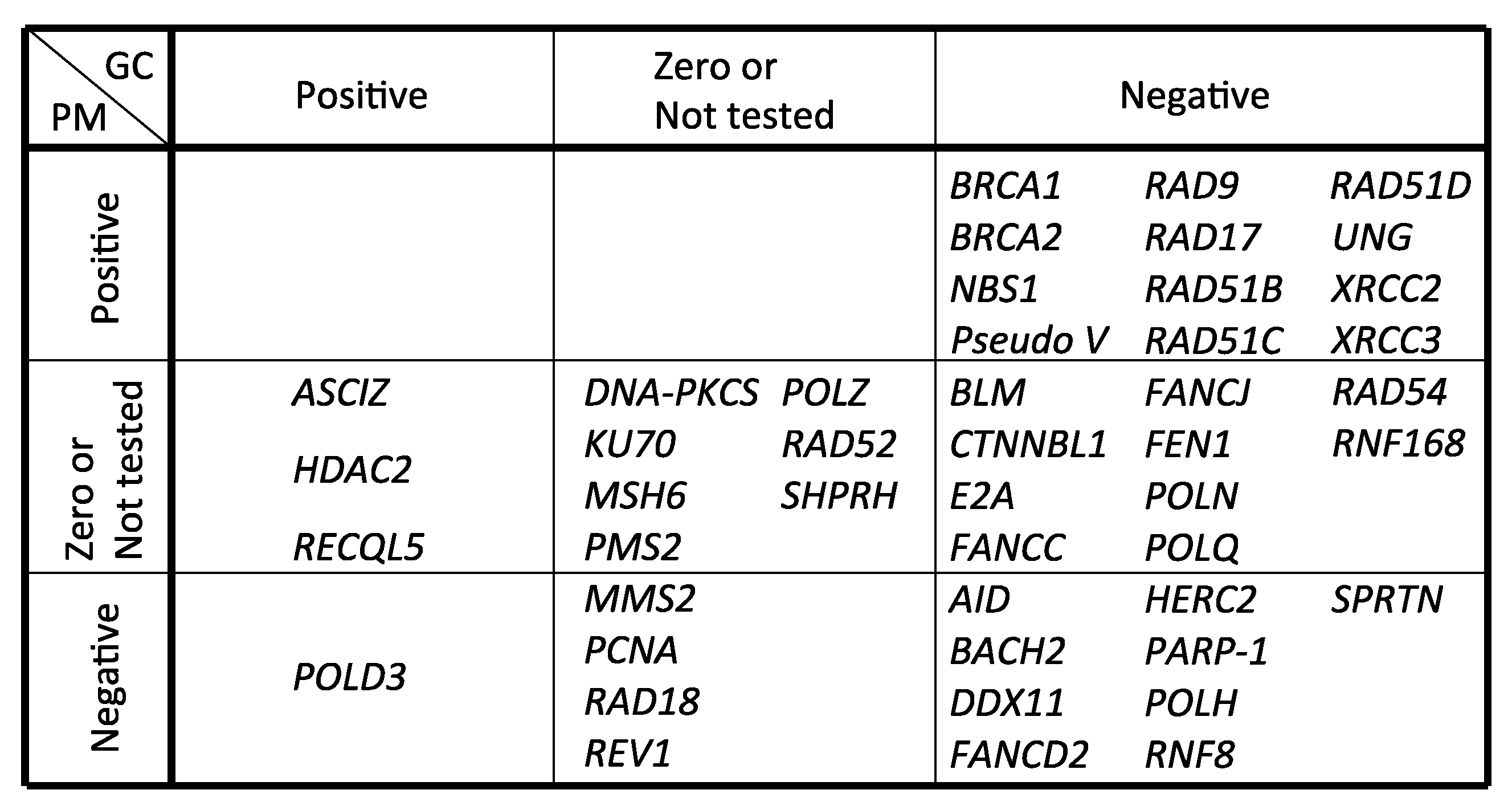
| Gene Name | GC | PM | Method | Reference |
|---|---|---|---|---|
| AID | − | − | IgM gain and sequencing | Arakawa et al., 2002 [105] |
| ASCIZ | + | 0 | IgM gain, mAID overexpression and sequencing | Oka et al., 2008 [106] |
| BACH2 | − | − | IgM gain and sequencing | Budzynska et al., 2017 [107] |
| BLM | − | NT | IgM gain | Kikuchi et al., 2009 [108] |
| BRCA1 | − | + | IgM loss and sequencing | Longerich et al., 2008 [42] |
| BRCA2 | − | + | IgM loss and sequencing | Hatanaka et al., 2005 [43] |
| CTNNBL1 | − | NT | IgM gain | Conticello et al., 2008 [109] |
| DDX11 | − | − | IgM gain, mAID overexpression and sequencing | Abe et al., 2018 [65] |
| DNA-PKCS | NT | 0 | IgM loss | Sale et al., 2001 [40] |
| E2A | − | 0 | IgM gain and sequencing | Kitao et al., 2008 [110] |
| FANCC | − | NT | IgM loss and sequencing | Pace et al., 2010 [111] |
| FANCD2 | − | − | IgM loss, gain and sequencing | Yamamoto et al., 2005 [45] |
| FANCJ | − | NT | IgM loss and sequencing | Kitao et al., 2011 [112] |
| FEN1 | − | NT | IgM gain and sequencing | Kikuchi et al., 2005 [113] |
| HDAC2 | + | 0 | IgM gain and sequencing | Lin et al., 2008 [114] |
| HERC2 | − | − | mAID overexpression and sequencing | Mohiuddin et al., 2016 [104] |
| KU70 | NT | 0 | IgM loss | Sale et al., 2001 [40] |
| MMS2 | 0 | − | IgM loss and sequencing | Simpson et al., 2005 [115] |
| MSH6 | 0 | 0 | IgM gain and sequencing | Campo et al., 2013 [116] |
| NBS1 (p70) | − | + | sIgM gain, mAID overexpression and sequencing | Nakahara et al., 2009 [117] |
| PARP-1 | − | − | IgM gain, mAID overexpression and sequencing | Paddock et al., 2010 [118] |
| PCNA (K164R) | NT | − | IgM loss and sequencing | Arakawa et al., 2006 [27] |
| PMS2 | 0 | 0 | IgM gain and sequencing | Campo et al., 2013 [116] |
| POLD3 | + | − | mAID overexpression and sequencing | Hirota et al., 2015 [16] |
| POLH | − | − | IgM gain and sequencing | Kawamoto et al., 2006 [46], Kohzaki et al., 2010 [17] |
| POLN | − | 0 | IgM gain, mAID overexpression and sequencing | Kohzaki et al., 2010 [17] |
| POLQ | − | 0 | IgM gain, mAID overexpression and sequencing | Kohzaki et al., 2010 [17] |
| POLH/POLN/POLQ | − | − | IgM gain, mAID overexpression and sequencing | Kohzaki et al., 2010 [17] |
| POLZ (REV3) | 0 | NT | IgM gain and sequencing | Okada et al., 2005 [83] |
| Pseudo V | − | + | IgM loss and sequencing | Arakawa et al., 2004 [28] |
| RAD9 | − | + | mAID overexpression and sequencing | Saberi et al., 2008 [38] |
| RAD17 | − | + | mAID overexpression and sequencing | Saberi et al., 2008 [38] |
| RAD18 | NT | − | IgM loss and sequencing | Arakawa et al., 2006 [27] |
| RAD51B | − | + | IgM loss and sequencing | Sale et al., 2001 [40] |
| RAD51C | − | + | IgM loss and sequencing | Hatanaka et al., 2005 [43] |
| RAD51D | − | + | IgM loss and sequencing | Hatanaka et al., 2005 [43] |
| RAD52 | NT | 0 | IgM loss | Sale et al., 2001 [40] |
| RAD54 | − | NT | IgM gain and sequencing | Bezzubova et al., 1997 [44] |
| RECQL5 | + | NT | IgM gain and sequencing | Hosono et al., 2014 [119] |
| RNF8 | − | − | mAID overexpression and sequencing | Mohiuddin et al., 2016 [104] |
| RNF168 | − | 0 | mAID overexpression and sequencing | Mohiuddin et al., 2016 [104] |
| SPRTN | − | − | IgM gain, mAID overexpression and sequencing | Nakazato et al., 2018 [78] |
| SHPRH | 0 | 0 | sIgM gain and sequencing | Tomi et al., 2014 [97] |
| REV1 | 0 | − | IgM loss and sequencing | Simpson et al., 2003 [120] |
| UNG | − | + | IgM gain, loss and sequencing | Saribasak et al., 2005 [25] |
| XRCC2 | − | + | IgM loss and sequencing | Sale et al., 2001 [40] |
| XRCC3 | − | + | IgM loss and sequencing | Sale et al., 2001 [40] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abe, T.; Branzei, D.; Hirota, K. DNA Damage Tolerance Mechanisms Revealed from the Analysis of Immunoglobulin V Gene Diversification in Avian DT40 Cells. Genes 2018, 9, 614. https://doi.org/10.3390/genes9120614
Abe T, Branzei D, Hirota K. DNA Damage Tolerance Mechanisms Revealed from the Analysis of Immunoglobulin V Gene Diversification in Avian DT40 Cells. Genes. 2018; 9(12):614. https://doi.org/10.3390/genes9120614
Chicago/Turabian StyleAbe, Takuya, Dana Branzei, and Kouji Hirota. 2018. "DNA Damage Tolerance Mechanisms Revealed from the Analysis of Immunoglobulin V Gene Diversification in Avian DT40 Cells" Genes 9, no. 12: 614. https://doi.org/10.3390/genes9120614
APA StyleAbe, T., Branzei, D., & Hirota, K. (2018). DNA Damage Tolerance Mechanisms Revealed from the Analysis of Immunoglobulin V Gene Diversification in Avian DT40 Cells. Genes, 9(12), 614. https://doi.org/10.3390/genes9120614




